Targeted Neurotransmitters Profiling Identifies Metabolic Signatures in Rat Brain by LC-MS/MS: Application in Insomnia, Depression and Alzheimer’s Disease
Abstract
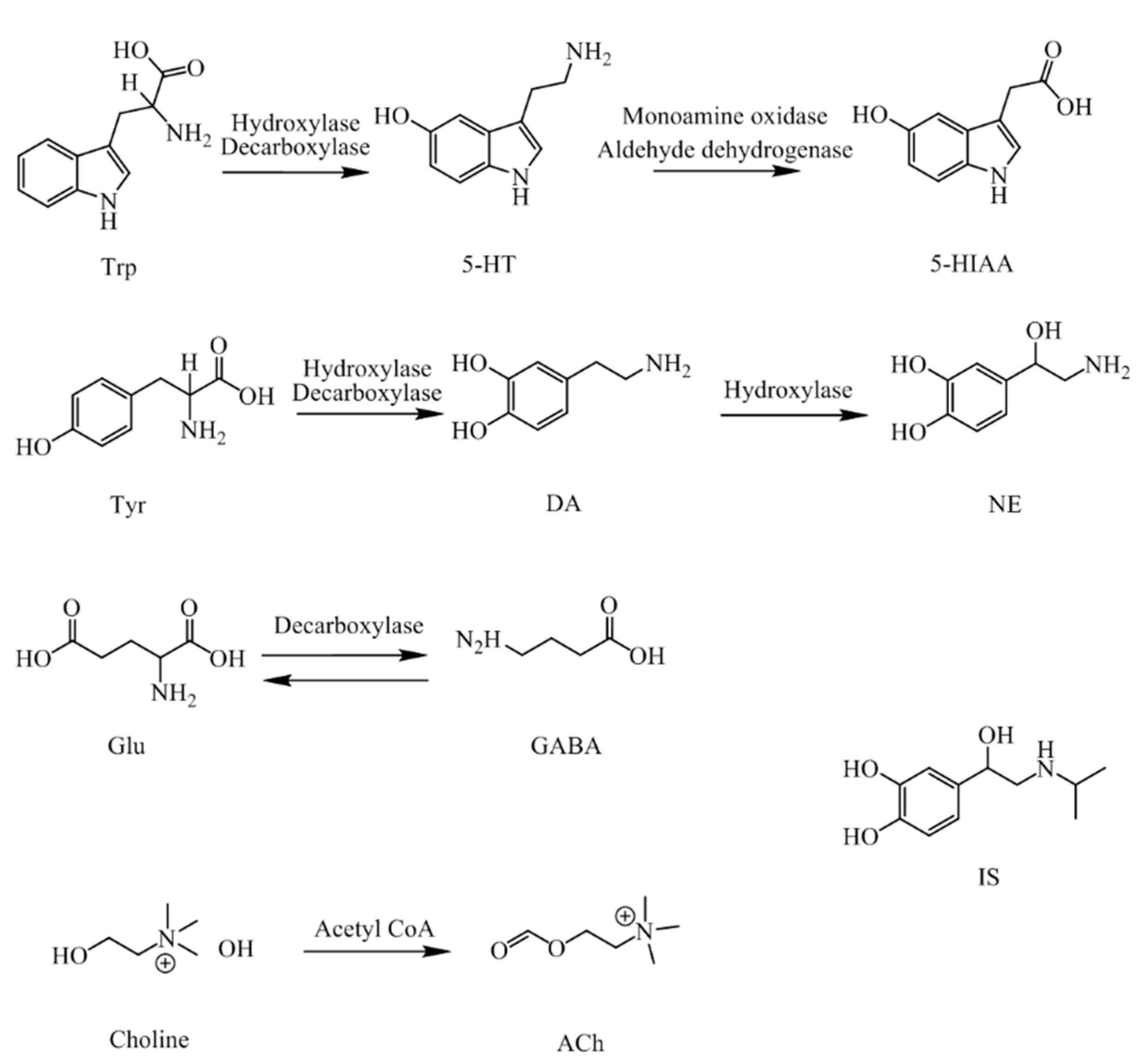
1. Introduction
2. Results and Discussion
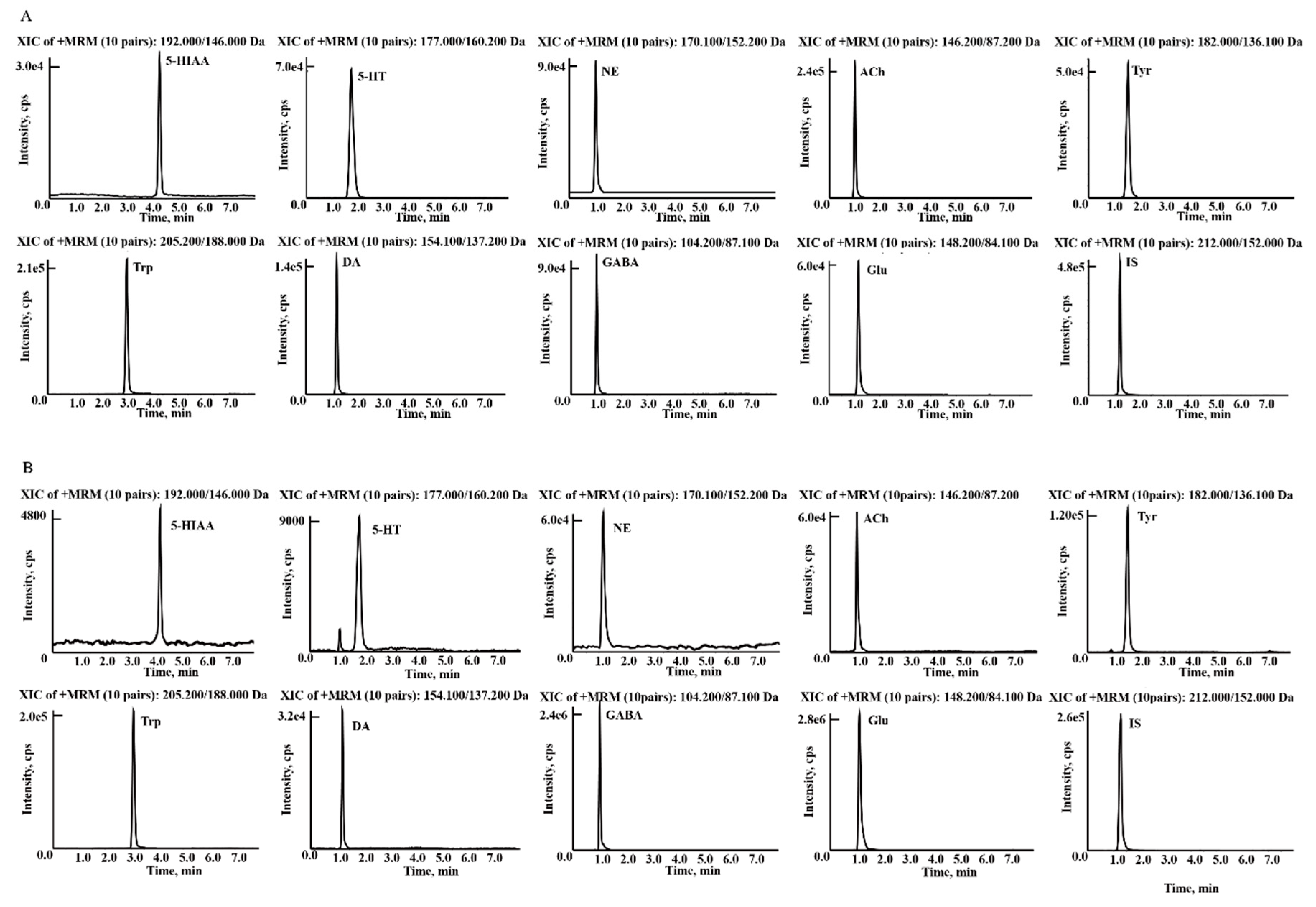
2.1. Optimisation of LC-MS/MS Parameters
2.2. Sample Preparation
2.3. Method Validation
2.3.1. Linearity and LLOQ
2.3.2. Accuracy and Precision
2.3.3. Recovery and Matrix Effect
2.3.4. Stability
2.4. Method Application
2.4.1. Determination of Neurotransmitters in Rat Brain
2.4.2. The Comparation of Insomnia, Depression and AD
3. Materials and Methods
3.1. Materials and Reagents
3.2. Animals and Treatment
3.3. Instruments and LC–MS/MS Conditions
3.4. Standard Solution and Quality Control Samples
3.5. Sample Preparation
3.6. Method Validation
3.7. Statistical Data Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wei, B.; Li, Q.; Fan, R.; Su, D.; Chen, X.; Jia, Y.; Bi, K. Determination of monoamine and amino acid neurotransmitters and their metabolites in rat brain samples by UFLC-MS/MS for the study of the sedative-hypnotic effects observed during treatment with S. chinensis. J. Pharm. Biomed. Anal. 2014, 88, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Si, L.; Wang, Y.; Wuyun, G.; Bao, L.; Agula, B. The effect of Mongolian medical acupuncture on cytokines and neurotransmitters in the brain tissue of insomniac rats. Eur. J. Integr. Med. 2015, 7, 492–498. [Google Scholar] [CrossRef]
- Strekalova, T.; Evans, M.; Chernopiatko, A.; Couch, Y.; Costa-Nunes, J.; Cespuglio, R.; Chesson, L.; Vignisse, J.; Steinbusch, H.W.; Anthony, D.C.; et al. Deuterium content of water increases depression susceptibility: The potential role of a serotonin-related mechanism. Behav. Brain Res. 2015, 277, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Yun, H.M.; Park, K.R.; Kim, E.C.; Kim, S.; Hong, J.T. Serotonin 6 receptor controls Alzheimer’s disease and depression. Oncotarget 2015, 6, 26716–26728. [Google Scholar] [CrossRef] [PubMed]
