Abstract
Cucurbita genus has received a renowned interest in the last years. This plant species, native to the Americas, has served worldwide folk medicine for treating gastrointestinal diseases and intestinal parasites, among other clinical conditions. These pharmacological effects have been increasingly correlated with their nutritional and phytochemical composition. Among those chemical constituents, carotenoids, tocopherols, phenols, terpenoids, saponins, sterols, fatty acids, and functional carbohydrates and polysaccharides are those occurring in higher abundance. However, more recently, a huge interest in a class of triterpenoids, cucurbitacins, has been stated, given its renowned biological attributes. In this sense, the present review aims to provide a detailed overview to the folk medicinal uses of Cucurbita plants, and even an in-depth insight on the latest advances with regards to its antimicrobial, antioxidant and anticancer effects. A special emphasis was also given to its clinical effectiveness in humans, specifically in blood glucose levels control in diabetic patients and pharmacotherapeutic effects in low urinary tract diseases.
1. Introduction
Cucurbita plants have been applied in different cultures as traditional medication. For instance, Native Americans have used pumpkins for the treatment of intestinal worms and urinary ailments, this therapeutic strategy being approved by American doctors in the early nineteenth century as an anthelmintic for worms annihilating [1]. Seeds are used as an anthelmintic, to treat issues of the urinary framework, high blood pressure, to prevent the development of kidney stones, to ease prostate disorders and even to improve the erysipelas skin contamination [2]. In southeastern Europe, Cucurbita pepo L. (pumpkin) seeds have been applied to heal irritable bladder and prostate enlargement. Specifically, in Germany, the use of pumpkin seeds was adopted for application by the authority for irritated bladder conditions and micturition problems of prostate enlargement, although the monograph written in 1985 noted a lack of pharmacological studies that could confirm its effective clinical effects. On the other hand, in the USA, the purchase of all such non-prescription medications for the therapy of prostate enlargement was banned in 1990. In traditional Chinese medicine, Cucurbita moschata Duchesne seeds were also applied for handling the parasitic diseases caused by worms, while Mexican herbalists have used Cucurbita ficifolia Bouché as a remedy for reducing blood sugar levels [3,4,5,6,7].
Indeed, increasing evidence has shown that cucurbits’ medicinal properties depend upon the chemical compounds present, which produce a specific physiological effect in the human body [8,9,10]. Specifically, cucurbits fruits are found to be beneficial in blood cleansing, purification of toxic substances and good for digestion, besides giving the required energy to improve human health. These species possess a higher amount of proteins, phytosterols [11,12], unsaturated fatty acids [13,14], vitamins (like carotenoids, tocopherols) [15] and microelements (e.g., zinc) [16]. Fruits, seeds and leaves from various Cucurbita members (pumpkin, watermelon, melon, cucumber squash, gourds, etc.) possess different pharmacological effects [17,18], such as antidiabetic [19,20,21], antiulcer, analgesic, nephroprotective [22] and anticancer activities [18]. In this sense, this review provides a detailed overview to the folk medicinal uses of Cucurbita plants, an in-depth insight on the latest advances regarding its antimicrobial, antioxidant and anticancer effects, and lastly, a special emphasis to its clinical effectiveness in humans, specifically in blood glucose levels control and low urinary tract diseases (Figure 1).
Figure 1.
Most pronounced and investigated biological effects of Cucurbita spp.
2. Cucurbita Plants: A Brief Overview to Its Ethnopharmacological Uses
Recent ethnopharmacological studies showed that C. pepo and Cucurbita maxima Duchesne are among the most commonly used Cucurbita plants for traditional medicinal treatments. As shown in Table 1, many different components of Cucurbita plants are applied in diverse regions of the globe for handling different diseases.

Table 1.
Cucurbita plants traditionally applied in the cures of different diseases in diverse regions of the world.
In particular, the positive health effects of C. maxima seeds are well-documented [23,25,26,27,28]. Raw C. maxima seeds are orally administered for the treatment of digestive disorders, such as intestinal worms [23,25], constipation [23] and vomiting blood and blood bile [26] by the local people in the Iberian Peninsula, Argentina and India, respectively. Also, sun-dried seeds of C. maxima are ingested in Mauritius for the treatment of renal failure [27], whereas raw seeds are consumed to treat prostatitis in the Agro Nocerino Sarnese in Campania, Southern Italy [28]. C. maxima seeds, fruits, flowers and leaves are also used as traditional medicine [24,26,27,29,30,31], where the treatment of urinary disorders, blood pressure regulation and prevention of constipation can be achieved with oral consumption of C. maxima fruits, and the wound healing with dermal application [26,27,30]. In Mkuranga district in Tanzania, C. maxima leaves are used for healing anemia [24], and in the Ashanti region in Ghana, this plant part is orally consumed for lung and head cancer treatment [31]. Furthermore, in Mauritius, C. maxima fruits are compressed externally on eyes against cataract [27], while in India the same petals are used to treat osteosarcoma [29]. Nonetheless, and to the authors knowledge, much is needed to support both the in vitro and in vivo biological effects of this plant, since most of the efforts has been made towards its agro-industrial applications.
With regards to C. pepo seeds, they are mainly regarded as agro-industrial wastes, while in some parts of the globe they are used raw, roasted or cooked, at a domestic scale [40]. Accordingly, in a study carried out in Ghimbi District in Southwest Ethiopia [32], it was reported that oral administration of cultivated seed of C. pepo is used as a gonorrhea therapy. Moreover, C. pepo seeds are also used as an herbal remedy by breast cancer patients in West Bank in Palestine [36]. In another study, conducted in Nkonkobe municipality in Eastern Cape, South Africa [35], it was indicated that arthritis and blood booster are treated with orally taken C. pepo leaves. C. pepo leaves are also used for the treatment of malaria and dandruff in the local government area in south-eastern Nigeria and Ghimbi District in Southwest Ethiopia, respectively [37,38]. In the latter study, it was also pointed out that the fruits of C. pepo are consumed to treat gastritis and stomachache [37]. Topical use of C. pepo fruit as an external antiseptic was reported in Ripollès district, the Pyrenees in Catalonia and Iberian Peninsula, whereas in the same location the flowers of this plant are used for antigenic, antidermatitic, antiecchymotic, antiophidian, antipyretic and anti-toxic purposes [34]. C. pepo, as the whole plant, is also applied in the folk medicine of Mesoamerica and Caribbean for the therapy of fitness due to its pancreatic lipase inhibition activity [33]. In addition to the above, the decoction prepared from the Cucurbita galeottii Cogn. seeds is used against mucous discharge in Mauritius [39].
3. Cucurbita Plants Phytochemical Composition
Carotenoids are highly present in the fruit of these plants, namely α-carotene, β-carotene, ζ-carotene, neoxanthin, violaxanthin, lutein, zeaxanthin, taraxanthin, luteoxanthin, auroxanthine, neurosporene, flavoxanthin, 5,6,5′,6′-diepoxy-β-carotene, phytofluene, α-cryptoxanthin and β-cryptoxanthin [41]. Total carotenoid content varied between 234.21 μg/g to 404.98 μg/g in C. moschata fruit [42], and 171.9 μg/g to 461.9 μg/g in C. pepo fruit [43]. There are also several publications on the carotenoid content of a number of Cucurbita plants such as C. moschata, C. pepo [42] and C. maxima [44]. Edible Cucurbita seeds are also rich in vitamin E (49.49 μg/g to 92.59 µg/g), γ-tocopherol is more abundant than α-tocopherol and the fruit contains less [45].
The study of Yang et al. [46] showed no flavonoid content (below detection limit: 0.05 mg/100 g) in either the immature or the mature fruit of C. maxima. Only the shoots and buds showed positive results. Sreeramulu and Raghunath [47] reported that average total phenolic content of C. maxima was 46.43 mg gallic acid equivalent (GAE)/100 g. In another study, C. maxima was analyzed for its flavonoid content and kaempferol was found to be the only flavonoid in this species at a concentration of 371.0 mg/kg of dry weight [48].
C. pepo was found to be very weak in polyphenol content. Only 0.02 mg GAE/100 mg sample has been found in its fresh fruit by Mongkolsilp et al. [49]. However, Iswaldi et al. [50] have reported for the first time a list of 34 polyphenols including a variety of flavonoids in the fruit of C. pepo., in addition to other unknown polar compounds. Besides, the flowers of C. pepo may contain considerable amount of phenolic compounds. Andjelkovic et al. [51] studied the phenolic content of six pumpkin (C. pepo) seed oils and identified the following compounds: Tyrosol, vanillic acid, vanillin, ferulic acid and luteolin. Among them, tyrosol was the most abundant compound ranging from 1.6 mg/kg to 17.7 mg/kg.
