Red Wine Consumption and Cardiovascular Health
Abstract
1. Introduction
2. Bioactive Components in Red Wine
2.1. Non-Flavonoid
2.1.1. Hydroxybenzoic Acids
2.1.2. Hydroxycinnamic Acids
2.1.3. Resveratrol
2.2. Flavonoids
2.2.1. Flavones
2.2.2. Flavan-3-ols
2.2.3. Flavonols
2.2.4. Anthocyanins
2.2.5. Tannins
2.2.6. Hydrolyzable tannins
2.2.7. Condensed tannins
3. Factors Influencing Bioactive Compounds and Composition of Wine
4. Putative Mechanisms of Action
4.1. Lipid Profile
4.2. Glucose Metabolism
4.3. Oxidative Stress
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yu, C.; Moore, B.M.; Kotchetkova, I.; Cordina, R.L.; Celermajer, D.S. Causes of death in a contemporary adult congenital heart disease cohort. Heart 2018, 104, 1678–1682. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Arranz, S.; Lamuela-Raventos, R.M.; Estruch, R. Effects of Wine, Alcohol and Polyphenols on Cardiovascular Disease Risk Factors: Evidences from Human Studies. Alcohol Alcohol. 2013, 48, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Steven, S.; Frenis, K.; Oelze, M.; Kalinovic, S.; Kuntic, M.; Jimenez, M.T.B.; Vujacic-Mirski, K.; Helmstädter, J.; Kröller-Schön, S.; Münzel, T.; et al. Vascular Inflammation and Oxidative Stress: Major Triggers for Cardiovascular Disease. Oxidative Med. Cell. Longev. 2019, 2019, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, B.; Aguilar, D.; Deswal, A.; Dunbar, S.B.; Francis, G.S.; Horwich, T.; Jessup, M.; Kosiborod, M.; Pritchett, A.M.; Ramasubbu, K.; et al. Contributory Risk and Management of Comorbidities of Hypertension, Obesity, Diabetes Mellitus, Hyperlipidemia, and Metabolic Syndrome in Chronic Heart Failure: A Scientific Statement From the American Heart Association. Circulation 2016, 134, 535. [Google Scholar] [CrossRef] [PubMed]
- Roerecke, M.; Rehm, J. Alcohol consumption, drinking patterns, and ischemic heart disease: a narrative review of meta-analyses and a systematic review and meta-analysis of the impact of heavy drinking occasions on risk for moderate drinkers. BMC Med. 2014, 12, 182. [Google Scholar] [CrossRef] [PubMed]
- DeSalvo, K.B.; Olson, R.; Casavale, K.O. Dietary guidelines for Americans. Jama 2016, 315, 457–458. [Google Scholar] [CrossRef]
- Di Renzo, L.; Marsella, L.T.; Carraro, A.; Valente, R.; Gualtieri, P.; Gratteri, S.; Tomasi, D.; Gaiotti, F.; De Lorenzo, A. Changes in LDL Oxidative Status and Oxidative and Inflammatory Gene Expression after Red Wine Intake in Healthy People: A Randomized Trial. Mediat. Inflamm. 2015, 2015, 1–13. [Google Scholar] [CrossRef]
- Annunziata, G.; Maisto, M.; Schisano, C.; Ciampaglia, R.; Narciso, V.; Hassan, S.T.; Tenore, G.C.; Novellino, E. Effect of grape pomace polyphenols with or without pectin on TMAO serum levels assessed by LC/MS-based assay: A preliminary clinical study on overweight/obese subjects. Front. Pharmacol. 2019, 10, 575. [Google Scholar] [CrossRef]
- Nova, E.; San Mauro-Martín, I.; Díaz-Prieto, L.E.; Marcos, A. Wine and beer within a moderate alcohol intake is associated with higher levels of HDL-c and adiponectin. Nutr. Res. 2019, 63, 42–50. [Google Scholar] [CrossRef]
- Golan, R.; Gepner, Y.; Shai, I.J.E.j.o.c.n. Wine and Health–New Evidence. Eur. J. Clin. Clin. Nutr. 2018, 72, 55–59. [Google Scholar] [CrossRef]
- Torres, A.; Cachofeiro, V.; Millán, J.; Lahera, V.; Nieto, M.; Martin, R.; Bello, E.; Alvarez-Sala, L.; Nieto, M. Red wine intake but not other alcoholic beverages increases total antioxidant capacity and improves pro-inflammatory profile after an oral fat diet in healthy volunteers. Revista Clínica Española 2015, 215, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Lamuela-Raventós, R.M.; Estruch, R. Mechanism of the Protective Effects of Wine Intake on Cardiovascular Disease. In Wine Safety, Consumer Preference, and Human Health; Moreno-Arribas, M.V., Suáldea, B.B., Eds.; Springer: Basel, Switzerland, 2016; pp. 231–239. [Google Scholar]
- Snopek, L.; Mlcek, J.; Sochorova, L.; Baron, M.; Hlavacova, I.; Jurikova, T.; Kizek, R.; Sedlackova, E.; Sochor, J. Contribution of Red Wine Consumption to Human Health Protection. Molcules 2018, 23, 1684. [Google Scholar] [CrossRef]
- Lippi, G.; Franchini, M.; Favaloro, E.J.; Targher, G. Moderate red wine consumption and cardiovascular disease risk: beyond the “French paradox”. In Proceedings of Seminars in thrombosis and hemostasis; Favaloro, E.J., Levi, M., Lisman, T., Kwaan, H.C., Schulman, S., Eds.; Thieme Medical Publishers: Stuttgart, Germany, 2010; pp. 059–070. [Google Scholar]
- Suo, H.; Tian, R.; Li, J.; Zhang, S.; Cui, Y.; Li, L.; Sun, B. Compositional characterization study on high -molecular -mass polymeric polyphenols in red wines by chemical degradation. Food Res. Int. 2019, 123, 440–449. [Google Scholar] [CrossRef] [PubMed]
