Curcumin Combination Chemotherapy: The Implication and Efficacy in Cancer
Abstract
1. Introduction
2. Mechanisms of Action of Oxidative Stress-Induced Cancer
3. Curcumin
4. Anticarcinogenic Potency and Molecular Mechanisms Induced by Curcumin
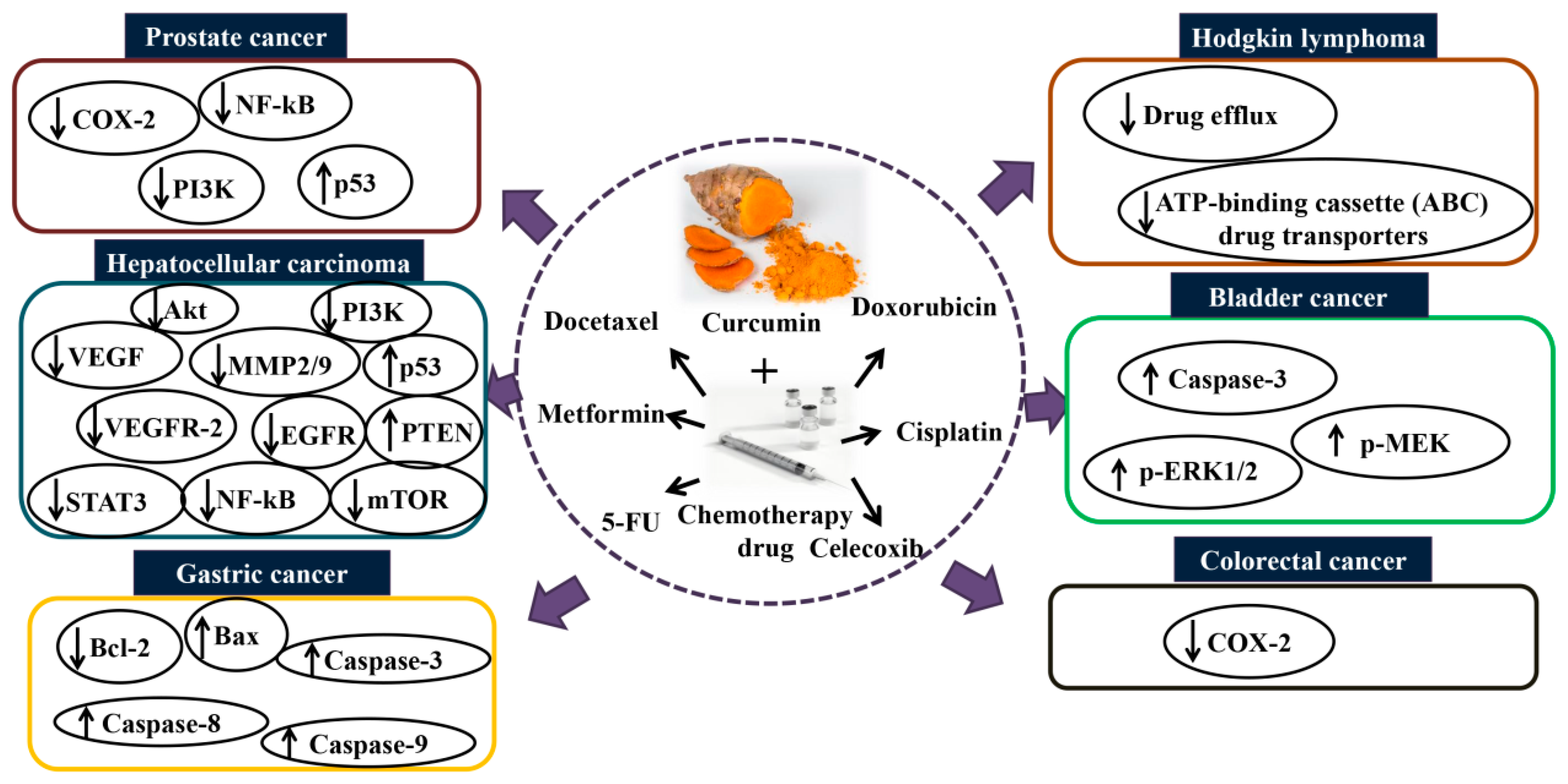
5. The Synergistic Effect of Curcumin Combination Chemotherapy
5.1. In Vitro and In Vivo
5.2. Clinical Trials
6. Nanoformulation of Curcumin Combination Chemotherapy
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Cancer. 2019. Available online: https://www.who.int/cancer/en/ (accessed on 25 February 2019).
- Seyed, M.A.; Jantan, I.; Bukhari, S.N.A.; Vijayaraghavan, K. A comprehensive review on the chemotherapeutic potential of piceatannol for cancer treatment, with mechanistic insights. J. Agric. Food Chem. 2016, 64, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.-J.; Chen, C.; Zhao, Y.; Wang, P.C. Circumventing tumor resistance to chemotherapy by nanotechnology. Methods Mol. Biol. 2010, 596, 467–488. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.-H.; Chen, M.-C.; Baskaran, R.; Lin, Y.M.; Day, C.H.; Lin, Y.J.; Tu, C.C.; Vijaya Padma, V.; Kuo, W.W.; Huang, C.Y. Oxaliplatin resistance in colorectal cancer cells is mediated via activation of ABCG2 to alleviate ER stress induced apoptosis. J. Cell. Physiol. 2018, 233, 5458–5467. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-T.; Hu, W.-T.; Zhou, J.-Y.; Tu, Y. Celecoxib enhances the radiosensitivity of HCT116 cells in a COX-2 independent manner by up-regulating BCCIP. Am. J. Transl. Res. 2017, 9, 1088–1100. [Google Scholar] [PubMed]
- Ahmad, S.; Panda, B.P.; Fahim, M.; Dhyani, N.; Dubey, K. Ameliorative effect of beraprost sodium on celecoxib induced cardiotoxicity in rats. Iran. J. Pharm. Res. 2018, 17, 155–163. [Google Scholar]
- Li, Q.; Yang, G.; Feng, M.; Zheng, S.; Cao, Z.; Qiu, J.; You, L.; Zheng, L.; Hu, Y.; Zhang, T.; et al. NF-κB in pancreatic cancer: Its key role in chemoresistance. Cancer Lett. 2018, 421, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Deveci, H.A.; Naziroğlu, M.; Nur, G. 5-Fluorouracil-induced mitochondrial oxidative cytotoxicity and apoptosis are increased in MCF-7 human breast cancer cells by TRPV1 channel activation but not Hypericum perforatum treatment. Mol. Cell. Biochem. 2018, 439, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Cai, D.; He, K.; Chang, S.; Tong, D.; Huang, C. MicroRNA-302b enhances the sensitivity of hepatocellular carcinoma cell lines to 5-FU via targeting Mcl-1 and DPYD. Int. J. Mol. Sci. 2015, 16, 23668–23682. [Google Scholar] [CrossRef]
- Chen, Z.; Penet, M.-F.; Krishnamachary, B.; Banerjee, S.R.; Pomper, M.G.; Bhujwalla, Z.M. PSMA-specific theranostic nanoplex for combination of TRAIL gene and 5-FC prodrug therapy of prostate cancer. Biomaterials 2016, 80, 57–67. [Google Scholar] [CrossRef]
- Lee, J.J.; Beumer, J.H.; Chu, E. Therapeutic drug monitoring of 5-fluorouracil. Cancer Chemother. Pharmacol. 2016, 78, 447–464. [Google Scholar] [CrossRef]
- Polk, A.; Vaage-Nilsen, M.; Vistisen, K.; Nielsen, D.L. Cardiotoxicity in cancer patients treated with 5-fluorouracil or capecitabine: A systematic review of incidence, manifestations and predisposing factors. Cancer Treat. Rev. 2013, 39, 974–984. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.L.; Norhaizan, M.E.; Huynh, K.; Heshu, S.R.; Yeap, S.K.; Hazilawati, H.; Roselina, K. Water extract of brewers’ rice induces apoptosis in human colorectal cancer cells via activation of caspase-3 and caspase-8 and downregulates the Wnt/β-catenin downstream signaling pathway in brewers’ rice-treated rats with azoxymethane-induced colon carcinogenesis. BMC Complement. Altern. Med. 2015, 15, 205. [Google Scholar]
- Pulido-Moran, M.; Moreno-Fernandez, J.; Ramirez-Tortosa, C.; Ramirez-Tortosa, M. Curcumin and health. Molecules 2016, 21, 264. [Google Scholar] [CrossRef] [PubMed]
- Barchitta, M.; Maugeri, A.; Favara, G.; Magnano San Lio, R.; Evola, G.; Agodi, A.; Basile, G. Nutrition and wound healing: An overview focusing on the beneficial effects of curcumin. Int. J. Mol. Sci. 2019, 20, 1119. [Google Scholar] [CrossRef] [PubMed]
- Mantzorou, M.; Pavlidou, E.; Vasios, G.; Tsagalioti, E.; Giaginis, C. Effects of curcumin consumption on human chronic diseases: A narrative review of the most recent clinical data. Phytother. Res. 2018, 32, 957–975. [Google Scholar] [CrossRef] [PubMed]
- Tan, R.-Z.; Liu, J.; Zhang, Y.-Y.; Wang, H.-L.; Li, J.-C.; Liu, Y.-H.; Xia, Z.; Zhang, Y.-W.; Yan, Y.; Lan, H.-Y.; et al. Curcumin relieved cisplatin-induced kidney inflammation through inhibiting Mincle-maintained M1 macrophage phenotype. Phytomedicine 2019, 52, 284–294. [Google Scholar] [CrossRef] [PubMed]
- Basnet, P.; Skalko, B.N. Curcumin: An anti-inflammatory molecule from a curry spice on the path to cancer treatment. Molecules 2011, 16, 4567–4598. [Google Scholar] [CrossRef]
- Creţu, E.; Trifan, A.; Vasincu, A.; Miron, A. Plant-derived anticancer agents-curcumin in cancer prevention and treatment. Revista MedicoChirugicala Societatii Medici Si Naturalisti Din Iasi 2012, 116, 1223–1229. [Google Scholar]
- Zhang, P.; Lai, Z.L.; Chen, H.F.; Zhang, M.; Wang, A.; Jia, T.; Sun, W.Q.; Zhu, X.M.; Chen, X.F.; Zhao, Z.; et al. Curcumin synergizes with 5-fluorouracil by impairing AMPK/ULK1-dependent autophagy, AKT activity and enhancing apoptosis in colon cancer cells with tumor growth inhibition in xenograft mice. J. Exp. Clin. Cancer Res. 2017, 36, 190. [Google Scholar] [CrossRef]
