Lamium Plants—A Comprehensive Review on Health Benefits and Biological Activities
Abstract
:1. Introduction
2. A Shot on the Biologically Active Compounds in Lamium Plants
2.1. The Phenolic and Terpenoid Compounds of Lamium Plants
2.2. Essential Oil Constituents of Lamium Plants
3. Traditional Medicinal Uses of Lamium Plants
4. Biological Activities Lamium Plants
4.1. In Vitro Studies
4.1.1. Antioxidant Activity
4.1.2. Antiviral Activity
4.1.3. Antimicrobial Activity
4.1.4. Anti-Inflammatory, Anti-Nociceptive Activity, and Pain Therapy
4.1.5. Cytotoxicity and Cytoprotective Activity
4.1.6. Antityrosinase Activity
4.2. In Vivo Studies
4.3. Clinical Studies
5. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Sen, T.; Samanta, S.K. Medicinal plants, human health and biodiversity: A broad review. Adv. Biochem. Eng. Biotechnol. 2015, 147, 59–110. [Google Scholar] [PubMed]
- Salehi, B.; Stojanović-Radić, Z.; Matejić, J.; Sharopov, F.; Antolak, H.; Kręgiel, D.; Sen, S.; Sharifi-Rad, M.; Acharya, K.; Sharifi-Rad, R.; et al. Plants of genus Mentha: From farm to food factory. Plants 2018, 7, 70. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Ozcelik, B.; Altın, G.; Daşkaya-Dikmen, C.; Martorell, M.; Ramírez-Alarcón, K.; Alarcón-Zapata, P.; Morais-Braga, M.F.B.; Carneiro, J.N.P.; Alves Borges Leal, A.L.; et al. Salvia spp. Plants-from farm to food applications and phytopharmacotherapy. Trends Food Sci. Technol. 2018, 80, 242–263. [Google Scholar] [CrossRef]
- Durazzo, A.; D’Addezio, L.; Camilli, E.; Piccinelli, R.; Turrini, A.; Marletta, L.; Marconi, S.; Lucarini, M.; Lisciani, S.; Gabrielli, P.; et al. From plant compounds to botanicals and back: A current snapshot. Molecules 2018, 23, 1844. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A. Extractable and Non-extractable polyphenols: An overview. In Non-Extractable Polyphenols and Carotenoids: Importance in Human Nutrition and Health; Saura-Calixto, F., Pérez-Jiménez, J., Eds.; Royal Society of Chemistry: London, UK, 2018; pp. 1–37. [Google Scholar]
- Daliu, P.; Santini, A.; Novellino, E. From pharmaceuticals to nutraceuticals: Bridging disease prevention and management. Expert Rev. Clin. Pharmacol. 2019, 12, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; Lucarini, M. A current shot and re-thinking of antioxidant research strategy. Braz. J. Anal. Chem. 2018, 5, 9–11. [Google Scholar] [CrossRef]
- Santini, A.; Tenore, G.C.; Novellino, E. Nutraceuticals: A paradigm of proactive medicine. Eur. J. Pharm. Sci. 2017, 96, 53–61. [Google Scholar] [CrossRef]
- Santini, A.; Novellino, E. Nutraceuticals-Shedding Light on the Grey Area between Pharmaceuticals and Food. Expert. Rev. Clin. Pharmacol. 2018, 11, 545–547. [Google Scholar] [CrossRef]
- Santini, A.; Cammarata, S.M.; Capone, G.; Ianaro, A.; Tenore, G.C.; Pani, L.; Novellino, E. Nutraceuticals: Opening the debate for a regulatory framework. Br. J. Clin. Pharmacol. 2018, 84, 659–672. [Google Scholar] [CrossRef]
- Raja, R.R. Medicinally potential plants of labiatae (lamiaceae) family, an overview. Res. J. Med. Plant. 2012, 6, 203–213. [Google Scholar] [CrossRef]
- The Plant List. Version 1.1. Available online: http://www.theplantlist.org/ (accessed on 29 April 2019).
- Tamokou, J.D.D.; Mbaveng, A.T.; Kuete, V. Antimicrobial activities of african medicinal spices and vegetables. In Medicinal Spices and Vegetables from Africa. Therapeutic Potential against Metabolic, Inflammatory, Infectious and Systemic Diseases; Kuete, V., Ed.; Academic Press: New York, NY, USA, 2017; p. 694. [Google Scholar]
- Lesjak, M.; Simin, N.; Orcic, D.; Franciskovic, M.; Knezevic, P.; Beara, I.; Aleksic, V.; Svircev, E.; Buzas, K.; Mimica-Dukic, N. Binary and tertiary mixtures of Satureja hortensis and Origanum vulgare essential oils as potent antimicrobial agents against Helicobacter pylori. Phytother. Res. 2016, 30, 476–484. [Google Scholar] [CrossRef] [PubMed]
- Waller, S.B.; Cleff, M.B.; Serra, E.F.; Silva, A.L.; Gomes, A.D.; de Mello, J.R.; de Faria, R.O.; Meireles, M.C. Plants from lamiaceae family as source of antifungal molecules in humane and veterinary medicine. Microb. Pathog. 2017, 104, 232–237. [Google Scholar] [CrossRef]
- Ghoneim, M.; Musa, A.; El-Hela, A.; Elokely, K. Evaluation and understanding the molecular basis of the antimethicillin-resistant Staphylococcus aureus activity of secondary metabolites isolated from Lamium amplexicaule. Pharmacogn. Mag. 2018, 14, 3–7. [Google Scholar]
- Bendiksby, M.; Brysting, A.K.; Thorbek, L.; Gussarova, G.; Ryding, O. Molecular phylogeny and taxonomy of the genus Lamium L. (lamiaceae): Disentangling origins of presumed allotetraploids. Taxon 2011, 60, 986–1000. [Google Scholar] [CrossRef]
- Mennema, J. A Taxonomic Revision of Lamium (Lamiaceae); Brill Archive: Leiden, The Netherlands, 1989. [Google Scholar]
- Baran, P.; Özdemdr, C. Morphological, anatomical and cytological studies on endemic Lamium pisidicum. Pak. J. Bot. 2013, 45, 73–85. [Google Scholar]
- Yalcin, F.N.; Kaya, D. Ethnobotany, pharmacology and phytochemistry of the genus Lamium (lamiaceae). FABAD J. Pharm. Sci. 2006, 31, 43–52. [Google Scholar]
- Yordanova, Z.P.; Zhiponova, M.K.; Iakimova, E.T.; Dimitrova, M.A.; Kapchina-Toteva, V.M. Revealing the reviving secret of the white dead nettle (Lamium album L.). Phytochem. Rev. 2014, 13, 375–389. [Google Scholar] [CrossRef]
- Czerwińska, M.E.; Swierczewska, A.; Wozniak, M.; Kiss, A.K. Bioassay-guided iisolation of iridoids and phenylpropanoids from aerial parts of Lamium album and their anti-inflammatory activity in human neutrophils. Planta Med. 2017, 83, 1011–1019. [Google Scholar]
