Phytochemical Characterization and Evaluation of the Antimicrobial, Antiproliferative and Pro-Apoptotic Potential of Ephedra alata Decne. Hydroalcoholic Extract against the MCF-7 Breast Cancer Cell Line
Abstract
1. Introduction
2. Results
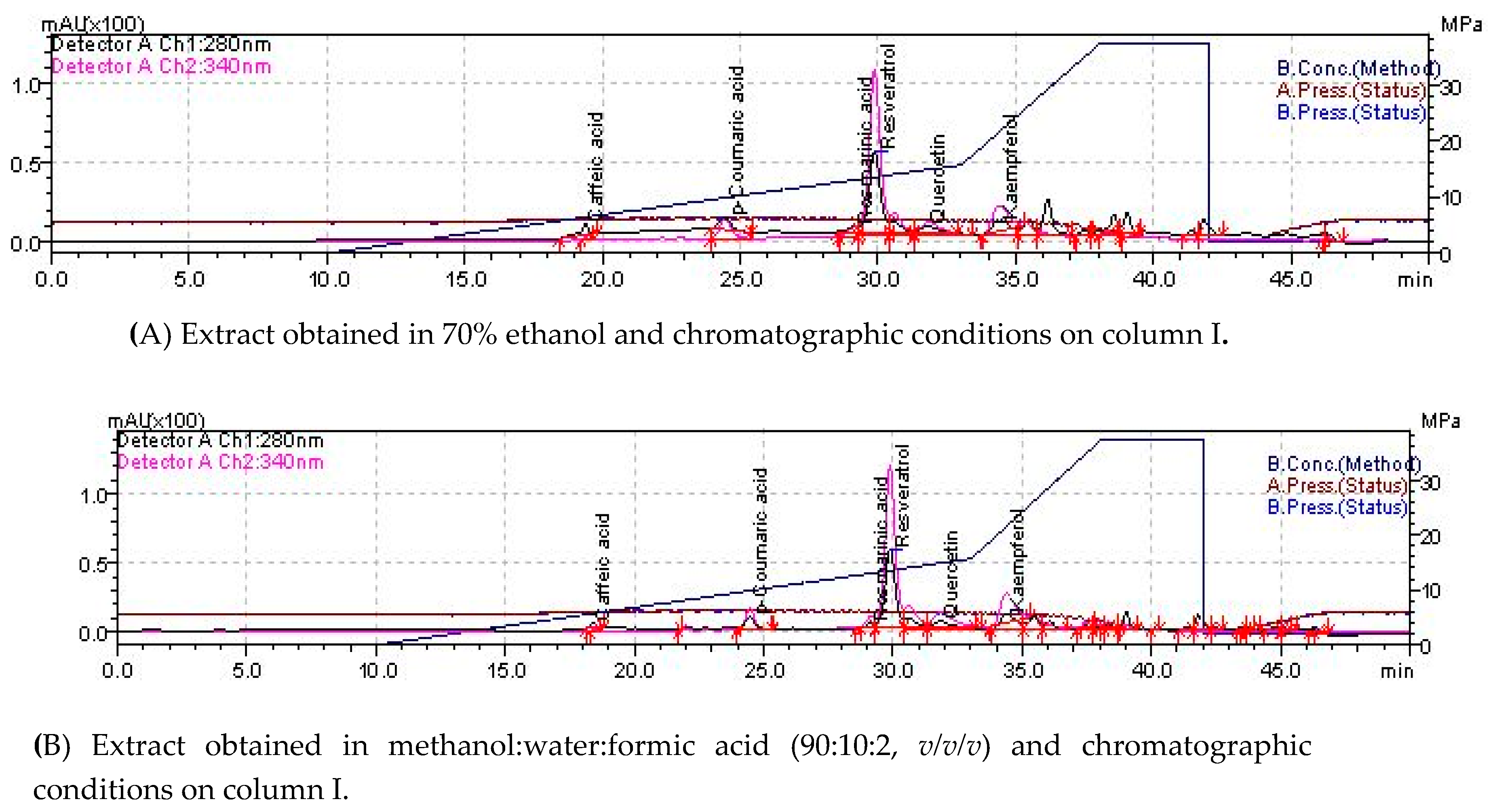
2.1. Phytochemical Composition
2.2. Antimicrobial Activity
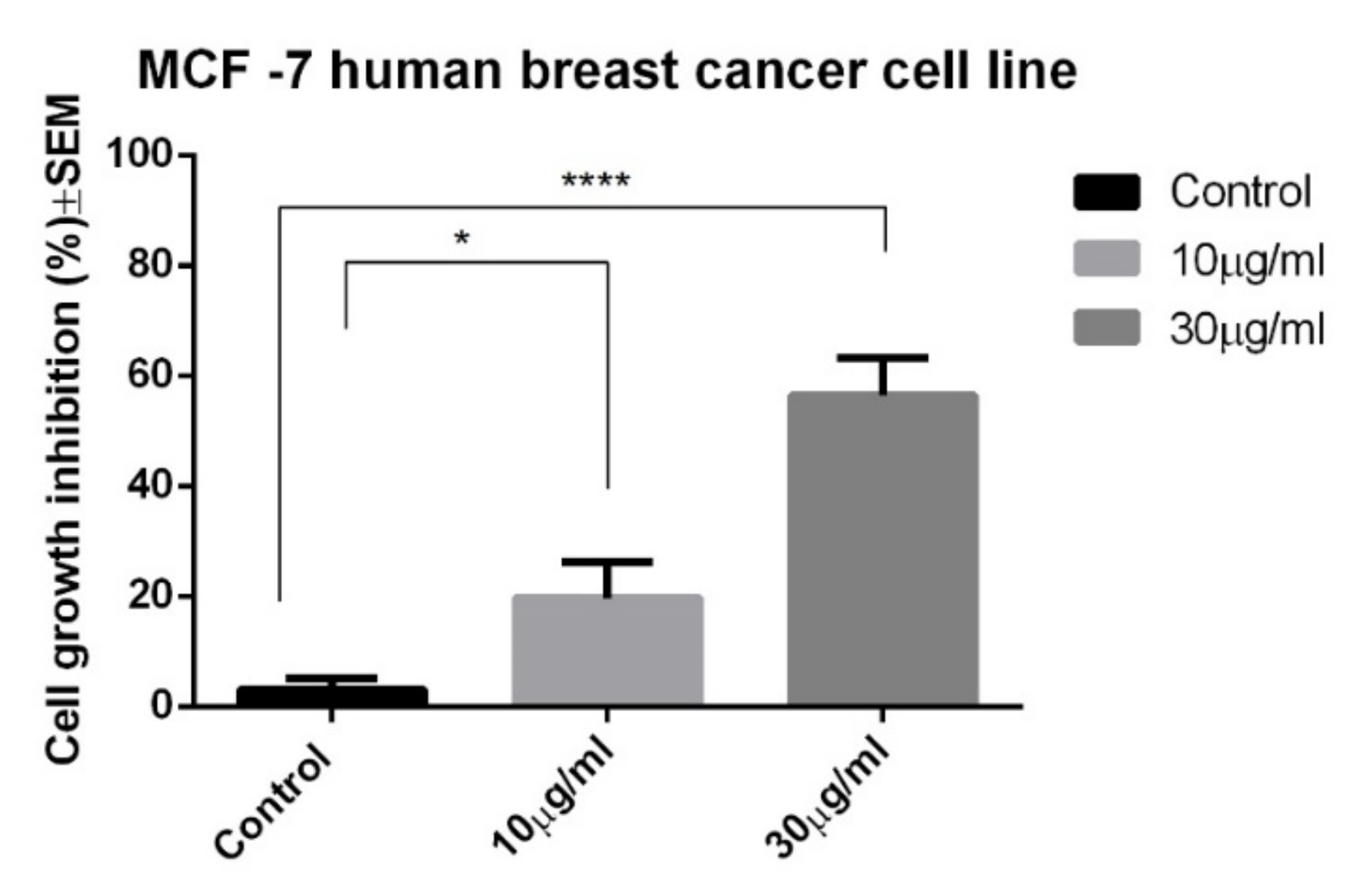
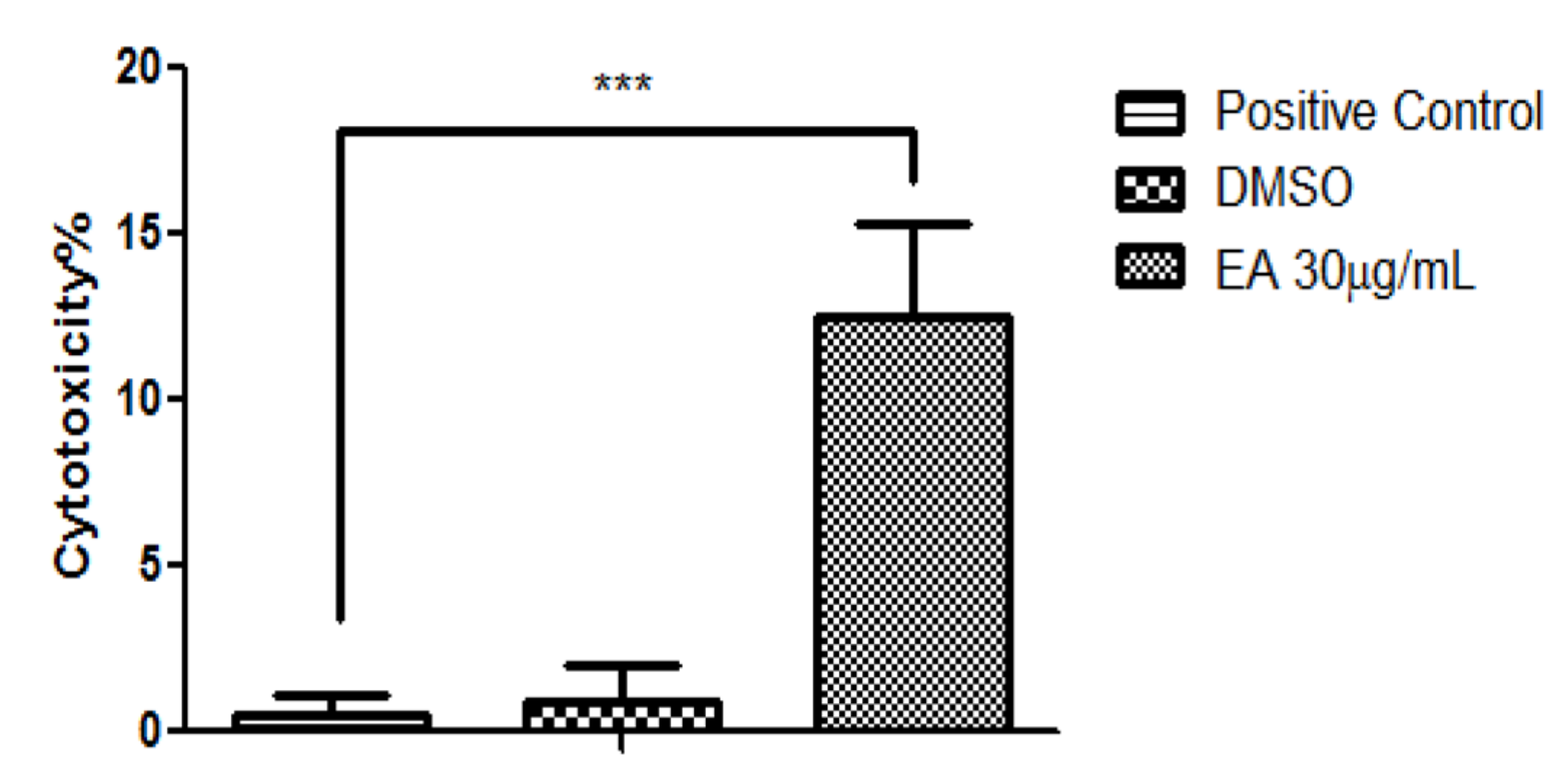
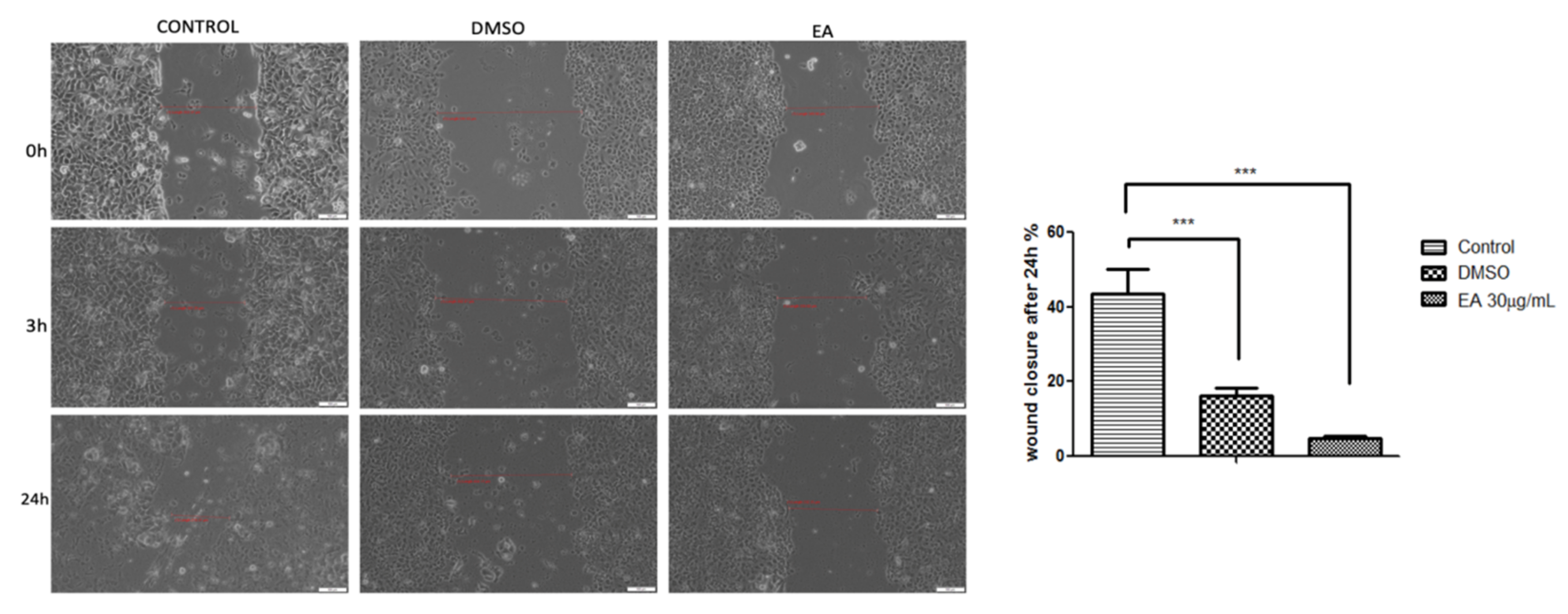
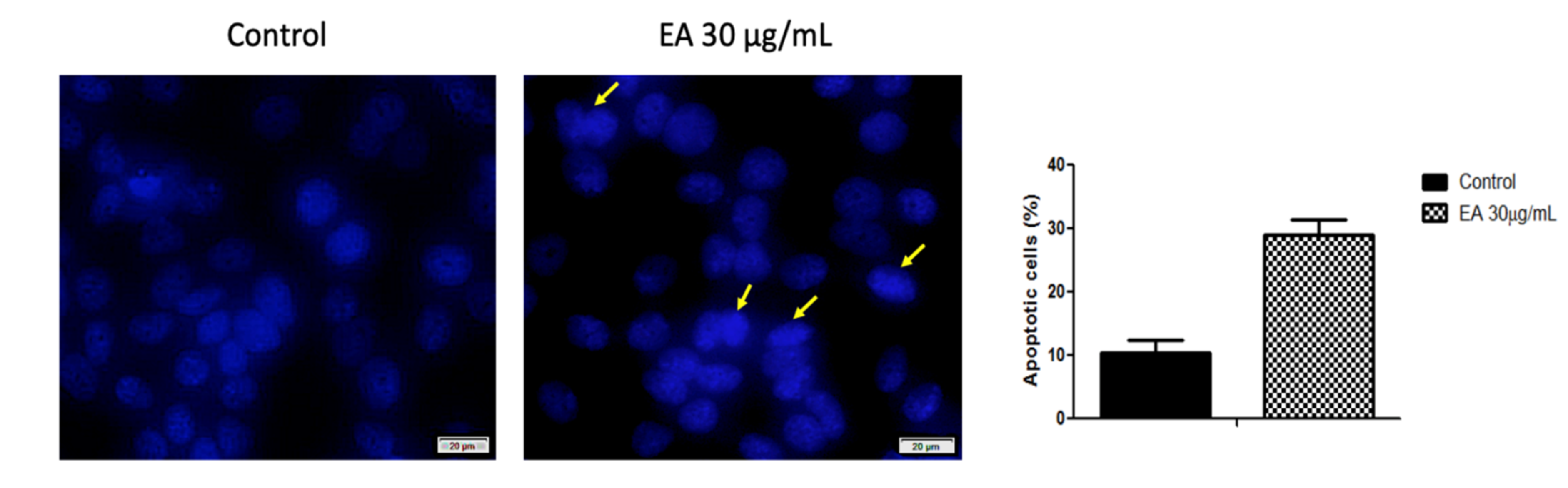
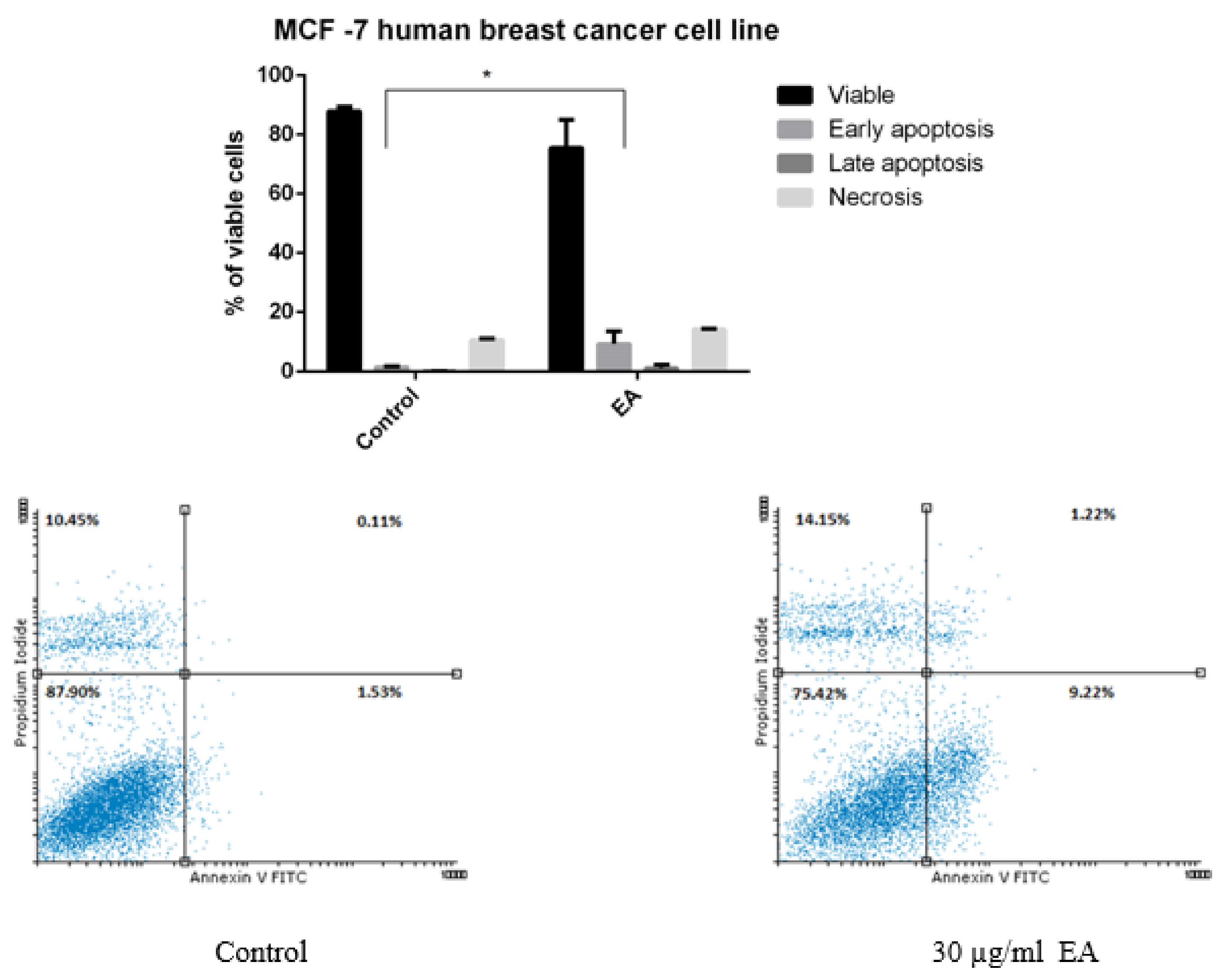
2.3. Anticancer Activity
3. Discussion
Antimicrobial Activity
4. Materials and Methods
4.1. Plant Materials
4.2. Determination of Total Polyphenols Content (TP)
4.3. Determination of Individual Polyphenols by LC-MS
4.4. Determination of the Total Antioxidant Activity (AA)
4.5. Determination of Total Alkaloids (TA)
4.6. In Vitro Antimicrobial Activity
4.7. Disk Diffusion Method
4.8. Determination of the Minimum Inhibitory Concentration (MIC) and the Minimum Bactericidal Concentration (MBC)
4.9. Cell Culture
4.10. Antiproliferative MTT Assay
4.11. Anti-Migratory Potential: A Wound Healing Technique
4.12. Determination of the Cytotoxic Potential by the Means of Lactate Dehydrogenase (LDH) Assay
4.13. DAPI: Cell Nuclei Staining
4.14. Annexin V-PI Assay
4.15. Statistics
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [PubMed]
- Rydin, C.; Pedersen, K.R.; Crane, P.R.; Friis, E.M. Former diversity of Ephedra (Gnetales): Evidence from Early Cretaceous seeds from Portugal and North America. Ann. Bot. 2006, 98, 123–140. [Google Scholar] [CrossRef] [PubMed]
- Hegazi, G.A.E.M.; El-Lamey, T.M. In vitro production of some phenolic compounds from Ephedra alata Decne. J. Appl. Environ. Biol. Sci. 2011, 1, 158–163. [Google Scholar]
- Caveney, S.; Charlet, D.A.; Freitag, H.; Maier-Stolte, M.; Starratt, A.N. New observations on the secondary chemistry of world Ephedra (Ephedraceae). Am. J. Bot. 2001, 88, 1199–1208. [Google Scholar] [CrossRef]
