Flavonoids from Brazilian Cerrado: Biosynthesis, Chemical and Biological Profile
Abstract
1. Introduction
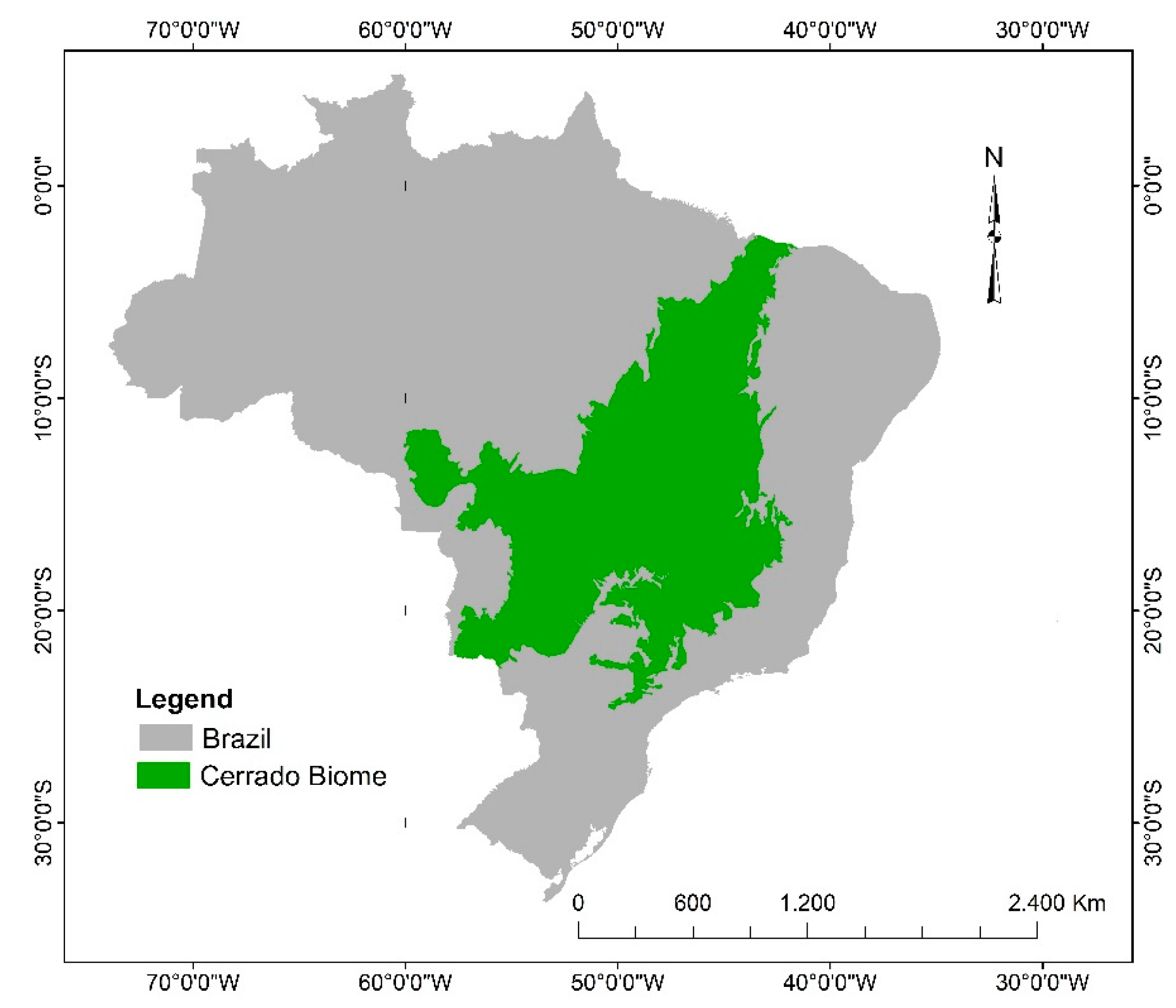
2. Characterization and Plant Diversity of the Brazilian Cerrado
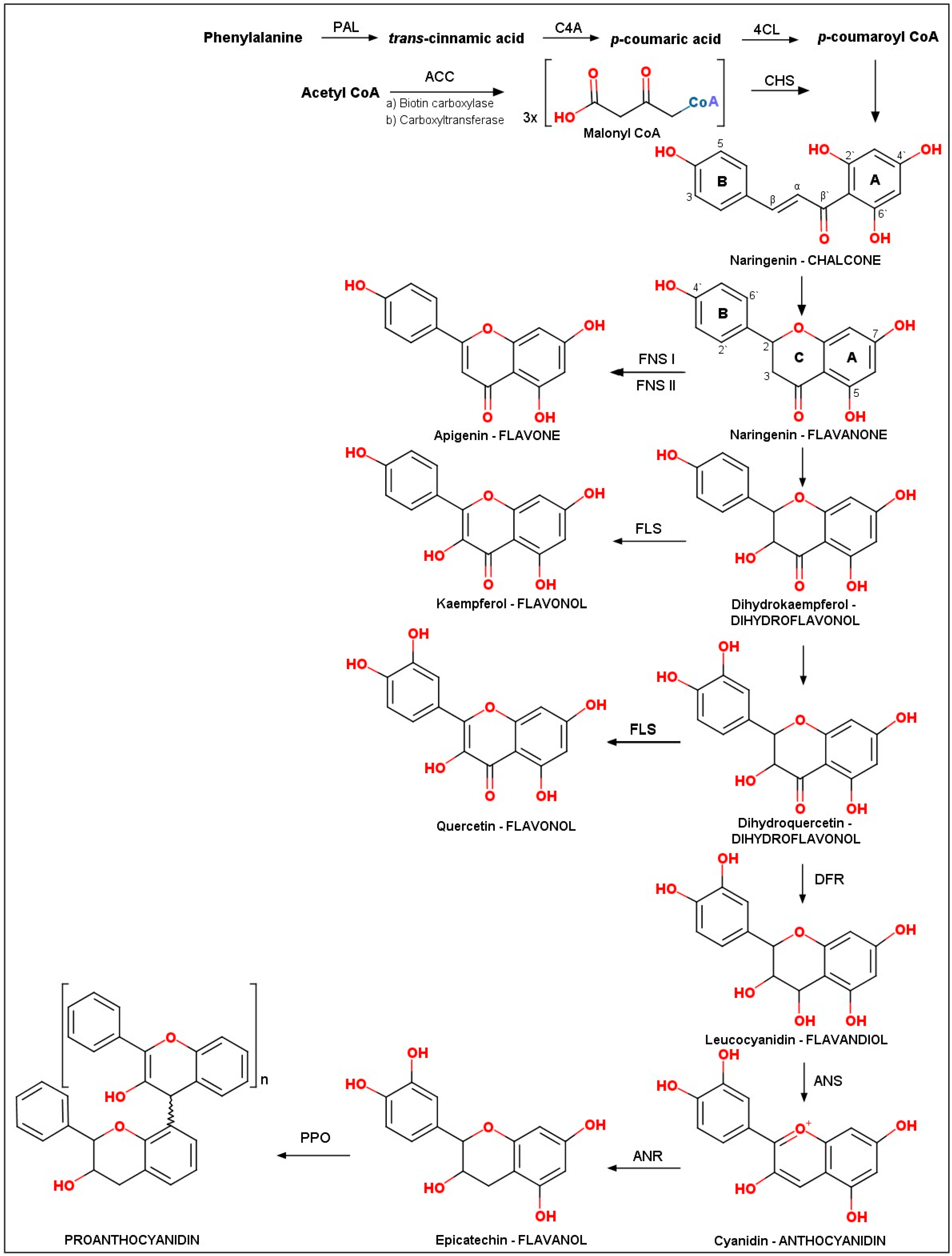
2.1. Biosynthesis of Flavonoids
2.2. Chemical Profile of Flavonoids
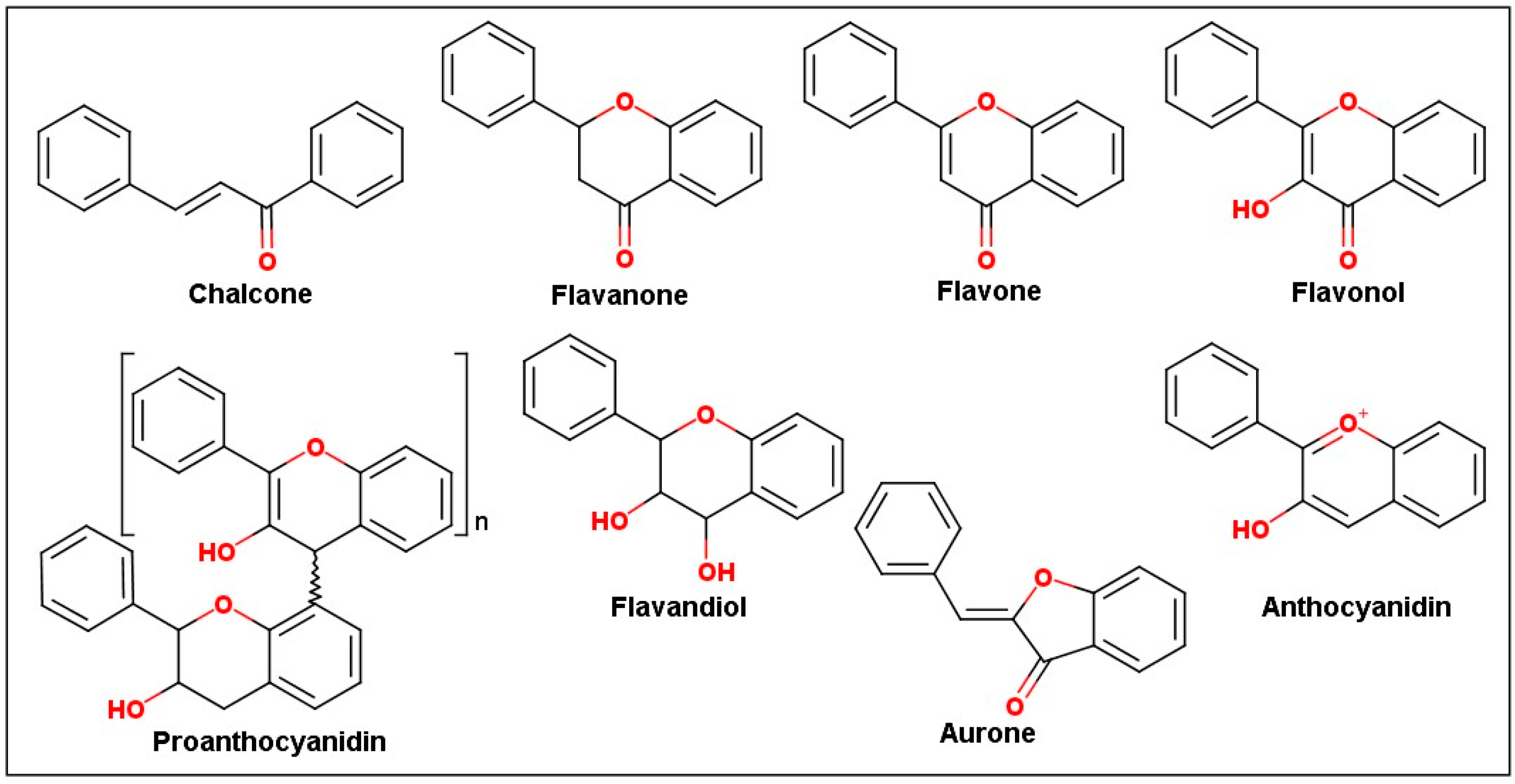
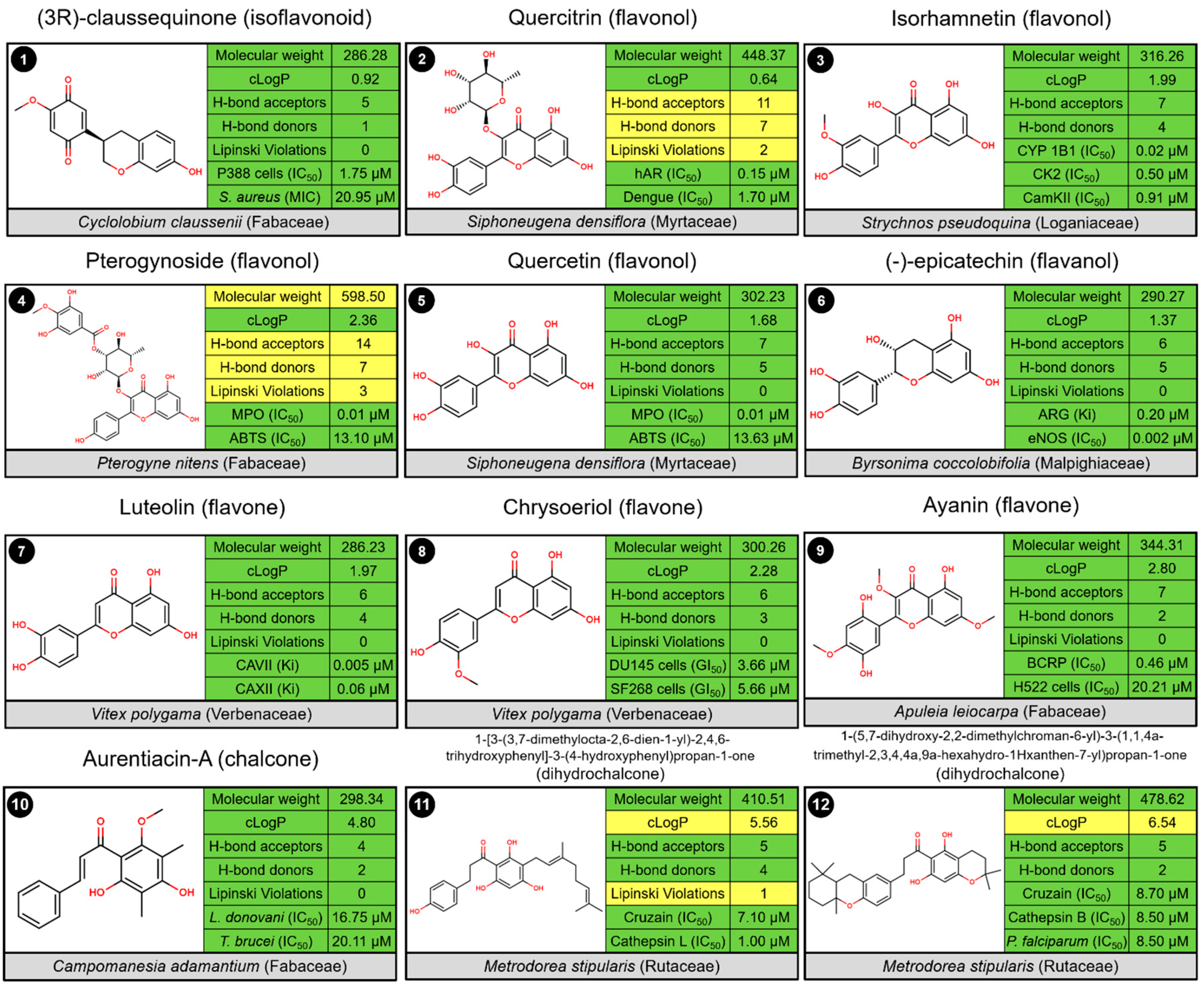
2.3. Biological Profile of Isolated Flavonoid
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Silva, J.F.; Farinas, M.R.; Felfili, J.M.; Klink, C.A. Spatial heterogeneity, land use and conservation in the Cerrado region of Brazil. J. Biogeogr. 2006, 33, 536–548. [Google Scholar] [CrossRef]
- Strassburg, B.B.N.; Brooks, T.; Feltran-Barbieri, R.; Iribarrem, A.; Crouzeilles, R.; Loyola, R.; Latawiec, A.E.; Oliveira-Filho, F.J.B.; Scaramuzza, C.A.M.; Scarano, F.R.; et al. Moment of truth for the Cerrado hotspot. Nat. Ecol. Evol. 2017, 1, 99. [Google Scholar] [CrossRef] [PubMed]
- Tohge, T.; de Souza, L.P.; Fernie, A.R. Current understanding of the pathways of flavonoid biosynthesis in model and crop plants. J. Exp. Bot. 2017, 68, 4013–4028. [Google Scholar] [CrossRef] [PubMed]
- Fank-de-Carvalho, S.M.; Somavilla, N.S.; Marchioretto, M.S.; Báo, S.N. Plant structure in the Brazilian neotropical savannah species. In Biodiversity in Ecosystems—Linking Structure and Function; Lo, Y., Blanco, J.A., Roy, S., Eds.; InTech: Rijeka, Croatia, 2015; pp. 425–459. [Google Scholar]
- Franzon, R.C.; Campos, L.Z.O.; Proença, C.E.B.; Sousa-Silva, J.C. Araças do Gênero Psidium: Principais Espécies, Ocorrência, Descrição e Usos; Embrapa Cerrados: Planaltina, Brazil, 2009; p. 48. [Google Scholar]
- Instituto Brasileiro de Geografia e Estatística (IBGE). Mapa de Biomas do Brasil; IBGE: Rio de Janeiro, Brazil, 2004. [Google Scholar]
- Ministério do Meio Ambiente (MMA). Diagnóstico Estratégico MacroZEE do Bioma Cerrado: Dinâmicas do Cerrado; MMA: São Paulo, Brazil, 2014. [Google Scholar]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, L.M. O conceito de cerrado. Rev. Bras. Bot. 1978, 1, 17–23. [Google Scholar]
- Ribeiro, J.F.; Walter, B.M.T. Fitofisionomias do bioma Cerrado. In Cerrado: Ambiente e Flora; Sano, S.M., Almeida, S.P., Eds.; Embrapa: Planaltina, Brazil, 1998; pp. 89–166. [Google Scholar]
- Rodrigues, V.E.G.; Carvalho, D.A. Plantas Medicinais no Domínio dos Cerrados; UFLA: Lavras, Brazil, 2001; p. 180. [Google Scholar]
- Almeida, S.P.; Proença, C.E.B.; Sano, S.M.; Ribeiro, J.F. Cerrado: Espécies Vegetais Úteis; Embrapa: Planaltina, Brazil, 1998; p. 464. [Google Scholar]
