Sabotage at the Powerhouse? Unraveling the Molecular Target of 2-Isopropylbenzaldehyde Thiosemicarbazone, a Specific Inhibitor of Aflatoxin Biosynthesis and Sclerotia Development in Aspergillus flavus, Using Yeast as a Model System
Abstract
1. Introduction
2. Results and Discussion
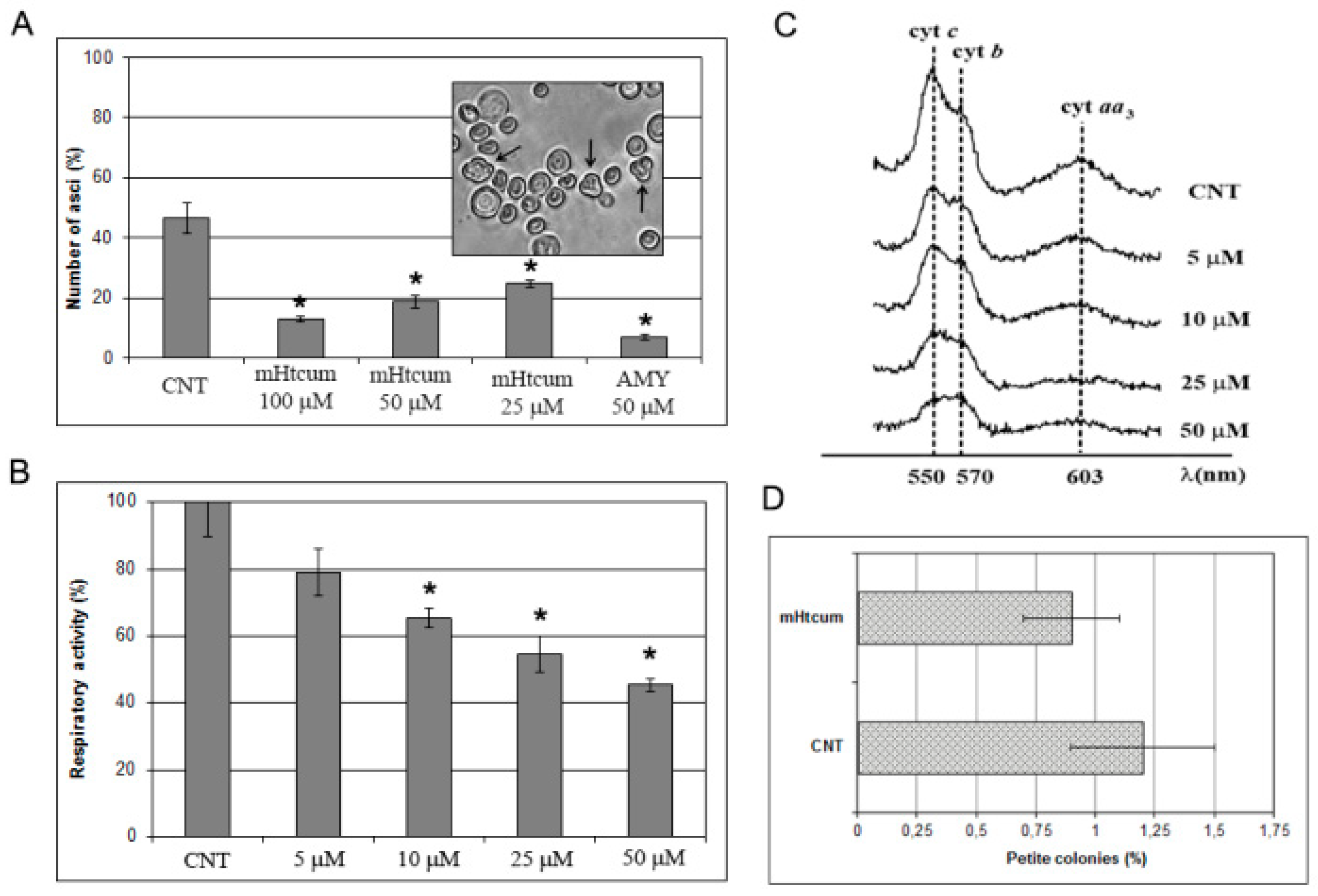
2.1. Effect of mHtcumon the Oxidative Carbon Source Utilization
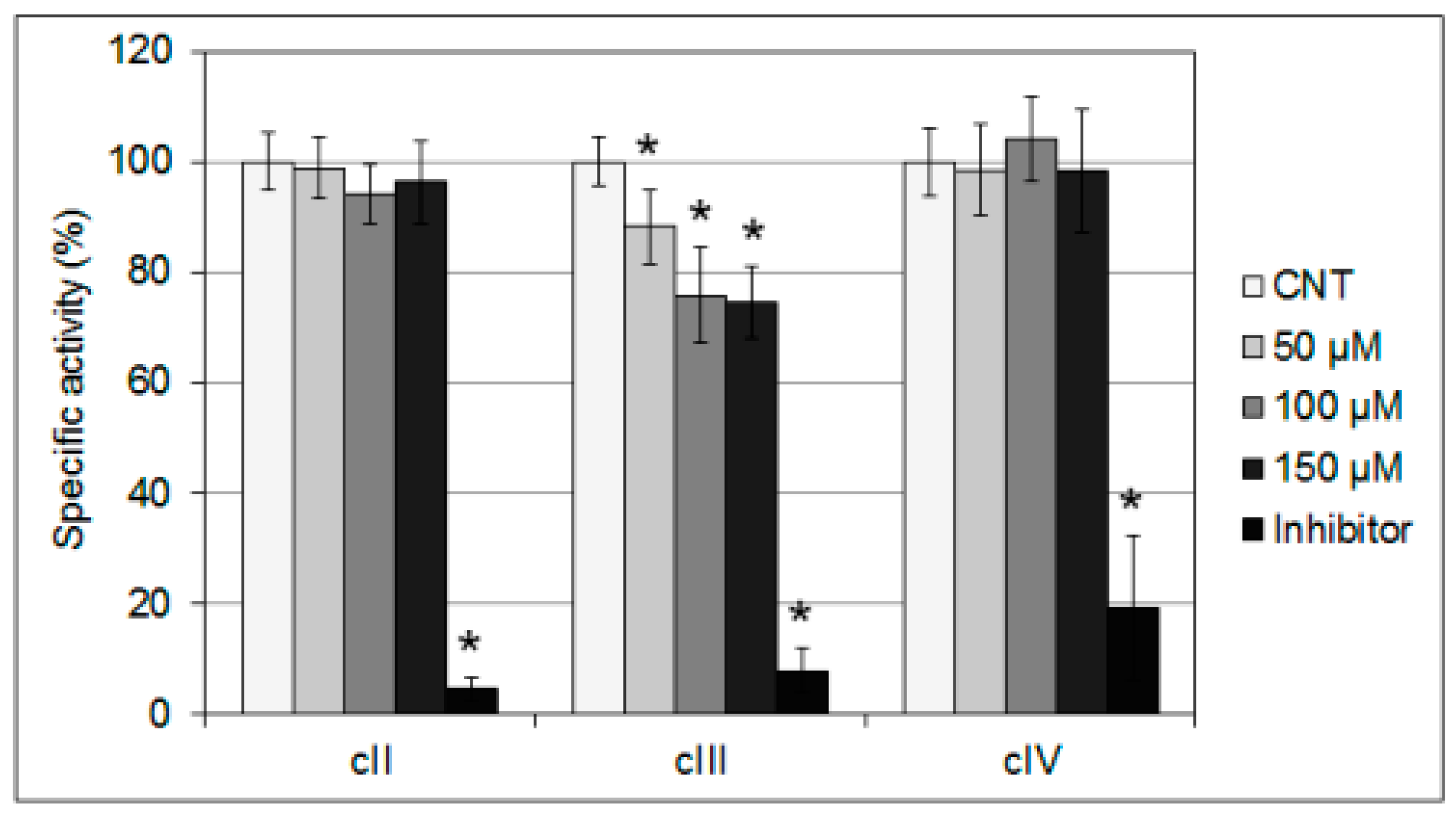
2.2. Interference of mHtcum on Mitochondrial Activity
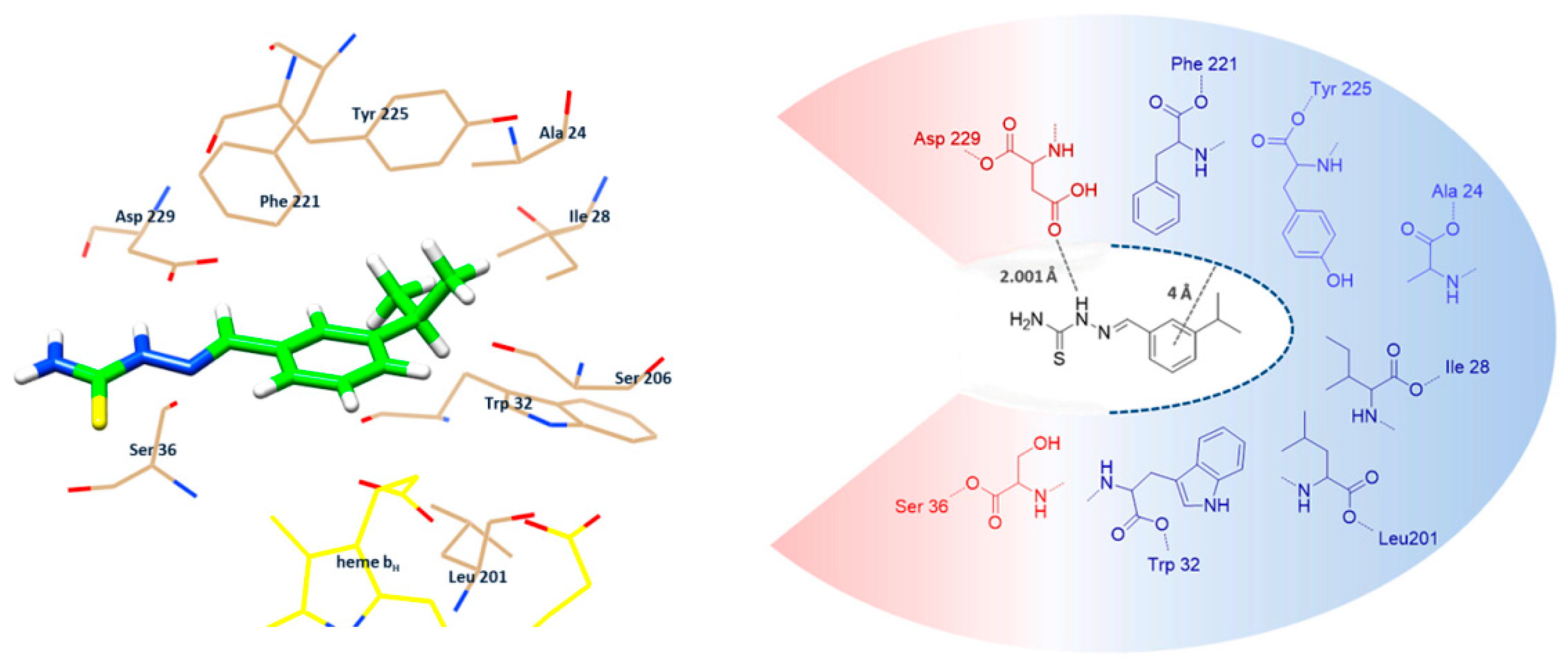
2.3. Comparative Molecular Docking Analysis
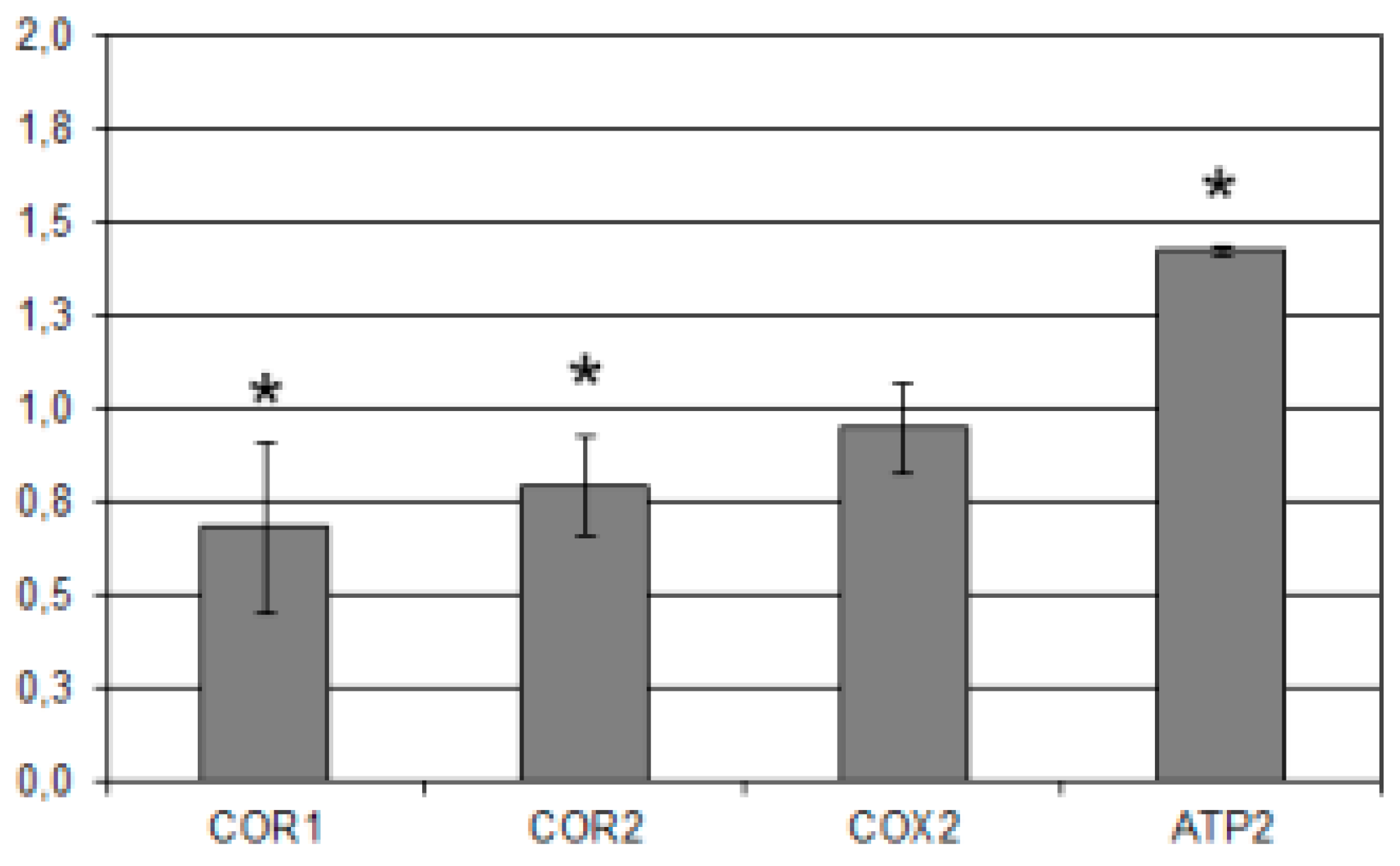
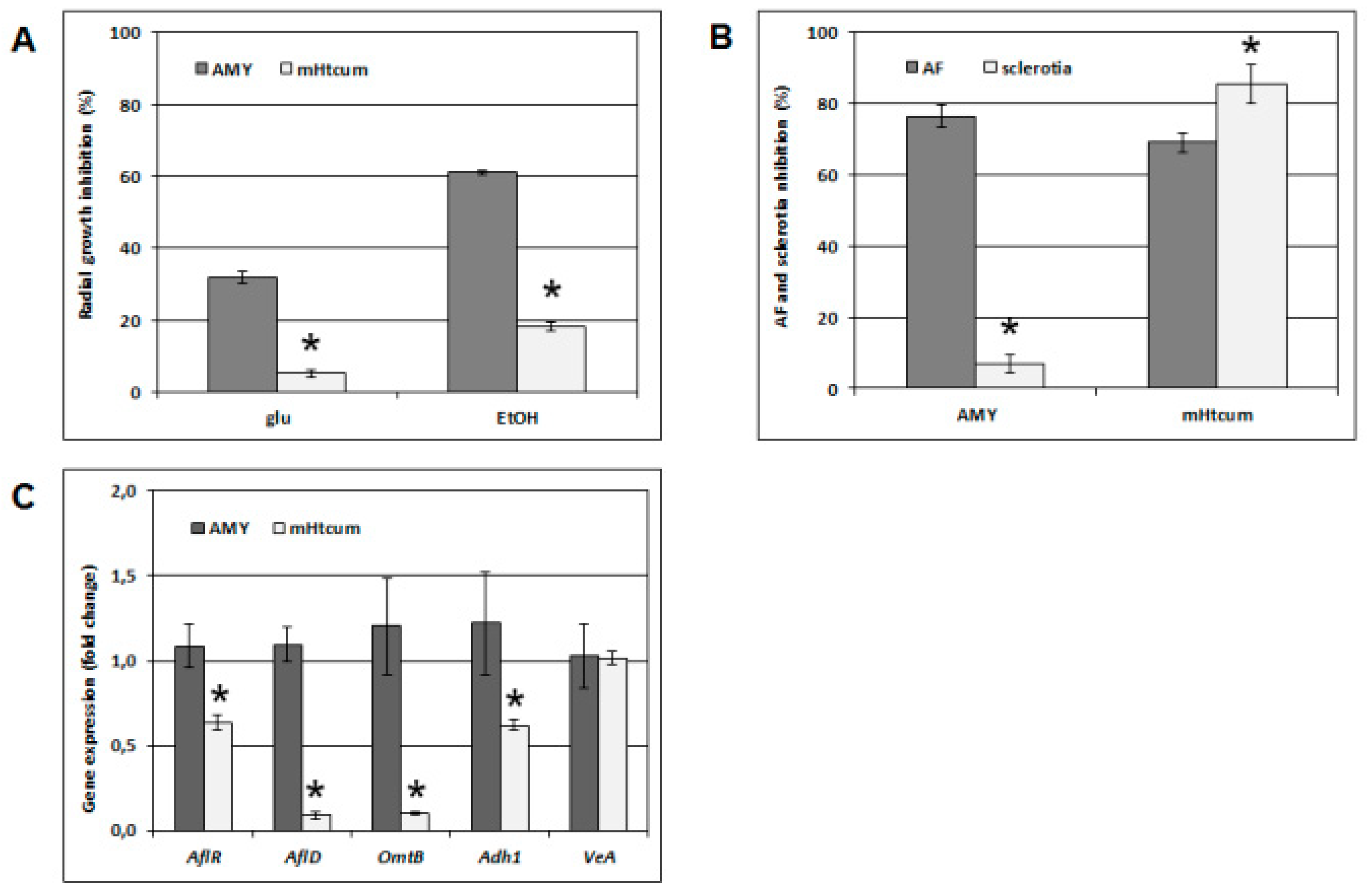
2.4. A. Flavus Growth, Development and Gene Expression
3. Materials and Methods
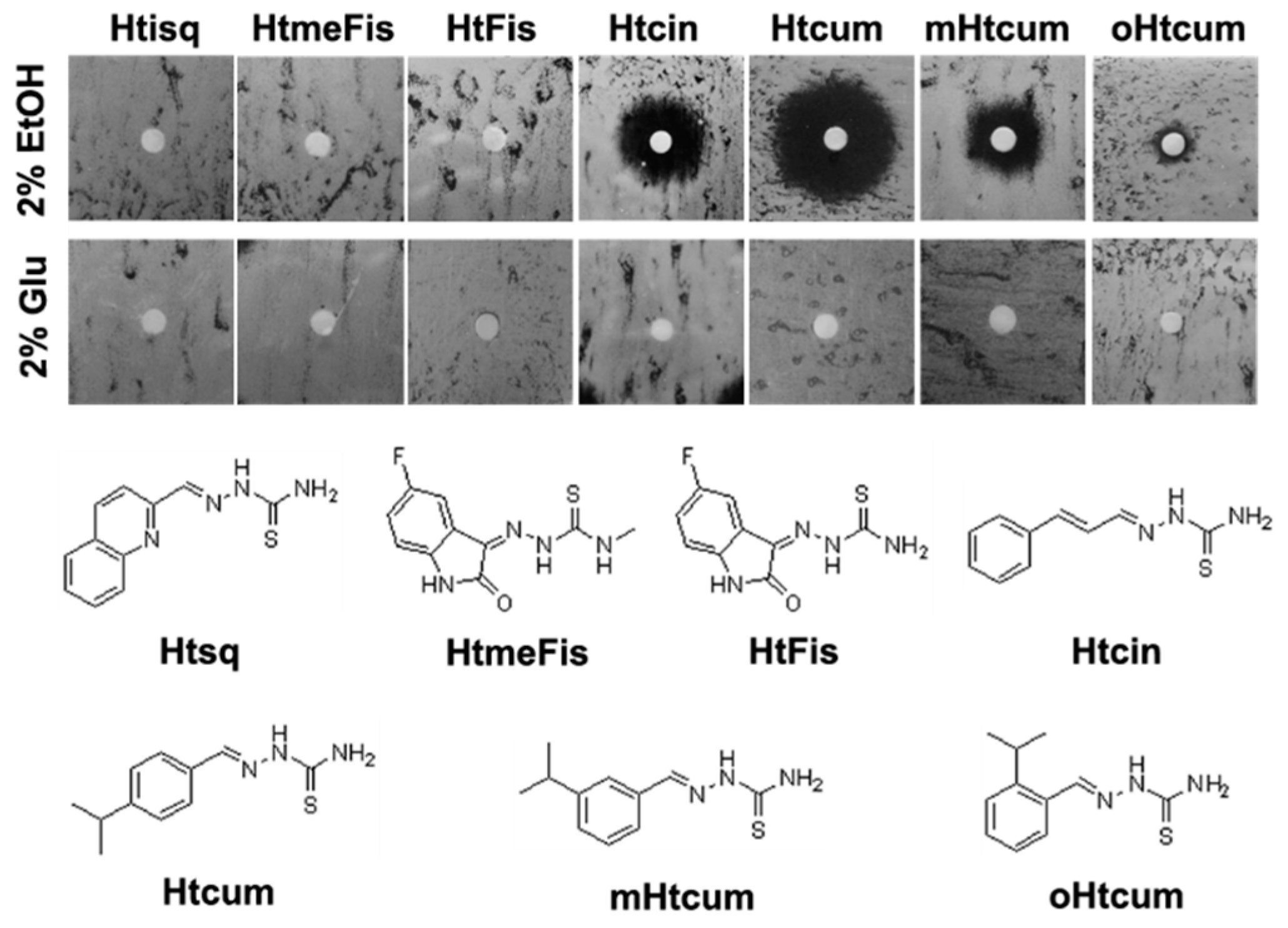
3.1. Thiosemicarbazones
3.2. Microbial Strains, Media and Culture Conditions
3.3. Agar Disk-Diffusion Testing
3.4. Spot Assay for Yeast Oxidative Growth
3.5. Asci Production in S. cerevisiae
3.6. Mitochondrial Respiratory Activity and Cytochrome Profiles
3.7. Mitochondrial DNA Stability
3.8. Isolation of Mitochondria, Gel Electrophoresis and Western Blot Analysis
3.9. Aflatoxin Production Assay
3.10. A. flavus Radial Growth Determination
3.11. Sclerotia Biogenesis Assay
3.12. RNA Extraction and Gene Expression Analysis
3.13. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, F.; Groopman, J.D.; Pestka, J.J. Public Health Impacts of Foodborne Mycotoxins. Annu. Rev. Food Sci. Technol. 2014, 5, 351–372. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.K.; Wilkinson, J.; Zablotowicz, R.; Accinelli, C.; Abel, C.; Bruns, H.; Weaver, M. Ecology of Aspergillus flavus, regulation of aflatoxin production, and management strategies to reduce aflatoxin contamination of corn. Toxin Rev. 2009, 28, 142–153. [Google Scholar] [CrossRef]