- Šeruga, M.; Novak, I.; Jakobek, L. Determination of polyphenols content and antioxidant activity of some red wines by differential pulse voltammetry, HPLC and spectrophotometric methods. Food Chem. 2011, 124, 1208–1216. [Google Scholar] [CrossRef]
- Stephan, L.S.; Almeida, E.D.; Markoski, M.M.; Garavaglia, J.; Marcadenti, A. Red Wine, Resveratrol and Atrial Fibrillation. Nutrients 2017, 9, 1190. [Google Scholar] [CrossRef] [PubMed]
- Doonan, B.; Iraj, S.; Pellegrino, L.; Hsieh, T.-C.; Wu, J.; Watson, R.R.; Zibadi, S. 22. The French paradox revisited: cardioprotection via hormesis, red wine and resveratrol. In Handbook of nutrition and diet in therapy of bone diseases; Watson, R.R., Zibadi, S., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2017; Volume 14, pp. 467–485, Wageningen Academic Publishers. [Google Scholar]
- Sakaki, J.; Melough, M.; Gil Lee, S.; Pounis, G.; Chun, O.K. Polyphenol-Rich Diets in Cardiovascular Disease Prevention. In Analysis in Nutrition Research; Pounis, G., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 259–298. [Google Scholar]
- Ivanova, V.; Stefova, M.; Chinnici, F. Determination of the polyphenol contents in Macedonian grapes and wines by standardized spectrophotometric methods. J. Serbian Chem. Soc. 2010, 75, 45–59. [Google Scholar] [CrossRef]
- Vázquez, E.S.; Segade, S.R.; Fernández, I.O. Effect of the winemaking technique on phenolic composition and chromatic characteristics in young red wines. Eur. Food Res. Technol. 2010, 231, 789–802. [Google Scholar] [CrossRef]
- Ivanova-Petropulos, V.; Hermosín-Gutíerrez, I.; Boros, B.; Stefova, M.; Stafilov, T.; Vojnoski, B.; Dörnyei, Á.; Kilár, F. Phenolic compounds and antioxidant activity of Macedonian red wines. J. Food Compos. Anal. 2015, 41, 1–14. [Google Scholar] [CrossRef]
- Cuzmar, P.D.; Salgado, E.; Ribalta-Pizarro, C.; Olaeta, J.A.; López, E.; Pastenes, C.; Cáceres-Mella, A. Phenolic composition and sensory characteristics of Cabernet Sauvignon wines: effect of water stress and harvest date. Int. J. Food Sci. Technol. 2018, 53, 1726–1735. [Google Scholar] [CrossRef]
- Rentzsch, M.; Wilkens, A.; Winterhalter, P. Non-flavonoid phenolic compounds. In Wine chemistry and biochemistry; Moreno-Arribas, M.V., Polo, C., Eds.; Springer: New York City, NY, USA, 2009; pp. 509–527. [Google Scholar]
- Lofrano, G.; Meriç, S. A Review On Occurrence, Measurement, Toxicity And Tannin Removal Processes From Wastewaters. Environ. Eng. Manag. J. 2019, 18, 109–123. [Google Scholar]
- Cozzolino, D. The role of visible and infrared spectroscopy combined with chemometrics to measure phenolic compounds in grape and wine samples. Molecules 2015, 20, 726–737. [Google Scholar] [CrossRef] [PubMed]
- Basli, A.; Soulet, S.; Chaher, N.; Mérillon, J.-M.; Chibane, M.; Monti, J.-P.; Richard, T. Wine Polyphenols: Potential Agents in Neuroprotection. Oxidative Med. Cell. Longev. 2012, 2012, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ferreira-Lima, N.; Vallverdú-Queralt, A.; Meudec, E.; Pinasseau, L.; Verbaere, A.; Bordignon-Luiz, M.T.; Le Guernevé, C.; Cheynier, V.; Sommerer, N. Quantification of hydroxycinnamic derivatives in wines by UHPLC-MRM-MS. Anal. Bioanal. Chem. 2018, 410, 3483–3490. [Google Scholar] [CrossRef] [PubMed]
- Del Prete, V.; Costantini, A.; Cecchini, F.; Morassut, M.; Garcia-Moruno, E. Occurrence of biogenic amines in wine: The role of grapes. Food Chem. 2009, 112, 474–481. [Google Scholar] [CrossRef]
- Lima, A.; Oliveira, C.; Santos, C.; Campos, F.M.; Couto, J.A. Phenolic composition of monovarietal red wines regarding volatile phenols and its precursors. Eur. Food Res. Technol. 2018, 244, 1985–1994. [Google Scholar] [CrossRef]
- Nardini, M.; Forte, M.; Vrhovsek, U.; Mattivi, F.; Viola, R.; Scaccini, C. White Wine Phenolics Are Absorbed and Extensively Metabolized in Humans. J. Agric. Food Chem. 2009, 57, 2711–2718. [Google Scholar] [CrossRef] [PubMed]
- Garrido, J.; Borges, F. Wine and grape polyphenols — A chemical perspective. Food Res. Int. 2013, 54, 1844–1858. [Google Scholar] [CrossRef]
- Kiselev, K.V. Perspectives for production and application of resveratrol. Appl. Microbiol. Biotechnol. 2011, 90, 417–425. [Google Scholar] [CrossRef]
- Vitaglione, P.; Sforza, S.; Rio, D. Occurrence, Bioavailability and Metabolism of Resveratrol; Spencer, J.P.E., Crozier, A., Eds.; CRC Pres: Boca Raton, FI, USA, 2012; pp. 167–182. [Google Scholar]
- Hussein, M.A. A convenient mechanism for the free radical scavenging activity of resveratrol. Int. J. Phytomedicine 2011, 3, 459. [Google Scholar]
- Paulo, L.; Domingues, F.; Queiroz, J.A.; Gallardo, E. Development and Validation of an Analytical Method for the Determination oftrans- andcis-Resveratrol in Wine: Analysis of Its Contents in 186 Portuguese Red Wines. J. Agric. Food Chem. 2011, 59, 2157–2168. [Google Scholar] [CrossRef]