- Rigonato, V.D. Resenha Do Livro “A Encruzilhada Socioambiental: Biodiversidade, Economia e Sustentabilidade no Cerrado”; Ateliê Geográfico: Goiânia, Brazil, 2011; Volume 5, pp. 321–323. [Google Scholar]
- Bailão, E.F.L.C.; Devilla, I.A.; da Conceição, E.C.; Borges, L.L. Bioactive Compounds Found in Brazilian Cerrado Fruits. Int. J. Mol. Sci. 2015, 16, 23760–23783. [Google Scholar] [CrossRef] [PubMed]
- Rosa, F.R.; Arruda, A.F.; Siqueira, E.M.A.; Arruda, S.F. Phytochemical Compounds and Antioxidant Capacity of Tucum-Do-Cerrado (Bactris setosa Mart), Brazil’s Native Fruit. Nutrients 2016, 8, 110. [Google Scholar] [CrossRef]
- Havsteen, B. Flavonoids, a class of natural products of high pharmacological potency. Biochem. Pharmacol. 1983, 32, 1141–1148. [Google Scholar] [CrossRef]
- Cechinel Filho, V. Estratégias para a obtenção de compostos farmacologicamente ativos a partir de plantas medicinais. Conceitos sobre modificação estrutural para otimização da atividade. Quím. Nova 1998, 21, 99–105. [Google Scholar] [CrossRef]
- Pilon, A.C.; Valli, M.; Dametto, A.C.; Pinto, M.E.F.; Freire, R.T.; Castro-Gamboa, I.; Andricopulo, A.D.; Bolzani, V.S. NuBBEDB: An updated database to uncover chemical and biological information from Brazilian biodiversity. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Gallo, M.B.; Silva, F.C.; Vieira, P.C.; Fernandes, J.B.; da Silva, M.F. New natural products from Siphoneugena densiflora Berg (Myrtaceae) and their chemotaxonomic significance. J. Braz. Chem. Soc. 2006, 17, 279–288. [Google Scholar] [CrossRef]
- Regasini, L.O.; Vellosa, J.C.R.; Silva, D.H.S.; Furlan, M.; de Oliveira, O.M.M.; Khalil, N.M.; Brunetti, I.L.; Young, M.C.M.; Barreiro, E.J.; Bolzani, V.S. Flavonols from Pterogyne nitens and their evaluation as myeloperoxidase inhibitors. Phytochemistry 2008, 69, 1739–1744. [Google Scholar] [CrossRef] [PubMed]
- Hamburger, M.O.; Cordell, G.A.; Tantivatana, P.; Ruangrungsi, N. Traditional Medicinal Plants of Thailand, VIII. Isoflavonoids of Dalbergia candenatensis. J. Nat. Prod. 1987, 50, 696–699. [Google Scholar] [CrossRef] [PubMed]
- Serpeloni, J.M.; Leal Specian, A.F.; Ribeiro, D.L.; Tuttis, K.; Vilegas, W.; Martínez-López, W.; Dokkedal, A.L.; Saldanha, L.L.; Cólus, I.M.S.; Varanda, E.A. Antimutagenicity and induction of antioxidant defense by flavonoid rich extract of Myrcia bella Cambess. in normal and tumor gastric cells. J. Ethnopharmacol. 2015, 176, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.M.; Souza, E.R.B.; Paula, J.A.M.; Borges, L.L.; Caramori, S.S. Chemical Characterization and Bioprospecting of Cashew Tree Polysaccharideof Brazilian Cerrado (Anacardium othonianum Rizz.), Anacardiaceae. Front. J. Soc. Technol. Environ. Sci. 2017, 6, 230–246. [Google Scholar]