- Zargari, A. Medicinal Plants; Tehran University Publications: Tehran, Iran, 1990; Volume 4. [Google Scholar]
- Matkowski, A.; Tasarz, P.; Szypula, E. Antioxidant activity of herb extracts from five medicinal plants from lamiaceae, subfamily lamioideae. J. Med. Plants Res. 2008, 2, 321–330. [Google Scholar]
- Akkol, E.K.; Yalçin, F.N.; Kaya, D.; Çaliş, I.; Yesilada, E.; Ersöz, T. In vivo anti-inflammatory and antinociceptive actions of some lamium species. J. Ethnopharmacol. 2008, 118, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Yalcin, F.N.; Kaya, D.; Kilic, E.; Ozalp, M.; Erspz, T.; Calis, I. Antimicrobial and free radical scavenging activities of some Lamium species from turkey. Hacet. Univ. J. Fac. Pharm. 2007, 27, 11–22. [Google Scholar]
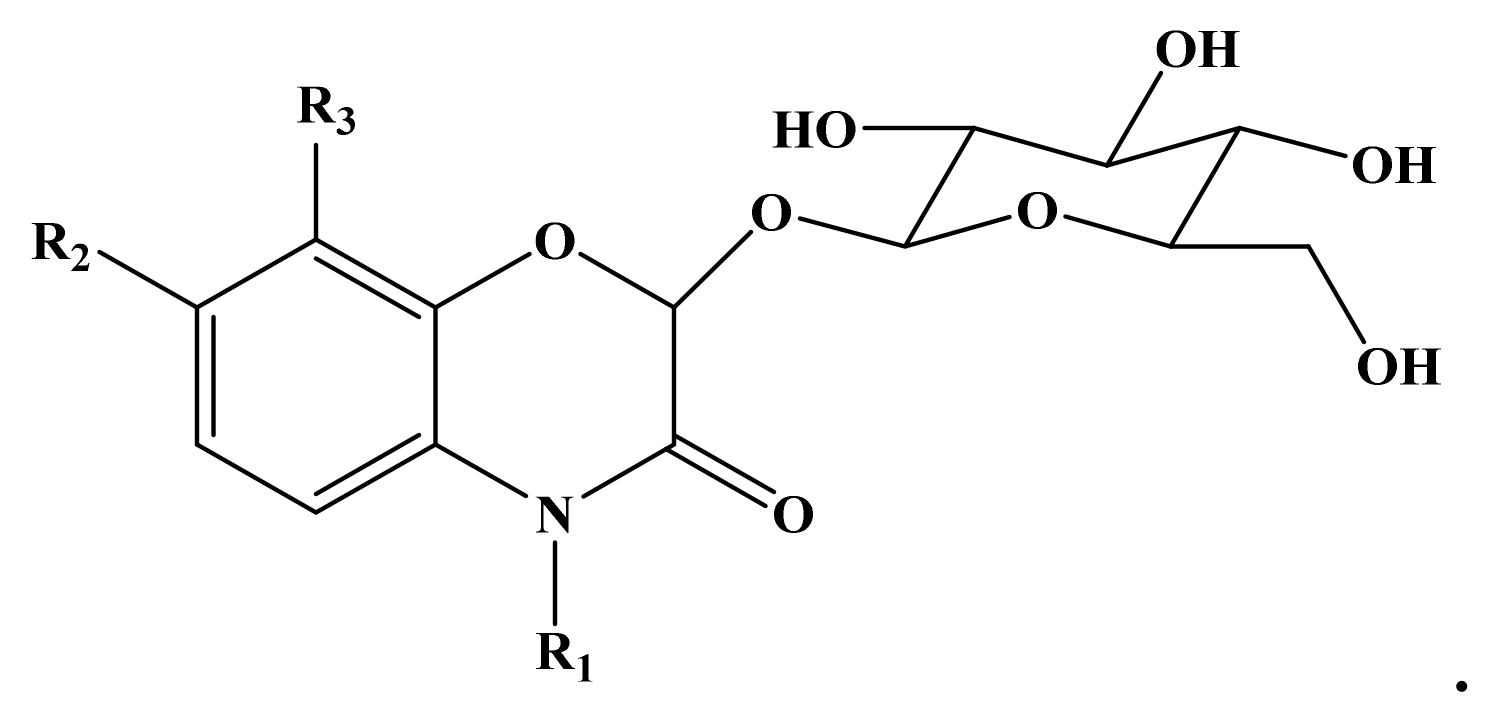
- Alipieva, K.I.; Taskova, R.M.; Evstatieva, L.N.; Handjieva, N.V.; Popov, S.S. Benzoxazinoids and iridoid glucosides from four lamium species. Phytochemistry 2003, 64, 1413–1417. [Google Scholar] [CrossRef] [PubMed]
- Budzianowski, J.; Skrzypczak, L. Phenylpropanoid esters from lamium album flowers. Phytochemistry 1995, 38, 997–1001. [Google Scholar] [CrossRef]
- Damtoft, S. Iridoid glucosides from lamium album. Phytochemistry 1991, 31, 175–178. [Google Scholar] [CrossRef]
- Damtoft, S.; Jensen, S.R. Hemialboside, a hemiterpene glucoside from lamium album. Phytochemistry 1995, 39, 923–924. [Google Scholar] [CrossRef]
- Savchenko, T.; Blackford, M.; Sarker, S.D.; Dinan, L. Phytoecdysteroids from Lamium spp: Identification and distribution within plants. Biochem. Syst. Ecol. 2001, 29, 891–900. [Google Scholar] [CrossRef]
- Carović-StanKo, K.; PeteK, M.; Martina, G.; Pintar, J.; Bedeković, D.; Ćustić, M.H.; Šatović, Z. Medicinal plants of the family lamiaceaeas functional foods—A review. Czech J. Food Sci. 2016, 34, 377. [Google Scholar] [CrossRef]
- Deng, Y.R.; He, L.; Li, W.Q.; Wang, H.Q. Studies on chemical constituents in herb of Lamium maculatum L. var Kansuense. Zhongguo Zhong Yao Za Zhi 2003, 28, 730–732. [Google Scholar]
- Nugroho, A.; Choi, J.K.; Park, J.H.; Lee, K.T.; Cha, B.C.; Park, H.J. Two new flavonol glycosides from Lamium amplexicaule L. And their in vitro free radical scavenging and tyrosinase inhibitory activities. Planta Med. 2009, 75, 364–366. [Google Scholar]
- Czerwinska, M.E.; Swierczewska, A.; Granica, S. Bioactive constituents of Lamium album L. As inhibitors of cytokine secretion in human neutrophils. Molecules 2018, 23, 2770. [Google Scholar] [CrossRef]
- Kurkin, V. Phenylpropanoids from medicinal plants: Distribution, classification, structural analysis, and biological activity. Chem. Nat. Comp. 2003, 39, 123–153. [Google Scholar] [CrossRef]
- Cao, J.; Yu, H.; Wu, Y.; Wang, X. Occurrence and Biological Activities of Phenylpropionyl Iridoids. Mini Rev. Med. Chem. 2019, 19, 292–309. [Google Scholar] [CrossRef]
- Damtoft, S.; Jensen, S.R.; Nielsen, B.J. Biosynthesis of iridoid glucosides in Lamium album. Phytochemistry 1991, 31, 135–137. [Google Scholar] [CrossRef]
- Alipieva, K.I.; Taskova, R.M.; Jensen, S.R.; Handjieva, N.V. Iridoid glucosides from Lamium album and Lamium maculatum (Lamiaceae). Biochem. Syst. Ecol. 2006, 34, 88–91. [Google Scholar] [CrossRef]
- Pereira, O.R.; Domingues, M.R.M.; Silva, A.M.S.; Cardoso, S.M. Phenolic constituents of lamium album: Focus on isoscutellarein derivatives. Food Res. Int. 2012, 48, 330–335. [Google Scholar] [CrossRef]
- Moerman, D.E. The medicinal flora of native north america: An analysis. J. Ethnopharmacol. 1991, 31, 1–42. [Google Scholar] [CrossRef]
- Flamini, G.; Cioni, P.L.; Morelli, I. Composition of the essential oils and in vivo emission of volatiles of four lamium species from italy: L. purpureum, L. hybridum, L. bifidum and L. amplexicaule. Food Chem. 2005, 91, 63–68. [Google Scholar] [CrossRef]
- Alipieva, K.; Evstatieva, L.; Handjieva, N.; Popov, S. Comparative analysis of the composition of flower volatiles from Lamium L. Species and Lamiastrum galeobdolon heist. Ex fabr. Z. Nat. C 2003, 58, 779–782. [Google Scholar] [CrossRef]
- Kurihara, F.; Kikuchi, M. On the constituents of the essential oil component from Lamium purpureum L. Yakugaku Zasshi 1976, 96, 1348–1351. [Google Scholar] [CrossRef]
- Hussain, A.I.; Anvar, F.; Sherazi, S.T.H.; Przybylski, R. Chemical composition, antioxidant and antimicrobial activities of basil (Ocimum basilicum) essential oils depends on seasonal variations. Food Chem. 2008, 108, 986–995. [Google Scholar] [CrossRef]
- Hussain, A.I.; Anvar, F.; Nigam, P.S.; Ashraf, M.; Gilani, A.H. Seasonal variation in content, chemical composition and antimicrobial and cytotoxic activities of essential oils from four mentha species. J. Sci. Food Agric. 2010, 90, 1827–1836. [Google Scholar] [CrossRef] [PubMed]
- Celiktas, O.Y.; Kocabas, E.E.H.; Bedir, E.; Sukan, F.V.; Ozek, T.; Baser, K.H.C. Antimicrobial activities of methanol extracts and essential oils of rosmarinus officinalis, depending on location and seasonal variations. Food Chem. 2007, 100, 553–559. [Google Scholar] [CrossRef]
- Ahmad, I.; Ahmad, M.S.A.; Ashraf, M.; Hussain, M.; Ashraf, M.Y. Seasonal variation in some medicinal and biochemical ingredients in Mentha longifolia (L.) huds. Pak. J. Bot. 2011, 43, 69–77. [Google Scholar]
- Singh, M.; Guleria, N. Influence of harvesting stage and inorganic and organic fertilizers on yield and oil composition of rosemary (Rosmarinus officinalis L.) in a semi-arid tropical climate. Ind. Crop. Prod. 2013, 42, 37–40. [Google Scholar] [CrossRef]
