Xanthommatin is Behind the Antioxidant Activity of the Skin of Dosidicus gigas
Abstract
1. Introduction
2. Results
2.1. Isolation of the Bioactive Pigments
2.2. Antioxidant Activity
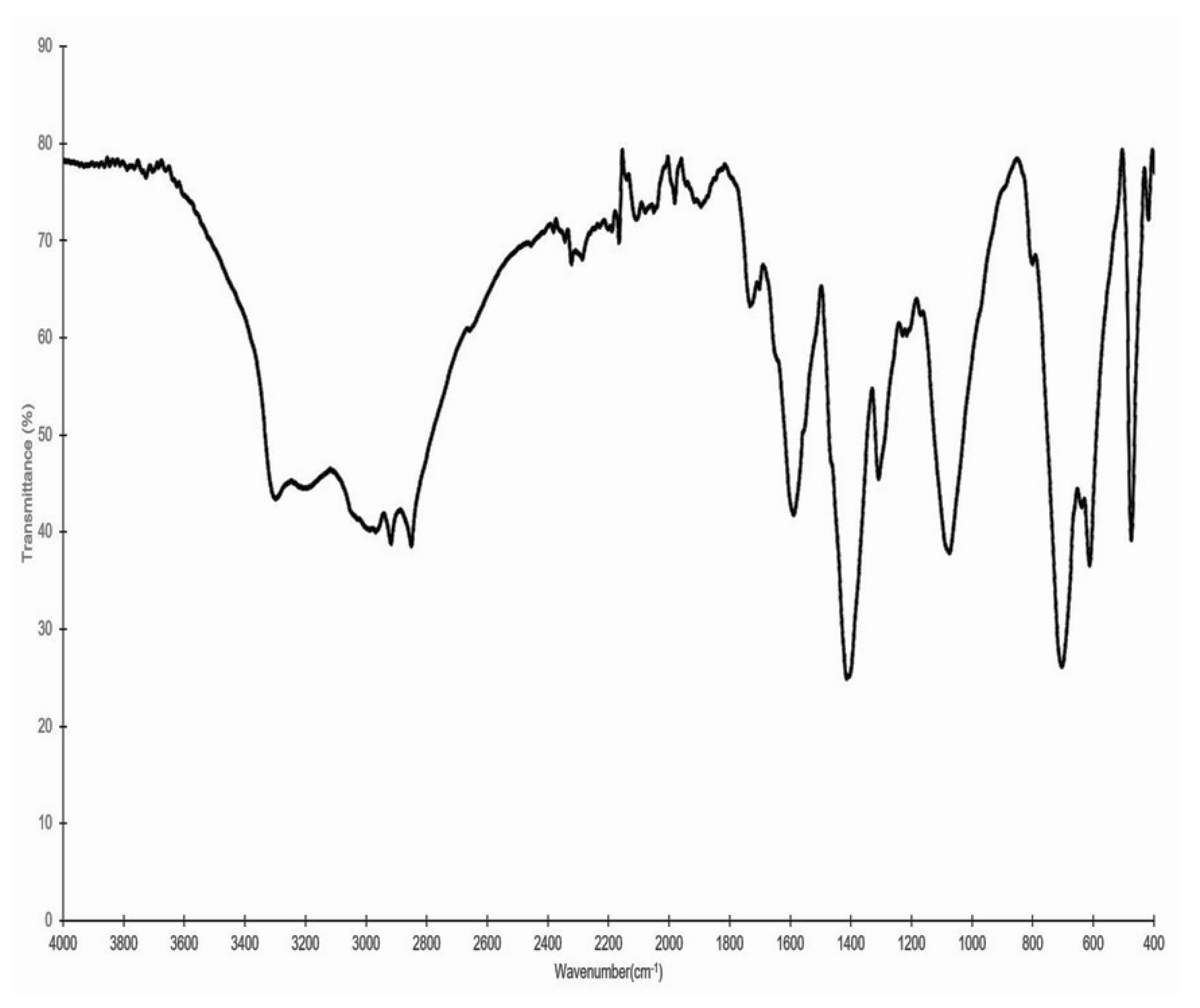
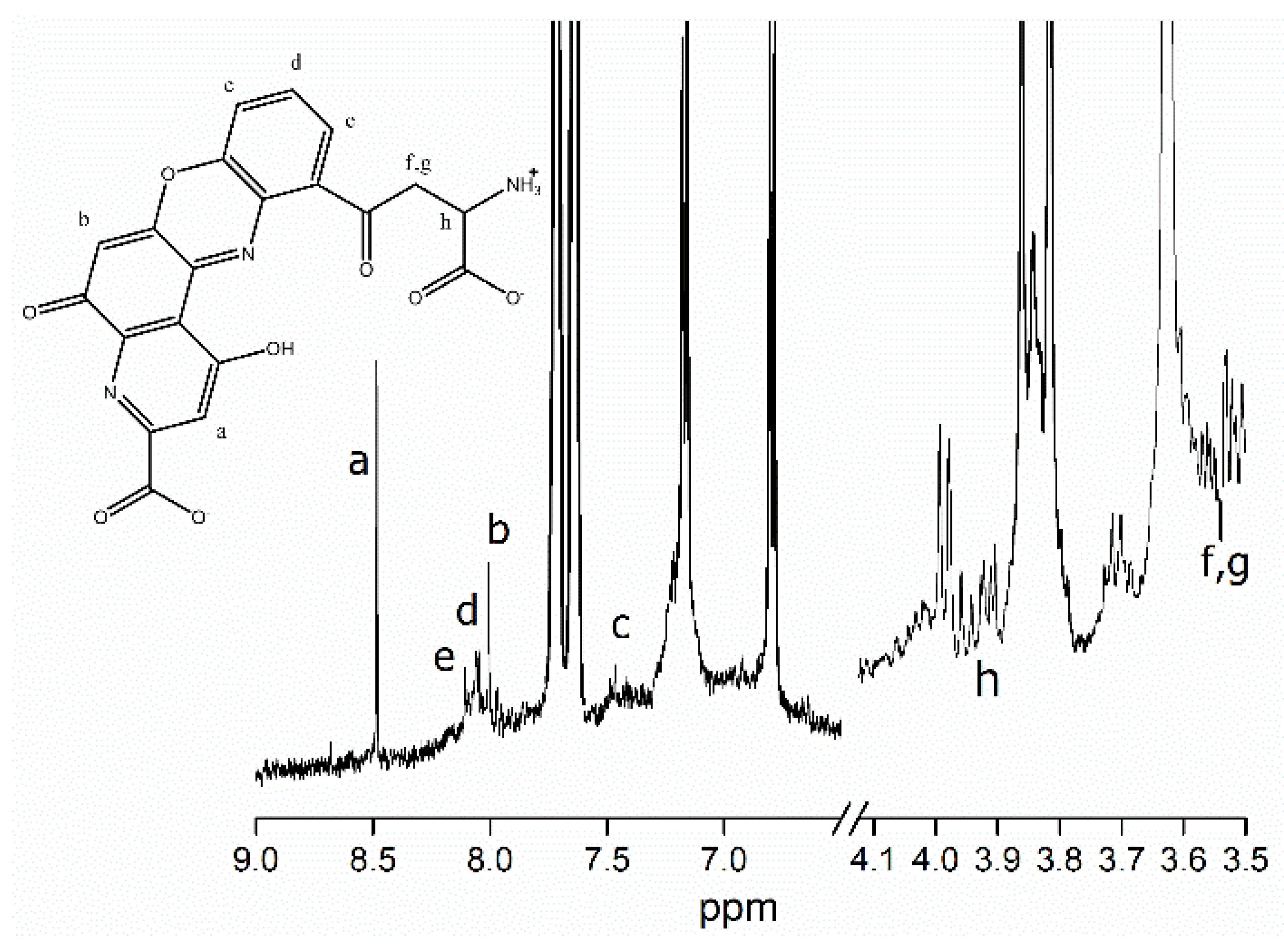
2.3. Structure Elucidation
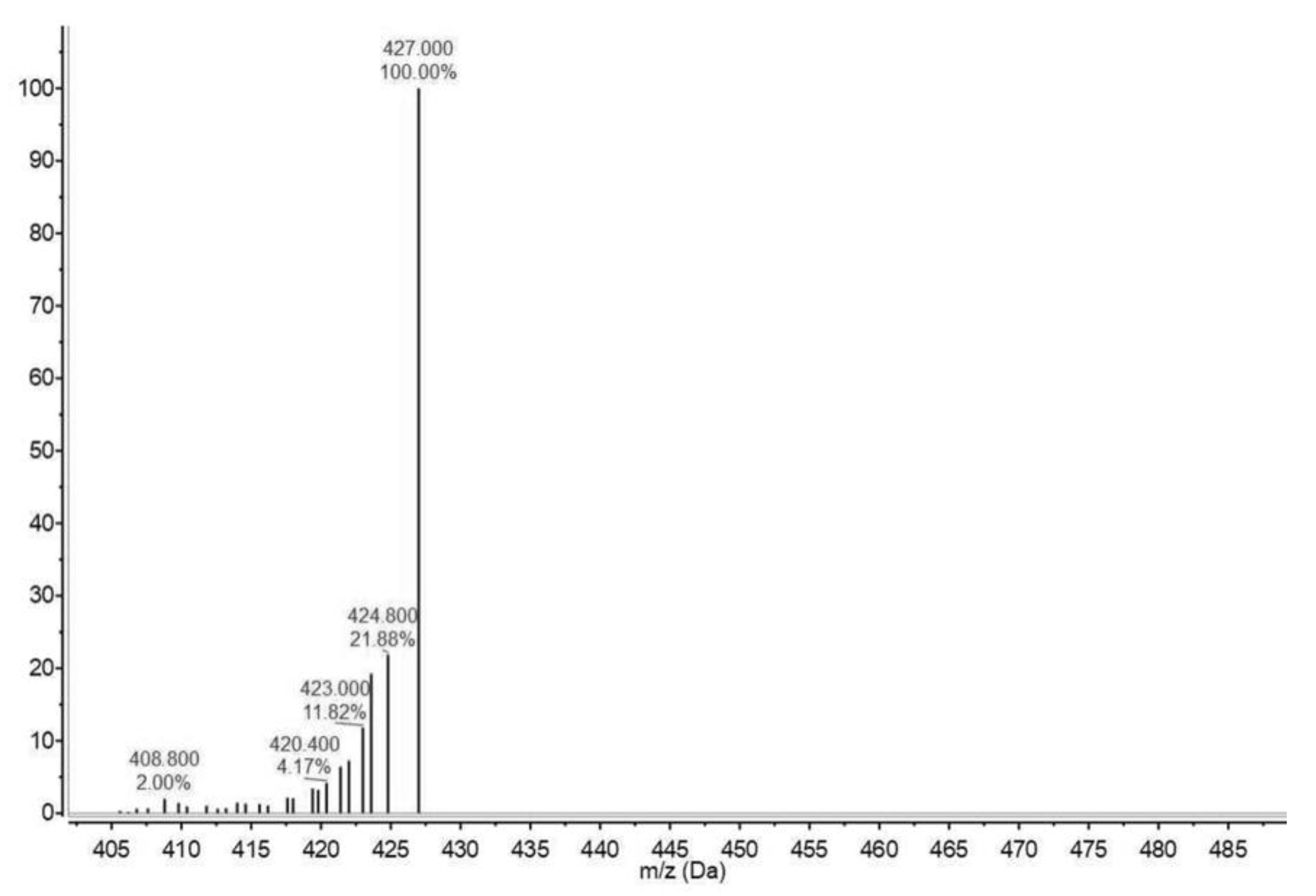
2.4. Electrospray Ionization–Mass Spectrometry
3. Discussion
4. Materials and Methods
4.1. Sample Preparation
4.2. Pigment Extraction
4.3. Fractioning by Open Column Chromatography
4.4. Thin-Layer Chromatography
4.5. Antioxidant Activity
4.6. Spectroscopic Methods
4.7. Electrospray Ionization–Mass Spectrometry
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Suleria, H.A.R.; Gobe, G.; Masci, P.; Osborne, S.A. Marine bioactive compounds and health promoting perspectives; innovation pathways for drug discovery. Trends Food Sci. Tech. 2016, 50, 44–55. [Google Scholar] [CrossRef]
- Suárez-Jimenez, G.M.; Robles-Sánches, R.M.; Yépiz-Plascencia, G.; Burgos-Hernández, A.; Ezquerra-Brauer, J.M. In vitro antioxidant, antimutagenic and antiproliferative activities of collagen hydrolysates of jumbo squid (Dosidicus gigas) byproducts. Food Sci. Technol. (Campinas). 2015, 35, 421–427. [Google Scholar] [CrossRef]
- Arias-Moscoso, J.L.; Maldonado-Arce, A.; Rouzaud-Sandez, O.; Márquez-Ríos, E.; Torres-Arreola, W.; Santacruz-Ortega, H.; Gaxiola-Cortés, M.G.; Ezquerra-Brauer, J.M. Physicochemical characterization of protein hydrolysates produced by Autolysis of jumbo squid (Dosidicus gigas) byproducts. Food Biophys. 2015, 10, 145–154. [Google Scholar] [CrossRef]