- Ferreyra, M.L.F.; Rius, S.P.; Casati, P. Flavonoids: Biosynthesis, biological functions, and biotechnological applications. Front. Plant Sci. 2012, 3, 222. [Google Scholar]
- Ribeiro, V.P.; Arruda, C.; Abd El-Salam, M.; Bastos, J.K. Brazilian medicinal plants with corroborated anti-inflammatory activities: A review. Pharm. Biol. 2018, 56, 253–268. [Google Scholar] [CrossRef] [PubMed]
- Teles, Y.C.F.; Souza, M.S.R.; de Souza, M.F.V. Sulphated Flavonoids: Biosynthesis, Structures, and Biological Activities. Molecules 2018, 23, 480. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Woo, J.B.; Ryu, S.I.; Moon, S.K.; Han, N.S.; Lee, S.B. Glucosylation of flavonol and flavanones by Bacillus cyclodextrin glucosyltransferase to enhance their solubility and stability. Food Chem. 2017, 229, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Saito, K.; Yonekura-Sakakibara, K.; Nakabayashi, R.; Higashi, Y.; Yamazaki, M.; Tohge, T.; Fernie, A.R. The flavonoid biosynthetic pathway in Arabidopsis: Structural and genetic diversity. Plant Physiol. Biochem. 2013, 72, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Liu, H.; Yang, J.; Gupta, V.K.; Jiang, Y. New insights on bioactivities and biosynthesis of flavonoid glycosides. Trends Food Sci. Technol. 2018, 79, 116–124. [Google Scholar] [CrossRef]
- Fraser, C.M.; Chapple, C. The phenylpropanoid pathway in Arabidopsis. Arab. Book 2011, 9, e0152. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Dudareva, N. The shikimate pathway and aromatic amino acid biosynthesis in plants. Annu. Rev. Plant Biol. 2012, 63, 73–105. [Google Scholar] [CrossRef]
- Hamberger, B.; Hahlbrock, K. The 4-coumarate: CoA ligase gene family in Arabidopsis thaliana comprises one rare, sinapate-activating and three commonly occurring isoenzymes. Proc. Natl. Acad. Sci. USA 2004, 101, 2209–2214. [Google Scholar] [CrossRef] [PubMed]
- Baud, S.; Guyon, V.; Kronenberger, J.; Wuilleme, S.; Miquel, M.; Caboche, M.; Lepiniec, L.; Rochat, C. Multifunctional acetyl-CoA carboxylase 1 is essential for very long chain fatty acid elongation and embryo development in Arabidopsis. Plant J. 2003, 33, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Lepiniec, L.; Debeaujon, I.; Routaboul, J.M.; Baudry, A.; Pourcel, L.; Nesi, N.; Caboche, M. Genetics and biochemistry of seed flavonoids. Annu. Rev. Plant Biol. 2006, 57, 405–430. [Google Scholar] [CrossRef] [PubMed]
- Winkel-Shirley, B. Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol. 2001, 126, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Davies, K.M.; Kathy, E.; Schwinn, K.E. Molecular Biology and Biotechnology of Flavonoid Biosynthesis. In Flavonoids Chemistry, Biochemistry and Applications; Andersen, O.M., Markham, K.R., Eds.; Taylor & Francis Group: London, UK, 2005; pp. 143–218. [Google Scholar]
