Broccoli or Sulforaphane: Is It the Source or Dose That Matters?
Abstract
1. Introduction
1.1. Epidemiology of Broccoli and Health
1.2. Discovery of Sulforaphane as a Bioactive Isothiocyanate
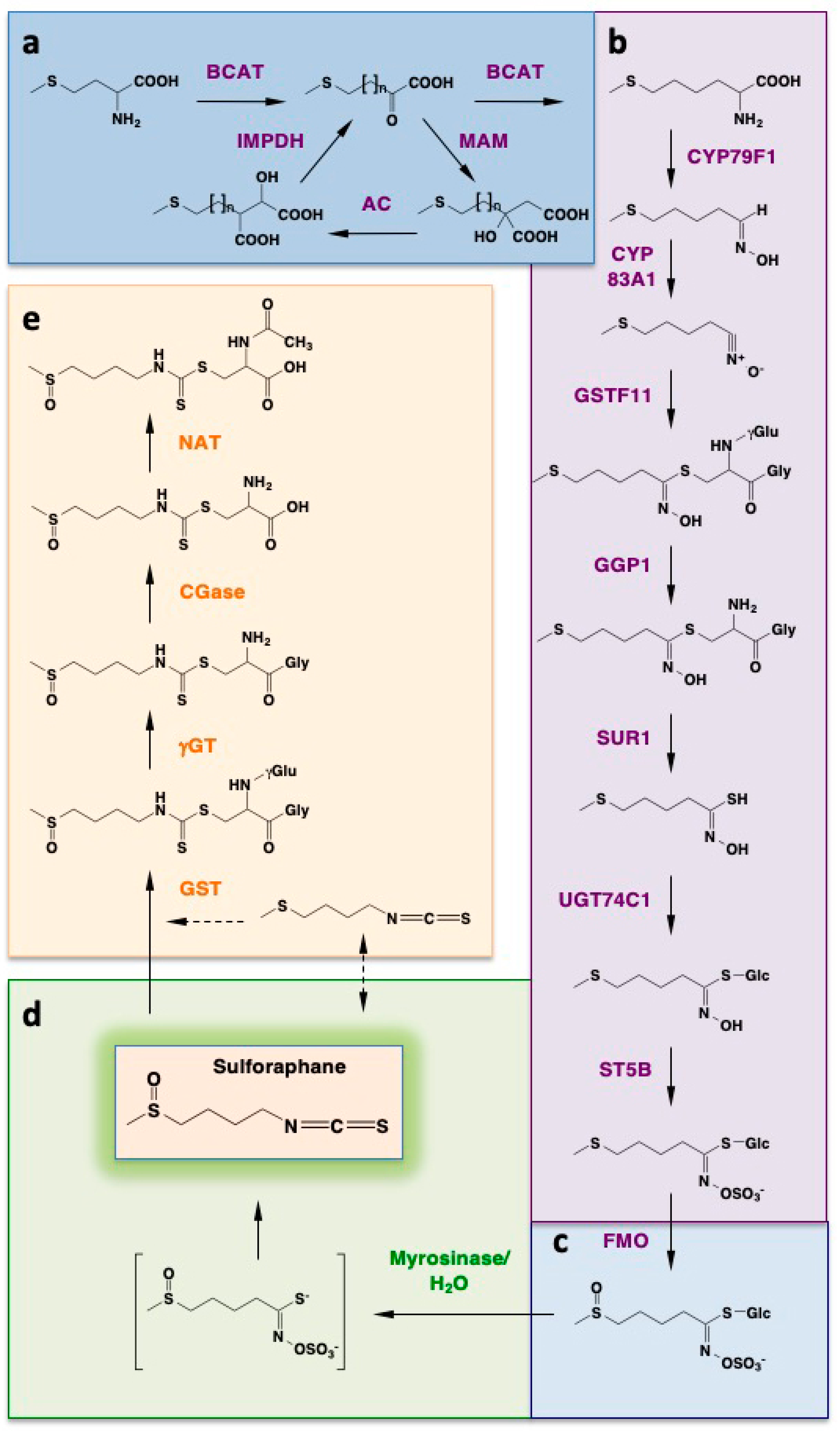
1.3. Biosynthesis and Function of Glucoraphanin in Broccoli
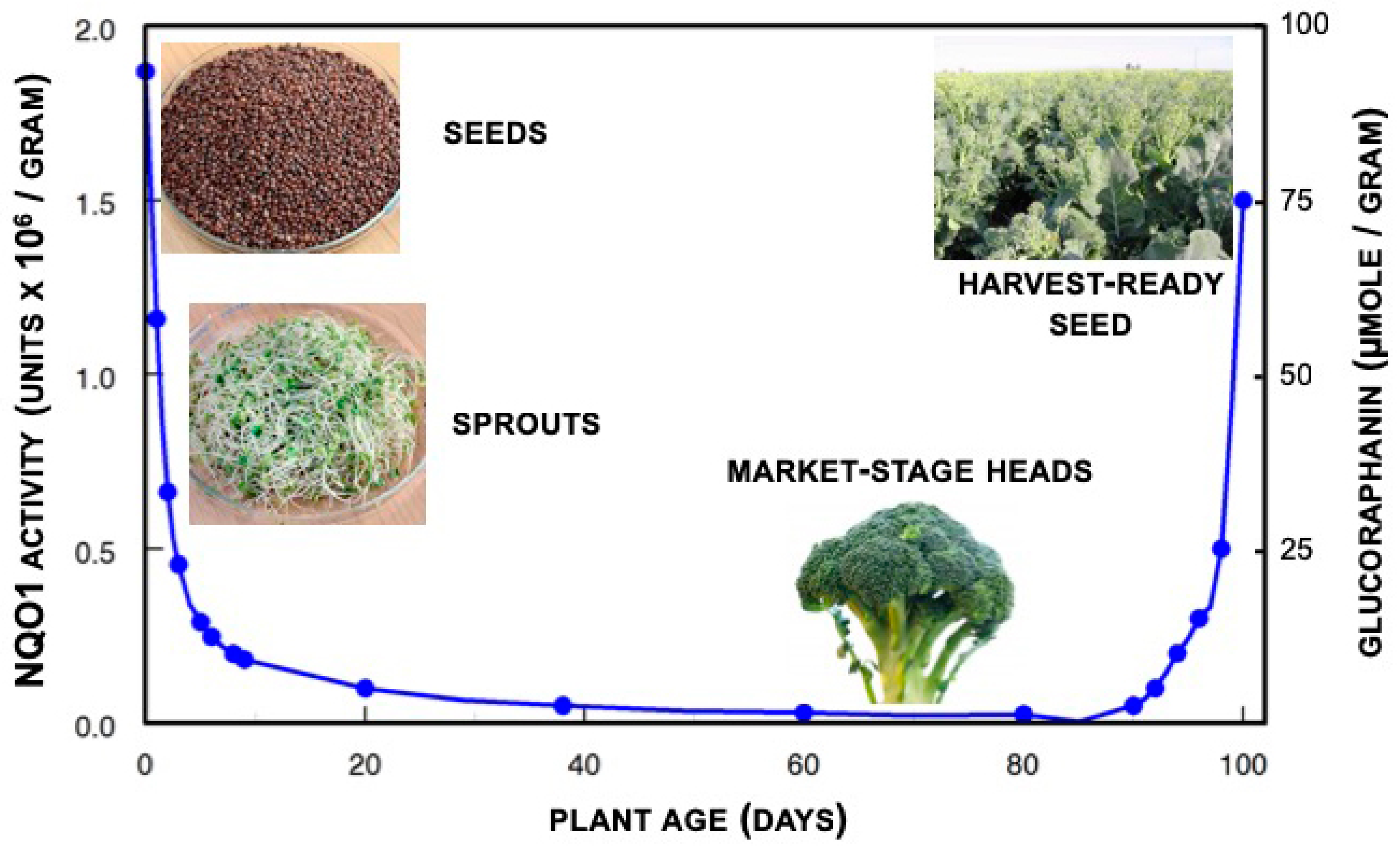
1.4. Glucoraphanin Levels in Broccoli
2. Broccoli-Based Intervention in Rodents
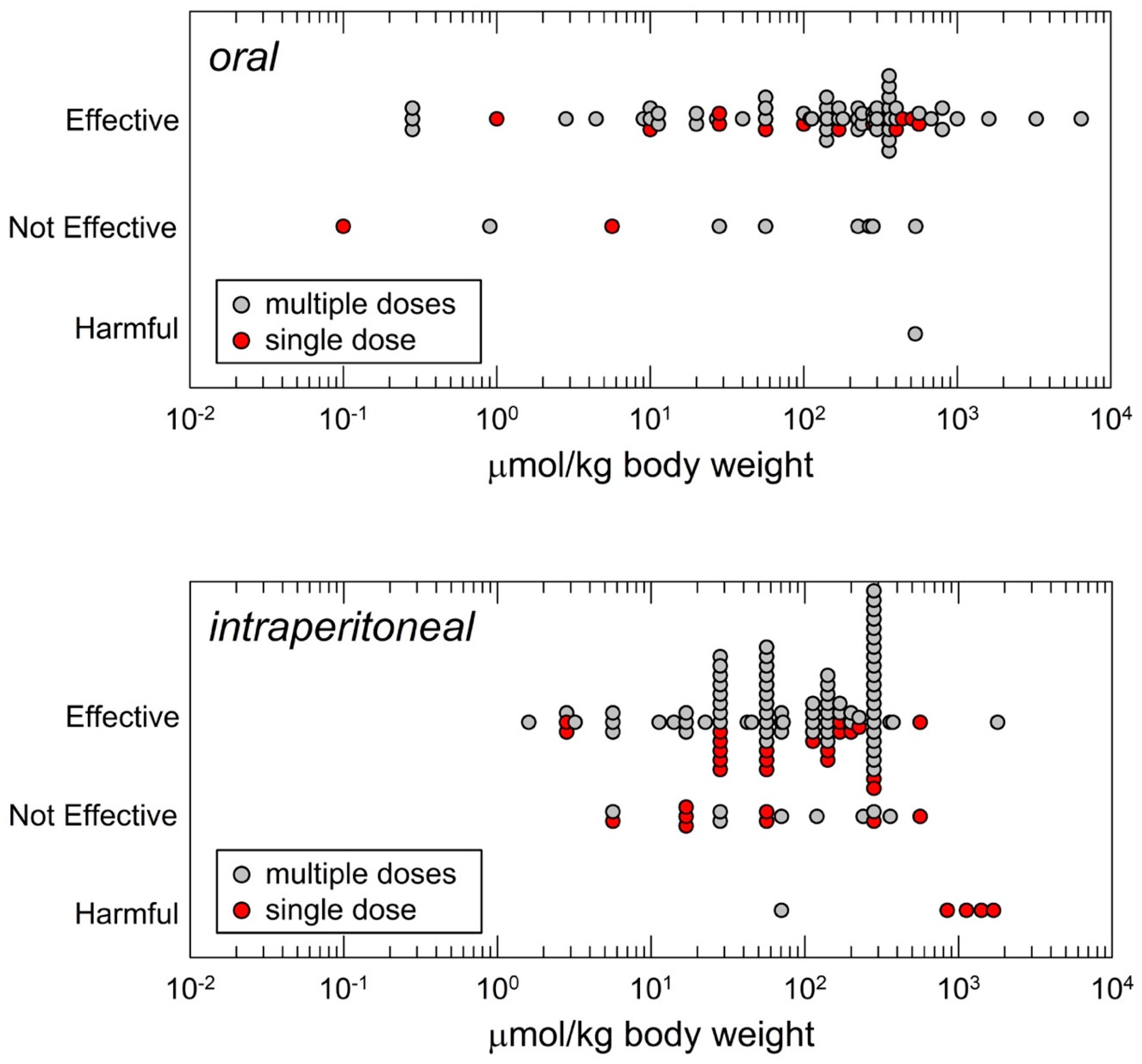
2.1. Formulation, Route of Administration and Dose
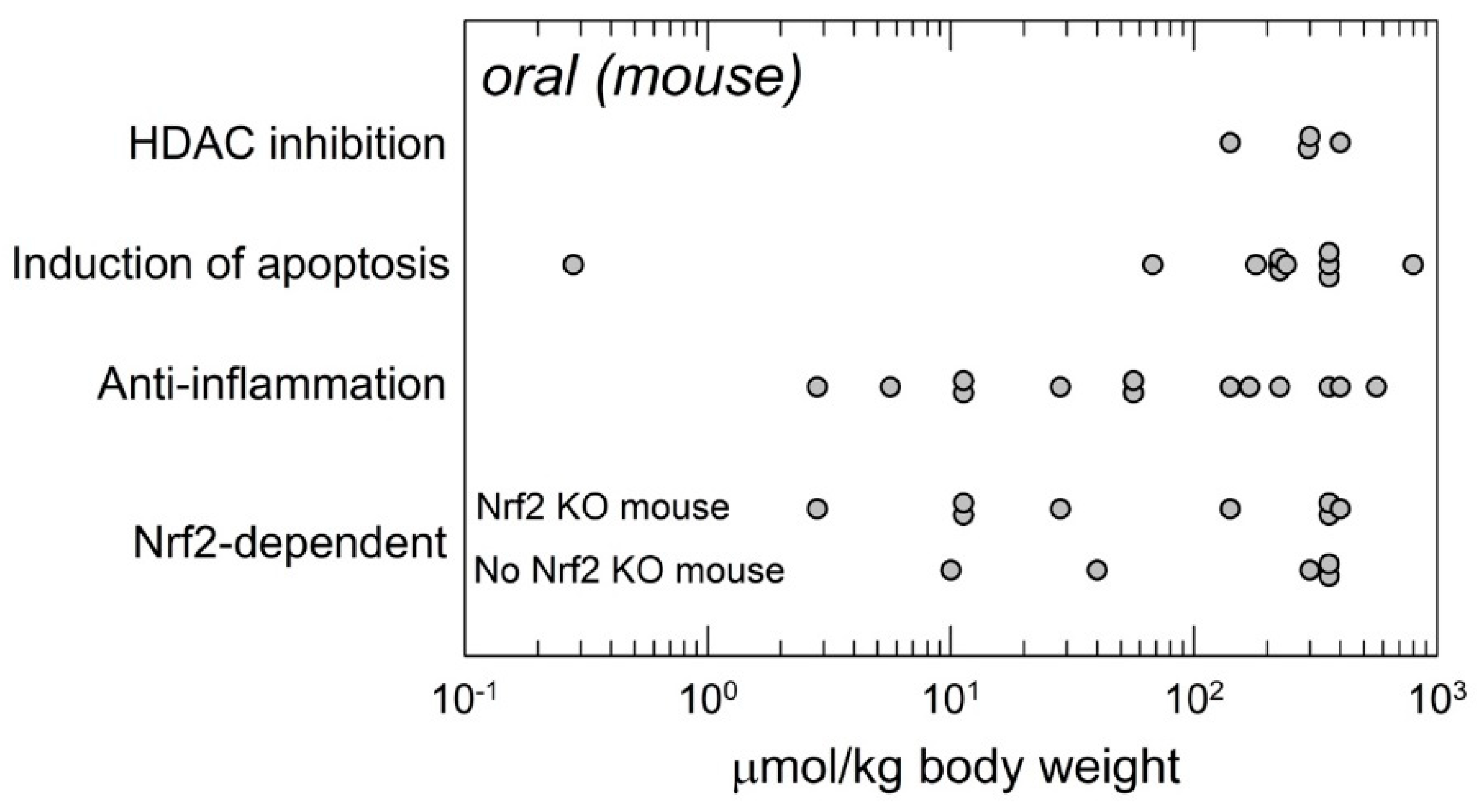
2.2. Efficacy Endpoints: Mechanisms Versus Dose, Risks and Benefits
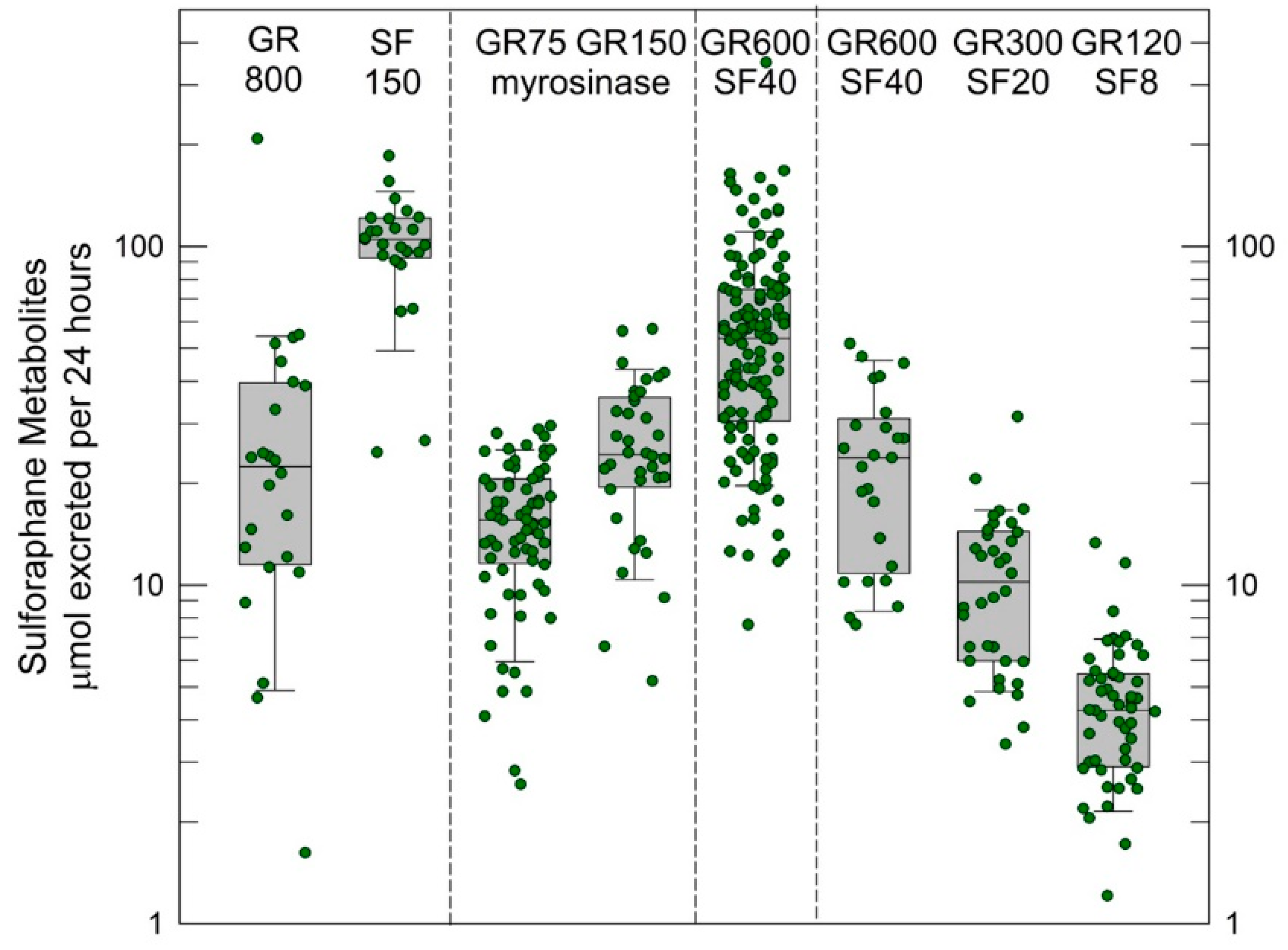
3. Broccoli-Based Clinical Trials
3.1. Sulforaphane Pharmacokinetics and Pharmacodynamics
3.2. Clinical Studies with Broccoli-Based Preparations: Efficacy
3.2.1. Aflatoxin Toxicity
3.2.2. Air Pollution Detoxification
3.2.3. Arthritis
3.2.4. Asthma and Atopic Allergic Responses
3.2.5. Cancer Biomarkers
3.2.6. COPD
3.2.7. CVD
3.2.8. Diabetes, Metabolic Syndrome, and Related Disorders
3.2.9. General
3.2.10. Helicobacter Pylori Infection
3.2.11. NASH/NAFLD
3.2.12. Neurodegenerative Conditions
3.2.13. Neurodevelopmental Conditions
3.2.14. Sickle Cell Disease
3.3. Summary of Clinical Studies with Sulforaphane, Glucoraphanin or Mixtures
4. Challenges Ahead
4.1. Plants Versus Discrete Isolates: Foods vs. Supplements
4.1.1. First
4.1.2. Second
4.1.3. Third
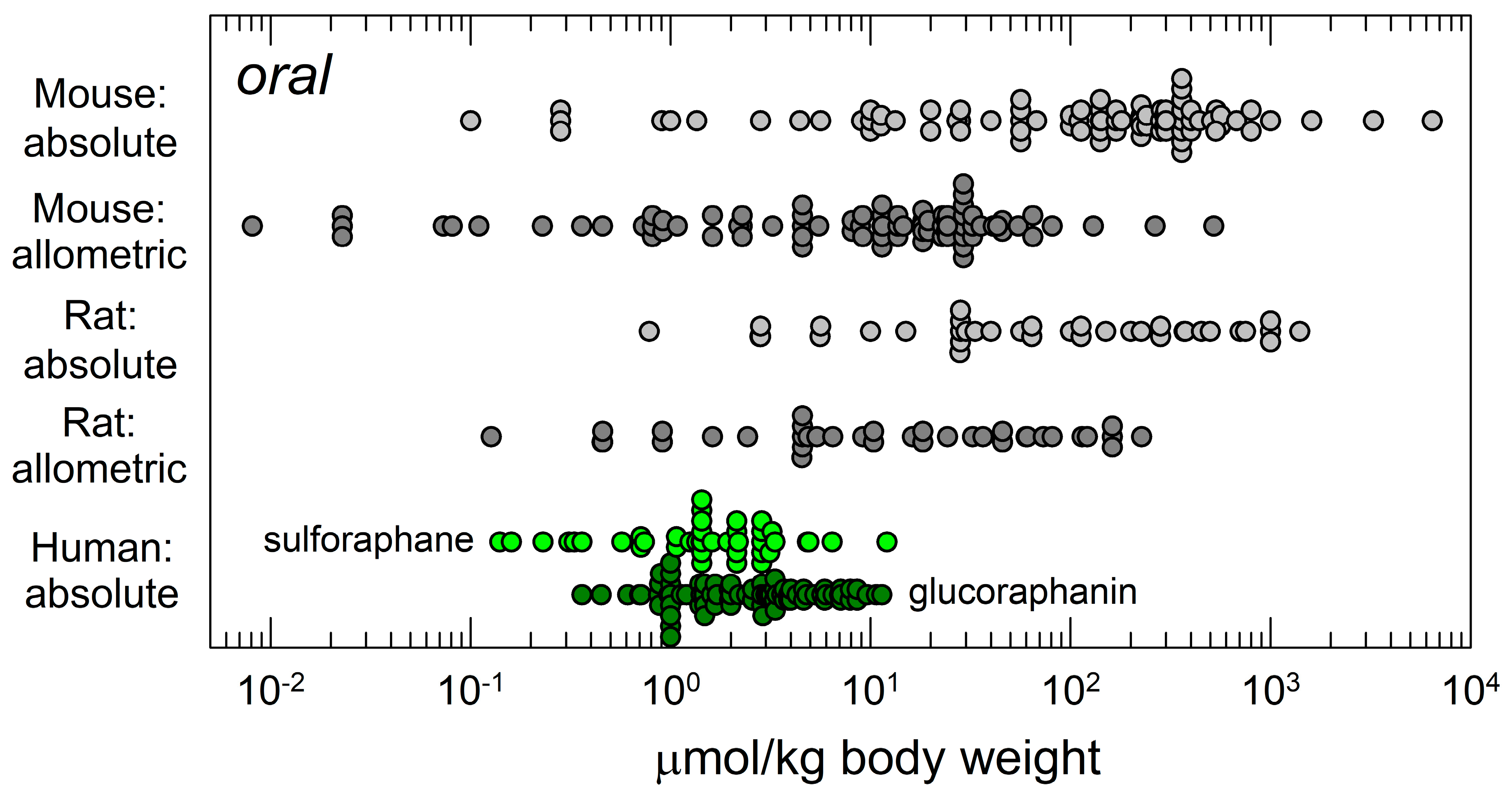
4.2. Optimizing the Definition of Dose
4.3. Integrating Animal and Clinical Studies
4.4. Better Efficacy Biomarkers
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest

Dedication
References
- Fahey, J. Brassica: Characteristics and properties. Encycl. Food Health 2016, 1, 469–477. [Google Scholar]