- Rada, P.; Hoebel, B.G. Acetylcholine in the accumbens is decreased by diazepam and increased by benzodiazepine withdrawal: A possible mechanism for dependency. Eur. J. Pharm. 2005, 508, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Marks, G.A.; Birabil, C.G. Enhancement of rapid eye movement sleep in the rat by cholinergic and adenosinergic agonists infused into the pontine reticular formation. Neuroscience 1998, 86, 29–37. [Google Scholar] [CrossRef]
- Coleman, C.G.; Lydic, R.; Baghdoyan, H.A. M2 muscarinic receptors in pontine reticular formation of C57BL/6J mouse contribute to rapid eye movement sleep generation. Neuroscience 2004, 126, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.L.; Miller, B.; Hill, M.A.; Neshkes, R. Neuropsychiatric aspects of multi-infarct dementia and dementia of the Alzheimer type. Arch. Neurol. 1987, 44, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Risch, S.C.; Cohen, R.M.; Janowsky, D.S.; Kalin, N.H.; Murphy, D.L. Mood and behavioral effects of physostigmine on humans are accompanied by elevations in plasma beta-endorphin and cortisol. Science 1980, 209, 1545–1546. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pi, Z.; Song, F.; Liu, Z. Ginsenosides attenuate d-galactose- and AlCl3-inducedspatial memory impairment by restoring the dysfunction of the neurotransmitter systems in the rat model of Alzheimer’s disease. J. Ethnopharmacol. 2016, 194, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Altamura, C.; Maes, M.; Dai, J.; Meltzer, H.Y. Plasma concentrations of excitatory amino acids, serine, glycine, taurine and histidine in major depression. Eur. Neuropsychopharmacol. 1995, 5, 71–75. [Google Scholar] [CrossRef]
- Fekkes, D.; Timmerman, L.; Pepplinkhuizen, L. Effects of clomipramine on plasma amino acids and serotonergic parameters in panic disorder and depression. Eur. Neuropsychopharmacol. 1997, 7, 235–239. [Google Scholar] [CrossRef]
- Bergquist, J.; Sciubisz, A.; Kaczor, A.; Silberring, J. Catecholamines and methods for their identification and quantitation in biological tissues and fluids. J. Neurosci. Methods 2002, 113, 1–13. [Google Scholar] [CrossRef]
