Biological Characterization and Inhibition of Streptococcus pyogenes ZUH1 Causing Chronic Cystitis by Crocus sativus Methanol Extract, Bee Honey Alone or in Combination with Antibiotics: An In Vitro Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation and Characterization of the ZUH1 Strain
2.2. Molecular Identification of the ZUH1 Strain
2.3. Antibiotic Susceptibility Test
2.4. Preparation of C. sativus Extracts
2.5. Preparation of BH Solutions and CFG
2.6. Bioassay of Inhibitory Activity of Plant Extracts, BH and CFG
2.7. Minimum Inhibitory Concentration (MIC) of C. sativus Methanol Extract (CSME)
2.8. Anti-Streptococcal Activity of the Combination of Antibiotics and Either CSME or BH
2.9. Instrumental Analysis of CSME
2.10. Transmission Electron Microscopy (TEM) of S. pyogenes ZUH1
2.11. Ethical Approval
3. Results
3.1. Biochemical and Molecular Characterization of S. pyogenes ZUH1
3.2. Antibiotic Susceptibility of S. pyogenes
3.3. Inhibition of S. Pyogenes by C. Sativus Extracts, BH and CFG
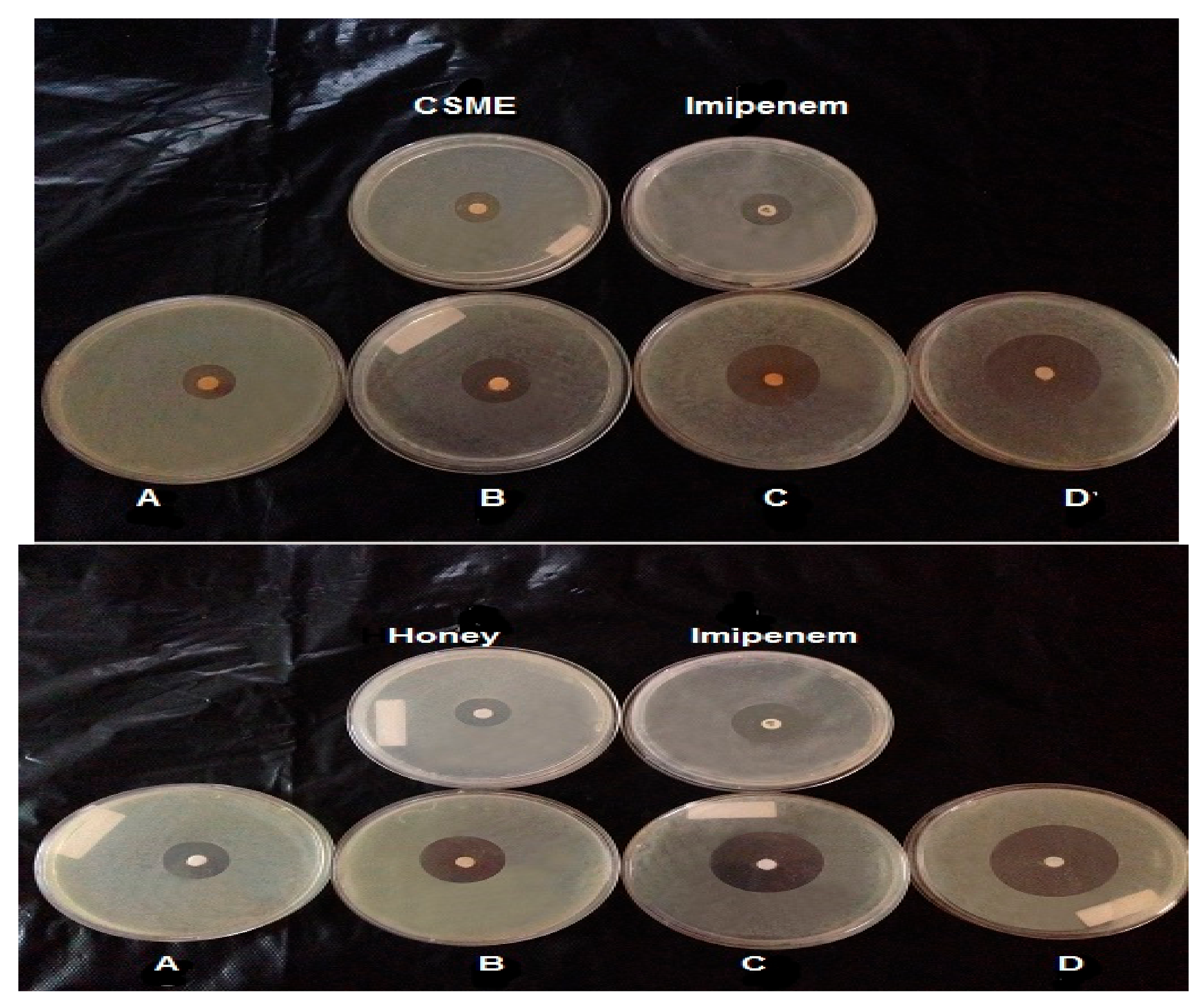
3.4. Anti-Streptococcal Effect of Combinations of Different Antibiotics and Either CSME or BH
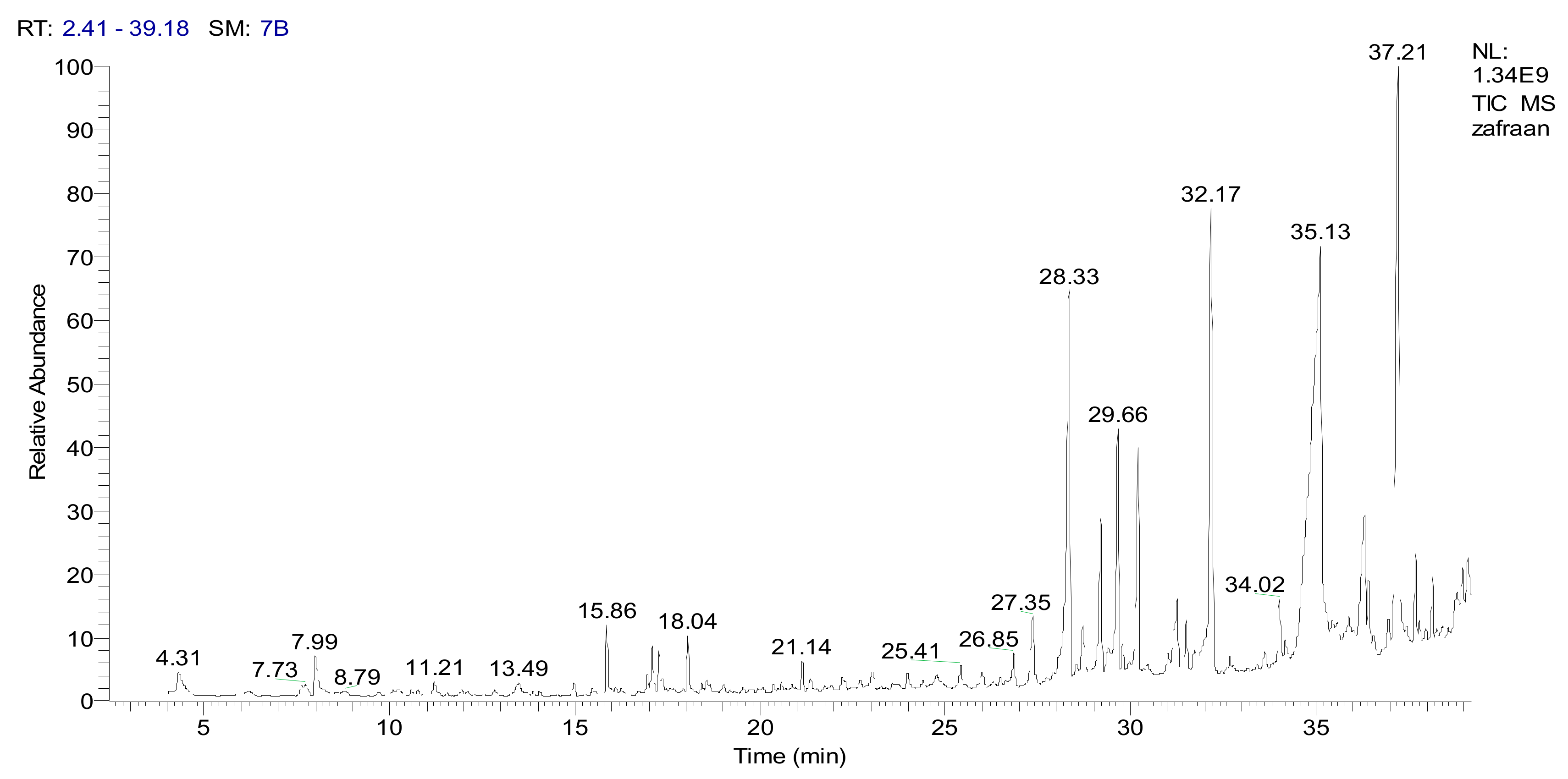
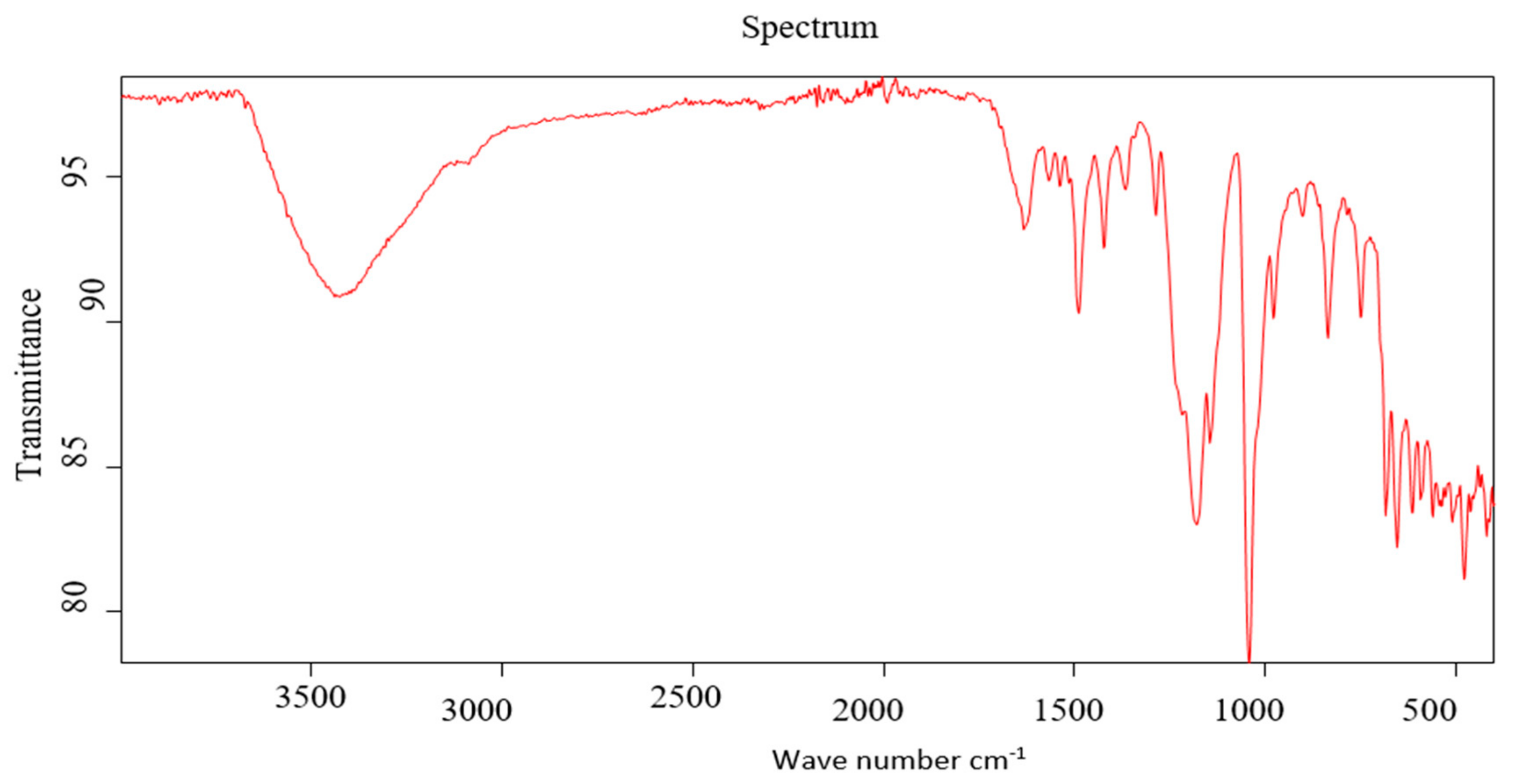
3.5. GC-MS and IR Analysis of CSME
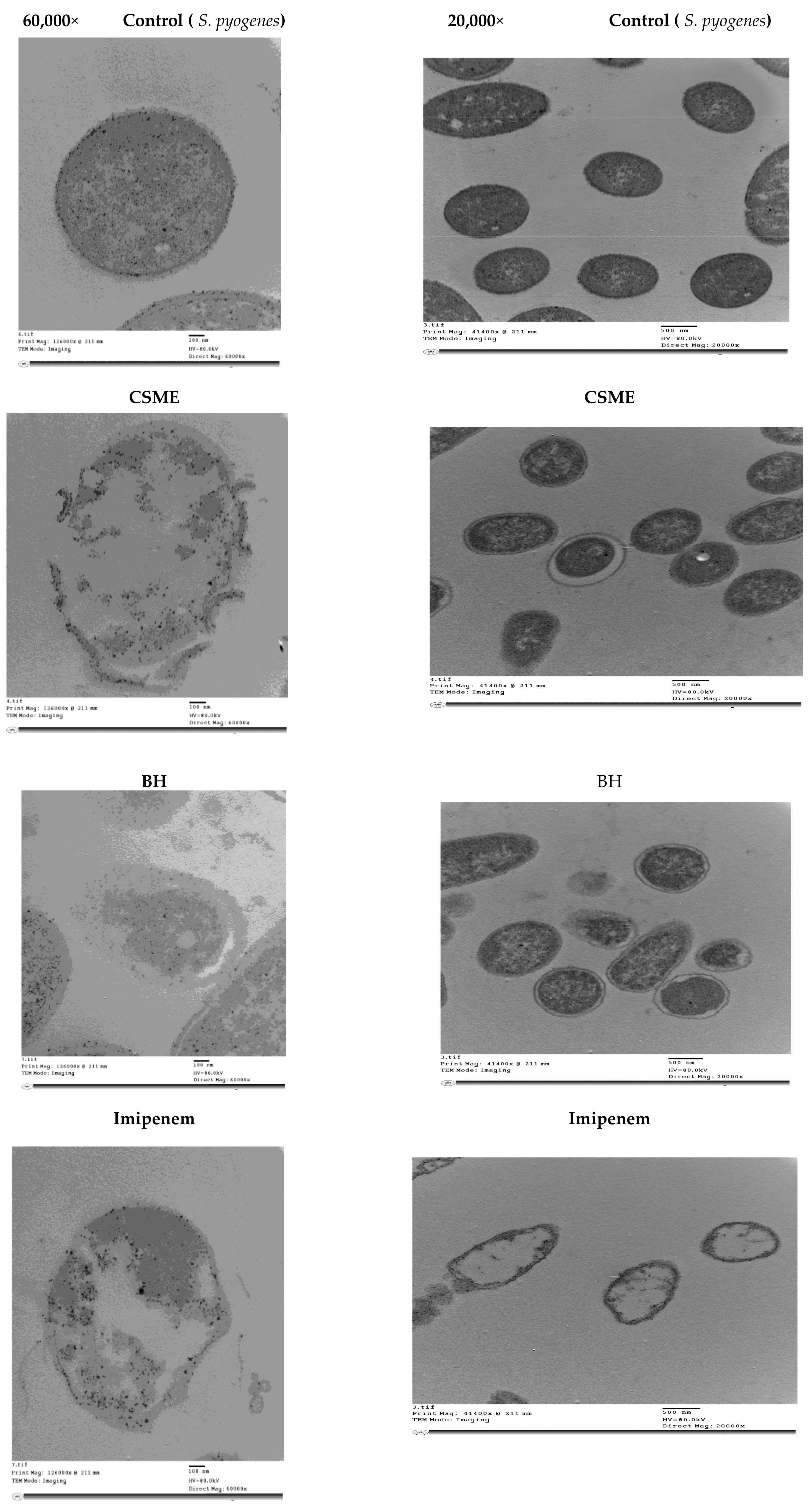
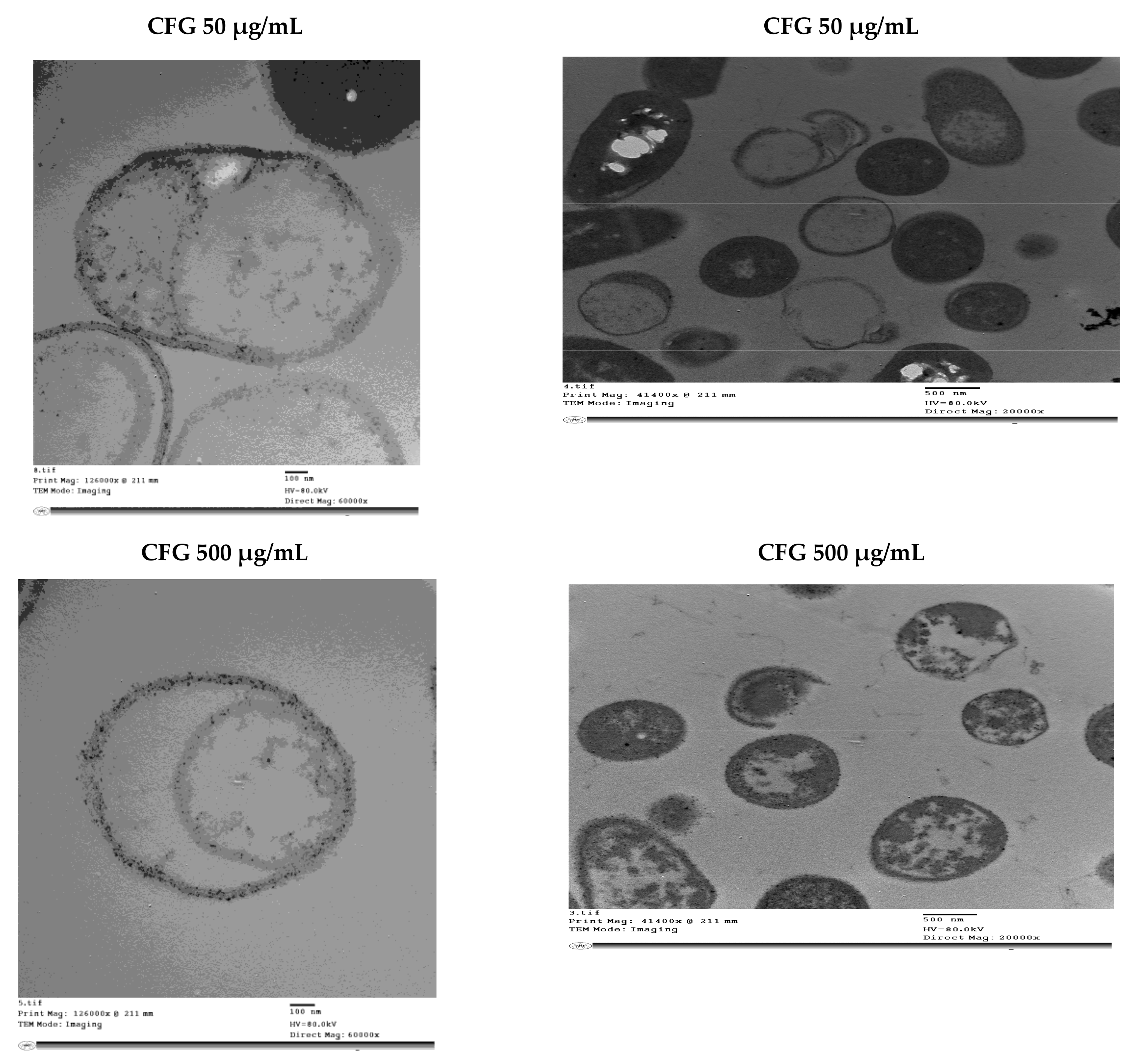
3.6. Transmission Electron Microscopy (TEM) Image Analysis of S. pyogenes Cells Treated with the Antibiotic Imipenem, CSME, BH and CFG
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gupta, P.D.; Birdi, T.J. Development of botanicals to combat antibiotic resistance. J. Ayurveda Integr. Med. 2017, 8, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Carapetis, J.R.; Steer, A.C.; Mulholland, E.K.; Weber, M. The global burden of group A streptococcal diseases. Lancet Infect. Dis. 2005, 5, 685–694. [Google Scholar] [CrossRef]
- Cunningham, M.W. Pathogenesis of group A streptococcal infections. Clin. Microbiol. Rev. 2000, 13, 470–511. [Google Scholar] [CrossRef] [PubMed]