- Nour, V.; Trandafir, I.; Muntean, C. Ultraviolet Irradiation of Trans-Resveratrol and HPLC Determination of Trans-Resveratrol and Cis-Resveratrol in Romanian Red Wines. J. Chromatogr. Sci. 2012, 50, 920–927. [Google Scholar] [CrossRef]
- Dudley, J.; Das, S.; Mukherjee, S.; Das, D.K. RETRACTED: Resveratrol, a unique phytoalexin present in red wine, delivers either survival signal or death signal to the ischemic myocardium depending on dose. J. Nutr. Biochem. 2009, 20, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Godelmann, R.; Fang, F.; Humpfer, E.; Schütz, B.; Bansbach, M.; Schäfer, H.; Spraul, M. Targeted and Nontargeted Wine Analysis by1H NMR Spectroscopy Combined with Multivariate Statistical Analysis. Differentiation of Important Parameters: Grape Variety, Geographical Origin, Year of Vintage. J. Agric. Food Chem. 2013, 61, 5610–5619. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.M.; Hsieh, T.c. Resveratrol: a cardioprotective substance. Ann. New York Acad. Sci. 2011, 1215, 16–21. [Google Scholar] [CrossRef]
- Sahebkar, A. Effects of resveratrol supplementation on plasma lipids: a systematic review and meta-analysis of randomized controlled trials. Nutr. Rev. 2013, 71, 822–835. [Google Scholar] [CrossRef] [PubMed]
- Penumathsa, S.V.; Maulik, N. Resveratrol: a promising agent in promoting cardioprotection against coronary heart disease. Can. J. Physiol. Pharmacol. 2009, 87, 275–286. [Google Scholar] [CrossRef]
- Sakata, Y.; Zhuang, H.; Kwansa, H.; Koehler, R.C.; Doré, S. Resveratrol protects against experimental stroke: putative neuroprotective role of heme oxygenase 1. Exp. Neurol. 2010, 224, 325–329. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Anti-oxidative action of resveratrol: Implications for human health. Arab. J. Chem. 2011, 4, 293–298. [Google Scholar] [CrossRef]
- Cheng, C.K.; Luo, J.; Lau, C.W.; Chen, Z.; Tian, X.Y.; Huang, Y. Pharmacological Basis and New Insights of Resveratrol Action in the Cardiovascular System. Br. J. Pharmacol. 2019. [Google Scholar] [CrossRef]
- Magyar, K.; Halmosi, R.; Palfi, A.; Feher, G.; Czopf, L.; Fulop, A.; Battyany, I.; Sumegi, B.; Toth, K.; Szabados, E. Cardioprotection by resveratrol: A human clinical trial in patients with stable coronary artery disease. Clin. Hemorheol. Microcirc. 2012, 50, 179–187. [Google Scholar]
- Romain, C.; Gaillet, S.; Carillon, J.; Vidé, J.; Ramos, J.; Izard, J.-C.; Cristol, J.-P.; Rouanet, J.-M. Vineatrol and Cardiovascular Disease: Beneficial Effects of a Vine-Shoot Phenolic Extract in a Hamster Atherosclerosis Model. J. Agric. Food Chem. 2012, 60, 11029–11036. [Google Scholar] [CrossRef] [PubMed]
- Fujitaka, K.; Otani, H.; Jo, F.; Jo, H.; Nomura, E.; Iwasaki, M.; Nishikawa, M.; Iwasaka, T.; Das, D.K. Modified resveratrol Longevinex improves endothelial function in adults with metabolic syndrome receiving standard treatment. Nutr. Res. 2011, 31, 842–847. [Google Scholar] [CrossRef] [PubMed]
- D’Archivio, M.; Filesi, C.; Varì, R.; Scazzocchio, B.; Masella, R. Bioavailability of the Polyphenols: Status and Controversies. Int. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Sang, S. Metabolism and pharmacokinetics of resveratrol and pterostilbene. BioFactors 2018, 44, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Gonzálvez, M.; Larrosa, M.; García-Almagro, F.J.; Avilés-Plaza, F.; Parra, S.; Yáñez-Gascón, M.J.; Ruiz-Ros, J.A.; García-Conesa, M.T.; Tomás-Barberán, F.A. Consumption of a grape extract supplement containing resveratrol decreases oxidized LDL and A po B in patients undergoing primary prevention of cardiovascular disease: A triple-blind, 6-month follow-up, placebo-controlled, randomized trial. Mol. Nutr. Food Res. 2012, 56, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Gonzálvez, M.; Larrosa, M.; Yáñez-Gascón, M.J.; García-Almagro, F.J.; Ruiz-Ros, J.A.; García-Conesa, M.T.; Tomás-Barberán, F.A.; Espin, J.C. One-Year Consumption of a Grape Nutraceutical Containing Resveratrol Improves the Inflammatory and Fibrinolytic Status of Patients in Primary Prevention of Cardiovascular Disease. Am. J. Cardiol. 2012, 110, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Aluyen, J.K.; Ton, Q.N.; Tran, T.; Yang, A.E.; Gottlieb, H.B.; Bellanger, R.A. Resveratrol: Potential as Anticancer Agent. J. Diet. Suppl. 2012, 9, 45–56. [Google Scholar] [CrossRef]
- Peng, X.L.; Qu, W.; Wang, L.Z.; Huang, B.Q.; Ying, C.J.; Sun, X.F.; Hao, L.P. Resveratrol Ameliorates High Glucose and High-Fat/Sucrose Diet-Induced Vascular Hyperpermeability Involving Cav-1/eNOS Regulation. PLOS ONE 2014, 9, e113716. [Google Scholar] [CrossRef]
- Oh, W.Y.; Shahidi, F. Antioxidant activity of resveratrol ester derivatives in food and biological model systems. Food Chem. 2018, 261, 267–273. [Google Scholar] [CrossRef]