- Medina, A.; Rodriguez, A.; Magan, N. Effect of climate change on Aspergillus flavus and aflatoxin B1 production. Front. Microbiol. 2014, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Battilani, P.; Toscano, P.; Van Der Fels-Klerx, H.J.; Moretti, A.; Camardo Leggieri, M.; Brera, C.; Rortais, A.; Goumperis, T.; Robinson, T. Aflatoxin B 1 contamination in maize in Europe increases due to climate change. Sci. Rep. 2016, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, M.; Mori, T. Detection of Aflatoxins in Autopsied Materials from a Patient Infected with Aspergillus flavus. Jpn. J. Med. Mycol. 1998, 39, 167–171. [Google Scholar] [CrossRef]
- Yamada, K.; Mori, T.; Irie, S.; Matsumura, M.; Nakayama, M.; Hirano, T.; Suda, K.; Oshimi, K. Systemic aspergillosis caused by an aflatoxin-producing strain of Aspergillus in a post-bone marrow transplant patient with acute myeloid leukemia. Rinsho Ketsueki 1998, 39, 1103–1108. [Google Scholar] [PubMed]
- Klich, M.A. Environmental and developmental factors influencing aflatoxin production by Aspergillus flavus and Aspergillus parasiticus. Mycoscience 2007, 48, 71–80. [Google Scholar] [CrossRef]
- Leema, G.; Kaliamurthy, J.; Geraldine, P.; Thomas, P.A. Keratitis due to Aspergillus flavus: Clinical profile, molecular identification of fungal strains and detection of aflatoxin production. Mol. Vis. 2010, 16, 843–854. [Google Scholar] [PubMed]
- Nidhina, N.; Bhavya, M.L.; Bhaskar, N.; Muthukumar, S.P.; Murthy, P.S. Aflatoxin production by Aspergillus flavus in rumen liquor and its implications. Food Control 2017, 71, 26–31. [Google Scholar] [CrossRef]
- Jiang, Y.; Jolly, P.; Ellis, W.; Wang, J.; Phillips, T.; Williams, J. Aflatoxin B1 albumin adduct levels and cellular immune status in Ghanaians. Int. Immunol. 2005, 17, 807–814. [Google Scholar] [CrossRef]
- Sakuda, S.; Ono, M.; Ikeda, H. Blasticidin A as an Inhibitor of Aflatoxin Production by Aspergillus parasiticus. J. Antibiot. 2000, 53, 1265–1271. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kondo, T.; Sakurada, M.; Okamoto, S.; Ono, M.; Tsukigi, H.; Suzuki, A.; Nagasawa, H.; Sakuda, S. Effects of aflastatin A, an inhibitor of aflatoxin production, on aflatoxin biosynthetic pathway and glucose metabolism in Aspergillus parasiticus. J. Antibiot. 2001, 54, 650–657. [Google Scholar] [CrossRef] [PubMed]