- Graham, S.; Dayal, H.; Swanson, M.; Mittelman, A.; Wilkinson, G. Diet in the epidemiology of cancer of the colon and rectum. J. Natl. Cancer Inst. 1978, 61, 709–714. [Google Scholar] [PubMed]
- Michaud, D.S.; Spiegelman, D.; Clinton, S.K.; Rimm, E.B.; Willett, W.C.; Giovannucci, E.L. Fruit and vegetable intake and incidence of bladder cancer in a male prospective cohort. J. Natl. Cancer Inst. 1999, 91, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.H.; Kristal, A.R.; Stanford, J.L. Fruit and vegetable intakes and prostate cancer risk. J. Natl. Cancer Inst. 2000, 92, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Panjwani, A.A.; Liu, H.; Fahey, J.W. Crucifers and related vegetables and supplements for neurologic disorders: What is the evidence? Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 451–457. [Google Scholar] [CrossRef]
- Benedict, A.L.; Mountney, A.; Hurtado, A.; Bryan, K.E.; Schnaar, R.L.; Dinkova-Kostova, A.T.; Talalay, P. Neuroprotective effects of sulforaphane after contusive spinal cord injury. J. Neurotrauma 2012, 29, 2576–2586. [Google Scholar] [CrossRef] [PubMed]
- Leone, A.; Diorio, G.; Sexton, W.; Schell, M.; Alexandrow, M.; Fahey, J.W.; Kumar, N.B. Sulforaphane for the chemoprevention of bladder cancer: Molecular mechanism targeted approach. Oncotarget 2017, 8, 35412–35424. [Google Scholar] [CrossRef]
- Palliyaguru, D.L.; Yuan, J.M.; Kensler, T.W.; Fahey, J.W. Isothiocyanates: Translating the power of plants to people. Mol. Nutr. Food Res. 2018, 62, e1700965. [Google Scholar] [CrossRef]
- Dayalan Naidu, S.; Suzuki, T.; Yamamoto, M.; Fahey, J.W.; Dinkova-Kostova, A.T. Phenethyl isothiocyanate, a dual activator of transcription factors NRF2 and HSF1. Mol. Nutr. Food Res. 2018, 62, e1700908. [Google Scholar] [CrossRef]
- Interactive Cancer Risk Matrix. 2019. Available online: https://www.wcrf.org/dietandcancer/interactive-cancer-risk-matrix (accessed on 6 October 2019).