- Markoski, M.M.; Garavaglia, J.; Oliveira, A.; Olivaes, J.; Marcadenti, A. Molecular Properties of Red Wine Compounds and Cardiometabolic Benefits. Nutr. Metab. Insights 2016, 9, 51–57. [Google Scholar] [CrossRef]
- Feng, W.; Hao, Z.; Li, M. Isolation and Structure Identification of Flavonoids; Justino, G.C., Ed.; IntechOpen: London, UK, 2017; pp. 17–43. [Google Scholar]
- Gutiérrez-Grijalva, E.; Picos-Salas, M.; Leyva-López, N.; Criollo-Mendoza, M.; Vazquez-Olivo, G.; Heredia, J. Flavonoids and phenolic acids from oregano: occurrence, biological activity and health benefits. Plants 2018, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Escobar-Cévoli, R.; Castro-Espín, C.; Béraud, V.; Buckland, G.; Zamora-Ros, R.; Béraud, G.B.V. An Overview of Global Flavonoid Intake and its Food Sources. Flavonoids Biosynth. Hum. Health 2017. [Google Scholar]
- Gutiérrez-Grijalva, E.P. 1. Review: dietary phenolic compounds, health benefits and bioaccessibility. Arch. Latinoam. Nutr. 2016, 66. [Google Scholar]
- Mozaffarian, D.; Wu, J.H. Flavonoids, Dairy Foods, and Cardiovascular and Metabolic Health: A Review of Emerging Biologic Pathways. Circ. Res. 2018, 122, 369–384. [Google Scholar] [CrossRef]
- Afroz, R.; Tanvir, E.; Little, P. Honey-derived flavonoids: natural products for the prevention of atherosclerosis and cardiovascular diseases. Clin. Exp. Pharmacol. 2016, 6. [Google Scholar]
- Bondonno, C.P.; Croft, K.D.; Ward, N.; Considine, M.J.; Hodgson, J.M. Dietary flavonoids and nitrate: effects on nitric oxide and vascular function. Nutr. Rev. 2015, 73, 216–235. [Google Scholar] [CrossRef]
- Parasuraman, S.; David, A.V.A.; Arulmoli, R. Overviews of biological importance of quercetin: A bioactive flavonoid. Pharmacogn. Rev. 2016, 10, 84–89. [Google Scholar] [CrossRef]
- Dalgaard, F.; Bondonno, N.; Murray, K.; Bondonno, C.P.; Lewis, J.R.; Croft, K.D.; Kyrø, C.; Gislason, G.; Scalbert, A.; Cassidy, A. Higher Habitual Flavonoid Intake Is Associated with Lower Atherosclerotic. Cardiovasc. Dis. Hosp. 2019.
- Kiokias, S.; Proestos, C.; Oreopoulou, V. Effect of Natural Food Antioxidants against LDL and DNA Oxidative Changes. Antioxidants 2018, 7, 133. [Google Scholar] [CrossRef]
- Kustrin, S.; Hettiarachchi, C.; Morton, D.; Ražić, S. Analysis of phenolics in wine by high performance thin-layer chromatography with gradient elution and high resolution plate imaging. J. Pharm. Biomed. Anal. 2014, 102C, 93–99. [Google Scholar]
- Barnaba, C.; Dellacassa, E.; Nicolini, G.; Nardin, T.; Malacarne, M.; Larcher, R. Identification and quantification of 56 targeted phenols in wines, spirits, and vinegars by online solid-phase extraction – ultrahigh-performance liquid chromatography – quadrupole-orbitrap mass spectrometry. J. Chromatogr. A 2015, 1423, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Banegil, M.; Hurtado-sánchez, M.C.; Galeano-Díaz, T.; Durán-Merás, I. Front-face fluorescence spectroscopy combined with second-order multivariate algorithms for the quantification of polyphenols in red wine samples. Food Chem. 2016, 220. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Kaur, M.; Silakari, O. Flavones: An important scaffold for medicinal chemistry. Eur. J. Med. Chem. 2014, 84, 206–239. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; He, J.; Gao, Y.; Xing, Y.; Hou, J.; Tian, J.J.C.t. Preventive Effect of Total Flavones of Choerospondiasaxillaries on Ischemia/Reperfusion-Induced Myocardial Infarction-Related MAPK Signaling Pathway. Cardiovasc. Toxicol. 2014, 14, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Keen, C.L.; Kelm, M. Flavanols and cardiovascular disease prevention. Eur. Hear. J. 2010, 31, 2583–2592. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Sanchez, I.; Maya, L.; Ceballos, G.; Villarreal, F. (-)-epicatechin activation of endothelial cell endothelial nitric oxide synthase, nitric oxide, and related signaling pathways. Hypertension 2010, 55, 1398–1405. [Google Scholar] [CrossRef]
- Perez-Vizcaino, F.; Duarte, J. Flavonols and cardiovascular disease. Mol. Asp. Med. 2010, 31, 478–494. [Google Scholar] [CrossRef]
- Annapurna, A.; Reddy, C.S.; Akondi, R.B.; Rao, S.R.C. Cardioprotective actions of two bioflavonoids, quercetin and rutin, in experimental myocardial infarction in both normal and streptozotocin-induced type I diabetic rats. J. Pharm. Pharmacol. 2009, 61, 1365–1374. [Google Scholar] [CrossRef]
- Castañeda, A.; Pacheco Hernandez, L.; Páez, E.; Rodriguez, J.; Galán-Vidal, C. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859–871. [Google Scholar] [CrossRef]