- Roze, L.V.; Arthur, A.E.; Hong, S.Y.; Chanda, A.; Linz, J.E. The initiation and pattern of spread of histone H4 acetylation parallel the order of transcriptional activation of genes in the aflatoxin cluster. Mol. Microbiol. 2007, 66, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Nesci, A.; Ferrari, L.; Etcheverry, M. Effect of synthetic antioxidants on stored maize grain mycoflora in situ. J. Sci. Food Agric. 2008, 88, 797–804. [Google Scholar] [CrossRef]
- Holmes, R.; Boston, R.S.; Payne, G. Diverse inhibitors of aflatoxin biosynthesis. Appl. Microbiol. Biotechnol. 2008, 78, 559–572. [Google Scholar] [CrossRef] [PubMed]
- Roze, L.V.; Chanda, A.; Laivenieks, M.; Beaudry, R.M.; Artymovich, K.; Koptina, A.V.; Awad, D.W.; Valeeva, D.; Jones, A.D.; Linz, J.E. Volatile profiling reveals intracellular metabolic changes in Aspergillus parasiticus: veA regulates branched chain amino acid and ethanol metabolism. BMC Biochem. 2010, 11, 33. [Google Scholar] [CrossRef] [PubMed]
- Sakuda, S. Mycotoxin production inhibitors from natural products. Mycotoxins 2010, 60, 79–86. [Google Scholar] [CrossRef]
- Kim, J.H.; Chan, K.L.; Mahoney, N.; Campbell, B.C. Antifungal activity of redox-active benzaldehydes that target cellular antioxidation. Ann. Clin. Microbiol. Antimicrob. 2011, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- Cantrell, C.L.; Dayan, F.E.; Duke, S.O. Natural products as sources for new pesticides. J. Nat. Prod. 2012, 75, 1231–1242. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Campbell, B.C.; Chan, K.L.; Mahoney, N.; Haff, R.P. Synergism of antifungal activity between mitochondrial respiration inhibitors and kojic acid. Molecules 2013, 18, 1564–1581. [Google Scholar] [CrossRef] [PubMed]