- Ibragic, S.; Sofić, E. Chemical composition of various Ephedra species. Bosn. J. Basic Med. Sci. 2015, 15, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Abourashed, E.A.; El-Alfy, A.T.; Khan, I.A.; Walker, L. Ephedra in perspective—A current review. Phytother. Res. 2003, 17, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, E.; Govêia, C.S.; Ladeira, L.C.d.A.; Nascimento, B.G.; Kluthcouski, S.M.C. Ephedrine versus phenylephrine: Prevention of hypotension during spinal block for cesarean section and effects on the fetus. Braz. J. Anesthesiol. 2009, 59, 11–20. [Google Scholar] [CrossRef]
- Cocan, I.; Alexa, E.; Danciu, C.; Radulov, I.; Galuscan, A.; Obistioiu, D.; Morvay, A.A.; Sumalan, R.M.; Poiana, M.A.; Pop, G.; et al. Phytochemical screening and biological activity of Lamiaceae family plant extracts. Exp. Ther. Med. 2018. [Google Scholar] [CrossRef]
- Jerbi, A.; Zehri, S.; Abdnnabi, R.; Gharsallah, N.; Kammoun, M. Essential oil composition, free-radical-scavenging and antibacterial effect from stems of Ephedra alata alenda in Tunisia. J. Essent. Oil Bear. Plants 2016, 19, 1503–1509. [Google Scholar] [CrossRef]
- Parsaeimehr, A.; Sargsyan, E.; Javidnia, K. A comparative study of the antibacterial, antifungal and antioxidant activity and total content of phenolic compounds of cell cultures and wild plants of three endemic species of Ephedra. Molecules 2010, 15, 1668–1678. [Google Scholar] [CrossRef]
- Khan, A.; Jan, G.; Khan, A.; Gul Jan, F.; Bahadur, A.; Danish, M. In vitro antioxidant and antimicrobial activities of Ephedra gerardiana (root and stem) crude extract and fractions. Evid.-Based Complement. Altern. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Pullela, S.V.; Takamatsu, S.; Khan, S.I.; Khan, IA. Isolation of lignans and biological activity studies of Ephedra viridis. Planta Med. 2005, 71, 789–791. [Google Scholar] [CrossRef] [PubMed]