- Bourcier, S.; Benoist, J.F.; Clerc, F.; Rigal, O.; Taghi, M.; Hoppilliard, Y. Detection of 28 neurotransmitters and related compounds in biological fluids by liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrum. RCM 2006, 20, 1405–1421. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Fang, C.; Smagin, G. Derivatization for the simultaneous LC/MS quantification of multiple neurotransmitters in extracellular fluid from rat brain microdialysis. J. Pharm. Biomed. Anal. 2014, 100, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Perry, M.; Li, Q.; Kennedy, R.T. Review of recent advances in analytical techniques for the determination of neurotransmitters. Anal. Chim. Acta 2009, 653, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Sanli, N.; Tague, S.E.; Lunte, C. Analysis of amino acid neurotransmitters from rat and mouse spinal cords by liquid chromatography with fluorescence detection. J. Pharm. Biomed. Anal. 2015, 107, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.; Zhong, X.; Greer, T.; Ye, H.; Li, L. Relative quantification of amine-containing metabolites using isobaric N,N-dimethyl leucine (DiLeu) reagents via LC-ESI-MS/MS and CE-ESI-MS/MS. Analyst 2015, 140, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Li, J.; Shi, H.L.; Wang, T.T.; Muhtar, W.; Du, M.; Zhang, B.B.; Wu, H.; Yang, L.; Hu, Z.B.; et al. Simultaneous quantification of seven hippocampal neurotransmitters in depression mice by LC-MS/MS. J. Neurosci. Methods 2014, 229, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Zhu, B.; Liu, F.; Lyu, C.; Zhang, S.; Yan, C.; Cheng, Y.; Wei, H. A rapid and simple method for the simultaneous determination of four endogenous monoamine neurotransmitters in rat brain using hydrophilic interaction liquid chromatography coupled with atmospheric-pressure chemical ionization tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci 2015, 1002, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Cheng, G. Approximative Bayes optimality linear discriminant analysis for Chinese handwriting character recognition. Neurocomputing 2016, 207, 346–353. [Google Scholar] [CrossRef]
