Biological Properties and Bioactive Components of Allium cepa L.: Focus on Potential Benefits in the Treatment of Obesity and Related Comorbidities
Abstract
1. Introduction
2. Allium cepa L.: Botany, Distribution and Phytochemistry
3. A. cepa Biological Activities
4. Beneficial Effects of Onion in the Treatment of Obesity
4.1. Pancreatic Lipase Inhibition
4.2. Adipogenesis Inhibition
4.3. Increase of Energy Expenditure
5. Effects on Obesity Related Comorbidities
5.1. Hyperlipidaemia
5.2. Diabetes
5.3. Hypertension
5.4. Cardiovascular Diseases
6. Allium cepa L. Bioactive Compounds with Anti-Obesity Properties
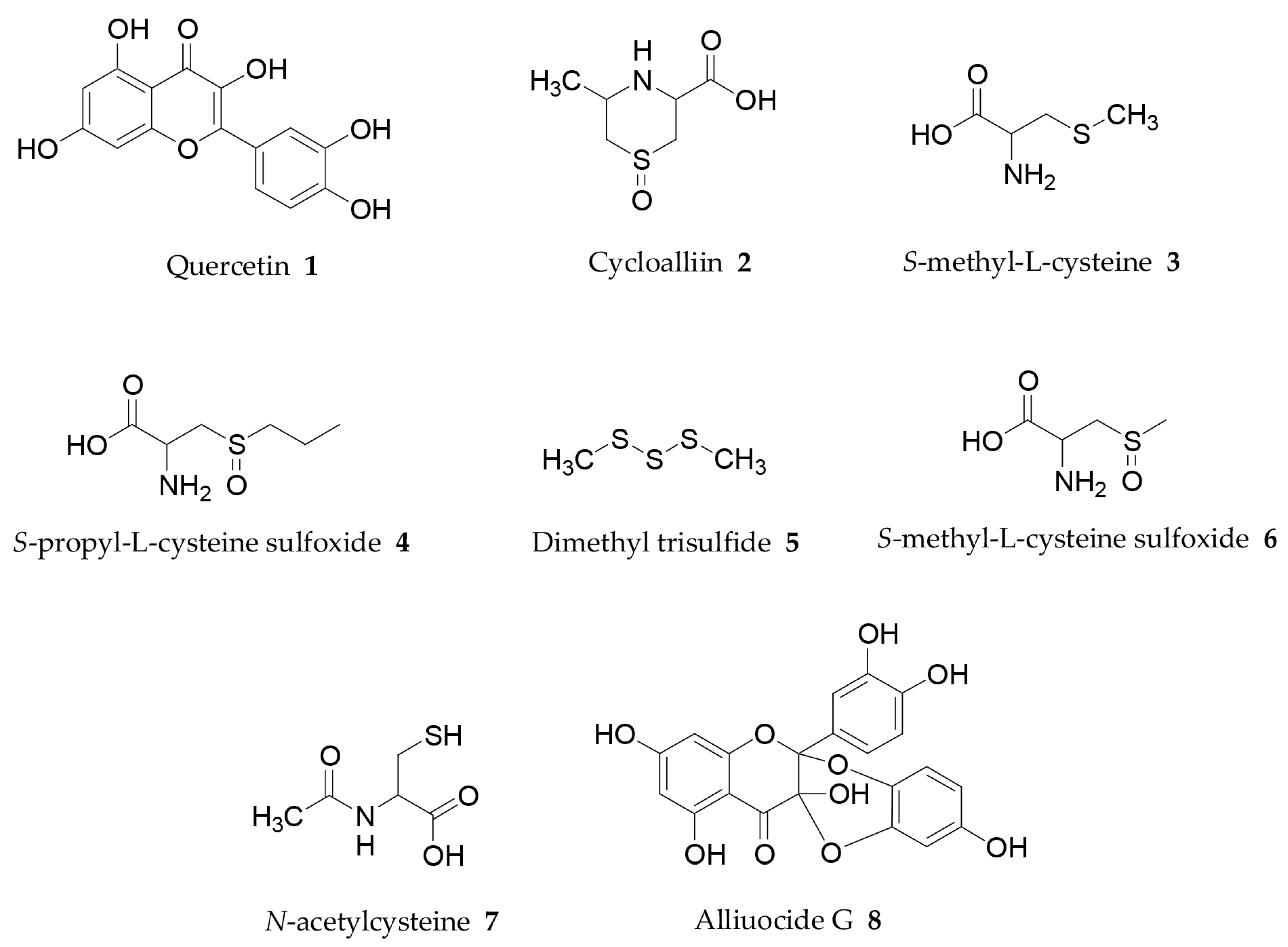
6.1. Quercetin
6.2. Sulphur-Containing Components
6.3. Flavonoid Alliuocide G
7. Negative Results
8. Conclusions
Funding
Conflicts of Interest
Abbreviations
| 3T3-L1 | mouse fibroblasts |
| ABP | ambulatory blood pressure |
| A1235A | human glioblastoma cell line |
| ABTS | 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) |
| B16F10 | murine melanoma cells |
| BMI | body mass index |
| BP | blood pressure |
| C/EBPα | CCAAT/enhancer-binding protein α |
| Caco-2 | human colorectal adenocarcinoma cells |
| CAT | catalase |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl radical |
| EPCs | endothelial progenitor cells |
| ER | extended release |
| FAO | Food and Agriculture Organization of the United Nations |
| FAS | fatty acid synthase |
| FFA | free fatty acids |
| FMD | flow-mediated dilation |
| FST | forced swimming test |
| Gbw | gram body weight |
| GPDH | glycerol 3-phosphate dehydrogenase |
| GSH | glutathione peroxidase |
| H9c2 | cardiac muscle cell line |
| HDL | high density lipoprotein |
| HepG2 | human hepatocellular carcinoma cells |
| HF | high fat diet |
| IL-6 | interleukin-6 |
| LDL | low-density lipoproteins |
| LPO | lipid peroxidase |
| MDA-MB-231 | human breast cancer |
| NO | nitric oxide |
| ORAC | oxygen radical absorbance capacity |
| PPAR-γ2 | peroxisome proliferator-activated receptor γ2 |
| SOD | superoxide dismutase |
| SR | sustained release |
| STZ | streptozotocin |
| TC | total cholesterol |
| TG | triacylglycerides |
| TNF-α | tumor necrosis factor-α |
| ZDF | Zucker diabetic fatty |
References
- Korner, J.; Aronne, L.J. The emerging science of body weight regulation and its impact on obesity treatment. J. Clin. Invest. 2003, 111, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.G.; Park, C.Y. Anti-obesity drugs: A review about their effects and safety. Diabetes Metab. J. 2012, 36, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, R.J.; Tschöp, M.H.; Wilding, J.P. Anti-obesity drugs: Past, present and future. Dis. Model. Mech. 2012, 5, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Karnani, A.; McFerran, B.; Mukhopadhyay, A. The obesity crisis as market failure: An analysis of systemic causes and corrective mechanisms. J. Assoc. Consum. Res. 2016, 1, 445–470. [Google Scholar] [CrossRef]