- Vakifahmetoglu-Norberg, H.; Ouchida, A.T.; Norberg, E. The role of mitochondria in metabolism and cell death. Biochem. Biophys. Res. Commun. 2017, 482, 426–431. [Google Scholar] [CrossRef]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. BioMed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, P.; Nandakumar, N.; Rengarajan, T.; Palaniswami, R.; Gnanadhas, E.N.; Lakshminarasaiah, U.; Gopas, J.; Nishigaki, I. Antioxidants and human diseases. Clin. Chim. Acta 2014, 436, 332–347. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.J.; Lee, E.K.; Kim, J.Y.; Zou, Y.; Sung, B.; Heo, H.S.; Kim, M.K.; Lee, J.; Kim, N.D.; Yu, B.P.; et al. Effect of short term calorie restriction on pro-inflammatory NF-kB and AP-1 in aged rat kidney. Inflamm. Res. 2009, 58, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Xiao, T.S. Innate immunity and inflammation. Cell. Mol. Immunol. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Qu, D.; Shen, L.S.; Liu, S.; Li, H.; Ma, Y.; Zhang, R.; Wu, K.; Yao, L.; Li, J.; Zhang, J. Chronic inflammation confers to the metabolic reprogramming associated with tumorigenesis of colorectal cancer. Cancer Biol. Ther. 2017, 18, 237–244. [Google Scholar] [CrossRef]
- Colotta, F.; Allavena, P.; Sica, A.; Garlanda, C.; Mantovani, A. Cancer-related inflammation, the seventh hallmark of cancer: Links to genetic instability. Carcinogenesis 2009, 30, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Raposo, T.P.; Beirão, B.C.; Pang, L.Y.; Queiroga, F.L.; Argyle, D.J. Infammation and cancer: Till death tears them apart. Vet. J. 2015, 205, 161–174. [Google Scholar] [CrossRef]
- Rakoff-Nahoum, S. Why cancer and inflammation? Yale J. Biol. Med. 2006, 79, 123–130. [Google Scholar]
- Mangino, G.; Chiantore, M.V.; Iuliano, M.; Fiorucci, G.; Romeo, G. Inflammatory microenvironment and human papillomavirus-induced carcinogenesis. Cytokine Growth Factor Rev. 2016, 30, 103–111. [Google Scholar] [CrossRef]
- D’lgnazio, L.; Batie, M.; Rocha, S. Hypoxia and inflammation in cancer, focus on HIF and NF-κB. Biomedicines 2017, 5, 21. [Google Scholar]
- Kunnumakkara, A.B.; Sailo, B.L.; Banik, K.; Harsha, C.; Prasad, S.; Gupta, S.C.; Bharti, A.C.; Aggarwal, B.B. Chronic diseases, inflammation, and spices: How are they linked? J. Transl. Med. 2018, 16, 14. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.; Carmody, R.J. NF-κB and the transcriptional control of inflammation. In Transcriptional Gene Regulation in Health and Disease; Loos, F., Ed.; Academic Press: Cambridge, MA, USA, 2018; Volume 335, pp. 41–84. [Google Scholar]
- Karin, M. The IkappaB kinase—A bridge between inflammation and cancer. Cell Res. 2008, 18, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A. The inflammation-cancer connection. FEBS J. 2018, 285, 638–640. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Singh, D.; Anwar, F.; Bhatt, P.C.; Al-Abbasi, F.; Kumar, V. Triterpenoids principle of Wedelia calendulacea attenuated diethynitrosamine-induced hepatocellular carcinoma via down-regulating oxidative stress, inflammation and pathology via NF-kB pathway. Inflammopharmacology 2018, 26, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Bromberg, J.; Wang, T.C. Inflammation and cancer: IL-6 and STAT3 complete the link. Cancer Cell 2009, 15, 79–80. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.Q.; Xu, F.; Lu, T.H.; Duan, Z.F.; Zhang, Z. Interleukin-6 signaling pathway in targeted therapy for cancer. Cancer Treat. Rev. 2012, 38, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-C.; Su, C.-Y.; Chen, M.-H.; Chen, W.-S.; Chen, C.-L.; Hsiao, M. Secretory RAB GTPase 3C modulates IL6-STAT3 pathway to promote colon cancer metastasis and is associated with poor prognosis. Mol. Cancer 2017, 16, 135. [Google Scholar] [CrossRef]
- Sung, B.; Prasad, S.; Yadav, V.R.; Aggarwal, B.B. Cancer cell signaling pathways-targeted by spice-derived nutraceuticals. Nutr. Cancer 2012, 64, 173–197. [Google Scholar] [CrossRef]
- Kim, S.J.; Yoon, S. Activated Rac1 regulates the degradation of IκBα and the nuclear translocation of STAT3-NFκB complexes in starved cancer cells. Exp. Mol. Med. 2016, 48, e231. [Google Scholar] [CrossRef]
- Chung, S.S.; Wu, Y.; Okobi, Q.; Adekoya, D.; Atefi, M.; Clarke, O.; Dutta, P.; Vadgama, J.V. Proinflammatory cytokines IL-6 and TNF-α increased telomerase activity through NF-κB/STAT1/STAT3 activation, and Withaferin A inhibited the signaling in colorectal cancer cells. Mediat. Inflamm. 2017, 2017, 5958429. [Google Scholar] [CrossRef]
- Stanley, J.; Jua, C.; Irene, K.; Gordon, W.; Mieko, T.; Bongo, S.; Ashley, V. Interleukin-6, a cytokine critical to mediation of inflammation, autoimmunity and allograft rejection: Therapeutic implications of IL-6 receptor blockade. Transplantation 2017, 101, 32–44. [Google Scholar] [CrossRef]
- Lee, H.A.; Koh, E.K.; Sung, J.E.; Kim, J.E.; Song, S.H.; Kim, D.S.; Son, H.J.; Lee, C.Y.; Lee, H.S.; Bae, C.J.; et al. Ethyl acetate extract from Asparagus cochinchinensis exerts anti-inflammatory effects in LPS-stimulated RAW264.7 macrophage cells by regulating COX-2/iNOS, inflammatory cytokine expression, MAP kinase pathways, the cell cycle and anti-oxidant activity. Mol. Med. Rep. 2017, 15, 1613–1623. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, H.; Shakeri, A.; Rashidi, B.; Jalili, A.; Banikazemi, Z.; Sahebkar, A. Phytosomal curcumin: A review of pharmacokinetic, experimental and clinical studies. Biomed. Pharmacother. 2017, 85, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Farooqui, T.; Farooqui, A.A. Chapter 2—Curcumin: Historical background, chemistry, pharmacological action, and potential therapeutic value. In Curcumin for Neurological and Psychiatric Disorders: Neurochemical and Pharmacological Properties; Academic Press: Cambridge, MA, USA, 2019; pp. 23–44. [Google Scholar]
- Singh, V.; Pal, M.; Gupta, S.; Tiwari, S.K.; Malkunje, L.; Das, S. Turmeric—A new treatment option for lichen planus: A pilot study. Natl. J. Maxillofac. Surg. 2013, 4, 198–201. [Google Scholar] [CrossRef] [PubMed]
