Analysis of Chemical Composition of Extractives by Acetone and the Chromatic Aberration of Teak (Tectona Grandis L.F.) from China
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Determination of Chemical Composition
2.3. Total Phenol Content
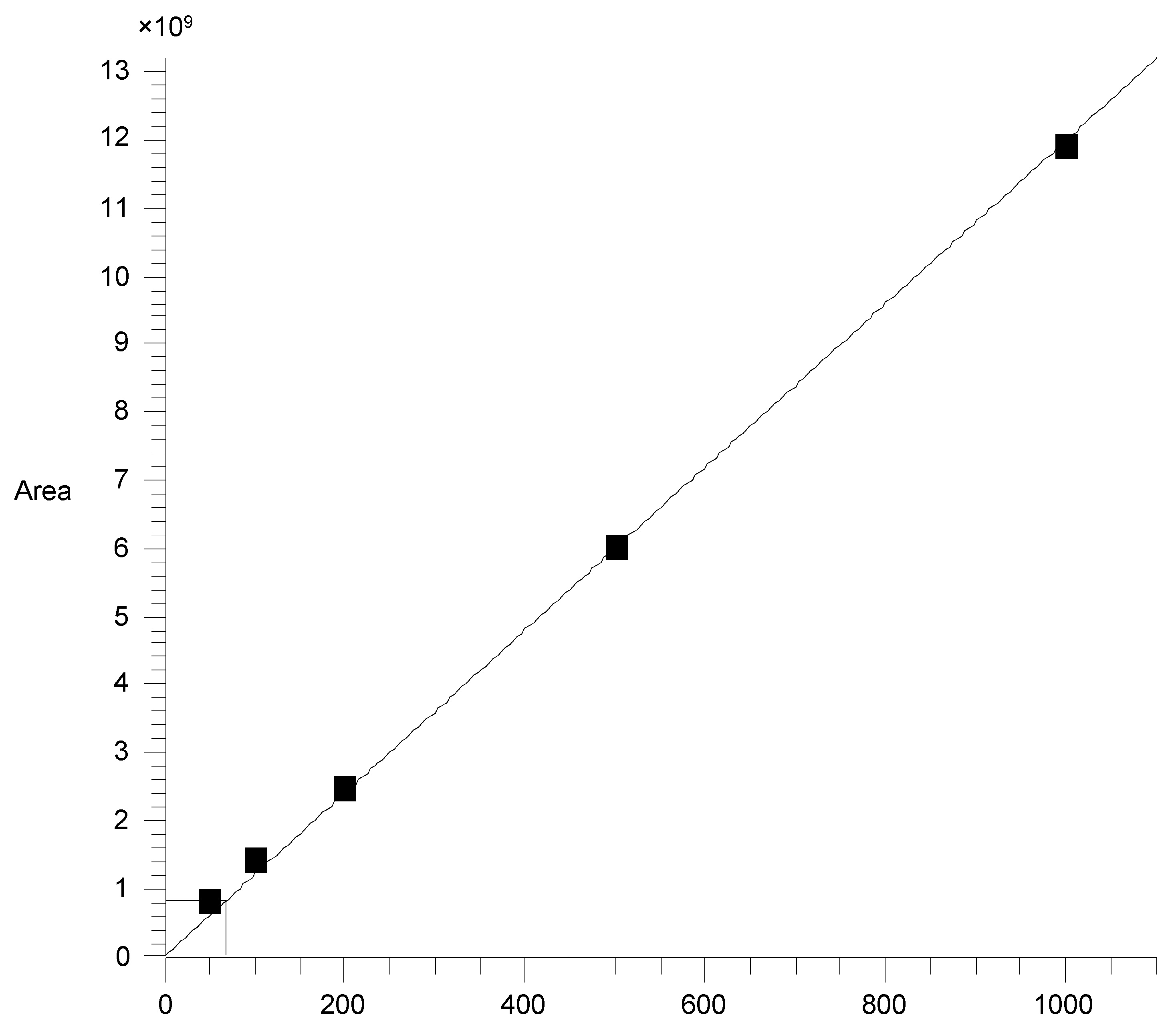
2.4. GC-MS Analysis
3. Results and Discussion
3.1. Components Analysis in Sapwood and Heartwood of Teak
3.2. GC–MS Analysis of Acetone Extractives
3.3. Discussion in the Chromatic Aberration Between Heartwood and Sapwood
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tanaka, N.; Hamazaki, T.; Vacharangkura, T. Distribution, growth and site requirements of teak [Tectona grandis]. Jpn. Agric. Res. Q. 1998, 32, 65–77. [Google Scholar]
- Francis, J.K.; Vozzo, J.A. Tectona grandis L.f. In Tropical Tree Seed Manual; USDA Forest Service: Washington, DC, USA, 2002; pp. 745–746. [Google Scholar]
- Lian, C.P.; Pan, B.; Wang, F.; Tang, J.; Yin, W.X. Research on Teak Light-induced Discoloration and Mechanism. For. Mach. Wood Eq. 2015, 43, 21–24. [Google Scholar]
- Nair, K.R.; Mukerji, H.K. A statistical study of the variability of physical and mechanical properties of Tectona grandis (Teak) grown at different localities of India and Burma and the effects of the variability on the choice of the sampling plan. Indian For. Rec. 1960, 1, 49. [Google Scholar]
- Ponneth, D.; Vasu, A.E.; Easwaran, J.C.; Mohandass, A.; Chauhan, S.S. Destructive and non-destructive evaluation of seven hardwoods and analysis of data correlation. Holzforschung 2014, 68, 951–956. [Google Scholar] [CrossRef]
- Seviset, S.; Piromgran, T.; Saributr, U.; Porncharoen, R.; Raerai, K.; Charoensettasilp, S. Mechanical Property of 9 Years Old Thinning of Teak Plantation in Thailand. MATEC Web Conf. 2017, 95, 03004. [Google Scholar] [CrossRef]