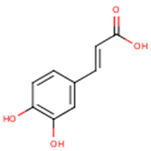
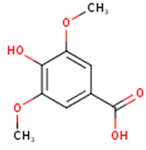
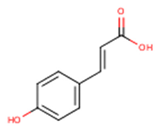
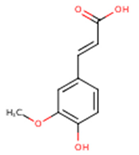
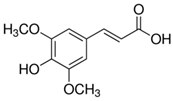
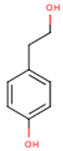
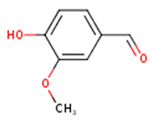
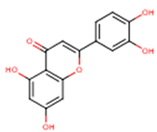
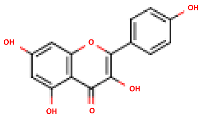
Peričin et al. [52] studied the phenolic acid content of C. pepo seeds. p-Hydroxybenzoic acid was found to be the prevailing phenolic acid, with 34.72%, 67.38% and 51.80% of the total phenolic acid content in whole dehulled seed, kernels and hulls, respectively. Aside from p-hydroxybenzoic acid, the most dominant phenolic compounds can be listed in a decreasing order of quantity as follows: Caffeic, ferulic and vanillic acids in whole dehulled seeds. Trans-synapic and protocatechuic acids, and p-hydroxybenzaldehyde were the abundant phenolic acids presented in the kernels of hulled pumpkin variety; the hulls comprised p-hydroxybenzaldehyde, vanillic and protocatechuic acids with considerable amounts. Table 2 presents the main phenolic compounds found in the Cucurbita spp. and their structures.

Table 2.
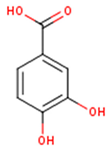
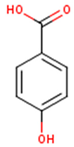
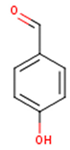
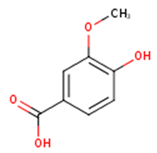
Main chemical structures of the phenolic compounds found in the Cucurbita spp.*.
4. Looking at Cucurbita Plants Biological Activity
4.1. Antimicrobial Activity of Cucurbita Plants
4.1.1. In Vitro Studies
Pumpkin extracts showed a positive activity towards bacterial and fungal infections. They were effective against gram-positive: Staphylococcus aureus, Bacillus subtilis, as well as gram-negative bacterium: Escherichia coli, Proteus vulgaris, Pseudomonas aeruginosa, Salmonella spp. or Klebsiella spp. Pumpkin extracts also showed antibacterial activity against water borne bacteria Vibrio cholerae as well as intestinal flagellated parasite Giardia lamblia, often isolated from surface water. Other studies documented that pumpkin extracts showed a wide range of antifungal activity against species from the Fusarium, Trichoderma, Aspergillus, Verticillium, Phytophora, Botrytis, Candida and Saccharomyces genera (Table 3). However, the mechanisms of antimicrobial activity of pumpkin extracts are still unknown, although it seems to exist a synergistic action between all extracted bioactive substances. It is well known that plant extracts exert biological effects more prominent than their isolated compounds. In fact, recent evidence has established that, in whole matrices, the major compounds interact with those in trace amounts to potentiate their own potential, to provide additional properties other than those often recommended and even to help counterbalance the side effects of these isolated compounds. In addition, and not the least important to emphasize, is that such minor compounds by strengthening the biological effects of a specific bioactive also reduces the dose required to achieve a similar effect.

Table 3.
Antimicrobial activity of Cucurbita spp. extracts evaluated in vitro.
4.1.2. In Vivo Studies
Not only pumpkin extracts, but also proteins and peptides isolated from Cucurbita spp. were identified and characterized in terms of antimicrobial activity. Three pumpkin proteins inhibited the growth of fungi Fusarium oxysporum, Verticillium dahliae and Saccharomyces cerevisiae [68]. The antifungal peptide—cucurmoschin—isolated from black pumpkin seeds also demonstrated inhibitory activity against mold growth: Botrytis cinerea, F. oxysporum and Mycosphaerella oxysporum [69]. The ribosome-inactivating protein extracted from C. moschata showed an antimicrobial effect towards phytopathogenic fungi Phytophora infestans as well as against bacteria Pseudomonas solanacearum and Erwinia amylovora [70]. Additionally, PR-5 protein isolated from leaves of pumpkin, demonstrated synergism with combination of nikkomycin, a chitin synthase inhibitor, towards to Candida albicans [71]. Protein Pr-1 isolated from pumpkin rind inhibited the growth of plant pathogenic fungi, namely B. cinerea, F. oxysporum, F. solani and Rhizoctonia solani, as well as the opportunistic pathogenic yeast C. albicans [72]. These results demonstrate that the proteins from pumpkin may be of importance to clinical microbiology with a wide range of therapeutic applications (Table 4). As the most prominent ones, and given the current evidence, namely regarding its ability to trigger fungal membranes damages and to improve the plasma membranes permeability, they can be effectively used to combat fungal infections and even to use in combination with current antifungal agents, both to improve its effectiveness and even to reduce its side effects.

Table 4.
In vitro antimicrobial activity of Cucurbita spp. proteins.
Pumpkin pulp, due to its antimicrobial properties, is widely used to relieve intestinal inflammation or stomach disorders [73] (Table 5). Pumpkin and its seeds, in the traditional world medicine, are often employed as an anti-helminthic remedy and for supportive therapy in functional diseases of the bladder as well as in the case of digestion problems. The usage of an extract of C. pepo cortex towards urinary tract infections may correspond to a new source of antibiotics against bacterial urinary tract infections [57]. Other studies represented the importance of oil from seeds of a pumpkin as a hopeful drug for treating wounds in vivo [74]. The researchers demonstrated a premium quality of pumpkin oil with a high quantity of polyunsaturated fatty acids, tocopherols that were able to perform efficient wound healing [74]. Morphometric evaluation and histological evidence in rats showed healed biopsies from pumpkin oil and a complete re-epithelialization with a recurrence of skin appendages and well re-growing collagen fibers out of cells inflammation.

Table 5.
Antimicrobial property of Cucurbita spp. and its importance in vivo.
Pumpkin-based foodstuff is well recognized as a source of anti-inflammatory remedies, which can be useful in arthritis treatment [75]. Pumpkin seed oil notably prevent adjuvant-induced arthritis in rats, similar to indomethacin, a well-known anti-inflammatory substance. Its clinical applicability as an antioxidant was also assessed on rheumatoid arthritis [76] and recently confirmed by Dixon [77].
4.2. Anticancer Activities of Cucurbita Plants
Cucurbitacins are a distinct class of triterpenoids characterized by a cucurbitane-based structure, which contributes to their diverse biological activities, particularly their anticancer potential. Cucurbitacins have been identified as major secondary metabolites within the Cucurbitaceae family. They possess a biogenetically derived 10α-cucurbit-5-ene [19(10→19β) abeo-10α-lanostane] skeleton, which is closely linked to their cytotoxic effects. Several studies have attributed both in vitro and in vivo cytotoxic effects to cucurbitacins [79,80]. Jayaprakasam et al. [81] demonstrated the anticancer properties of cucurbitacins B, D, E and I, isolated from Cucurbita andreana Naudin, against colon, breast, lung, and central nervous system cancer cell lines. Among these, cucurbitacin B has been extensively investigated, with multiple studies confirming its efficacy in various cancer models, including in vivo tumor xenografts [82,83,84,85]. The precise mechanisms underlying its anticancer activity remain debated. The suppression of the oncogene Signal Transducer and Activator of Transcription 3 (STAT3) appears to play a key role in tumor growth inhibition [86], although alternative mechanisms may also contribute. Cancer remains a leading cause of mortality, accounting for approximately 12% of global deaths. Current therapeutic options include chemotherapy, surgical interventions, and radiation therapy. However, chemotherapy is often limited by drug resistance, toxicity, side effects, and insufficient selectivity for tumor cells [87]. Consequently, there is significant interest in exploring plant-derived bioactive compounds as promising sources for novel anticancer agents.
