Optimization of Antioxidant and Skin-Whitening Compounds Extraction Condition from Tenebrio molitor Larvae (Mealworm)
Abstract
:1. Introduction
2. Results
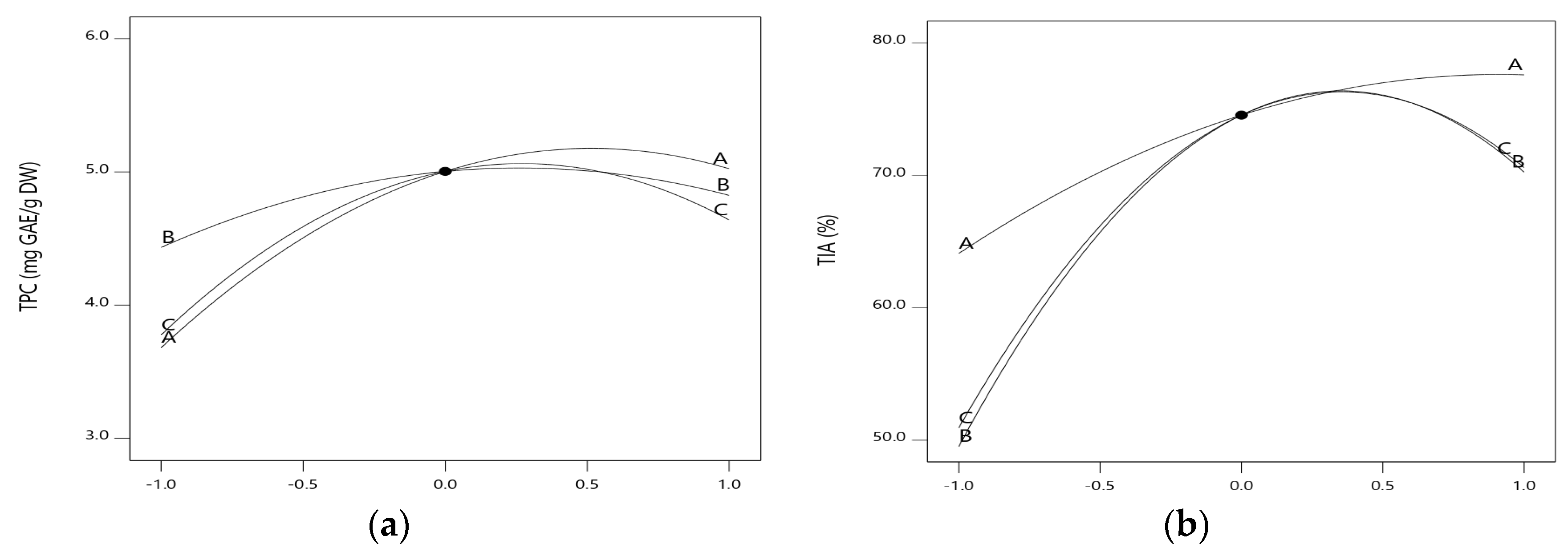
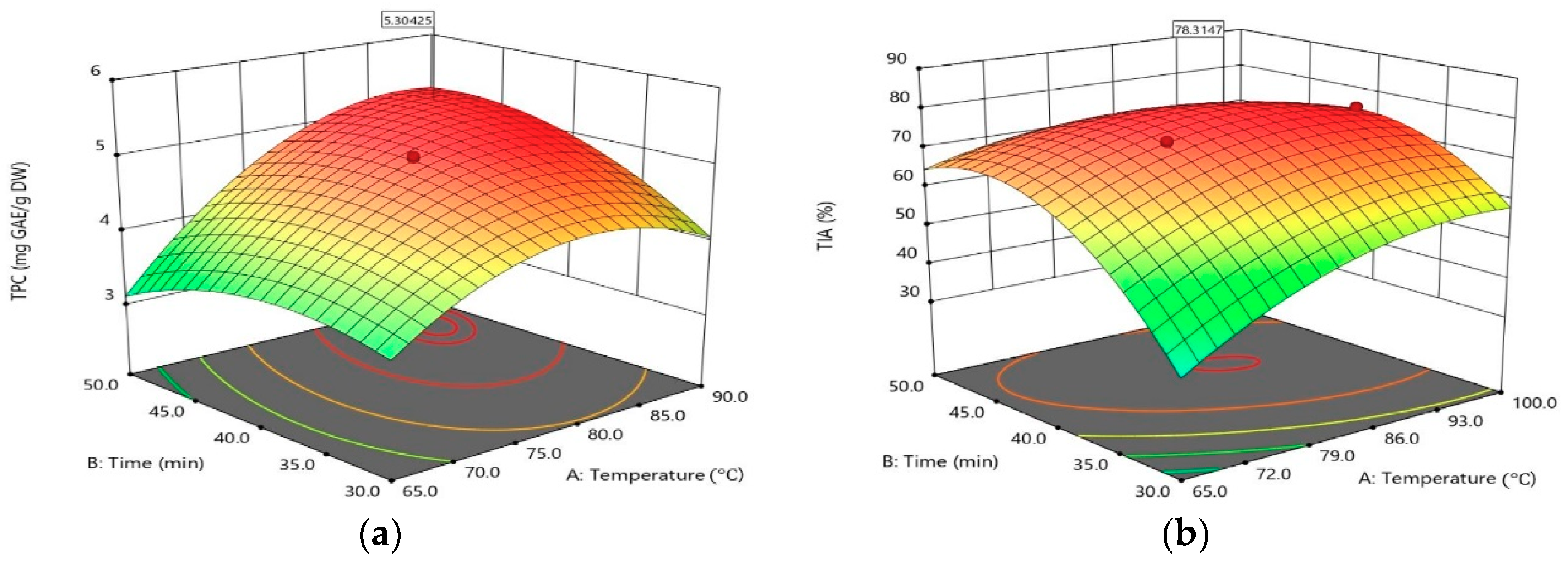
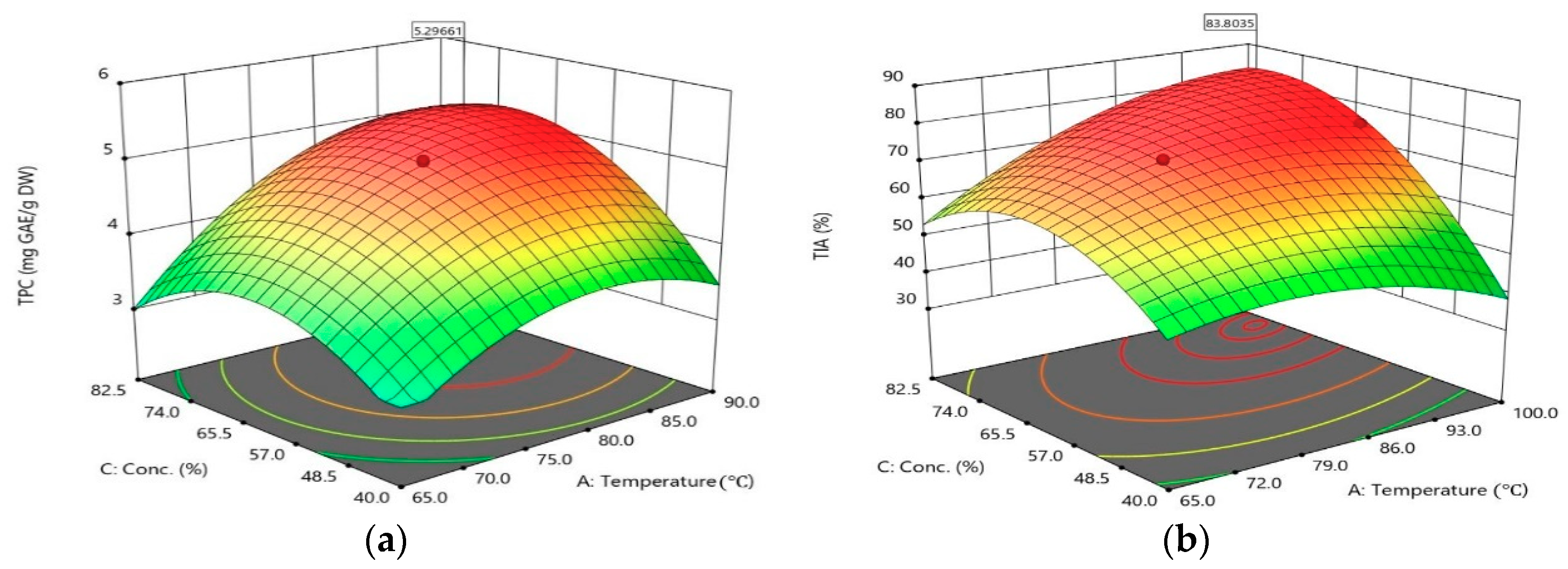
2.1. Opimization of Extraction Variables
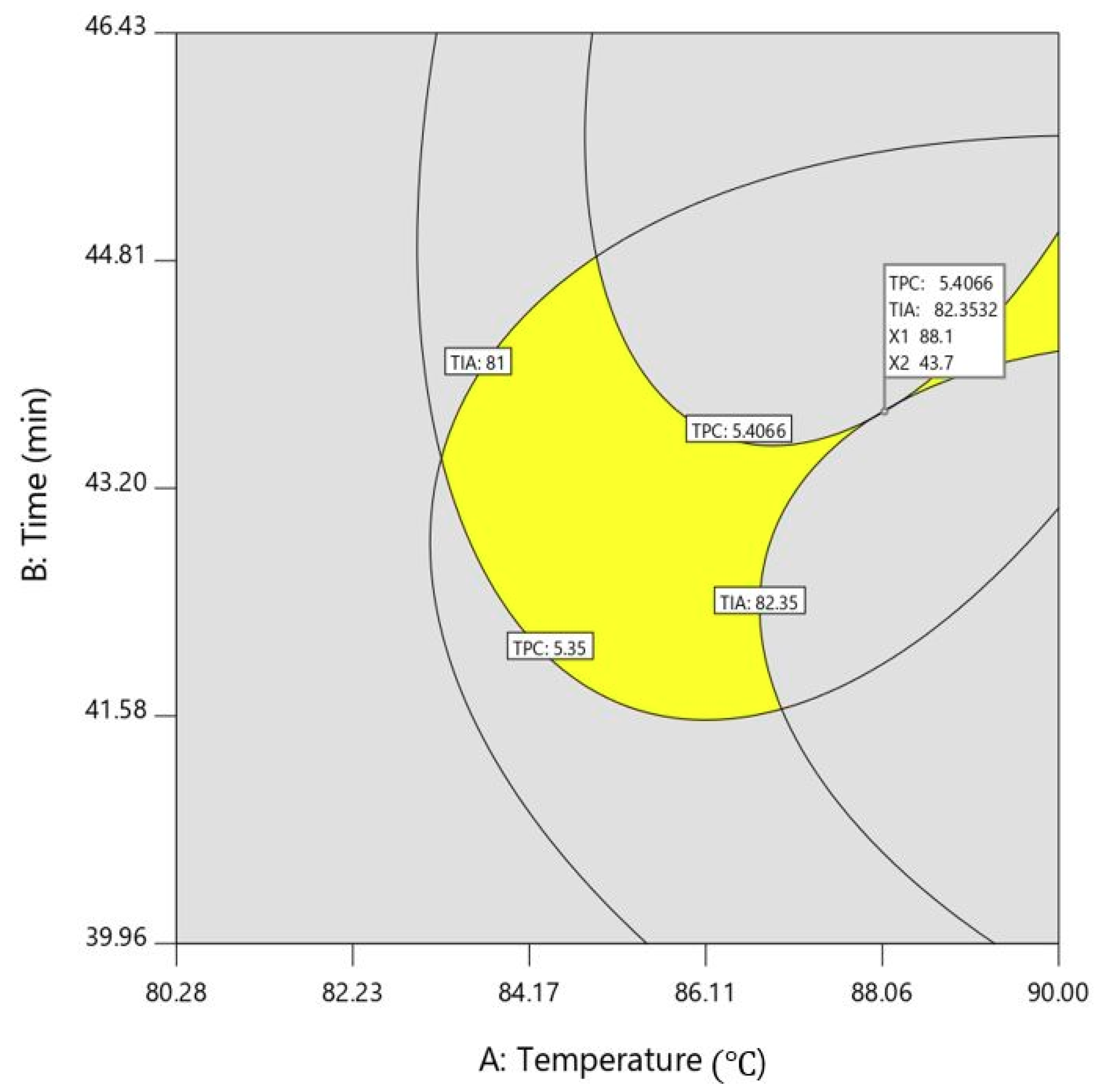
2.2. Process Optimization and Validation
2.3. Antioxidant Activity (Radical Scavenging Activity)
3. Discussion
4. Materials and Methods
4.1. Mealworms
4.2. Extraction
4.3. Determination of Total Phenolic Compounds (TPC)
4.4. Determination of Tyrosinase Inhibition Activity (TIA)
4.5. DPPH Radical Scavenging Assay
4.6. Experimental Design
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Premalatha, M.; Abbasi, T.; Abbasi, T.; Abbasi, S.A. Energy-efficient food production to reduce global warming and ecodegradation: The use of edible insects. Renew. Sustain. Energy Rev. 2011, 15, 4357–4360. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, J.; Wu, W.M.; Zhao, J.; Song, Y.; Gao, L.; Yang, R.; Jiang, L. Biodegradation and mineralization of polystyrene plastic-eating mealworms: Part 1. Chemical and physical characterization and isotopic tests. Environ. Sci. Technol. 2015, 49, 12080–12086. [Google Scholar] [CrossRef] [PubMed]
- Huis, A.V.; Ltterbeeck, J.V.; Klunder, H.; Mertens, E.; Halloran, A.; Muir, G.; Vantomme, P. Edible insects-Future prospects for food and feed security. FAO For. Pap. 2013, 171, 1–201. [Google Scholar]