- Shome, S.; Talukdar, A.D.; Choudhury, M.D.; Bhattacharya, M.K.; Upadhyaya, H. Curcumin as potential therapeutic natural product: A nanobiotechnological perspective. J. Pharm. Pharmacol. 2016, 68, 1481–1500. [Google Scholar] [CrossRef] [PubMed]
- Benzer, F.; Kandemir, F.M.; Ozkaraca, M.; Kucukler, S.; Caglayan, C. Curcumin ameliorates doxorubicin-induced cardiotoxicity by abrogation of inflammation, apoptosis, oxidative DNA damage, and protein oxidation in rats. J. Biochem. Mol. Toxicol. 2018, 32, e22030. [Google Scholar] [CrossRef]
- Campbell, M.S.; Fleenor, B.S. The emerging role of curcumin for improving vascular dysfunction: A review. Crit. Rev. Food Sci. Nutr. 2017, 58, 2790–2799. [Google Scholar] [CrossRef]
- Serafini, M.M.; Catanzaro, M.; Rosini, M.; Racchi, M.; Lanni, C. Curcumin in Alzheimer’s disease: Can we think to new strategies and perspectives for this molecule? Pharmacol. Res. 2017, 124, 146–155. [Google Scholar] [CrossRef]
- Shehzad, A.; Qureshi, M.; Anwar, M.N.; Lee, Y.S. Multifunctional curcumin mediate multitherapeutic effects. J. Food Sci. 2017, 82, 2006–2015. [Google Scholar] [CrossRef]
- Khamis, A.A.; Sharshar, A.H.; Mahmoud, A.H.; Mohamed, T.M. The antioxidant activity of curcumin extract against HepG2 cells. Int. J. Adv. Res. Sci. Technol. 2018, 7, 115–125. [Google Scholar]
- Goel, A.; Kunnumakkara, A.B.; Aggarwal, B.B. Curcumin as “Curecumin”: From kitchen to clinic. Biochem. Pharmacol. 2008, 75, 787–809. [Google Scholar] [CrossRef]
- Byun, S.-Y.; Kim, D.-B.; Kim, E. Curcumin ameliorates the tumor-enhancing effects of a high-protein diet in an azoxymethane-induced mouse model of colon carcinogenesis. Nutr. Res. 2015, 35, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Hossain, D.; Bhattacharyya, S.; Das, T.; Sa, G. Curcumin: The multi-targeted therapy for cancer regression. Front. Biosci. 2011, 4, 335–355. [Google Scholar]
- Chiang, I.; Wang, W.-S.; Liu, H.-C.; Yang, S.-T.; Tang, N.-Y.; Chung, J.-G. Curcumin alters gene expression-associated DNA damage, cell cycle, cell survival and cell migration and invasion in NCI-H460 human lung cancer cells in vitro. Oncol. Rep. 2015, 34, 1853–1874. [Google Scholar] [CrossRef]
- Momtazi, A.A.; Shahabipour, F.; Khatibi, S.; Johnston, T.P.; Pirro, M.; Sahebkar, A. Curcumin as a MicroRNA regulator in cancer: A review. Rev. Physiol. Biochem. Pharmacol. 2016, 171, 1–38. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, H.; Naseri, G.; Rezaee, R.; Mohammadi, M.; Banikazemi, Z.; Mirzaei, H.R.; Salehi, H.; Peyvandi, M.; Pawelek, J.M.; Sahebkar, A. Curcumin: A new candidate for melanomatherapy? Int. J. Cancer 2016, 139, 1683–1695. [Google Scholar] [CrossRef] [PubMed]
- Hoesel, B.; Schmid, J.A. The complexity of NF-κB signaling in inflammation and cancer. Mol. Cancer 2013, 12, 86. [Google Scholar] [CrossRef]
- Taniguchi, K.; Karin, M. NF-κB, inflammation, immunity and cancer: Coming of age. Nat. Rev. Immunol. 2018, 18, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Mortezaee, K.; Salehi, E.; Mirtavoos-mahyari, H.; Motevaseli, E.; Najafi, M.; Farhood, B.; Rosengren, R.J.; Sahebkar, A. Mechanisms of apoptosis modulation by curcumin: Implications for cancer therapy. J. Cell. Physiol. 2019, 234, 12537–12550. [Google Scholar] [CrossRef]
- Bimonte, S.; Barbieri, A.; Leongito, M.; Piccirillo, M.; Giudice, A.; Pivonello, C.; de Angelis, C.; Granata, V.; Palaia, R.; Izzo, F.; et al. Curcumin anticancer studies in pancreatic cancer. Nutrients 2016, 8, 433. [Google Scholar] [CrossRef]
- Gupta, S.C.; Patchva, S.; Aggarwal, B.B. Therapeutic roles of curcumin: Lessons learned from clinical trials. AAPS J. 2013, 15, 195–218. [Google Scholar] [CrossRef] [PubMed]
- Goradel, N.H.; Hour, F.G.; Negahdari, B.; Malekshahi, Z.V.; Hashemzehi, M.; Masoudifar, A.; Mirzaei, H. Stem cell therapy: A new therapeutic option for cardiovascular diseases. J. Cell. Biochem. 2017, 119, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Hashemi Goradel, N.; Ghiyami-Hoor, F.; Jahangiri, S.; Negahdari, B.; Sahebkar, A.; Masoudifar, A.; Mirzaei, H. Nanoparticles as new tools for inhibition of cancer angiogenesis. J. Cell. Physiol. 2018, 233, 2902–2910. [Google Scholar] [CrossRef] [PubMed]
- Rupaimoole, R.; Slack, F.J. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nat. Rev. Drug Discov. 2017, 16, 203–222. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, H.; Masoudifar, A.; Sahebkar, A.; Zare, N.; Sadri Nahand, J.; Rashidi, B.; Mehrabian, E.; Mohammadi, M.; Mirzaei, H.R.; Jaafari, M.R. MicroRNA: A novel target of curcumin in cancer therapy. J. Cell. Physiol. 2018, 233, 3004–3015. [Google Scholar] [CrossRef] [PubMed]
- Kronski, E.; Fiori, M.E.; Barbieri, O.; Astigiano, S.; Mirisola, V.; Killian, P.H.; Bruno, A.; Pagani, A.; Rovera, F.; Pfeffer, U.; et al. MiR181b is induced by the chemopreventive polyphenol curcumin and inhibits breast cancer metastasis via down-regulation of the inflammatory cytokines CXCL1 and -2. Mol. Oncol. 2014, 8, 581–595. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Qiao, F.; Wang, Y.; Xu, Y.; Shang, Y. Curcumin inhibits cell proliferation and induces apoptosis of human non-small cell lung cancer cells through the upregulation of miR-192-5p and suppression of PI3K/Akt signaling pathway. Oncol. Rep. 2015, 34, 2782–2789. [Google Scholar] [CrossRef] [PubMed]
- Schwertheim, S.; Wein, F.; Lennartz, K.; Worm, K.; Schmid, K.W.; Sheu-Grabellus, S.Y. Curcumin induces G2/M arrest, apoptosis, NF-kappaB inhibition, and expression of differentiation genes in thyroid carcinoma cells. J. Cancer Res. Clin. Oncol. 2017, 143, 1143–1154. [Google Scholar] [CrossRef]
