Proteomics Research on the Protective Effect of Mangiferin on H9C2 Cell Injury Induced by H2O2
Abstract
:1. Introduction
2. Results
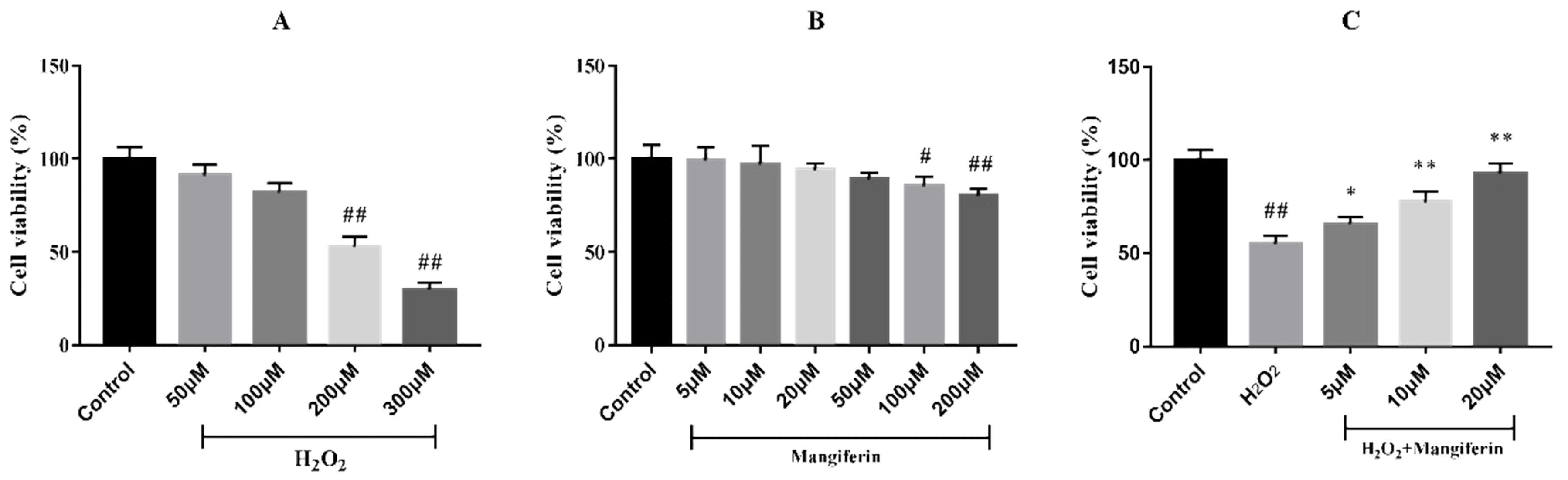
2.1. Mangiferin Protected the H9C2 Cell Injury Induced by H2O2
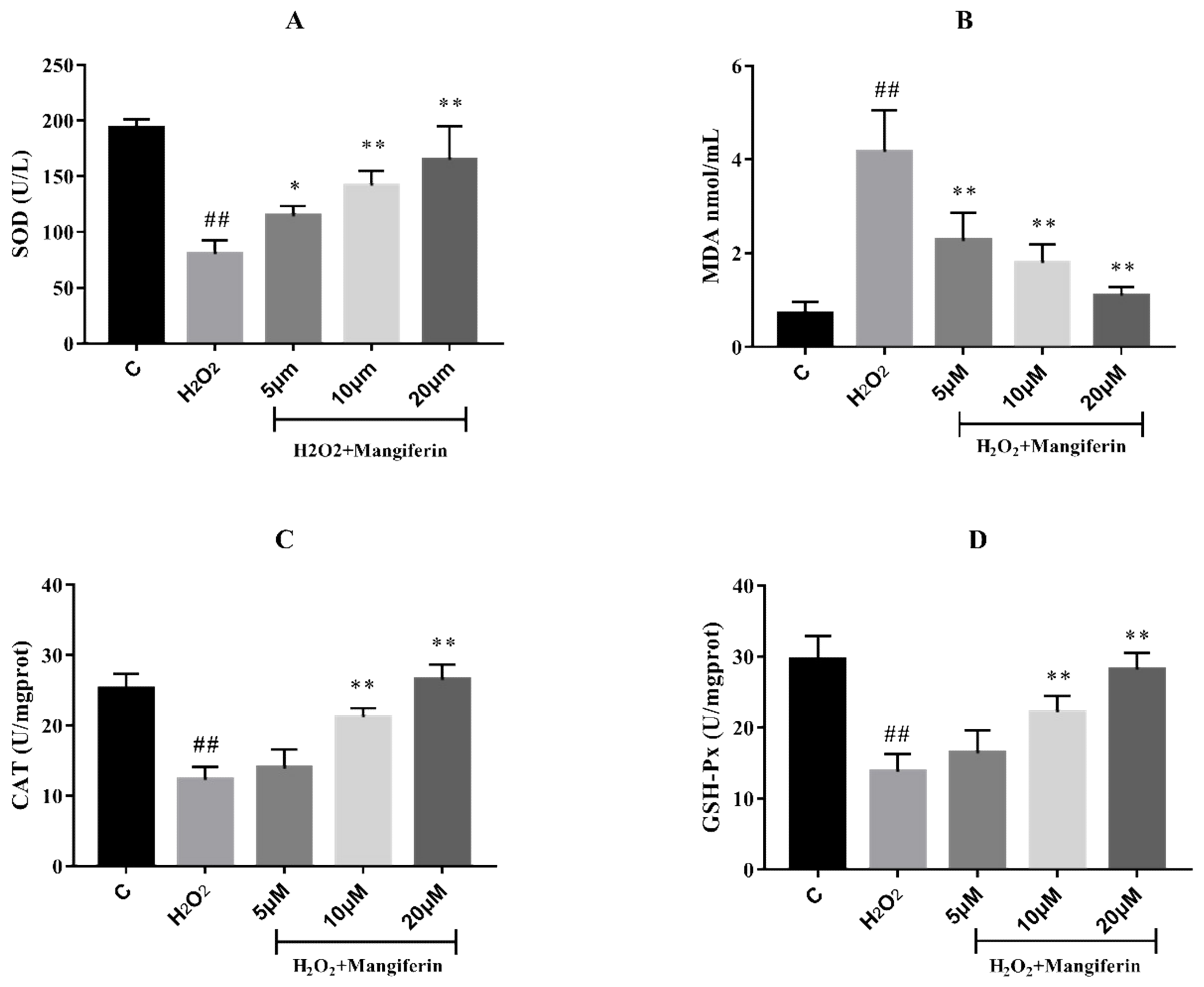
2.2. The Anti-Oxidation Effect of Mangiferin
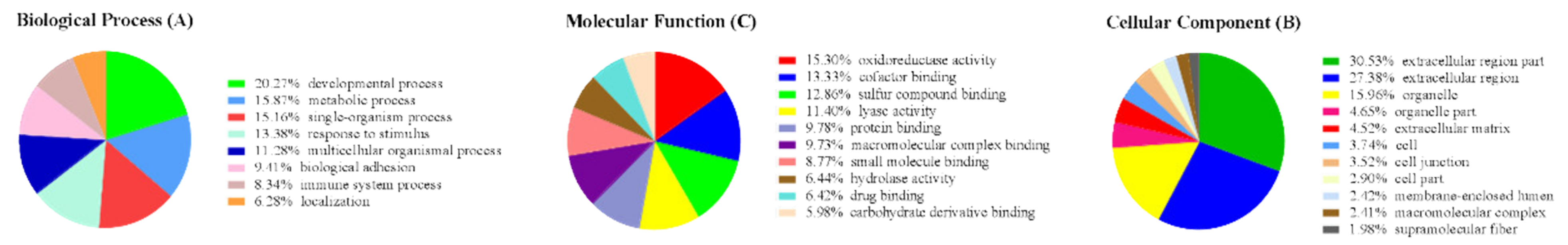
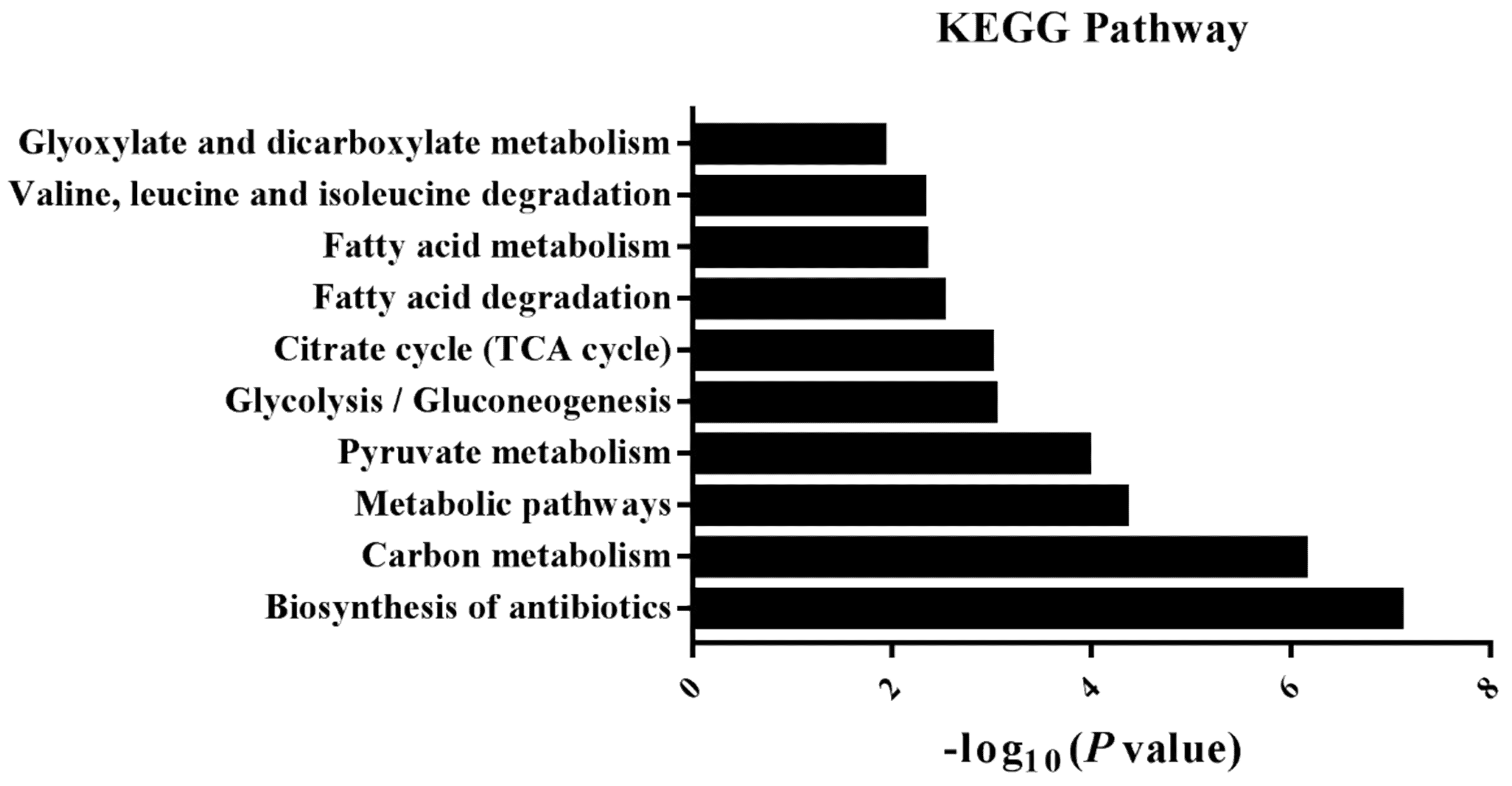
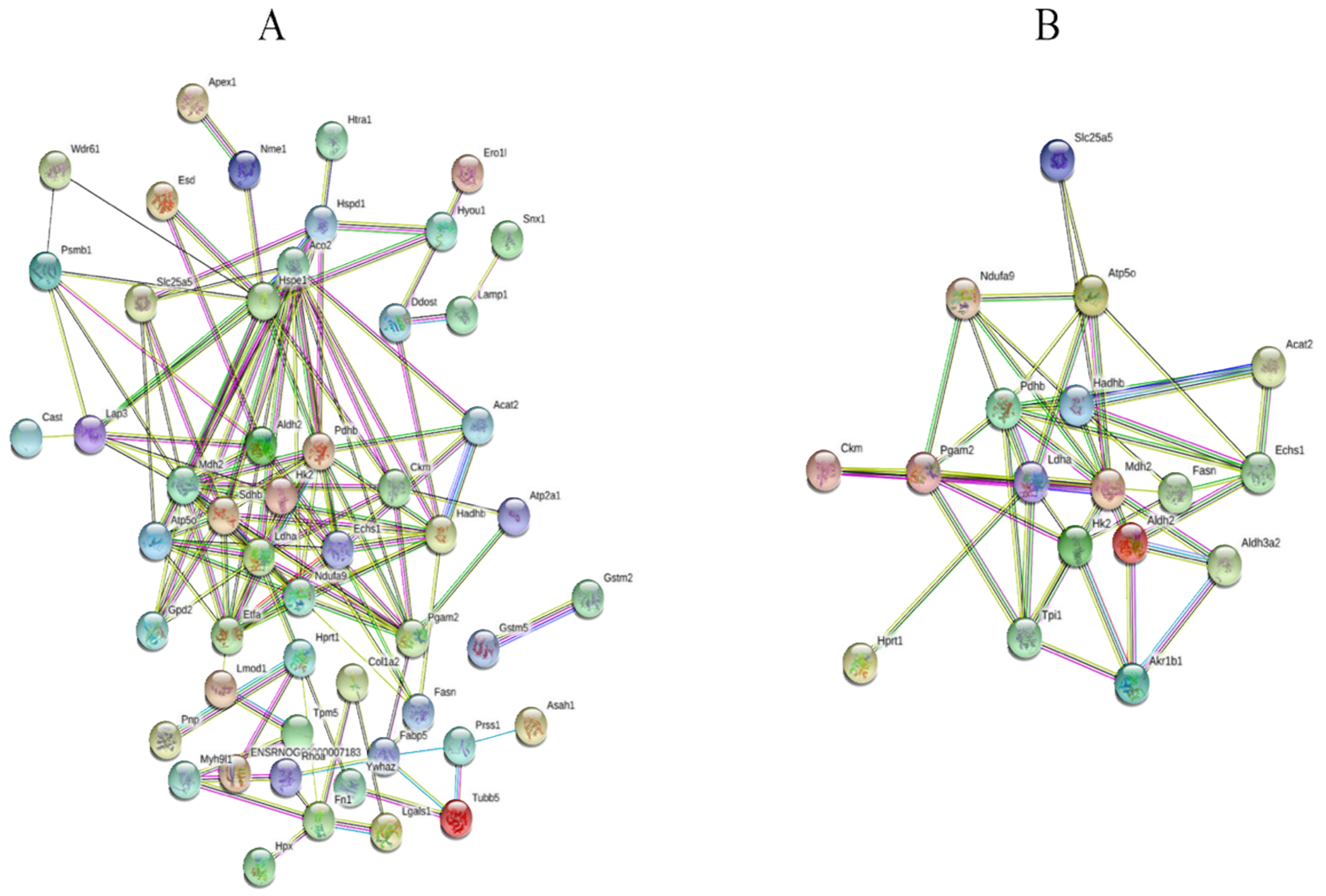
2.3. Effect of Mangiferin on Protein Expression Profiles in H9C2 Cell Induced by H2O2
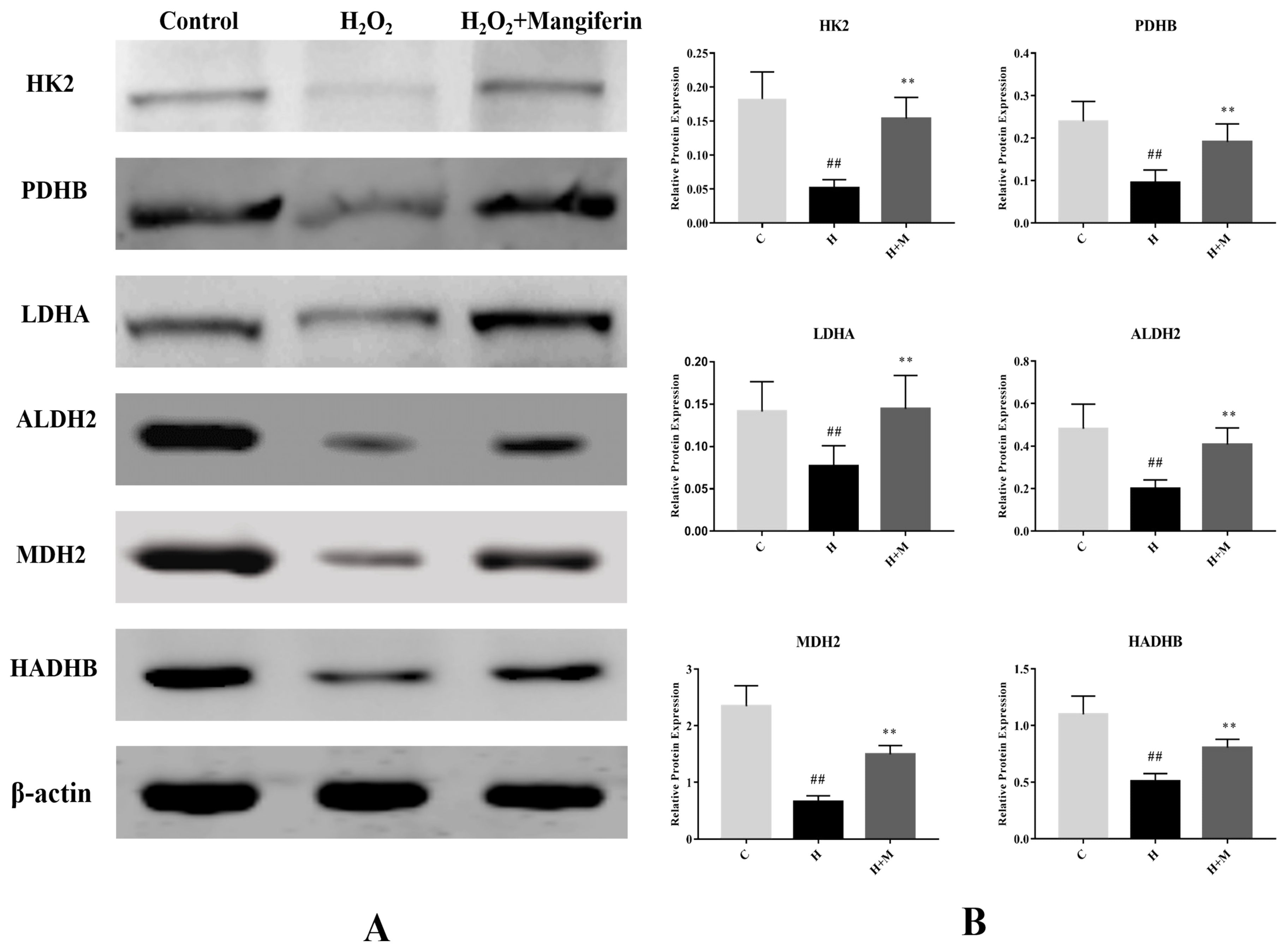
2.4. Validation of Selected Proteins by Western Blot
3. Discussion
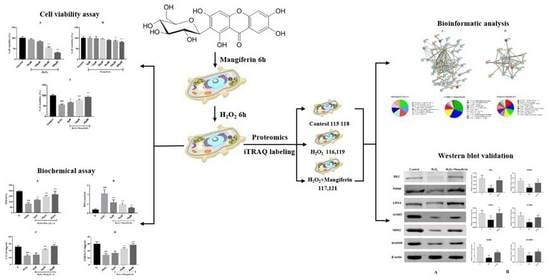
4. Materials and Methods
4.1. Reagents and Chemicals
4.2. Cell Culture and Oxidative Injury Induced by H2O2
4.3. Cell Viability Assays
4.4. Biochemical Analysis of H9C2 Cells
4.5. Protein Preparation and iTRAQ Labelling
4.6. High-pH Reverse-Phase Liquid Chromatography Fraction
4.7. Nano LC-MS/MS Analysis
4.8. Protein Identification and Bioinformatic Analysis
4.9. Western Blot Validation
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ALDH2 | Aldehyde dehydrogenase |
| CAT | Catalase |
| DMSO | Dimethyl sulfoxide |
| GSH-Px | Glutathione peroxidase |
| H2O2 | Hydrogen peroxide |
| HADHB | Trifunctional enzyme subunit beta |
| HK2 | Hexokinase-2 |
| LDHA | L-lactate dehydrogenase A chain |
| MDA | Malonaldehyde |
| MDH2 | Malate dehydrogenase |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| PDHB | Pyruvate dehydrogenase E1 component subunit beta |
| SOD | Superoxide dismutase |
References
- Song, L.N.; Yang, H.; Wang, H.X.; Tian, C.; Liu, Y.; Zeng, X.J.; Gao, E.; Kang, Y.M.; Du, J.; Li, H.H. Inhibition of 12/15 lipoxygenase by baicalein reduces myocardial ischemia/reperfusion injury via modulation of multiple signaling pathways. Apoptosis 2014, 19, 567–580. [Google Scholar] [CrossRef]