- Freemark, M. Childhood Obesity in the Modern Age: Global Trends, Determinants, Complications and Costs. In Pediatric Obesity; Freemark, M., Ed.; Humana Press, Cham.: New York, NY, USA, 2018; pp. 3–24. ISBN 978-3-319-68192-4. [Google Scholar]
- Meldrum, D.R.; Morris, M.A.; Gambone, J.C. Obesity pandemic: Causes, consequences and solutions—But do we have the will? Fertil. Steril. 2017, 107, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Uzunlulu, M.; Caklili, O.T.; Oguz, A. Association between metabolic syndrome and cancer. Ann. Nutr. MeTable 2016, 68, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Barone, I.; Giordano, C.; Bonofiglio, D.; Andò, S.; Catalano, S. Leptin, obesity and breast cancer: Progress to understanding the molecular connections. Curr. Opin. Pharmacol. 2016, 31, 83–89. [Google Scholar] [CrossRef] [PubMed]
- O’neill, S.; O’driscoll, L. Metabolic syndrome: A closer look at the growing epidemic and its associated pathologies. Obes. Rev. 2015, 16, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Hussain, M.E. Obesity and diabetes: An update. Diabetes Metab. Syndr. 2017, 11, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Kazemipoor, M.; Cordell, G.A.; Sarker, M.M.R.; Radzi, C.W.J.B.W.M.; Hajifaraji, M.; En Kiat, P. Alternative treatments for weight loss: Safety/risks and effectiveness of anti-obesity medicinal plants. Int. J. Food Prop. 2015, 18, 1942–1963. [Google Scholar] [CrossRef]
- Derosa, G.; Maffioli, P. Anti-obesity drugs: A review about their effects and their safety. Expert Opin. Drug Saf. 2012, 11, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Padwal, R.S.; Majumdar, S.R. Drug treatments for obesity: Orlistat, sibutramine and rimonabant. The Lancet 2007, 369, 71–77. [Google Scholar] [CrossRef]
- Krentz, A.J.; Fujioka, K.; Hompesch, M. Evolution of pharmacological obesity treatments: Focus on adverse side-effect profiles. Diabetes Obes. MeTable 2016, 8, 558–570. [Google Scholar] [CrossRef] [PubMed]
- Brandt, K.; Christensen, L.P.; Hansen-Møller, J.; Hansen, S.L.; Haraldsdottir, J.; Jespersen, L.; Purup, S.; Kharazmi, A.; Barkholt, V.; Frøkiærf, H.; et al. Health promoting compounds in vegetables and fruits: A systematic approach for identifying plant components with impact on human health. Trends Food Sci. Technol. 2004, 15, 384–393. [Google Scholar] [CrossRef]
- Raskin, I.; Ribnicky, D.M.; Komarnytsky, S.; Ilic, N.; Poulev, A.; Borisjuk, N.; Brinker, A.; Moreno, D.A.; Ripoll, C.; Yakoby, N.; et al. Plants and human health in the twenty-first century. Trends Biotechnol. 2002, 20, 522–531. [Google Scholar] [CrossRef]
- Srivastava, G.; Apovian, C. Future pharmacotherapy for obesity: New anti-obesity drugs on the horizon. Curr. Obes. Rep. 2018, 7, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Gamboa-Gómez, C.I.; Rocha-Guzmán, N.E.; Gallegos-Infante, J.A.; Moreno-Jiménez, M.R.; Vázquez-Cabral, B.D.; González-Laredo, R.F. Plants with potential use on obesity and its complications. EXCLI J. 2015, 14, 809–831. [Google Scholar] [PubMed]
- Wang, S.; Moustaid-Moussa, N.; Chen, L.; Mo, H.; Shastri, A.; Su, R.; Bapat, P.; Kwun, I.; Shen, C.-L. Novel insights of dietary polyphenols and obesity. J. Nutr. Biochem. 2014, 25, 1–18. [Google Scholar] [CrossRef]