- Roze, L.V.; Hong, S.-Y.; Linz, J.E. Aflatoxin biosynthesis: Current frontiers. Ann. Rev. Food Sci. Technol. 2013, 4, 293–311. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Mo, H.; Chen, Y.; Ding, D.; Hu, L. Inhibition of Aflatoxin Synthesis in Aspergillus flavus by Three Structurally Modified Lentinans. Int. J. Mol. Sci. 2014, 15, 3860–3870. [Google Scholar] [CrossRef] [PubMed]
- Sakuda, S.; Prabowo, D.F.; Takagi, K.; Shiomi, K.; Mori, M.; Omura, S.; Nagasawa, H. Inhibitory effects of respiration inhibitors on aflatoxin production. Toxins 2014, 6, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Xing, F.; Selvaraj, J.N.; Liu, X.; Wang, L.; Hua, H.; Zhou, L.; Zhao, Y.; Wang, Y.; Liu, Y. Inhibitory Effect of Cinnamaldehyde, Citral, and Eugenol on Aflatoxin Biosynthetic Gene Expression and Aflatoxin B1 Biosynthesis in Aspergillus flavus. J. Food Sci. 2015, 80, 2917–2924. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Yazawa, S.; Nakagawa, Y.; Sasaki, Y.; Yajima, S. Effects of alkyl parabens on plant pathogenic fungi. Bioorg. Med. Chem. Lett. 2015, 25, 1774–1777. [Google Scholar] [CrossRef]
- Jahanshiri, Z.; Shams-Ghahfarokhi, M.; Allameh, A.; Razzaghi-Abyaneh, M. Inhibitory effect of eugenol on aflatoxin B1 production in Aspergillus parasiticus by downregulating the expression of major genes in the toxin biosynthetic pathway. World J. Microbiol. Biotechnol. 2015, 31, 1071–1078. [Google Scholar] [CrossRef]
- Caceres, I.; Khoury, R.E.; Medina, Á.; Lippi, Y.; Naylies, C.; Atoui, A.; Khoury, A.E.; Oswald, I.; Bailly, J.-D.; Puel, O. Deciphering the Anti-Aflatoxinogenic Properties of Eugenol Using a Large-Scale q-PCR Approach. Toxins 2016, 8, 123. [Google Scholar] [CrossRef]
- Furukawa, T.; Iimura, K.; Kimura, T.; Yamamoto, T.; Sakuda, S. Inhibitory Activities of Alkyl Syringates and Related Compounds on Aflatoxin Production. Toxins 2016, 8, 177. [Google Scholar] [CrossRef]