- Borek, A.L.; Obszanska, K.; Hryniewicz, W.; Sitkiewicz, I. Detection of Streptococcus pyogenes virulence factors by multiplex PCR. Virulence 2012, 3, 1–5. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Osman, A.; Daidamony, G.; Sitohy, M.; Khalifa, M.; Enan, G. Soybean glycinin basic subunit inhibits methicillin resistant-vancomycin intermediate Staphylococcus aureus (MRSA-VISA) in vitro. Int. J. Appl. Res. Nat. Prod. 2016, 9, 17–26. [Google Scholar]
- Enan, G.; Hamdy, S.; Abdel-Shafi, S.; Al-Mohammadi, A.R. Biological characteristics and inhibition by both natural agents and antibiotics. Res. J. Med. Sci. 2016, 10, 573–586. [Google Scholar]
- Abdel-Shafi, S.; Al-Mohammadi, A.R.; Osman, A.; Enan, G.; Abdel-Hameid, S.; Sitohy, M. Characterization and Antibacterial Activity of 7S and11S Globulins Isolated from Cowpea Seed Protein. Molecules 2019, 24, 1082. [Google Scholar] [CrossRef]
- Subramani, R.; Narayanasamy, M.; Feussner, K.D. Plant-derived antimicrobials to fight against multi-drug-resistant human pathogens. 3 Biotech 2017, 7, 172. [Google Scholar] [CrossRef]
- Cheesman, M.J.; Ilanko, A.; Blonk, B.; Cock, I.E. Developing New Antimicrobial Therapies: Are Synergistic Combinations of Plant Extracts/Compounds with Conventional Antibiotics the Solution? Pharm. Rev. 2017, 11, 57–72. [Google Scholar] [CrossRef]
- Gislene, G.F.; Locatelli, N.J.; Paulo, C.F.; Giuliana, L.S. Antibacterial activity of plant extracts and phytochemicals on antibiotics-resistant bacteria. Braz. J. Microbiol. 2000, 31, 247–256. [Google Scholar]
- Abdel-Shafi, S.; Osman, A.; Enan, G.; El-Nemer, M.; Sitohy, M. Antibacterial activity of methylated egg white proteins against pathogenic G+ and G− bacteria matching antibiotics. Springerplus 2016, 5, 983. [Google Scholar] [CrossRef] [PubMed]