- Fahey, J.W.; Haristoy, X.; Dolan, P.M.; Kensler, T.W.; Scholtus, I.; Stephenson, K.K.; Talalay, P.; Lozniewski, A. Sulforaphane inhibits extracellular, intracellular, and antibiotic-resistant strains of Helicobacter pylori and prevents benzo[a]pyrene-induced stomach tumors. Proc. Natl. Acad. Sci. USA 2002, 99, 7610–7615. [Google Scholar] [CrossRef]
- Fahey, J.W.; Talalay, P. Antioxidant functions of sulforaphane: A potent inducer of Phase II detoxication enzymes. Food Chem. Toxicol. 1999, 37, 973–979. [Google Scholar] [CrossRef]
- Zhang, Y.; Talalay, P.; Cho, C.G.; Posner, G.H. A major inducer of anticarcinogenic protective enzymes from broccoli: Isolation and elucidation of structure. Proc. Natl. Acad. Sci. USA 1992, 89, 2399–2403. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kensler, T.W.; Cho, C.G.; Posner, G.H.; Talalay, P. Anticarcinogenic activities of sulforaphane and structurally related synthetic norbornyl isothiocyanates. Proc. Natl. Acad. Sci. USA 1994, 91, 3147–3150. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Zhang, Y.; Talalay, P. Broccoli sprouts: An exceptionally rich source of inducers of enzymes that protect against chemical carcinogens. Proc. Natl. Acad. Sci. USA 1997, 94, 10367–10372. [Google Scholar] [CrossRef] [PubMed]
- Posner, G.H.; Cho, C.G.; Green, J.V.; Zhang, Y.; Talalay, P. Design and synthesis of bifunctional isothiocyanate analogs of sulforaphane: Correlation between structure and potency as inducers of anticarcinogenic detoxication enzymes. J. Med. Chem. 1994, 37, 170–176. [Google Scholar] [CrossRef]
- Fahey, J.W.; Dinkova-Kostova, A.T.; Stephenson, K.K.; Talalay, P. The “Prochaska” microtiter plate bioassay for inducers of NQO1. Methods Enzym. 2004, 382, 243–258. [Google Scholar]
- Dinkova-Kostova, A.T.; Fahey, J.W.; Kostov, R.V.; Kensler, T.W. KEAP1 and done? Targeting the NRF2 pathway with sulforaphane. Trends Food Sci. Technol. 2017, 69, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Dinkova-Kostova, A.T.; Kostov, R.V. Glucosinolates and isothiocyanates in health and disease. Trends Mol. Med. 2012, 18, 337–347. [Google Scholar] [CrossRef]
- Cuadrado, A.; Rojo, A.I.; Wells, G.; Hayes, J.D.; Cousin, S.P.; Rumsey, W.L.; Attucks, O.C.; Franklin, S.; Levonen, A.L.; Kensler, T.W.; et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019, 18, 295–317. [Google Scholar] [CrossRef]
- Jiang, X.; Liu, Y.; Ma, L.X.; Ji, R.; Qu, Y.Q.; Xin, Y.; Lv, G.Y. Chemopreventive activity of sulforaphane. Drug Des. Dev. 2018, 12, 2905–2913. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, L. Discovery and development of sulforaphane as a cancer chemopreventive phytochemical. Acta Pharmacol. Sin. 2007, 28, 1343–1354. [Google Scholar] [CrossRef] [PubMed]
- Sonderby, I.E.; Geu-Flores, F.; Halkier, B.A. Biosynthesis of glucosinolates--gene discovery and beyond. Trends Plant. Sci 2010, 15, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, M.D.; Olsen, C.E.; Halkier, B.A. Production of the cancer-preventive glucoraphanin in tobacco. Mol. Plant. 2010, 3, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Huseby, S.; Koprivova, A.; Lee, B.R.; Saha, S.; Mithen, R.; Wold, A.B.; Bengtsson, G.B.; Kopriva, S. Diurnal and light regulation of sulphur assimilation and glucosinolate biosynthesis in Arabidopsis. J. Exp. Bot. 2013, 64, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, T.A.; Fahey, J.W.; Wade, K.L.; Stephenson, K.K.; Talalay, P. Human metabolism and excretion of cancer chemoprotective glucosinolates and isothiocyanates of cruciferous vegetables. Cancer Epidemiol. Biomark. Prev. 1998, 7, 1091–1100. [Google Scholar]
- Ho, C.L.; Tan, H.Q.; Chua, K.J.; Kang, A.; Lim, K.H.; Ling, K.L.; Yew, W.S.; Lee, Y.S.; Thiery, J.P.; Chang, M.W. Engineered commensal microbes for diet-mediated colorectal-cancer chemoprevention. Nat. Biomed. Eng. 2018, 2, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Kiddle, G.; Bennett, R.N.; Wallsgrove, R.M. Local and systemic changes in glucosinolates in Chinese and European cultivars of oilseed rape (Brassica napus L.) after inoculation with Sclerotinia sclerotiorum (stem rot). Ann. Appl. Biol. 1999, 134, 45–58. [Google Scholar] [CrossRef]
- Ishimoto, H.; Fukushi, Y.; Yoshida, T.; Tahara, S. Rhizopus and Fusarium are selected as dominant fungal genera in rhizospheres of Brassicaceae. J. Chem. Ecol. 2000, 26, 2387–2399. [Google Scholar] [CrossRef]
- Del Carmen Martinez-Ballesta, M.; Moreno, D.A.; Carvajal, M. The physiological importance of glucosinolates on plant response to abiotic stress in Brassica. Int. J. Mol. Sci. 2013, 14, 11607–11625. [Google Scholar] [CrossRef] [PubMed]
- Moreira, X.; Abdala-Roberts, L.; Gols, R.; Francisco, M. Plant domestication decreases both constitutive and induced chemical defences by direct selection against defensive traits. Sci. Rep. 2018, 8, 12678. [Google Scholar] [CrossRef] [PubMed]
- Traka, M.H.; Saha, S.; Huseby, S.; Kopriva, S.; Walley, P.G.; Barker, G.C.; Moore, J.; Mero, G.; van den Bosch, F.; Constant, H.; et al. Genetic regulation of glucoraphanin accumulation in Beneforte broccoli. New Phytol. 2013, 198, 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- Urbancsok, J.; Bones, A.M.; Kissen, R. Arabidopsis mutants impaired in glutathione biosynthesis exhibit higher sensitivity towards the glucosinolate hydrolysis product allyl-isothiocyanate. Sci. Rep. 2018, 8, 9809. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Stephenson, K.K. Cancer chemoprotective effects of cruciferous vegetables. Hortscience 1999, 34, 1159–1163. [Google Scholar] [CrossRef]
- Farnham, M.W.; Stephenson, K.K.; Fahey, J.W. The capacity of broccoli to induce a mammalian chemoprotective enzyme varies among inbred lines. J. Amer. Soc. Hort. Sci. 2000, 125, 482–488. [Google Scholar] [CrossRef]
- Farnham, M.W.; Wilson, P.E.; Stephenson, K.K.; Fahey, J.W. Genetic and environmental effects on glucosinolate content and chemoprotective potency of broccoli. Plant. Breed. 2004, 123, 60–65. [Google Scholar] [CrossRef]
- Farnham, M.W.; Stephenson, K.K.; Fahey, J.W. Glucoraphanin level in broccoli seed is largely determined by genotype. Hortscience 2005, 40, 50–53. [Google Scholar] [CrossRef]
- Bachmanov, A.A.; Reed, D.R.; Beauchamp, G.K.; Tordoff, M.G. Food intake, water intake, and drinking spout side preference of 28 mouse strains. Behav. Genet. 2002, 32, 435–443. [Google Scholar] [CrossRef]
- Mohammed, A.; Fox, J.T.; Miller, M.S. Cancer Chemoprevention: Preclinical In Vivo Alternate Dosing Strategies to Reduce Drug Toxicities. Toxicol. Sci. 2019, 170, 251–259. [Google Scholar] [CrossRef]
- Zhang, Y. The molecular basis that unifies the metabolism, cellular uptake and chemopreventive activities of dietary isothiocyanates. Carcinogenesis 2012, 33, 2–9. [Google Scholar] [CrossRef]
- Ramirez, C.N.; Li, W.; Zhang, C.; Wu, R.; Su, S.; Wang, C.; Gao, L.; Yin, R.; Kong, A.N. In vitro-in vivo dose response of ursolic acid, sulforaphane, PEITC, and curcumin in cancer prevention. AAPS J. 2017, 20, 19. [Google Scholar] [CrossRef]
- Paunkov, A.; Chartoumpekis, D.V.; Ziros, P.G.; Sykiotis, G.P. A bibliometric review of the Keap1/Nrf2 pathway and its related antioxidant compounds. Antioxidants 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Socala, K.; Nieoczym, D.; Kowalczuk-Vasilev, E.; Wyska, E.; Wlaz, P. Increased seizure susceptibility and other toxicity symptoms following acute sulforaphane treatment in mice. Toxicol. Appl. Pharm. 2017, 326, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Poulton, E.J.; Levy, L.; Lampe, J.W.; Shen, D.D.; Tracy, J.; Shuhart, M.C.; Thummel, K.E.; Eaton, D.L. Sulforaphane is not an effective antagonist of the human pregnane X-receptor in vivo. Toxicol. Appl. Pharm. 2013, 266, 122–131. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shorey, L.E.; Madeen, E.P.; Atwell, L.L.; Ho, E.; Lohr, C.V.; Pereira, C.B.; Dashwood, R.H.; Williams, D.E. Differential modulation of dibenzo[def,p]chrysene transplacental carcinogenesis: Maternal diets rich in indole-3-carbinol versus sulforaphane. Toxicol. Appl. Pharm. 2013, 270, 60–69. [Google Scholar] [CrossRef]
- Tao, S.; Rojo de la Vega, M.; Chapman, E.; Ooi, A.; Zhang, D.D. The effects of NRF2 modulation on the initiation and progression of chemically and genetically induced lung cancer. Mol. Carcinog. 2018, 57, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Kombairaju, P.; Ma, J.; Thimmulappa, R.K.; Yan, S.G.; Gabrielson, E.; Singh, A.; Biswal, S. Prolonged sulforaphane treatment does not enhance tumorigenesis in oncogenic K-ras and xenograft mouse models of lung cancer. J. Carcinog. 2012, 11, 8. [Google Scholar]
- Bheemreddy, R.M.; Jeffery, E.H. The metabolic fate of purified glucoraphanin in F344 rats. J. Agric. Food Chem. 2007, 55, 2861–2866. [Google Scholar] [CrossRef]
- Melchini, A.; Traka, M.H. Biological profile of erucin: A new promising anticancer agent from cruciferous vegetables. Toxins 2010, 2, 593–612. [Google Scholar] [CrossRef]
- Zhang, Y.; Wade, K.L.; Prestera, T.; Talalay, P. Quantitative determination of isothiocyanates, dithiocarbamates, carbon disulfide, and related thiocarbonyl compounds by cyclocondensation with 1,2-benzenedithiol. Anal. Biochem. 1996, 239, 160–167. [Google Scholar] [CrossRef]
- Kerns, M.L.; DePianto, D.; Dinkova-Kostova, A.T.; Talalay, P.; Coulombe, P.A. Reprogramming of keratin biosynthesis by sulforaphane restores skin integrity in epidermolysis bullosa simplex. Proc. Natl. Acad. Sci. USA 2007, 104, 14460–14465. [Google Scholar] [CrossRef]
- Sivapalan, T.; Melchini, A.; Saha, S.; Needs, P.W.; Traka, M.H.; Tapp, H.; Dainty, J.R.; Mithen, R.F. Bioavailability of Glucoraphanin and Sulforaphane from High-Glucoraphanin Broccoli. Mol. Nutr. Food Res. 2018, 62, e1700911. [Google Scholar] [CrossRef] [PubMed]
- Gasper, A.V.; Al-Janobi, A.; Smith, J.A.; Bacon, J.R.; Fortun, P.; Atherton, C.; Taylor, M.A.; Hawkey, C.J.; Barrett, D.A.; Mithen, R.F. Glutathione S-transferase M1 polymorphism and metabolism of sulforaphane from standard and high-glucosinolate broccoli. Am. J. Clin. Nutr. 2005, 82, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Egner, P.A.; Kensler, T.W.; Chen, J.G.; Gange, S.J.; Groopman, J.D.; Friesen, M.D. Quantification of sulforaphane mercapturic acid pathway conjugates in human urine by high-performance liquid chromatography and isotope-dilution tandem mass spectrometry. Chem. Res. Toxicol. 2008, 21, 1991–1996. [Google Scholar] [CrossRef]