- Hausenloy, D.J.; Yellon, D.M. Myocardial ischemia-reperfusion injury: A neglected therapeutic target. J. Clin. Invest. 2013, 123, 92–100. [Google Scholar] [CrossRef]
- Kalogeris, T.; Bao, Y.M.; Korthuis, R.J. Mitochondrial reactive oxygen species: A double edged sword in ischemia/reperfusion vs preconditioning. Redox Biol. 2014, 2, 702–714. [Google Scholar] [CrossRef]
- Turer, A.; Hill, J. Pathogenesis of myocardial ischemia-reperfusion injury and rationale for therapy. J. Am. Coll. Cardiol. 2010, 106, 360–368. [Google Scholar] [CrossRef]
- Loke, K.E.; McConnell, P.I.; Tuzman, J.M.; Shesely, E.G.; Smith, C.J.; Stackpole, C.J.; Thompson, C.I.; Kaley, G.; Wolin, M.S.; Hintze, T.H. Endogenous endothelial nitric oxide synthase–derived nitric oxide is a physiological regulator of myocardial oxygen consumption. Circ. Res. 1999, 84, 840–845. [Google Scholar] [CrossRef]
- Xia, Y.; Zweier, J.L. Substrate control of free radical generation from xanthine oxidase in the postischemic heart. J. Biol. Chem. 1995, 270, 18797–18803. [Google Scholar] [CrossRef]
- Chou, H.C.; Chen, Y.W.; Lee, T.R.; Wu, F.S.; Chan, H.T.; Lyu, P.C.; Timms, J.F.; Chan, H.L. Proteomics study of oxidative stress and Src kinase inhibition in H9C2 cardiomyocytes: A cell model of heart ischemia–reperfusion injury and treatment. Free Radic. Biol. Med. 2010, 49, 96–108. [Google Scholar] [CrossRef]
- Zhao, X.; Dou, M.; Zhang, Z.; Zhang, D.; Huang, C. Protective effect of Dendrobium officinale polysaccharides on H2O2-induced injury in H9c2 cardiomyocytes. Biomed. Pharmacother. 2017, 94, 72–78. [Google Scholar] [CrossRef]
- Liu, Y.W.; Cheng, Y.Q.; Liu, X.L.; Hao, Y.C.; Li, Y.; Zhu, X.; Zhang, F.; Yin, X.X. Mangiferin upregulates glyoxalase 1 through activation of Nrf2/are signaling in central neurons cultured with high glucose. Mol. Neurobiol. 2017, 54, 4060–4070. [Google Scholar] [CrossRef]