- Sánchez-Ilárduya, M.B.; Sánchez-Fernández, C.; Garmón-Lobato, S.; Abad-García, B.; Berrueta, L.A.; Gallo, B.; Vicente, F. Detection of non-coloured anthocyanin–flavanol derivatives in Rioja aged red wines by liquid chromatography–mass spectrometry. Talanta 2014, 121, 81–88. [Google Scholar] [CrossRef]
- Basalekou, M.; Kyraleou, M.; Pappas, C.; Tarantilis, P.; Kotseridis, Y.; Kallithraka, S. Proanthocyanidin content as an astringency estimation tool and maturation index in red and white winemaking technology. Food Chem. 2019, 299, 125135. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-M.; Yoon, Y.; Yoon, H.; Park, H.-M.; Song, S.; Yeum, K.-J.J.N. Dietary anthocyanins against obesity and inflammation. Nutrients 2017, 9, 1089. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; Bertoia, M.; Chiuve, S.; Flint, A.; Forman, J.; Rimm, E.B. Habitual intake of anthocyanins and flavanones and risk of cardiovascular disease in men. Am. J. Clin. Nutr. 2016, 104, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; Mukamal, K.J.; Liu, L.; Franz, M.; Eliassen, A.H.; Rimm, E.B. High anthocyanin intake is associated with a reduced risk of myocardial infarction in young and middle-aged women. Circulation 2013, 127, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; Rimm, E.B.; O’Reilly, É.J.; Logroscino, G.; Kay, C.; Chiuve, S.E.; Rexrode, K.M. Dietary flavonoids and risk of stroke in women. Stroke 2012, 43, 946–951. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Huang, X.; Zhang, Y.; Wang, Y.; Liu, Y.; Sun, R.; Xia, M. Anthocyanin Supplementation Improves HDL-Associated Paraoxonase 1 Activity and Enhances Cholesterol Efflux Capacity in Subjects With Hypercholesterolemia. J. Clin. Endocrinol. Metab. 2014, 99, 561–569. [Google Scholar] [CrossRef] [PubMed]
- McCullough, M.L.; Peterson, J.J.; Patel, R.; Jacques, P.F.; Shah, R.; Dwyer, J.T. Flavonoid intake and cardiovascular disease mortality in a prospective cohort of US adults. Am. J. Clin. Nutr. 2012, 95, 454–464. [Google Scholar] [CrossRef]
- Cassidy, A.; Rogers, G.; Peterson, J.J.; Dwyer, J.T.; Lin, H.; Jacques, P.F. Higher dietary anthocyanin and flavonol intakes are associated with anti-inflammatory effects in a population of US adults. Am. J. Clin. Nutr. 2015, 102, 172–181. [Google Scholar] [CrossRef]
- Huang, W.-Y.; Liu, Y.-M.; Wang, J.; Wang, X.-N.; Li, C.-Y. Anti-Inflammatory Effect of the Blueberry Anthocyanins Malvidin-3-Glucoside and Malvidin-3-Galactoside in Endothelial Cells. Molecules 2014, 19, 12827–12841. [Google Scholar] [CrossRef]
- Chira, K.; Pacella, N.; Jourdes, M.; Teissedre, P.-L. Chemical and sensory evaluation of Bordeaux wines (Cabernet-Sauvignon and Merlot) and correlation with wine age. Food Chem. 2011, 126, 1971–1977. [Google Scholar] [CrossRef]
- Ghosh, D.J.I.J.P.R.R. Tannins from foods to combat diseases. Int. J. Pharma Res. Rev. 2015, 4, 40–44. [Google Scholar]
- De Morais Cardoso, L.; Pinheiro, S.S.; Martino, H.S.D.; Pinheiro-Sant’Ana, H.M. Sorghum (Sorghum bicolor L.): Nutrients, bioactive compounds, and potential impact on human health. Crit. Rev. Food Sci. Nutr. 2017, 57, 372–390. [Google Scholar] [CrossRef] [PubMed]
- Stark, T.; Wollmann, N.; Wenker, K.; Lösch, S.; Glabasnia, A.; Hofmann, T. Matrix-Calibrated LC-MS/MS Quantitation and Sensory Evaluation of Oak Ellagitannins and Their Transformation Products in Red Wines. J. Agric. Food Chem. 2010, 58, 6360–6369. [Google Scholar] [CrossRef] [PubMed]
- García-Estévez, I.; Escribano-Bailón, M.T.; Rivas-Gonzalo, J.-C.; Alcalde-Eon, C. Validation of a Mass Spectrometry Method To Quantify Oak Ellagitannins in Wine Samples. J. Agric. Food Chem. 2012, 60, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Jourdes, M.; Michel, J.; Saucier, C.; Quideau, S.; Teissedre, P.-L. Identification, amounts, and kinetics of extraction of C-glucosidic ellagitannins during wine aging in oak barrels or in stainless steel tanks with oak chips. Anal. Bioanal. Chem. 2011, 401, 1531–1539. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Guo, A.; Zhang, Y.; Wang, H.; Liu, Y.; Li, H.J. A review on astringency and bitterness perception of tannins in wine. Trends Food Sci. Technol. 2014, 40, 6–19. [Google Scholar] [CrossRef]
- Lu, S.-C.; Liao, W.-R.; Chen, S.-F. Quantification of Trans-resveratrol in Red Wines Using QuEChERS Extraction Combined with Liquid Chromatography–Tandem Mass Spectrometry. Anal. Sci. 2018, 34, 439–444. [Google Scholar] [CrossRef]
- Guerrero, R.F.; García-Parrilla, M.C.; Puertas, B.; Cantos-Villar, E. Wine, resveratrol and health: a review. Nat. Prod. Commun. 2009, 4, 1934578X0900400503. [Google Scholar] [CrossRef]
- Caruana, M.; Cauchi, R.; Vassallo, N. Putative Role of Red Wine Polyphenols against Brain Pathology in Alzheimer’s and Parkinson’s Disease. Front. Nutr. 2016, 3, 31. [Google Scholar] [CrossRef]