- Rizanti, D.E.; Darmawan, W.; George, B.; Merlin, A.; Dumarcay, S.; Chapuis, H.; Gérardin, C.; Gelhaye, E.; Raharivelomanana, P.; Sari, R.K. Comparison of teak wood properties according to forest management: Short versus long rotation. Ann. For. Sci. 2018, 75, 39. [Google Scholar] [CrossRef]
- Huang, G.; Liang, K.; Zhou, Z.; Yang, G.; Muralidharan, E.M. Variation in Photosynthetic Traits and Correlation with Growth in Teak (Tectona grandis Linn.) Clones. Forests 2019, 10, 44. [Google Scholar] [CrossRef]
- Guzmán, N.; Moya, R.; Murillo, O. Evaluation of Bent Trees in Juvenile Teak (Tectona grandis L.f.) Plantations in Costa Rica: Effects on Tree Morphology and Wood Properties. Forests 2017, 8, 79. [Google Scholar] [CrossRef]
- Zhou, Z.Z.; Liu, S.C.; Liang, K.N.; Ma, H.M.; Huang, G.H. Growth and mineral nutrient analysis of teak (Tectona grandis) grown on acidic soils in south China. J. Forest. Res. 2017, 28, 503–511. [Google Scholar] [CrossRef]
- Yin, X.; Huang, A.; Zhang, S.; Liu, R.; Ma, F. Identification of Three Dalbergia Species Based on Differences in Extractive Components. Molecules 2018, 23, 2163. [Google Scholar] [CrossRef]
- Gardini, C.; Urso, E.; Guerrini, M.; Van Herpen, R.; De Wit, P.; Naggi, A. Characterization of Danaparoid Complex Extractive Drug by an Orthogonal Analytical Approach. Molecules 2017, 22, 1116. [Google Scholar] [CrossRef]
- Musumeci, L.E.; Ryona, I.; Pan, B.S.; Loscos, N.; Feng, H.; Cleary, M.T.; Sacks, G.L. Quantification of Polyfunctional Thiols in Wine by HS-SPME-GC-MS Following Extractive Alkylation. Molecules 2015, 20, 12280–12299. [Google Scholar] [CrossRef]
- Belt, T.; Hänninen, T.; Rautkari, L. Antioxidant activity of Scots pine heartwood and knot extractives and implications for resistance to brown rot. Holzforschung 2017, 71, 527–534. [Google Scholar] [CrossRef]
- Kallbom, S.; Moghaddam, M.; Walinder, M. Liquid sorption, swelling and surface energy properties of unmodified and thermally modified Scots pine heartwood after extraction. Holzforschung 2018, 72, 251–258. [Google Scholar] [CrossRef]