- Muruganandan, S.; Gupta, S.; Kataria, M.; Lal, J.; Gupta, P. Mangiferin protects the streptozotocin-induced oxidative damage to cardiac and renal tissues in rats. Toxicology 2002, 176, 165–173. [Google Scholar] [CrossRef]
- Miura, T.; Ichiki, H.; Iwamoto, N.; Kato, M.; Kubo, M.; Sasaki, H.; Okadia, M.; Ishida, T.; Seino, Y.; Tanigawa, K. Antidiabetic activity of the rhizoma of Anemarrhena asphodeloides and active components, mangiferin and its glucoside. Biol. Pharm. Bull. 2001, 24, 1009–1011. [Google Scholar] [CrossRef]
- Miura, T.; Ichiki, H.; Hashimoto, I.; Iwamoto, N.; Kao, M.; Kubo, M.; Ishihara, E.; Komatsu, Y.; Okada, M.; Ishida, T. Antidiabetic activity of a xanthone compound, mangiferin. Phytomedicine 2001, 8, 85–87. [Google Scholar] [CrossRef]
- Telang, M.; Dhulap, S.; Mandhare, A.; Hirwani, R. Therapeutic and cosmetic applications of mangiferin: A patent review. Expert Opin Ther Pat. 2013, 23, 1561–1580. [Google Scholar] [CrossRef]
- Pal, P.B.; Sinha, K.; Sil, P.C. Mangiferin Attenuates Diabetic Nephropathy by Inhibiting Oxidative Stress Mediated Signaling Cascade, TNFα Related and Mitochondrial Dependent Apoptotic Pathways in Streptozotocin-Induced Diabetic Rats. PLoS ONE 2014, 9, e107220. [Google Scholar] [CrossRef]
- Shi, C.X.; Li, X.J.; Yuan, L.L.; Cheng, B.H.; Xu, X.D. Protective effects of mangiferin on the models of BRL cell oxidative stress induced by H2O2. Anat. Res. 2017, 39, 110–114. [Google Scholar]
- Chao, W.; Xian, W.G. Cardioprotective Effects of Mangiferin on Myocardial in Schemia Reperfusion Injury in Rats. Chin. J. Arterioscler. 2008, 16, 697–700. [Google Scholar]
- Suchal, K.; Malik, S.; Gamad, N.; Malhotra, R.K.; Goyal, S.N.; Ojha, S.; Kumari, S.; Bhatia, J.; Arya, D.S. Mangiferin protect myocardial insults through modulation of MAPK/TGF-β pathways. Eur. J. Pharmacol. 2016, 776, 34–43. [Google Scholar] [CrossRef]