- Ma, J.; Zhang, J.; Shen, Z. Two unusual isoflavonoids from Campylotropis hirtella—A new biosynthesis route of flavonoids. Tetrahedron Lett. 2017, 58, 1462–1466. [Google Scholar] [CrossRef]
- Pedroso, R.C.; Branquinho, N.A.; Hara, A.C.; Costa, A.C.; Silva, F.G.; Pimenta, L.P.; Silva, M.L.; Cunha, W.R.; Pauletti, P.M.; Januario, A.H. Impact of light quality on flavonoid production and growth of Hyptis marrubioides Epling seedlings cultivated in vitro. Rev. Bras. Farmacogn. 2017, 27, 466–470. [Google Scholar] [CrossRef]
- Wang, R.E.; Kao, J.L.-F.; Hilliard, C.A.; Pandita, R.K.; Roti Roti, J.L.; Hunt, C.R.; Taylor, J.-S. Inhibition of Heat Shock Induction of Heat Shock Protein 70 and Enhancement of Heat Shock Protein 27 Phosphorylation by Quercetin Derivatives. J. Med. Chem. 2009, 52, 1912–1921. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, H.B.; Campos, D.M.B.; Vaz, W.F.; Garro, F.L.T. Inovação e Biotecnologia na Biodiversidade do Cerrado. Fronteiras: J. Soc. Technol. Environ. Sci. 2016, 5, 162–180. [Google Scholar] [CrossRef]
- Linares, F.; Procopio, E.Q.; Galindo, M.A.; Romero, M.A.; Navarro, J.A.R.; Barea, E. Molecular architecture of redox-active half-sandwich Ru(II) cyclic assemblies. Interactions with biomolecules and anticancer activity. CrystEngComm 2010, 12, 2343–2346. [Google Scholar] [CrossRef]
- Babu, N.J.; Sanphui, P.; Nath, N.K.; Khandavilli, U.B.R.; Nangia, A. Temozolomide hydrochloride dihydrate. CrystEngComm 2013, 15, 666–671. [Google Scholar] [CrossRef]
- Baisch, U.; Vella-Zarb, L. Towards understanding P-gp resistance: A case study of the antitumour drug cabazitaxel. CrystEngComm 2014, 16, 10161–10164. [Google Scholar] [CrossRef]
- Therrien, B. Biologically relevant arene ruthenium metalla-assemblies. Crystengcomm. 2015, 17, 484–491. [Google Scholar] [CrossRef]
- Gupta, G.; Oggu, G.S.; Nagesh, N.; Bokara, K.K.; Therrien, B. Anticancer activity of large metalla-assemblies built from half-sandwich complexes. CrystEngComm 2016, 18, 4952–4957. [Google Scholar] [CrossRef]
- Ajibola Adeyemo, A.; Shettar, A.; Bhat, I.A.; Kondaiah, P.; Mukherjee, P.S. Self-Assembly of Discrete RuII8 Molecular Cages and Their in Vitro Anticancer Activity. Inorg. Chem. 2017, 56, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Fahmy, H.; Khalifa, N.; Ismail, M.; El-Sahrawy, H.; Nossier, E. Biological Validation of Novel Polysubstituted Pyrazole Candidates with in Vitro Anticancer Activities. Molecules 2016, 21, 271. [Google Scholar] [CrossRef]