- Cheng, A.L.; Hsu, C.H.; Lin, J.K.; Hsu, M.M.; Ho, Y.F.; Shen, T.S.; Ko, J.Y.; Lin, J.T.; Lin, B.R.; Ming-Shiang, W.; et al. Phase I clinical trial of curcumin, a chemopreventive agent, in patients with high-risk or pre-malignant lesions. Anticancer Res. 2001, 21, 2895–2900. [Google Scholar]
- Kang, Y.; Hu, W.; Bai, E.; Zheng, H.; Liu, Z.; Wu, J.; Jin, R.; Zhao, C.; Liang, G. Curcumin sensitizes human gastric cancer cells to 5-fluorouracil through inhibition of the NFκB survival-signaling pathway. Onco Targets Ther. 2016, 9, 7373–7384. [Google Scholar] [CrossRef]
- Yue, Q.; Gao, G.; Zou, G.; Yu, H.; Zheng, X. Natural products as adjunctive treatment for pancreatic cancer: Recent trends and advancements. BioMed Res. Int. 2017, 2017, 8412508. [Google Scholar] [CrossRef] [PubMed]
- Tannock, I.F.; de Wit, R.; Berry, W.R.; Horti, J.; Pluzanska, A.; Chi, K.N.; Oudard, S.; Théodore, C.; James, N.D.; Turesson, N.D.; et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 2004, 351, 1502–1512. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.; Wang, Q.; Shi, Y.; Huang, Y.; Zhang, J.; Zhang, X.; Lin, G. Nanotechnology-mediated immunochemotherapy combined with docetaxel and PD-L1 antibody increase therapeutic effects and decrease systemic toxicity. J. Control. Release 2018, 286, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Singh, S.K.; Chowdhury, I.; Lillard, J.W., Jr.; Singh, R. Combinatorial effect of curcumin with docetaxel modulates apoptotic and cell survival molecules in prostate cancer. Front. Biosci. 2017, 9, 235–245. [Google Scholar]
- Christofides, E.A. Practical insights into improving adherence to metformin therapy in patients with type 2 diabetes mellitus. Clin. Diabetes 2019, 37. [Google Scholar] [CrossRef]
- Donadon, V.; Balbi, M.; Mas, M.D.; Casarin, P.; Zanette, G. Metformin and reduced risk of hepatocellular carcinoma in diabetic patients with chronic liver disease. Liver Int. 2010, 30, 750–758. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.; Gandhi, J.; Joshi, G.; Smith, N.L.; Khan, S.A. The anticancer potential of metformin on prostate cancer. Prostate Cancer Prostatic Dis. 2019. [Google Scholar] [CrossRef] [PubMed]
- Hadad, S.M.; Coates, P.; Jordan, L.B.; Dowling, R.J.; Chang, M.C.; Done, S.J.; Purdie, C.A.; Goodwin, P.J.; Stambolic, V.; Moulder-Thompson, S.; et al. Evidence for biological effects of metformin in operable breast cancer: Biomarker analysis in a preoperative window of opportunity randomized trial. Breast Cancer Res. Treat. 2015, 150, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-H.; Zhang, Y.; Cheng, Y.-N.; Gong, F.-L.; Cao, Z.-Q.; Yu, L.-G.; Guo, X.-L. Metformin incombination with curcumin inhibits the growth, metastasis, and angiogenesis of hepatocellular carcinoma in vitro and in vivo. Mol. Carcinog. 2018, 57, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Kuang, M.; Gong, Y.; Cao, C.; Chen, J.; Tang, C. Survival benefit and safety of the combinations of FOLFOXIRI ± bevacizumab versus the combinations of FOLFIRI ± bevacizumab as first-line treatment for unresectable metastatic colorectal cancer: A meta-analysis. Onco Targets Ther. 2016, 9, 4833–4842. [Google Scholar] [CrossRef]
- Heydari, K.; Saidijam, M.; Sharifi, M.R.; Dermani, F.K.; Soleimani Asl, S.; Shabab, N.; Najafi, R. The effect of miR-200c inhibition on chemosensitivity (5- FluoroUracil) in colorectal cancer. Pathol. Oncol. Res. 2018, 24, 145–151. [Google Scholar] [CrossRef]
- Meng, X.; Fu, R. miR-206 regulates 5-FU resistance by targeting Bcl-2 in colon cancer cells. Onco Targets Ther. 2018, 11, 1757–1765. [Google Scholar] [CrossRef] [PubMed]
- Shakibaei, M.; Buhrmann, C.; Kraehe, P.; Shayan, P.; Lueders, C.; Goel, A. Curcumin chemosensitizes 5-fluorouracil resistant MMR-deficient human colon cancer cells in high density cultures. PLoS ONE 2014, 9, e85397. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wang, W.; Li, P.; Zheng, Z.; Tu, Y.; Zhang, Y.; You, T. Curcumin enhances the effects of 5-fluorouracil and oxaliplatin in inducing gastric cancer cell apoptosis both in vitro and in vivo. Oncol. Res. 2016, 23, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Toden, S.; Okugawa, Y.; Jascur, T.; Wodarz, D.; Komarova, N.L.; Buhrmann, C.; Shakibaei, M.; Boland, C.R.; Goel, A. Curcumin mediates chemosensitization to 5-fluorouracil through miRNA-induced suppression of epithelial-to-mesenchymal transition in chemoresistant colorectal cancer. Carcinogenesis 2015, 36, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Vinod, B.S.; Antony, J.; Nair, H.H.; Puliyappadamba, V.T.; Saikia, M.; Narayanan, S.S.; Bevin, A.; Anto, R.J. Mechanistic evaluation of the signaling events regulating curcumin-mediated chemosensitization of breast cancer cells to 5-fluorouracil. Cell Death Dis. 2013, 4, e505. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Huang, S.; Wei, Y.; Cao, S.; Pi, C.; Feng, T.; Liang, J.; Zhao, L.; Ren, G. Curcumin enhances the anticancer effect of 5-fluorouracil against gastric cancer through down-regulation of COX-2 and NF-κB signaling pathways. J. Cancer 2017, 8, 3697–3706. [Google Scholar] [CrossRef] [PubMed]
- Zong, L.; Cheng, G.; Liu, S.; Pi, Z.; Liu, Z.; Song, F. Reversal of multidrug resistance in breast cancer cells by a combination of ursolic acid with doxorubicin. J. Pharm. Biomed. Anal. 2019, 165, 268–275. [Google Scholar] [CrossRef]
- Abouzeid, A.H.; Pate, N.R.; Rachman, I.M.; Senn, S.; Torchilin, V.P. Anti-cancer activity of anti-GLUT1 antibody-targeted polymeric micelles co-loaded with curcumin and doxorubicin. J. Drug Target. 2013, 21, 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.L.; Shen, Y.M.; Zhang, Q.W.; Li, Y.L.; Luo, M.; Liu, Z.; Li, Y.; Qian, Z.Y.; Gao, X.; Shi, H.S. Codelivery of curcumin and doxorubicin by MPEG-PCL results in improved efficacy of systemically administered chemotherapy in mice with lung cancer. Int. J. Nanomed. 2013, 8, 3521–3531. [Google Scholar] [CrossRef]