- Nakagaki, B.J.; DeFoliart, G.R. Comparison of diets for mass-rearing Acheta domesticus with values reported for livestock. J. Econ. Entomol. 1991, 84, 891–896. [Google Scholar] [CrossRef]
- Mlcek, J.; Rob, O.; Borkovcova, M.; Bednarova, M. A Comprehensive Look at the Possibilities of Edible Insects as Food in Europe-A Review. Pol. J. Food Nutr. Sci. 2014, 64, 147–157. [Google Scholar] [CrossRef]
- Maeda, K.; Fukuda, M. In vitro effectiveness of several whitening cosmetic components in human melanocytes. J. Soc. Cosmet. Chem. 1991, 42, 361–368. [Google Scholar]
- Whittaker, J.R. Food Enzymes, Structure and Mechanism; Chapman and Hall: New York, NY, USA, 1995. [Google Scholar]
- Solano, F.; Briganti, S.; Picardo, M.; Ghanem, G. Hypopigmenting agents: An updated review on biological, chemical and clinical aspects. Pigment Cell Melanoma Res. 2006, 19, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2400–2475. [Google Scholar] [CrossRef] [PubMed]
- Frenk, E. Treatment of melasma with depigmenting agents. In Melasma: New Approaches to Treatment; Martin Dunitz: London, UK, 1995. [Google Scholar]
- Hunt, K.J.; Hung, S.K.; Ernst, E. Botanical extracts as anti-aging preparations for the skin: A systematic review. Drugs Aging 2010, 27, 973–985. [Google Scholar] [CrossRef] [PubMed]
- Antignac, E.; Nohynek, G.J.; Re, T.; Clouzeau, J.; Toutain, H. Safety of botanical ingredients in personal care products/cosmetics. Food Chem. Toxicol. 2011, 49, 324–341. [Google Scholar] [CrossRef] [PubMed]