- Karimi, E.; Oskoueian, E.; Hendra, R.; Jaafar, H.Z. Evaluation of Crocus sativus L. stigma phenolic and flavonoid compounds and its antioxidant activity. Molecules 2010, 15, 6244–6256. [Google Scholar] [CrossRef] [PubMed]
- Asil, H. GC-MS analysis of volatile components of safranbolu and Kirikhan saffron (Crocus sativus L.) prepared by ultrasonic extraction. Fresenius Environ. Bull. 2018, 27, 9557–9563. [Google Scholar]
- Urbani, E.; Blasi, F.; Chiesi, C.; Maurizi, A.; Cossignani, L. Characterization of Volatile Fraction of Saffron from Central Italy (Cascia, Umbria). Int. J. Food Prop. 2015, 18, 2223–2230. [Google Scholar] [CrossRef]
- Okmen, G.; Kardas, S.; Bayrak, D.; Arslan, A.; Cakar, H. The antibacterial activities of Crocus sativus against mastitis pathogens and its antioxidant activities. World J. Pharm. Pharm. Sci. 2016, 5, 146–156. [Google Scholar]
- Farrell, D.; Qlobinson, M. Nutrient and blood agaricultural. J. Appl. Bacteriol. 1972, 25, 625–630. [Google Scholar] [CrossRef]
- George, C.K.; Willis, L.J. Comparison of the streptozyme test with the Antistreptolysin O, Antideoxyribonuclease B, And Antihyaluronidase Tests. Appl. Microbiol. 1970, 21, 227–259. [Google Scholar]
- Musantra, A. Use of lipase in the resolution of racemic ibuprofen. Appl. Microbiol. Biotechnol. 1992, 38, 61–66. [Google Scholar]
- Norris, J.R.; Ribbons, D.W. Methods in Microbiology; Academic Press: London, UK; New York, NY, USA, 1971; Volume 6, pp. 24–28. [Google Scholar]
- Pamela, A.S.; Dennis, E.O.; Barbara, E.I. A more Sensitive plate assay for detection of protease production by Pseudomonas aeruginosa. J. Clin. Microbiol. 1979, 9, 538–540. [Google Scholar]
- Williams, J.G.; Kubelik, A.R.; Livak, K.J.; Rafalski, J.A.; Tingey, S.V. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res. 1990, 18, 6531–6535. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twentieth Informational Upplement. CLSI Document M100-S20; Clinical and Laboratory Standards Institute (CLSI): Wayne, PA, USA, 2010. [Google Scholar]