- Meydani, M.; Hasan, S.T. Dietary polyphenols and obesity. Nutrients 2010, 2, 737–751. [Google Scholar] [CrossRef]
- Birari, R.B.; Bhutani, K.K. Pancreatic lipase inhibitors from natural sources: Unexplored potential. Drug Discov. Today 2007, 12, 879–889. [Google Scholar] [CrossRef]
- Singh, G.; Suresh, S.; Bayineni, V.K.; Kadeppagari, R.K. Lipase inhibitors from plants and their medical applications. Int. J. Pharm. Pharm. Sci. 2015, 7, 1–5. [Google Scholar]
- Marrelli, M.; Conforti, F.; Araniti, F.; Statti, G.A. Effects of saponins on lipid metabolism: A review of potential health benefits in the treatment of obesity. Molecules 2016, 21, 1404. [Google Scholar] [CrossRef] [PubMed]
- Hasani-Ranjbar, S.; Nayebi, N.; Larijani, B.; Abdollahi, M. A systematic review of the efficacy and safety of herbal medicines used in the treatment of obesity. World J. Gastroenterol. 2009, 15, 3073–3085. [Google Scholar] [CrossRef] [PubMed]
- Hasani-Ranjbar, S.; Jouyandeh, Z.; Abdollahi, M. A systematic review of anti-obesity medicinal plants-an update. J. Diabetes Metab. Disord. 2013, 12, 28. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.W. Possible anti-obesity therapeutics from nature–A review. Phytochemistry 2010, 71, 1625–1641. [Google Scholar] [CrossRef] [PubMed]
- Bahmani, M.; Eftekhari, Z.; Saki, K.; Fazeli-Moghadam, E.; Jelodari, M.; Rafieian-Kopaei, M. Obesity phytotherapy: Review of native herbs used in traditional medicine for obesity. J. Evid. Based Complementary Altern. Med. 2016, 21, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Torres-Fuentes, C.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. A natural solution for obesity: Bioactives for the prevention and treatment of weight gain. A review. Nutr. Neurosci. 2015, 18, 49–65. [Google Scholar] [CrossRef]
- González-Castejón, M.; Rodriguez-Casado, A. Dietary phytochemicals and their potential effects on obesity: A review. Pharmacol. Res. 2011, 64, 438–455. [Google Scholar] [CrossRef]
- Fritsch, R.M.; Friesen, N. Evolution, domestication and taxonomy. In Allium crop science: Recent advances; Rabinowitch, H.D., Currah, L., Eds.; CABI publishing: Stratford-upon-Avon, UK, 2002; pp. 5–30. ISBN 0-85199-510-1. [Google Scholar]
- Breu, W. Allium cepa L. (onion) Part 1: Chemistry and analysis. Phytomedicine 1996, 3, 293–306. [Google Scholar] [CrossRef]
- Angiosperm Phylogeny Group. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG III. Bot. J. Linn. Soc. 2009, 161, 105–121. [Google Scholar]
- Peruzzi, L.; Carta, A.; Altinordu, F. Chromosome diversity and evolution in Allium (Allioideae, Amaryllidaceae). Plant. Biosyst. 2017, 151, 212–220. [Google Scholar] [CrossRef]
- Lim, T.K. Modified Stems, Roots, Bulbs. In Edible Medicinal and Non-Medicinal Plants; Springer: Dordrecht, The Netherlands, 2015; Volume 9, pp. 124–203. ISBN 978-94-017-9511-1. [Google Scholar]
- FAO. Available online: http://www.fao.org/faostat/en/#data/QC/visualize (accessed on 4 November 2018).