- Cornblatt, B.S.; Ye, L.; Dinkova-Kostova, A.T.; Erb, M.; Fahey, J.W.; Singh, N.K.; Chen, M.S.; Stierer, T.; Garrett-Mayer, E.; Argani, P.; et al. Preclinical and clinical evaluation of sulforaphane for chemoprevention in the breast. Carcinogenesis 2007, 28, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Dinkova-Kostova, A.T.; Wade, K.L.; Zhang, Y.; Shapiro, T.A.; Talalay, P. Quantitative determination of dithiocarbamates in human plasma, serum, erythrocytes and urine: Pharmacokinetics of broccoli sprout isothiocyanates in humans. Clin. Chim. Acta 2002, 316, 43–53. [Google Scholar] [CrossRef]
- Atwell, L.L.; Hsu, A.; Wong, C.P.; Stevens, J.F.; Bella, D.; Yu, T.W.; Pereira, C.B.; Lohr, C.V.; Christensen, J.M.; Dashwood, R.H.; et al. Absorption and chemopreventive targets of sulforaphane in humans following consumption of broccoli sprouts or a myrosinase-treated broccoli sprout extract. Mol. Nutr. Food Res. 2015, 59, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Garzotto, M.; Davis, E.W., 2nd; Mori, M.; Stoller, W.A.; Farris, P.E.; Wong, C.P.; Beaver, L.M.; Thomas, G.V.; Williams, D.E.; et al. Sulforaphane bioavailability and chemopreventive activity in men presenting for biopsy of the prostate gland: A randomized controlled trial. Nutr. Cancer 2019, 1–14. [Google Scholar] [CrossRef]
- Davidson, R.; Gardner, S.; Jupp, O.; Bullough, A.; Butters, S.; Watts, L.; Donell, S.; Traka, M.; Saha, S.; Mithen, R.; et al. Isothiocyanates are detected in human synovial fluid following broccoli consumption and can affect the tissues of the knee joint. Sci. Rep. 2017, 7, 3398. [Google Scholar] [CrossRef]
- Shapiro, T.A.; Fahey, J.W.; Wade, K.L.; Stephenson, K.K.; Talalay, P. Chemoprotective glucosinolates and isothiocyanates of broccoli sprouts: Metabolism and excretion in humans. Cancer Epidemiol. Biomark. Prev. 2001, 10, 501–508. [Google Scholar]
- Shapiro, T.A.; Fahey, J.W.; Dinkova-Kostova, A.T.; Holtzclaw, W.D.; Stephenson, K.K.; Wade, K.L.; Ye, L.; Talalay, P. Safety, tolerance, and metabolism of broccoli sprout glucosinolates and isothiocyanates: A clinical phase I study. Nutr. Cancer 2006, 55, 53–62. [Google Scholar] [CrossRef]
- Chartoumpekis, D.V.; Ziros, P.G.; Chen, J.G.; Groopman, J.D.; Kensler, T.W.; Sykiotis, G.P. Broccoli sprout beverage is safe for thyroid hormonal and autoimmune status: Results of a 12-week randomized trial. Food Chem. Toxicol. 2019, 126, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Egner, P.A.; Chen, J.G.; Wang, J.B.; Wu, Y.; Sun, Y.; Lu, J.H.; Zhu, J.; Zhang, Y.H.; Chen, Y.S.; Friesen, M.D.; et al. Bioavailability of Sulforaphane from two broccoli sprout beverages: Results of a short-term, cross-over clinical trial in Qidong, China. Cancer Prev. Res. (Phila) 2011, 4, 384–395. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Hullar, M.A.; Beresford, S.A.; Lampe, J.W. Variation of glucoraphanin metabolism in vivo and ex vivo by human gut bacteria. Br. J. Nutr. 2011, 106, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Wehage, S.L.; Holtzclaw, W.D.; Kensler, T.W.; Egner, P.A.; Shapiro, T.A.; Talalay, P. Protection of humans by plant glucosinolates: Efficiency of conversion of glucosinolates to isothiocyanates by the gastrointestinal microflora. Cancer Prev. Res. (Phila) 2012, 5, 603–611. [Google Scholar] [CrossRef]
- Fahey, J.W.; Wade, K.L.; Wehage, S.L.; Holtzclaw, W.D.; Liu, H.; Talalay, P.; Fuchs, E.; Stephenson, K.K. Stabilized sulforaphane for clinical use: Phytochemical delivery efficiency. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Wade, K.L.; Stephenson, K.K.; Panjwani, A.A.; Liu, H.; Cornblatt, G.; Cornblatt, B.S.; Ownby, S.L.; Fuchs, E.; Holtzclaw, W.D.; et al. Bioavailability of sulforaphane following ingestion of glucoraphanin-rich broccoli sprout and seed extracts with active myrosinase: A pilot study of the effects of proton pump inhibitor administration. Nutrients 2019, 11. [Google Scholar] [CrossRef]
- Fahey, J.W.; Holtzclaw, W.D.; Wehage, S.L.; Wade, K.L.; Stephenson, K.K.; Talalay, P. Sulforaphane bioavailability from glucoraphanin-rich broccoli: Control by active endogenous myrosinase. PLoS ONE 2015, 10, e0140963. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.G.; Johnson, J.; Egner, P.; Ng, D.; Zhu, J.; Wang, J.B.; Xue, X.F.; Sun, Y.; Zhang, Y.H.; Lu, L.L.; et al. Dose-dependent detoxication of the airborne pollutant benzene in a randomized trial of broccoli sprout beverage in Qidong, China. Am. J. Clin. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Bogaards, J.J.; Verhagen, H.; Willems, M.I.; van Poppel, G.; van Bladeren, P.J. Consumption of Brussels sprouts results in elevated alpha-class glutathione S-transferase levels in human blood plasma. Carcinogenesis 1994, 15, 1073–1075. [Google Scholar] [CrossRef]
- Sreerama, L.; Hedge, M.W.; Sladek, N.E. Identification of a class 3 aldehyde dehydrogenase in human saliva and increased levels of this enzyme, glutathione S-transferases, and DT-diaphorase in the saliva of subjects who continually ingest large quantities of coffee or broccoli. Clin. Cancer Res. 1995, 1, 1153–1163. [Google Scholar]
- Bauman, J.E.; Zang, Y.; Sen, M.; Li, C.; Wang, L.; Egner, P.A.; Fahey, J.W.; Normolle, D.P.; Grandis, J.R.; Kensler, T.W.; et al. Prevention of carcinogen-induced oral cancer by sulforaphane. Cancer Prev. Res. (Phila) 2016, 9, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.H.; Reynolds, C.; Brooker, A.; Talalay, P.; Fahey, J.W. Sulforaphane improves the bronchoprotective response in asthmatics through Nrf2-mediated gene pathways. Respir. Res. 2015, 16, 106. [Google Scholar] [CrossRef] [PubMed]
- Doss, J.F.; Jonassaint, J.C.; Garrett, M.E.; Ashley-Koch, A.E.; Telen, M.J.; Chi, J.T. Phase 1 Study of a sulforaphane-containing broccoli sprout homogenate for sickle cell disease. PLoS ONE 2016, 11, e0152895. [Google Scholar] [CrossRef] [PubMed]
- Dinkova-Kostova, A.T.; Fahey, J.W.; Wade, K.L.; Jenkins, S.N.; Shapiro, T.A.; Fuchs, E.J.; Kerns, M.L.; Talalay, P. Induction of the phase 2 response in mouse and human skin by sulforaphane-containing broccoli sprout extracts. Cancer Epidemiol Biomark. Prev. 2007, 16, 847–851. [Google Scholar] [CrossRef]
- Riedl, M.A.; Saxon, A.; Diaz-Sanchez, D. Oral sulforaphane increases Phase II antioxidant enzymes in the human upper airway. Clin. Immunol. 2009, 130, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Kensler, T.W.; Ng, D.; Carmella, S.G.; Chen, M.; Jacobson, L.P.; Munoz, A.; Egner, P.A.; Chen, J.G.; Qian, G.S.; Chen, T.Y.; et al. Modulation of the metabolism of airborne pollutants by glucoraphanin-rich and sulforaphane-rich broccoli sprout beverages in Qidong, China. Carcinogenesis 2012, 33, 101–107. [Google Scholar] [CrossRef]
- Egner, P.A.; Chen, J.G.; Zarth, A.T.; Ng, D.K.; Wang, J.B.; Kensler, K.H.; Jacobson, L.P.; Munoz, A.; Johnson, J.L.; Groopman, J.D.; et al. Rapid and sustainable detoxication of airborne pollutants by broccoli sprout beverage: Results of a randomized clinical trial in China. Cancer Prev. Res. (Phila) 2014, 7, 813–823. [Google Scholar] [CrossRef]
- Traka, M.; Gasper, A.V.; Melchini, A.; Bacon, J.R.; Needs, P.W.; Frost, V.; Chantry, A.; Jones, A.M.; Ortori, C.A.; Barrett, D.A.; et al. Broccoli consumption interacts with GSTM1 to perturb oncogenic signalling pathways in the prostate. PloS ONE 2008, 3, e2568. [Google Scholar] [CrossRef]
- Traka, M.H.; Melchini, A.; Coode-Bate, J.; Al Kadhi, O.; Saha, S.; Defernez, M.; Troncoso-Rey, P.; Kibblewhite, H.; O’Neill, C.M.; Bernuzzi, F.; et al. Transcriptional changes in prostate of men on active surveillance after a 12-mo glucoraphanin-rich broccoli intervention-results from the Effect of Sulforaphane on prostate CAncer PrEvention (ESCAPE) randomized controlled trial. Am. J. Clin. Nutr. 2019, 109, 1133–1144. [Google Scholar] [CrossRef]
- Sedlak, T.W.; Nucifora, L.G.; Koga, M.; Shaffer, L.S.; Higgs, C.; Tanaka, T.; Wang, A.M.; Coughlin, J.M.; Barker, P.B.; Fahey, J.W.; et al. Sulforaphane augments glutathione and influences brain metabolites in human subjects: A clinical pilot study. Mol. Neuropsychiatry 2018, 3, 214–222. [Google Scholar] [CrossRef]
- Armah, C.N.; Traka, M.H.; Dainty, J.R.; Defernez, M.; Janssens, A.; Leung, W.; Doleman, J.F.; Potter, J.F.; Mithen, R.F. A diet rich in high-glucoraphanin broccoli interacts with genotype to reduce discordance in plasma metabolite profiles by modulating mitochondrial function. Am. J. Clin. Nutr. 2013, 98, 712–722. [Google Scholar] [CrossRef] [PubMed]
- Talalay, P.; Fahey, J.W.; Healy, Z.R.; Wehage, S.L.; Benedict, A.L.; Min, C.; Dinkova-Kostova, A.T. Sulforaphane mobilizes cellular defenses that protect skin against damage by UV radiation. Proc. Natl. Acad. Sci. USA 2007, 104, 17500–17505. [Google Scholar] [CrossRef] [PubMed]
- Knatko, E.V.; Ibbotson, S.H.; Zhang, Y.; Higgins, M.; Fahey, J.W.; Talalay, P.; Dawe, R.S.; Ferguson, J.; Huang, J.T.; Clarke, R.; et al. Nrf2 activation protects against solar-simulated ultraviolet radiation in mice and humans. Cancer Prev Res. (Phila) 2015, 8, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Armah, C.N.; Derdemezis, C.; Traka, M.H.; Dainty, J.R.; Doleman, J.F.; Saha, S.; Leung, W.; Potter, J.F.; Lovegrove, J.A.; Mithen, R.F. Diet rich in high glucoraphanin broccoli reduces plasma LDL cholesterol: Evidence from randomised controlled trials. Mol. Nutr. Food Res. 2015, 59, 918–926. [Google Scholar] [CrossRef] [PubMed]
- Axelsson, A.S.; Tubbs, E.; Mecham, B.; Chacko, S.; Nenonen, H.A.; Tang, Y.; Fahey, J.W.; Derry, J.M.J.; Wollheim, C.B.; Wierup, N.; et al. Sulforaphane reduces hepatic glucose production and improves glucose control in patients with type 2 diabetes. Sci. Transl. Med. 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Connors, S.L.; Macklin, E.A.; Smith, K.D.; Fahey, J.W.; Talalay, P.; Zimmerman, A.W. Sulforaphane treatment of autism spectrum disorder (ASD). Proc. Natl. Acad. Sci. USA 2014, 111, 15550–15555. [Google Scholar] [CrossRef]
- Chen, J.; Zhu, J.; Wang, G.; Groopman, J.D.; Kensler, T.W. Qidong: A crucible for studies on liver cancer etiology and prevention. Cancer Biol. Med. 2019, 16, 24–37. [Google Scholar]
- Egner, P.A.; Wang, J.B.; Zhu, Y.R.; Jacobson, L.P.; Ng, D.; Munoz, A.; Fahey, J.W.; Chen, J.G.; Chen, T.Y.; Qian, G.S.; et al. Prevention of liver cancer in Qidong, China: Lessons from aflatoxin biomarker studies. Prog. Chem. 2013, 25, 1454–1461. [Google Scholar]
- Kensler, T.W.; Chen, J.G.; Egner, P.A.; Fahey, J.W.; Jacobson, L.P.; Stephenson, K.K.; Ye, L.; Coady, J.L.; Wang, J.B.; Wu, Y.; et al. Effects of glucosinolate-rich broccoli sprouts on urinary levels of aflatoxin-DNA adducts and phenanthrene tetraols in a randomized clinical trial in He Zuo township, Qidong, People’s Republic of China. Cancer Epidemiol Biomark. Prev. 2005, 14, 2605–2613. [Google Scholar] [CrossRef]