- Baumann, L.; Woolery-Lloyd, H.; Friedman, A. Natural ingredients in cosmetic dermatology. J. Drugs Dermatol. 2009, 8, 5–9. [Google Scholar]
- Yang, Y.F.; Lai, X.Y.; Lai, G.Y.; Jian, Z.D.; Ni, H.; Chen, F. Purification and characterization of a tyrosinase inhibitor from camellia pollen. J. Funct. Foods 2016, 27, 140–149. [Google Scholar] [CrossRef]
- Nithitanakool, S.; Pithayanukul, P.; Bavovada, R.; Saparpakorn, P. Molecular docking studies and anti-tyrosinase activity of Thai mango seed kernel extract. Molecules 2009, 14, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Saewan, N.; Koysomboon, S.; Chantrapromma, K. Anti-tyrosinase and anti-cancer activities of flavonoids from Blumea balsamifera DC. J. Med. Plants Res. 2011, 5, 1018–1025. [Google Scholar]
- Masamoto, Y.; Ando, H.; Murata, Y.; Shimoshi, Y.; Tada, M.; Takahata, K. Mushroom tyrosinase inhibitory activity of Esculetin Isolated from Seeds of Euphorbia lathyris L. Biosci. Biotechol. Biochem. 2003, 67, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Youshizaki, Y. Nobiletin as a tyrosinase inhibitor from the peel of Citrus fruit. Biol. Pharm. Bull. 2002, 25, 806–808. [Google Scholar] [CrossRef] [PubMed]
- Husni, A.; Jeon, J.S.; Um, B.H.; Han, N.S.; Chung, D. Tyrosinase inhibitory by water and ethanol extracts of far eastern sea cucumber, Stichopus japonicus. J. Sci. Food Agric. 2011, 91, 1541–1547. [Google Scholar] [CrossRef] [PubMed]