- Sun, Q.; Shang, B.; Wang, L.; Lu, Z.; Liu, Y. Cinnamaldehyde inhibits fungal growth and aflatoxin B1 biosynthesis by modulating the oxidative stress response of Aspergillus flavus. Appl. Microbiol. Biotechnol. 2016, 100, 1355–1364. [Google Scholar] [CrossRef]
- Iimura, K.; Furukawa, T.; Yamamoto, T.; Negishi, L.; Suzuki, M.; Sakuda, S. The Mode of Action of Cyclo(l-Ala-l-Pro) in Inhibiting Aflatoxin Production of Aspergillus flavus. Toxins 2017, 9, 219. [Google Scholar] [CrossRef]
- Fountain, J.C.; Koh, J.; Yang, L.; Pandey, M.K.; Nayak, S.N.; Bajaj, P.; Zhuang, W.J.; Chen, Z.Y.; Kemerait, R.C.; Lee, R.D.; et al. Proteome analysis of Aspergillus flavus isolate-specific responses to oxidative stress in relationship to aflatoxin production capability. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Degola, F.; Morcia, C.; Bisceglie, F.; Mussi, F.; Tumino, G.; Ghizzoni, R.; Pelosi, G.; Terzi, V.; Buschini, A.; Restivo, F.M.; et al. In vitro evaluation of the activity of thiosemicarbazone derivatives against mycotoxigenic fungi affecting cereals. Int. J. Food Microbiol. 2015, 200, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Degola, F.; Bisceglie, F.; Pioli, M.; Palmano, S.; Elviri, L.; Pelosi, G.; Lodi, T.; Restivo, F.M. Structural modification of cuminaldehyde thiosemicarbazone increases inhibition specificity toward aflatoxin biosynthesis and sclerotia development in Aspergillus flavus. Appl. Microbiol. Biotechnol. 2017, 101, 6683–6696. [Google Scholar] [CrossRef] [PubMed]
- Rogolino, D.; Gatti, A.; Carcelli, M.; Pelosi, G.; Bisceglie, F.; Restivo, F.M.; Degola, F.; Buschini, A.; Montalbano, S.; Feretti, D.; et al. Thiosemicarbazone scaffold for the design of antifungal and antiaflatoxigenic agents: Evaluation of ligands and related copper complexes. Sci. Rep. 2017. [Google Scholar] [CrossRef] [PubMed]
- Weidberg, H.; Moretto, F.; Spedale, G.; Amon, A.; van Werven, F.J. Nutrient Control of Yeast Gametogenesis Is Mediated by TORC1, PKA and Energy Availability. PLoS Genet. 2016, 12, 1–26. [Google Scholar] [CrossRef]