- Duan, J.; Mansour, H.; Zhang, Y.; Deng, X.; Chen, Y.; Wang, J.; Pan, Y.; Zhao, J. Reversion of multidrug resistance by co-encapsulation of doxorubicin and curcumin in chitosan/poly(butyl cyanoacrylate) nanoparticles. Int. J. Pharm. 2012, 426, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Guorgui, J.; Wang, R.; Mattheolabakis, G.; Mackenzie, G.G. Curcumin formulated in solid lipid nanoparticles has enhanced efficacy in Hodgkin’s lymphoma in mice. Arch. Biochem. Biophys. 2018, 648, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Wang, J.; Guo, Q.; Tu, P. Simultaneous determination of doxorubicin and curcumin in rat plasma by LC–MS/MS and its application to pharmacokinetic study. J. Pharm. Biomed. Anal. 2015, 111, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, D.S. Challenges in the treatment of bladder cancer. Ann. Oncol. 2006, 17, v106–v112. [Google Scholar] [CrossRef] [PubMed]
- Yoon, C.Y.; Park, M.J.; Lee, J.S.; Lee, S.C.; Oh, J.J.; Park, H.; Chung, C.W.; Abdullajanov, M.M.; Jeong, S.J.; Hing, S.K.; et al. The histone deacetylase inhibitor trichostatin A synergistically resensitizes a cisplatin resistant human bladder cancer cell line. J. Urol. 2011, 185, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Yadav, A.; Hideg, K.; Kuppusamy, P.; Teknos, T.N.; Kumar, P. A novel curcumin analog (H-4073) enhances the therapeutic efficacy of cisplatin treatment in head and neck cancer. PLoS ONE 2014, 9, e93208. [Google Scholar] [CrossRef] [PubMed]
- Siddik, Z.H. Cisplatin: Mode of cytotoxic action and molecular basis of resistance. Oncogene 2003, 22, 7265–7279. [Google Scholar] [CrossRef] [PubMed]
- Park, B.H.; Lim, J.E.; Jeon, H.G.; Seo, S., II; Lee, H.M.; Choi, H.Y.; Jeon, S.S.; Jeong, B.C. Curcumin potentiates antitumor activity of cisplatin in bladder cancer cell lines via ROS-mediated activation of ERK1/2. Oncotarget 2016, 7, 63870–63886. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Shen, H.; Yin, X.; Long, L.; Xie, C.; Liu, Y.; Hui, L.; Lin, X.; Fang, Y.; Cao, Y.; et al. MiR-186 regulation of Twist1 and ovarian cancer sensitivity to cisplatin. Oncogene 2016, 35, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Hong, S.-W.; Kim, S.; Kim, D.; Hur, D.Y.; Jin, D.H.; Kim, B.; Kim, Y.S. Cyclooxygenase-2 expression is induced by celecoxib treatment in lung cancer cells and is transferred to neighbor cells via exosomes. Int. J. Oncol. 2018, 52, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S.D.; McMurray, J.J.; Pfeffer, M.A.; Wittes, J.; Fowler, R.; Finn, P.; Anderson, W.F.; Zauber, A.; Hawk, E.; Bertagnolli, M.; et al. Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. N. Engl. J. Med. 2005, 352, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Lev-Ari, S.; Strier, L.; Kazanov, D.; Madar-Shapiro, L.; Dvory-Sobol, H.; Pinchuk, I.; Marian, B.; Lichtenberg, D.; Arber, N. Celecoxib and curcumin synergistically inhibit the growth of colorectal cancer cells. Clin. Cancer Res. 2005, 11, 6738–6744. [Google Scholar] [CrossRef] [PubMed]
- Waghela, B.N.; Sharma, A.; Dhumale, S.; Pandey, S.M.; Pathak, C. Curcumin conjugated with PLGA potentiates sustainability, anti-proliferative activity and apoptosis in human colon carcinoma cells. PLoS ONE 2015, 10, e0117526. [Google Scholar] [CrossRef] [PubMed]
- Shankar, T.B.; Shantha, N.V.; Ramesh, H.P.; Murthy, I.A.; Murthy, V.S. Toxicity studies on turmeric (Curcuma longa): Acute toxicity studies in rats, guineapigs and monkeys. Indian J. Exp. Biol. 1980, 18, 73–75. [Google Scholar] [PubMed]
- Aggarwal, B.B.; Harikumar, K.B. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int. J. Biochem. Cell Biol. 2009, 41, 40–59. [Google Scholar] [CrossRef] [PubMed]
- Bayet-Robert, M.; Kwiatowski, F.; Leheurteur, M.; Gachon, F.; Planchat, E.; Abrial, C.; Mouret-Reynier, M.A.; Durando, X.; Barthomeuf, C.; Chollet, P. Phase I dose escalation trial of docetaxel plus curcumin in patients with advanced and metastatic breast cancer. Cancer Biol. Ther. 2010, 9, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Epelbaum, R.; Schaffer, M.; Vizel, B.; Badmaev, V.; Bar-Sela, G. Curcumin and gemcitabine in patients with advanced pancreatic cancer. Nutr. Cancer 2010, 62, 1137–1141. [Google Scholar] [CrossRef] [PubMed]
- Ghalaut, V.S.; Sangwan, L.; Dahiya, K.; Ghalaut, P.S.; Dhankhar, R.; Saharan, R. Effect of imatinib therapy with and without turmeric powder on nitric oxide levels in chronic myeloid leukemia. J. Oncol. Pharm. Pract. 2012, 18, 186–190. [Google Scholar] [CrossRef]
- Kanai, M.; Yoshimura, K.; Asada, M.; Imaizumi, A.; Suzuki, C.; Matsumoto, S.; Nishimura, T.; Mori, Y.; Masui, T.; Kawaguchi, Y.; et al. A phase I/II study of gemcitabine-based chemotherapy plus curcumin for patients with gemcitabine-resistant pancreatic cancer. Cancer Chemother. Pharmacol. 2011, 68, 157–164. [Google Scholar] [CrossRef] [PubMed]
- James, M.I.; Iwuji, C.; Irving, G.; Karmokar, A.; Higgins, J.A.; Griffin-Teal, N.; Thomas, A.; Greaves, P.; Cai, H.; Patel, S.R.; et al. Curcumin inhibits cancer stem cell phenotypes in ex vivo models of colorectal liver metastases, and is clinically safe and tolerable in combination with FOLFOX chemotherapy. Cancer Lett. 2015, 364, 135–141. [Google Scholar] [CrossRef]