- Fu, W.D.; Wang, Y.; Zheng, B.; Liao, M.F.; Zhang, W.J. Isolation and characterization of pepsin-soluble collagen from the skin of Peru squid (Dosidicus gigas). J. Aquat. Food Prod. Technol. 2013, 22, 270–280. [Google Scholar] [CrossRef]
- Chan-Higuera, J.E.; Robles-Sánchez, R.M.; Burgos-Hernández, A.; Márquez-Ríos, E.; Velázquez-Contreras, C.A.; Ezquerra-Brauer, J.M. Squid by-product gelatines: Effect on oxidative stress biomarkers in healthy rats. Czech J. Food Sci. 2016, 34, 105–110. [Google Scholar] [CrossRef]
- Cuevas-Acuña, D.A.; Robles-Sánchez, R.M.; Torres-Arreola, W.; Márquez-Ríos, E.; Ezquerra-Brauer, J.M. Collagen from jumbo squid fin: Extracting conditions and influence of the protease system on collagen hydrolysate antioxidant activity. CYTA-J. Food. 2016, 14, 193–199. [Google Scholar] [CrossRef]
- Aubourg, S.P.; Torres-Arreola, W.; Trigo, M.; Ezquerra-Brauer, J.M. Partial characterization of jumbo squid skin pigment extract and its antioxidant potential in a marine oil system. Eur. J. Lipid Sci. Tech. 2016, 118, 1293–1304. [Google Scholar] [CrossRef]
- Sutherland, R.L.; Mathger, L.M.; Hanlon, R.T.; Urbas, A.M.; Stone, M.O. Cephalopod coloration model. II. Multiple layer skin effects. J. Opt. Soc. Am. A. 2008, 25, 2044–2054. [Google Scholar] [CrossRef]
- Riou, M.; Christides, J.-P. Cryptic Color Change in a Crab Spider (Misumena vatia): Identification and quantification of precursors and ommochrome pigments by HPLC. J. Chem. Ecol. 2010, 36, 412–423. [Google Scholar] [CrossRef]