- Mirza, S.P. Quantitative mass spectrometry-based approaches in cardiovascular research. Circ.-Cardiovasc. Gene. 2012, 5, 453–477. [Google Scholar] [CrossRef]
- Mesaros, C.; Blair, I.A. Mass spectrometry-based approaches to targeted quantitative proteomics in cardiovascular disease. Clin. Proteomics. 2016, 13, 20–37. [Google Scholar] [CrossRef]
- Mokou, M.; Lygirou, V.; Vlahou, A.; Mischak, H. Proteomics in cardiovascular disease: Recent progress and clinical implication and implementation. Expert Rev. Proteomic. 2017, 14, 117–136. [Google Scholar] [CrossRef]
- Sun, G.; Ye, N.; Dai, D.; Chen, Y.; Li, C.; Sun, Y. The protective role of the TOPK/PBK pathway in myocardial ischemia/reperfusion and H2O2-induced injury in H9C2 cardiomyocytes. Int. J. Mol. Sci. 2016, 17, 267–280. [Google Scholar] [CrossRef]
- Kumar, D.; Jugdutt, B.I. Apoptosis and oxidants in the heart. J. Lab. Clin. Med. 2003, 142, 288–297. [Google Scholar] [CrossRef]
- Circu, M.L.; Aw, T.Y. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic. Biol. Med. 2010, 48, 749–762. [Google Scholar] [CrossRef]
- Lenčo, J.; Lenčová-Popelová, O.; Link, M.; Jirkovská, A.; Tambor, V.; Potůčková, E.; Stulík, J.; Šimůnek, T.; Štěrba, M. Proteomic investigation of embryonic rat heart-derived H9c2 cell line sheds new light on the molecular phenotype of the popular cell model. Exp. Cell Res. 2015, 339, 174–186. [Google Scholar] [CrossRef]
- Xu, J.; Tang, Y.; Bei, Y.; Ding, S.; Che, L.; Yao, J.; Wang, H.; Lv, D.; Xiao, J. miR-19b attenuates H2O2-induced apoptosis in rat H9C2 cardiomyocytes via targeting PTEN. Oncotarget 2016, 7, 10870–10878. [Google Scholar]
- Mates, J. Effects of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicology 2000, 153, 83–104. [Google Scholar] [CrossRef]