- Böttcher, C.; Krähmer, A.; Stürtz, M.; Widder, S.; Schulz, H. Effect of cultivar and cultivation year on the metabolite profile of onion bulbs (Allium cepa l.). J. Agric. Food Chem. 2018, 66, 3229–3238. [Google Scholar] [CrossRef] [PubMed]
- Tedesco, I.; Carbone, V.; Spagnuolo, C.; Minasi, P.; Russo, G.L. Identification and quantification of flavonoids from two southern Italian cultivars of Allium cepa L., Tropea (Red Onion) and Montoro (Copper Onion) and their capacity to protect human erythrocytes from oxidative stress. J. Agric. Food Chem. 2015, 63, 5229–5238. [Google Scholar] [CrossRef] [PubMed]
- Marotti, M.; Piccaglia, R. Characterization of flavonoids in different cultivars of onion (Allium cepa L.). J. Food Sci. 2002, 67, 1229–1232. [Google Scholar] [CrossRef]
- Bennett, M.D.; Johnston, S.; Hodnett, G.L.; Price, H.J. Allium cepa L. cultivars from four continents compared by flow cytometry show nuclear DNA constancy. Ann. Bot. 2000, 85, 351–357. [Google Scholar] [CrossRef]
- Griffiths, G.; Trueman, L.; Crowther, T.; Thomas, B.; Smith, B. Onions—A global benefit to health. Phytother. Res. 2002, 16, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Bystrická, J.; Musilová, J.; Vollmannová, A.; Timoracká, M.; Kavalcová, P. Bioactive components of onion (Allium cepa L.)—A Review. Acta Aliment. 2013, 42, 11–22. [Google Scholar] [CrossRef]
- Teshika, J.D.; Zakariyyah, A.M.; Zaynab, T.; Zengin, G.; Rengasamy, K.R.; Pandian, S.K.; Fawzi, M.M. Traditional and modern uses of onion bulb (Allium cepa L.): A systematic review. Crit. Rev. Food Sci. Nutr. 2018. [Google Scholar] [CrossRef]
- Lanzotti, V. The analysis of onion and garlic. J. Chromatogr. A 2006, 1112, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Nuutila, A.M.; Puupponen-Pimiä, R.; Aarni, M.; Oksman-Caldentey, K.M. Comparison of antioxidant activities of onion and garlic extracts by inhibition of lipid peroxidation and radical scavenging activity. Food Chem. 2003, 81, 485–493. [Google Scholar] [CrossRef]
- Prakash, D.; Singh, B.N.; Upadhyay, G. Antioxidant and free radical scavenging activities of phenols from onion (Allium cepa). Food Chem. 2007, 102, 1389–1393. [Google Scholar] [CrossRef]
- Hosseini, B.; Saedisomeolia, A.; Allman-Farinelli, M. Association between antioxidant intake/status and obesity: A systematic review of observational studies. Biol. Trace Elem. Res. 2017, 175, 287–297. [Google Scholar] [CrossRef]
- Hassan, H.A.; El-Gharib, N.E. Obesity and clinical riskiness relationship: Therapeutic management by dietary antioxidant supplementation—A review. Appl. Biochem. Biotechnol. 2015, 176, 647–669. [Google Scholar] [CrossRef] [PubMed]
- Benkeblia, N. Free-radical scavenging capacity and antioxidant properties of some selected onions (Allium cepa L.) and garlic (Allium sativum L.) extracts. Braz. Arch. Biol. Techn. 2005, 48, 753–759. [Google Scholar] [CrossRef]
- Škerget, M.; Majhenič, L.; Bezjak, M.; Knez, Ž. Antioxidant, radical scavenging and antimicrobial activities of red onion (Allium cepa L) skin and edible part extracts. Chem. Biochem. Eng. Q. 2009, 23, 435–444. [Google Scholar]
- Santas, J.; Almajano, M.P.; Carbó, R. Antimicrobial and antioxidant activity of crude onion (Allium cepa, L.) extracts. Int, J. Food Sci Technol. 2010, 45, 403–409. [Google Scholar] [CrossRef]
- Singh, B.N.; Singh, B.R.; Singh, R.L.; Prakash, D.; Singh, D.P.; Sarma, B.K.; Upadhyay, G.; Singh, H.B. Polyphenolics from various extracts/fractions of red onion (Allium cepa) peel with potent antioxidant and antimutagenic activities. Food Chem. Toxicol. 2009, 47, 1161–1167. [Google Scholar] [CrossRef]
- Nile, S.H.; Park, S.W. Total phenolics, antioxidant and xanthine oxidase inhibitory activity of three coloured onions (Allium cepa L.). Front. Life Sci. 2013, 7, 224–228. [Google Scholar] [CrossRef]
- Benmalek, Y.; Yahia, O.A.; Belkebir, A.; Fardeau, M.L. Anti-microbial and anti-oxidant activities of Illicium verum, Crataegus oxyacantha ssp monogyna and Allium cepa red and white varieties. Bioengineered 2013, 4, 244–248. [Google Scholar] [CrossRef]