- Vinardell, M.; Mitjans, M. Antitumor Activities of Metal Oxide Nanoparticles. Nanomaterials 2015, 5, 1004–1021. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. CA. Cancer statistics, 2016. Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Cryst. 2016, B72, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997, 23, 3–25. [Google Scholar] [CrossRef]
- Vaz, W.F.; Custodio, J.M.F.; Silveira, R.G.; Castro, A.N.; Campos, E.M.; Anjos, M.M.; Oliveira, G.R.; Valverde, C.; Napolitano, H.B. Synthesis, characterization, and third-order nonlinear optical properties of a new neolignane analogue. RSC Adv. Int. J. Furth. Chem. Sci. 2016, 79215–79227. [Google Scholar] [CrossRef]
- Spackman, M.A.; McKinnon, J.J. Fingerprinting intermolecular interactions in molecular crystals. CrystEngComm 2002, 4, 378–392. [Google Scholar] [CrossRef]
- Martin, A.D.; Britton, J.; Easun, T.L.; Blake, A.J.; Lewis, W.; Schröder, M. Hirshfeld Surface Investigation of Structure-Directing Interactions within Dipicolinic Acid Derivatives. Cryst. Growth Des. 2015, 15, 1697–1706. [Google Scholar] [CrossRef]
- The International Plant Names Index. 2012. Available online: http://www.ipni.org (accessed on 10 March 2019).
- Silva, K.L.; Cechinel Filho, V. Plantas do gênero Bauhinia: Composição química e potencial farmacológico. Quím. Nova 2002, 25, 449–454. [Google Scholar] [CrossRef]
- Estrela, F.N. Atividades Antiulcerogênica e Antimicrobiana da espécie Spiranthera odoratissima A. ST. HIL. (RUTACEAE). Ph.D. Thesis, Universidade Federal de Uberlândia, Uberlândia, Brazil, 2016. [Google Scholar]
- Ayres, M.C.C.; Escórcio, S.P.; Costa, D.A.; Chaves, M.H.; Vieira Júnior, G.M.; Cavalheiro, A.J. Químicos das folhas de Qualea grandiflora: Atribuição dos dados de RMN de dois flavonóides glicosilados acilados diastereoisoméricos. Quím. Nova 2008, 31, 1481–1484. [Google Scholar] [CrossRef]
- Silva, L.M.P.; Alves, J.S.F.; Siqueira, E.M.S.; Souza Neto, M.A.; Abreu, L.S.; Tavares, J.F.; Porto, D.L.; Ferreira, L.S.; Demarque, D.P.; Lopes, N.P.; et al. Isolation and Identification of the Five Novel Flavonoids from Genipa americana Leaves. Molecules 2018, 23, 2521. [Google Scholar] [CrossRef]
- Gaulton, A.; Bellis, L.J.; Bento, A.P.; Chambers, J.; Davies, M.; Hersey, A.; Light, Y.; McGlinchey, S.; Michalovich, D.; Al-Lazikani, B.; et al. ChEMBL: A large-scale bioactivity database for drug discovery. Nucleic Acids Res. 2012, 40, D1100–D1107. [Google Scholar] [CrossRef]
- Gottlieb, O.R.; de Oliveira, A.B.; Machado Gonçalves, T.M.; de Oliveira, G.G.; Pereira, S.A. Isoflavonoids from Cyclolobium species. Phytochemistry 1975, 14, 2495–2499. [Google Scholar] [CrossRef]
- Dhagat, U.; Endo, S.; Hara, A.; El-Kabbani, O. Inhibition of 3(17)α-hydroxysteroid dehydrogenase (AKR1C21) by aldose reductase inhibitors. Bioorganic Med. Chem. 2008, 16, 3245–3254. [Google Scholar] [CrossRef] [PubMed]