- Laskowska, A.; Sobczak, J.W. Surface chemical composition and roughness as factors affecting the wettability of thermo-mechanically modified oak (Quercus robur L.). Holzforschung 2018, 72, 993–1000. [Google Scholar] [CrossRef]
- Hisamochi, R.; Watanabe, Y.; Sano, M.; Nakatsuka, T.; Kurita, N.; Matsuo-Ueda, M.; Yamamoto, H.; Tazuru, S.; Sugiyama, J.; Subiyanto, B. Cellulose oxygen isotopic composition of teak (Tectona grandis) collected from Java Island: A tool for dendrochronological and dendroclimatological analysis. Dendrochronologia 2018, 52, 80–86. [Google Scholar] [CrossRef]
- Brocco, V.F.; Paes, J.B.; Costa, L.G.A.D.; Brazolin, S.; Arantes, M.D.C. Potential of teak heartwood extracts as a natural wood preservative. J. Clean. Prod. 2017, 142, 2039–2099. [Google Scholar] [CrossRef]
- Leonardo, F.V.S.; Rocha, H.F.; Mendoza, Z.M.S.H. Chemical compounds in teak. Pesqui. Florest. Bras. 2015, 35, 315–322. [Google Scholar]
- Lukmandaru, G.; Ashitani, T. Color and chemical characterization of partially black-streaked heartwood in teak (Tectona grandis). J. Forest. Res. 2009, 20, 377. [Google Scholar] [CrossRef]
- Nakano, J.; Takama, H.; Masano, J.; Ishizu, A. Wood Chemistry; Bao, H.; Li, Z.Z., Translators; China Forestry Publishing House: Beijing, China, 1989. [Google Scholar]
- Shi, S.; He, F. Analysis and Detection of Pulp and Paper Making; China Light Industry Press: Beijing, China, 2009. [Google Scholar]
- Vieitez, I.; Maceiras, L.; Jachmanián, I.; Alborés, S. Antioxidant and antibacterial activity of different extracts from herbs obtained by maceration or supercritical technology. J. Supercrit. Fluids 2018, 133, 58–64. [Google Scholar] [CrossRef]
- Walker, J.C.F. Primary Wood Processing: Principles and Practice; Chapman & Hall: London, UK, 1993. [Google Scholar]
- Standard Test Method for Preparation of Extractive-Free Wood; ASTM International: West Conshohocken, PA, USA, 2001.