- Shamim, G.; Ranjan, S.K.; Pandey, D.M.; Ramani, R. Biochemistry and biosynthesis of insect pigments. Eur. J. Entomol. 2014, 111, 149–164. [Google Scholar] [CrossRef]
- Ostrovsky, M.A.; Sakina, N.L.; Dontsov, A.E. An antioxidative role of ocular screening pigments. Vision Res. 1987, 27, 893–899. [Google Scholar] [CrossRef]
- Egorov, S.Y.; Dontsov, A.E.; Krasnovsky, A.A.J.; Ostrovsky, M.A. Quenching of singlet molecular oxygen by screening pigments– melanin and ommochromes. Biofizika. 1987, 32, 685–686. [Google Scholar]
- Ezquerra-Brauer, J.M.; Miranda, J.M.; Cepeda, A.; Barros-Velazquez, J.; Aubourg, S.P. Effect of jumbo squid (Dosidicus gigas) skin extract on the microbial activity in chilled mackerel (Scomber scombrus). LWT-Food Sci. Technol. 2016, 72, 134–140. [Google Scholar] [CrossRef]
- Ezquerra-Brauer, J.M.; Miranda, J.M.; Chan-Higuera, J.E.; Barros-Velázquez, J.; Aubourg, S.P. New icing media for quality enhancement of chilled hake (Merluccius merluccius) using a jumbo squid (Dosidicus gigas) skin extract. J. Sci. Food Agric. 2017, 97, 3412–3419. [Google Scholar] [CrossRef] [PubMed]
- Giménez, B.; Alemán, A.; Montero, P.; Gómez-Guillén, M.C. Antioxidant and functional properties of gelatin hydrolysates obtained from skin of sole and squid. Food Chem. 2009, 114, 976–983. [Google Scholar] [CrossRef]
- Bolognese, A.; Scherillo, G. Occurrence and characterization of a labile xanthommatin precursor in some invertebrates. Experientia 1974, 30, 225–226. [Google Scholar] [CrossRef] [PubMed]
- Bolognese, A.; Liberatore, R. Photochemistry of ommochrome pigments. J. Hetercocyclic Chem. 1988, 25, 1243–1246. [Google Scholar] [CrossRef]