- Puglisi, P.P.; Zennaro, E. Erythromycin inhibition of sporulation in Saccharomyces cerevisiae. Experientia 1971, 27, 963–964. [Google Scholar] [CrossRef]
- Alexandre, A.; Lehninger, A.L. Bypasses of the antimycin a block of mitochondrial electron transport in relation to ubisemiquinone function. Biochim. Biophys. Acta. 1984, 767, 120–129. [Google Scholar] [CrossRef]
- Balaban, R.S.; Nemoto, S.; Finkel, T. Mitochondria, oxidants, and aging. Cell 2005, 120, 483–495. [Google Scholar] [CrossRef]
- Campo, M.L.; Kinnally, K.W.; Tedeschi, H. The effect of antimycin A on mouse liver inner mitochondrial membrane channel activity. J. Biol. Chem. 1992, 267, 8123–8127. [Google Scholar]
- De Graaf, C.; Pospisil, P.; Pos, W.; Folkers, G.; Vermeulenet, N.P.E. Binding Mode Prediction of Cytochrome P450 and Thymidine Kinase Protein−Ligand Complexes by Consideration of Water and Rescoring in Automated Docking. J. Med. Chem. 2005, 48, 2308–2318. [Google Scholar] [CrossRef]
- Nissink, J.W.M.; Murray, C.; Hartshorn, M.; Verdonk, M.L.; Cole, J.C.; Taylor, R. A New Test Set for Validating Predictions of Protein–Ligand Interaction. Proteins 2002, 49, 457–471. [Google Scholar] [CrossRef]
- Kyte, J.; Doolittle, R.F. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 1982, 157, 105–132. [Google Scholar] [CrossRef]
- Verdonk, M.L.; Cole, J.C.; Hartshorn, M.J.; Murray, C.W.; Taylor, R.D. Improved protein-ligand docking using GOLD. Proteins 2003, 52, 609–623. [Google Scholar] [CrossRef] [PubMed]
- Eldridge, M.D.; Murray, C.W.; Auton, T.R.; Paolini, G.V.; Mee, R.P. Empirical scoring functions: I. The development of a fast empirical scoring function to estimate the binding affinity of ligands in receptor complexes. J. Comput.-Aided Mol. Des. 1997, 11, 425–445. [Google Scholar] [CrossRef] [PubMed]
- Degola, F.; Berni, E.; Restivo, F.M. Laboratory tests for assessing efficacy of atoxigenic Aspergillus flavus strains as biocontrol agents. Int. J. Food Microbiol. 2011, 146, 235–343. [Google Scholar] [CrossRef] [PubMed]
