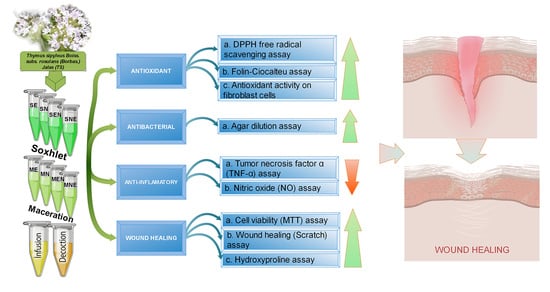
In Vitro Evaluation of Antioxidant, Anti-Inflammatory, Antimicrobial and Wound Healing Potential of Thymus Sipyleus Boiss. Subsp. Rosulans (Borbas) Jalas
Abstract
1. Introduction
2. Results and Discussion
2.1. Antioxidant Activities of TS Extracts
2.2. Antibacterial Activities of TS Extracts
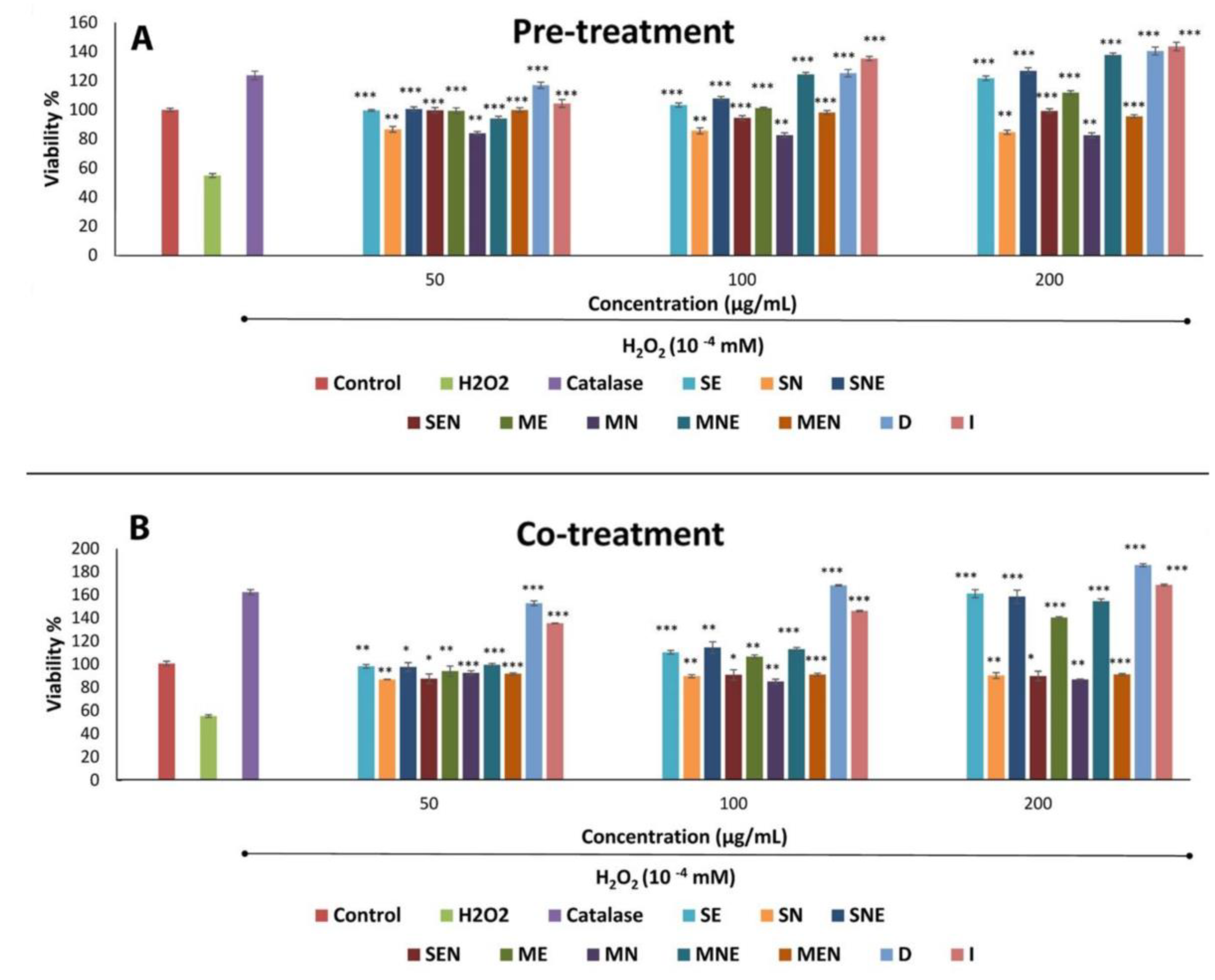
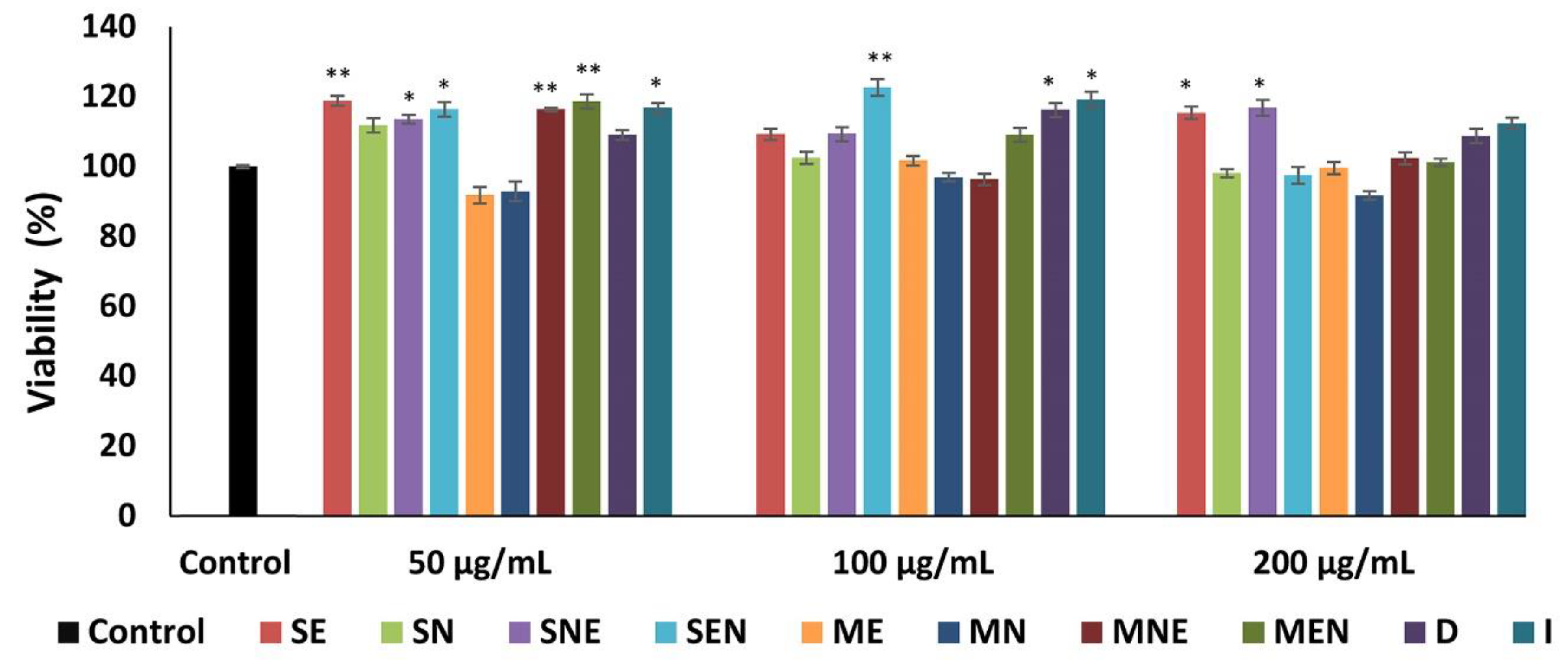
2.3. Effects of TS Extracts on Cell Proliferation
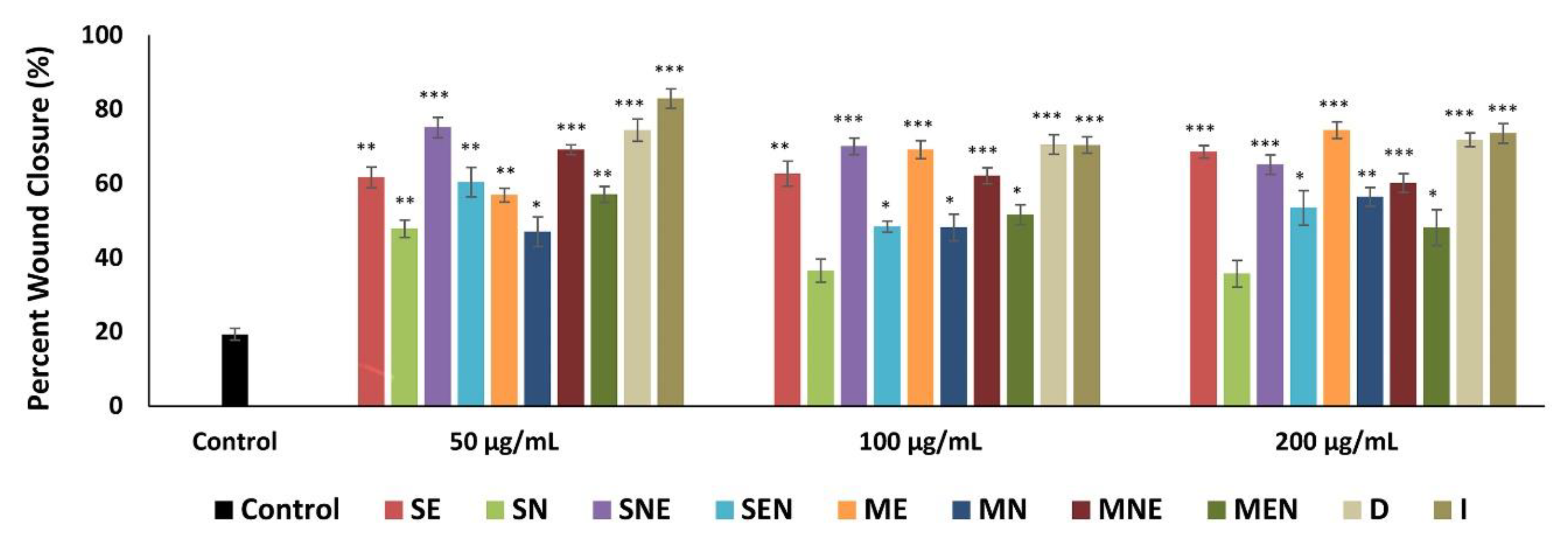
2.4. Scratch Wound Healing of Fibroblasts in the Presence of TS Extracts
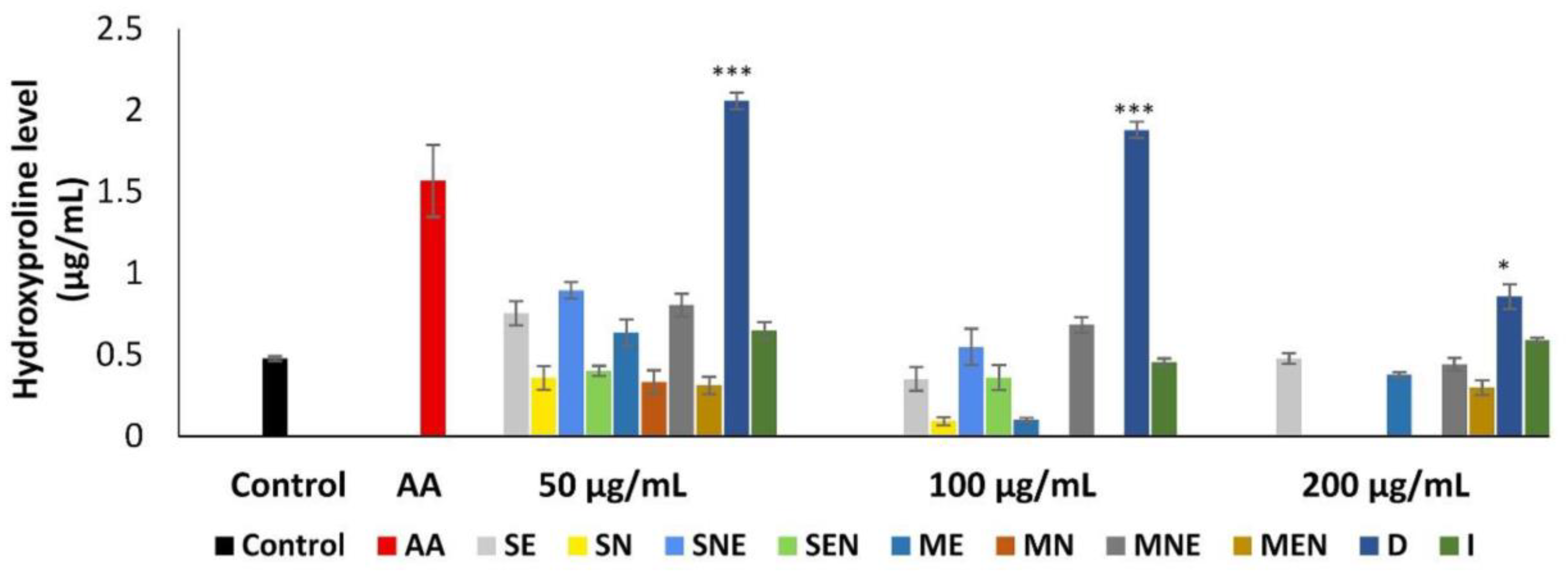
2.5. Effects of TS Extracts on Hydroxyproline Level
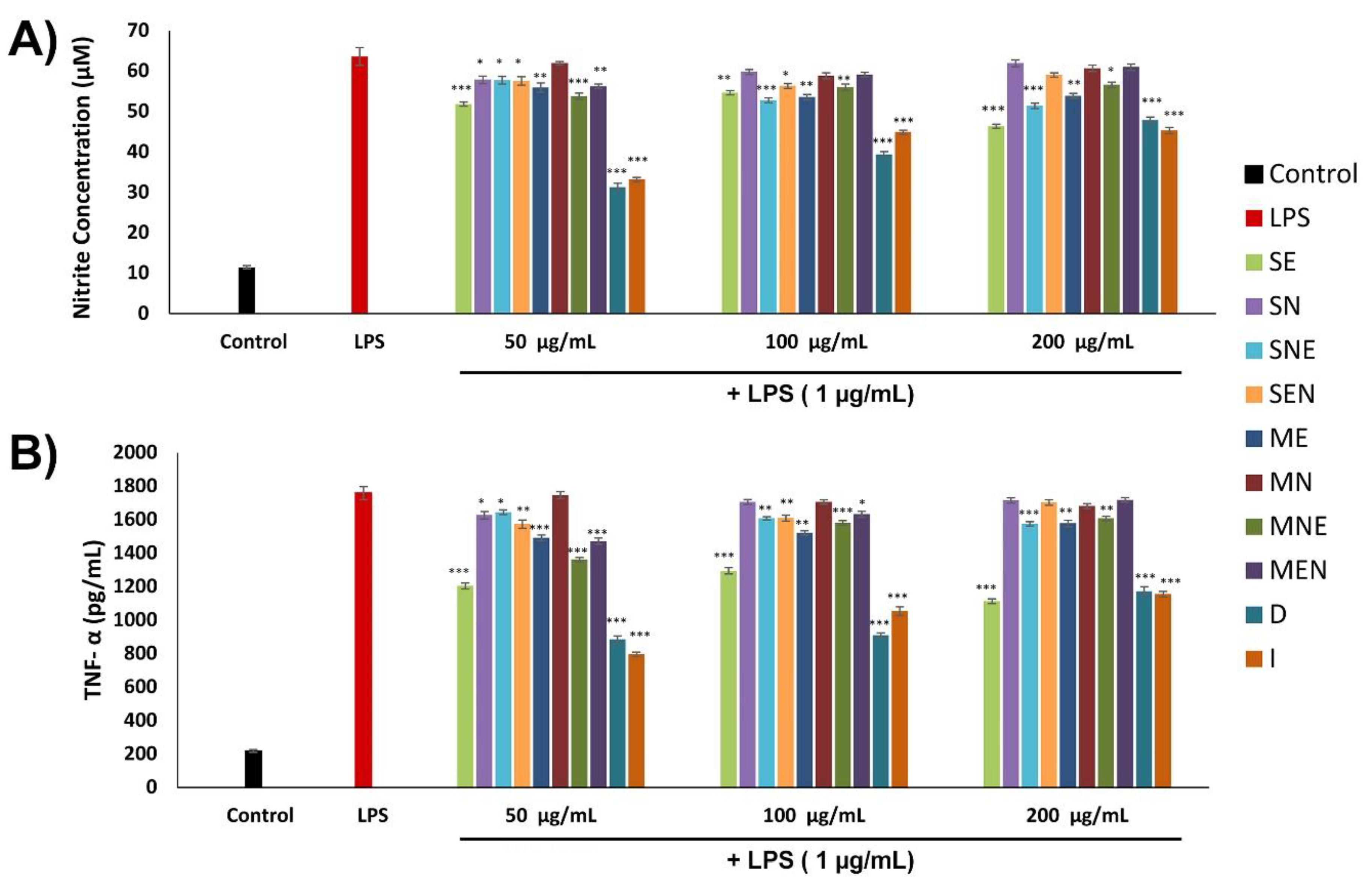
2.6. Anti-Inflammatory Activities of TS Extracts
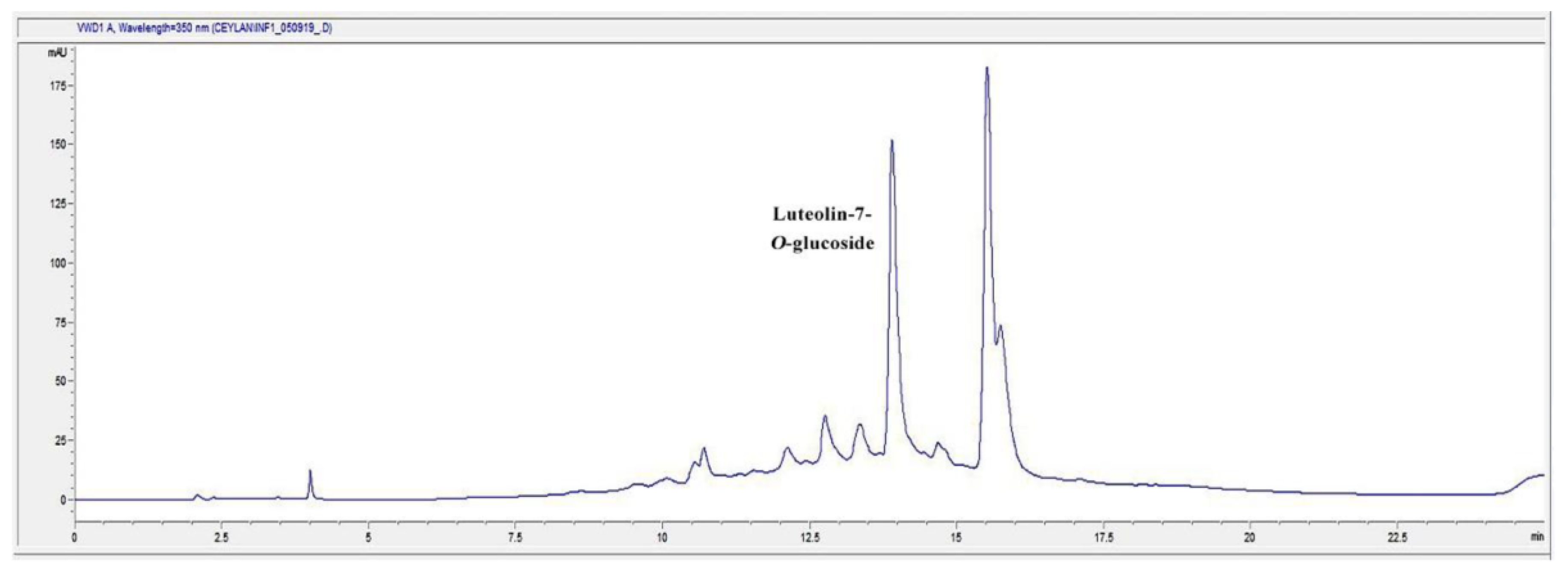
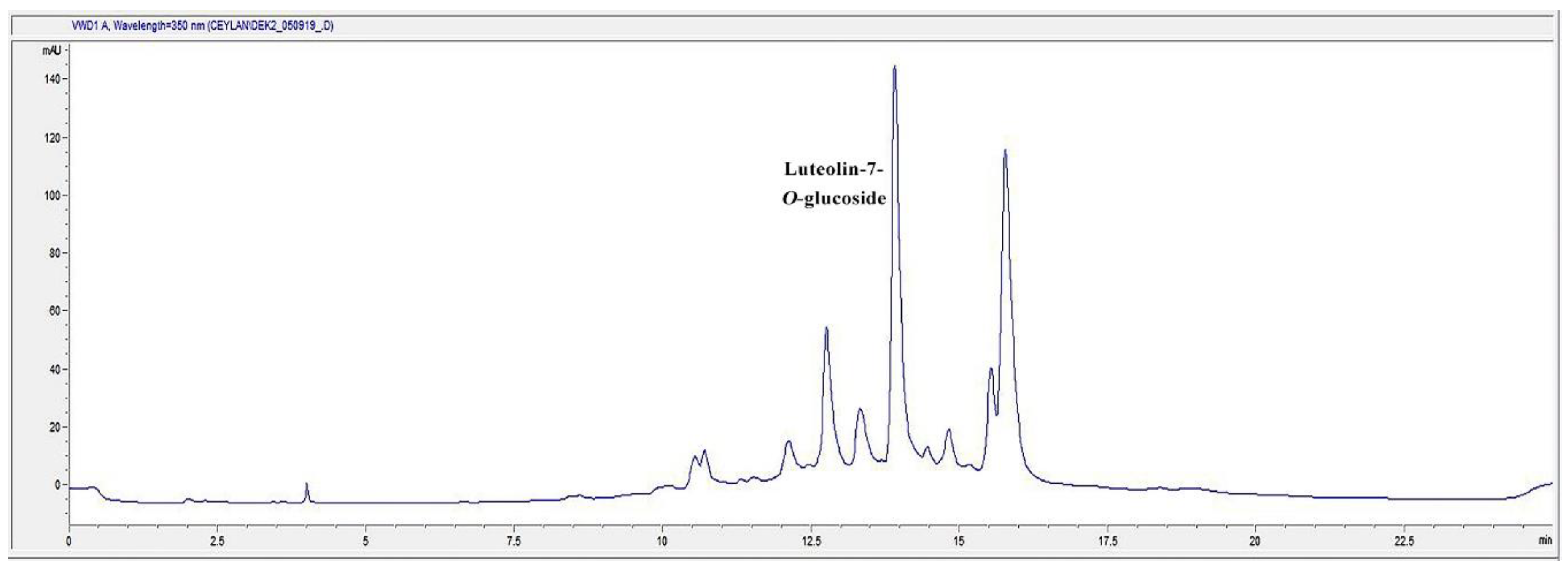
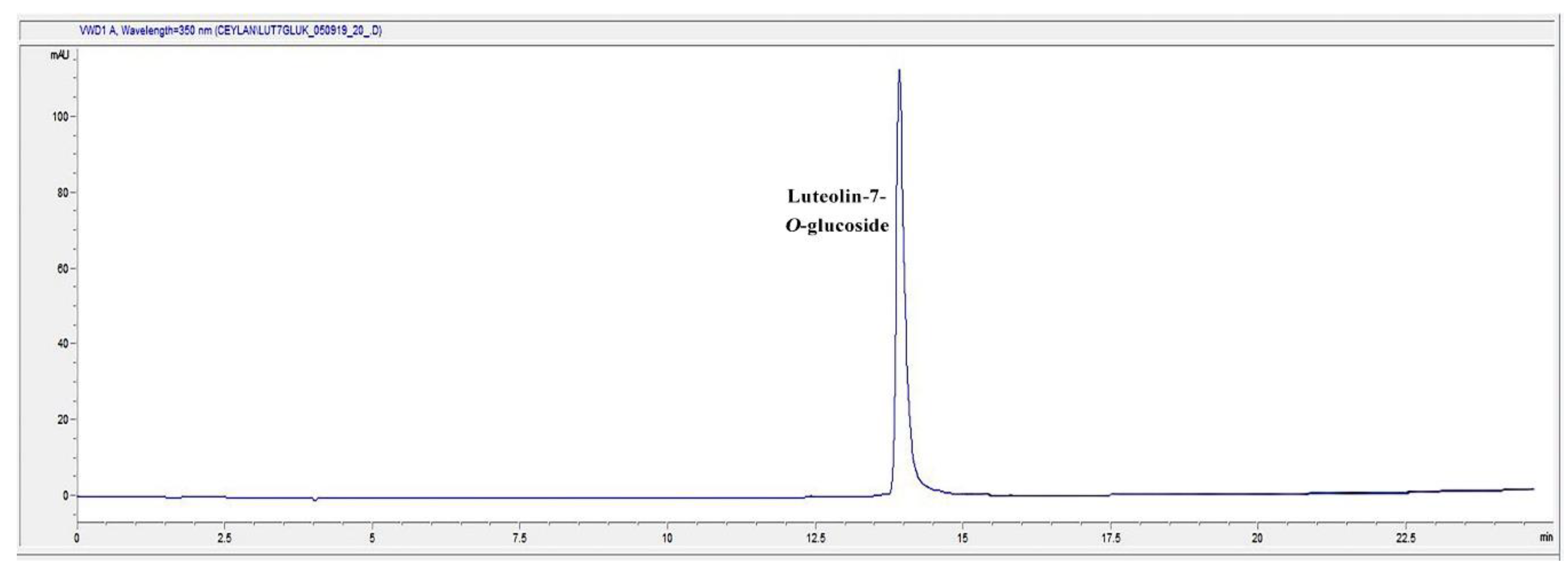
2.7. HPLC Analysis of the Infusion (I) and Decoction (D) Extracts for Luteolin-7-O-glucoside Content