- Wang, L.E.; Bai, Y.J.; Shi, X.R.; Cui, X.Y.; Cui, S.Y.; Zhang, F.; Zhang, Q.Y.; Zhao, Y.Y.; Zhang, Y.H. Spinosin, a C-glycoside flavonoid from semen Zizhiphi Spinozae, potentiated pentobarbital-induced sleep via the serotonergic system. Pharmacol. Biochem. Behav. 2008, 90, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Shahar, S.; Hassan, J.; Sundar, V.V.; Kong, A.Y.; Ping Chin, S.; Ahmad, S.A.; Kuan Lee, L. Determinants of depression and insomnia among institutionalized elderly people in Malaysia. Asian J. Psychiatry 2011, 4, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Livingston, G.; Blizard, B.; Mann, A. Does sleep disturbance predict depression in elderly people? A study in inner London. Br. J. Gen. Pract. 1993, 43, 445–448. [Google Scholar] [PubMed]
- Zailinawati, A.; Ariff, K.; Nurjahan, M.; Teng, C. Epidemiology of insomnia in Malaysian adults: A community-based survey in 4 urban areas. Asia-Pac. J. Pub. Health 2008, 20, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Doraiswamy, P.M.; Xiong, G.L. Pharmacological strategies for the prevention of Alzheimer’s disease. Expert Opin. Pharmacother. 2006, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Farlow, M.R. Utilizing combination therapy in the treatment of Alzheimer’s disease. Expert Rev. Neurother. 2004, 4, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Toledo, J.B.; Shaw, L.M.; Trojanowski, J.Q. Plasma amyloid beta measurements-a desired but elusive Alzheimer’s disease biomarker. Alzheimer’s Res. Ther. 2013, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Benedict, C.; Byberg, L.; Cedernaes, J.; Hogenkamp, P.S.; Giedratis, V.; Kilander, L.; Lind, L.; Lannfelt, L.; Schiöth, H.B. Self-reported sleep disturbance is associated with Alzheimer’s disease risk in men. Alzheimer’s Dementia 2015, 11, 1090–1097. [Google Scholar] [CrossRef] [PubMed]
- Reid, I.; Forbes, N.; Stewart, C.; Matthews, K. Chronic mild stress and depressive disorder: A useful new model? Psychopharmacology 1997, 134, 365–367. [Google Scholar] [CrossRef] [PubMed]