- Pastorelli, D.; Fabricio, A.S.C.; Giovanis, P.; D’lppolito, S.; Fiduccia, P.; Soldà, C.; Buda, A.; Sperti, C.; Bardini, R.; Da Dalt, G.; et al. Phytosome complex of curcumin as complementary therapy of advanced pancreatic cancer improves safety and efficacy of gemcitabine: Results of a prospective phase II trial. Pharmacol. Res. 2018, 132, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Desai, P.; Ann, D.; Wang, J.; Prabhu, S. Pancreatic cancer: Recent advances in nanoformulation-based therapies. Crit. Rev. Ther. Drug Carr. Syst. 2019, 36, 59–91. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Chan, C.; Lin, W. Nanoparticle-mediated immunogenic cell death enables and potentiates cancer immunotherapy. Angew. Chem. Int. Ed. Engl. 2019, 58, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Liu, X.; Zhang, C.; Zeng, X. Food macromolecule based nanodelivery systems for enhancing the bioavailability of polyphenols. J. Food Drug Anal. 2017, 25, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Rachmawati, H.; Pradana, A.T.; Safitri, D.; Adnyana, I.K. Multiple functions of dalpha-tocopherol polyethylene glycol 1000 succinate (TPGS) as curcumin nanoparticle stabilizer: In vivo kinetic profile and anti-ulcerative colitis analysis in animal model. Pharmaceutics 2017, 9, 24. [Google Scholar] [CrossRef] [PubMed]
- Rachmawati, H.; Safitri, D.; Pradana, A.T.; Adnyana, I.K. TPGS-stabilized curcumin nanoparticles exhibit superior effect on carrageenan-induced inflammation in wistar rat. Pharmaceutics 2016, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Esmatabadi, M.J.D.; Motamedrad, M.; Sadeghizadeh, M. Down-regulation of lncRNA, GAS5 decreases chemotherapeutic effect of dendrosomal curcumin (DNC) in breast cancer cells. Phytomedicine 2018, 42, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Guo, Y.; Song, Y.; Shang, C. Long noncoding RNA GAS5 inhibits malignant proliferation and chemotherapy resistance to doxorubicin in bladder transitional cell carcinoma. Cancer Chemother. Pharmacol. 2017, 79, 49–55. [Google Scholar] [CrossRef]
- Greil, R.; Greil-Ressler, S.; Weiss, L.; Schönlieb, C.; Magnes, T.; Radl, B.; Bolder, G.T.; Vcelar, B.; Sordillo, P.P. A phase 1 dose-escalation study on the safety, tolerability and activity of liposomal curcumin (Lipocurc™) in patients with locally advanced or metastatic cancer. Cancer Chemother. Pharmacol. 2018, 82, 695–706. [Google Scholar] [CrossRef]
- Ma, W.; Guo, Q.; Li, Y.; Wang, X.; Wang, J.; Tu, P. Co-assembly of doxorubicin and curcumin targeted micelles for synergistic delivery and improving anti-tumor efficacy. Eur. J. Pharm. Biopharm. 2017, 112, 209–223. [Google Scholar] [CrossRef]
- Li, P.-Y.; Lai, P.-S.; Hung, W.-C.; Syu, W.-J. Poly (l-lactide)-vitamin E TPGS nanoparticles enhanced the cytotoxicity of doxorubicin in drug-resistant MCF-7 breast cancer cells. Biomacromolecules 2010, 11, 2576–2582. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Si, X.; Han, M.K.; Viennois, E.; Zhang, M.; Merlin, D. Co-delivery of camptothecin and curcumin by cationic polymeric nanoparticles for synergistic colon cancer combination chemotherapy. J. Mater. Chem. B Mater. Biol. Med. 2015, 3, 7724–7733. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.; Phung, C.D.; Thapa, R.K.; Pham, T.T.; Tran, T.H.; Jeong, J.H.; Ku, S.K.; Choi, H.G.; Yong, C.S.; Kim, J.O. Multifunctional nanoparticles as somatostatin receptor-targeting delivery system of polyaniline and methotrexate for combined chemo-photothermal therapy. Acta Biomater. 2018, 68, 154–167. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.-F.; Liang, W.-C.; Feng, L.; Pang, J.X.; Waye, M.M.; Zhang, J.F.; Fu, W.M. H19 mediates methotrexate resistance in colorectal cancer through activating Wnt/β-catenin pathway. Exp. Cell Res. 2017, 350, 312–317. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, M.; Safa, K.D.; Salehi, R. Co-delivery of doxorubicin and methotrexate by dendritic chitosan-g-mPEG as a magnetic nanocarrier for multi-drug delivery in combination chemotherapy. Polym. Chem. 2017, 8, 7333–7350. [Google Scholar] [CrossRef]
- Zhao, X.; Chen, Q.W.; Liu, W.; Li, Y.; Tang, H.; Liu, X.; Yang, X. Codelivery of doxorubicin and curcumin with lipid nanoparticles results in improved efficacy of chemotherapy in liver cancer. Int. J. Nanomed. 2015, 10, 257–270. [Google Scholar] [CrossRef]
- Ruttala, H.B.; Ko, Y.T. Liposomal co-delivery of curcumin and albumin/paclitaxel nanoparticle for enhanced synergistic antitumor efficacy. Colloids Surf. B Biointerfaces 2015, 128, 419–426. [Google Scholar] [CrossRef]
- Cui, T.; Zhang, S.; Sun, H. Co-delivery of doxorubicin and pH-sensitive curcumin prodrug by transferrin-targeted nanoparticles for breast cancer treatment. Oncol. Rep. 2017, 37, 1253–1260. [Google Scholar] [CrossRef]
- Sesarman, A.; Tefas, L.; Sylvester, B.; Licarete, E.; Rauca, V.; Luput, L.; Patras, L.; Banciu, M.; Porfire, A. Anti-angiogenic and anti-inflammatory effects of long-circulating liposomes co-encapsulating curcumin and doxorubicin on C26 murine colon cancer cells. Pharmacol. Rep. 2018, 70, 331–339. [Google Scholar] [CrossRef]
- Motevalli, S.M.; Eltahan, A.S.; Liu, L.; Magrini, A.; Rosato, N.; Guo, W.; Bottini, M.; Liang, X.-J. Co-encapsulation of curcumin and doxorubicin in albumin nanoparticles blocks the adaptive treatment tolerance of cancer cells. Biophys. Rep. 2019, 5, 19–30. [Google Scholar] [CrossRef]
- Ni, W.; Li, Z.; Liu, Z.; Ji, Y.; Wu, L.; Sun, S.; Jian, X.; Gao, X. Dual-targeting nanoparticles: Codelivery of curcumin and 5-fluorouracil for synergistic treatment of hepatocarcinoma. J. Pharm. Sci. 2019, 108, 1284–1295. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Setua, S.; Kumari, S.; Dan, N.; Massey, A.; Hafeez, B.B.; Yallapu, M.M.; Stiles, Z.E.; Alabkaa, A.; Yue, J.; et al. Superparamagnetic iron oxide nanoparticles of curcumin enhance gemcitabine therapeutic response in pancreatic cancer. Biomaterials 2019, 208, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Chen, Q.; Li, Y.; Tang, H.; Liu, W.; Yang, X. Doxorubicin and curcumin co-delivery by lipid nanoparticles for enhanced treatment of diethylnitrosamine-induced hepatocellular carcinoma in mice. Eur. J. Pharm. Biopharm. 2015, 93, 27–36. [Google Scholar] [CrossRef] [PubMed]