- Kador, P.F.; Kinoshita, J.H.; Sharpless, N.E. Aldose reductase inhibitors: A potential new class of agents for the pharmacological control of certain diabetic complications. J. Med. Chem. 1985, 28, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, J.H.; Nishimura, C. The involvement of aldose reductase in diabetic complications. Diabetes/Metab. Rev. 1988, 4, 323–337. [Google Scholar] [CrossRef]
- Coulerie, P.; Maciuk, A.; Eydoux, C.; Hnawia, E.; Lebouvier, N.; Figadère, B.; Guillemot, J.C.; Nour, M. New inhibitors of the DENV-NS5 RdRp from Carpolepis laurifolia as potential antiviral drugs for dengue treatment. Rec. Nat. Prod. 2014, 8, 286–289. [Google Scholar]
- Nicoletti, M.; Goulart, M.O.F.; de Lima, R.A.; Goulart, A.E.; Monache, F.D.; Bettolo, G.B.M. Flavonoids and Alkaloids from Strychnos pseudoquina. J. Nat. Prod. 1984, 47, 953–957. [Google Scholar] [CrossRef] [PubMed]
- Takemura, H.; Itoh, T.; Yamamoto, K.; Sakakibara, H.; Shimoi, K. Selective inhibition of methoxyflavonoids on human CYP1B1 activity. Bioorganic Med. Chem. 2010, 18, 6310–6315. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, L.R.; Ramalho, S.D.; Burger, M.C.; Nebo, L.; Fernandes, J.B.; da Silva, M.F.; Iemma, M.R.; Correa, C.J.; Souza, D.H.; Lima, M.I.; et al. Isolation of Arginase Inhibitors from the Bioactivity-Guided Fractionation of Byrsonima coccolobifolia Leaves and Stems. J. Nat. Prod. 2014, 77, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Ulloa, A.; Cid, A.; Rubio-Gayosso, I.; Ceballos, G.; Villarreal, F.; Ramirez-Sanchez, I. Effects of (−)-epicatechin and derivatives on nitric oxide mediated induction of mitochondrial proteins. Bioorganic Med. Chem. Lett. 2013, 23, 4441–4446. [Google Scholar] [CrossRef] [PubMed]
- Leitão, S.G.; Delle Monache, F. 2″-O-caffeoylorientin from Vitex polygama. Phytochemistry 1998, 49, 2167–2169. [Google Scholar] [CrossRef]
- Karioti, A.; Ceruso, M.; Carta, F.; Bilia, A.-R.; Supuran, C.T. New natural product carbonic anhydrase inhibitors incorporating phenol moieties. Bioorganic Med. Chem. 2015, 23, 7219–7225. [Google Scholar] [CrossRef] [PubMed]
- Pettit, G.R.; Meng, Y.; Stevenson, C.A.; Doubek, D.L.; Knight, J.C.; Cichacz, Z.; Pettit, R.K.; Chapuis, J.-C.; Schmidt, J.M. Isolation and Structure of Palstatin from the Amazon Tree Hymeneae p alustris 1. J. Nat. Prod. 2003, 66, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Braz Filho, R.; Gottlieb, O.R. The flavones of Apuleia leiocarpa. Phytochemistry 1971, 10, 2433–2450. [Google Scholar] [CrossRef]