- Degola, F.; Dall’Asta, C.; Restivo, F.M. Development of a simple and high-throughput method for detecting aflatoxins production in culture media. Lett. Appl. Microbiol. 2012, 55, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Thomas, B.J.; Rothstein, R. The genetic control of direct-repeat recombination in Saccharomyces: The effect of rad52 and rad1 on mitotic recombination at GAL10, a transcriptionally regulated gene. Genetics 1989, 123, 725–738. [Google Scholar] [PubMed]
- Baruffini, E.; Ferrero, I.; Foury, F. In vivo analysis of mtDNA replication defects in yeast. Methods 2010, 51, 426–436. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of proteins utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Barrientos, A.; Fontanesi, F.; Diaz, F. Evaluation of the mitochondrial respiratory chain and oxidative phosphorylation system using polarography and spectrophotometric enzyme assays. Curr. Protoc. Hum. Genet. 2009. [Google Scholar] [CrossRef]
- Soto, I.C.; Fontanesi, F.; Valledor, M.; Horn, D.; Singh, R.; Barrientos, A. Synthesis of cytochrome c oxidase subunit 1 is translationally downregulated in the absence of functional F1F0-ATP synthase. Biochim. Biophys. Acta 2009, 1793, 1776–1786. [Google Scholar] [CrossRef] [PubMed]
- Roze, L.V.; Laivenieks, M.; Hong, S.-Y.; Wee, J.; Wong, S.S.; Vanos, B.; Awad, D.; Ehrlich, K.C.; Linz, J.E. Aflatoxin biosynthesis is a novel source of reactive oxygen species—A potential redox signal to initiate resistance to oxidative stress? Toxins 2015, 7, 1411–1430. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |

| Oligo Name | Sequence (5′3′) | Target Gene |
|---|---|---|
| tubFw-RT | TACCATGGACGCCGTCCG | Tub1 |
| tubRev-RT | GACGGACAACATCGACAAC | |
| OmtB F | GCCTTGACATGGAAACCATC | AflO |
| OmtBRev-RT | TCCACTGCTCAATCGCATG | |
| Adh1 FwRT | CTAAACCAGGACCAGATGAG | Adh1 |
| Adh1 RevRT | TCCCTCGTGTCCACCTAC | |
| Nor1 F | ACGGATCACTTAGCCAGCAC | AflD |