- Kim, M.H.; Jo, S.H.; Jang, H.D.; Lee, M.S.; Kwon, Y.I. Antioxidant activity and α-glucosidase inhibitory potential of onion (Allium cepa L.) extracts. Food Sci. Biotechnol. 2010, 19, 159–164. [Google Scholar] [CrossRef]
- Lee, K.A.; Kim, K.T.; Kim, H.J.; Chung, M.S.; Chang, P.S.; Park, H.; Pai, H.D. Antioxidant activities of onion (Allium cepa L.) peel extracts produced by ethanol, hot water and subcritical water extraction. Food Sci. Biotechnol. 2014, 23, 615–621. [Google Scholar] [CrossRef]
- Ye, C.L.; Dai, D.H.; Hu, W.L. Antimicrobial and antioxidant activities of the essential oil from onion (Allium cepa L.). Food control 2013, 30, 48–53. [Google Scholar] [CrossRef]
- Campos, K.E.; Diniz, Y.S.; Cataneo, A.C.; Faine, L.A.; Alves, M.J.Q.F.; Novelli, E.L.B. Hypoglycaemic and antioxidant effects of onion, Allium cepa: Dietary onion addition, antioxidant activity and hypoglycaemic effects on diabetic rats. Int. J. Food Sci. Nutr. 2003, 54, 241–246. [Google Scholar] [CrossRef]
- Baragob, A.E.; Al-Wabel, N.A.; Ahmed, N.A.; Babiker, M.F.; Abdalkarim, A.S.; Elboshra, M.I. Study to investigate the pancreatic regeneration and evaluation of the antidiabetic and antihyperlipidemic potential of aerial parts of Allium cepa. Biochem. Biotechnol. Res. 2015, 3, 19–29. [Google Scholar]
- Ren, F.; Reilly, K.; Kerry, J.P.; Gaffney, M.; Hossain, M.; Rai, D.K. Higher antioxidant activity, total flavonols and specific quercetin glucosides in two different onion (Allium cepa L.) varieties grown under organic production: Results from a 6-year field study. J. Agric. Food Chem. 2017, 65, 5122–5132. [Google Scholar] [PubMed]
- Roldán, E.; Sánchez-Moreno, C.; de Ancos, B.; Cano, M.P. Characterisation of onion (Allium cepa L.) by-products as food ingredients with antioxidant and antibrowning properties. Food Chem. 2008, 108, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Gawlik-Dziki, U.; Świeca, M.; Dziki, D.; Baraniak, B.; Tomiło, J.; Czyż, J. Quality and antioxidant properties of breads enriched with dry onion (Allium cepa L.) skin. Food Chem. 2013, 138, 1621–1628. [Google Scholar] [CrossRef] [PubMed]
- Helen, A.; Krishnakumar, K.; Vijayammal, P.L.; Augusti, K.T. Antioxidant effect of onion oil (Allium cepa. Linn) on the damages induced by nicotine in rats as compared to alpha-tocopherol. Toxicol. Lett. 2000, 116, 61–68. [Google Scholar] [CrossRef]
- Siddiq, M.; Roidoung, S.; Sogi, D.S.; Dolan, K.D. Total phenolics, antioxidant properties and quality of fresh-cut onions (Allium cepa L.) treated with mild-heat. Food Chem. 2013, 136, 803–806. [Google Scholar] [CrossRef]
- Ma, Y.L.; Zhu, D.Y.; Thakur, K.; Wang, C.H.; Wang, H.; Ren, Y.F.; Zhang, J.G.; Wei, Z.J. Antioxidant and antibacterial evaluation of polysaccharides sequentially extracted from onion (Allium cepa L.). Int. J. Biol. Macromol. 2018, 111, 92–101. [Google Scholar] [CrossRef]
- Arung, E.T.; Furuta, S.; Ishikawa, H.; Kusuma, I.W.; Shimizu, K.; Kondo, R. Anti-melanogenesis properties of quercetin-and its derivative-rich extract from Allium cepa. Food Chem. 2011, 124, 1024–1028. [Google Scholar] [CrossRef]
- Khaki, A.; Fathiazad, F.; Nouri, M.; Khaki, A.A.; Khamenehi, H.J.; Hamadeh, M. Evaluation of androgenic activity of allium cepa on spermatogenesis in the rat. Folia Morphol. 2009, 68, 45–51. [Google Scholar]
- Naseri, M.K.G.; Yahyavi, H.; Arabian, M. Antispasmodic activity of onion (Allium cepa L.) peel extract on rat ileum. Iran. J. Pharm. Res. 2008, 7, 155–159. [Google Scholar]
- Sakakibara, H.; Yoshino, S.; Kawai, Y.; Terao, J. Antidepressant-like effect of onion (Allium cepa L.) powder in a rat behavioural model of depression. Biosci. Biotechnol. Biochem. 2008, 72, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, G.D.; Rose, D.P. Breast cancer and obesity: An update. Nutr. Cancer 2003, 45, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Fader, A.N.; Arriba, L.N.; Frasure, H.E.; von Gruenigen, V.E. Endometrial cancer and obesity: Epidemiology, biomarkers, prevention and survivorship. Gynecol. Oncol. 2009, 114, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Alshaker, H.; Sacco, K.; Alfraidi, A.; Muhammad, A.; Winkler, M.; Pchejetski, D. Leptin signalling, obesity and prostate cancer: Molecular and clinical perspective on the old dilemma. Oncotarget 2015, 6, 35556. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, A.A.; Woodward, M.; Huxley, R. Obesity and risk of colorectal cancer: A meta-analysis of 31 studies with 70,000 events. Cancer Epidem. Biomar. 2007, 16, 2533–2547. [Google Scholar] [CrossRef]