3. Materials and Methods
3.1. Plant Material
3.2. Preparation of Extracts
3.3. DPPH Radical Scavenging Assay
3.4. Folin-Ciocalteu Assay
3.5. Agar Dilution Assay
3.6. Cell Culture and Reagents
3.7. Cell Viability Assay
3.8. In Vitro Wound Healing (Scratch) Assay
3.9. Nitric Oxide (NO) Assay
3.10. Tumor Necrosis Factor-Alpha (TNF-α) Assay
3.11. Hydroxyproline Assay
3.12. Antioxidant Activity of Extracts on 3T3-Swiss Albino Mouse Fibroblast Cells
3.13. Phytochemical Analysis
3.14. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sen, C.K. Human Wounds and Its Burden: An Updated Compendium of Estimates. Adv. Wound Care 2019, 8, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Kokane, D.D.; More, R.Y.; Kale, M.B.; Nehete, M.N.; Mehendale, P.C.; Gadgoli, C.H. Evaluation of wound healing activity of root of Mimosa pudica. J. Ethnopharmacol. 2009, 124, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Thakur, R.; Jain, N.; Pathak, R.; Sandhu, S.S. Practices in Wound Healing Studies of Plants. Evid. Based Complement. Altern. Med. 2011, 2011, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, A.A.; Pauzi, N.A.S.; Arulselvan, P.; Abas, F.; Fakurazi, S. In VitroWound Healing Potential and Identification of Bioactive Compounds fromMoringa oleiferaLam. BioMed Res. Int. 2013, 2013, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Süntar, I.; Küpeli Akkol, E.; Nahar, L.; Sarker, S.D. Wound healing and antioxidant properties: Do they coexist in plants? Free Rad. Antiox. 2012, 2, 1–7. [Google Scholar] [CrossRef]
- Sarmento, P.D.A.; Ataíde, T.D.R.; Barbosa, A.P.F.; De Araújo-Júnior, J.X.; Lúcio, I.M.L.; Bastos, M.L.D.A. Evaluation of the extract of Zeyheria tuberculosa with a view to products for wound healing. Rev. Latino-Americana de Enferm. 2014, 22, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Premoli, M.; Bonini, S.A.; Maccarinelli, G.; Gianoncelli, A.; Memo, M.; Mastinu, A. Cannabimimetic plants: Are they new cannabinoidergic modulators? Planta 2019, 249, 1681–1694. [Google Scholar] [CrossRef]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef]
- Anlas, C.; Bakirel, T.; Ustun-Alkan, F.; Celik, B.; Baran, M.Y.; Ustuner, O.; Kuruuzum-Uz, A. In vitro evaluation of the therapeutic potential of Anatolian kermes oak (Quercus coccifera L.) as an alternative wound healing agent. Ind. Crop. Prod. 2019, 137, 24–32. [Google Scholar] [CrossRef]
- Pereira, R.F.; Bártolo, P.J. Traditional Therapies for Skin Wound Healing. Adv. Wound Care 2016, 5, 208–229. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Traditional Medicine Strategy: 2014–2023; World Health Organization: Geneva, Switzerland; Available online: https://apps.who.int/iris/bitstream/handle/10665/92455/9789241506090_eng.pdf;jsessionid=3EEF37EEC1AE12ACD5DF9F8C103DBDDB?sequence=1 (accessed on 3 July 2019).