- Alade, P.I.; Irobi, O.N. Antimicrobial activities of crude leaf extracts of Acalypha wilkesiana. J. Ethnopharmacol. 1993, 39, 171–174. [Google Scholar] [CrossRef]
- Moussa, A.; Noureddine, D.; Mohamed, H.S.; Abdelmelek, M.; Saad, A. Antibacterial activity of various honey types of Algeria against Staphylococcus aureus and Streptococcus pyogenes. Asian Pac. J. Trop. Med. 2012, 5, 773–776. [Google Scholar] [CrossRef]
- Ebran, N.; Julien, S.; Orange, N.; Auperin, B.; Molle, G. Isolation and characterization of novel glycoproteins from fish epidermal mucus: Correlation between their pore-forming properties and their antibacterial activities. Biochim. Biophys. Acta 2000, 1467, 271–280. [Google Scholar] [CrossRef]
- Saeed, S.; Naim, A.; Tariq, B. A study on prevalence of multidrug resistant Gram negative bacteria. Int. J. Biol. Bio. 2007, 4, 71–74. [Google Scholar]
- Ralph, A.P.; Carapetis, J.R. Group a streptococcal diseases and their global burden. Curr. Top. Microbiol. Immunol. 2013, 368, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G.F.; Karen, K.F. The Streptococci in: Jawetz, Melnick and Adelberg Medical Microbiology, 25th ed.; Brooks, G.F., Karen, K.C., Butel, J.S., Morse, S.A., Mietzner, T.A., Eds.; McGraw Hill: New York, NY, USA, 2010; Volume 14, pp. 198–200. [Google Scholar]
- Kenneth, J.R.; Lawrence, W.D. The Streptococci and Enterococci. Sherris MEDICAL Microbiology, 5th ed.; Kenneth, J.R., George, C.R., Sherrisch, J.C., Ahmed, N., Lawrence, W.D., Plorde, J.J., Eds.; McGraw-Hill Companies: New York, NY, USA, 2010; Volume 25, pp. 443–469. [Google Scholar]
- Holt, J.G.; Krieg, N.R.; Sneath, P.H.A.; Staley, J.T.; Williams, T.; Hensyl, W.R. Bergey’s Manual of Systematic Bacteriology, 9th ed.; Williams Wilkins: Baltimore, MS, USA, 1994. [Google Scholar]