- Salman, M.; Abdel-Hameed, E.S.S.; Bazaid, S.A.; Dadi, M.M. Chemical composition for hydrodistillation essential oil of Mentha longifolia by gas chromatography-mass spectrometry from north regions in kingdom of saudi arabia. Pharma Chem. 2015, 7, 34–40. [Google Scholar]
- Kapchina-Toteva, V.; Dimitrova, M.A.; Stefanova, M.; Koleva, D.; Kostov, K.; Yordanova, Z.P.; Stefanov, D.; Zhiponova, M.K. Adaptive changes in photosynthetic performance and secondary metabolites during white dead nettle micropropagation. J. Plant Physiol. 2014, 171, 1344–1353. [Google Scholar] [CrossRef] [PubMed]
- Sajjadi, S.E.; Ghannadi, A. Analysis of the essential oil of Lamium amplexicaule L. from northeastern iran. J. Essent. Oil Bear. Plants 2012, 15, 577–581. [Google Scholar] [CrossRef]
- Jones, C.D.; Woods, K.E.; Setzer, W.N. A chemical ecological investigation of the allelopathic potential of Lamium amplexicaule and Lamium purpureum. Open J. Ecol. 2012, 2, 167–177. [Google Scholar] [CrossRef]
- Nickavar, B.; Mojab, F.; Bamasian, S. Volatile components from aerial parts of Lamium amplexicaule from Iran. J. Essent. Oil-Bear. Plants 2013, 11, 36–40. [Google Scholar] [CrossRef]
- Abu-ziada, M.E.A.; Mashaly, I.A.; Abdelgawed, A.M.; Asmeda, A.A. Ecology and phytochemistry of Lamium amplexicaule L. J. Environ. Sci. 2014, 43, 311–327. [Google Scholar]
- Layka, S.; Kara-Ali, A.; Sultan, A. A morphological, anatomical and chemical study on Lamium amplexicaule L. (lamiaceae). Tishreen Univ. J. Res. Sci. Stud. 2011, 33, 176–194. [Google Scholar]
- Rastitelnye Resursy Sssr; Nauka: St. Petersburg, FL, USA, 1991; Volume 6.
- El-Sayed, Z.I.A. Chemical composition, antimicrobial and insecticidal activities of the essential oil of Lamium maculatum L. Grown in egypt. Biosci. Biotechnol. Res. Asia 2008, 5, 65–72. [Google Scholar]
- El-Sattar, A.; Handjieva, N.; Popov, S.; Evstatieva, L. Volatile constituents from Lamium maculatum leaves and Nepeta mussini roots. C. R. Acad. Bulg. Sci. 1993, 46, 37–39. [Google Scholar]
- Roussis, V.; Chinou, I.; Perdetzoglou, D.; Loukis, A. Identification and bacteriostatic activity of the essential oil of Lamium garganicum L. ssp. Laevigatum arcangeli. J. Essent. Oil Res. 1996, 8, 291–293. [Google Scholar] [CrossRef]
- Morteza-Semnani, K.; Saeedi, M.; Akbarzadeh, M. Chemical composition of the essential oil of the flowering aerial parts of Lamium album L. J. Essent. Oil Bear. Plants 2016, 19, 773–777. [Google Scholar] [CrossRef]
- Mickene, R.; Bakutis, B.; Maruska, A.; Ragazinskiene, O.; Kaskoniene, V. Effect of volatile secondary metabolites of Monarda didyma L., Lamium album L. And Myrrhis odorata L. Plants against micromycetes of indoor environments of animals. Veterinariia 2014, 68, 48–54. [Google Scholar]
- Kovalvoya, A.; Ilyina, T.; Kolesnik, Y. Study of component composition of the essential oil of leaves Lamium album. Pharmacology 2013, 1, 80–82. [Google Scholar]
- Layka, S.; Kara-Ali, A.; Sultan, A. A morphological and chemical study of two species of Lamium L.: Lamium moschatum mill. And Lamium striatum sibth. Et smith. Belonging to lamiaceae family. Tishreen Univ. Off. Website 2009, 31, 133–147. [Google Scholar]
- Turner, N.J.; Luczaj, L.J.; Migliorini, P.; Pieroni, A.; Dreon, A.L.; Sacchetti, L.E. Edible and tended wild plants, traditional ecological knowledge and agroecology. Crit. Rev. Plant Sci. 2011, 30, 198–225. [Google Scholar] [CrossRef]
- Heinrich, M.; Müller, W.E.; Galli, C. Local Mediterranean Food Plants and Nutraceuticals; Karger: Basel, Switzerland, 2006; Volume 59, p. 186. [Google Scholar]
- Ninomiya, K.; Nishida, S.; Matsura, Y.; Asada, M.; Kawahara, Y.; Yoshikawa, M.; Nishida, N.; Matsuura, Y. Fat-Metabolism Improving Agent for Use in Food/Drink for Improving Fat Metabolism and Preventing/Treating Lifestyle Related Disease e.g. Diabetes, Contains Polar Solvent Extract of Herb e.g. Rose Hip Fruit, Mugwort or Safflower; MORI-Non-Standard, Morishita Jintan KK; China, 2006; p. 19. [Google Scholar]
- Xu, F. Chinese Medicine e.g. for Treating Arthropathy, Comprises Broad Cocklebur, Vervain, Condyle Grass, Motherwort, Saxifrage, Cactus, Mulberry Branch, White Dead Nettle, Boston Ivy, Folium Photiniae, Water Pepper and Chinese Fever Vine; XUFF-Individual; 2008; p. 10. [Google Scholar]
- Picuric-Jovanovic, K.; Milovanovic, M.; Budincevic, M.; Vrbaski, Z. Antioxydative wirkung von lamium purpureum als nahrungsmittelzusatzstoff. In Acta of the 6th Symposium “Vitamine und Zusatzstoffe in der Ernahrung von Mensch und Tier”; Friedrich-Schiller Universitat: Jena, Germany, 1997. [Google Scholar]
- Bremness, L. The Complete Book of Herbs; Dorling Kindersley: London, UK, 1995. [Google Scholar]
- Cui, S.Y.; Chen, X.G.; Hu, Z. Identification and determination of ecdysone and phenylpropanoid glucoside and flavonoids in Lamium maculatum by capillary zone electrophoresis. Biomed. Chromatogr. 2003, 17, 477–482. [Google Scholar]
- Malik, Z.A.; Bhat, J.A.; Ballabha, R.; Bussmann, R.W.; Bhatt, A.B. Ethnomedicinal plants traditionally used in health care practices by inhabitants of western himalaya. J. Ethnopharmacol. 2015, 172, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Bahmani, M.; Zargaran, A. Ethno-botanical medicines used for urinary stones in the Urmia, Northwest Iran. Eur. J. Integr. Med. 2015, 7, 657–662. [Google Scholar] [CrossRef]
- Pieroni, A.; Sõukand, R.; Quave, C.L.; Hajdari, A.; Mustafa, B. Traditional food uses of wild plants among the gorani of south kosovo. Appetite 2017, 108, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Tetik, F.; Civelek, S.; Cakilcioglu, U. Traditional uses of some medicinal plants in malatya (Turkey). J. Ethnopharmacol. 2013, 146, 331–346. [Google Scholar] [CrossRef]