- European Parliament and of the Council. Directive 2004/24/EC. Amendment, as Regards Traditional Herbal Medicinal Products, of the Directive 2001/83/EC on the Community Code Relating to Medicinal Products for Human Use. Strasbourg, France. Available online: https://eurlex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2004:136:0085:0090:en:PDF (accessed on 10 July 2019).
- Özgen, U.; Mavi, A.; Terzi, Z.; Kazaz, C.; Asçı, A.; Kaya, Y.; Seçen, H. Relationship between chemical structure and antioxidant activity of luteolin and its glycosides isolated from Thymus sipyleus subsp. sipyleus var. Sipyleus. Rec. Nat. Prod. 2011, 5, 12–21. [Google Scholar] [CrossRef]
- Özcan, S.; Toprak, G.; Torun, C.; Vural, C. Thymus sipyleus Boiss subsp. rosulans (Borbas) J. Jalas’ın organik ekstrakt ve uçucu yağlarının antimikrobiyal aktivitesi. Bibad 2008, 1, 17–22. [Google Scholar]
- Gökbulut, A. Validated RP-HPLC Method for Quantification of Phenolic Compounds in Methanol Extracts of Aerial Parts and Roots of Thymus sipyleus and Evaluation of Antioxidant Potential. Trop. J. Pharm. Res. 2015, 14, 1871. [Google Scholar] [CrossRef]
- Koyuncu, O.; Yaylacı, Ö.K.; Öztürk, D.; Potoğlu Erkara, İ.; Savaroğlu, F.; Akcoşkun, Ö.; Ardıç, M. Risk categories and ethnobotanical features of the Lamiaceae taxa growing naturally in Osmaneli (Bilecik/Turkey) and environs. Biol. Divers. Conserv. 2010, 3, 31–45. [Google Scholar]
- Abarca-Vargas, R.; Malacara, C.F.P.; Petricevich, V.L. Characterization of Chemical Compounds with Antioxidant and Cytotoxic Activities in Bougainvillea x buttiana Holttum and Standl, (var. Rose) Extracts. Antioxidants 2016, 5, 45. [Google Scholar] [CrossRef] [PubMed]
- Thouri, A.; Chahdoura, H.; El Arem, A.; Hichri, A.O.; Ben Hassin, R.; Achour, L. Effect of solvents extraction on phytochemical components and biological activities of Tunisian date seeds (var. Korkobbi and Arechti). BMC Complement. Altern. Med. 2017, 17, 379. [Google Scholar] [CrossRef] [PubMed]
- Azahar, N.F.; Gani, S.S.A.; Mokhtar, N.F.M. Optimization of phenolics and flavonoids extraction conditions of Curcuma Zedoaria leaves using response surface methodology. Chem. Central J. 2017, 11, 96. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, R.; Pasalar, P.; Kamalinejad, M.; Dehpour, A.R.; Tavangar, S.M.; Paknejad, M.; Mehrabani Natanzi, M.; Nourbakhsh, M.; Ahmadi Ashtiani, H.R.; Akbari, M.; et al. The effect of silymarin (Silybum marianum) on human skin fibroblasts in an in vitro wound healing model. Pharm. Biol. 2013, 51, 298–303. [Google Scholar] [CrossRef]
- Barku, V.Y.A.; Boye, A.; Erzah, F.; Tsamenyi, P. In-vitro Antioxidant and Wound Healing Properties of Combretum dolichopetalum Engl. and Diels (Combretaceae). J. Appl. Pharm. Sci. 2016, 6, 185–192. [Google Scholar] [CrossRef][Green Version]
- Agyare, C.; Dwobeng, A.S.; Agyepong, N.; Boakye, Y.D.; Mensah, K.B.; Ayande, P.G.; Adarkwa-Yiadom, M. Antimicrobial, antioxidant, and wound healing properties of Kigelia africana (Lam.) Beneth. and Strophanthus hispidus DC. Adv. Pharmacol. Sci. 2013, 2013, 692613. [Google Scholar]
- Vittorazzi, C.; Endringer, D.C.; Andrade, T.U.; Scherer, R.; Fronza, M. Antioxidant, antimicrobial and wound healing properties of Struthanthus vulgaris. Pharm. Biol. 2016, 54, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Tepe, B.; Sökmen, M.; Akpulat, H.; Daferera, D.; Polissiou, M.; Sokmen, A.; Akpulat, H.A. Antioxidative activity of the essential oils of Thymus sipyleus subsp. sipyleus var. sipyleus and Thymus sipyleus subsp. sipyleus var. rosulans. J. Food Eng. 2005, 66, 447–454. [Google Scholar] [CrossRef]
- Piluzza, G.; Bullitta, S. Correlations between phenolic content and antioxidant properties in twenty-four plant species of traditional ethnoveterinary use in the Mediterranean area. Pharm. Boil. 2011, 49, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Khatoon, M.; Islam, E.; Islam, R.; Rahman, A.A.; Alam, A.K.; Khondkar, P.; Rashid, M.; Parvin, S. Estimation of total phenol and in vitro antioxidant activity of Albizia procera leaves. BMC Res. Notes 2013, 6, 121. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.G.; Yun, C.K.; Lee, H.S. Screening and comparison of antioxidant activity of solvent extracts of herbal medicines used in Korea. J. Ethnopharmacol. 2003, 87, 231–236. [Google Scholar] [CrossRef]
- Sultana, B.; Anwar, F.; Ashraf, M. Effect of Extraction Solvent/Technique on the Antioxidant Activity of Selected Medicinal Plant Extracts. Molecules 2009, 14, 2167–2180. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.-P.; Chen, Y.-F.; Zhang, J.-L.; You, M.-M.; Wang, K.; Hu, F.-L. Mechanisms underlying the wound healing potential of propolis based on its in vitro antioxidant activity. Phytomedicine 2017, 34, 76–84. [Google Scholar] [CrossRef]
- Houghton, P.; Hylands, P.; Mensah, A.; Hensel, A.; Deters, A. In vitro tests and ethnopharmacological investigations: Wound healing as an example. J. Ethnopharmacol. 2005, 100, 100–107. [Google Scholar] [CrossRef]
- Pitz, H.D.S.; Pereira, A.; Blasius, M.B.; Voytena, A.P.L.; Affonso, R.C.L.; Fanan, S.; Trevisan, A.C.D.; Ribeiro-Do-Valle, R.M.; Maraschin, M. In Vitro Evaluation of the Antioxidant Activity and Wound Healing Properties of Jaboticaba (Plinia peruviana) Fruit Peel Hydroalcoholic Extract. Oxidative Med. Cell. Longev. 2016, 2016, 1–6. [Google Scholar] [CrossRef]