- Vijayasarathy, K.; Naidu, K.S.; Sastry, B. Melatonin metabolite 6-Sulfatoxymelatonin, Cu/Zn superoxide dismutase, oxidized LDL and malondialdehyde in unstable angina. Int. J. Cardiol. 2010, 144, 315–317. [Google Scholar] [CrossRef]
- Sheng, Y.; Abreu, I.A.; Cabelli, D.E.; Maroney, M.J.; Miller, A.-F.; Teixeira, M.; Valentine, J.S. Superoxide dismutases and superoxide reductases. Chem Rev. 2014, 114, 3854–3918. [Google Scholar] [CrossRef]
- Chelikani, P.; Fita, I.; Loewen, P.C. Diversity of structures and properties among catalases. Cell Mol. Life Sci. 2004, 61, 192–208. [Google Scholar] [CrossRef]
- Nielsen, F.; Mikkelsen, B.B.; Nielsen, J.B.; Andersen, H.R.; Grandjean, P. Plasma malondialdehyde as biomarker for oxidative stress: Reference interval and effects of life-style factors. Clin. Chem. 1997, 43, 1209–1214. [Google Scholar]
- Lopaschuk, G. Regulation of carbohydrate metabolism in ischemia and reperfusion. Am. Heart J. 2000, 139, 115–119. [Google Scholar] [CrossRef]
- Kyu Kim, H.; Thu, V.T.; Heo, H.J.; Kim, N.; Han, J. Cardiac proteomic responses to ischemia–reperfusion injury and ischemic preconditioning. Expert Rev. Proteomic. 2011, 8, 241–261. [Google Scholar] [CrossRef]
- Xu, Z.W.; Chen, X.; Jin, X.H.; Meng, X.Y.; Zhou, X.; Fan, F.X.; Mao, S.Y.; Wang, Y.; Zhang, W.C.; Shan, N.N. SILAC-based proteomic analysis reveals that salidroside antagonizes cobalt chloride-induced hypoxic effects by restoring the tricarboxylic acid cycle in cardiomyocytes. J. Proteomics. 2016, 130, 211–220. [Google Scholar] [CrossRef]
- Von Lewinski, D.; Gasser, R.; Rainer, P.P.; Huber, M.S.; Wilhelm, B.; Roessl, U.; Haas, T.; Wasler, A.; Grimm, M.; Bisping, E. Functional effects of glucose transporters in human ventricular myocardium. Eur. J. Heart Fail. 2010, 12, 106–113. [Google Scholar] [CrossRef]