- De Feo, V.; Aquino, R.; Menghini, A.; Ramundo, E.; Senatore, F. Traditional phytotherapy in the Peninsula Sorrentina, Campania, Southern Italy. J. Ethnopharmacol. 1992, 36, 113–125. [Google Scholar] [CrossRef]
- Vokou, D.; Katradi, K.; Kokkini, S. Ethnobotanical survey of zagori (epirus, greece), a renowned centre of folk medicine in the past. J. Ethnopharmacol. 1993, 39, 187–196. [Google Scholar] [CrossRef]
- Ugulu, I.; Baslar, S.; Yorek, N.; Dogan, Y. The investigation and quantitative ethnobotanical evaluation of medicinal plants used around Izmir province, Turkey. J. Med. Plants Res. 2009, 3, 345–367. [Google Scholar]
- Grudzinskaya, L.M.; Gemedzhieva, N.G.; Nelina, N.B.; Karzhaubekova, Z.Z. Annotated List of Medicinal Plants of Kazakhstan; Almaty, Kazakhstan, 2014; Volume 20, p. 91. [Google Scholar]
- Sokolov, P.D. Plant. Resources of the USSR: Flowering Plants, Their Chemical Composition, Use. (Family. Hippuridaceae-Lobeliaceae); Nauka: St. Petersburg, Russia, 1991; pp. 38–39. [Google Scholar]
- Rehman, K.; Mashwani, Z.U.; Khan, M.A.; Ullah, Z.; Chaudhary, H.J. An ethnobotanical perspective of traditional medicinal plants from the khattak tribe of chonthra karak, pakistan. J. Ethnopharmacol. 2015, 165, 251–259. [Google Scholar] [CrossRef]
- Jan, H.A.; Ahmad, S.W.L.; Jan, S.; Ahmad, N.; Ullah, N. Ethnomedicinal survey of medicinal plants of chinglai valley, buner district, pakistan. Eur. J. Integr. Med. 2017, 13, 64–74. [Google Scholar]
- Naghibi, F.; Mosaddegh, M.; Motamed, S.M.; Ghorbani, A. Labiatae family in folk medicine in iran: From ethnobotany to pharmacology. Iran. J. Pharm. Res. 2005, 2, 63–79. [Google Scholar]
- Tomas-Barberan, F.A.; Gil, M.L. Chemistry and Natural Distribution of Flavonoids in the Labiatae; Royal Botanical Gardens: London, UK, 1992. [Google Scholar]
- Offiah, V.N.; Chikwendu, U.A. Antidiarrhoeal effects of Ocimum gratissimum leaf extract in experimental animals. J. Ethnopharmacol. 1999, 68, 327–330. [Google Scholar] [CrossRef]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 2012, 5, 9. [Google Scholar] [CrossRef]
- Gülçin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef]
- Zucca, P.; Sanjust, E.; Trogu, E.; Sollai, F.; Rescigno, A. Evaluation of antioxidant capacity of antioxidant-declared beverages marketed in italy. Ital. J. Food Sci. 2010, 22, 313–319. [Google Scholar]
- Belkhir, M.; Dhaouadi, K.; Rosa, A.; Atzeri, A.; Nieddu, M.; Tuberoso, C.I.G.; Rescigno, A.; Amri, M.; Fattouch, S. Protective effects of azarole polyphenolic extracts against oxidative damage using in vitro biomolecular and cellular models. Ind. Crop. Prod. 2016, 86, 239–250. [Google Scholar] [CrossRef]
- Orrù, R.; Zucca, P.; Falzoi, M.; Atzori, E.; Rescigno, A.; Padiglia, A. First step towards the biomolecular characterization of pompia, an endemic citrus-like fruit from sardinia (italy). Plant Biosyst. 2017, 151, 464–473. [Google Scholar] [CrossRef]
- Apak, R.A.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant activity/capacity measurement. 2. Hydrogen atom transfer (hat)-based, mixed-mode (electron transfer (et)/hat), and lipid peroxidation assays. J. Agric. Food Chem. 2016, 64, 1028–1045. [Google Scholar] [CrossRef]
- Apak, R.a.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant activity/capacity measurement. 1. Classification, physicochemical principles, mechanisms, and electron transfer (et)-based assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Capanoglu, E.; Shahidi, F. Measurement of Antioxidant Activity and Capacity: Recent Trends and Applications; John Wiley & Sons: New York, NY, USA, 2017. [Google Scholar]
- Tabart, J.; Kevers, C.; Pincemail, J.; Defraigne, J.-O.; Dommes, J. Comparative antioxidant capacities of phenolic compounds measured by various tests. Food Chem. 2009, 113, 1226–1233. [Google Scholar] [CrossRef]
- Tirzitis, G.; Bartosz, G. Determination of antiradical and antioxidant activity: Basic principles and new insights. Acta Biochim. Pol. 2010, 57, 139–142. [Google Scholar] [CrossRef]
- Leopoldini, M.; Marino, T.; Russo, N.; Toscano, M. Antioxidant properties of phenolic compounds: H-atom versus electron transfer mechanism. J. Phys. Chem. A 2004, 108, 4916–4922. [Google Scholar] [CrossRef]
- Leopoldini, M.; Marino, T.; Russo, N.; Toscano, M. Density functional computations of the energetic and spectroscopic parameters of quercetin and its radicals in the gas phase and in solvent. Theor. Chem. Acc. 2004, 111, 210–216. [Google Scholar] [CrossRef]
- Ahmed, J.K.; Salih, H.A.; Hadi, A. Anthocyanins in red beet juice act as scavengers for heavy metals ions such as lead and cadmium. Int. J. Sci. Technol. 2013, 2, 269–274. [Google Scholar]
- Rosa, A.; Nieddu, M.; Piras, A.; Atzeri, A.; Putzu, D.; Rescigno, A. Maltese mushroom (Cynomorium coccineum L.) as source of oil with potential anticancer activity. Nutrients 2015, 7, 849–864. [Google Scholar] [CrossRef] [PubMed]
- Zucca, P.; Rosa, A.; Tuberoso, C.I.G.; Piras, A.; Rinaldi, A.C.; Sanjust, E.; Dessì, M.A.; Rescigno, A. Evaluation of antioxidant potential of “maltese mushroom” (Cynomorium coccineum) by means of multiple chemical and biological assays. Nutrients 2013, 5, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Gorinstein, S.; Böhm, V.; Schaich, K.M.; Özyürek, M.; Güçlü, K. Methods of measurement and evaluation of natural antioxidant capacity/activity (iupac technical report). Pure Appl. Chem. 2013, 85, 957–998. [Google Scholar] [CrossRef]