- Heber, D.; Li, Z.; Garcia-Lloret, M.; Wong, A.M.; Lee, T.Y.; Thames, G.; Krak, M.; Zhang, Y.; Nel, A. Sulforaphane-rich broccoli sprout extract attenuates nasal allergic response to diesel exhaust particles. Food Funct. 2014, 5, 35–41. [Google Scholar] [CrossRef]
- Noah, T.L.; Zhang, H.; Zhou, H.; Glista-Baker, E.; Muller, L.; Bauer, R.N.; Meyer, M.; Murphy, P.C.; Jones, S.; Letang, B.; et al. Effect of broccoli sprouts on nasal response to live attenuated influenza virus in smokers: A randomized, double-blind study. PLoS ONE 2014, 9, e98671. [Google Scholar] [CrossRef] [PubMed]
- Healy, Z.R.; Lee, N.H.; Gao, X.; Goldring, M.B.; Talalay, P.; Kensler, T.W.; Konstantopoulos, K. Divergent responses of chondrocytes and endothelial cells to shear stress: Cross-talk among COX-2, the phase 2 response, and apoptosis. Proc. Natl. Acad. Sci. USA 2005, 102, 14010–14015. [Google Scholar] [CrossRef] [PubMed]
- Meyer, M.; Kesic, M.J.; Clarke, J.; Ho, E.; Simmen, R.C.; Diaz-Sanchez, D.; Noah, T.L.; Jaspers, I. Sulforaphane induces SLPI secretion in the nasal mucosa. Respir. Med. 2013, 107, 472–475. [Google Scholar] [CrossRef] [PubMed]
- Sudini, K.; Diette, G.B.; Breysse, P.N.; McCormack, M.C.; Bull, D.; Biswal, S.; Zhai, S.; Brereton, N.; Peng, R.D.; Matsui, E.C. A randomized controlled trial of the effect of broccoli sprouts on antioxidant gene expression and airway inflammation in asthmatics. J. Allergy Clin. Immunol. Pr. 2016, 4, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Muller, L.; Meyer, M.; Bauer, R.N.; Zhou, H.; Zhang, H.; Jones, S.; Robinette, C.; Noah, T.L.; Jaspers, I. Effect of broccoli sprouts and live attenuated influenza virus on peripheral blood natural killer cells: A randomized, double-blind study. PLoS ONE 2016, 11, e0147742. [Google Scholar] [CrossRef] [PubMed]
- Duran, C.G.; Burbank, A.J.; Mills, K.H.; Duckworth, H.R.; Aleman, M.M.; Kesic, M.J.; Peden, D.B.; Pan, Y.; Zhou, H.; Hernandez, M.L. A proof-of-concept clinical study examining the NRF2 activator sulforaphane against neutrophilic airway inflammation. Respir. Res. 2016, 17, 89. [Google Scholar] [CrossRef]
- Atwell, L.L.; Zhang, Z.; Mori, M.; Farris, P.; Vetto, J.T.; Naik, A.M.; Oh, K.Y.; Thuillier, P.; Ho, E.; Shannon, J. Sulforaphane bioavailability and chemopreventive activity in women scheduled for breast biopsy. Cancer Prev. Res. (Phila) 2015, 8, 1184–1191. [Google Scholar] [CrossRef] [PubMed]
- Riso, P.; Martini, D.; Visioli, F.; Martinetti, A.; Porrini, M. Effect of broccoli intake on markers related to oxidative stress and cancer risk in healthy smokers and nonsmokers. Nutr. Cancer 2009, 61, 232–237. [Google Scholar] [CrossRef]
- Gasper, A.V.; Traka, M.; Bacon, J.R.; Smith, J.A.; Taylor, M.A.; Hawkey, C.J.; Barrett, D.A.; Mithen, R.F. Consuming broccoli does not induce genes associated with xenobiotic metabolism and cell cycle control in human gastric mucosa. J. Nutr. 2007, 137, 1718–1724. [Google Scholar] [CrossRef]
- Walters, D.G.; Young, P.J.; Agus, C.; Knize, M.G.; Boobis, A.R.; Gooderham, N.J.; Lake, B.G. Cruciferous vegetable consumption alters the metabolism of the dietary carcinogen 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) in humans. Carcinogenesis 2004, 25, 1659–1669. [Google Scholar] [CrossRef]
- Cipolla, B.G.; Mandron, E.; Lefort, J.M.; Coadou, Y.; Della Negra, E.; Corbel, L.; Le Scodan, R.; Azzouzi, A.R.; Mottet, N. Effect of sulforaphane in men with biochemical recurrence after radical prostatectomy. Cancer Prev Res. (Phila) 2015, 8, 712–719. [Google Scholar] [CrossRef]
- Alumkal, J.J.; Slottke, R.; Schwartzman, J.; Cherala, G.; Munar, M.; Graff, J.N.; Beer, T.M.; Ryan, C.W.; Koop, D.R.; Gibbs, A.; et al. A phase II study of sulforaphane-rich broccoli sprout extracts in men with recurrent prostate cancer. Invest. New Drugs 2015, 33, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Tahata, S.; Singh, S.V.; Lin, Y.; Hahm, E.R.; Beumer, J.H.; Christner, S.M.; Rao, U.N.; Sander, C.; Tarhini, A.A.; Tawbi, H.; et al. Evaluation of biodistribution of sulforaphane after administration of oral broccoli sprout extract in melanoma patients with multiple atypical nevi. Cancer Prev. Res. (Phila) 2018, 11, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Wise, R.A.; Holbrook, J.T.; Criner, G.; Sethi, S.; Rayapudi, S.; Sudini, K.R.; Sugar, E.A.; Burke, A.; Thimmulappa, R.; Singh, A.; et al. Lack of effect of oral sulforaphane administration on Nrf2 expression in COPD: A randomized, double-blind, placebo controlled trial. PLoS ONE 2016, 11, e0163716. [Google Scholar] [CrossRef]
- Sidhaye, V.K.; Holbrook, J.T.; Burke, A.; Sudini, K.R.; Sethi, S.; Criner, G.J.; Fahey, J.W.; Berenson, C.S.; Jacobs, M.R.; Thimmulappa, R.; et al. Compartmentalization of anti-oxidant and anti-inflammatory gene expression in current and former smokers with COPD. Respir. Res. 2019, 20, 190. [Google Scholar] [CrossRef] [PubMed]
- Murashima, M.; Watanabe, S.; Zhuo, X.G.; Uehara, M.; Kurashige, A. Phase 1 study of multiple biomarkers for metabolism and oxidative stress after one-week intake of broccoli sprouts. Biofactors 2004, 22, 271–275. [Google Scholar] [CrossRef]
- Christiansen, B.; Bellostas Muguerza, N.; Petersen, A.M.; Kveiborg, B.; Madsen, C.R.; Thomas, H.; Ihlemann, N.; Sorensen, J.C.; Kober, L.; Sorensen, H.; et al. Ingestion of broccoli sprouts does not improve endothelial function in humans with hypertension. PLoS ONE 2010, 5, e12461. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Chillon, M.T.; Carazo-Diaz, C.; Prieto-Merino, D.; Zafrilla, P.; Moreno, D.A.; Villano, D. Effects of long-term consumption of broccoli sprouts on inflammatory markers in overweight subjects. Clin. Nutr. 2019, 38, 745–752. [Google Scholar] [CrossRef]
- Mirmiran, P.; Bahadoran, Z.; Hosseinpanah, F.; Keyzad, A.; Azizi, F. Effects of broccoli sprout with high sulforaphane concentration on inflammatory markers in type 2 diabetic patients: A randomized double-blind placebo-controlled clinical trial. J. Funct. Foods 2012, 4, 837–841. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Mirmiran, P.; Hosseinpanah, F.; Hedayati, M.; Hosseinpour-Niazi, S.; Azizi, F. Broccoli sprouts reduce oxidative stress in type 2 diabetes: A randomized double-blind clinical trial. Eur. J. Clin. Nutr. 2011, 65, 972–977. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Tohidi, M.; Nazeri, P.; Mehran, M.; Azizi, F.; Mirmiran, P. Effect of broccoli sprouts on insulin resistance in type 2 diabetic patients: A randomized double-blind clinical trial. Int. J. Food Sci. Nutr. 2012, 63, 767–771. [Google Scholar] [CrossRef] [PubMed]
- Healy, Z.R.; Liu, H.; Holtzclaw, W.D.; Talalay, P. Inactivation of tautomerase activity of macrophage migration inhibitory factor by sulforaphane: A potential biomarker for anti-inflammatory intervention. Cancer Epidemiol. Biomark. Prev. 2011, 20, 1516–1523. [Google Scholar] [CrossRef] [PubMed]
- Ushida, Y.S.H.; Yanaka, A. Low-dose of the sulforaphane precursor glucoraphanin as a dietary supplement induces chemoprotective enzymes in humans. Food Nutr. Sci. 2015, 6, 62147. [Google Scholar] [CrossRef]
- Galan, M.V.; Kishan, A.A.; Silverman, A.L. Oral broccoli sprouts for the treatment of Helicobacter pylori infection: A preliminary report. Dig. Dis. Sci. 2004, 49, 1088–1090. [Google Scholar] [CrossRef] [PubMed]
- Yanaka, A.; Fahey, J.W.; Fukumoto, A.; Nakayama, M.; Inoue, S.; Zhang, S.; Tauchi, M.; Suzuki, H.; Hyodo, I.; Yamamoto, M. Dietary sulforaphane-rich broccoli sprouts reduce colonization and attenuate gastritis in Helicobacter pylori-infected mice and humans. Cancer Prev. Res. (Phila) 2009, 2, 353–360. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Mirmiran, P.; Yeganeh, M.Z.; Hosseinpanah, F.; Zojaji, H.; Azizi, F. Complementary and alternative medicinal effects of broccoli sprouts powder on Helicobacter pylori eradication rate in type 2 diabetic patients: A randomized clinical trial. J. Funct. Foods 2014, 7, 390–397. [Google Scholar] [CrossRef]
- Kikuchi, M.; Ushida, Y.; Shiozawa, H.; Umeda, R.; Tsuruya, K.; Aoki, Y.; Suganuma, H.; Nishizaki, Y. Sulforaphane-rich broccoli sprout extract improves hepatic abnormalities in male subjects. World J. Gastroenterol. 2015, 21, 12457–12467. [Google Scholar] [CrossRef]
- Lynch, R.; Diggins, E.L.; Connors, S.L.; Zimmerman, A.W.; Singh, K.; Liu, H.; Talalay, P.; Fahey, J.W. Sulforaphane from broccoli reduces symptoms of autism: A follow-up case series from a randomized double-blind study. Glob. Adv. Health Med. 2017, 6, 2164957X17735826. [Google Scholar] [CrossRef]
- Bent, S.; Lawton, B.; Warren, T.; Widjaja, F.; Dang, K.; Fahey, J.W.; Cornblatt, B.; Kinchen, J.M.; Delucchi, K.; Hendren, R.L. Identification of urinary metabolites that correlate with clinical improvements in children with autism treated with sulforaphane from broccoli. Mol. Autism. 2018, 9, 35. [Google Scholar] [CrossRef]
- Matile, P. The mustard oil bomb - compartmentation of the myrosinase system. Biochem. Physiol. Pfl. 1980, 175, 722–731. [Google Scholar] [CrossRef]
- Fahey, J.W.; Wade, K.L.; Stephenson, K.K.; Shi, Y.; Liu, H.; Panjwani, A.A.; Warrick, C.R.; Olson, M.E. A strategy to deliver precise oral doses of the glucosinolates or isothiocyanates from moringa oleifera leaves for use in clinical studies. Nutrients 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Bierwirth, J.E.; Oftedal, K.N.; Civille, G.V.; Fahey, J.W. Flavor misattribution: A novel approach to improving compliance and blinding in food-based clinical interventions. NFS J. 2015, 1, 24–30. [Google Scholar] [CrossRef]
- Conaway, C.C.; Getahun, S.M.; Liebes, L.L.; Pusateri, D.J.; Topham, D.K.; Botero-Omary, M.; Chung, F.L. Disposition of glucosinolates and sulforaphane in humans after ingestion of steamed and fresh broccoli. Nutr. Cancer 2000, 38, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Myzak, M.C.; Tong, P.; Dashwood, W.M.; Dashwood, R.H.; Ho, E. Sulforaphane retards the growth of human PC-3 xenografts and inhibits HDAC activity in human subjects. Exp. Biol Med. (Maywood) 2007, 232, 227–234. [Google Scholar] [PubMed]
- Rungapamestry, V.; Duncan, A.J.; Fuller, Z.; Ratcliffe, B. Effect of meal composition and cooking duration on the fate of sulforaphane following consumption of broccoli by healthy human subjects. Br. J. Nutr. 2007, 97, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, M.; Klopping-Ketelaars, I.W.; van den Berg, R.; Vaes, W.H. Bioavailability and kinetics of sulforaphane in humans after consumption of cooked versus raw broccoli. J. Agric. Food Chem. 2008, 56, 10505–10509. [Google Scholar] [CrossRef] [PubMed]