- Calle, E.E.; Kaaks, R. Overweight, obesity and cancer: Epidemiological evidence and proposed mechanisms. Nat. Rev. Cancer 2004, 4, 579. [Google Scholar] [CrossRef]
- Wang, Y.; Tian, W.X.; Ma, X.F. Inhibitory effects of onion (Allium cepa L.) extract on proliferation of cancer cells and adipocytes via inhibiting fatty acid synthase. Asian Pac. J. Cancer Prev. 2012, 13, 5573–5579. [Google Scholar] [CrossRef]
- Fredotović, Ž.; Šprung, M.; Soldo, B.; Ljubenkov, I.; Budić-Leto, I.; Bilušić, T.; Čikeš-Čulić, V.; Puizina, J. Chemical composition and biological activity of Allium cepa L. and Allium × cornutum (Clementi ex Visiani 1842) methanolic extracts. Molecules 2017, 22, 448. [Google Scholar] [CrossRef]
- Shrivastava, S.; Ganesh, N. Tumor inhibition and Cytotoxicity assay by aqueous extract of onion (Allium cepa) & Garlic (Allium sativum): An in-vitro analysis. Int. J. Phytomed. 2010, 2, 80–84. [Google Scholar]
- Yang, J.; Meyers, K.J.; van der Heide, J.; Liu, R.H. Varietal differences in phenolic content and antioxidant and antiproliferative activities of onions. J. Agric. Food Chem. 2004, 52, 6787–6793. [Google Scholar] [CrossRef] [PubMed]
- Millet, A.; Lamy, E.; Jonas, D.; Stintzing, F.; Mersch-Sundermann, V.; Merfort, I. Fermentation enhances the biological activity of Allium cepa bulb extracts. J. Agric. Food Chem. 2012, 60, 2148–2156. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y. Effects of Onion (Allium cepa) skin extract on pancreatic lipase and body weight-related parameters. Food Sci. Biotechnol. 2007, 16, 434–438. [Google Scholar]
- Slanc, P.; Doljak, B.; Kreft, S.; Lunder, M.; Janeš, D.; Štrukelj, B. Screening of selected food and medicinal plant extracts for pancreatic lipase inhibition. Phytother. Res. 2009, 23, 874–877. [Google Scholar] [CrossRef] [PubMed]
- Trisat, K.; Wong-on, M.; Lapphanichayakool, P.; Tiyaboonchai, W.; Limpeanchob, N. Vegetable juices and fibers reduce lipid digestion or absorption by inhibiting pancreatic lipase, cholesterol solubility and bile acid binding. Int. J. Vegetable Sci. 2017, 23, 260–269. [Google Scholar] [CrossRef]
- Yoshinari, O.; Shiojima, Y.; Igarashi, K. Anti-obesity effects of onion extract in Zucker diabetic fatty rats. Nutrients 2012, 4, 1518–1526. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.; Do, H.J.; Kim, O.Y.; Shin, M. J Antiobesity effects of quercetin-rich onion peel extract on the differentiation of 3T3-L1 preadipocytes and the adipogenesis in high fat-fed rats. Food Chem. Toxicol. 2013, 58, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Bae, C.R.; Park, Y.K.; Cha, Y.S. Quercetin-rich onion peel extract suppresses adipogenesis by down-regulating adipogenic transcription factors and gene expression in 3T3-L1 adipocytes. J. Sci. Food Agric. 2014, 94, 2655–2660. [Google Scholar] [CrossRef]
- Lee, S.G.; Parks, J.S.; Kang, H.W. Quercetin, a functional compound of onion peel, remodels white adipocytes to brown-like adipocytes. J. Nutr. Biochem. 2017, 42, 62–71. [Google Scholar] [CrossRef]
- Kim, O.Y.; Lee, S.M.; Do, H.; Moon, J.; Lee, K.H.; Cha, Y.J.; Shin, M.J. Influence of quercetin-rich onion peel extracts on adipokine expression in the visceral adipose tissue of rats. Phytother. Res. 2012, 26, 432–437. [Google Scholar] [CrossRef]
- Lata, S.; Saxena, K.K.; Bhasin, V.; Saxena, R.S.; Kumar, A.; Srivastava, V.K. Beneficial effects of Allium sativum, Allium cepa and Commiphora mukul on experimental hyperlipidaemia and atherosclerosis--a comparative evaluation. J. Postgrad. Med. 1991, 37, 132–135. [Google Scholar] [PubMed]
- Lee, K.H.; Kim, Y.; Park, E.; Hwang, H.J. Effect of onion powder supplementation on lipid metabolism in high fat-cholesterol fed SD Rats. J. Food Sci. Nutr. 2008, 13, 71–76. [Google Scholar] [CrossRef]