In Vitro Anticancer/Antitumor Effects
To date, over forty cucurbitacins have been isolated from the Cucurbitaceae family and other related medicinal plants. The proapoptotic effects of cucurbitacins are attributed to their ability to modulate gene expression, transcriptional activity via nuclear signaling, and mitochondrial membrane potential. Additionally, cucurbitacins can either activate or inhibit key apoptotic regulators. They are potent inhibitors of the JAK/STAT signaling pathway and also impact alternative apoptotic pathways, including PARP cleavage, MAPK signaling, and caspase-3 activation. Cucurbitacins have been shown to downregulate JAK3 and pSTAT3 levels, as well as several STAT3-regulated proteins involved in cell cycle progression, such as Bcl-2, Mcl-1, cyclin D3, and Bcl-xL [88]. C. pepo alcohol extract demonstrated cytotoxic activity against HepG2 and CT26 cancer cell lines, with IC50 values of 132.6 µg/mL and 167.2 µg/mL, respectively. Similarly, the ethanol extract of C. pepo exhibited a dose-dependent inhibitory effect on HeLa cell proliferation [89]. Cucurbita glycosides A and B, isolated from C. pepo ethanol extract, exhibited cytotoxic activity in vitro against HeLa cells, with IC50 values of 17.2 µg/mL and 28.5 µg/mL, respectively [90]. Cucurbitacins B and E, isolated from C. pepo cv dayangua, demonstrated antiproliferative effects against MCF-7, HCT-116, SF-268, A549, and NCI-H460 cancer cell lines [81]. The antiproliferative effects of 23,24-dihydrocucurbitacin F on human prostate cancer (PCa) cells may be attributed to the induction of cofilin–actin rod formation, leading to actin aggregation, cytokinesis failure, and cell cycle arrest at the G2/M phase, followed by apoptosis [91]. Furthermore, 23,24-dihydrocucurbitacin F has been shown to inhibit Epstein–Barr virus activation, induced by the tumor-promoting agent 12-O-tetradecanoylphorbol-13-acetate (TPA), and exerts significant antitumor-promoting effects in murine skin cancer models [88].
Treatment with cucurbitacins B and E resulted in apoptosis and cell cycle arrest in MDA-MB-231 and MCF-7 breast cancer cell lines. Additionally, these compounds modulated the expression of proteins involved in cell cycle regulation in both estrogen-independent (MDA-MB-231) and estrogen-dependent (MCF-7) human breast cancer cell lines. Cucurbitacin B was found to inhibit tumor growth and exert cytotoxic effects on SKBR-3 and MCF-7 breast cancer cell lines, primarily through G2/M phase arrest and apoptosis. Cucurbitacin B treatment also downregulated the expression of Cyclin D1, c-Myc, and β-catenin and prevented the nuclear translocation of β-catenin and galectin-3. Western blot analysis revealed increased PARP cleavage, suggesting caspase activation, and a reduction in Wnt signaling-related proteins such as galectin-3, β-catenin, c-Myc, and cyclin D1, along with modifications in phosphorylated GSK-3β levels [92].
Cucurbitacin E disrupted the cytoskeletal architecture of actin and vimentin, thereby inhibiting prostate cancer cell proliferation. Additionally, cucurbitacins suppressed endothelial cell proliferation by disrupting F-actin and tubulin microfilaments. Moreover, they exhibited antiangiogenic and antimetastatic properties by reducing T-lymphocyte proliferation and cellular motility [93]. Current research suggests that secondary metabolites from C. pepo possess significant anticancer activity, highlighting their potential role in the development of novel chemotherapeutic agents for tumor prevention and treatment.
5. Clinical Effectiveness of Cucurbita Plants in Humans
5.1. Control of Blood Glucose Level in Diabetic Patients
Diabetes mellitus is a chronic disease characterized by changes in saccharide, lipid and protein metabolism resulting from a deficiency in insulin secretion from the pancreas, insulin resistance or both. The main clinical symptom is represented by increased blood sugar levels (hyperglycemia) that uncontrolled lead in time to a wide spectrum of complications [94]. Natural therapeutic alternatives to allopathic treatment always attracted the researchers to the intention of finding new drugs with fewer side effects [95,96,97,98,99,100]. Thus, the hypoglycemic effect of Cucurbita species (Table 6) is known and used for long traditional medicine in many countries, like China, India, Iran and Mexico [101,102,103,104].

Table 6.
Pharmacotherapeutic effects of Cucurbita plants in human clinical studies.
Mahmoodpoor et al. [106] in a recent study performed on patients with severe diabetes from the Intensive Care Unit showed the hypoglycemic effect of C. maxima pulp. The subjects received five grams of C. maxima powder per 12 h for three consecutive days. After the treatment, it was observed a decrease of serum glucose levels from 214.9 mg/dL to 214.9 mg/dL associated with a reduction of insulin doses from 48.05 IU to 39.5 IU [106]. C. ficifolia also showed a good hypoglycemic effect when the extract was administered in doses of 4 mL/kg to patients with type 2 diabetes and moderately elevated blood glucose level [105]. Five hours after administration, the mean of serum glucose level decreased from 217.2 mg/dL to 150.8 mg/dL [105].
The most important hypoglycemic active substances in pumpkin are non-pectines polysaccharides and pectines from pulp, proteins and oil obtained from seeds [107,108,109]. Alenazi et al. [118] reported a clinical case of a 12-year-old Asian diabetic patient that ate every day for four months 200 g of pumpkin. After two months of daily pumpkin consumption, a decrease of glycosylated hemoglobin (HbA1C) from 10.8% to 8.5% was observed [118]. The same positive hypoglycemic effect was also revealed in another study by Jain et al. [119]. Fourteen patients diagnosed with type 2 diabetes received C. ficifolia juice for 40 days, and glycosylated hemoglobin decreased with 22.5% [119]. Shi et al. [120] investigated the antidiabetic activity of pumpkin carbohydrate granules in patients with type 2 diabetes compared to a control placebo group. After one month of treatment, both blood and urine glucose levels were significantly decreased compared with the placebo control group [120]. The results of a randomized, placebo-controlled trial conducted showed that a rich diet in pumpkin (C. maxima) seeds significantly reduced postprandial blood glucose of adults with normal glycaemia [121]. This study included 25 normoglycemic adults who consumed daily 65 g of pumpkin seeds [121]. Possible mechanisms of antihyperglycemic action of Cucurbita species are not fully understood but several studies investigated this subject in the last decades. Zhang et al. [122] demonstrated that C. moschata heteropolysaccharides regenerate pancreatic islets by stimulating proliferation of pancreatic β-cells. Quanhong et al. [123] showed that polysaccharides bounded by protein (polysaccharide 41.21% and protein 10.13%) increase glucose tolerance level and reduce hyperglycemia. In the light of these results, supplements with natural extracts from Cucurbita plants can be considered as alternative hypoglycemic products and further multicenter randomized studies can confirm these results.
5.2. Pharmacotherapeutic Effects in Low Urinary Tract Diseases
Benign prostatic hyperplasia (BPH) represents an increase in the volume of the prostate under the influence of androgenic hormones, and 70% of aging men suffer from this condition. Since clinical evolution of urinary signs is slow, prevention of BPH is useful, phytotherapy being an alternative way [124]. For example, oil obtained from C. pepo seeds is traditionally used to treat urinary symptoms in BPH as the daily frequency of urination, nycturia, time of the bladder emptying and residual volume [110,111]. The main mechanism through which these effects are obtained is represented by the inhibition of 5-α-reductase. This enzyme is required to convert testosterone to dihydrotestosterone, which has a higher affinity than testosterone for androgen receptors. As a result, protein synthesis increases the volume of the prostate implicitly [113].
In a multicenter clinical trial, thousands of patients diagnosed with BPH were treated with capsules containing 500 mg of C. pepo seeds extracts. Their quality of life has been significantly improved by reducing the urinary symptoms of BPH [125].
Other modern studies have shown pharmacotherapeutic synergism in BPH when C. Pepo is administrated simultaneously with other plants. Thus, the combination with Serenoa repens (W. Bartram) Small significantly improved the urinary symptoms of BPH and decreased blood dihydrotestosterone levels [111]. Hong et al. [112] obtained similar results on urinary symptoms in Korean men with BPH treated with 320 mg of C. pepo plus 320 mg of S. repens. They also observed a decrease in prostatic antigen levels after the treatment, but without changes in prostate volume [112]. In a randomized Phase II clinical trial carried out by Coulson et al. [126] the efficacy of the ProstateEZE Max formulation obtained from a mixture of plants traditionally used in treating BPH was evaluated. ProstateEZE is a natural formulation containing C. pepo, S. repens, Pygeum africanum Hook.f., E. parviflorum Schreb. and lycopene. Fifty-seven male patients diagnosed with BPH were selected in the study. Thirty-two of them received a capsule of ProstateEZE Max daily for three consecutive months, and 25 patients were treated with a placebo. In patients treated with Prostate EZE, the clinical symptoms of BPH decreased by 35.9% compared with only 8.3% for the placebo. The frequency of nocturnal urination was reduced with 39.3% in subjects treated for three months with ProstateEZE compared to the placebo group [126].