- Pick, A.; Müller, H.; Mayer, R.; Haenisch, B.; Pajeva, I.K.; Weigt, M.; Bönisch, H.; Müller, C.E.; Wiese, M. Structure–activity relationships of flavonoids as inhibitors of breast cancer resistance protein (BCRP). Bioorganic Med. Chem. 2011, 19, 2090–2102. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.-H.; Li, N.-G.; Tang, Y.-P.; Shi, Q.-P.; Zhang, W.; Zhang, P.-X.; Dong, Z.-X.; Li, W.; Zhang, X.; Fu, H.-A.; et al. Synthesis, biological evaluation and SAR analysis of O-alkylated analogs of quercetin for anticancer. Bioorganic Med. Chem. Lett. 2014, 24, 4424–4427. [Google Scholar] [CrossRef] [PubMed]
- Lescano, C.H.; Freitas de Lima, F.; Mendes-Silvério, C.B.; Justo, A.F.O.; da Silva Baldivia, D.; Vieira, C.P.; Sanjinez-Argandoña, E.J.; Cardoso, C.A.L.; Mónica, F.Z.; Pires de Oliveira, I. Effect of Polyphenols from Campomanesia adamantium on Platelet Aggregation and Inhibition of Cyclooxygenases: Molecular Docking and in Vitro Analysis. Front. Pharmacol. 2018, 9, 617. [Google Scholar] [CrossRef] [PubMed]
- Salem, M.M.; Werbovetz, K.A. Antiprotozoal Compounds from Psorothamnus p olydenius. J. Nat. Prod. 2005, 68, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Burger, M.C.M.; Fernandes, J.B.; da Silva, M.F.G.F.; Escalante, A.; Prudhomme, J.; Le Roch, K.G.; Izidoro, M.A.; Vieira, P.C. Structures and bioactivities of dihydrochalcones from Metrodorea stipularis. J. Nat. Prod. 2014, 77, 2418–2422. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Species Name | Family | Flavonoids |
|---|---|---|
| Secondatia densiflora A.DC. | Apocynaceae | Pterogynoside; quercetin |
| Tithonia diversifolia (Hemsl) | Asteraceae | Luteolin |
| Apuleia leiocarpa (Vogel) J.F.Macbr. | Fabaceae | Ayanin |
| Bauhinia candicans Benth. | Fabaceae | Kaempferol 3-O-rutinoside; Kaempferol 3-O-rutinosidium |
| Bauhinia forficata Link. | Fabaceae | kaempferitrine |
| Bauhinia guianensis Aubl. | Fabaceae | 4-hydroxy-7-methoxyflavone |
| Bauhinia manca Standl. | Fabaceae | Apigenin; Chrisoeriol; Luteolin 5,3-dimethoxy; Kaempferol; Isoliquiritigenin; 2-methoxy isoliquiritigenin; 4-methoxy isoliquiritigenin; Echinatine; 2,4-dihydroxy-4-methoxy-dihydrochalcone; (2S)-Narigenin (2S)-Eriodiethiol; (2S)-Lithiocygenin; (2S)-Lithiocygenin 7-methoxy; (2S)-Lithiocygenin 4-methoxy; (2S)-7,4-Dihydroxyflavone; (2S)-7,3-Dimethoxy-4-hydroxy-flavone; (2S)-3,4-Dimethoxy-7-hydroxy-flavone; (2S)-7,4-Dimethoxy-3-hydroxy-flavone |
| Bauhinia megalandra Griseb. | Fabaceae | 5,7,5′-trihydroxy-2′-O-rhamnosyl-flavone; 5,7,2′-trihydroxy-5′-O-rhamnosyl flavone |
| Bauhinia purpúrea DC. ex Walp. | Fabaceae | Soquercitrin; Quercetin; Astragalin |
| Bauhinia reticulata DC. | Fabaceae | Quercetin |
| Bauhinia splendens Kunth | Fabaceae | Bausplendin, Quercetin, Routine |
| Bauhinia tomentosa Náves ex Fern.-Vill. | Fabaceae | Isoquercitrin; Quercetin; Routine |