- Chorti, E.; Guidoni, S.; Ferrandino, A.; Novello, V.J. Effect of different cluster sunlight exposure levels on ripening and anthocyanin accumulation in Nebbiolo grapes. American Society for Enology and Viticulture 2010, 61, 23–30. [Google Scholar]
- Xu, C.; Zhang, Y.; Zhu, L.; Huang, Y.; Lu, J.J. Influence of growing season on phenolic compounds and antioxidant properties of grape berries from vines grown in subtropical climate. J. Agric. Food Chem. 2011, 59, 1078–1086. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, F.; Ning, J.; Liu, X.; Zhang, Z.; Yang, S. Predicting the anthocyanin content of wine grapes by NIR hyperspectral imaging. Food Chem. 2015, 172, 788–793. [Google Scholar] [CrossRef] [PubMed]
- Haseeb, S.; Alexander, B.; Baranchuk, A. Wine and cardiovascular health: A comprehensive review. Circulation 2017, 136, 1434–1448. [Google Scholar] [CrossRef] [PubMed]
- Casassa, L.F.; Harbertson, J.F. Extraction, Evolution, and Sensory Impact of Phenolic Compounds During Red Wine Maceration. Annu. Rev. Food Sci. Technol. 2014, 5, 83–109. [Google Scholar] [CrossRef] [PubMed]
- Saucier, C. How do wine polyphenols evolve during wine ageing? Cerevisia 2010, 35, 11–15. [Google Scholar] [CrossRef]
- Muccillo, L.; Gambuti, A.; Frusciante, L.; Iorizzo, M.; Moio, L.; Raieta, K.; Rinaldi, A.; Colantuoni, V.; Aversano, R. Biochemical features of native red wines and genetic diversity of the corresponding grape varieties from Campania region. Food Chem. 2014, 143, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Niculescu, V.-C.; Paun, N.; Ionete, R.-E. The Evolution of Polyphenols from Grapes to Wines; Justino, G.C., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Sanz, M.; De Simón, B.F.; Esteruelas, E.; Muñoz, Á.M.; Cadahía, E.; Hernández, M.T.; Estrella, I.; Martínez, J.; De Simón, M.B.F. Polyphenols in red wine aged in acacia (Robinia pseudoacacia) and oak (Quercus petraea) wood barrels. Anal. Chim. Acta 2012, 732, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Arriola, L.; Martinez-Camblor, P.; Larrañaga, N.; Basterretxea, M.; Amiano, P.; Moreno-Iribas, C.; Carracedo, R.; Agudo, A.; Ardanaz, E.; Barricarte, A. Alcohol intake and the risk of coronary heart disease in the Spanish EPIC cohort study. Heart 2010, 96, 124–130. [Google Scholar] [CrossRef]
- E Ronksley, P.; E Brien, S.; Turner, B.J.; Mukamal, K.J.; A Ghali, W. Association of alcohol consumption with selected cardiovascular disease outcomes: a systematic review and meta-analysis. BMJ 2011, 342, d671. [Google Scholar] [CrossRef]
- Djoussé, L.; Lee, I.-M.; Buring, J.E.; Gaziano, J.M. Alcohol consumption and risk of cardiovascular disease and mortality in women: potential mediating mechanisms. Circulation 2009, 120, 237. [Google Scholar] [CrossRef]
- Pavlidou, E.; Mantzorou, M.; Fasoulas, A.; Tryfonos, C.; Petridis, D.; Giaginis, C. Wine: An Aspiring Agent in Promoting Longevity and Preventing Chronic Diseases. Diseases 2018, 6, 73. [Google Scholar] [CrossRef] [PubMed]
- Da Luz, P.L.; Coimbra, S.; Favarato, D.; Albuquerque, C.; Mochiduky, R.I.; Rochitte, C.E.; Hojaij, E.; Gonsalves, C.R.L.; Laurindo, F.R. Coronary artery plaque burden and calcium scores in healthy men adhering to long-term wine drinking or alcohol abstinence %J Brazilian. J. Med. Biol. Res. 2014, 47, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Marques-Vidal, P.; Bochud, M.; Paccaud, F.; Waterworth, D.; Bergmann, S.; Preisig, M.; Waeber, G.; Vollenweider, P. No interaction between alcohol consumption and HDL-related genes on HDL cholesterol levels. Atherosclerosis 2010, 211, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Kim, K. Association of Alcohol Consumption with Lipid Profile in Hypertensive Men. Alcohol Alcohol. 2012, 47, 282–287. [Google Scholar] [CrossRef]
- Magnus, P.; Bakke, E.; Hoff, D.A.; Høiseth, G.; Graff-Iversen, S.; Peggy Knudsen, G.; Myhre, R.; Trygve Normann, P.; Næss, Ø.; Tambs, K. Controlling for high-density lipoprotein cholesterol does not affect the magnitude of the relationship between alcohol and coronary heart disease. Circulation 2011, 124, 2296–2302. [Google Scholar] [CrossRef] [PubMed]
- E Brien, S.; E Ronksley, P.; Turner, B.J.; Mukamal, K.J.; A Ghali, W. Effect of alcohol consumption on biological markers associated with risk of coronary heart disease: systematic review and meta-analysis of interventional studies. BMJ 2011, 342, d636. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Urpi-Sarda, M.; Ros, E.; Valderas-Martínez, P.; Casas, R.; Arranz, S.; Guillén, M.; Lamuela-Raventos, R.M.; Llorach, R.; Andres-Lacueva, C.; et al. Effects of red wine polyphenols and alcohol on glucose metabolism and the lipid profile: A randomized clinical trial. Clin. Nutr. 2013, 32, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Paul, K.; Bairwa, N.K.; Kazal, H.; Batta, A.J. To Study the effects of Alcohol on Lipid Profile on Basis of Amount, Type and Duration of Alcohol Consumption. Indian J. Public Health Res. Dev. 2015, 6, 302–307. [Google Scholar] [CrossRef]