Due to these beneficial therapeutic effects of Cucurbita plants in BPH, the European Medicines Agency approved the use of C. pepo for both BPH and other bladder disorders, such as urinary stress incontinence in women [127].
Urinary stress incontinence occurs when pelvic muscles that support the bladder and the sphincter muscle, which controls the urinary flow, are weakened. This disorder is associated with aging in women. The main symptom is urinary incontinence [115]. The seeds extract of C. pepo have a therapeutic effect in this condition through a double mechanism. Directly by relaxing the bladder muscles leading to a decrease in nycturia and indirectly through a hormonal mechanism by inhibiting 5-α reductase. This inhibition determines the anabolic effects that strengthen the bladder sphincter muscles [115,127]. The main chemical compounds in the pumpkin seeds that explain these effects are sterols (sitosterol, spinasterol) and fatty oil, which contain oleic, linoleic, palmitic acids and tocopherol) [114]. Gažová et al. [128] demonstrated these effects in a study of 86 women with urinary incontinence stress who were treated for twelve weeks with the preparation of a plant mix: C. pepo, Equisetum arvense L. and Linum usitatissimum L. Episodes of urinary incontinence during the day were reduced to 35% and nocturnal urinary frequency to 54% [128].
Overactive bladder syndrome (OAB) is characterized by the frequent urge to urinate during the day and night, followed by an involuntary loss of urine [116]. A human clinical trial conducted by Shim et al. (2014) investigated the efficacy and utility of Cucuflavone (tablets with a mixture of plant extracts 87.5% C. pepo seeds and 12.5% soy) in reducing OAB symptoms [116]. The active compounds of Cucuflavone are phenols (pyrogallol) and isoflavones (genistein, daidzin). One hundred and twenty patients were included in the study, divided into two groups: The Cucuflavone group and the placebo group. Patients from Cucuflavone group received two tablets twice a day (a total of 875 mg of C. pepo seed extract and 125 mg of soy extract daily) for twelve weeks. The final results of the investigation showed that urinary incontinence, the frequency of daily and nocturnal urination was statistically significantly reduced compared to the initial parameters [116]. In a recent investigation, Nishimura et al. obtained similar results. They confirmed the efficacy of C. maxima seeds oil on urinary disorders in OAB. Forty-five subjects with OAB were included and treated daily with 10 g of C. maxima seed oil for twelve weeks. At the end of the investigation, the frequency of average daily urination was reduced from 10.96 to 8.00 [117].
6. Conclusions and Future Perspectives
In short, the use of Cucurbita species and their active constituents in various clinical and pharmacological studies revealed the presence of multiple, effective and useful compounds, which provide the opportunity for further production of antidiabetic, analgesic, anti-inflammatory and cardioprotective drugs and foods. Indeed, the use of Cucurbita plants in the treatment of several diseases, including gastrointestinal disorders, intestinal parasites and hypertension, dates from a long time ago. The antimicrobial and antioxidant properties of these species have triggered a huge interest for multiple applications. First of all, free radicals are generated through various metabolic activities in the body, ultimately resulting in various deleterious diseases [99]. These diseases can be treated by supplementation of cucurbits as activities of some cucurbits are comparable with commercially available antibiotics. The present review markedly highlights that Cucurbita species have preventive and therapeutic abilities for treatment of different diseases. The presence of active phytochemicals in Cucurbita species further strengthens the opportunity for their application as an upcoming anticancer, antidiabetic, analgesic, anti-inflammatory and cardioprotective drugs, as well as foods. Finally, and not the least important, the application of Cucurbitaceae members in public health, as nutraceuticals is associated with great availability and a good safety profile.
Author Contributions
All authors contributed equally to this work. J.S.-R., M.M., N.M., and R.C., critically reviewed the manuscript. All the authors read and approved the final manuscript.
Funding
This research received no external funding.
Acknowledgments
This work was supported by CONICYT PIA/APOYO CCTE AFB170007. N. Martins would like to thank the Portuguese Foundation for Science and Technology (FCT-Portugal) for the Strategic project ref. UID/BIM/04293/2013 and “NORTE2020—Northern Regional Operational Program” (NORTE-01-0145-FEDER-000012) and C.F.R. for the project UID/EQU/00511/2019—Laboratory for Process Engineering, Environment, Biotechnology, and Energy—LEPABE funded by national funds through FCT/MCTES (PIDDAC).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Marie-Magdeleine, C.; Hoste, H.; Mahieu, M.; Varo, H.; Archimede, H. In vitro effects of Cucurbita moschata seed extracts on Haemonchus contortus. Vet. Parasitol. 2009, 161, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.M.; Yang, S.T. A preliminary study on the cultivating technique of Cucurbita pepo cv Dayangua. Spec. Econ. Amin. Plant 2000, 3, 28–34. [Google Scholar]
- Adnan, M.; Gul, S.; Batool, S.; Fatima, B.; Rehman, A.; Yaqoob, S.; Shabir, H.; Yousaf, T.; Mussarat, S.; Ali, N.; et al. A review on the ethnobotany, phytochemistry, pharmacology and nutritional composition of Cucurbita pepo L. J. Phytopharm. 2017, 6, 133–139. [Google Scholar]
- Andolfo, G.; Di Donato, A.; Darrudi, R.; Errico, A.; Aiese Cigliano, R.; Ercolano, M.R. Draft of Zucchini (Cucurbita pepo L.) Proteome: A Resource for Genetic and Genomic Studies. Front. Genet. 2017, 8, 181. [Google Scholar] [CrossRef] [PubMed]
- Dubey, S.D. Overview on Cucurbita maxima. Int. J. Phytopharm. 2012, 2, 68–71. [Google Scholar] [CrossRef]
- Paris, H.S. Historical records, origins, and development of the edible cultivar groups of Cucurbita pepo (Cucurbitaceae). Econ. Bot. 1989, 43, 423–443. [Google Scholar] [CrossRef]
- Ratnam, N. A review on Cucurbita pepo. Int. J. Pharm. Phytochem. Res. 2017, 9, 1190–1194. [Google Scholar] [CrossRef]
- Salehi, B.; Valussi, M.; Jugran, A.K.; Martorell, M.; Ramírez-Alarcón, K.; Stojanović-Radić, Z.Z.; Antolak, H.; Kręgiel, D.; Mileski, K.S.; Sharifi-Rad, M.; et al. Nepeta species: From farm to food applications and phytotherapy. Trends Food Sci. Technol. 2018, 80, 104–122. [Google Scholar] [CrossRef]
- Mishra, A.P.; Sharifi-Rad, M.; Shariati, M.A.; Mabkhot, Y.N.; Al-Showiman, S.S.; Rauf, A.; Salehi, B.; Župunski, M.; Sharifi-Rad, M.; Gusain, P.; et al. Bioactive compounds and health benefits of edible Rumex species-A review. Cell. Mol. Biol. 2018, 64, 27–34. [Google Scholar] [CrossRef]