- Ramchoun, M.; Harnafi, H.; Alem, C.; Benlyas, M.; Elrhaffari, L.; Amrani, S. Study on antioxidant and hypolipidemic effects of polyphenol-rich extracts from Thymus vulgaris and Lavendula multifida. Pharmacognosy Res. 2009, 1, 106–112. [Google Scholar]
- Kulisic, T.; Radonic, A.; Milos, M. Antioxidant properties of Thyme (Thymus Vulgaris, L.) and wild thyme (Thymus Serpyllum, L.) essential oils. Ital. J. Food Sci. 2005, 17, 315–324. [Google Scholar]
- Roby, M.H.H.; Sarhan, M.A.; Selim, K.A.H.; Khalel, K.I. Evaluation of antioxidant activity, total phenols and phenolic compounds in thyme (Thymus vulgaris L.), sage (Salvia officinalis L.), and marjoram (Origanum majorana L.) extracts. Ind. Crop. Prod. 2013, 43, 827–831. [Google Scholar] [CrossRef]
- Eruygur, N.; Ataş, M.; Çevir, Ö; Tekin, M. Investigating of Phytochemicals, Antioxidant, Antimicrobial and Proliferative Properties of Different Extracts of Thymus spathulifolius Hausskn. and Velen. Endemic Medicinal Plant from Sivas, Turkey. Int. J. Second. Metab. 2017, 4, 155–166. [Google Scholar] [CrossRef]
- Guo, S.; DiPietro, L.A. Factors affecting wound healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.; Harding, K.G. Bacteria and wound healing. Curr. Opin. Infect. Dis. 2004, 17, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Benz, R.; Bauer, K. Permeation of hydrophilic molecules through the outer membrane of gram-negative bacteria. Review on bacterial porins. Eur J. Biochem. 1988, 176, 1–19. [Google Scholar] [CrossRef]
- Ababutain, I.M. Antimicrobial activity of ethanolic extracts from some medicinal plant. AJBAS 2011, 5, 678–683. [Google Scholar]
- Bouyahya, A.; Bakri, Y.; Et-Touys, A.; Talbaoui, A.; Khouchlaa, A.; Idrissi, A.E.Y.E.; Abrini, J.; Dakka, N. In vitro Screening of Antibacterial and Antioxidant Activities of Essential Oils from Four Moroccan Medicinal Plants. Microbiol. Res. J. Int. 2017, 18, 1–10. [Google Scholar] [CrossRef]
- Inouye, S.; Yamaguchi, H.; Takizawa, T. Screening of the antibacterial effects of a variety of essential oils on respiratory tract pathogens, using a modified dilution assay method. J. Infect. Chemother. 2001, 7, 251–254. [Google Scholar] [CrossRef]
- Duarte, M.C.T.; Figueira, G.M.; Sartoratto, A.; Rehder, V.L.G.; Delarmelina, C. Anti-Candida activity of Brazilian medicinal plants. J. Ethnopharmacol. 2005, 97, 305–311. [Google Scholar] [CrossRef]
- Fabry, W.; O Okemo, P.; Ansorg, R. Antibacterial activity of East African medicinal plants. J. Ethnopharmacol. 1998, 60, 79–84. [Google Scholar] [CrossRef]
- Mummed, B.; Abraha, A.; Feyera, T.; Nigusse, A.; Assefa, S. In Vitro Antibacterial Activity of Selected Medicinal Plants in the Traditional Treatment of Skin and Wound Infections in Eastern Ethiopia. BioMed Res. Int. 2018, 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Salawu, S.O.; Ogundare, A.O.; Ola-Salawu, B.B.; Akindahunsi, A.A. Antimicrobial activities of phenolic containing extracts of some tropical vegetables. Afr. J. Pharm. Pharmacol. 2011, 5, 486–492. [Google Scholar]
- Kuhnt, M.; Pröbstle, A.; Rimpler, H.; Bauer, R.; Heinrich, M. Biological and Pharmacological Activities and Further Constituents ofHyptis verticillata. Planta Medica 1995, 61, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Memar, M.Y.; Raei, P.; Alizadeh, N.; Aghdam, M.A.; Kafil, H.S. Carvacrol and thymol: Strong antimicrobial agents against resistant isolates. Rev. Med. Microbiol. 2017, 28, 63–68. [Google Scholar] [CrossRef]
- Çetin, B.; Çakmakçı, S.; Çakmakçı, R. The investigation of antimicrobial activity of thyme and oregano essential oils. Turk. J. Agric. For. 2011, 35, 145–154. [Google Scholar]
- Singh, D.; Singh, D.; Choi, S.M.; Zo, S.M.; Painuli, R.M.; Kwon, S.W.; Han, S.S. Effect of Extracts of Terminalia chebula on Proliferation of Keratinocytes and Fibroblasts Cells: An Alternative Approach for Wound Healing. Evid. Based Complement. Altern. Med. 2014, 2014, 1–13. [Google Scholar] [CrossRef]
- Muniandy, K.; Gothai, S.; Tan, W.S.; Kumar, S.S.; Esa, N.M.; Chandramohan, G.; Al-Numair, K.S.; Arulselvan, P. In Vitro Wound Healing Potential of Stem Extract of Alternanthera sessilis. Evid. Based Complement. Altern. Med. 2018, 2018, 3142073. [Google Scholar] [CrossRef]
- Varga, E.; Bardocz, A.; Belak, A.; Maraz, A.; Boros, B.; Felinger, A.; Böszörmenyi, A.; Horvath, G. Antimicrobial activity and chemical composition of thyme essential oils and the polyphenolic content of different Thymus extracts. Farmacia 2015, 63, 357–361. [Google Scholar]
- Panah, K.G.; Hesaraki, S.; Farahpour, M.R. Histopathological evaluation of Thymus Vulgaris on wound healing. Indian, J. Fund. App. Life Sci. 2014, 4, 3538–3544. [Google Scholar]
- Khosravitabar, F.; Abrishamchi, P.; Bahrami, A.R.; Matin, M.M.; Ejtehadi, H.; Varasteh-Kojourian, M. Enhanced cutaneous wound healing by the leaf extract of Achillea eriophora D.C. using the in vitro scratch assay. J. Sci. I. R. Iran. 2017, 28, 305–312. [Google Scholar]