- Ono, K.; Hiraide, M.; Amari, M. Determination of lignin, holocellulose, and organic solvent extractives in fresh leaf, litterfall, and organic material on forest floor using near-infrared reflectance spectroscopy. J. Forest. Res. 2003, 8, 191–198. [Google Scholar] [CrossRef]
- Chen, F.S.; Yuan, S.S. Research progress on the removal of acetone extract from plant raw materials. Hunan Papermak. 2015, 2, 29–31. [Google Scholar]
- Liu, Y.X.; Zhao, G.J. Wood Science; China Forestry Publishing House: Beijing, China, 2012. [Google Scholar]
- Deng, Z. Development of an alternative solvent to replace benzene in the determination of organic soluble extractives in wood. World Pulp Pap. 2006, 25, 32–34. [Google Scholar]
- Lukmandaru, G.; Takahashi, K. Radial distribution of quinones in plantation teak (Tectona grandis L.f.). Ann. For. Sci. 2009, 66, 605. [Google Scholar] [CrossRef]
- Windeisen, E.; Klassen, A.; Wegener, G. On the chemical characterisation of plantation teakwood from Panama. Holz als Roh- und Werkstoff 2003, 61, 416–418. [Google Scholar] [CrossRef]
- Tolvaj, L.; Faix, O. Artificial Ageing of Wood Monitored by DRIFT Spectroscopy and CIE L*a*b* Color Measurements. 1. Effect of UV Light. Holzforschung 1995, 49, 397–404. [Google Scholar] [CrossRef]
- Sehlstedt-persson, M. Colour responses to heat-treatment of extractives and sap from pine and spruce. In Proceedings of the 8th International IUFRO Wood Drying Conference, Brasov, Romania, 24–29 August 2003; pp. 459–464. [Google Scholar]
- Johansson, C.I.; Saddler, J.N.; Beatson, R.P. Characterization of the polyphenolics related to the colour of western red cedar (Thuja plicata Donn) heartwood. Holzforschung 2000, 54, 246–254. [Google Scholar] [CrossRef]
- Li, H.; Zeng, D.; Huang, Z.Z. Analysis of extractive compounds in teakwood before and after irradiation by chromatography-mass spectrometry. J. Central South. Univ. Forest. Technol. 2018, 38, 122–128. [Google Scholar]

| Experiment Reagent | Manufacturers |
|---|---|
| Distilled water | Guangzhou Watson’s Food and Beverage Co., Ltd., Guangzhou, China |
| Sodium hydroxide (analytical grade) | Beijing Chemical Plant, Beijing, China |