- Lopaschuk, G.D.; Belke, D.D.; Gamble, J.; Toshiyuki, I.; Schönekess, B.O. Regulation of fatty acid oxidation in the mammalian heart in health and disease. Biochim. Biophys. Acta (BBA)-Lipids Lipid Metab. 1994, 1213, 263–276. [Google Scholar] [CrossRef]
- Oliver, E.F.; Opie, L. Effects of glucose and fatty acids on myocardial ischaemia and arrhythmias. Lancet 1994, 343, 155–158. [Google Scholar] [CrossRef]
- Zhang, T.J.; Guo, R.X.; Li, X.; Wang, Y.W.; Li, Y.J. Tetrandrine cardioprotection in ischemia–reperfusion (I/R) injury via JAK3/STAT3/Hexokinase II. Eur. J. Pharmacol. 2017, 813, 153–160. [Google Scholar] [CrossRef]
- Wilson, J.E. Isozymes of mammalian hexokinase: Structure, subcellular localization and metabolic function. J. Exp. Biol. 2003, 206, 2049–2057. [Google Scholar] [CrossRef]
- Miyamoto, S.; Murphy, A.N.; Brown, J.H. Akt mediates mitochondrial protection in cardiomyocytes through phosphorylation of mitochondrial hexokinase-II. Cell Death Differ. 2008, 15, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Zuurbier, C.J.; Eerbeek, O.O.; Meijer, A.J. Ischemic preconditioning, insulin and morphine all cause hexokinase redistribution. Am. J. Physiol-Heart C. 2005, 289, 496–499. [Google Scholar] [CrossRef] [PubMed]
- Daisuke, K.; Yoji Andrew, M.; Koh, N. Prolyl-hydroxylase PHD3 interacts with pyruvate dehydrogenase (PDH)-E1β and regulates the cellular PDH activity. Biochem. Biophys. Res. Commun. 2014, 451, 288–294. [Google Scholar]
- Quinlan, C.L.; Goncalves, R.L.S.; Martin, H.M.; Nagendra, Y.; Bunik, V.I.; Brand, M.D. The 2-oxoacid dehydrogenase complexes in mitochondria can produce superoxide/hydrogen peroxide at much higher rates than complex I. J. Biol. Chem. 2014, 289, 8312–8325. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Neely, J.R. Effects of ischemia and reperfusion on pyruvate dehydrogenase activity in isolated rat hearts. J. Mol. Cell Cardiol. 