- Suvorov, A.N.; Ferretti, J.J. Physical and genetic chromosomal map of an M type 1 strain of Streptococcus pyogenes. J. Bacteriol. 1996, 178, 5546–5549. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Golińska, E.; van der Linden, M.; Więcek, G.; Mikołajczyk, D.; Machul, A.; Samet, A.; Piórkowska, A.; Dorycka, M.; Heczko, P.B.; Strus, M. Virulence factors of Streptococcus pyogenes strains from women in peri-labor with invasive infections. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 747–754. [Google Scholar] [CrossRef]
- Kim, K.S.; Kaplan, E.L. Association of penicillin tolerance with failure to eradicate group A streptococci from patients with pharyngities. J. Pediatr. 1985, 107, 681–684. [Google Scholar] [CrossRef]
- Ikebe, T.; Hirasawa, K.; Suzuki RIsobe, J.; Tanaka, D.; Katsukawa, C.; Kawahara, R.; Tomita, M.; Ogata, K.; Endoh, M.; Okuno, R.; et al. Antimicrobial susceptibility survey of Streptococcus pyogenes isolated in Japan from patients with severe invasive group A Streptococcal infections. Antimicrob. Agents Chemother. 2005, 49, 788–790. [Google Scholar] [CrossRef]
- Dmitriev, A.V.; Chaussee, M.S. The Streptococcus pyogenes proteome: Maps, virulence factors and vaccine candidates. Future Microbiol. 2010, 5, 1539–1551. [Google Scholar] [CrossRef]
- Camara, M.; Dieng, A.; Boye, C.S. Antibiotic susceptibility of streptococcus pyogenes isolated from respiratory tract infections in dakar, senegal. Microbiol. Insights 2013, 6, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.; Sekuloski, S.; Batzloff, M.R. Novel strategies for controlling Streptococcus pyogenes infection and associated diseases: From potential peptide vaccines to antibody immunotherapy. Immunol. Cell Biol. 2009, 87, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Dubey, D.; Sahu, M.C.; Rath, S.; Paty, B.P.; Debata, N.K.; Padhy, R.N. Antimicrobial activity of medicinal plants used by aborigines of Kalahandi, Orissa, India against multidrug resistant bacteria. Asian Pac. J. Trop. Bio. 2012, 8, 800–807. [Google Scholar] [CrossRef]