- Luthria, D.L. Significance of sample preparation in developing analytical methodologies for accurate estimation of bioactive compounds in functional foods. J. Sci. Food Agric. 2006, 86, 2266–2272. [Google Scholar] [CrossRef]
- Durazzo, A. Study approach of antioxidant properties in foods: Update and considerations. Foods 2017, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Lagouri, V.; Alexandri, G. Antioxidant properties of greek O. dictamnus and R. officinalis methanol and aqueous extracts—HPLC determination of phenolic acids. Int. J. Food Prop. 2013, 16, 549–562. [Google Scholar] [CrossRef]
- Trouillas, P.; Calliste, C.A.; Allais, D.P.; Simon, A.; Marfak, A.; Delage, C.; Duroux, J.L. Antioxidant, anti-inflammatory and antiproliferative properties of sixteen water plant extracts used in the limousin countryside as herbal teas. Food Chem. 2003, 80, 399–407. [Google Scholar] [CrossRef]
- Matkowski, A.; Piotrowska, M. Antioxidant and free radical scavenging activities of some medicinal plants from the lamiaceae. Fitoterapia 2006, 77, 346–353. [Google Scholar] [CrossRef]
- Yumrutas, O.; Saygideger, S.D. Determination of in vitro antioxidant activities of different extracts of marrubium parviflorum fish et mey. and Lamium amplexicaule L. From south east of turkey. J. Med. Plants Res. 2010, 4, 2164–2172. [Google Scholar]
- Li, A.S.H.; Bandy, B.; Tsang, S.S.; Davison, A.J. DNA-breaking versus DNA-protecting activity of four phenolic compounds in vitro. Free Radic. Res. 2000, 33, 551–566. [Google Scholar] [CrossRef]
- Mira, L.; Fernandez, M.T.; Santos, M.; Rocha, R.; Florêncio, M.H.; Jennings, K.R. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res. 2002, 36, 1199–1208. [Google Scholar] [CrossRef]
- Bubueanu, C.; Gheorghe, C.; Pirvu, L.; Bubueanu, G. Antioxidant activity of butanolic extracts of Romanian native species—Lamium album and Lamium purpureum. Rom. Biotechnol. Lett. 2013, 18, 7255–7262. [Google Scholar]
- Vladimir-Knežević, S.; Blažeković, B.; Kindl, M.; Vladić, J.; Lower-Nedza, A.; Brantner, A. Acetylcholinesterase inhibitory, antioxidant and phytochemical properties of selected medicinal plants of the lamiaceae family. Molecules 2014, 19, 767–782. [Google Scholar] [CrossRef] [PubMed]
- Danila, D.; Adriana, T.; Camelia, S.; Valentin, G.; Anca, M. Antioxidant activity of methanolic extracts of Lamium album and Lamium maculatum species from wild populations in the Romanian eastern Carpathians. Planta Med. 2015, 81. [Google Scholar] [CrossRef]
- Eghbaliferiz, S.; Iranshahi, M. Prooxidant activity of polyphenols, flavonoids, anthocyanins and carotenoids: Updated review of mechanisms and catalyzing metals. Phytother. Res. 2016, 30, 1379–1391. [Google Scholar] [CrossRef]
- Bhat, S.H.; Azmi, A.S.; Hadi, S.M. Prooxidant DNA breakage induced by caffeic acid in human peripheral lymphocytes: Involvement of endogenous copper and a putative mechanism for anticancer properties. Toxicol. Appl. Pharmacol. 2007, 218, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Sofic, E.; Prior, R.L. Antioxidant and prooxidant behavior of flavonoids: Structure-activity relationships. Free Radic. Boil. Med. 1997, 22, 749–760. [Google Scholar] [CrossRef]
- Procházková, D.; Boušová, I.; Wilhelmová, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef]
- Lin, L.T.; Hsu, W.C.; Lin, C.C. Antiviral natural products and herbal medicines. J. Tradit. Complement. Med. 2014, 4, 24–35. [Google Scholar] [CrossRef]
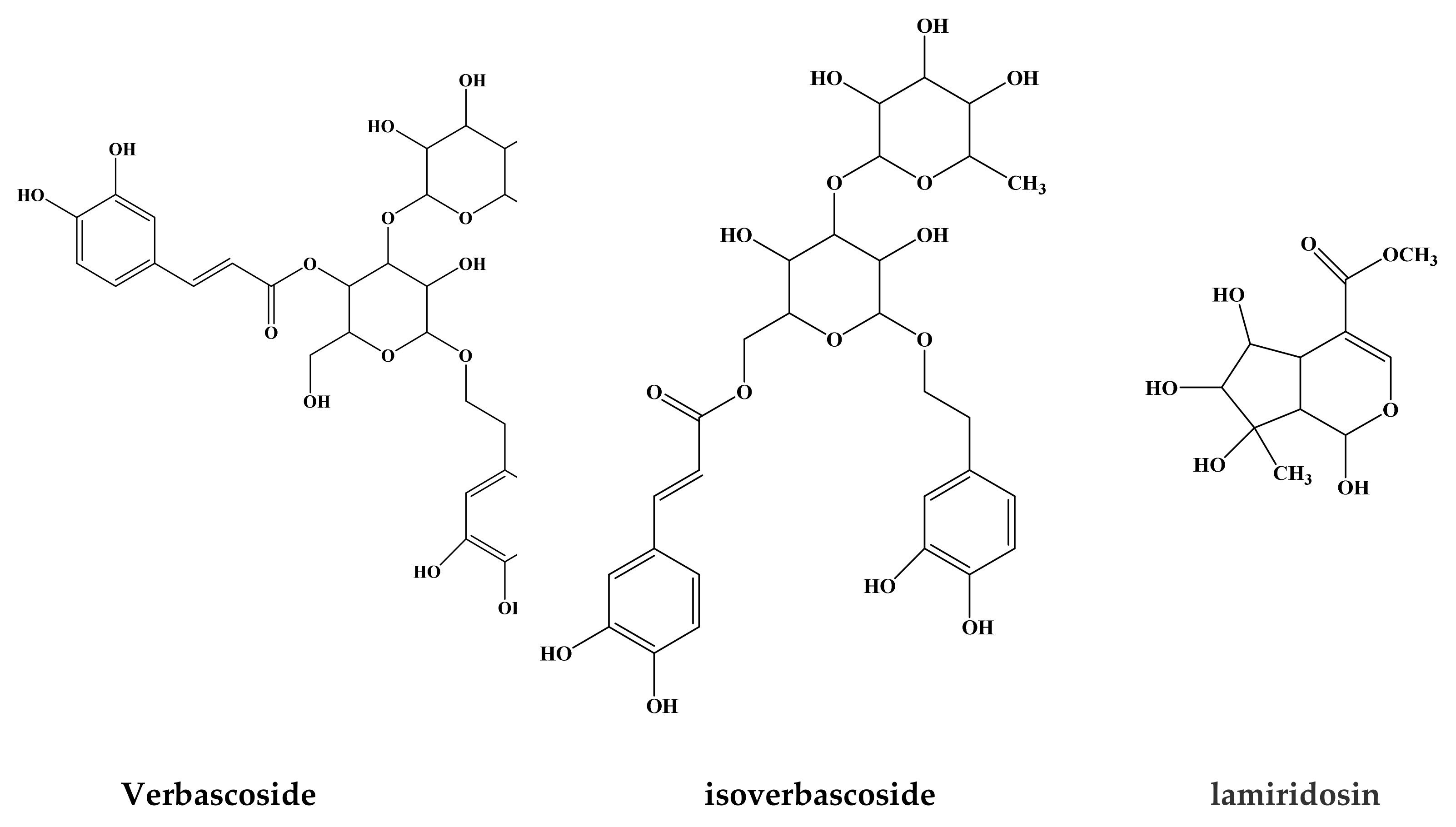
- Zhang, H.; Rothwangl, K.; Mesecar, A.D.; Sabahi, A.; Rong, L.; Fong, H.H.S. Lamiridosins, hepatitis c virus entry inhibitors from Lamium album. J. Nat. Prod. 2009, 72, 2158–2162. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, M.J.; Piras, A.; Porcedda, S.; Marongiu, B.; Falconieri, D.; Cavaleiro, C.; Rescigno, A.; Rosa, A.; Salgueiro, L. Antifungal activity of extracts from Cynomorium coccineum growing wild in sardinia island (Italy). Nat. Prod. Res. 2015, 29, 2247–2250. [Google Scholar] [CrossRef]
- Dulger, B. Antifungal activity of lamium tenuiflorum against some medical yeast candida and cryptococcus species. Pharm. Biol. 2009, 47, 467–470. [Google Scholar] [CrossRef]
- Erbil, N.; Alan, Y.; Digrak, M. Antimicrobial and antioxidant properties of Lamium galactophyllum boiss & reuter, L. macrodon boiss & huet and L. amplexicaule from Turkish Flora. Asian J. Chem. 2014, 26, 549–554. [Google Scholar]
- Chipeva, V.A.; Petrova, D.C.; Geneva, M.E.; Dimitrova, M.A.; Moncheva, P.A.; Kapchina-Toteva, V.M. Antimicrobial activity of extracts from in vivo and in vitro propagated Lamium album L. Plants. Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 559–562. [Google Scholar] [CrossRef]
- Kvietys, P.R.; Granger, D.N. Role of reactive oxygen and nitrogen species in the vascular responses to inflammation. Free Radic. Boil. Med. 2012, 52, 556–592. [Google Scholar] [CrossRef]
- Mitjavila, M.T.; Moreno, J.J. The effects of polyphenols on oxidative stress and the arachidonic acid cascade. Implications for the prevention/treatment of high prevalence diseases. Biochem. Pharmacol. 2012, 84, 1113–1122. [Google Scholar] [CrossRef]
- Lv, Q.W.; Zhang, W.; Shi, Q.; Zheng, W.J.; Li, X.; Chen, H.; Wu, Q.J.; Jiang, W.L.; Li, H.B.; Gong, L.; et al. Comparison of tripterygium wilfordii hook f with methotrexate in the treatment of active rheumatoid arthritis (Trifra): A randomised, controlled clinical trial. Ann. Rheum. Dis. 2015, 74, 1078–1086. [Google Scholar] [CrossRef] [PubMed]
- Zarei, L.; Naji-Haddadi, S.; Pourjabali, M.; Naghdi, N.; Tasbih-Forosh, M.; Shahsavari, S. Systematic review of anti-rheumatic medicinal plants: An overview of the effectiveness of articular tissues and joint pain associated with rheumatoid arthritis. J. Pharm. Sci. Res. 2017, 9, 547–551. [Google Scholar]