1983, 15, 359–367. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, G.; Gao, X. Attenuation of miR-34a protects cardiomyocytes against hypoxic stress through maintenance of glycolysis. Biosci Rep. 2017, 37, BSR20170925. [Google Scholar] [CrossRef]
- Luo, C.; Wang, H.; Chen, X.; Cui, Y.; Li, H.; Long, J.; Mo, X.; Liu, J. Protection of H9c2 rat cardiomyoblasts against oxidative insults by total paeony glucosides from Radix Paeoniae Rubrae. Phytomedicine 2013, 21, 20–24. [Google Scholar] [CrossRef]
- Luo, X.J.; Liu, B.; Ma, Q.L.; Peng, J. Mitochondrial aldehyde dehydrogenase, a potential drug target for protection of heart and brain from ischemia/reperfusion injury. Curr. Drug Targets. 2014, 15, 948–955. [Google Scholar]
- Mali, V.R.; Deshpande, M.; Pan, G.; Thandavarayan, R.A.; Palaniyandi, S.S. Impaired ALDH2 activity decreases the mitochondrial respiration in H9C2 cardiomyocytes. Cell Signal. 2016, 28, 1–6. [Google Scholar] [CrossRef]
- Wang, H.; Kang, P.; Hongwei, Y.E.; Ying, Y.U.; Wang, X.; Qin, G. Anti-apoptotic role of mitochondrial aldehyde dehydrogenase 2 in myocardial ischemia/reperfusion injury in diabetic rats. South Med. J. 2012, 32, 345–348. [Google Scholar]
- Hussain, C.; Nicolaj, B.; Colin, C.; Cedric, M.; Jonathan, D.; Ormerod, J.O.M.; Rebekka, J.; Hans Erik, B.T.; Andrew, R.; Schmidt, M.R. Aldehyde dehydrogenase-2 inhibition blocks remote preconditioning in experimental and human models. Basic Res. Cardiol. 2013, 108, 343. [Google Scholar]
- Janero, D.R.; Hreniuk, D. Suppression of TCA cycle activity in the cardiac muscle cell by hydroperoxide-induced oxidant stress. Am. J. Physiol. 1996, 270, 1735–1742. [Google Scholar] [CrossRef]