| Sulfuric acid (analytical grade) | Beijing Chemical Plant, Beijing, China |
| Benzene | Tianjin Fuchen Chemical Reagent Factory, Tianjin, China |
| Methanol (HPLC(high-performance liquid chromatography) grade) | Mreda Technology Inc, California, USA |
| Acetone (HPLC grade) | Duksan Pure Chemicals Co. Ltd., Ansansi Gyunggido, Korea |
| Moisture (%) | Acid-Insoluble Lignin (%) | Holocellulose (%) | α-Cellulose (%) | pH Value | |
|---|---|---|---|---|---|
| Heartwood | 3.90 | 26.76 | 58.27 | 34.97 | 5.79 |
| Sapwood | 4.25 | 27.73 | 62.23 | 35.53 | 5.84 |
| Hot Water (%) | Cold Water (%) | 1% NaOH (%) | Alcohol-benzene (%) | Acetone (%) | Total Phenol (%) | ||
|---|---|---|---|---|---|---|---|
| Hot Water | Cold Water | ||||||
| Heartwood | 10.22 | 6.40 | 22.34 | 14.54 | 10.13 | 1.35 | 0.71 |
| Sapwood | 9.36 | 6.07 | 24.06 | 4.67 | 2.37 | 0.93 | 0.33 |
| NO. | Categories | Retention Time (min) | Retention Index | Component | GC Content (μg/g) | |
|---|---|---|---|---|---|---|
| Heartwood | Sapwood | |||||
| 1 | Alkane | 5.61 | 570 | 2-methyl-pentane | 45.01 | 13.89 |
| 2 | 5.79 | 584 | 3-methyl-pentane | 56.94 | 17.89 | |
| 3 | 6 | 601 | N-hexane | 207.26 | 73.03 | |
| 4 | 6.44 | 630 | Methyl-cyclopentane | 88.56 | 27.99 | |
| 5 | 7.02 | 664 | Cyclohexane | 65.32 | 19.78 | |
| 6 | 7.61 | 691 | Isooctane | 589.54 | 129.64 | |
| 7 | 8.65 | 736 | 2,2,3-trimethylpentane | 7.54 | / | |
| 8 | 9.06 | 630 | 2,3,4-trimethylpentane | 11.72 | 4.00 | |
| 9 | 28.38 | 1360 | 5,8-diethyldodecane | 4.82 | / | |
| 10 | 28.53 | 1387 | 9-n-hexylheptadecane | 5.02 | / | |
| 11 | Olefin | 8.99 | 750 | 2,3-dimethyl-1-hexene | 5.65 | 4.21 |
| 12 | 41.85 | 2835 | All-trans-squalene | 569.65 | 36.83 | |
| 13 | Benzene series | 28.16 | 1323 | 1,3,5-triisopropyl-benzene | 4.61 | / |
| 14 | Alcohol | 33.41 | 2970 | Estriol | 23.66 | 118.49 |
| 15 | Ketones | 10.77 | 811 | Acetonyldimethylcarbinol | 163.09 | 182.46 |
| 16 | 29.7 | 1927 | 6-(1-Hydroxymethylvinyl)-4,8a-dimethyl-3,5,6,7,8,8a-hexahydro-1H-naohthalen-2-one | 13.40 | 2.53 | |
| 17 | 30.19 | 1941 | 2-Acetyl-3-methyl-3-phenyl-2,3-dihudro-5H-indazol-5-one | 145.50 | / | |