- Hassanein, S.M.; Gebreel, H.M.; Hassan, A.A. Honey compared with some antibiotics against bacteria isolated from burn-wound infections of patients in Ain Shams University Hospital. J. Am. Sci. 2010, 6, 301–320. [Google Scholar]
- Enan, G.; Abdel-Shafi, S.; Ouda, S.; Negm, S. Characterization of probiotic lactic acid bacteria to be used as starter culture and protective cultures for diary fermentations. Int. J. Probiotics Prebiotics 2013, 8, 157–163. [Google Scholar]
- Kiran, I.; Ozşen, O.; Celik, T.; Ilhan, S.; Gürsu, B.Y.; Demirci, F. Microbial transformations of isophorone by Alternaria alternata and Neurospora crassa. Nat. Prod. Commun. 2013, 8, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Patra, J.K.; Das, G.; Baek, K.H. Chemical Composition and Antioxidant and Antibacterial Activities of an esential Oil Extracted from an Edible Seaweed, Laminaria japonica L. Molecules 2015, 20, 12093–12113. [Google Scholar] [CrossRef]
- Ramos-Nino, M.E.; Ramirez-Rodriguez, C.A.; Clifford, M.N.; Adams, M.R. QSARs for the effect of benzaldehydes on foodborne bacteria and the role of sulfhydryl groups as targets of their antibacterial activity. J. Appl. Microbiol. 1998, 24, 207–212. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
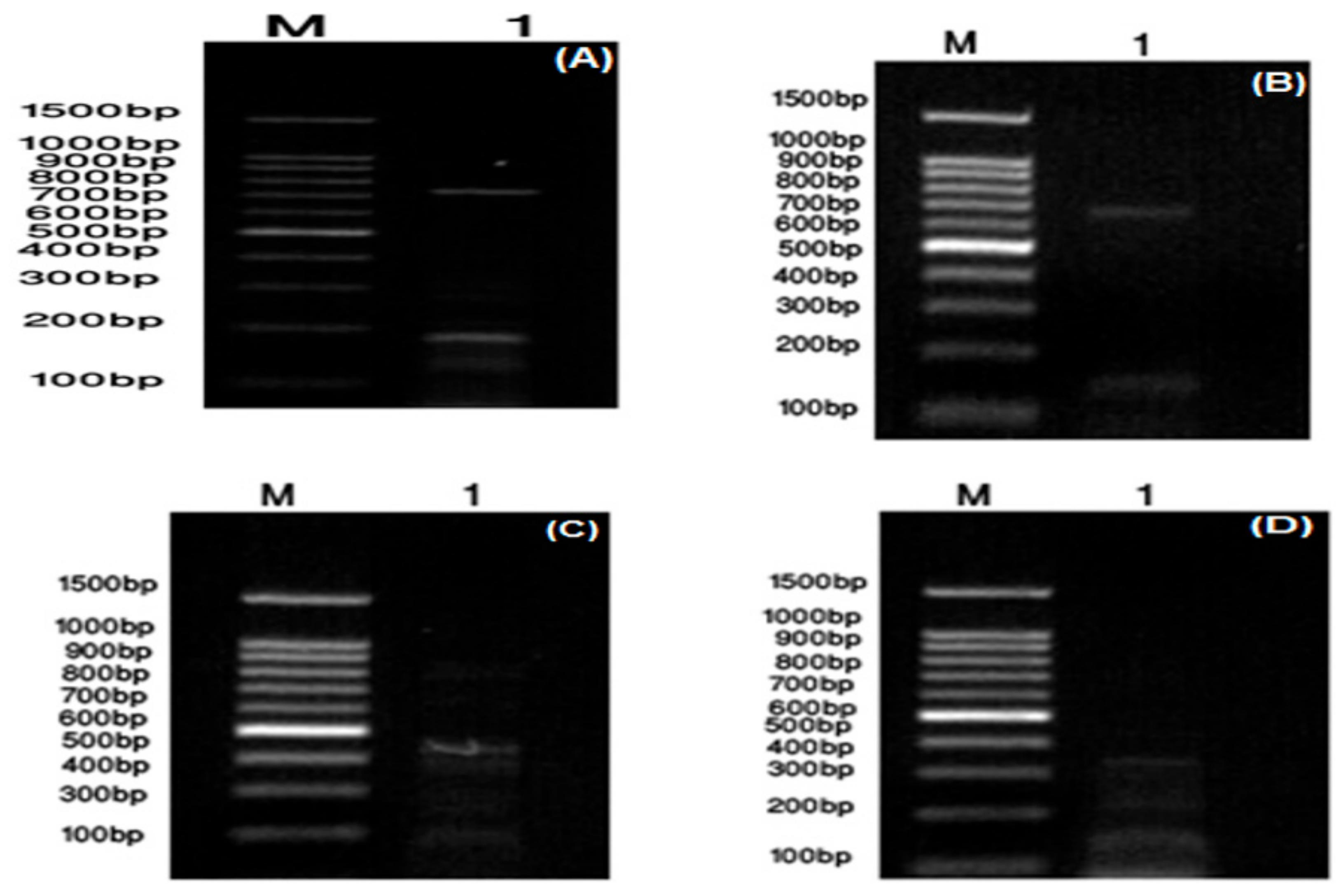
| Detected Virulence Factors | Primer Sequence (Forward) | Primer Sequence (Reverse) | Size of the PCR Product (bp) |
|---|---|---|---|
| spyCEP | GATCCGGCCCATCAAAGCAT | AGCTGCCACTGATGTTGGTG | 786 |
| ssa | AAGAATACTCGTTGTAGCATGTGT | AATATTGCTCCAGGTGCGGG | 678 |
| sic | TTACGTTGCTGATGGTGTATATGGT | TTTGATAGAGGGTTTTCAGCTGGC | 150 |
| sdaB | TATAGCGCATGCCGCCTTTT | TGATGGCGCAAGCAAGTACC | 440 |
| speG | TGGAAGTCAATTAGCTTATGCAG | GCGAACAACCTCAGAGGGCAAA | 384 |
| Types of Antibiotic | IZD (mm) |
|---|---|
| Tetracycline (30 μg/mL) | 16.00 e ± 1.5 (S) |
| Piperacillin (100 μg/mL) | 7.00 g ± 0.33 (I) |
| Clindamycin (2 μg/mL) | 10.00 f ± 0.25 (I) |
| Penicillin G (10 μg/mL) | 19.00 d ± 0.41 (S) |
| Ceftazidime (30 μg/mL) | 25.00 b ± 0.11 (S) |
| Cefoxitin (30 μg/mL) | 24.00 b ± 0.65 (S) |