- Siddhuraju, P.; Becker, K. Antioxidant Properties of Various Solvent Extracts of Total Phenolic Constituents from Three Different Agroclimatic Origins of Drumstick Tree (Moringa oleiferaLam.) Leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef] [PubMed]
- Grada, A.; Otero-Vinas, M.; Prieto-Castrillo, F.; Obagi, Z.; Falanga, V. Research Techniques Made Simple: Analysis of Collective Cell Migration Using the Wound Healing Assay. J. Investig. Dermatol. 2017, 137, e11–e16. [Google Scholar] [CrossRef] [PubMed]
- Girija, D.M.; Kalachaveedu, M.; Subbarayan, R.; Jenifer, P.; Rao, S.R. Aristolochia bracteolata Enhances Wound Healing in vitro through Anti-inflammatory and Proliferative Effect on Human Dermal Fibroblasts and Keratinocytes. Pharmacogn. J. 2017, 9, 129–136. [Google Scholar] [CrossRef]
- Bainbridge, P. Wound healing and the role of fibroblasts. J. Wound Care 2013, 22, 407–412. [Google Scholar] [PubMed]
- Lai, H.Y.; Lim, Y.Y.; Kim, K.H. Potential dermal wound healing agent in Blechnum orientale Linn. BMC Complement. Altern. Med. 2011, 11, 62. [Google Scholar] [CrossRef]
- Udupa, A.; Kulkarni, D.; Udupa, S. Effect of Tridax Procumbens Extracts on Wound Healing. Int. J. Pharmacogn. 1995, 33, 37–40. [Google Scholar] [CrossRef]
- Stipcevic, T.; Piljac, J.; Berghe, D.V. Effect of Different Flavonoids on Collagen Synthesis in Human Fibroblasts. Plant Foods Hum. Nutr. 2006, 61, 27–32. [Google Scholar] [CrossRef]
- Da Silva, S.M.M.; Costa, C.R.R.; Gelfuso, G.M.; Guerra, E.N.S.; Nóbrega, Y.K.D.M.; Gomes, S.M.; Pic-Taylor, A.; Fonseca-Bazzo, Y.M.; Silveira, D.; Magalhães, P.D.O. Wound Healing Effect of Essential Oil Extracted from Eugenia dysenterica DC (Myrtaceae) Leaves. Molecules 2018, 24, 2. [Google Scholar] [CrossRef]
- Schwentker, A.; Vodovotz, Y.; Weller, R.; Billiar, T.R. Nitric oxide and wound repair: role of cytokines? Nitric Oxide 2002, 7, 1–10. [Google Scholar] [CrossRef]
- Sudsai, T.; Wattanapiromsakul, C.; Nakpheng, T.; Tewtrakul, S. Evaluation of the wound healing property of Boesenbergia longiflora rhizomes. J. Ethnopharmacol. 2013, 150, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Geller, F.C.; Teixeira, M.R.; Pereira, A.B.D.; Dourado, L.P.A.; Souza, D.G.; Braga, F.C.; Simões, C.M.O. Evaluation of the Wound Healing Properties ofHancornia speciosaLeaves. Phytotherapy Res. 2015, 29, 1887–1893. [Google Scholar] [CrossRef] [PubMed]
- Wedler, J.; Daubitz, T.; Schlotterbeck, G.; Butterweck, V. In Vitro Anti-Inflammatory and Wound-Healing Potential of a Phyllostachys edulis Leaf Extract – Identification of Isoorientin as an Active Compound. Planta Medica 2014, 80, 1678–1684. [Google Scholar] [CrossRef] [PubMed]
- Afonso, A.F.; Pereira, O.R.; Válega, M.; Silva, A.M.S.; Cardoso, S.M. Metabolites and Biological Activities of Thymus zygis, Thymus pulegioides, and Thymus fragrantissimus Grown under Organic Cultivation. Molecules 2018, 23, 1514. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.-R.; Chung, K.-S.; Cheon, S.-Y.; Lee, M.; Hwang, S.; Hwang, S.N.; Rhee, K.-J.; An, H.-J. Rosmarinic acid suppresses colonic inflammation in dextran sulphate sodium (DSS)-induced mice via dual inhibition of NF-κB and STAT3 activation. Sci. Rep. 2017, 7, 46252. [Google Scholar] [CrossRef]
- Cao, W.; Hu, C.; Wu, L.; Xu, L.; Jiang, W. Rosmarinic acid inhibits inflammation and angiogenesis of hepatocellular carcinoma by suppression of NF-κB signaling in H22 tumor-bearing mice. J. Pharmacol. Sci. 2016, 132, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.-B.; Yuan, Y.-J.; Zhang, Q.-H.; Li, H.; Dai, J.-L.; Min, J.-K. Salvianolic Acid B Suppresses Inflammatory Mediator Levels by Downregulating NF-κB in a Rat Model of Rheumatoid Arthritis. Med Sci. Monit. 2018, 24, 2524–2532. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K. The general case for redox control of wound repair. Wound Repair Regen. 2003, 11, 431–438. [Google Scholar] [CrossRef]
- García-Lafuente, A.; Guillamón, E.; Villares, A.; Rostagno, M.A.; Martínez, J.A. Flavonoids as anti-inflammatory agents: Implications in cancer and cardiovascular disease. Inflamm. Res. 2009, 58, 537–552. [Google Scholar] [CrossRef]
- Siqueira, M.; Li, J.; Chehab, L.; Desta, T.; Chino, T.; Krothpali, N.; Behl, Y.; Alikhani, M.; Yang, J.; Braasch, C.; et al. Impaired wound healing in mouse models of diabetes is mediated by TNF-α dysregulation and associated with enhanced activation of forkhead box O1 (FOXO1). Diabetologia 2010, 53, 378–388. [Google Scholar] [CrossRef]
- Aziz, N.; Kim, M.-Y.; Cho, J.Y. Anti-inflammatory effects of luteolin: A review of in vitro, in vivo, and in silico studies. J. Ethnopharmacol. 2018, 225, 342–358. [Google Scholar] [CrossRef] [PubMed]
- Özay, Y.; Güzel, S.; Erdoğdu, I.H.; Yıldırım, Z.; Pehlivanoglu, B.; Türk, B.A.; Darcan, S. Evaluation of the Wound Healing Properties of Luteolin Ointments on Excision and Incision Wound Models in Diabetic and Non-Diabetic Rats. Rec. Nat. Prod. 2018, 12, 350–366. [Google Scholar] [CrossRef]
- Bayrami, Z.; Khalighi-Sigaroodi, F.; Rahimi, R.; Farzaei, M.H.; Hodjat, M.; Baeeri, M.; Rahimifard, M.; Navaei-nigjeh, M.; Abdollahi, M.; Hajiaghaee, R. In vitro wound healing activity of luteolin. Res. J. Pharmacogn. 2017, 4, 7. [Google Scholar]