- Fapohunda, S.; Adewumi, A.; Jegede, D. Cucurbitaceae - the family that nourishes and heals. MicroMedicine 2018, 6, 85–93. [Google Scholar]
- Phillips, K.M.; Ruggio, D.M.; Ashraf-Khorassani, M. Phytosterol composition of nuts and seeds commonly consumed in the United States. J. Agric. Food Chem. 2005, 53, 9436–9445. [Google Scholar] [CrossRef] [PubMed]
- Ryan, E.; Galvin, K.; O’Connor, T.P.; Maguire, A.R.; O’Brien, N.M. Phytosterol, squalene, tocopherol content and fatty acid profile of selected seeds, grains, and legumes. Plant Foods Hum. Nutr. 2007, 62, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Applequist, W.L.; Avula, B.; Schaneberg, B.T.; Wang, Y.H.; Khan, I.A. Comparative fatty acid content of seeds of four Cucurbita species grown in a common (shared) garden. J. Food Compos. Anal. 2006, 19, 606–611. [Google Scholar] [CrossRef]
- Sabudak, T. Fatty acid composition of seed and leaf oils of pumpkin, walnut, almond, maize, sunflower and melon. Chem. Nat. Compd. 2007, 43, 465–467. [Google Scholar] [CrossRef]
- Stevenson, D.G.; Eller, F.J.; Wang, L.; Jane, J.L.; Wang, T.; Inglett, G.E. Oil and tocopherol content and composition of pumpkin seed oil in 12 cultivars. J. Agric. Food Chem. 2007, 55, 4005–4013. [Google Scholar] [CrossRef] [PubMed]
- Glew, R.H.; Glew, R.S.; Chuang, L.T.; Huang, Y.S.; Millson, M.; Constans, D.; Vanderjagt, D.J. Amino acid, mineral and fatty acid content of pumpkin seeds (Cucurbita spp) and Cyperus esculentus nuts in the Republic of Niger. Plant Foods Hum. Nutr. 2006, 61, 49–54. [Google Scholar] [CrossRef]
- Talukdar, S.N.; Hossain, M.N. Phytochemical, Phytotherapeutical and Pharmacological Study of Momordica dioica. Evid.-Based Complement. Altern. Med. 2014, 2014, 806082. [Google Scholar] [CrossRef]
- Vijayakumar, M.; Eswaran, M.B.; Ojha, S.K.; Rao, C.V.; Rawat, A.K.S. Antiulcer activity of hydroalcohol extract of Momordica dioica roxb. Fruit. Indian J. Pharm. Sci. 2011, 73, 572–577. [Google Scholar] [CrossRef]
- Chandrasekar, B.; Mukherjee, B.; Mukherjee, S.K. Blood sugar lowering potentiality of selected Cucurbitaceae plants of Indian origin. Indian J. Med Res. 1989, 90, 300–305. [Google Scholar] [PubMed]
- Huseini, H.F.; Darvishzadeh, F.; Heshmat, R.; Jafariazar, Z.; Raza, M.; Larijani, B. The clinical investigation of Citrullus colocynthis (L.) schrad fruit in treatment of type II diabetic patients: A randomized, double blind, placebo-controlled clinical trial. Phytother. Res. 2009, 23, 1186–1189. [Google Scholar] [CrossRef]
- Rashidi, A.A.; Mirhashemi, S.M.; Taghizadeh, M.; Sarkhail, P. Iranian medicinal plants for diabetes mellitus: A systematic review. Pak. J. Biol. Sci. 2013, 16, 401–411. [Google Scholar]
- Jain, A.; Singhai, A.K. Effect of Momordica dioica Roxb on gentamicin model of acute renal failure. Nat. Prod. Res. 2010, 24, 1379–1389. [Google Scholar] [CrossRef]
- Menendez-Baceta, G.; Aceituno-Mata, L.; Molina, M.; Reyes-García, V.; Tardío, J.; Pardo-De-Santayana, M. Medicinal plants traditionally used in the northwest of the Basque Country (Biscay and Alava), Iberian Peninsula. J. Ethnopharmacol. 2014, 152, 113–134. [Google Scholar] [CrossRef]
- Peter, E.L.; Rumisha, S.F.; Mashoto, K.O.; Malebo, H.M. Ethno-medicinal knowledge and plants traditionally used to treat anemia in Tanzania: A cross sectional survey. J. Ethnopharmacol. 2014, 154, 767–773. [Google Scholar] [CrossRef]
- Kujawska, M.; Pieroni, A. Plants used as food and medicine by polish migrants in Misiones, Argentina. Ecol. Food Nutr. 2015, 54, 255–279. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Morvin Yabesh, J.E.; Prabhu, S.; Manikandan, R.; Muralidharan, B. Quantitative ethnomedicinal study of plants used in the Nelliyampathy hills of Kerala, India. J. Ethnopharmacol. 2015, 161, 238–254. [Google Scholar] [CrossRef] [PubMed]
- Mahomoodally, M.F.; Mootoosamy, A.; Wambugu, S. Traditional therapies used to manage diabetes and related complications in Mauritius: A comparative ethnoreligious study. Evid.-Based Complement. Altern. Med. 2016, 2016, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Motti, R.; Motti, P. An ethnobotanical survey of useful plants in the agro Nocerino Sarnese (Campania, southern Italy). Hum. Ecol. 2017, 45, 865–878. [Google Scholar] [CrossRef]
- Nayak, D.; Ashe, S.; Rauta, P.R.; Nayak, B. Assessment of antioxidant, antimicrobial and anti-osteosarcoma potential of four traditionally used Indian medicinal plants. J. Appl. Biomed. 2017, 15, 119–132. [Google Scholar] [CrossRef]
- Ramzan, S.; Soelberg, J.; Jäger, A.K.; Cantarero-Arévalo, L. Traditional medicine among people of Pakistani descent in the capital region of Copenhagen. J. Ethnopharmacol. 2017, 196, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Agyare, C.; Spiegler, V.; Asase, A.; Scholz, M.; Hempel, G.; Hensel, A. An ethnopharmacological survey of medicinal plants traditionally used for cancer treatment in the Ashanti region, Ghana. J. Ethnopharmacol. 2018, 212, 137–152. [Google Scholar] [CrossRef] [PubMed]
- Balcha, A. Medicinal plants used in traditional medicine by Oromo people, Ghimbi District, Southwest Ethiopia. J. Ethnobiol. Ethnomed. 2014, 10, 1–15. [Google Scholar]
- Alonso-Castro, A.J.; Dominguez, F.; Zapata-Morales, J.R.; Carranza-Alvarez, C. Plants used in the traditional medicine of Mesoamerica (Mexico and Central America) and the Caribbean for the treatment of obesity. J. Ethnopharmacol. 2015, 175, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Rigat, M.; Vallès, J.; Dambrosio, U.; Gras, A.; Iglésias, J.; Garnatje, T. Plants with topical uses in the Ripollès district (Pyrenees, Catalonia, Iberian Peninsula): Ethnobotanical survey and pharmacological validation in the literature. J. Ethnopharmacol. 2015, 164, 162–179. [Google Scholar] [CrossRef] [PubMed]
- Asowata-Ayodele, A.M.; Afolayan, A.J.; Otunola, G.A. Ethnobotanical survey of culinary herbs and spices used in the traditional medicinal system of Nkonkobe Municipality, Eastern Cape, South Africa. South Afr. J. Bot. 2016, 104, 69–75. [Google Scholar] [CrossRef]
- Jaradat, N.A.; Shawahna, R.; Eid, A.M.; Al-Ramahi, R.; Asma, M.K.; Zaid, A.N. Herbal remedies use by breast cancer patients in the West Bank of Palestine. J. Ethnopharmacol. 2016, 178, 1–8. [Google Scholar] [CrossRef]
- Meragiaw, M.; Asfaw, Z.; Argaw, M. The Status of Ethnobotanical Knowledge of Medicinal Plants and the Impacts of Resettlement in Delanta, Northwestern Wello, Northern Ethiopia. Evid.-Based Complement. Altern. Med. 2016, 2016, 5060247. [Google Scholar] [CrossRef]
- Odoh, U.E.; Uzor, P.F.; Eze, C.L.; Akunne, T.C.; Onyegbulam, C.M.; Osadebe, P.O. Medicinal plants used by the people of Nsukka Local Government Area, south-eastern Nigeria for the treatment of malaria: An ethnobotanical survey. J. Ethnopharmacol. 2018, 218, 1–15. [Google Scholar] [CrossRef]