| Cefoperazone (75 μg/mL) | 24.00 b ± 0.13 (S) |
| Imipenem (10 μg/mL) | 36.00 a ± 0.15 (S) |
| Amikacin (30 μg/mL) | 21.00 c ± 0.26 (S) |
| Ampicillin (10 μg/mL) | R |
| Aztreonam (10 μg/mL) | R |
| Norfloxacin (10 μg/mL) | R |
| Concentrations of CSME (µg/mL) | 0 | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 |
|---|---|---|---|---|---|---|---|---|---|---|
| IZD (mm) | 0.00 g | 0.00 g | 5.00 f ± 0.25 | 10.00 e ± 0.22 | 12.00 e ± 0.15 | 15.00 d ± 0.16 | 18.00 c ± 0.45 | 24.00 b ± 0.36 | 26.00 b ± 0.75 | 32.00 a ± 0.11 |
| Concentrations of BH (g/100 mL) | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | 100 |
| IZD (mm) | 0.00 f | 8.00 e ±0.35 | 15.00 d ± 0.25 | 18.00 d ± 0.29 | 25.00 c ± 0.71 | 26.00 c ± 0.11 | 28.00 bc ± 0.23 | 30.00 b ± 0.43 | 36.00 a ±0.13 | 38.00 a ± 0.43 |
| Concentrations of CFG (µg/mL) | 0 | 10 | 25 | 50 | 100 | 250 | 500 | |||
| IZD (mm) | 0.00 d | 0.00 d | 0.00 d | 19.00 c ±0.13 | 24.00 b ±0.45 | 30.00 a ±0.34 | 32.00 a ± 0.65 | |||
| Antibiotic | IZD of Antibiotics Only | IZD of Antibiotics with BH * | IZD of Antibiotic with CSME ** |
|---|---|---|---|
| Ceftazidime (30 μg/mL) | 26.00 b ± 0.25 | 35.00 c ± 0.11 | 30.00 c ± 0.16 |
| Cefoperazone (75 μg/mL) | 25.00 b ± 0.23 | 34.00 c ± 0.35 | 32.00 b ± 0.76 |
| Cefoxitin (30 μg/mL) | 25.00 b ± 0.34 | 38.00 b ± 0.54 | 33.00 b ± 0.24 |
| Imipenem (10 μg/mL) | 34.00 a ± 0.23 | 42.00 a ± 0.51 | 36.00 a ± 0.55 |
| No. | Compounds Name | M.W. | Molecular Formula | M.+ Parent Ion | Area | Base Peak | Classification |
|---|---|---|---|---|---|---|---|
| 1 | Isophorone | 138.0 | C9H14O | 138.0 | 2.50 | 29.0 | Cyclic ketone |
| 2 | 3-Hydroxy-7,8-dihydro-á-1-ionol | 208.0 | C13H20O2 | 209.0 (M+1) | 1.24 | 43.0 | Alcohol |
| 3 | 2-Naphthalenemethanol, decahydro-à,à,4a-trimethyl-8-methylene | 222.0 | C15H26O | 222.0 | 4.25 | 59.0 | Alcohol |
| 4 | Phenyl acetate | 338 | C22H42O | 338.0 | 4.25 | 43.0 | Ester |