- Crescenzi, O.; Correale, G.; Bolognese, A.; Piscopo, V.; Parrilli, M.; Barone, V. Observed and calculated 1H- and 13C-NMR chemical shifts of substituted 5H-pyrido[3,2-a]- and 5H-pyrido[2,3-a]phenoxazin-5-ones and of some 3H-phenoxazin-3-one derivatives. Org. Biomol. Chem. 2004, 2, 1577–1581. [Google Scholar] [CrossRef]
- Figon, F.; Casas, J. Ommochromes in invertebrates: Biochemistry and cell biology. Biol. Rev. 2019, 94, 156–183. [Google Scholar] [CrossRef]
- Sawada, H.; Tsusué, M.; Iino, T. Identification of ommin in the integument of the silkworm, Bombyx mori. Biol. Chem. Hoppe-Seyler. 1994, 375, 425–427. [Google Scholar]
- Kato, T.; Sawada, H.; Yamamoto, T.; Mase, K.; Nakagoshi, M. Pigment pattern formation in the quail mutant of the silkworm, Bombyx mori: Parallel increase of pteridine biosynthesis and pigmentation of melanin and ommochromes. Pigment Cell Res. 2006, 19, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Aybastier, O.; Şahin, S.; Demir, C. Response surface optimized ultrasonic-assisted extraction of quercetin and isolation of phenolic compounds from Hypericum perforatum L. by column chromatography. Sep. Sci. Technol. 2013, 48, 1665–1674. [Google Scholar] [CrossRef]
- Romero, Y.; Martinez, A. Antiradical capacity of ommochromes. J. Mol. Model. 2015, 21, 220. [Google Scholar] [CrossRef] [PubMed]
- Lian, X.; Liu, L.; Dong, S.; Wu, H.; Zhao, J.; Han, Y. Two new monascus red pigments produced by Shandong Zhonghui Food Company in China. Eur. Food Res.Technol. 2015, 240, 719–724. [Google Scholar] [CrossRef]