| AflD-RevRT | ACGGTGCTTTTGGGACGTTG | |
| AflJ-gF | GAACGCTGATTGCCAATGCC | AflS |
| AflS RevRT | GATTCATCCAGAGGGATAC | |
| VeA FwRT | CGAGACGGAAGCCTCCGT | VeA |
| VeA RevRT | TGGAGGATCGACTGGACGA |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dallabona, C.; Pioli, M.; Spadola, G.; Orsoni, N.; Bisceglie, F.; Lodi, T.; Pelosi, G.; Restivo, F.M.; Degola, F. Sabotage at the Powerhouse? Unraveling the Molecular Target of 2-Isopropylbenzaldehyde Thiosemicarbazone, a Specific Inhibitor of Aflatoxin Biosynthesis and Sclerotia Development in Aspergillus flavus, Using Yeast as a Model System. Molecules 2019, 24, 2971. https://doi.org/10.3390/molecules24162971
Dallabona C, Pioli M, Spadola G, Orsoni N, Bisceglie F, Lodi T, Pelosi G, Restivo FM, Degola F. Sabotage at the Powerhouse? Unraveling the Molecular Target of 2-Isopropylbenzaldehyde Thiosemicarbazone, a Specific Inhibitor of Aflatoxin Biosynthesis and Sclerotia Development in Aspergillus flavus, Using Yeast as a Model System. Molecules. 2019; 24(16):2971. https://doi.org/10.3390/molecules24162971
Chicago/Turabian StyleDallabona, Cristina, Marianna Pioli, Giorgio Spadola, Nicolò Orsoni, Franco Bisceglie, Tiziana Lodi, Giorgio Pelosi, Francesco Maria Restivo, and Francesca Degola. 2019. "Sabotage at the Powerhouse? Unraveling the Molecular Target of 2-Isopropylbenzaldehyde Thiosemicarbazone, a Specific Inhibitor of Aflatoxin Biosynthesis and Sclerotia Development in Aspergillus flavus, Using Yeast as a Model System" Molecules 24, no. 16: 2971. https://doi.org/10.3390/molecules24162971
APA StyleDallabona, C., Pioli, M., Spadola, G., Orsoni, N., Bisceglie, F., Lodi, T., Pelosi, G., Restivo, F. M., & Degola, F. (2019). Sabotage at the Powerhouse? Unraveling the Molecular Target of 2-Isopropylbenzaldehyde Thiosemicarbazone, a Specific Inhibitor of Aflatoxin Biosynthesis and Sclerotia Development in Aspergillus flavus, Using Yeast as a Model System. Molecules, 24(16), 2971. https://doi.org/10.3390/molecules24162971