- Nam, N.-H.; Lee, C.-W.; Hong, D.-H.; Kim, H.-M.; Bae, K.-H.; Ahn, B.-Z. Antiinvasive, antiangiogenic and antitumour activity of Ephedra sinica extract. Phytother. Res. 2003, 17, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, N.; Falah, A.; Fatima, J.; Hussein, K.; Akram, H.; Ali, C.; Hassan, R. Antibacterial, antioxidant and antiproliferative activities of the hydroalcoholic extract of the lebanese plant: Ephedra Campylopoda. Int. Res. J. Pharm. 2016, 7, 23–29. [Google Scholar] [CrossRef]
- Attard, E.; Vella, K. Effects of ephedrine and Ephedra fragilis crude extracts on human peripheral lymphocytes. Pharmacogn. Res. 2009, 1, 38–42. [Google Scholar]
- Al-Qarawi, A.A.; Abd-Allah, E.F.; Hashem, A. Ephedra alata as biologically-based strategy inhibit aflatoxigenic seedborne mold. Afr. J. Microbiol. Res. 2011, 5, 2297–2303. [Google Scholar] [CrossRef]
- Jaradat, N.; Hussen, F.; Anas, A.A. Preliminary phytochemical screening, quantitative estimation of total flavonoids, total phenols and antioxidant activity of Ephedra alata Decne. J. Mater. Environ. Sci. 2015, 6, 1771–1778. [Google Scholar]
- Al-Rimawi, F.; Abu-Lafi, S.; Abbadi, J.; Alamarneh, A.A.A.; Sawahreh, R.A.; Odeh, I. Analysis of phenolic and flavonoids of wild Ephedra alata plant extracts by LC/PDA and LC/MS and their antioxidant activity. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 130–141. [Google Scholar] [CrossRef]
- Amakura, Y.; Yoshimura, M.; Yamakami, S.; Yoshida, T.; Wakana, D.; Hyuga, M.; Hyuga, S.; Hanawa, T.; Goda, Y. Characterization of Phenolic Constituents from Ephedra Herb Extract. Molecules 2013, 18, 5326–5334. [Google Scholar] [CrossRef]
- Ziani, B.E.C.; Heleno, S.A.; Bachari, K.; Dias, M.I.; Ferreira, I.C.F. Phenolic compounds characterization by LC-DAD-ESI/MSn and bioactive properties of Thymus algeriensis Boiss. & Reut. and Ephedra alata Decne. Food Res. Int. 2018. [Google Scholar] [CrossRef]