| 5 | Tetradecanoic acid | 228.0 | C14H28O2 | 228.0 | 6.65 | 73.0 | Acid |
| 6 | 1,3-Cyclohexadiene-1-carboxaldehyde, 2,6,6-trimethyl-(Safranal) | 150 | C10H14O | 150 | 23.12 | 39.0 | Aldehyde |
| 7 | 6-Hydroxy-4,4,7a-trimethyl-5,6,7,7a-tetrahydrobenzofuran-2 (4H)-one | 196.0 | C11H16O3 | 196.0 | 5.12 | 111.0&43.0 | Lactone (cyclic esters) |
| 8 | 2,5,7,8-Tetramethyl-2-(4,8,12-Trimethyltridecyl)-6-chromanol | 430.0 | C29H50O2 | 430.0 | 3.12 | 165.0 | Natural product (cyclic ether) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdel-Shafi, S.; Al-Mohammadi, A.-R.; Hamdi, S.; Moustafa, A.H.; Enan, G. Biological Characterization and Inhibition of Streptococcus pyogenes ZUH1 Causing Chronic Cystitis by Crocus sativus Methanol Extract, Bee Honey Alone or in Combination with Antibiotics: An In Vitro Study. Molecules 2019, 24, 2903. https://doi.org/10.3390/molecules24162903
Abdel-Shafi S, Al-Mohammadi A-R, Hamdi S, Moustafa AH, Enan G. Biological Characterization and Inhibition of Streptococcus pyogenes ZUH1 Causing Chronic Cystitis by Crocus sativus Methanol Extract, Bee Honey Alone or in Combination with Antibiotics: An In Vitro Study. Molecules. 2019; 24(16):2903. https://doi.org/10.3390/molecules24162903
Chicago/Turabian StyleAbdel-Shafi, Seham, Abdul-Raouf Al-Mohammadi, Sara Hamdi, Ahmed H. Moustafa, and Gamal Enan. 2019. "Biological Characterization and Inhibition of Streptococcus pyogenes ZUH1 Causing Chronic Cystitis by Crocus sativus Methanol Extract, Bee Honey Alone or in Combination with Antibiotics: An In Vitro Study" Molecules 24, no. 16: 2903. https://doi.org/10.3390/molecules24162903
APA StyleAbdel-Shafi, S., Al-Mohammadi, A.-R., Hamdi, S., Moustafa, A. H., & Enan, G. (2019). Biological Characterization and Inhibition of Streptococcus pyogenes ZUH1 Causing Chronic Cystitis by Crocus sativus Methanol Extract, Bee Honey Alone or in Combination with Antibiotics: An In Vitro Study. Molecules, 24(16), 2903. https://doi.org/10.3390/molecules24162903