- Cai, Z.; Wu, J.; Chen, L.; Guo, W.; Li, J.; Wang, J.; Zhang, Q. Purification and characterisation of aquamarine blue pigment from the shells of abalone (Haliotis discus hannai Ino). Food Chem. 2011, 128, 129–133. [Google Scholar] [CrossRef]
- Topal, F.; Nar, M.; Gocer, H.; Kalin, P.; Kocyigit, U.M.; Gulcin, I.; Alwasel, S.H. Antioxidant activity of taxifolin: An activity-structure relationship. J. Enzyme Inhib. Med. Chem. 2016, 31, 674–683. [Google Scholar] [CrossRef]
- Blanco-Diaz, M.T.; Del Rio-Celestino, M.; Martinez-Valdivieso, D.; Font, R. Use of visible and near-infrared spectroscopy for predicting antioxidant compounds in summer squash (Cucurbita pepo ssp pepo). Food Chem. 2014, 164, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Williams, T.L.; Martin, C.A.; Figueroa-Navedo, A.M.; Deravi, L.F. Xanthommatin-based electrochromic displays inspired by nature. ACS Appl. Mater. Interfaces. 2018, 10, 43177–43183. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Ezymol. 1999, 299, 15–27. [Google Scholar]
| Fraction | ABTS2 (IC50 mg mL−1) | FRAP2 (IC50 mg mL−1) | DPPH2 (IC50 mg mL−1) |
|---|---|---|---|
| F1 | 2.12 ± 0.11a | 6.54 ± 0.06c | 6.49 ± 0.04e |
| F2 | 2.77 ± 0.08c | 4.15 ± 0.11b | 4.67 ± 0.03c |
| F3 | 2.07 ± 0.11a | 2.52 ± 0.20a | 3.56 ± 0.08b |
| F4 | 10.2 ± 0.02e | 12.34 ± 0.33d | 8.69 ± 0.09e |
| F5 | 6.02 ± 0.05d | 6.89 ± 0.07c | 9.52 ± 1.01e |
| F6 | 2.25 ± 0.02b | 3.98 ± 0.10b | 5.34 ± 0.05d |
| F7 | 2.08 ± 0.02a | 2.25 ± 0.09a | 2.60 ± 0.04a |
| F8 | 5.78 ± 1.02d | 7.30 ± 0.03c | 3.44 ± 0.09b |
| Mixture of solvents | Proportion |
|---|---|
| Ethyl acetate/Methanol | 60:40 |