| Bauhinia vahlii Wight & Arn. | Fabaceae | Quercetin; Quercetin-3-glucoside; Kaempferol; Agathisflavone |
| Bauhinia. variegata L. | Fabaceae | Narigenin-5,7-dimethoxy-4-rhamnoglycoside; Kaempferol-3-galactoside; Kaempferol-3-ramno-glucoside |
| Cyclolobium claussenii Benth. | Fabaceae | (3R)-claussequinone |
| Pterogyne nitens Tull. | Fabaceae | Pterogynoside; quercetin |
| Strychnos pseudoquina A. St. Hil. | Loganiaceae | isorhamnetin |
| Byrsonima coccolobifolia Kunth | Malpighiaceae | (−)-epicatechin |
| Campomanesia adamantium (Cambess.) O.Berg | Myrtaceae | Aurentiacin-A |
| Siphoneugena densiflora O.Berg. | Myrtaceae | Quercetin |
| Metrodorea stipularis Mart. | Myrtaceae | 1-[3-(3,7-dimethylocta-2,6-dien-1-yl)-2,4,6-trihydroxyphenyl]-3-(4-hydroxyphenyl)propan-1-one 1-(5,7-dihydroxy-2,2-dimethylchroman-6-yl)-3-(1,1,4a-trimethyl-2,3,4,4a,9a-hexahydro-1H-xanthen-7-yl)propan-1-one |
| Genipa americana L | Rubiaceae | Quercetin-3-O-robinoside, Kaempferol-3-O-robinoside, Isorhamnetin-3-O-robinoside, Kaempferol-3-O-robinoside-7-O-rhamnoside (robinin), Isorhamnetin-3-O-robinoside-7-rhamnoside. |
| Spiranthera odoratissima A. St.-Hil. | Rutaceae | Kaempferol-3-ramno-glucoside |
| Vitex polygama Cham. | Verbenaceae | Luteolin; chrysoeriol |
| Qualea grandiflora Mart. | Vochysiaceae | kanferol-3-OAL-(4”-Zp-cumaroyl) -raminoside (2), squalene, phytol, lupeol, A-amyrin, B-amirin, sitosterol, ursolic and oleanolic acids and sitosterol 3-OBD-glucopyranoside. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peixoto, J.d.C.; Neves, B.J.; Vasconcelos, F.G.; Napolitano, H.B.; Barbalho, M.G.d.S.; Silva, S.D.e.; Rosseto, L.P. Flavonoids from Brazilian Cerrado: Biosynthesis, Chemical and Biological Profile. Molecules 2019, 24, 2891. https://doi.org/10.3390/molecules24162891
Peixoto JdC, Neves BJ, Vasconcelos FG, Napolitano HB, Barbalho MGdS, Silva SDe, Rosseto LP. Flavonoids from Brazilian Cerrado: Biosynthesis, Chemical and Biological Profile. Molecules. 2019; 24(16):2891. https://doi.org/10.3390/molecules24162891
Chicago/Turabian StylePeixoto, Josana de Castro, Bruno Junior Neves, Flávia Gonçalves Vasconcelos, Hamilton Barbosa Napolitano, Maria Gonçalves da Silva Barbalho, Sandro Dutra e Silva, and Lucimar Pinheiro Rosseto. 2019. "Flavonoids from Brazilian Cerrado: Biosynthesis, Chemical and Biological Profile" Molecules 24, no. 16: 2891. https://doi.org/10.3390/molecules24162891
APA StylePeixoto, J. d. C., Neves, B. J., Vasconcelos, F. G., Napolitano, H. B., Barbalho, M. G. d. S., Silva, S. D. e., & Rosseto, L. P. (2019). Flavonoids from Brazilian Cerrado: Biosynthesis, Chemical and Biological Profile. Molecules, 24(16), 2891. https://doi.org/10.3390/molecules24162891