- Suroowan, S.; Mahomoodally, M.F. A comparative ethnopharmacological analysis of traditional medicine used against respiratory tract diseases in Mauritius. J. Ethnopharmacol. 2016, 177, 61–80. [Google Scholar] [CrossRef]
- Peiretti, P.G.; Meineri, G.; Gai, F.; Longato, E.; Amarowicz, R. Antioxidative activities and phenolic compounds of pumpkin (Cucurbita pepo) seeds and amaranth (Amaranthus caudatus) grain extracts. Nat. Prod. Res. 2017, 31, 2178–2182. [Google Scholar] [CrossRef] [PubMed]
- Azevedo-Meleiro, C.H.; Rodriguez-Amaya, D.B. Qualitative and quantitative differences in carotenoid composition among Cucurbita moschata, Cucurbita maxima, and Cucurbita pepo. J. Agric. Food Chem. 2007, 55, 4027–4033. [Google Scholar] [CrossRef] [PubMed]
- Maria, L.; Carvalho, J.D.; Barros, P.; Luiz, R.; Godoy, D.O.; Pacheco, S.; Henrique, P.; Luiz, J.; Carvalho, V.D.; Regini, M.; et al. Total carotenoid content, α-carotene and β-carotene, of landrace pumpkins (Cucurbita moschata Duch): A preliminary study. Food Res. Int. 2012, 47, 337–340. [Google Scholar]
- Perez Gutierrez, R.M. Review of Cucurbita pepo (Pumpkin) its Phytochemistry and Pharmacology. Med. Chem. 2016, 6, 12–21. [Google Scholar] [CrossRef]
- Chandrika, U.G.; Basnayake, B.M.L.B.; Athukorala, I.; Colombagama, P.W.N.M.; Goonetilleke, A. Carotenoid Content and In Vitro Bioaccessibility of Lutein in Some Leafy Vegetables Popular in Sri Lanka. J. Nutr. Sci. Vitaminol. 2010, 56, 203–207. [Google Scholar] [CrossRef]
- Mi, Y.K.; Eun, J.K.; Young-Nam, K.; Changsun, C.; Bo-Hieu, L. Comparison of the chemical compositions and nutritive values of various pumpkin (Cucurbitaceae) species and parts. Nutr. Res. Pract. 2012, 6, 21–27. [Google Scholar] [CrossRef]
- Yang, R.Y.; Lin, S.; Kuo, G. Content and distribution of flavonoids among 91 edible plant species. Asia Pac. J. Clin. Nutr. 2008, 17, 275–279. [Google Scholar]
- Sreeramulu, D.; Raghunath, M. Antioxidant activity and phenolic content of roots, tubers and vegetables commonly consumed in India. Food Res. Int. 2010, 43, 1017–1020. [Google Scholar] [CrossRef]
- Koo, M.H.; Suhaila, M. Flavonoid (Myricetin, Quercetin, Kaempferol, Luteolin and Apigenin) Content of Edible Tropical Plants. J. Agric. Food Chem. 2001, 49, 3106–3112. [Google Scholar]
- Mongkolsilp, S.; Pongbupakit, I.; Sae-Lee, N.; Sitthihaworm, W.; Article, O. Radical Scavenging Activity and Total Phenolic Content of Medicinal Plants Used in Primary Health Care Savitree Mongkolsilp, Isara Pongbupakit, Nittaya Sae-Lee and Worapan Sitthithaworn. Swu J. Pharm. Sci. 2004, 9, 32–35. [Google Scholar]
- Iswaldi, I.; Gómez-Caravaca, A.M.; Lozano-Sánchez, J.; Arráez-Román, D.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Profiling of phenolic and other polar compounds in zucchini (Cucurbita pepo L.) by reverse-phase high-performance liquid chromatography coupled to quadrupole time-of-flight mass spectrometry. Food Res. Int. 2013, 50, 77–84. [Google Scholar] [CrossRef]
- Andjelkovic, M.; Van Camp, J.; Trawka, A.; Verhé, R. Phenolic compounds and some quality parameters of pumpkin seed oil. Eur. J. Lipid Sci. Technol. 2010, 112, 208–217. [Google Scholar] [CrossRef]
- Peričin, D.; Krimer, V.; Trivić, S.; Radulović, L. The distribution of phenolic acids in pumpkin’s hull-less seed, skin, oil cake meal, dehulled kernel and hull. Food Chem. 2009, 113, 450–456. [Google Scholar] [CrossRef]
- El-Kamali, H.H.; Mahjoub, S.A.T. Antibacterial activity of Francoeuria crispa, Pulicaria undulata, Ziziphus spina-christi and Cucurbita pepo against seven standard pathogenic bacteria. Ethnobot. Leafl. 2009, 13, 722–733. [Google Scholar]
- Dubey, A.; Mishra, N.; Singh, N. Antimicrobial activity of some selected vegetables. Int. J. Appl. Biol. Pharm. Technol. 2010, 1, 994–999. [Google Scholar]
- Sood, A.; Kaur, P.; Gupta, R. Phytochemical screening and antimicrobial assay of various seeds extract of Cucurbitaceae family. Int. J. Appl. Biol. Pharm. Technol. 2012, 3, 401–409. [Google Scholar]
- Grzybek, M.; Kukula-Koch, W.; Strachecka, A.; Jaworska, A.; Phiri, A.M.; Paleolog, J.; Tomczuk, K. Evaluation of anthelmintic activity and composition of pumpkin (Cucurbita pepo L.) seed extracts—in vitro and in vivo studies. Int. J. Mol. Sci. 2016, 17, 1456. [Google Scholar] [CrossRef]
- Al-Ghazal, A.T. Evaluation of Antibacterial Effect of Cucurbita pepo (Yakten) Extracts on Multi-antibiotic Resistance Bacterial Strains Isolated From Human Urinary Tract Infections. Rafidain J. Sci. 2012, 23, 1–7. [Google Scholar]
- Chonoko, U.G.; Rufai, A.B. Phytochemical screening and antibacterial activity of Curbita pepo (Pumpkin) against Staphylococcus aureus and Salmonella typhi. J. Pure Appl. Sci. 2011, 4, 145–147. [Google Scholar]
- Jasim, S.; Alwan, A.N.; Altimimi, H.W.; Kareem, K.H. Evaluation of antimicrobial activity of flavonoids extract from Cucurbita pepo leaves. Bas. J. Vet. Res. 2010, 9, 10–17. [Google Scholar]
- Noumedem, J.A.K.; Mihasan, M.; Lacmata, S.T.; Stefan, M.; Kuiate, J.R.; Kuete, V. Antibacterial activities of the methanol extracts of ten Cameroonian vegetables against Gram-negative multidrug-resistant bacteria. BMC Complement. Altern. Med. 2013, 13, 26. [Google Scholar] [CrossRef]
- Dar, A.H.; Sofi, S.A. Pumpkin the functional and therapeutic ingredient: A review. Int. J. Food Sci. Nutr. 2017, 2, 165–170. [Google Scholar]
- Abed El-Aziz, A.; Abed El-Aziz, H. Antimicrobial proteins and oil seeds from pumpkin. Nat. Sci. 2011, 9, 105–119. [Google Scholar]
- Elhadi, I.M.; Koko, S.W.; Dahab, M.M.; El Imam, J.M.; El Mageed, M.A.E. Antigiardial activity of some Cucurbita species and Lagenaria siceraria. J. For. Prod. Ind. 2013, 2, 43–47. [Google Scholar]
- Muruganantham, N.; Solomon, S.; Senthamilselvi, M.M. Anti-cancer activity of Cucumis sativus (cucumber) flowers against human liver cancer. Int. J. Pharm. Clin. Res. 2016, 8, 39–41. [Google Scholar]
- Geetha, S. Antimicrobial activity of selected vegetable peels against water borne pathogens. Int. J. Adv. Pharm. Biol. Chem. 2014, 3, 937–940. [Google Scholar]
- Kabbashi, A.S.; Koko, W.S.; Mohammed, S.E.A.; Musa, N.; Elbadri, E.; Dahab, M.M.; Mohammed, A.K. In vitro a moebicidal, antimicrobial and antioxidant activities of the plants Adansonia digitata and Cucurbit maxima. Adv. Med. Plant Res. 2014, 2, 50–57. [Google Scholar]
- Ravishankar, K.; Kiranmayi, G.V.N.; Appa Reddy, G.V.; Sowjanya, V.V.L.; Baba Sainadh, V.; Lakshmi, V.G.; Prasad, T. Preliminary phytochemical screening and In-vitro antibacterial activity of Cucurbita maxima seed extract. Int. J. Res. Pharm. Chem. 2012, 2, 86–91. [Google Scholar]
- Cassel, C.K. Policy challenges and clinical practices. Hosp. Pract. 1993, 28, 9–10. [Google Scholar] [CrossRef]
- Wang, H.X.; Ng, T.B. Isolation of cucurmoschin, a novel antifungal peptide abundant in arginine, glutamate and glycine residues from black pumpkin seeds. Peptides 2003, 24, 969–972. [Google Scholar] [CrossRef]