- Randall, C.; Randall, H.; Dobbs, F.; Hutton, C.; Sanders, H. Randomized controlled trial of nettle sting for treatment of base-of-thumb pain. J. R. Soc. Med. 2000, 93, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Uritu, C.M.; Mihai, C.T.; Stanciu, G.D.; Dodi, G.; Alexa-Stratulat, T.; Luca, A.; Leon-Constantin, M.M.; Stefanescu, R.; Bild, V.; Melnic, S.; et al. Medicinal plants of the family lamiaceae in pain therapy: A review. Pain Res. Manag. 2018, 2018, 7801543. [Google Scholar] [CrossRef] [PubMed]
- Veleva, R.; Petkova, B.; Moskova-Doumanova, V.; Doumanov, J.; Dimitrova, M.; Koleva, P.; Mladenova, K.; Petrova, S.; Yordanova, Z.; Kapchina-Toteva, V. Changes in the functional characteristics of tumor and normal cells after treatment with extracts of white dead-nettle. Biotechnol. Biotechnol. Equip. 2015, 29, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Krishnaiah, D.; Sarbatly, R.; Nithyanandam, R. A review of the antioxidant potential of medicinal plant species. Food Bioprod. Process. 2011, 89, 217–233. [Google Scholar] [CrossRef]
- Santoro, A.; Bianco, G.; Picerno, P.; Aquino, R.P.; Autore, G.; Marzocco, S.; Gazzerro, P.; Lioi, M.B.; Bifulco, M. Verminoside- and verbascoside-induced genotoxicity on human lymphocytes: Involvement of parp-1 and p53 proteins. Toxicol. Lett. 2008, 178, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Perucatti, A.; Genualdo, V.; Pauciullo, A.; Iorio, C.; Incarnato, D.; Rossetti, C.; Vizzarri, F.; Palazzo, M.; Casamassima, D.; Iannuzzi, L.; et al. Cytogenetic tests reveal no toxicity in lymphocytes of rabbit (Oryctolagus cuniculus, 2n = 44) feed in presence of verbascoside and/or lycopene. Food Chem. Toxicol. 2018, 114, 311–315. [Google Scholar] [CrossRef]
- Paduch, R.; Wójciak-Kosior, M.; Matysik, G. Investigation of biological activity of lamii albi flos extracts. J. Ethnopharmacol. 2007, 110, 69–75. [Google Scholar] [CrossRef]
- Moskova-Doumanova, V.; Miteva, G.; Dimitrova, M.; Topouzova-Hristova, T.; Kapchina, V. Methanol and chloroform extracts from Lamium album L. Affect cell properties of a549 cancer lung cell line. Biotechnol. Biotechnol. Equip. 2014, 26, 120–125. [Google Scholar] [CrossRef]
- Paduch, R.; Woźniak, A. The effect of Lamium album extract on cultivated human corneal epithelial cells (10.014 prsv-t). J. Ophthalmic Vis. Res. 2015, 10, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Richter, A.; Jander, G. Beyond defense: Multiple functions of benzoxazinoids in maize metabolism. Plant Cell Physiol. 2018, 59, 1528–1533. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, K.B.; Tanwir, F.; Gregersen, P.L.; Steffensen, S.K.; Jensen, B.M.; Poulsen, L.K.; Nielsen, C.H.; Høyer, S.; Borre, M.; Fomsgaard, I.S. Benzoxazinoids: Cereal phytochemicals with putative therapeutic and health-protecting properties. Mol. Nutr. Food Res. 2015, 59, 1324–1338. [Google Scholar] [CrossRef] [PubMed]
- Rescigno, A.; Sollai, F.; Rinaldi, A.C.; Soddu, G.; Sanjust, E. Polyphenol oxidase activity staining in polyacrylamide electrophoresis gels. J. Biochem. Biophys. Methods 1997, 34, 155–159. [Google Scholar] [CrossRef]
- Rescigno, A.; Sanjust, E.; Pedulli, G.F.; Valgimigli, L. Spectrophotometric method for the determination of polyphenol oxidase activity by coupling of 4-tert-butyl-O-benzoquinone and 4-amino-N,N-diethylaniline. Anal. Lett. 1999, 32, 2007–2017. [Google Scholar] [CrossRef]
- Asthana, S.; Zucca, P.; Vargiu, A.V.; Sanjust, E.; Ruggerone, P.; Rescigno, A. Structure-activity relationship study of hydroxycoumarins and mushroom tyrosinase. J. Agric. Food Chem. 2015, 63, 7236–7244. [Google Scholar] [CrossRef]
- Rescigno, A.; Sanjust, E.; Soddu, G.; Rinaldi, A.C.; Sollai, F.; Curreli, N.; Rinaldi, A. Effect of 3-hydroxyanthranilic acid on mushroom tyrosinase activity. Biochim. Biophys. Acta-Protein Struct. Mol. 1998, 1384, 268–276. [Google Scholar] [CrossRef]
- Rescigno, A.; Casañola-Martin, G.M.; Sanjust, E.; Zucca, P.; Marrero-Ponce, Y. Vanilloid derivatives as tyrosinase inhibitors driven by virtual screening-based QSAR models. Drug Test. Anal. 2011, 3, 176–181. [Google Scholar] [CrossRef]
- Schlich, M.; Fornasier, M.; Nieddu, M.; Sinico, C.; Murgia, S.; Rescigno, A. 3-hydroxycoumarin loaded vesicles for recombinant human tyrosinase inhibition in topical applications. Colloids Surf. B Biointerfaces 2018, 171, 675–681. [Google Scholar] [CrossRef]
- Kim, D.; Park, J.; Kim, J.; Han, C.; Yoon, J.; Kim, N.; Seo, J.; Lee, C. Flavonoids as mushroom tyrosinase inhibitors: A fluorescence quenching study. J. Agric. Food Chem. 2006, 54, 935–941. [Google Scholar] [CrossRef]
- Xie, L.P.; Chen, Q.X.; Huang, H.; Wang, H.Z.; Zhang, R.Q. Inhibitory effects of some flavonoids on the activity of mushroom tyrosinase. Biochemistry (Moscow) 2003, 68, 487–491. [Google Scholar] [CrossRef]
- Etsassala, N.G.; Waryo, T.; Popoola, O.K.; Adeloye, A.O.; Iwuoha, E.I.; Hussein, A.A. Electrochemical screening and evaluation of lamiaceae plant species from South Africa with potential tyrosinase activity. Sensors 2019, 19, 1035. [Google Scholar] [CrossRef] [PubMed]
- Petukhova, N.M.; Buryakina, A.V.; Avenirova, E.L.; Burakova, M.A.; Drozhzhina, E.V. Studies of the biological activity of an oil extract of the snakeflower Lamium album. Pharm. Chem. J. 2008, 42, 354–356. [Google Scholar] [CrossRef]
- National Sunflower Association (N.S.A.). Four Types of Sunflower Oil. Available online: https://www.sunflowernsa.com/oil/Four-Types-of-Sunflower-Oil/ (accessed on 29 April 2019).