- Manosroi, A.; Boonpisuttinant, K.; Winitchai, S.; Manosroi, W.; Manosroi, J. Free radical scavenging and tyrosinase inhibition Activity of oils and sericin extracted from Thai native silkworms (Bombyx mori). Pharm. Biol. 2010, 48, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.S.; Yang, M.R.; No, G.R.; Kim, D.H.; Bang, W.Y.; Kim, S.W. Identification of antioxidant properties of various extracts from mealworm, Tenebrio molitor, for development of functional food. Korean Soc. Biotechnol. Bioeng. 2015, 10, 555. [Google Scholar]
- Alves, A.V.; Sanjinez-Argandona, E.J.; Linzmeier, A.M.; Cardoso, C.A.L.; Macedo, M.L.R. Food Value of mealworm grown on Acrocomia aculeata pulp flour. PLoS ONE 2016, 14, 11. [Google Scholar] [CrossRef] [PubMed]
- Meetali, D.; Dulal, C.B.; Dipali, D.; Rajlakshmi, D. Antioxidant and antigenotoxic effects of pupae of the muga silkworm Antheraea assamensis. Food Biosci. 2014, 5, 108–114. [Google Scholar]
- Kim, J.W.; Mazza, G. Optimization of extraction of phenolic compounds from flax shives by pressurized low-polarity water. J. Agric. Food Chem. 2006, 54, 7575–7584. [Google Scholar] [CrossRef] [PubMed]
- Sifaoui, I.N.; Chammem, M.; Mejri, M. Optimization of phenolic compounds extraction from olive leaves using experimental design methodology. J. Mater. Environ. Sci. 2016, 7, 1119–1127. [Google Scholar]
- Zhou, Y.; Gao, F.; Zhao, Y.; Lu, J. Study on the extraction kinetics of phenolic compounds from petroleum refinery waste lye. J. Saudi Chem. Soc. 2014, 18, 589–592. [Google Scholar] [CrossRef]