| Types of Cancer | Treatment | Participants | Findings | References |
|---|---|---|---|---|
| Advanced or metastatic breast cancer | Curcumin (500 mg/day) and escalated until a dose-limiting toxicity + docetaxel (100 mg/m2) for 7 days every 3 weeks | 14 patients | Improves biological and clinical responses | [109] |
| Pancreatic cancer | Curcumin (8000 mg/day) + gemcitabine (1000 mg/m2) weekly | 17 patients | Time to tumor progression was 1-12 months and overall survival was 1-24 months | [110] |
| Chronic myeloid leukemia | Imatinib (400 mg twice a day for 6 weeks) Group B [Turmeric powder (5 g three times/day) + imatinib (400 mg twice a day)] for 6 weeks | 50 patients | The suppressive effect of nitric oxide levels was noted at Group B | [111] |
| Pancreatic cancer | Curcumin (1000 mg/day) + gemcitabine (1000 mg/m2 on day 1 and 8) and 60 mg/m2 of S-1 orally for 14 consecutive days every 3 weeks | 21 patients | Median survival time after initiation of curcumin was 161 days and 1-year survival rate was 19% | [112] |
| Colorectal liver metastases | 5 µM curcumin + 2 µM oxaliplatin + 5 µM 5-FU | 12 patients | Curcumin enhanced the FOLFOX-based chemotherapy | [113] |
| Pancreatic cancer | Curcumin (2000 mg/die continuously (4 capsules, each of 500 mg, every day) + gemcitabine (10 mg/m2) | 44 patients (13 locally advanced and 31 metastatic) | Median progression-free survival and overall survival were 8.4 and 10.2 months, respectively | [114] |
| Types of Cancer | Co-Delivery System | Treatment | Cancer Cell Lines | Findings | References |
|---|---|---|---|---|---|
| Colon cancer | Cationic polymeric nanoparticles | Camptothecin and curcumin | Colon-26 cells | Enhances synergistic effects of anticancer activity | [125] |
| Liver cancer | Lipid nanoparticle | Curcumin and doxorubicin | HepG2 cells | Enhance cytotoxicity and decrease inhibitory concentration in HepG2 cells | [129] |
| Breast cancer | Liposomal | Paclitaxel and curcumin | MCF-7 cell lines | Effectively kills the cancer cells compared to individual treatment | [130] |
| Breast cancer | Dendritic chitosan grafted methoxy polyethylene glycol (mPEG) coated magnetic nanoparticles | Doxorubicin and methotrexate | MCF7 cell lines | Enhances synergistic effects and alleviates adverse outcome | [128] |
| Breast cancer | Transferrin-decorated nanoparticles | Curcumin and doxorubicin | MCF-7 cells | Combination of transferrin-poly(ethylene glycol)-curcumin (Tf-PEG-CUR) and doxorubicin nanoparticle exhibited higher cytotoxicity in MCF-7 cells compared with Tf-PEG-CUR nanoparticle alone | [131] |
| Colon cancer | PEGylated long-circulating liposomes | Curcumin and doxorubicin | C26 murine colon cancer cells | Exerts strong antiproliferative effects via inhibition of the angiogenic/inflammatory proteins such as TNF-α, TIMP-2, and IL-6 | [132] |
| Breast cancer | Albumin nanoparticles | Curcumin and doxorubicin | MCF-7 resistant breast cancer cells | Shows lower viability than the cells treated with a combination of curcumin and albumin nanoparticle or combination of doxorubicin and albumin nanoparticle | [133] |
| Liver cancer | Biotin-/lactobionic acid–modified poly(ethylene glycol)-poly(lactic-co-glycolic acid)-poly(ethylene glycol) (BLPP) copolymer | Curcumin and 5-FU | HepG2 cells | Exhibits higher cellular uptake, strong cytotoxicity for tumor cells | [134] |
| Pancreatic cancer | Iron oxide nanoparticles | Curcumin and gemcitabine | Human pancreatic cancer (HPAF-II and Panc-1) cell lines | Effectively delivers bioactive curcumin to pancreatic cells, simultaneously enhances gemcitabine uptake | [135] |
| Types of Cancer | Co-Delivery System | Treatment | Animal Model | Findings | References |
|---|---|---|---|---|---|
| Lung cancer | Methoxy poly(ethylene glycol)-poly(caprolactone) (MPEG-PCL) micelles | Curcumin and doxorubicin (5 mg/kg) intravenous tail injection every 5 days until the control mice became weak | The female C57 mice (aged 6-8 weeks) (n = 40) mice were injected subcutaneously with 100 µL of LL/2 cell suspension (1 × 106) into the right flank. The tumor-bearing mice were randomly divided when the mean tumor diameter reached about 6 mm | 1. Tumors in the groups treated with curcumin and doxorubicin/MPEG-PCL were smaller than those receiving the other treatments (p < 0.05) 2. Inhibited the growth of subcutaneous LL/2 lung carcinoma (p < 0.05) 3. Induced apoptosis of tumor tissue and inhibited tumor angiogenesis, as shown in TUNEL assay and CD31 staining (p < 0.05) | [93] |
| Liver cancer | Lipid nanoparticle | Curcumin and doxorubicin (2 mg/kg doxorubicin) for 20 weeks, intravenously injected once a week | Diethylnitrosamine-induced hepatocellular carcinoma mice (n = 32) 24 mice were administrated by oral administration of diethylnitrosamine solution in sesame oil (0.1 g/mL) at 40 mg/kg once a week for 15 weeks. 8 mice were administrated with sesame oil only (normal mice) | 1. The liver/body weight (p < 0.05) and serum ALT and AST levels (p < 0.01) were significantly decreased in curcumin and doxorubicin-lipid nanoparticle group 2. The mRNA and protein levels of Bax/Bcl-2 (p < 0.01) were all increased in tumor tissue from curcumin and doxorubicin-lipid nanoparticle group compared to the doxorubicin-lipid nanoparticle group 3. The immunohistochemistry analysis induced expression of caspase-3 4. The expression of c-myc and PCNA decreased significantly in curcumin and doxorubicin-lipid nanoparticle (p < 0.01) compared to the control group 5. The mRNA and protein levels of VEGF in curcumin and doxorubicin-lipid nanoparticle were significantly decreased compared to the control group (p < 0.05) 6. The mRNA levels of MDR1 and Bcl-2, as well as the protein levels of P-gp and Bcl-2 were all decreased in curcumin and doxorubicin-lipid nanoparticle group compared to the doxorubicin-lipid nanoparticle group (p < 0.01) | [136] |