- Barbieri, L.; Polito, L.; Bolognesi, A.; Ciani, M.; Pelosi, E.; Farini, V.; Stirpe, F. Ribosome-inactivating proteins in edible plants and purification and characterization of a new ribosome-inactivating protein from Cucurbita moschata. Biochim. Biophys. Acta 2006, 760, 783–792. [Google Scholar] [CrossRef]
- Cheong, N.E.; Choi, Y.O.; Kim, W.Y.; Bae, I.S.; Cho, M.J.; Hwang, I.; Lee, S.Y. Purification and characterization of an antifungal PR-5 protein from pumpkin leaves. Mol. Cells 1997, 7, 214–219. [Google Scholar]
- Park, S.C.; Lee, J.R.; Kim, J.Y.; Hwang, I.; Nah, J.W.; Cheong, H.; Hahm, K.S. Pr-1, a novel antifungal protein from pumpkin rinds. Biotechnol. Lett. 2009, 32, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Karanja, J.; Mugendi, J.; Muchugi, A.; Karanja, J.K.; Mugendi, B.J.; Khamis, F.M.; Muchugi, A.N. Nutritional evaluation of some kenyan pumpkins (Cucurbita spp.). Int. J. Agric. For. 2016, 4, 195–200. [Google Scholar]
- Bardaa, S.; Ben Halima, N.; Aloui, F.; Ben Mansour, R.; Jabeur, H.; Bouaziz, M.; Sahnoun, Z. Oil from pumpkin (Cucurbita pepo L.) seeds: Evaluation of its functional properties on wound healing in rats. Lipids Health Dis. 2016, 15, 783–792. [Google Scholar] [CrossRef]
- Seo, J.S.; Burri, B.J.; Quan, Z.; Neidlinger, T.R. Extraction and chromatography of carotenoids from pumpkin. J. Chromatogr. A 2005, 1073, 371–375. [Google Scholar] [CrossRef] [PubMed]
- van Vugt, R.M.; Rijken, P.J.; Rietveld, A.G.; van Vugt, A.C.; Dijkmans, B.A.C. Antioxidant intervention in rheumatoid arthritis: Results of an open pilot study. Clin. Rheumatol. 2008, 27, 771–775. [Google Scholar] [CrossRef]
- Dixon, W.G. Rheumatoid arthritis: Biological drugs and risk of infection. Lancet 2015, 386, 224–225. [Google Scholar] [CrossRef]
- Fokou, E.A.M. Preliminary nutritional evaluation of five species of egusi seeds in Cameroon. Afr. J. Food Agric. Nutr. Dev. 2004, 4, 1–11. [Google Scholar] [CrossRef]
- Duncan, K.L.K.; Duncan, M.D.; Alley, M.C.; Sausville, E.A. Cucurbitacin E-induced disruption of the actin and vimentin cytoskeleton in prostate carcinoma cells. Biochem. Pharmacol. 1996, 52, 1553–1560. [Google Scholar] [CrossRef]
- Fang, X.; Phoebe, C.H.; Pezzuto, J.M.; Fong, H.H.; Farnsworth, N.R.; Yellin, B.; Hecht, S.M. Plant anticancer agents, XXXIV. Cucurbitacins from Elaeocarpus dolichostylus. J. Nat. Prod. 1984, 47, 988–993. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakasam, B.; Seeram, N.P.; Nair, M.G. Anticancer and antiinflammatory activities of cucurbitacins from Cucurbita andreana. Cancer Lett. 2003, 189, 11–16. [Google Scholar] [CrossRef]
- Chan, K.T.; Meng, F.Y.; Li, Q.; Ho, C.Y.; Lam, T.S.; To, Y.; Toh, M. Cucurbitacin B induces apoptosis and S phase cell cycle arrest in BEL-7402 human hepatocellular carcinoma cells and is effective via oral administration. Cancer Lett. 2010, 294, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, M.; Zhang, H.; Sun, C.; Deng, Y. Inhibitory effects of cucurbitacin B on laryngeal squamous cell carcinoma. Eur. Arch. Oto-Rhino-Laryngol. 2008, 265, 1225–1232. [Google Scholar] [CrossRef]
- Wakimoto, N.; Yin, D.; O’Kelly, J.; Haritunians, T.; Karlan, B.; Said, J.; Koeffler, H.P. Cucurbitacin B has a potent antiproliferative effect on breast cancer cells in vitro and in vivo. Cancer Sci. 2008, 99, 1793–1797. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, H.; Sun, C.; Shan, X.; Yang, X.; Li-Ling, J.; Deng, Y. Targeted constitutive activation of signal transducer and activator of transcription 3 in human hepatocellular carcinoma cells by cucurbitacin B. Cancer Chemother. Pharmacol. 2009, 63, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.T.; Li, K.; Liu, S.L.; Chu, K.H.; Toh, M.; Xie, W.D. Cucurbitacin B inhibits STAT3 and the Raf/MEK/ERK pathway in leukemia cell line K562. Cancer Lett. 2010, 289, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Herrero, E.; Fernández-Medarde, A. Advanced targeted therapies in cancer: Drug nanocarriers, the future of chemotherapy. Eur. J. Pharm. Biopharm. 2015, 93, 52–79. [Google Scholar] [CrossRef]
- Konoshima, T.; Takasaki, M.; Kozuka, M.; Nagao, T.; Okabe, H.; Irino, N.; Nishino, H. Inhibitory effects of cucurbitane triterpenoids on Epstein-Barr virus activation and two-stage carcinogenesis of skin tumor. Biol. Pharm. Bull. 1994, 18, 284–287. [Google Scholar] [CrossRef]
- Shokrzadeh, M.; Azadbakht, M.; Ahangar, N.; Hashemi, A.; Saravi, S. Cytotoxicity of hydro-alcoholic extracts of Cucurbita pepo and Solanum nigrum on HepG2 and CT26 cancer cell lines. Pharmacogn. Mag. 2010, 6, 176. [Google Scholar] [CrossRef]
- Wang, D.C.; Xiang, H.; Li, D.; Gao, H.; Cai, H.; Wu, L.J.; Deng, X.M. Purine-containing cucurbitane triterpenoids from Cucurbita pepo cv dayangua. Phytochemistry 2008, 69, 1434–1438. [Google Scholar] [CrossRef]
- Ren, S.; Ouyang, D.Y.; Saltis, M.; Xu, L.H.; Zha, Q.B.; Cai, J.Y.; He, X.H. Anti-proliferative effect of 23,24-dihydrocucurbitacin F on human prostate cancer cells through induction of actin aggregation and cofilin-actin rod formation. Cancer Chemother. Pharmacol. 2012, 70, 415–424. [Google Scholar] [CrossRef]
- Dakeng, S.; Duangmano, S.; Jiratchariyakul, W.; U-Pratya, Y.; Bögler, O.; Patmasiriwat, P. Inhibition of Wnt signaling by cucurbitacin B in breast cancer cells: Reduction of Wnt-associated proteins and reduced translocation of galectin-3-mediated β-catenin to the nucleus. J. Cell. Biochem. 2012, 113, 49–60. [Google Scholar] [CrossRef]
- Attard, E.; Cuschieri, A.; Scicluna-Spiteri, A.; Brincat, M.P. The effects of cucurbitacin E on two lymphocyte models. Pharm. Biol. 2004, 42, 170–175. [Google Scholar] [CrossRef]
- Alam, U.; Asghar, O.; Azmi, S.; Malik, R.A. General aspects of diabetes mellitus. Handb. Clin. Neurol. 2014, 126, 211–222. [Google Scholar] [PubMed]
- Sharifi-Rad, M.; Fokou, P.V.T.; Sharopov, F.; Martorell, M.; Ademiluyi, A.O.; Rajkovic, J.; Salehi, B.; Martins, N.; Iriti, M.; Sharifi-Rad, J. Antiulcer agents: From plant extracts to phytochemicals in healing promotion. Molecules 2018, 23, 1751. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.P.; Saklani, S.; Salehi, B.; Parcha, V.; Sharifi-Rad, M.; Milella, L.; Iriti, M.; Sharifi-Rad, J.; Srivastava, M. Satyrium nepalense, a high altitude medicinal orchid of Indian Himalayan region: Chemical profile and biological activities of tuber extracts. Cell. Mol. Biol. 2018, 64, 35–43. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Nazaruk, J.; Polito, L.; Morais-Braga, M.F.B.; Rocha, J.E.; Coutinho, H.D.M.; Salehi, B.; Tabanelli, G.; Montanari, C.; del Mar Contreras, M.; et al. Matricaria genus as a source of antimicrobial agents: From farm to pharmacy and food applications. Microbiol. Res. 2018, 215, 76–88. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Tayeboon, G.S.; Niknam, F.; Sharifi-Rad, M.; Mohajeri, M.; Salehi, B.; Iriti, M.; Sharifi-Rad, M. Veronica persica Poir. extract - antibacterial, antifungal and scolicidal activities, and inhibitory potential on acetylcholinesterase, tyrosinase, lipoxygenase and xanthine oxidase. Cell. Mol. Biol. 2018, 64, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Ozcelik, B.; Altın, G.; Daşkaya-Dikmen, C.; Martorell, M.; Ramírez-Alarcón, K.; Alarcón-Zapata, P.; Morais-Braga, M.F.B.; Carneiro, J.N.P.; Alves Borges Leal, A.L.; et al. Salvia spp. plants-from farm to food applications and phytopharmacotherapy. Trends Food Sci. Technol. 2018, 80, 242–263. [Google Scholar] [CrossRef]