- Grippo, A.A.; Capps, K.; Rougeau, B.; Gurley, B.J. Analysis of flavonoid phytoestrogens in botanical and ephedra-containing dietary supplements. Ann. Pharmacother. 2007, 41, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Ghanem, S.; El-Magly, U.I.A. Antimicrobial activity and tentative identification of active compounds from the medicinal Ephedra alata male plant. J. Taibah Univ. Med. Sci. 2008, 3, 7–15. [Google Scholar] [CrossRef]
- Chebouat, E.; Dadamoussa, B.; Gharabli, S.; Gherraf, N.; Allaoui, M.; Cheriti, A.; Lahham, A.; Zellagui, A. Assessment of antimicrobial activity of flavonoids extract from Ephedra alata. Der Pharmacia Lettre 2014, 6, 27–30. [Google Scholar]
- Al-Awaida, W.; Al-Hourani, B.J.; Akash, M.; Talib, W.H.; Zein, S.; Falah, R.R. In vitro anticancer, anti-inflammatory, and antioxidant potentials of Ephedra aphylla. J. Cancer Res. Ther. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kallassy, H.; Fayyad-Kazan, M.; Makki, R.; EL-Makhour, Y.; Rammal, H.; Leger, D.Y.; Sol, V.; Fayyad-Kazan, H.; Liagre, B.; Badran, B. Chemical composition and antioxidant, anti-inflammatory, and antiproliferative activities of Lebanese Ephedra Campylopoda plant. Med. Sci. Monit. Basic Res. 2017, 23, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Oshima, N.; Yamashita, T.; Hyuga, S.; Hyuga, M.; Kamakura, H.; Yoshimura, M.; Maruyama, T.; Hakamatsuka, T.; Amakura, Y.; Hanawa, T.; et al. Efficiently prepared ephedrine alkaloids-free Ephedra Herb extract: A putative marker and antiproliferative effects. J. Nat. Med. 2016, 70, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Kmail, A.; Lyoussi, B.; Zaid, H.; Saad, B. In vitro assessments of cytotoxic and cytostatic effects of Asparagus aphyllus, Crataegus aronia, and Ephedra alata in monocultures and co-cultures of Hepg2 and THP-1-derived macrophages. Pharmacogn. Commun. 2017, 7, 24–33. [Google Scholar] [CrossRef]