| Ethyl acetate/Methanol | 40:60 |
| Ethyl acetate/Methanol | 20:80 |
| Methanol | 100 |
| Acetic acid/Water | 5:95 |
| Acetic acid/Water | 10:90 |
| Ammonium hydroxide/Water | 4:96 |
| Ammonium hydroxide/Water | 8:92 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan-Higuera, J.E.; Santacruz-Ortega, H.d.C.; Carbonell-Barrachina, Á.A.; Burgos-Hernández, A.; Robles-Sánchez, R.M.; Cruz-Ramírez, S.G.; Ezquerra-Brauer, J.M. Xanthommatin is Behind the Antioxidant Activity of the Skin of Dosidicus gigas. Molecules 2019, 24, 3420. https://doi.org/10.3390/molecules24193420
Chan-Higuera JE, Santacruz-Ortega HdC, Carbonell-Barrachina ÁA, Burgos-Hernández A, Robles-Sánchez RM, Cruz-Ramírez SG, Ezquerra-Brauer JM. Xanthommatin is Behind the Antioxidant Activity of the Skin of Dosidicus gigas. Molecules. 2019; 24(19):3420. https://doi.org/10.3390/molecules24193420
Chicago/Turabian StyleChan-Higuera, Jesús Enrique, Hisila del Carmen Santacruz-Ortega, Ángel A. Carbonell-Barrachina, Armando Burgos-Hernández, Rosario Maribel Robles-Sánchez, Susana Gabriela Cruz-Ramírez, and Josafat Marina Ezquerra-Brauer. 2019. "Xanthommatin is Behind the Antioxidant Activity of the Skin of Dosidicus gigas" Molecules 24, no. 19: 3420. https://doi.org/10.3390/molecules24193420
APA StyleChan-Higuera, J. E., Santacruz-Ortega, H. d. C., Carbonell-Barrachina, Á. A., Burgos-Hernández, A., Robles-Sánchez, R. M., Cruz-Ramírez, S. G., & Ezquerra-Brauer, J. M. (2019). Xanthommatin is Behind the Antioxidant Activity of the Skin of Dosidicus gigas. Molecules, 24(19), 3420. https://doi.org/10.3390/molecules24193420