- Tretter, L.; Adam, V.V. Inhibition of Krebs cycle enzymes by hydrogen peroxide: A key role of [alpha]-ketoglutarate dehydrogenase in limiting NADH production under oxidative stress. J. Neurosci. 2000, 20, 8972–8979. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.D.; Qiu, J.; Zhao, G.X.; Qie, L.Y.; Wei, X.B.; Gao, H.Q. Quantitative Mitochondrial Proteomics Study on Protective Mechanism of Grape Seed Proanthocyanidin Extracts Against Ischemia/Reperfusion Heart Injury in Rat. Chem. Res. Chin. Univ. 2012, 28, 1035–1040. [Google Scholar]
- Kim, N.; Lee, Y.; Kim, H.; Joo, H.; Youm, J.B.; Park, W.S.; Warda, M.; Cuong, D.V.; Han, J. Potential biomarkers for ischemic heart damage identified in mitochondrial proteins by comparative proteomics. Proteomics 2010, 6, 1237–1249. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guan, W.; Liu, Y.; Liu, Y.; Wang, Q.; Ye, H.-L.; Cheng, Y.-G.; Kuang, H.-X.; Jiang, X.-C.; Yang, B.-Y. Proteomics Research on the Protective Effect of Mangiferin on H9C2 Cell Injury Induced by H2O2. Molecules 2019, 24, 1911. https://doi.org/10.3390/molecules24101911
Guan W, Liu Y, Liu Y, Wang Q, Ye H-L, Cheng Y-G, Kuang H-X, Jiang X-C, Yang B-Y. Proteomics Research on the Protective Effect of Mangiferin on H9C2 Cell Injury Induced by H2O2. Molecules. 2019; 24(10):1911. https://doi.org/10.3390/molecules24101911
Chicago/Turabian StyleGuan, Wei, Yan Liu, Yuan Liu, Qi Wang, Hong-Liang Ye, Yan-Gang Cheng, Hai-Xue Kuang, Xi-Cheng Jiang, and Bing-You Yang. 2019. "Proteomics Research on the Protective Effect of Mangiferin on H9C2 Cell Injury Induced by H2O2" Molecules 24, no. 10: 1911. https://doi.org/10.3390/molecules24101911
APA StyleGuan, W., Liu, Y., Liu, Y., Wang, Q., Ye, H.-L., Cheng, Y.-G., Kuang, H.-X., Jiang, X.-C., & Yang, B.-Y. (2019). Proteomics Research on the Protective Effect of Mangiferin on H9C2 Cell Injury Induced by H2O2. Molecules, 24(10), 1911. https://doi.org/10.3390/molecules24101911