- Yuan, T.; Fan, W.B.; Cong, Y.; Xu, H.D.; Li, C.J.; Meng, J.; Bao, N.R.; Zhao, J.N. Linoleic acid induces red blood cells and hemoglobin damage via oxidative mechanism. Int. J. Clin. Exp. Pathol. 2015, 8, 5044–5052. [Google Scholar] [PubMed]
- Jabbar, A.; Raza, M.A.; Iqbal, Z.; Khan, M.N. An inventory of the ethnobotanicals used as anthelmintics in the Southern Punjab (Pakistan). J. Ethnopharmacol. 2006, 108, 152–154. [Google Scholar] [CrossRef] [PubMed]
- Shapira, M.Y.; Raphaelovich, Y.; Gilad, L.; Or, R.; Dumb, A.J.; Ingber, A. Treatment of atopic dermatitis with herbal combination of eleutherococcus, Achillea millefolium, and Lamium album has no advantage over placebo: A double blind, placebo-controlled, randomized trial. J. Am. Acad. Dermatol. 2005, 52, 691–693. [Google Scholar] [CrossRef]
| Plant Name | Place/Country of Collection | Parts Used | Extraction Method Used | % Yield | Main Components | References |
|---|---|---|---|---|---|---|
| (1) Lamium amplexicaule L. | Khorassan-e Razavi province in northeastern Iran | aerial parts (flowers and leaves) * | Hydro-distillation | 0.1 (w/w) | Trans-phytol (44.8%), octadecanol (12.0%), hexadecanoic acid (11.8%) and hexahydrofarnesyl acetone (10.6%) | [52] |
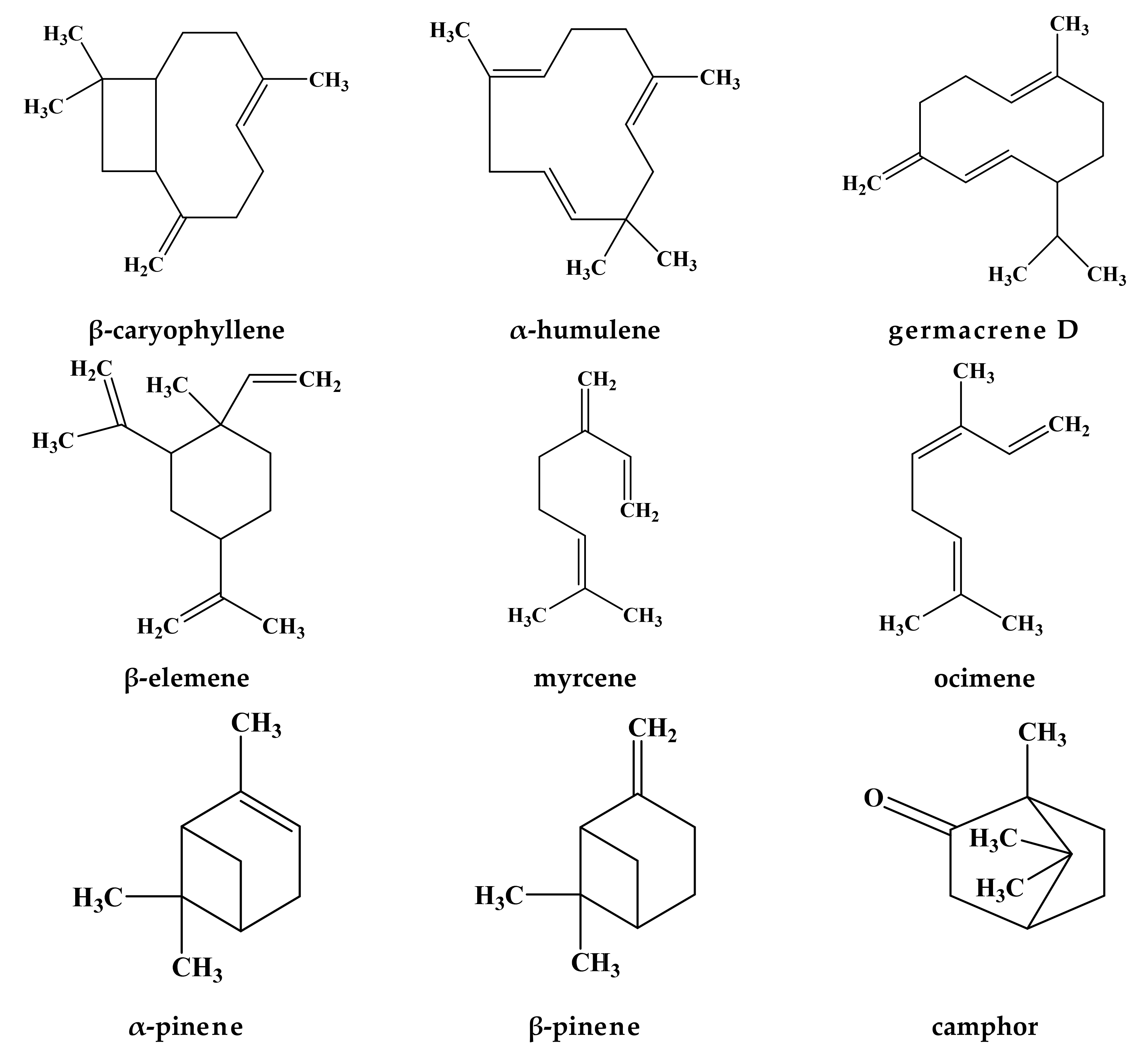
| Huntsville, Alabama, USA | aerial parts ** | Hydro-distillation | NA | Germacrene D (18.5–34.9%), (E)-caryophyllene (2.5–11.9%), α-pinene (2.2–16.2%), β-pinene (2.0–10.6%) and 1-octen-3-ol (3.5–8.0%) | [53] | |
| Pergole, Arcidosso Municipality, South Tuscany, Italy | Flowering aerial parts | Hydro-distillation | NA | Trans-chrysanthenyl acetate (41.1%), germacrene D (28.9%) and α-pinene (6.8%), ocimene (0.8%) | [42] | |
| Northeast of Tehran, Iran | aerial parts * | Hydro-distillation | 0.1(v/w) | Germacrene-D (22.3%) and camphor (18.1%) | [54] | |
| El Dakahlyia governorate, Egypt | Leaves | NA | NA | Isophytol (14.8%), 9,12,15-ocadecanoic acid methyl ester (19.2%), 6,10,14-trimethyl-2-pentadecanone (8.0%), dibutyl phathalate (6.1%), nonacosane (5.5%), hexadecanoic acid (3.4%) and nonyl phenol (3.2%) | [55] | |
| Syria | NA | Dry evaporation | NA | Imedazol and pyrimidene | [56] | |
| NA | NA | NA | 0.09 | NA | [57] | |
| (2) Lamium purpureum L. | Huntsville, Alabama, USA | aerial parts ** | Hydro-distillation | NA | Germacrene D (15.0–46.3%), α-pinene (4.1–15.3%), β-pinene (6.3–16.3%), and 1-octen-3-ol (4.2–15.3%), β-elemene (3.7–16.0%) | [53] |
| Pergole (Arcidosso Municipality, South Tuscany, Italy | Flowering aerial parts | Hydro-distillation | NA | Germacrene D (35.4%), β-pinene (26.8%) and α-pinene (13.4%), ocimene (2.9%) | [42] | |
| Japan | Aerial parts | Steam distillation | NA | 1-Octen-3-ol, cis-3-hexen-1-ol, phenethyl alcohol, benzyl alcohol, phenol, 0-, m-, and p-cresols, guaiacol, eugenol | [44] | |
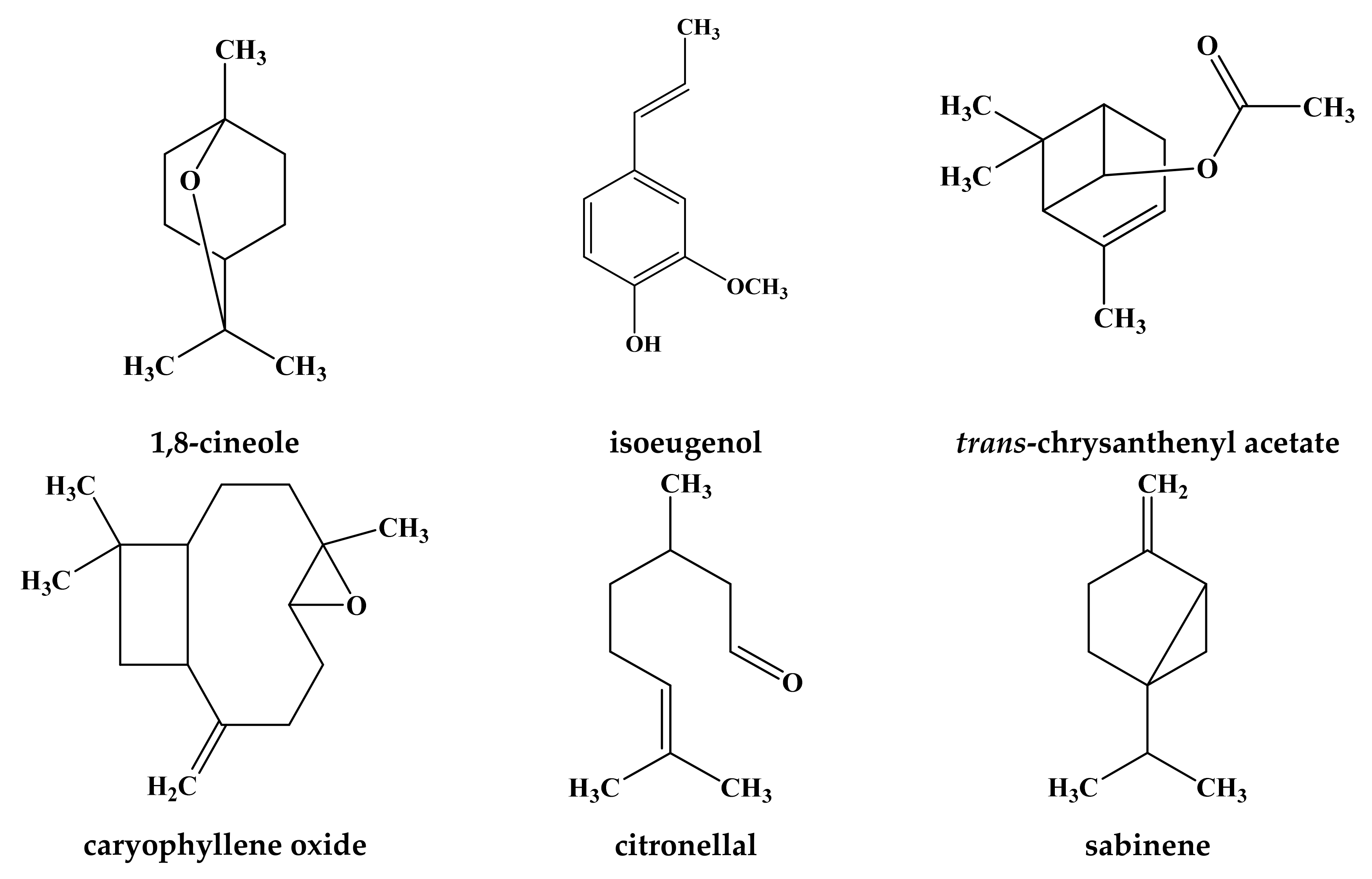
| (3) Lamium maculatum L. | Experimental station of Faculty of Pharmacy, Zagazig University, Egypt | aerial parts ** | Hydro-distillaion | 0.35 (v/w) | β-caryophyllene (14.8%), caryophyllene oxide (13.8%), Z,E-α-franesene (10.1%), dihydroedulan I (9.13%), α-humulene (6.1%), bornyl formate (6.0%) and α-bisabolene (5.3%) | [58] |