- Hanlon, N.; Coldham, N.; Gielbert, A.; Sauer, M.J.; Ioannides, C. Repeated intake of broccoli does not lead to higher plasma levels of sulforaphane in human volunteers. Cancer Lett. 2009, 284, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J.D.; Riedl, K.; Bella, D.; Schwartz, S.J.; Stevens, J.F.; Ho, E. Comparison of isothiocyanate metabolite levels and histone deacetylase activity in human subjects consuming broccoli sprouts or broccoli supplement. J. Agric. Food Chem. 2011, 59, 10955–10963. [Google Scholar] [CrossRef]
- Hauder, J.; Winkler, S.; Bub, A.; Rufer, C.E.; Pignitter, M.; Somoza, V. LC-MS/MS quantification of sulforaphane and indole-3-carbinol metabolites in human plasma and urine after dietary intake of selenium-fortified broccoli. J. Agric. Food Chem. 2011, 59, 8047–8057. [Google Scholar] [CrossRef] [PubMed]
- Cramer, J.M.; Teran-Garcia, M.; Jeffery, E.H. Enhancing sulforaphane absorption and excretion in healthy men through the combined consumption of fresh broccoli sprouts and a glucoraphanin-rich powder. Br. J. Nutr. 2012, 107, 1333–1338. [Google Scholar] [CrossRef]
- Saha, S.; Hollands, W.; Teucher, B.; Needs, P.W.; Narbad, A.; Ortori, C.A.; Barrett, D.A.; Rossiter, J.T.; Mithen, R.F.; Kroon, P.A. Isothiocyanate concentrations and interconversion of sulforaphane to erucin in human subjects after consumption of commercial frozen broccoli compared to fresh broccoli. Mol. Nutr. Food Res. 2012, 56, 1906–1916. [Google Scholar] [CrossRef] [PubMed]
- Baier, S.R.; Zbasnik, R.; Schlegel, V.; Zempleni, J. Off-target effects of sulforaphane include the derepression of long terminal repeats through histone acetylation events. J. Nutr. Biochem. 2014, 25, 665–668. [Google Scholar] [CrossRef]
- Chang, Y.W.; Jang, J.Y.; Kim, Y.H.; Kim, J.W.; Shim, J.J. The effects of broccoli sprout extract containing sulforaphane on lipid peroxidation and helicobacter pylori infection in the gastric mucosa. Gut. Liver 2015, 9, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Medina, S.; Dominguez-Perles, R.; Moreno, D.A.; Garcia-Viguera, C.; Ferreres, F.; Gil, J.I.; Gil-Izquierdo, A. The intake of broccoli sprouts modulates the inflammatory and vascular prostanoids but not the oxidative stress-related isoprostanes in healthy humans. Food Chem. 2015, 173, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Shiina, A.; Kanahara, N.; Sasaki, T.; Oda, Y.; Hashimoto, T.; Hasegawa, T.; Yoshida, T.; Iyo, M.; Hashimoto, K. An open study of sulforaphane-rich broccoli sprout extract in patients with schizophrenia. Clin. Psychopharmacol. Neurosci. 2015, 13, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Housley, L.; Magana, A.A.; Hsu, A.; Beaver, L.M.; Wong, C.P.; Stevens, J.F.; Choi, J.; Jiang, Y.; Bella, D.; Williams, D.E.; et al. Untargeted metabolomic screen reveals changes in human plasma metabolite profiles following consumption of fresh broccoli sprouts. Mol. Nutr. Food Res. 2018, 62, e1700665. [Google Scholar] [CrossRef] [PubMed]
- Okunade, O.; Niranjan, K.; Ghawi, S.K.; Kuhnle, G.; Methven, L. Supplementation of the diet by exogenous myrosinase via mustard seeds to increase the bioavailability of sulforaphane in healthy human subjects after the consumption of cooked broccoli. Mol. Nutr. Food Res. 2018, 62, e1700980. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Kensler, T.W. Role of dietary supplements/nutraceuticals in chemoprevention through induction of cytoprotective enzymes. Chem. Res. Toxicol. 2007, 20, 572–576. [Google Scholar] [CrossRef]
- Fahey, J.W.; Kensler, T.W. Frugal medicine: Health-span extension through green chemoprevention. Am. Med. Assoc. Virtual. Mentor. 2013, 15, 311–318. [Google Scholar]
- Value added to GDP by agriculture and related industries, 2007–2017. 2019.
- Fahey, J.W.; Talalay, P.; Kensler, T.W. Notes from the field: “Green” chemoprevention as frugal medicine. Cancer Prev Res. 2012, 5, 179–188. [Google Scholar] [CrossRef]
- Talalay, P.; Talalay, P. The importance of using scientific principles in the development of medicinal agents from plants. Acad Med. 2001, 76, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, A.; Sharfstein, J.M. Breaking the gridlock: Regulation of dietary supplements in the United States. Drug Test. Anal. 2016, 8, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic. Clin. Pharm. 2016, 7, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Talalay, P. A fascination with enzymes: The journey not the arrival matters. J. Biol. Chem. 2005, 280, 28829–28847. [Google Scholar] [CrossRef] [PubMed]
| Compound Delivered2 | Delivery Format3 | Non-Fresh Product Source4 | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Year1 | First Author | Study Population | SF | GR | GR+ Myr | Tabs/Caps | Powder/Other | Fresh | Commercial | Academic | Treatment5 | Sample No | Dose as Reported, Converted to μmol6 | μmol/kg BW7 | Results | Ref |
| 1998 | Shapiro | Healthy | x | x | x | x | 250 g fresh broccoli (180 μmol GR) or 144 μmol GS or 118 μmol isothiocyanates (ITC) | 6 | 180 μmol GR | 2.57 (GR) | PK following GR or GS or ITC ingestion: Reproducibility and dose-dependence as measured in urine using the cyclocondensation | [26] | ||||
| 2000 | Conaway | Healthy | x | x | x | 200 g fresh or steamed broccoli w/ 220 and 200 μmol ITCs respectively | 12 | 220 & 200 μmol of total ITCs (measured and assumed to be primarily SF) | 3.14/2.86 (SF) | Bioavailability of ITCs from fresh broccoli is greater than that from cooked broccoli | [124] | |||||
| 2002 | Ye | Healthy | x | x | x | 200 μmol SF | 4 | broccoli extracts containing predominantly SF; 200 μmol total ITC | 2.86 (SF) | Development of a sensitive and specific method for quantifying levels of ITC and their metabolites in human plasma, serum, and erythrocytes | [56] | |||||
| 2004 | Murashima | Healthy | x | x | 100 g fresh broccoli sprouts/d x 7 d | 12 | we calculate a maximum of 500 μmol GR/d | 7.1 (GR) | Improvement of lipid metabolism; HDL cholesterol increased significantly only among females | [107] | ||||||
| 2004 | Walters | Healthy | x | x | 250 g broccoli or brussels sprouts (cooked), per day for 12 days | 20 | (est.) ca. 25 μmol GR | 0.36 (GR) | Induced metabolism of PhIP in humans | [101] | ||||||
| 2005 | Gasper | Healthy | x | x | 100 g florets from standard broccoli and high-glucosinolate broccoli, one dose | 16 | 150 mL of 107.5 or 345.8 μM “sulforaphane metabolites” broccoli soup | 0.23 or 0.74 (SF) | GSTM1 genotypes have significant effect on the SF metabolism | [53] | ||||||
| 2005 | Kensler | Healthy | x | x | x | broccoli sprout extract (BSE) containing 400 umol or <3 umol GR (placebo), daily for 2 weeks. | 200 | <3 μmol GR/day or 400 μmol GR/day | 0.043/5.71 | Decreased urinary excretion of dithiocarbamates and aflatoxin-DNA adducts and trans, anti-phenanthrene tetraol in urine by broccoli sprout glucosinolates. | [90] | |||||
| 2004 | Galan | Helicobacter pylori infected adults | x | x | x | x | 14, 28, or 56 g broccoli sprouts 2x/d x 7 days | 7 | up to 280 μmol GR/day | 1, 2, 4 (GR) | 7 of 9 patients were stool antigen negative immediately after the completion of therapy and six remained negative at day 35. | [115] | ||||
| 2006 | Shapiro | Healthy | x | x | x | BSE containing 25 μmol GR, 100 μmol GR, or 25 μmol SF, 3x/d x 7 d | 12 | 75 & 300 μmol GR/d; 75 μmol SF/d | 1.97 & 4.29 (GR); 1.07 (SF) | Cumulative excretion of SF metabolites similar regardless of GR dose, & much higher when taking SF; diurnal cycling obser. | [61] | |||||
| 2007 | Cornblatt | Reduction mammaplasty | x | x | SF-rich beverage (containing 200 μmole SF) | 8 | 200 μmol SF once | 2.86 (SF) | Measured serum and mammary tissue levels of SF metabolites | [55] | ||||||
| 2007 | Gasper | Healthy | x | x | “standard” and “HG” broccoli florets microwaved gently to make soup | 16 | 683 and 2296 μM SF measured | 1.42/4.92 (SF) | Consumption of high glucosinolate broccoli resulted in up-regulation of several xenobiotic metabolizing genes in gastric mucosal tissue | [100] | ||||||
| 2007 | Myzak | Healthy | x | x | 68 g broccoli sprouts (approximately 105 mg SF) | 3 | 847 μmol SF if all converted from GR using author′s estimate; 136 μmol SF using our estimate | 12.1 (auth)/1.9 (us) (SF) | HDAC activity was significantly inhibited in PBMC | [125] | ||||||
| 2007 | Rungapamestry | Healthy | x | x | x | x | x | 150 g lightly cooked broccoli or fully cooked broccoli or a broccoli seed extract with added mustard seed | 12 | 62 & 71.7 μmol GR/d or 2.7 μmol SF | 0.89/1.0 (GR) | Estimated yield of SF was ~ 3-fold higher after consumption of lightly cooked broccoli than fully cooked broccoli. Meal matrix did not significantly influence the hydrolysis of GR and its excretion as SF | [126] | |||
| 2008 | Traka | Diagnosed with high-grade prostatic intraepithelial neoplasia (PIN) | x | x | 150 g broccoli /d | 22 | 79.3 μmol GR/d | 1.13 (GR) | Showed complex change in signaling pathways associated with inflammation and carcinogenesis: Modified by GSTM1 genotype in broccoli feeding group | [79] | ||||||
| 2008 | Vermeulen | Healthy | x | x | x | 200 g crushed raw or cooked broccoli | 8 | 9.92 μmol SF or 61.4 μmol GR/d | 0.14 (SF)/0.88 (GR) | 10x greater bioavailability of SF w/ crushed, raw vs. microwaved broccoli | [127] | |||||
| 2009 | Hanlon | Healthy | x | x | 300 mL of homogenized raw broccoli w/ 3.9 mg SF, for 10 consecutive days | 6 | 22 μmol SF/d | 0.31 (SF) | Repeated intake of broccoli had no impact on the pharmacokinetic behavior or plasma levels of SF | [128] | ||||||
| 2009 | Riedl | Healthy | x | x | 150, 175, and 200 g broccoli sprout homogenate mixed with daikon sprouts homogenate, once daily for 3 days | 65 | 75 μmol/87.5 μmol /100 μmol SF/day (est) | 1.07/1.25/1.43 (SF) | Increased Phase II enzyme expression in nasal lavage cells occurred in a dose-dependent manner | [76] | ||||||
| 2009 | Riso | Healthy (10 smokers and 10 nonsmokers) | x | x | 200 g blanched broccoli x 10 d | 20 | 200 μmol total ITC equivalents (presumed by us to be </=50 μmol GR | 0.71 (GR) | ↓ Strand breaks with broccoli diet in smokers/nonsmokers. ↓ oxidized purines only in smokers. Broccoli intake did not modify HDAC activity or IGF-I serum levels | [99] | ||||||
| 2009 | Yanaka | H. pylori–infected | x | x | x | GR-rich broccoli sprouts for 8 weeks | 47 | 420 μmol GR (+ mryrosinase) | 6 (GR) | Decreased levels of urease measured by the urea breath test and H. pylori stool antigen; decreased serum biomarkers of gastric inflammation | [116] | |||||
| 2010 | Christiansen | Hypertensive, without diabetes & with normal cholesterol | x | x | x | 10 g dried broccoli sprouts/d | 40 | 259 μmol GR (measured) | 3.7 (GR) | Daily ingestion of 10 g dried broccoli sprouts does not improve endothelial function in the presence of hypertension | [108] | |||||