- De Vry, J.; Schreiber, R. The chronic mild stress depression model: Future developments from a drug discovery perspective. Psychopharmacology 1997, 134, 349–350. [Google Scholar] [CrossRef] [PubMed]
- Borbely, A.A.; Neuhaus, H.U.; Tobler, I. Effect of p-chlorophenylalanine and tryptophan on sleep, EEG and motor activity in the rat. Behav. Brain Res. 1981, 2, 1–22. [Google Scholar] [CrossRef]
- Wang, Q.; Xu, Z.; Tang, J.; Sun, J.; Gao, J.; Wu, T.; Xiao, M. Voluntary exercise counteracts Abeta25-35-induced memory impairment in mice. Behav. Brain Res. 2013, 256, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Huitron-Resendiz, S.; Rios, C.; Rojas, P.; Mexicano, G.; Ayala-Guerrero, F. Effect of p-chlorophenylalanine (PCPA) on sleep and monoamines content in the brain of a lizard species. Brain Res. 1997, 761, 19–24. [Google Scholar] [CrossRef]
- Shen, Y.X.; Yang, J.; Wei, W.; Liu, L.H.; Xu, S.Y. Induction of learning and memory dysfunction by beta-amyloid peptide fragment 25∼35 in rats. Chin. Pharmacol. Bull. 2001, 17, 26–29. [Google Scholar] [CrossRef]
- Willner, P.; Towell, A.; Sampson, D.; Sophokleous, S.; Muscat, R. Reduction of sucrose preference by chronic unpredictable mild stress, and its restoration by a tricyclic antidepressant. Psychopharmacology 1987, 93, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Bioanalytical Method Validation Guidance for Industry. Available online: https://www.fda.gov/downloads/drugs/guidances/ucm070107.Pdf (accessed on 3 June 2018).
Sample Availability: Samples of the compounds mentioned in the manuscript are available from the authors. |
| Analyte | Linear Regression Equation | Linear Range (ng/mL) | Correlation Coefficient (R2) | LLOQ | |
|---|---|---|---|---|---|
| Accuracy (RE%) | Precision (RSD%) | ||||
| 5-HT | y = 3.675 × 10−4 x + 9.17 × 10−4 | 2.0~64 | 0.9979 | 10.8 | 11.5 |
| 5-HIAA | y = 1.568 × 10−4 x + 9.85 × 10−4 | 2.0~64 | 0.9920 | −5.9 | 10.2 |
| Glu | y = 1.617 × 10−1 x + 1.954 | 1.0 × 103~3.2 × 104 | 0.9928 | 3.4 | 8.1 |
| GABA | y = 2.792 × 10−1 x + 1.325 | 1.0 × 103~3.2 × 104 | 0.9935 | −6.9 | 7.8 |
| DA | y = 8.126 × 10−4 x + 1.353 × 10−3 | 20~6.4 × 102 | 0.9983 | 11.9 | 9.7 |