- Ojieh, A.E.; Adegor, E.C.; Okolo, A.C.; Ewhre, O.L.; Njoku, I.P.; Onyekpe, C.U. Hypoglycaemic and hypolipidaemic effect of Allium cepa in streptozotocin-induced diabetes. Int. J. Sci. Eng. Res. 2015, 6, 23–29. [Google Scholar]
- Benítez, V.; Mollá, E.; Martín-Cabrejas, M.A.; Aguilera, Y.; López-Andréu, F.J.; Esteban, R.M. Onion (Allium cepa L.) by-products as source of dietary fibre: Physicochemical properties and effect on serum lipid levels in high-fat fed rats. Eur. Food Res. Technol. 2012, 234, 617–625. [Google Scholar] [CrossRef]
- Henagan, T.M.; Cefalu, W.T.; Ribnicky, D.M.; Noland, R.C.; Dunville, K.; Campbell, W.W.; Stewart, L.K.; Forney, L.A.; Gettys, T.W.; Chang, J.S.; et al. In vivo effects of dietary quercetin and quercetin-rich red onion extract on skeletal muscle mitochondria, metabolism and insulin sensitivity. Genes Nutr. 2015, 10, 2. [Google Scholar] [CrossRef]
- Brüll, V.; Burak, C.; Stoffel-Wagner, B.; Wolffram, S.; Nickenig, G.; Müller, C.; Langguth, P.; Alteheld, B.; Fimmers, R.; Naaf, S.; et al. Effects of a quercetin-rich onion skin extract on 24 h ambulatory blood pressure and endothelial function in overweight-to-obese patients with (pre-) hypertension: A randomised double-blinded placebo-controlled cross-over trial. Br. J. Nutr. 2015, 114, 1263–1277. [Google Scholar] [CrossRef]
- Choi, E.Y.; Lee, H.; Woo, J.S.; Jang, H.H.; Hwang, S.J.; Kim, H.S.; Kim, W.S.; Kim, Y.S.; Choue, R.; Cha, Y.J.; et al. Effect of onion peel extract on endothelial function and endothelial progenitor cells in overweight and obese individuals. Nutrition 2015, 31, 1131–1135. [Google Scholar] [CrossRef]
- Park, S.; Kim, M.Y.; Lee, D.H.; Lee, S.H.; Baik, E.J.; Moon, C.H.; Park, S.W.; Ko, E.Y.; Oh, S.R.; Jung, Y.S. Methanolic extract of onion (Allium cepa) attenuates ischemia/hypoxia-induced apoptosis in cardiomyocytes via antioxidant effect. Eur. J. Nutr. 2009, 48, 235–242. [Google Scholar] [CrossRef]
- Kazak, L.; Chouchani, E.T.; Lu, G.Z.; Jedrychowski, M.P.; Bare, C.J.; Mina, A.I.; Kumari, M.; Zhang, S.; Vuckovic, I.; Laznik-Bogoslavski, D.; et al. Genetic depletion of adipocyte creatine metabolism inhibits diet-induced thermogenesis and drives obesity. Cell Metab. 2017, 26, 660–671. [Google Scholar] [CrossRef]
- Westerterp, K.R. Diet induced thermogenesis. Nutr. Metab. 2004, 1, 5. [Google Scholar] [CrossRef]
- Chen, Y.; Zeng, X.; Huang, X.; Serag, S.; Woolf, C.J.; Spiegelman, B.M. Crosstalk between KCNK3-mediated ion current and adrenergic signaling regulates adipose thermogenesis and obesity. Cell 2017, 171, 836–848. [Google Scholar] [CrossRef] [PubMed]
- Bastien, M.; Poirier, P.; Lemieux, I.; Després, J.P. Overview of epidemiology and contribution of obesity to cardiovascular disease. Prog. Cardiovasc. Dis. 2014, 56, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Franssen, R.; Monajemi, H.; Stroes, E.S.; Kastelein, J.J. Obesity and dyslipidaemia. Med. Clin. N. Am. 2011, 95, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J. A comprehensive review on metabolic syndrome. Cardiol. Res. Pract. 2014, 2014, 943162. [Google Scholar] [CrossRef] [PubMed]
- Fantuzzi, G. Adipose tissue, adipokines and inflammation. J. Allergy Clin. Immunol. 2005, 115, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Akash, M.S.H.; Rehman, K.; Chen, S. Spice plant Allium cepa: Dietary supplement for treatment of type 2 diabetes mellitus. Nutrition 2014, 30, 1128–1137. [Google Scholar] [CrossRef] [PubMed]
- Sowers, J.R. Obesity as a cardiovascular risk factor. Am. J. Med. 2003, 115, 37–41. [Google Scholar] [CrossRef]
- Van Gaal, L.F.; Mertens, I.L.; Christophe, E. Mechanisms linking obesity with cardiovascular disease. Nature 2006, 444, 875–880. [Google Scholar] [CrossRef]
- Kendler, B.S. Garlic (Allium sativum) and onion (Allium cepa): A review of their relationship to cardiovascular disease. Prev. Med. 1987, 16, 670–685. [Google Scholar] [CrossRef]
- Kaur, G.; Meena, C. Amelioration of obesity, glucose intolerance and oxidative stress in high-fat diet and low-dose streptozotocin-induced diabetic rats by combination consisting of “curcumin with piperine and quercetin”. ISRN Pharmacol. 2012, 2012, 957283. [Google Scholar] [CrossRef]