| 2011 | Bahadoran | Type 2 disbetes | x | x | x | 5 g or 10 g broccoli sprouts powder (BroccoPhane) said to contain SF, daily for 4 weeks (we have assayed previously and found this not to be the case) | 81 | 113 or 225 μmol SF/d based only on manufacturer′s claims | 1.61/3.22 (SF) | Significant decrease in malondialdehyde, oxidized low density lipoprotein cholesterol, oxidative stress index & significant increase in total serum antioxidant capacity | [111] | |||||
| 2011 | Clarke | Healthy non-smokers | x | x | x | x | x | x | 68 g fresh broccoli sprouts or supplement purported to contain GR + Myr from 3 g dried broccoli sprouts | 24 | ~ <220 μmol GR | 3.1 (GR) | Consumption of fresh sprouts with active myrosinase provides 5x more bioavailable SF; SF and erucin metabolites are readily interconverted | [129] | ||
| 2011 | Egner | Healthy | x | x | x | x | SF-rich beverage (containing 150 μmole SF) or GR-rich beverage (containing 800 μmole GR) daily for 7 days. | 69 | 800 μmol GR/d, 150 μmol SF /d | 11.4 (GR)/2.14 (SF) | Bioavailability of SF (measured as urinary metabolites) was substantially greater with the SF-rich than GR rich beverage | [63] | ||||
| 2011 | Healy | Healthy | x | x | x | 561 mg BSE powder (200 μmol GR) | 4 | 200 μmol GR/d | 2.86 (GR) | Consumption of BSE showed inactivation of urinary MIF tautomerase activity in urine | [113] | |||||
| 2011 | Hauder | Healthy male non-smoking (50-82 y.o.) | x | x | 200 g of blanched, regular or selenium-fertilized broccoli, daily, for 4 weeks | 76 | broccoli contained either 133 or 226 μg/g of GR | 0.87 or 1.48 (GR) | Dietary intake of selenium-fertilized broccoli increased serum selenium concentration, but affected neither glucosinolate concentrations in broccoli nor their metabolite levels in plasma and urine compared to regular broccoli. | [130] | ||||||
| 2012 | Bahadoran | Type 2 diabetes | x | x | x | 10 g or 5 g broccoli sprouts powder (BroccoPhane) said to contain SF, daily for 4 weeks. | 81 | 113 or 225 μmol SF/day based only on manufacturer′s claims | 1.61/3.22 (SF) | Consumption of 10 g/d resulted in a significant decrease in serum insulin concentration and HOMA-IR | [112] | |||||
| 2012 | Cramer | Healthy | x | x | x | x | 2 g GR powder or 42 g fresh broccoli sprouts/d; once a week for 4 weeks | 4 | 120 μmol GR/d or 70 μmol GR (fresh sprouts with active myrosinase)/d | 1.71/1 (GR) | Adding fresh broccoli sprouts (as a source of myrosinase) to GR synergistically enhanced absorption and excretion | [131] | ||||
| 2012 | Fahey | Healthy | x | x | x | 200 μmol GR | 131 | 200 μmol GR | 2.86 (GR) | Extreme inter-individual range of conversion efficiencies (1–40% of administered dose of GR) attributed to differences in microbiomes as well as circadian rhythm; within-individual differences were less pronounced | [65] | |||||
| 2012 | Kensler | Healthy | x | x | x | x | Cross-over design: GR-rich beverage (800 μmol GR) → SF-rich beverage (150 μmol SF)/SF-rich beverage (150 μmol SF) →GR-rich beverage (800 μmol GR) | 50 | 800 μmol GR/day, 150 μmol SF /d | 11.4 (GR)/2.14 (SF) | Statistically significant increases in the levels of excretion of glutathione-derived conjugates of benzene and acrolein, but not crotonaldehyde in groups receiving SF-rich, GR-rich beverages or both compared to preintervention values | [77] | ||||
| 2012 | Mirmiran | Type 2 diabetes | x | x | x | x | 5 or 10 g/d of BroccoPhane powder (BSP), reported to be rich in SF, -- daily x 4 wks (we have assayed previously and found this not to be the case) | 81 | 113 or 225 μmol SF/day based only on manufacturer′s claims | 1.61/3.22 (SF) | Sera of BSP treatment groups showed ↓ hs-CRP, and non-significant ↓ IL-6 and TNF-α | [110] | ||||
| 2012 | Saha | Healthy | x | x | x | Broccoli soups produced from fresh or frozen broccoli florets | 18 | 4.16 mg SF/serv (23.5 μmol) or 18.6 mg GR/serv. (42.7 μmol) | 0.33 (SF)/0.61 (GR) | SF bioavailability was~10x higher in fresh compared to frozen broccoli | [132] | |||||
| 2013 | Armah | 10-y CVD risk profile | x | x | 400 g standard or “HG” broccoli or placebo vegetable (peas) for 12 wk | 54 | 6.9 or 21.6 μmol GR/g dry wt broccoli | 1.48/4.63 (GR) | No significant differences on markers of CVD risk | [82] | ||||||
| 2013 | Meyer | Healthy | x | x | SF-rich homogenate prepared from 200 g BroccoSprouts (assayed for SF) daily for 3 consecutive days | 12 | 100 μmol SF/day (our est.) | 1.43 (SF) | Homogenate significantly increased secretory leukocyte protease inhibitor levels in nasal lavage fluid | [94] | ||||||
| 2013 | Poulton | Healthy | x | x | BSE (450 µmol SF/d x 7 d delivered in cheese-based soup | 23 | 450 μmol SF/d | 6.43 (SF) | No effect on CYP3A4 activity | [44] | ||||||
| 2014 | Bahadoran | Type 2 diabetic with H. pylori infection | x | x | x | 6 g/d of Cyvex broccoli sprouts powder (BSP), reported to be rich in SF, (we have assayed previously and found this not to be the case), daily, in combination with other drugs, for up to 28 d | 86 | Maximum possible is 135 μmol SF/day | 1.93 (SF) | BSP ↓ H. pylori load in diabetic patients, but no effect on gastric inflammatory markers | [117] | |||||
| 2014 | Baier | Healthy | x | x | 34, 68 or 102 g Broccoli sprouts | 8 | 170, 340, and 680 μmol GR/day | 2.4/4.8/9.6 (GR) | Dose dependent elevation in LTR (long terminal repeats) mRNA & histone acetylation in circulating leukocytes | [133] | ||||||
| 2014 | Egner | Healthy | x | x | x | x | broccoli sprouts beverages containing GR-rich and SF-rich powders (600 μmol GR + 40 μmol SF), daily, for 150 days | 267 | 600 umol GR/day + 40 μmol SF/day | 8.57 (GR) + 0.57 (SF) | Significant increases in the levels of excretion of the glutathione-derived conjugates of benzene and acrolein in urine in broccoli sprout beverage treatment group | [78] | ||||
| 2014 | Heber | Healthy | x | x | x | 1.25 g BSE suspended in juice (100 μmol SF/d x 4 d) | 29 | 100 μmol SF/d | 1.43 (SF) | Subjects challenged w/ repeated nasal diesel exhaust particles: WBC decreased 54% by daily BSE | [91] | |||||
| 2014 | Noah | Healthy | x | x | smokers/nonsmokers - broccoli sprout homogenate | 16/35 | 100 μmol SF/d (est) | 1.43 (SF) | Post BSH, live attenuated influenza virus-induced inflammatory markers reduced; NQO1 increased, in smokers but not non-smokers | [92] | ||||||
| 2014 | Singh | ♂,13–27 y.o. with moderate to severe Autism Spectrum Disorder | x | x | x | 50 - 150 µmol SF/d x 18 weeks; dosed by BW | 44 | 50 μmol SF/d - 150 μmol SF/d | 0.71 - 2.14 (SF) | SF showed substantial declines (improvement of behavior) in Aberrant Behavior Checklist and Social Responsiveness Scale scores | [87] | |||||
| 2015 | Alumkal | Prostate cancer | x | x | x | SF-rich broccoli sprout extract; 200 μmol/d x 20 weeks | 20 | 200 μmol SF/d | 2.86 (SF) | Did not lead to ≥50% PSA declines | [103] | |||||
| 2015 | Armah | 10 Year cardiovascular risk profile of between 10 and 20% | x | x | 400 g standard or “HG” broccoli or placebo vegetable (peas) for 12 wk | 130 | 6.9 or 21.6 μmol GR/g dry wt broccoli | 1.48/4.63 (GR) | High glucoraphanin (HG) broccoli diet ↓ plasma LDL-C more than standard broccoli | [85] | ||||||
| 2015 | Atwell | Abnormal mammograms; scheduled for breast biopsy | x | x | x | broccoli seed extract containing GR (BroccoMax); [we have assayed previously and found the myrosinase to be inactive and the GR titer to not be as represented], 2 capsules, 3x/d, x 2-8 wk | 54 | 180 mg GR/day (= 413 μmol GR/d) | 5.9 (GR) | ↓ PBMC HDAC activity; pre-to-post changes in Ki-67 and HDAC3; NSD in tissue biomarkers between placebo and treatment group | [98] | |||||
| 2015 | Atwell | Healthy | x | x | x | x | x | fresh broccoli sprouts (containing 200 μmol GR) or SF-rich BSE (containing 200 μmol SF) daily, consumed every 12 h. | 20 | 200 μmol SF/d; 200 μmol GR/d | 3.33 (SF); 3.33 (GR) | 3 x higher SF metabolite levels in plasma and urine in sprout consumers compared to SF-rich BSE consumers | [57] | |||
| 2015 | Brown | Moderate asthma | x | x | x | 440 mg SF-rich BSE (100 μmol SF/d x 14 d) | 45 | 100 μmol SF/d | 1.43 (SF) | Individuals in whom SF treatment enhanced the forced expiratory volume response to methacholine, had increased expression of Nrf2-regulated antioxidant and anti-inflammatory genes in peripheral blood mononuclear cells: SF treatment resulted in significant reduction in airway resistance and increased small airway luminal area | [73] | |||||
| 2015 | Chang | H. pylori | x | x | x | SF capsule twice daily for 4 weeks. | 67 | 2 mg SF/day (11.3 μmol SF/d) [we do not trust this company′s representation of dose] | 0.16 (SF) | No significant difference in urea breath test values or ammonia concentration; SF did ↓ gastric mucosal malondialdehyde but not glutathione levels | [134] | |||||
| 2015 | Cipolla | Prostate cancer patients post- radical prostatectomy | x | x | x | SF tablets (2 tablets containing 10 mg stabilized SF extracted from broccoli seeds, 3 times a day) for 6 months | 78 | 60 mg SF /day (339 μmol SF/d) | 4.83 (SF) | Decreased prostate-specific antigen (PSA) levels | [102] | |||||
| 2015 | Fahey | Healthy | x | x | x | x | x | x | 50, 69, 100, 200, 230 μmol GR in various dose forms including comparing commercial tablets to investigator-prepared materials and GR with active myrosinase | 20 | 50, 69, 100, 200 & 230 μmol GR | 0.7/1.0/1.4/2.9/3.3 (GR) | Inter- and intra-individual variabilities, when GR delivered in teas, juices, or gelatin capsules; established effect of adding active myrosinase to the “dose” | [68] | ||
| 2015 | Kikuchi | Diagnosis of fatty liver with elevated liver function markers | x | 3 broccoli sprout capsules (containing 30 mg GR) for 2 months | 55 | 69 μmol GR/d | 1 (GR) | ↓ serum ALT, AST, γ-GTP | [118] | |||||||
| 2015 | Medina | Healthy | x | x | 30 g or 60 g of broccoli sprouts | 24 | ~117 or 234 μmol GR (measured)/d as well as much smaller amounts of SF (measured) | 1.67/3.34 (GR) | Broccoli sprouts modulated excretion of biomarkers linked to inflammation and vascular reactions without exerting a significant influence on the oxidation of phospholipids | [135] | ||||||
| 2015 | Shiina | Schizophrenia | x | x | x | 30 mg GR/d x 8 weeks | 10 | 30 mg GR/d (69 μmol GR/d) | 1 (GR) | Mean score in the Accuracy component of the One Card Learning Task increased significantly after the trial. | [136] | |||||
| 2015 | Ushida | Healthy | x | x | x | dried broccoli sprout capsules (3 or 6 capsules rich in GR) | 21 | 68.7 or 137.4 μmol GR/day | 0.98/1.96 (GR) | Serum activities of GST and NQO1 were dose-dependently and synchronously elevated | [114] | |||||