| ACh | y = 1.802 × 10−3 x + 1.571 × 10−2 | 20~6.4 × 102 | 0.9976 | 7.9 | 10.5 |
| Trp | y = 1.063 × 10−3 x + 1.042 × 10−1 | 20~6.4 × 102 | 0.9961 | 7.9 | 3.7 |
| Tyr | y = 6.268 × 10−4 x + 4.080 × 10−2 | 1.0 × 102~3.2 × 103 | 0.9944 | 9.1 | 6.3 |
| NE | y = 1.536 × 10−3x + 2.270 × 10−2 | 1.0 × 102~3.2 × 103 | 0.9974 | −7.4 | 5.6 |
| Analytes | Concentration (ng mL−1) | Intra-Day RSD (%) | Inter-Day RSD (%) | Accuracy (RE%) | Recovery (%, mean ± SD) | Matrix Effect (%, mean ± SD) |
|---|---|---|---|---|---|---|
| 5-HT | 4 | 3.1 | 3.6 | −11.4 | 105.6 ± 7.4 | 98.5 ± 9.5 |
| 16 | 5.2 | 1.6 | −9.0 | 99.0 ± 9.4 | 98.0 ± 1.9 | |
| 50 | 4.8 | 7.2 | 7.3 | 102.0 ± 8.8 | 98.7 ± 2.0 | |
| 5-HIAA | 4 | 8.9 | 14.7 | 1.5 | 97.0 ± 6.7 | 92.2 ± 8.6 |
| 16 | 7.8 | 13.7 | 4.2 | 95.5 ± 6.5 | 92.2 ± 6.3 | |
| 50 | 6.7 | 10.6 | 7.0 | 97.5 ± 7.2 | 90.2 ± 5.7 | |
| Glu | 2000 | 6.5 | 2.5 | 3.0 | 102.9 ± 9.8 | 101.4 ± 8.5 |
| 8000 | 4.4 | 1.0 | 8.4 | 99.5 ± 6.4 | 95.8 ± 4.4 | |
| 25,000 | 3.9 | 4.4 | 10.9 | 98.9 ± 9.9 | 97.1 ± 5.9 | |
| GABA | 2000 | 6.9 | 6.0 | −1.7 | 102.2 ± 8.1 | 95.4 ± 10.8 |
| 8000 | 4.9 | 5.8 | 2.3 | 105.1 ± 4.7 | 98.3 ± 5.2 | |
| 25,000 | 9.4 | 7.4 | 12.9 | 92.6 ± 9.0 | 99.2 ± 4.0 | |
| DA | 40 | 5.4 | 3.2 | 8.0 | 98.1 ± 8.0 | 90.1 ± 6.8 |
| 160 | 2.9 | 2.1 | 12.0 | 95.2 ± 7.9 | 94.6 ± 3.8 | |
| 500 | 13.4 | 0.7 | 13.4 | 94.4 ± 6.8 | 99.8 ± 5.4 | |
| NE | 200 | 6.4 | 2.8 | 12.9 | 96.4 ± 9.5 | 102.2 ± 7.0 |
| 800 | 5.4 | 4.9 | −13.4 | 90.6 ± 4.3 | 104.3 ± 6.2 | |
| 2500 | 7.9 | 5.4 | 4.3 | 95.0 ± 5.2 | 97.0 ± 4.7 | |
| ACh | 40 | 5.4 | 1.2 | 4.6 | 92.3 ± 12.5 | 99.5 ± 7.7 |
| 160 | 2.4 | 3.1 | 12.6 | 95.5 ± 9.4 | 91.3 ± 3.6 | |
| 500 | 11.4 | 7.0 | 9.3 | 96.9 ± 7.8 | 103.4 ± 2.3 | |
| Trp | 40 | 9.4 | 3.5 | −5.4 | 93.2 ± 9.8 | 101.0 ± 9.7 |
| 160 | 7.2 | 2.2 | 1.7 | 101.4 ± 8.0 | 96.9 ± 6.2 | |
| 500 | 4.1 | 0.8 | 4.1 | 98.9 ± 6.0 | 101.9 ± 5.6 | |
| Tyr | 200 | 6.0 | 7.1 | −1.9 | 95.5 ± 8.9 | 102.4 ± 6.0 |
| 800 | 3.2 | 6.2 | 7.8 | 98.6 ± 7.8 | 101.4 ± 1.7 | |
| 2500 | 4.2 | 4.2 | 8.8 | 96.7 ± 6.5 | 97.3 ± 7.9 |
| Analytes | Concentration (ng mL−1) | 8 h, 4 °C | 8 h, Room Temperature | 3 Freeze-Thaw Cycles | −80 °C for 3 Months | ||||