| 18 | 31.07 | 1966 | 2,3-Dimethyl-1,4,4a,9a-tetrahydro-9,10-anthracenedione | 1228.70 | 30.31 | |
| 19 | 31.25 | 1971 | Tetracyclo [10.2.1.0(2,11).0(4,9)] petadeca-2(11),6,13-triene-5,8-dione | 107.40 | / | |
| 20 | 31.85 | 1988 | 2-(3-Hydroxyphenyl)-1H-indene-1,3(2H)-dione | 320.73 | 4.42 | |
| 21 | 32.14 | 1966 | 2-Acetyl-3-methyl-3-phenyl-2,3-dihydro-5H-indazol-5-one | 58.41 | / | |
| 22 | 36.46 | 3387 | 2-(2-Nitro-1-phenyl-2-propenyl)cyclohexanone | 266.51 | / | |
| 23 | Aldehyde | 22.17 | 1285 | 1,3-benzodioxole-5-carboxaldehyde (Piperonal) | 20.94 | / |
| 24 | 26.49 | 1640 | (2E)-3-(1,3-benzodioxol-5-yl)-2-propenal | 47.10 | / | |
| 25 | 32.62 | 2679 | 9,10-dioxo-9,10-dihydro-1-anthracenecarbaldehyde | 170.20 | 4.21 | |
| 26 | 33.15 | 2846 | 1-methyl-1,2,3,4,4a,9,10,10a-octahydro-1-phenanthrenecarbaldehyde | 62.81 | / | |
| 27 | Acid | 7.2 | 702 | Propanoic acid | 9.63 | / |
| 28 | 29.17 | 1912 | 1,4-Dihydroxy-3-(3-methyl-2-butenyl)-2-naphthoic acid | 23.66 | 7.16 | |
| 29 | Ester | 8.33 | 726 | Methyl isocyanate | 11.72 | / |
| 30 | 12.7 | 878 | Acrylic acid butyl ester | 3.35 | / | |
| 31 | 28.77 | 1901 | Butyl(2-chlorocyclohexyl) methyl phthalate | 4.82 | 2.95 | |
| 32 | acid anhydride | 21.61 | 1322 | 1,3-isobenzofurandione (Phthalic anhydride) | 3.35 | / |
| 33 | Phenol | 27.31 | 1675 | 4-((1E)-3-hydroxy-1-propenyl)-2-methoxyphenol | 7.33 | / |
| 34 | 29.53 | 1922 | 4-tert-butyl-2-phenyl-phenol | 2675.75 | 23.99 | |
| 35 | 30.62 | 1953 | 4-tert-butyl-2-phenyl-phenol (isomer) | 2230.46 | 21.68 | |
| 36 | Quinone | 23.31 | 1427 | 1,4-naphthoquinone | 20.73 | / |
| 37 | 24.84 | 1469 | Menadione | 15.28 | / | |
| 38 | 30.05 | 1937 | Anthraquinone | 101.54 | 2.10 | |
| 39 | 30.48 | 1949 | 2-hydroxy-3-(3-methyl-2-butenyl)-1,4-Naphthoquinone (Lapachol) | 227.36 | 31.36 | |
| 40 | 31.39 | 1975 | 2-methyl-anthraquinone | 3019.30 | 516.46 | |
| 41 | 32.53 | 2855 | 1-Hydroxy-4-methylanthra-9,10-quinone | 94.21 | 14.73 | |
| 42 | 34.73 | 3064 | 2-(Hydroxymethyl)anthraquinone | 419.96 | 13.05 | |
| 43 | Heterocycle | 26.09 | 1603 | 3,4-methylenedioxybenzhydrazide | 3.56 | / |
| 44 | 26.27 | 1612 | Dibenzo-p-dioxin | 4.61 | / | |