- Mendelovich, M.; Shoshan, M.; Fridlender, M.; Mazuz, M.; Namder, D.; Nallathambi, R.; Selvaraj, G.; Kumari, P.; Ion, A.; Wininger, S.; et al. Effect of Ephedra foeminea active compounds on cell viability and actin structures in cancer cell lines. J. Med. Plant Res. 2017, 11, 690–702. [Google Scholar] [CrossRef]
- Jaradat, N.A.; Shawahna, R.; Eid, A.M.; Al-Ramahi, R.; Asma, M.K.; Zaid, AN. Herbal remedies use by breast cancer patients in the West Bank of Palestine. J. Ethnopharmacol. 2016, 1–8. [Google Scholar] [CrossRef]
- Jaradat, N.A.; Al-Ramahi, R.; Zaid, A.N.; Ayesh, O.I.; Eid, A.M. Ethnopharmacological survey of herbal remedies used for treatment of various types of cancer and their methods of preparations in the West Bank-Palestine. BMC Complement. Altern. Med. 2016, 16, 93. [Google Scholar] [CrossRef]
- Abu-Darwish, M.S.; Efferth, T. Medicinal plants from Near East for cancer therapy. Front. Pharmacol. 2018, 9, 56. [Google Scholar] [CrossRef] [PubMed]
- Atak, M.; Mavi, K.; Uremis, I. Bio-herbicidal effects of oregano and rosemary essential oils on germination and seedling growth of bread wheat cultivars and weeds. Rom. Biotechnol. Lett. 2016, 21, 11149–11159. [Google Scholar]
- Pavel, I.Z.; Danciu, C.; Oprean, C.; Dehelean, C.A.; Muntean, D.; Csuk, R.; Muntean, D.M. In vitro evaluation of the antimicrobial ability and cytotoxicity on two melanoma cell lines of a benzylamide derivative of maslinic acid. Anal. Cell. Pathol. (Amst.) 2016, 2016, 2787623. [Google Scholar] [CrossRef] [PubMed]
- Oprean, C.; Zambori, C.; Borcan, F.; Soica, C.; Zupko, I.; Minorics, R.; Bojin, F.; Ambrus, R.; Muntean, D.; Danciu, C.; et al. Anti-proliferative and antibacterial in vitro evaluation of the polyurethane nanostructures incorporating pentacyclic triterpeneS. Pharm. Biol. 2016, 54, 2714–2722. [Google Scholar] [CrossRef] [PubMed]
- Wikler, M.A.; Low, D.E.; Cockerill, F.R.; Sheehan, D.J.; Craig, W.A.; Tenover, F.C. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard, 7th, ed.; Document M7-A7; CLSI: Wayne, PA, USA, 2006. [Google Scholar]