- Waszkowiak, K.; Gliszczyńska-Świgło, A. Binary ethanol–water solvents affect phenolic profile and antioxidant capacity of flaxseed extracts. Eur. Food Res. Technol. 2016, 242, 777–786. [Google Scholar] [CrossRef]
- Lim, T.Y.; Lim, Y.Y.; Yule, C.M. Evaluation of antioxidant, antibacterial and anti-tyrosinase activities of four Macaranga species. Food Chem. 2009, 114, 594–599. [Google Scholar] [CrossRef]
- Saha, S.; Verma, R.J. Antioxidant activity of polyphenolic extract of Terminalia chebula Retzius fruits. J. Taibah Univ. Sci. 2016, 10, 805–812. [Google Scholar] [CrossRef]
Sample Availability: All samples are available from the authors. |
| IC50 Value (mg/mL) | Extract Solvent | |
|---|---|---|
| Camellia pollen | 0.05 | ethanol |
| Thai mango seed kernel | 98,630 | ethanol |
| Blumea balsamifera DC leave | 0.319, 0.345 | Hexane, water |
| Euphorbia lathyris L. | 0.28 | methanol |
| Peel of Citrus Fruit | 2.42 | methanol |
| Far Eastern sea cucumber | 0.49–0.61, 1.80–1.99 | Ethanol, water |
| Sericin extract of silkworm | 1200–18,760 | water |
| X1 | X2 | X3 | Actual | Predicted | |||
|---|---|---|---|---|---|---|---|
| Y1 | Y2 | Y1 | Y2 | ||||
| Run# | Temp. (°C) | Time (min) | Ethanol Conc. (v/v%) | TPC (mg GAE/g DW) | TIA (%) | TPC (mg GAE/g DW) | TIA (%) |
| 1 | 77.5 | 40.0 | 61.3 | 5.07 | 75.2 | 5.01 | 74.6 |
| 2 | 65.0 | 50.0 | 40.0 | 2.37 | 46.2 | 2.21 | 47.8 |
| 3 | 77.5 | 40.0 | 61.3 | 4.92 | 73.3 | 5.01 | 74.6 |