- Brasnyó, P.; Molnár, G.A.; Mohás, M.; Markó, L.; Laczy, B.; Cseh, J.; Mikolás, E.; Szijártó, I.A.; Mérei, Á.; Halmai, R.; et al. Resveratrol improves insulin sensitivity, reduces oxidative stress and activates the Akt pathway in type 2 diabetic patients. Br. J. Nutr. 2011, 106, 383–389. [Google Scholar] [CrossRef]
- European Commission. Commision Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off J. Eur. Union 2006, 364, 365/5–365/24. [Google Scholar]
- Baliunas, D.O.; Taylor, B.J.; Irving, H.; Roerecke, M.; Patra, J.; Mohapatra, S.; Rehm, J. Alcohol as a risk factor for type 2 diabetes: a systematic review and meta-analysis. Diabetes Care 2009, 32, 2123–2132. [Google Scholar] [CrossRef] [PubMed]
- A Mekary, R.; Rimm, E.B.; Giovannucci, E.; Stampfer, M.J.; Willett, W.C.; Ludwig, D.S.; Hu, F.B. Joint association of glycemic load and alcohol intake with type 2 diabetes incidence in women. Am. J. Clin. Nutr. 2011, 94, 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- Ramadori, G.; Gautron, L.; Fujikawa, T.; Vianna, C.R.; Elmquist, J.K.; Coppari, R. Central administration of resveratrol improves diet-induced diabetes. Endocrinology 2009, 150, 5326–5333. [Google Scholar] [CrossRef] [PubMed]
- Tangney, C.C.; Rasmussen, H.E.J.C.a.r. Polyphenols, inflammation, and cardiovascular disease. Curr. Atheroscler. Rep. 2013, 15, 324. [Google Scholar] [CrossRef] [PubMed]
- Holthoff, J.H.; Woodling, K.A.; Doerge, D.R.; Burns, S.T.; Hinson, J.A.; Mayeux, P.R. Resveratrol, a dietary polyphenolic phytoalexin, is a functional scavenger of peroxynitrite. Biochem. Pharmacol. 2010, 80, 1260–1265. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K. Resveratrol, Wine, and Atherosclerosis. Int. J. Angiol. 2012, 21, 7–18. [Google Scholar] [CrossRef]
- Estruch, R.; Sacanella, E.; Mota, F.; Chiva-Blanch, G.; Antúnez, E.; Casals, E.; Deulofeu, R.; Rotilio, D.; Andres-Lacueva, C.; Lamuela-Raventos, R.M. Moderate consumption of red wine, but not gin, decreases erythrocyte superoxide dismutase activity: A randomised cross-over trial. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 46–53. [Google Scholar] [CrossRef]
- Chiva-Blanch, G.; Urpi-Sarda, M.; Ros, E.; Arranz, S.; Valderas-Martínez, P.; Casas, R.; Sacanella, E.; Llorach, R.; Lamuela-Raventos, R.M.; Andres-Lacueva, C.; et al. Dealcoholized Red Wine Decreases Systolic and Diastolic Blood Pressure and Increases Plasma Nitric Oxide. Circ. Res. 2012, 111, 1065–1068. [Google Scholar] [CrossRef]
- Egert, S.; Bosy-Westphal, A.; Seiberl, J.; Kürbitz, C.; Settler, U.; Plachta-Danielzik, S.; Wagner, A.E.; Frank, J.; Schrezenmeir, J.; Rimbach, G.; et al. Quercetin reduces systolic blood pressure and plasma oxidised low-density lipoprotein concentrations in overweight subjects with a high-cardiovascular disease risk phenotype: a double-blinded, placebo-controlled cross-over study. Br. J. Nutr. 2009, 102, 1065–1074. [Google Scholar] [CrossRef]
- Bulut, D.; Jelich, U.; Dacanay-Schwarz, R.; Mügge, A. Red Wine Ingestion Prevents Microparticle Formation After a Single High-Fat Meal—A Crossover Study in Healthy Humans. J. Cardiovasc. Pharmacol. 2013, 61, 489–494. [Google Scholar] [CrossRef]
- Trpkovic, A.; Resanovic, I.; Stanimirovic, J.; Radak, D.; Mousa, S.A.; Cenic-Milosevic, D.; Jevremovic, D.; Isenovic, E.R. Oxidized low-density lipoprotein as a biomarker of cardiovascular diseases. Crit. Rev. Clin. Lab. Sci. 2015, 52, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Bandeali, S.; Farmer, J. High-Density Lipoprotein and Atherosclerosis: The Role of Antioxidant Activity. Curr. Atheroscler. Rep. 2012, 14, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Lafta, M.A.J.J.N.S.R. A comparative study for some atherogenic indices in sera of myocardial infarction, ischemic heart disease patients and control. J. Nat. Sci. Res. 2014, 121–128. [Google Scholar]
| Group | Subclass | Main Representatives | Range in mg/L | Characteristic Structure |
|---|---|---|---|---|
| Non-flavonoid | ||||
| Hydroxybenzoic acids | Gallic, ellagic, parahydroxybenzoic, protocatechuic, vanillicand syringic acids | 0–218.0 | 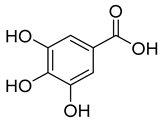 Gallic acid | |
| Hydroxycinnamic acids | Coutaric, caftaric, and fertaric acids | 60.0–334.0 |  Caffeic acid | |
| Stilbenes | Resveratrol | 0.1–7.0 | 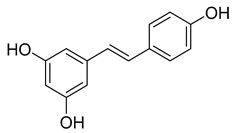 Resveratrol | |
| Flavonoids | ||||
| Flavones | Luteolin | 0.2–1.0 | 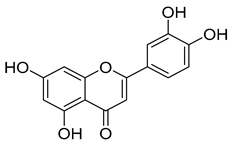 Luteolin | |
| Flavan-3-ols | Catechin and epicatechin | 50.0–120.0 | 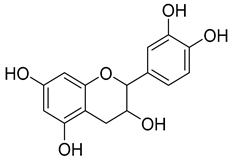 Catechin | |
| Flavonols | Myricetin, quercetin, kaempferol, and rutin | 12.7–130.0 | 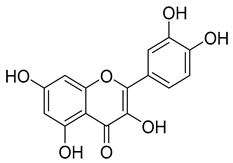 Quercetin | |