|---|---|---|---|---|---|---|---|---|---|
| RE (%) | RSD (%) | RE (%) | RSD (%) | RE (%) | RSD (%) | RE (%) | RSD (%) | ||
| 5-HT | 4 | 3.0 | 4.5 | −4.9 | 3.3 | −2.9 | 4.1 | 4.2 | 2.7 |
| 50 | 1.3 | 2.0 | −2.1 | 2.3 | 0.8 | 3.0 | −1.0 | 5.7 | |
| 5-HIAA | 4 | −9.3 | 1.6 | −9.0 | 4.5 | 2.3 | 1.0 | 1.0 | 2.2 |
| 50 | −7.4 | 7.4 | −3.7 | 5.2 | −1.1 | 6.9 | −4.6 | 2.3 | |
| Glu | 2000 | −5.0 | 3.6 | −5.9 | 6.0 | −2.0 | 1.8 | 4.1 | 6.4 |
| 25,000 | 8.5 | 5.2 | 6.6 | 3.7 | −2.0 | 6.3 | −6.6 | 3.5 | |
| GABA | 2000 | −4.0 | 2.6 | 3.8 | 2.2 | 3.1 | 1.4 | 1.5 | 1.9 |
| 25,000 | 2.7 | 4.6 | 6.7 | 1.6 | 5.9 | 6.6 | −3.1 | 1.5 | |
| DA | 40 | 5.7 | 9.5 | -9.6 | 8.8 | 1.7 | 0.7 | −2.8 | 5.8 |
| 500 | 2.3 | 7.1 | 1.9 | 6.6 | −1.2 | 5.0 | 4.5 | 4.2 | |
| NE | 200 | −3.2 | 7.2 | 2.7 | 5.0 | −2.7 | 2.8 | −2.5 | 3.6 |
| 2500 | −3.2 | 5.0 | −4.7 | 2.1 | 3.6 | 2.8 | −1.2 | 2.9 | |
| ACh | 40 | 8.7 | 4.4 | −5.8 | 2.4 | −2.9 | 1.7 | 2.2 | 1.2 |
| 500 | −6.0 | 5.6 | −1.5 | 2.5 | −2.6 | 7.7 | −5.3 | 1.6 | |
| Trp | 40 | 1.1 | 3.2 | 11.4 | 5.5 | 2.7 | 2.1 | −1.9 | 0.9 |
| 500 | −3.9 | 7.0 | 5.9 | 2.7 | 1.1 | 4.7 | −0.9 | 2.4 | |
| Tyr | 200 | 2.6 | 3.8 | 2.9 | 5.8 | 5.1 | 3.7 | −2.7 | 2.1 |
| 2500 | −7.8 | 3.5 | −5.7 | 6.0 | 2.1 | 5.4 | −1.2 | 1.3 | |
| Analyte (ng mg−1) | Control Group | Insomnia Group | Depression Group | AD Group |
|---|---|---|---|---|
| 5-HT | 1.551 ± 0.262 | 0.449 ± 0.113 aa | 0.940 ± 0.061 aa, bb | 0886 ± 0.087 aa,bb |
| 5-HIAA | 0.960 ± 0.263 | 0.556 ± 0.135 aa | 0.324 ± 0.121 aa | 0.766 ± 0.306 c |
| Glu | 1219.476 ± 94.828 | 948.926 ± 144.407 aa | 988.866 ± 161.352 a | 1288.012 ± 56.815 bb,cc |
| GABA | 861.936 ± 83.004 | 840.986 ± 139.038 | 896.462 ± 71.449 | 623.404 ± 140.555 a,bb,cc |
| DA | 7.225 ± 0.873 | 10.315 ± 0.824 aa | 4.022 ± 0.972 aa,bb | 5.353 ± 1.016 aa,bb,cc |
| NE | 22.616 ± 3.435 | 53.086 ± 3.267 aa | 19.771 ± 2.776 a,bb | 17.602 ± 2.372 aa,bb |
| ACh | 3.787 ± 0.506 | 5.059 ± 0.590 aa | 2.510 ± 0.273 aa,bb | 3.132 ± 0.322 aa,bb,c |
| Trp | 21.837 ± 2.311 | 19.405 ± 2.265 a | 18.135 ± 1.769 aa | 14.791 ± 1.721 aa,bb,cc |