- Salehi, B.; Sharopov, F.; Martorell, M.; Rajkovic, J.; Ademiluyi, A.O.; Sharifi-Rad, M.; Fokou, P.V.T.; Martins, N.; Iriti, M.; Sharifi-Rad, J. Phytochemicals in Helicobacter pylori infections: What are we doing now? Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Caili, F.; Huan, S.; Quanhong, L. A review on pharmacological activities and utilization technologies of pumpkin. Plant Foods Hum. Nutr. 2006, 61, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Andrade-Cetto, A.; Heinrich, M. Mexican plants with hypoglycaemic effect used in the treatment of diabetes. J. Ethnopharmacol. 2005, 99, 325–348. [Google Scholar] [CrossRef]
- Jia, W.; Gao, W.; Tang, L. Antidiabetic herbal drugs officially approved in China. Phytother. Res. 2003, 17, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Maiti, K.; Mukherjee, K.; Houghton, P.J. Leads from Indian medicinal plants with hypoglycemic potentials. J. Ethnopharmacol. 2006, 106, 1–28. [Google Scholar] [CrossRef]
- Acosta-Patiño, J.L.; Jiménez-Balderas, E.; Juárez-Oropeza, M.A.; Díaz-Zagoya, J.C. Hypoglycemic action of Cucurbita ficifolia on Type 2 diabetic patients with moderately high blood glucose levels. J. Ethnopharmacol. 2001, 77, 99–101. [Google Scholar] [CrossRef]
- Mahmoodpoor, A.; Medghalchi, M.; Nazemiyeh, H.; Asgharian, P.; Shadvar, K.; Hamishehkar, H. Effect of Cucurbita maxima on control of blood glucose in diabetic critically ill patients. Adv. Pharm. Bull. 2018, 8, 347–351. [Google Scholar] [CrossRef]
- Adams, G.G.; Imran, S.; Wang, S.; Mohammad, A.; Kok, S.; Gray, D.A.; Harding, S.E. The hypoglycaemic effect of pumpkins as anti-diabetic and functional medicines. Food Res. Int. 2011, 44, 862–867. [Google Scholar] [CrossRef]
- Cai, T.; Li, Q.; Yan, H.; Li, N. Study on the hypoglycemic action of pumpkin seed protein. J. Chin. Inst. Food Sci. Technol. 2003, 3, 7–11. [Google Scholar]
- Xiong, X.; Cao, J. Study of extraction and isolation of effective pumpkin polysaccharide component and its reducing glycemia function. Chin. J. Mod. Appl. Pharm. 2001, 18, 662–664. [Google Scholar]
- Gossell-Williams, M.; Davis, A.; O’Connor, N. Inhibition of testosterone-induced hyperplasia of the prostate of sprague-dawley rats by pumpkin seed oil. J. Med. Food 2006, 9, 284–286. [Google Scholar] [CrossRef]
- PDR for Herbal Medicines, 4th ed.; Thomson Healthcare: Montvale, NJ, USA, 2007.
- Hong, H.; Kim, C.S.; Maeng, S. Effects of pumpkin seed oil and saw palmetto oil in Korean men with symptomatic benign prostatic hyperplasia. Nutr. Res. Pract. 2009, 3, 323. [Google Scholar] [CrossRef] [PubMed]
- Ramak, P.; Mahboubi, M. The beneficial effects of pumpkin (Cucurbita pepo L.) seed oil for health condition of men. Food Rev. Int. 2018, 1–11. [Google Scholar] [CrossRef]
- Rezig, L.; Chouaibi, M.; Msaada, K.; Hamdi, S. Chemical composition and profile characterisation of pumpkin (Cucurbita maxima) seed oil. Ind. Crop. Prod. 2012, 37, 82–87. [Google Scholar] [CrossRef]
- Sogabe, H.; Terado, T. Open clinical study of effects of pumpkin seed extract/soybean germ extract vixture-containing processed food on nocturia. Jpn. J. Med. Pharm. Sci. 2001, 46, 727–737. [Google Scholar]
- Shim, B.; Jeong, H.; Lee, S.; Hwang, S.; Moon, B.; Storni, C. A randomized double-blind placebo-controlled clinical trial of a product containing pumpkin seed extract and soy germ extract to improve overactive bladder-related voiding dysfunction and quality of life. J. Funct. Foods 2014, 8, 111–117. [Google Scholar] [CrossRef]
- Nishimura, M.; Ohkawara, T.; Sato, H.; Takeda, H.; Nishihira, J. Pumpkin seed oil extracted from Cucurbita maxima improves urinary disorder in human overactive bladder. J. Tradit. Complement. Med. 2014, 4, 72–74. [Google Scholar] [CrossRef] [PubMed]
- Alenazi, B.; Deeb, A.; Alrowaili, A.; Alkhaldi, A.; Alanazi, A. Does pumpkin affect glycemic control in diabetic patient. Case report and literature review. Eur. J. Pharm. Med. Res. 2017, 4, 42–45. [Google Scholar]
- Jain, A.; Mishra, M.; Yadav, D.; Khatarker, D.; Jadaun, P.; Tiwari, A.; Prasad, G. Evaluation of the antihyperglycemic, antilipidemic and antioxidant potential of Cucurbita ficifolia in human type 2 diabetes. Prog. Nutr. 2018, 20, 191–198. [Google Scholar]
- Shi, Y.; Xiong, X.; Cao, J.; Kang, M. Effect of pumpkin polysaccharide granules on glycemic control in type 2 diabetes. Cent. South Pharm. 2003, 1, 275–276. [Google Scholar]
- Cândido, F.G.; de Oliveira, F.C.E.; Lima, M.F.C.; Pinto, C.A.; da Silva, L.L.; Martino, H.S.D.; Alfenas, R.C.G. Addition of pooled pumpkin seed to mixed meals reduced postprandial glycemia: A randomized placebo-controlled clinical trial. Nutr. Res. 2018, 56, 90–97. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, P.; Zhang, Y.; Jin, H.; Zhu, L.; Li, J.; Yao, H. Effects of polysaccharide from pumpkin on biochemical indicator and pancreatic tissue of the diabetic rabbits. Int. J. Biol. Macromol. 2013, 62, 574–581. [Google Scholar] [CrossRef]
- Li, Q.; Fu, C.; Rui, Y.; Hu, G.; Cai, T. Effects of protein-bound polysaccharide isolated from pumpkin on insulin in diabetic rats. Plant Foods Hum. Nutr. 2005, 60, 13–16. [Google Scholar]
- Allkanjari, O.; Vitalone, A. What do we know about phytotherapy of benign prostatic hyperplasia? Life Sci. 2015, 126, 42–56. [Google Scholar] [CrossRef] [PubMed]
- Schiebel-Schlosser, G.; Friederich, M. Phytotherapy of BPH with pumpkin seeds-a multicenter clinical trial. Phytotherapy 1998, 19, 71–76. [Google Scholar]
- Coulson, S.; Rao, A.; Beck, S.L.; Steels, E.; Gramotnev, H.; Vitetta, L. A phase II randomised double-blind placebo-controlled clinical trial investigating the efficacy and safety of ProstateEZE Max: A herbal medicine preparation for the management of symptoms of benign prostatic hypertrophy. Complementary Ther. Med. 2013, 21, 172–179. [Google Scholar] [CrossRef]
- Assessment Report on Cucurbita pepo L. Semen; European Medicines Agency: Amsterdam, The Netherlands, 2013.
- Gažová, A.; Valášková, S.; Žufková, V.; Castejon, A.M.; Kyselovič, J. Clinical study of effectiveness and safety of CELcomplex®containing Cucurbita pepo seed extract and flax and casuarina on stress urinary incontinence in women. J. Tradit. Complement. Med. 2018, 9, 138–142. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).