- Szabó, J.; Jerkovics, N.; Schneider, G.; Wölfling, J.; Bózsity, N.; Minorics, R.; Zupkó, I.; Mernyák, E. Synthesis and in vitro antiproliferative evaluation ofC-13 epimers of triazolyl-D-secoestrone alcohols: The first potent 13a-D-secoestrone derivative. Molecules 2016, 21, 611. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Liang, C.C.; Park, A.Y.; Guan, J.L. In vitro scratch assay: A convenient and inexpensive method for analysis of cell migration in vitro. Nat. Protoc. 2007, 2, 329–333. [Google Scholar] [CrossRef]
- Moacă, E.A.; Farcaş, C.; Ghiţu, A.; Coricovac, D.; Popovici, R.; Cărăba-Meiţă, N.L.; Ardelean, F.; Antal, D.S.; Dehelean, C.; Avram, Ş. A Comparative Study of Melissa officinalis Leaves and Stems Ethanolic Extracts in terms of Antioxidant, Cytotoxic, and Antiproliferative Potential. Evid. Based Complement. Alternat. Med. 2018, 7860456. [Google Scholar] [CrossRef]
- Felice, F.; Zambito, Y.; Belardinelli, E.; Fabiano, A.; Santoni, T.; Di Stefano, R. Effect of different chitosan derivatives on in vitro scratch wound assay: A comparative study. Int. J. Biol. Macromol. 2015, 76, 236–241. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
| Sample | TP (mgGAE/g Extract) | AA (μmol Trolox/g Extract) | TA (mg Ephedrine/g Extract) |
|---|---|---|---|
| EA | 156.226 ± 0.5 | 7453.18 ± 2.5 | 17.57 ± 0.6 |
| Compounds | Retention Time Column I (min) | Retention Time Column II (min) | m/z Signal | EA in Hydroethanolic Extract | EA in Hydromethanolic Extract Chromatographic Column I | Calibration Curve | |
|---|---|---|---|---|---|---|---|
| Column I | Column II | ||||||
| µg/mL ± SD (µg/mg) | µg/mL ± SD (µg/mg) | (µg/mL ± SD) (µg/mg) | |||||
| Gallic acid | 4.826 | 4.745 | 169 | nd | nd | nd | y = 8.470 e-006x (r = 0.9996); |
| Protocatecuic acid | 11.982 | 11.774 | 153 | nd | nd | nd | y = 8.036 e-006x (r = 0.9990); |
| Caffeic acid | 18.605 | 18.396 | 179 | 0.709 ± 0.279 (0.014) | nd | 0.389 ± 0.49 (0.008) | y = 7.110 e-006x (r = 0.9990); |