| 45 | 28.3 | 1844 | 5-methoxy-7-phenyl-bicyclo [3.2.0] hept-2-en-6-one, (Z, exo+ endo) | 14.86 | / | |
| 46 | 28.61 | 1876 | 1,2-benzisothiazol-3-amine tbdms | 6.70 | / | |
| 47 | 28.92 | 1905 | 2-Hydroxydibenzofuran | 26.59 | 4.00 | |
| 48 | 30.28 | 1944 | 4a-methyl-1-methylene-1,2,3,4,4a,9,10,10a-octahydrophenanthrene | 50.87 | 45.25 | |
| 49 | 35.79 | 3241 | Lochnerine | 26.80 | / | |
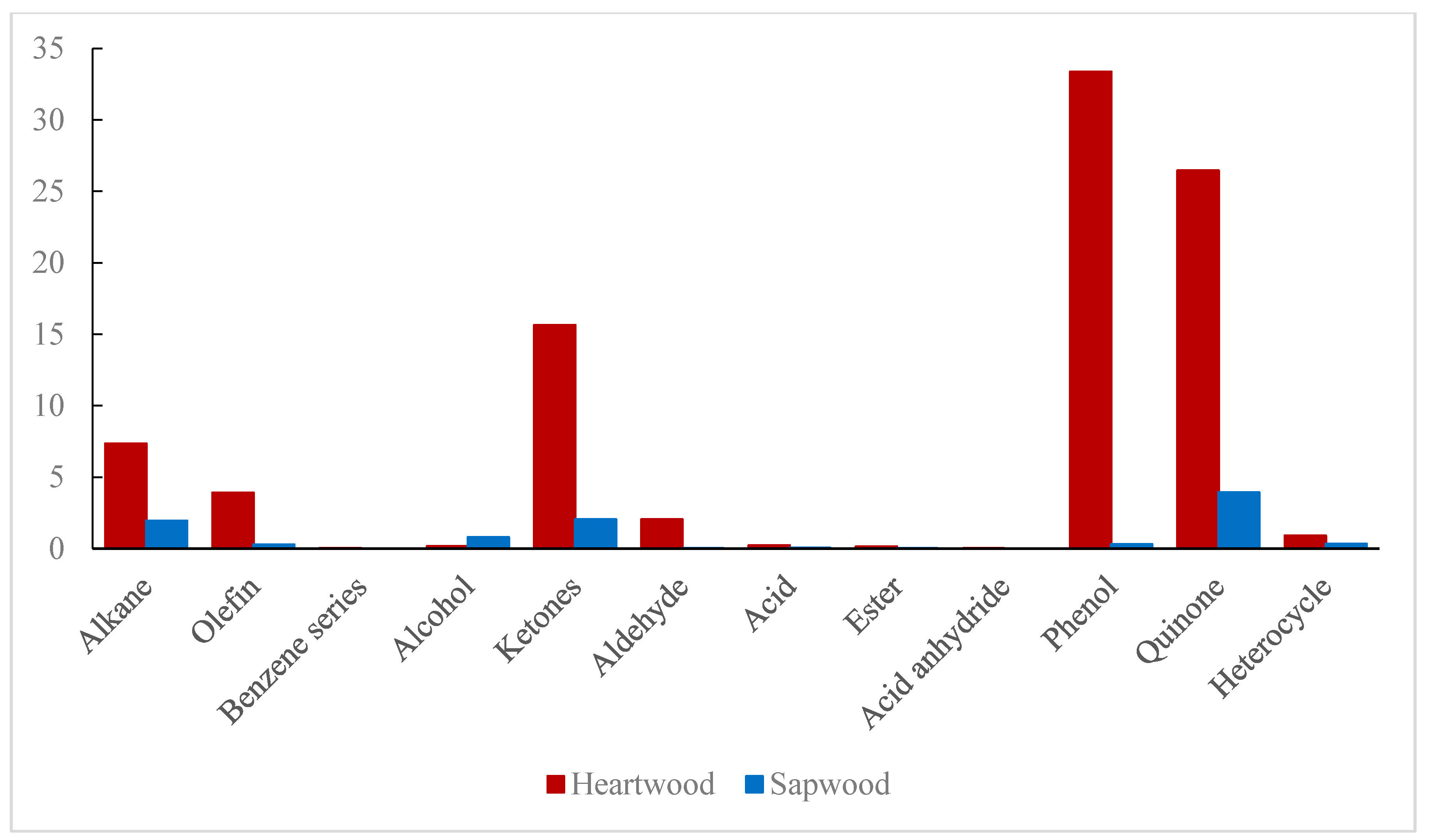
| Heartwood (μg/g) | Sapwood (μg/g) | |
|---|---|---|
| Alkane | 1081.73 | 286.22 |
| Olefin | 575.30 | 41.04 |
| Benzene series | 4.61 | 0.00 |
| Alcohol | 23.66 | 118.49 |
| Ketones | 2303.73 | 301.79 |
| Aldehyde | 301.05 | 4.21 |
| Acid | 33.29 | 7.16 |
| Ester | 19.89 | 2.95 |
| Acid anhydride | 3.35 | 0.00 |
| Phenol | 4913.54 | 45.67 |
| Quinone | 3898.38 | 577.70 |
| Heterocycle | 133.99 | 49.25 |
| Structures | Heartwood ug/g | Sapwood ug/g | |
|---|---|---|---|
| 4-tert-butyl-2-phenyl-phenol |  | 4906.21 | 45.67 |
| 2-methyl-Anthraquinone | 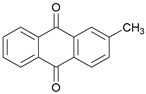 | 3019.30 | 516.46 |
| 2,3-Dimethyl-1,4,4a,9a-tetrahydro-9,10-anthracenedione# | 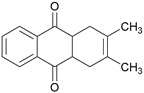 | 1228.70 | 30.31 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, H.; Liu, R.; Long, L. Analysis of Chemical Composition of Extractives by Acetone and the Chromatic Aberration of Teak (Tectona Grandis L.F.) from China. Molecules 2019, 24, 1989. https://doi.org/10.3390/molecules24101989
Qiu H, Liu R, Long L. Analysis of Chemical Composition of Extractives by Acetone and the Chromatic Aberration of Teak (Tectona Grandis L.F.) from China. Molecules. 2019; 24(10):1989. https://doi.org/10.3390/molecules24101989
Chicago/Turabian StyleQiu, Hongyun, Ru Liu, and Ling Long. 2019. "Analysis of Chemical Composition of Extractives by Acetone and the Chromatic Aberration of Teak (Tectona Grandis L.F.) from China" Molecules 24, no. 10: 1989. https://doi.org/10.3390/molecules24101989
APA StyleQiu, H., Liu, R., & Long, L. (2019). Analysis of Chemical Composition of Extractives by Acetone and the Chromatic Aberration of Teak (Tectona Grandis L.F.) from China. Molecules, 24(10), 1989. https://doi.org/10.3390/molecules24101989