- Schmidt, C.; Fronza, M.; Goettert, M.; Geller, F.; Luik, S.; Flores, E.; Bittencourt, C.; Zanetti, G.; Heinzmann, B.; Laufer, S.; et al. Biological studies on Brazilian plants used in wound healing. J. Ethnopharmacol. 2009, 122, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-B.; Jiang, Y.; Wong, C.-C.; Cheng, K.-W.; Chen, F. Evaluation of two methods for the extraction of antioxidants from medicinal plants. Anal. Bioanal. Chem. 2007, 388, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Albayrak, S.; Aksoy, A.; Sagdıc, O.; Albayrak, S. Antioxidant and antimicrobial activities of different extracts of some medicinal herbs consumed as tea and spices in Turkey. J. Food. Biochem. 2012, 36, 547–554. [Google Scholar] [CrossRef]
- Yang, S.-A.; Jeon, S.-K.; Lee, E.-J.; Im, N.-K.; Jhee, K.-H.; Lee, S.-P.; Lee, I.-S. Radical Scavenging Activity of the Essential Oil of Silver Fir (Abies alba). J. Clin. Biochem. Nutr. 2009, 44, 253–259. [Google Scholar] [CrossRef]
- Domínguez, M.; Nieto, A.; Marin, J.C.; Keck, A.-S.; Jeffery, E.; Cespedes, C.L.; Dominguez-Lopez, M. Antioxidant Activities of Extracts fromBarkleyanthus salicifolius(Asteraceae) andPenstemon gentianoides(Scrophulariaceae)†. J. Agric. Food Chem. 2005, 53, 5889–5895. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Sixteenth Informational Supplement. CLSI document M100-S16 [ISBN 1-56238-588-7]; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2006. [Google Scholar]
- Chen, L.-G.; Yang, L.-L.; Wang, C.-C. Anti-inflammatory activity of mangostins from Garcinia mangostana. Food Chem. Toxicol. 2008, 46, 688–693. [Google Scholar] [CrossRef]
- Jorge, M.P.; Madjarof, C.; Ruiz, A.L.T.G.; Fernandes, A.T.; Rodrigues, R.A.F.; Sousa, I.M.D.O.; Foglio, M.A.; De Carvalho, J.E. Evaluation of wound healing properties of Arrabidaea chica Verlot extract. J. Ethnopharmacol. 2008, 118, 361–366. [Google Scholar] [CrossRef]
- Annan, K.; Dickson, R. Evaluation of wound healing actions of Hoslundia opposita Vahl, Anthocleista nobilis G. Don and Balanites aegyptiaca L. J. Sci. Technol. 2008, 28, 26–35. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |

| Extracts | DPPH Scavenging Activity | Total Phenolic Content |
|---|---|---|
| EC50 (μg/mL) | mg GAE/g Extract | |
| SE | 121.63 ± 1.67 | 113.9 ± 2.17 |
| SN | N.D. a | N.D. |
| SNE | 100.21 ± 1.19 | 78.15 ± 1.17 |
| SEN | N.D. | N.D. |
| ME | 104.91 ± 1.04 | 109 ± 2.44 |
| MN | N.D. | N.D. |
| MNE | 95.19 ± 1.62 | 90.75 ± 1.29 |
| MEN | N.D. | N.D. |
| D | 43.5 ± 1.02 | 147.6 ± 3.8 |
| I | 87.38 ± 1.73 | 118.5 ± 2.3 |
| Ascorbic acid b | 27.63 ± 1.12 | − |
| Extracts | Minimum Inhibitory Concentrations (MICs) (mg/ mL) | ||||||
|---|---|---|---|---|---|---|---|
| Escherichia coli | Klebsiella pneumonia | Pseudomonas aeruginosa | Bacillus subtilis | Bacillus cereus | Staphylococcus aureus | Staphylococcus epidermidis | |
| SE | 2 | 2 | N.I. a | 2 | 8 | 0.5 | 1 |
| SN | 1 | N.I. | 0.125 | 4 | N.I. | 2 | 0.25 |
| SNE | N.I. | N.I. | 8 | 2 | 4 | 2 | 2 |
| SEN | N.I. | N.I. | N.I. | N.I. | N.I. | N.I. | N.I. |
| ME | N.I. | 8 | N.I. | 2 | N.I. | 1 | 1 |
| MN | N.I. | N.I. | N.I. | N.I. | N.I. | N.I. | N.I. |
| MNE | 8 | N.I. | N.I. | 2 | N.I. | 8 | 8 |
| MEN | N.I. | N.I. | N.I. | N.I. | N.I. | N.I. | N.I. |
| D | 8 | 8 | 4 | 1 | 8 | 2 | 1 |
| I | N.I. | N.I. | 4 | 4 | 8 | 2 | 1 |
| Gentamicin | 0.00025 | 0.00025 | 0.00025 | 0.0005 | 0.0008 | 0.0005 | 0.0005 |
| Extract | The Amount of Luteolin-7-O-glucoside (mg/100 mg Extract) |
|---|---|
| Infusion (I) | 0.4050 ± 0.0013 |
| Decoction (D) | 0.4599 ± 0.0018 |
| Time (min) | Solution A | Solution B | Flow rate (mL/min) |
|---|---|---|---|
| 0–10 | 5%→35% | 95%→65% | 0.8 |
| 10–15 | 35%→40% | 65%→60% | 0.6 |
| 15–18 | 40% | 60% | 0.8 |
| 18–20 | 40%→42% | 60%→58% | 0.8 |
| 20–25 | 42%→100% | 58%→0% | 0.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ustuner, O.; Anlas, C.; Bakirel, T.; Ustun-Alkan, F.; Diren Sigirci, B.; Ak, S.; Akpulat, H.A.; Donmez, C.; Koca-Caliskan, U. In Vitro Evaluation of Antioxidant, Anti-Inflammatory, Antimicrobial and Wound Healing Potential of Thymus Sipyleus Boiss. Subsp. Rosulans (Borbas) Jalas. Molecules 2019, 24, 3353. https://doi.org/10.3390/molecules24183353
Ustuner O, Anlas C, Bakirel T, Ustun-Alkan F, Diren Sigirci B, Ak S, Akpulat HA, Donmez C, Koca-Caliskan U. In Vitro Evaluation of Antioxidant, Anti-Inflammatory, Antimicrobial and Wound Healing Potential of Thymus Sipyleus Boiss. Subsp. Rosulans (Borbas) Jalas. Molecules. 2019; 24(18):3353. https://doi.org/10.3390/molecules24183353
Chicago/Turabian StyleUstuner, Oya, Ceren Anlas, Tulay Bakirel, Fulya Ustun-Alkan, Belgi Diren Sigirci, Seyyal Ak, Huseyin Askin Akpulat, Ceylan Donmez, and Ufuk Koca-Caliskan. 2019. "In Vitro Evaluation of Antioxidant, Anti-Inflammatory, Antimicrobial and Wound Healing Potential of Thymus Sipyleus Boiss. Subsp. Rosulans (Borbas) Jalas" Molecules 24, no. 18: 3353. https://doi.org/10.3390/molecules24183353
APA StyleUstuner, O., Anlas, C., Bakirel, T., Ustun-Alkan, F., Diren Sigirci, B., Ak, S., Akpulat, H. A., Donmez, C., & Koca-Caliskan, U. (2019). In Vitro Evaluation of Antioxidant, Anti-Inflammatory, Antimicrobial and Wound Healing Potential of Thymus Sipyleus Boiss. Subsp. Rosulans (Borbas) Jalas. Molecules, 24(18), 3353. https://doi.org/10.3390/molecules24183353