| Epicatechin | 22.910 | 23.242 | 289 | nd | 7.736 ± 0.297 (0.155) | nd | y = 3.881 e-005x (r = 0.9996); |
| p-coumaric acid | 24.374 | 24.454 | 163 | 0.241 ± 0.028 (0.005) | nd | 0.261 ± 0.021 (0.005) | y = 1.1566 e-006x (r = 0.9997); |
| Ferulic acid | 26.333 | 24.883 | 193 | nd | nd | nd | y = 1.172 e-006x (r = 0.9999); |
| Rutin | 27.290 | 26.086 | 609 | nd | nd | nd | y = 1.813 e-005x (r = 0.9999) |
| Rosmarinic acid | 28.252 | 29.129 | 359 | 0.915 ± 0.008 (0.018) | 0.397 ± 0.002 (0.008) | 0.807 ± 0.049 (0.016) | y = 1.018 e-006x (r = 0.9982); |
| Resveratrol | 29.749 | 29.887 | 227 | 9.403 ± 0.008 (0.188) | 12.963 ± 0.454 (0.259) | 10.387 ± 0.422 (0.207) | y = 6.388 e-006x (r = 0.9945); |
| Quercitin | 32.591 | 31.841 | 301 | 2.873 ± 0.70 (0.057) | 2.387 ± 0.06 (0.048) | 4.572 ± 0.384 (0.091) | y = 1.001 e-005x (r = 0.9992); |
| Kaempferol | 34.470 | 34.378 | 285 | 24.297 ± 2.73 (0.485) | 6.814 ± 0.179 (0.136) | 28.675 ± 2.579 (2.867) | y = 3.273 e-005x (r = 0.9990). |
| Compound | Klebsiella Pneumonia | Shigella flexneri | Salmonella Enterica | Escherichia coli | Pseudomonas Aeruginosa | Staphylococcus Aureus | Enterococcus Faecalis | Candida Albicans | Candida Parapsilosis |
|---|---|---|---|---|---|---|---|---|---|
| EA | 7 mm | 7 mm | 7 mm | 7 mm | 7 mm | 9 mm | 7 mm | 10 mm | 10 mm |
| Species | MIC (μg/mL) | MBC (μg/mL) |
|---|---|---|
| K. pneumoniae | 200 | - |
| S. flexne | 200 | - |
| S. enterica | 200 | - |
| E. coli | 200 | - |
| P. aeruginosa | 200 | - |
| S. aureus | 50 | 100 |
| E. faecalis | 100 | 200 |
| C. albicans | 50 | 100 |
| C. parapsilosis | 50 | 100 |
| Bacterial Species | ATCC | Producer |
|---|---|---|
| Salmonella enterica serotype typhimurium | 14,028 | Thermo Scientific |
| Shigella flexneri serotype 2b | 12,022 | Thermo Scientific |
| Enterococcus faecalis | 51,299 | Thermo Scientific |
| Escherichia coli | 25,922 | Thermo Scientific |
| Klebsiella pneumoniae | 700,603 | Thermo Scientific |