| NA | Leaves | NA | NA | Hexahydrofarnesylacetone (22%) | [59] | |
| (4) Lamium hybridum Vill | Pergole (Arcidosso Municipality, South Tuscany, Italy | Flowering aerial parts | Hydro-distillation | NA | Germacrene D (39.0%), (Z)-ocimene (8.7%), methyl salicylate (7.5%) and β-caryophyllene (6.1%), ocimene (11.6%) | [42] |
| (5) Lamium bifidum Cyr. | Pergole (Arcidosso Municipality, South Tuscany, Italy | Flowering aerial parts | Hydro-distillation | NA | Germacrene D (34.9%), sabinene (12.4%), β-caryophyllene (11.5%), α-humulene (6.8%) | [42] |
| (6) Lamium garganicum L. subsp. laevigatum Arcangeli | Athens, Greece | aerial parts *** | Hydro-distillation | 0.31 | 1,8-cineole (47.5%), citronellal (25.1%) and isoeugenol (11.8%) | [60] |
| (7) Lamium album L. | Behshahr, Mazandaran Province, North of Iran | Flowering aerial parts | Hydro-distillation | 0.2 (w/w) | 6,10,14-trimethyl-2-pentadecanone (10.2%) and 4-hydroxy-4-methyl-2-pentanone (9.1%) | [61] |
| Experimental field of the Kaunas Botanical Garden of Vytautas Magnus University, Lithuania | Plants in the vegetation period | Supercritical carbon dioxide extraction method | NA | Prenol, farnesene-beta- E, tridecanol n, dodecanoic acid n, hexadecane-n, squalene, tetradecanol-n, undecane–n, benzoate-isopentyl, dodecanoate -butyl, phytone, neophytadiene | [62] | |
| NA | Aerial part | NA | 0.04–0.46 | NA | [57] | |
| NA | Flowers | NA | 0.05 | NA | [57] | |
| Kharkiv region, Ukraine | Leaves | NA | NA | α-Terpeniol, linalool, squalene, spatulenol, α-Bisabolol | [63] | |
| (8) Lamium moschatum Mill. | NA | Flowers | Steam-washed | NA | Caryophyllene | [64] |
| (9) Lamium striatum Sibth. et. Smith | Flowers | Steam-washed | NA | Carboxylic acids | [64] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salehi, B.; Armstrong, L.; Rescigno, A.; Yeskaliyeva, B.; Seitimova, G.; Beyatli, A.; Sharmeen, J.; Mahomoodally, M.F.; Sharopov, F.; Durazzo, A.; et al. Lamium Plants—A Comprehensive Review on Health Benefits and Biological Activities. Molecules 2019, 24, 1913. https://doi.org/10.3390/molecules24101913
Salehi B, Armstrong L, Rescigno A, Yeskaliyeva B, Seitimova G, Beyatli A, Sharmeen J, Mahomoodally MF, Sharopov F, Durazzo A, et al. Lamium Plants—A Comprehensive Review on Health Benefits and Biological Activities. Molecules. 2019; 24(10):1913. https://doi.org/10.3390/molecules24101913
Chicago/Turabian StyleSalehi, Bahare, Lorene Armstrong, Antonio Rescigno, Balakyz Yeskaliyeva, Gulnaz Seitimova, Ahmet Beyatli, Jugreet Sharmeen, Mohamad Fawzi Mahomoodally, Farukh Sharopov, Alessandra Durazzo, and et al. 2019. "Lamium Plants—A Comprehensive Review on Health Benefits and Biological Activities" Molecules 24, no. 10: 1913. https://doi.org/10.3390/molecules24101913
APA StyleSalehi, B., Armstrong, L., Rescigno, A., Yeskaliyeva, B., Seitimova, G., Beyatli, A., Sharmeen, J., Mahomoodally, M. F., Sharopov, F., Durazzo, A., Lucarini, M., Santini, A., Abenavoli, L., Capasso, R., & Sharifi-Rad, J. (2019). Lamium Plants—A Comprehensive Review on Health Benefits and Biological Activities. Molecules, 24(10), 1913. https://doi.org/10.3390/molecules24101913