- Souza, G.A.; Ebaid, G.X.; Seiva, F.R.; Rocha, K.H.; Galhardi, C.M.; Mani, F.; Novelli, E.L. N-Acetylcysteine an Allium plant compound improves high-sucrose diet-induced obesity and related effects. Evid. Based Complement. Alternat. Med. 2011, 2011, 643269. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, G.A. Alliuocide G, a new flavonoid with potent α-amylase inhibitory activity from Allium cepa L. Arkivoc 2008, 11, 202–209. [Google Scholar]
- Ueda, Y.; Tsubuku, T.; Miyajima, R. Composition of sulphur-containing components in onion and their flavor characters. Biosci. Biotechnol. Biochem. 1994, 58, 108–110. [Google Scholar] [CrossRef] [PubMed]
- Brüll, V.; Burak, C.; Stoffel-Wagner, B.; Wolffram, S.; Nickenig, G.; Müller, C.; Langguth, P.; Alteheld, B.; Fimmers, R.; Stehle, P.; et al. Acute intake of quercetin from onion skin extract does not influence postprandial blood pressure and endothelial function in overweight-to-obese adults with hypertension: A randomized, double-blind, placebo-controlled, crossover trial. Eur. J. Nutr. 2017, 56, 1347–1357. [Google Scholar] [CrossRef] [PubMed]
| Effect | Extract | Test Subjects | Ref. |
|---|---|---|---|
| Pancreatic lipase inhibition | Peel extract obtained with fermented alcohol | - | [79] |
| Leaves hydroalcoholic extract | - | [80] | |
| Onion juice | - | [81] | |
| Adipogenesis inhibition | Heated fresh onion at 92 °C | Rat white preadipocyte cells | [82] |
| Quercetin-rich onion peel 60% aqueous ethanol extract | 3T3-L1 preadipocyte cells | [83] | |
| Quercetin-rich onion peel hydroalcoholic extract | 3T3-L1 preadipocyte cells | [84] | |
| Energy expenditure increase | Onion peels 60% aqueous ethanol extract | 3T3-L1 preadipocyte cells | [85] |
| Influence on expression of inflammatory mediators from the adipose tissue | Onion peel hydroalcoholic extract | Dawley rats | [86] |
| Antihyperlipidemic activity | Petroleum extract | Rats | [87] |
| Peel extract | Mice | [79] | |
| Onion powder | Male SD rats | [88] | |
| Ethanolic extract and fractions | STZ-induced diabetic rats | [58] | |
| Bulbs juice | STZ-induced diabetic male Wistar rats | [89] | |
| Different onion by-products | Rats | [90] | |
| Heated fresh onion at 92 °C | ZDF rats | [82] | |
| Hypoglycaemic potential | Ethanolic extract and fractions | STZ-induced diabetic rats | [58] |
| Bulbs juice | STZ-induced diabetic male Wistar rats | [89] | |
| Peeled onions extracted with 80% ethanol | C57BL/6J mice | [91] | |
| Bulbs infusion | STZ-induced diabetic rats | [57] | |
| Blood pressure lowering effect | Onion skins ethanolic extract | Overweight-to-obese patients with hypertension | [92] |
| Benefits on endothelial function | Hydroalcoholic extract obtained at 50 °C | Healthy overweight and obese patients | [93] |
| Preventive effect on ischemic heart injury | Onion peeled bulbs hydroalcoholic extract | Heart-derived H9c2 cells and rats | [94] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marrelli, M.; Amodeo, V.; Statti, G.; Conforti, F. Biological Properties and Bioactive Components of Allium cepa L.: Focus on Potential Benefits in the Treatment of Obesity and Related Comorbidities. Molecules 2019, 24, 119. https://doi.org/10.3390/molecules24010119
Marrelli M, Amodeo V, Statti G, Conforti F. Biological Properties and Bioactive Components of Allium cepa L.: Focus on Potential Benefits in the Treatment of Obesity and Related Comorbidities. Molecules. 2019; 24(1):119. https://doi.org/10.3390/molecules24010119
Chicago/Turabian StyleMarrelli, Mariangela, Valentina Amodeo, Giancarlo Statti, and Filomena Conforti. 2019. "Biological Properties and Bioactive Components of Allium cepa L.: Focus on Potential Benefits in the Treatment of Obesity and Related Comorbidities" Molecules 24, no. 1: 119. https://doi.org/10.3390/molecules24010119
APA StyleMarrelli, M., Amodeo, V., Statti, G., & Conforti, F. (2019). Biological Properties and Bioactive Components of Allium cepa L.: Focus on Potential Benefits in the Treatment of Obesity and Related Comorbidities. Molecules, 24(1), 119. https://doi.org/10.3390/molecules24010119