| 2016 | Bauman | Healthy | x | x | 150 μmol SF oral swallowed or held in mouth, or 600 μmol GR swallowed | 10 | 8.57 (GR)/2.14 (SF) | Clinical support for good mucosal bioactivity and pharmacodynamic activity | [72] | |||||||
| 2016 | Doss | Sickle cell disease (SCD) | x | x | x | broccoli sprout homogenate (BSH) made from 50, 100, or 150 g fresh BroccoSprouts | 16 | 250, 500, or 750 μmol GR [calculated maximum delivery] | 3.6/7.1/10.7 (GR) | Homogenate is safe in SCD subjects; only modest changes in NRF2-mediated gene expression | [74] | |||||
| 2016 | Duran | Healthy | x | x | Homogenate prepared from 111 g BroccoSprouts once daily for 3 days | 16 | 555 μmol GR/day | 7.9 (GR) | On last treatment day, subjects were exposed to ozone with intermittent moderate exercise to induce airway inflammation: Homogenate did not induce expression of antioxidant genes in blood and nasal epithelial cells | [97] | ||||||
| 2016 | Müller | Healthy | x | x | Broccoli sprout homogenate shake prepared from 200 g sprout (about 100μmol of SF per dose) or alfalfa sprout homogenate for control, for 4 days | 42 | 100 μmol SF/day (est.) | 1.43 (SF) | BSH supplementation increased live attenuated influenza virus-induced granzyme B production in NK cells compared to control | [96] | ||||||
| 2016 | Sudini | Asthma | x | x | 100 g fresh broccoli sprouts (BS) /d or 100 g of alfalfa sprouts (placebo) x 3 consecutive d | 40 | no GR or SF measurements made or imputed; max poss. expected to be 500 μmol GR/d | 7.14 (GR) | No induction of cytoprotective antioxidant genes in either PBMCs or nasal epithelial cells or ↓ oxidative stress and inflammatory markers in urine and serum; no improved lung function. | [95] | ||||||
| 2016 | Wise | COPD | x | x | x | SF extracted from broccoli sprouts (25 μmol/150 μmol) daily for four weeks | 89 | 25 μmol SF/d; 150 μmol SF/d | 0.36 & 2.14 (SF) | SF did not stimulate expression of Nrf2 target genes or have an effect on levels of other anti-oxidants or markers of inflammation. | [105] | |||||
| 2017 | Axelsson | Diabetics (well regulated, and dysregulated) | x | x | x | 5410 ppm of SF x 5 g/d | 97 | 153 μmol SF/d | 2.18 (SF) | SF reduced fasting blood glucose (hepatic gluconeogenesis) & glycated hemoglobin (HbA1c) in obese patients with dysregulated T2D | [86] | |||||
| 2017 | Davidson | Osteoarthritis | x | x | 100 g high glucosinolate (HG) broccoli/d x 14 d | 40 | 180 μmol GR/d | 2.57 (GR) | ITCs detected in synovial fluid of HG group, but not the low glucosinolate group. | [59] | ||||||
| 2017 | Fahey | Healthy | x | x | x | x | x | 94.4 μmol SF/d or 200 μmol α-cyclodextrin enrobed SF/d | 10 | 94.4 μmol SF/d | 1.35 & 2.86 (SF) | PK and tolerance of α-cyclodextrin enrobed SF (for theoretical stabilization) was compared to that of SF along, in the commercial “stabilized SF” product Prostaphane™ | [66] | |||
| 2018 | Sedlak | Healthy | x | x | x | 100 μmol SF/d x 7 d taken in 2 gel caps per day | 9 | 100 μmol SF/d | 1.43 (SF) | Correlation between blood and thalamic GSH post- and pre-SF treatment ratios and a consistent increase in brain GSH levels (7 Tesla MRI) | [81] | |||||
| 2018 | Tahata | Melanoma | x | x | x | Broccoli sprout extract-SF standardized for 50, 100, or 200 µmol SF for 28 days | 17 | 50 μmol /100 umol/200 μmol SF/day | 0.71/1.43/2.86 (SF) | Oral BSE-SF is well tolerated at 50, 100, and 200 μmol /day attaining blood plasma and skin biopsy levels reasonable for pharmacodynamic action | [104] | |||||
| 2018 | Bent | Children with ASD and related neurodevelopmental disorders | x | x | 6 to 15 Avmacol tablets (222 μmol GR to 555 μmol GR) daily for 12 weeks, depending on BW | 15 | 222 umol GR/day - 555 umol GR/day | 3.17 - 7.93 (GR) | Mean scores on both symptom measures showed improvements (decreases) over the study period, which was correlated with urinary metabolites | [120] | ||||||
| 2018 | Housley | Healthy | x | x | fresh broccoli sprouts (containing 200 μmol GR) | 10 | 200 μmol GR/d | 2.86 (GR) | Untargeted metabolomic screen of human plasma following consumption of fresh broccoli sprouts | [137] | ||||||
| 2018 | Okunade | Healthy | x | x | x | 200 g raw, cooked broccoli ± 1 g mustard powder | 12 | 62, 32, or 257 μmol GR/d based on conversion of author′s dry wt., to (our) fresh wt. basis | 0.88/0.45/3.7 (GR) | ↑ urinary SF-NAC when cooked broccoli consumed with mustard powder | [138] | |||||
| 2018 | Sivapalan | Healthy | x | x | 300 g Myb28B/B (standard) or Myb28B/V (Beneforte) or Myb28V/V broccoli soup; single dose | 10 | 84, 280, or 452 μmol GR/d | 1.2/4.0/6.5 (GR) | Three different Myb28 genotypes of broccoli related with delivery of sulforaphane to the systemic circulation. | [52] | ||||||
| 2019 | Chartoumpekis | Healthy women | x | x | x | x | Broccoli sprouts beverages containing GR-rich and SF-rich powders (600 μmol GR + 40 μmol SF), daily, for 150 days | 45 | 600 umol GR/day + 40 μmol SF/day | 8.57 (GR) + 0.57 (SF) | Measurement of thyroglobulin, TSK, free thyroxine, and others | [62] | ||||
| 2019 | Lopez Chillon | Healthy, overweight | x | x | 30 g/d of fresh broccoli sprouts, for 10 wks followed by 10 wks of washout | 40 | 117 μmol GR/d (measured) | 1.67 (GR) | Reduced IL-6 and CRP following broccoli sprout consumption | [109] | ||||||
| 2019 | Chen | Healthy | x | x | x | x | Broccoli sprout beverage (contained GR and SF), full dose or half dose or fifth dose, daily for 10 consecutive days | 170 | 600 μmol GR + 40 µmol SF or 300 μmol GR + 20 µmol SF or 125μmol GR + 8 µmol SF/day | 600/40, 300/20, or 125/8 (GR + SF) | Benzene mercapturic acids in urine was increased in high dose treated group, but not in half dose and one-fifth dose | [69] | ||||
| 2019 | Fahey | Healthy | x | x | x | Avmacol - (GR with active myrosinase; 6 tablets211 μ, mol GR, single dose, n=20); and 8 Avmacol tablets (369 μmol GR), on 4 separate days, enteric-coated and not-coated | 20 & 16 | 211 and 369 μmol GR/day | 3.01/5.27 (GR) | Gastric acidity somewhat attenuates activity of oral myrosinase reducing conversion of GR to SF, and thus SF bioavailability; cytoprotection, antioxidant and detoxification gene expression increased with increasing SF bioavailability. | [67] | |||||
| 2019 | Traka | Men with low to intermed. risk prostate cancer | x | x | broccoli | 61 | 72 - 492 μmol GR | 1 - 7 (GR) | Affected gene expression in the prostates of men under active surveillance, consistent with prevention | [80] | ||||||
| 2020 | Bauman | Head and neck cancer survivors | x | x | Avmacol 50 or 100 mg GR (115 or 230 μmol GR) | 36 | 115 or 230 μmol GR | 1.6-3.3 | Ongoing; “Preventing Recurrence in Patients With Tobacco-Related Head and Neck Squamous Cell Cancer” | NCT03182959 | ||||||
| 2020 | Kim | H. pylori infected adults (18-75 y.o.) | x | broccoli sprout extract | 360 | -- | Ongoing; “The Effect of Broccoli Sprout Extract and Probiotics for Eradication of Helicobacter Pylori” | NCT03220542 | ||||||||
| 2020 | Bauman | Smokers | x | x | Avmacol - 4 or 8 tablets/d | 61 | (our calculation) ~ 138 and 275 μmol GR | 2.0/4.0 (GR) | Ongoing; Decreasing Toxicity in Heavy Smokers | NCT03402230 | ||||||
| 2020 | Dickerson | Adults (18-65 y.o.) with schizophrenia | x | x | x | Avmacol | 64 | weight-based, about 1.4 μmol/kg BW | 1.42 (GR) | Ongoing; amelioration of symptoms of schizophrenia | NCT02810964 | |||||
| 2020 | Hua/Davis | Children (3-15 y.o.) on the autism spectrum | x | x | x | Avmacol | 110 | -- | Ongoing; amelioration of symptoms of autism spectrum disorder (ASD) | NCT02879110 | ||||||
| 2020 | Hua/Davis | Adults with 1st episode or early onset schizophrenia | x | x | x | Avmacol | 180 | -- | Ongoing; amelioration of symptoms of schizophrenia | NCT02880462 | ||||||
| 2020 | Johnson | Young adults (13-30 y.o.), on the autism spectrum | x | x | x | Avmacol | 45 | weight-based, about 1.5 μmol/kg BW | 1.47 (GR) | Ongoing; amelioration of symptoms of autism spectrum disorder (ASD) | NCT02677051 | |||||
| 2020 | Li | Veterans with allergic rhinitis | x | x | broccoli sprout extract | 475 | Phase 2 RCT; BSE paired with fluticasone or normal saline spray | -- | Ongoing; “Effects of Broccoli Sprout Extract on Allergy Rhinitis” | NCT 0288 5025 | ||||||
| 2020 | Politte | Young men (13-30 y.o.), on the autism spectrum | x | x | x | Avmacol | 48 | weight-based, about 1.4 μmol/kg BW | 1.4 (GR) | Ongoing; amelioration of symptoms of autism spectrum disorder (ASD) | NCT 0290 9959 | |||||
| 2020 | Tex Tech | Doxirubicin-naïve women with breast cancer | x | x | Avmacol (2 - 8 tablets/d) x 12 weeks; weight based | 70 | 68.8 - 275 μmol GR | 1 - 3.9 (GR) | Ongoing; “Effects of the SF on doxorubicin-associated cardiac dysfunction” | NCT 0393 4905 | ||||||
| 2020 | Wang | Adults at risk for psychosis | x | x | x | Chinese commercial “GR + Myros. “ supplement (Zhiyinguosu) | 300 | 52 wk, daily about 411 μmol GR with active myrosinase | 5.9 (GR) | Ongoing; 1° planned outcome -- conversion to psychosis | NCT 0393 2136 | |||||
| 2020 | Wu | 1st episode or early onset schizophrenia (SZ) | x | x | Avmacol | 180 | -- | Ongoing; “A 6-month Study to Evaluate SF add-on Effects in Treatment of SZ” | NCT 0288 0462 | |||||||
| 2020 | Yuan | Former Smokers | x | x | Avmacol | 72 | 120 μmol 2x/d | 3.4 (GR) | Ongoing; lung cancer prevention in former smokers | NCT 0323 2138 | ||||||
| 2020 | Zandberg | Head & neck cancer patients post-curative treatment | x | x | x | Avmacol; escalating daily doses, from 2 to 4 to 8 tabs per day for a month each | 36 | 69, 138, or 275 μmol GR | 1/2/3.9 (GR) | Ongoing | NCT 0326 8993 | |||||
| 2020 | Zimmerman | Children (3-12 y.o.), on the autism spectrum | x | x | x | Avmacol | 60 | weight-based, about 2.2 μmol/kg BW | 2.2 (GR) | Ongoing; amelioration of symptoms of autism spectrum disorder | NCT 0256 1481 | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yagishita, Y.; Fahey, J.W.; Dinkova-Kostova, A.T.; Kensler, T.W. Broccoli or Sulforaphane: Is It the Source or Dose That Matters? Molecules 2019, 24, 3593. https://doi.org/10.3390/molecules24193593
Yagishita Y, Fahey JW, Dinkova-Kostova AT, Kensler TW. Broccoli or Sulforaphane: Is It the Source or Dose That Matters? Molecules. 2019; 24(19):3593. https://doi.org/10.3390/molecules24193593
Chicago/Turabian StyleYagishita, Yoko, Jed W. Fahey, Albena T. Dinkova-Kostova, and Thomas W. Kensler. 2019. "Broccoli or Sulforaphane: Is It the Source or Dose That Matters?" Molecules 24, no. 19: 3593. https://doi.org/10.3390/molecules24193593
APA StyleYagishita, Y., Fahey, J. W., Dinkova-Kostova, A. T., & Kensler, T. W. (2019). Broccoli or Sulforaphane: Is It the Source or Dose That Matters? Molecules, 24(19), 3593. https://doi.org/10.3390/molecules24193593