| Anthocyanins | Malvidin, cyanidin, peonidin, delphinidin, pelargonidin, petunidin | 90.0–400.0 | 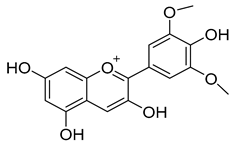 Malvidin |
| Study Model | Outcome(s) | Study Characteristics | Main Findings | References |
|---|---|---|---|---|
| Drinkers vs. abstainers | HDL-C level and changes in the coronary vasculature | Benefits of moderate consumption of red wine | Drinkers displayed a significantly higher HDL-C level and a protective effect on coronary lesions | [110] |
| Men | HDL-C level | The study included 5409 subjects categorized in abstainers, moderate, high, and very high alcohol drinkers | Alcohol consumption increased HDL-C levels | [111] |
| Hypertensive population | Lipid profile | Benefits of moderate consumption of alcohol | Alcohol consumption was negatively associated with prevalence of low HDL-C levels, whereas the prevalence of high triglyceride levels increased with increasing amounts of alcohol intake | [112] |
| Cohort study | Cardioprotective function | Increasing HDL-C levels as the mechanism used by alcohol to exert its cardioprotective function | Increasing HDL-C levels is not a relevant mechanism by which ethanol exerts its cardioprotective effect | [113] |
| Meta-analysis | Lipid profile | Effect of moderate alcohol consumption on lipid profile | Alcohol consumption increased the levels of HDL-C, apolipoprotein A1, and adiponectin but not triglycerides levels. | [114] |
| Study Model | Outcome (s) | Study Characteristics | Main Findings | References |
|---|---|---|---|---|
| Men | Glucose metabolism | Light–moderate alcohol consumption (red wine, dealcoholized red wine, and gin) | Dealcoholized red wine decreased plasma insulin and HOMA-IR values | [115] |
| Drinkers vs. abstainers | Glucose level and diabetes | Benefits of moderate consumption of red win | Drinkers showed a lower incidence of diabetes and lower glucose levels compared to abstainers | [110] |
| T2D | Glucose metabolism | Effect of resveratrol on glucose metabolism | Resveratrol improved insulin resistance and increased AKT phosphorylation | [117] |
| Meta-analysis | T2D incidence | Effect of moderate alcohol consumption on the incidence of T2D | Light–moderate alcohol consumption decreased the incidence of T2D | [119] |
| Cohort study | GL and incidence of T2D | Impact of alcohol consumption and positive association between GL and T2D | High alcohol intake (≥15 g/day) attenuates the effect of GL on T2D incidence | [120] |
| Obese and diabetic mice | Glucose metabolism | Impact of intracerebroventricular infusion of resveratrol on glucose metabolism | Normalized hyperglycemia and improved hyperinsulinemia mediated by activating SIRT 1 expressed in the brain | [121] |
| Study Model | Outcome(s) | Study Characteristics | Main Findings | References |
|---|---|---|---|---|
| Men | Lag phase time of LDL particles | Benefits of moderate consumption of red wine, dealcoholized red wine, and gin. | Red wine consumption showed increased lag phase time of LDL oxidation up to 11.0 min | [125] |
| High cardiovascular risk | Plasma nitric oxide, systolic and diastolic pressure | Effects of alcoholic and dealcoholized red wine and gin on plasma NO and blood pressure | Dealcoholized red wine reduced systolic and diastolic blood pressure and increase plasma NO concentration | [126] |
| Overweight or obese subjects | Concentrations of oxidized LDL | Changes in markers of oxidative stress following 150 mg/day of quercetin supplementation | Quercetin significantly decreased plasma concentrations of oxidized LDL | [127] |
| Men | Circulating microparticles | Benefits of moderate consumption of red win, dealcoholized red wine and gin during a high-fat meal | Red wine consumption decreased circulating microparticles | [128] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castaldo, L.; Narváez, A.; Izzo, L.; Graziani, G.; Gaspari, A.; Di Minno, G.; Ritieni, A. Red Wine Consumption and Cardiovascular Health. Molecules 2019, 24, 3626. https://doi.org/10.3390/molecules24193626
Castaldo L, Narváez A, Izzo L, Graziani G, Gaspari A, Di Minno G, Ritieni A. Red Wine Consumption and Cardiovascular Health. Molecules. 2019; 24(19):3626. https://doi.org/10.3390/molecules24193626
Chicago/Turabian StyleCastaldo, Luigi, Alfonso Narváez, Luana Izzo, Giulia Graziani, Anna Gaspari, Giovanni Di Minno, and Alberto Ritieni. 2019. "Red Wine Consumption and Cardiovascular Health" Molecules 24, no. 19: 3626. https://doi.org/10.3390/molecules24193626
APA StyleCastaldo, L., Narváez, A., Izzo, L., Graziani, G., Gaspari, A., Di Minno, G., & Ritieni, A. (2019). Red Wine Consumption and Cardiovascular Health. Molecules, 24(19), 3626. https://doi.org/10.3390/molecules24193626