| Breast cancer | Polymeric micelles | Curcumin (10 mg/kg) and doxorubicin (10 mg/kg) for 12 days | The female BALB/c mice (n = 60) were injected with 1 × 106 4T1 cells into the right axilla of mice. When the tumor reached about 100 mm3, 4T1 tumor-bearing mice were randomly divided into 6 groups (n = 10) | 1. Curcumin and doxorubicin polymeric micelles treated group exhibited a considerable tumor inhibition compared to the saline-treated group (p < 0.001) 2. The AST, LDH, CK, and CKMB were significantly reduced compared to the mice treated with doxorubicin polymeric micelles (p < 0.05) 3. No pathological damages were found in heart, liver, spleen, lung, and kidney in the mice treated with curcumin and doxorubicin polymeric micelles by using H&E staining 4. Tumors treated with curcumin and doxorubicin polymeric micelles had enhanced dark brown spots by using TUNEL assay, indicating that drug encapsulated in micelles could enhance tumor cell apoptosis and showed better antitumor effects | [123] |
| Breast cancer | Transferrin-poly(ethylene glycol) | Curcumin (50 mg/kg) and doxorubicin (50 mg/kg) were injected into the mice by tail vein for 7 weeks | BALB/c mice were inoculated subcutaneously with 1 × 106 MCF-7 cells. MCF-7 tumor xenografts were grown in BALB/c mice and estrogen was provided as a β-estradiol pellet 1 week prior to the injection of the cells. The tumors were allowed to develop on the posterolateral side of the mice for 1 week prior to treatment to obtain the breast cancer-bearing animal model. The mice were randomly divided into 6 groups | Compared with curcumin and doxorubicin, transferrin-poly(ethylene glycol)-curcumin/doxorubicin nanoparticles presented a remarkably higher inhibition effect towards tumor growth (p < 0.05) | [131] |
| Liver cancer | Biotin-/lactobionic acid–modified poly(ethylene glycol)-poly(lactic-co-glycolic acid)-poly(ethylene glycol) (BLPP) copolymer | Curcumin (10 mg/kg) and 5-FU (4 mg/kg). The mice were injected once at an interval of 2 days of a total of 4 injections through the tail vein for 30 days | The BALB/c nude mice (n = 18) were inoculated subcutaneously with HepG2 cells (2 × 106). The mice were randomly divided into 6 groups (n = 3) when the tumor volume reached about 50 mm3 | 1. The tumors in BLPP/curcumin +5-FU nanoparticle were approximately 8 times smaller than the tumor volume observed in the control (phosphate-buffered saline) group (p < 0.001) 2. The mice treated with BLPP/curcumin +5-FU nanoparticles induced tumor apoptosis or necrosis significantly compared to the BLPP/curcumin nanoparticle (p < 0.05) 3. The western blotting analysis showed that BLPP/curcumin + 5-FU nanoparticle significantly decreases the DPYD expression compared to the BLPP/5-FU nanoparticle and the control groups (p < 0.05) 4. p53 protein expression was higher in BLPP/curcumin groups than in BLPP/5-FU nanoparticle group (p < 0.05) 5. Bcl-2 protein expression of BLPP/curcumin + 5-FU was lower than that of the BLPP/5-FU nanoparticle, BLPP/curcumin nanoparticle, and control groups; while the expression of cytochrome c was higher (p < 0.05) | [134] |
| Pancreatic cancer | Superparamagnetic iron oxide nanoparticle (SPION) formulation of curcumin (SP-CUR) | 2 treatments groups [Curcumin (100 µg dissolved in 100 μL of 0.1% Tween 20) and gemcitabine (300 μg dissolved in 50 μL of phosphate-buffered saline)] and another two groups were treated with an intraperitoneal injection of 100 μg curcumin loaded SP-CUR and combination with gemcitabine, respectively. Treatments were administered twice weekly for 7 weeks | HPAF-II cells (1.0 × 106) and human pancreatic stromal cells (stromal component; 0.5 × 106) were suspended in 50 μL of HBSS media containing 1% (v/v) matrigel and injected into the parenchyma of the pancreas in old male athymic nude mice (6 weeks old). Five days later, mice were randomly divided into five groups (n = 8) | 1. The bioluminescence imaging results showed a significant (p < 0.05) decrease in the pancreatic tumor volume of SP-CUR + gemcitabine-treated mice compared to the vehicle-treated mice 2. SP-CUR + gemcitabine-treated mice showed a significant decrease in the tumor weight of pancreas compared to the gemcitabine alone (p < 0.0001) 3. None of the mice treated with SP-CUR + gemcitabine were recorded for distant metastasis 4. Immunoblotting and immunohistochemistry analyses showed that SP-CUR + gemcitabine inhibited SHH, NF-қB, Gli-1 and Gli-2 expression 5. SP-CUR + gemcitabine reduced the amount of α-SMA, N-cadherin, and SMO and upregulated hCNT | [135] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, B.L.; Norhaizan, M.E. Curcumin Combination Chemotherapy: The Implication and Efficacy in Cancer. Molecules 2019, 24, 2527. https://doi.org/10.3390/molecules24142527
Tan BL, Norhaizan ME. Curcumin Combination Chemotherapy: The Implication and Efficacy in Cancer. Molecules. 2019; 24(14):2527. https://doi.org/10.3390/molecules24142527
Chicago/Turabian StyleTan, Bee Ling, and Mohd Esa Norhaizan. 2019. "Curcumin Combination Chemotherapy: The Implication and Efficacy in Cancer" Molecules 24, no. 14: 2527. https://doi.org/10.3390/molecules24142527
APA StyleTan, B. L., & Norhaizan, M. E. (2019). Curcumin Combination Chemotherapy: The Implication and Efficacy in Cancer. Molecules, 24(14), 2527. https://doi.org/10.3390/molecules24142527