| Pseudomonas aeruginosa | 27,853 | Thermo Scientific |
| Staphylococcus aureus | 25,923 | Thermo Scientific |
| Candida albicans | 10,231 | Thermo Scientific |
| Candida parapsilosis | 22,019 | Thermo Scientific |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Danciu, C.; Muntean, D.; Alexa, E.; Farcas, C.; Oprean, C.; Zupko, I.; Bor, A.; Minda, D.; Proks, M.; Buda, V.; et al. Phytochemical Characterization and Evaluation of the Antimicrobial, Antiproliferative and Pro-Apoptotic Potential of Ephedra alata Decne. Hydroalcoholic Extract against the MCF-7 Breast Cancer Cell Line. Molecules 2019, 24, 13. https://doi.org/10.3390/molecules24010013
Danciu C, Muntean D, Alexa E, Farcas C, Oprean C, Zupko I, Bor A, Minda D, Proks M, Buda V, et al. Phytochemical Characterization and Evaluation of the Antimicrobial, Antiproliferative and Pro-Apoptotic Potential of Ephedra alata Decne. Hydroalcoholic Extract against the MCF-7 Breast Cancer Cell Line. Molecules. 2019; 24(1):13. https://doi.org/10.3390/molecules24010013
Chicago/Turabian StyleDanciu, Corina, Delia Muntean, Ersilia Alexa, Claudia Farcas, Camelia Oprean, Istvan Zupko, Andrea Bor, Daliana Minda, Maria Proks, Valentina Buda, and et al. 2019. "Phytochemical Characterization and Evaluation of the Antimicrobial, Antiproliferative and Pro-Apoptotic Potential of Ephedra alata Decne. Hydroalcoholic Extract against the MCF-7 Breast Cancer Cell Line" Molecules 24, no. 1: 13. https://doi.org/10.3390/molecules24010013
APA StyleDanciu, C., Muntean, D., Alexa, E., Farcas, C., Oprean, C., Zupko, I., Bor, A., Minda, D., Proks, M., Buda, V., Hancianu, M., Cioanca, O., Soica, C., Popescu, S., & Dehelean, C. A. (2019). Phytochemical Characterization and Evaluation of the Antimicrobial, Antiproliferative and Pro-Apoptotic Potential of Ephedra alata Decne. Hydroalcoholic Extract against the MCF-7 Breast Cancer Cell Line. Molecules, 24(1), 13. https://doi.org/10.3390/molecules24010013