| Tyr | 39.254 ± 5.862 | 74.728 ± 10.452 aa | 19.644 ± 2.120 aa,bb | 29.533 ± 6.896 a,bb,c |
| Group | ||||
|---|---|---|---|---|
| Blank | Insomnia | Depression | AD | |
| DA | 7.740 | 10.723 | 4.310 | 5.692 |
| ACh | 16.614 | 21.920 | 11.152 | 13.836 |
| Tyr | 0.760 | 1.533 | 0.367 | 0.571 |
| Constant | −75.047 | −169.858 | −27.946 | −47.148 |
| Analyte | Q1 Mass (Da) | Q3 Mass (Da) | DP | EP | CE | CXP |
|---|---|---|---|---|---|---|
| 5-HT | 177.1 | 160.2 | 38 | 10 | 10 | 7 |
| 5-HIAA | 192.0 | 146.0 | 25 | 9 | 20 | 8 |
| Glu | 148.2 | 84.1 | 41 | 10 | 24 | 15 |
| GABA | 104.2 | 87.1 | 26 | 14 | 15 | 16 |
| DA | 154.1 | 137.2 | 37 | 100 | 13 | 8 |
| NE | 170.1 | 152.2 | 33 | 5 | 11 | 9 |
| ACh | 146.2 | 87.2 | 52 | 3 | 20 | 4 |
| Trp | 205.2 | 188.0 | 40 | 4 | 14 | 12 |
| Tyr | 182.0 | 136.1 | 52 | 8 | 25 | 7 |
| IS | 212.0 | 152.0 | 46 | 8 | 20 | 9 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, H.; Wang, Z.; Zhu, L.; Sui, Z.; Bi, W.; Liu, R.; Bi, K.; Li, Q. Targeted Neurotransmitters Profiling Identifies Metabolic Signatures in Rat Brain by LC-MS/MS: Application in Insomnia, Depression and Alzheimer’s Disease. Molecules 2018, 23, 2375. https://doi.org/10.3390/molecules23092375
Xu H, Wang Z, Zhu L, Sui Z, Bi W, Liu R, Bi K, Li Q. Targeted Neurotransmitters Profiling Identifies Metabolic Signatures in Rat Brain by LC-MS/MS: Application in Insomnia, Depression and Alzheimer’s Disease. Molecules. 2018; 23(9):2375. https://doi.org/10.3390/molecules23092375
Chicago/Turabian StyleXu, Huarong, Zhenru Wang, Lin Zhu, Zhenyu Sui, Wenchuan Bi, Ran Liu, Kaishun Bi, and Qing Li. 2018. "Targeted Neurotransmitters Profiling Identifies Metabolic Signatures in Rat Brain by LC-MS/MS: Application in Insomnia, Depression and Alzheimer’s Disease" Molecules 23, no. 9: 2375. https://doi.org/10.3390/molecules23092375
APA StyleXu, H., Wang, Z., Zhu, L., Sui, Z., Bi, W., Liu, R., Bi, K., & Li, Q. (2018). Targeted Neurotransmitters Profiling Identifies Metabolic Signatures in Rat Brain by LC-MS/MS: Application in Insomnia, Depression and Alzheimer’s Disease. Molecules, 23(9), 2375. https://doi.org/10.3390/molecules23092375