| 4 | 65.0 | 30.0 | 40.0 | 2.86 | 19.7 | 2.61 | 17.7 |
| 5 | 77.5 | 40.0 | 61.3 | 5.04 | 74.7 | 5.01 | 74.6 |
| 6 | 65.0 | 50.0 | 82.5 | 2.74 | 49.6 | 2.41 | 53.6 |
| 7 | 77.5 | 40.0 | 97.0 | 3.54 | 56.9 | 3.48 | 52.1 |
| 8 | 90.0 | 50.0 | 82.5 | 5.09 | 61.2 | 5.20 | 71.6 |
| 9 | 77.5 | 40.0 | 25.5 | 1.87 | 17.1 | 2.03 | 19.0 |
| 10 | 65.0 | 30.0 | 82.5 | 2.73 | 22.1 | 2.82 | 23.4 |
| 11 | 77.5 | 23.2 | 61.3 | 3.76 | 13.3 | 3.62 | 15.7 |
| 12 | 90.0 | 30.0 | 40.0 | 2.33 | 22.2 | 2.50 | 26.7 |
| 13 | 90.0 | 50.0 | 40.0 | 3.76 | 43.6 | 3.69 | 38.0 |
| 14 | 98.5 | 40.0 | 61.3 | 4.55 | 76.3 | 4.30 | 75.5 |
| 15 | 56.5 | 40.0 | 61.3 | 1.68 | 54.7 | 2.04 | 52.7 |
| 16 | 77.5 | 56.8 | 61.3 | 4.04 | 55.7 | 4.28 | 50.6 |
| 17 | 90.0 | 30.0 | 82.5 | 3.84 | 66.2 | 4.02 | 60.2 |
| TPC | TIA | |||||
|---|---|---|---|---|---|---|
| Sum of Squares | F Value | p Level | Sum of Squares | F Value | p Level | |
| Model | 20.64 | 27.54 | 0.0001 | 7687.94 | 29.10 | <0.0001 |
| X1 | 6.15 | 73.86 | <0.0001 | 620.95 | 21.15 | 0.0025 |
| X2 | 0.5211 | 6.26 | 0.0409 | 1467.43 | 49.98 | 0.0002 |
| X3 | 2.53 | 30.43 | 0.0009 | 1320.85 | 44.99 | 0.0003 |
| X1X2 | 1.26 | 15.08 | 0.0060 | 177.02 | 6.03 | 0.0438 |
| X1X3 | 0.8563 | 10.29 | 0.0149 | 387.31 | 13.19 | 0.0084 |
| X2X3 | 0.0133 | 0.1602 | 0.7009 * | 79.91 | 2.72 | 0.1430 * |
| X12 | 4.79 | 57.48 | 0.0001 | 155.31 | 5.29 | 0.0550 |
| X22 | 1.59 | 19.05 | 0.0033 | 2428.74 | 82.73 | <0.0001 |
| X32 | 7.14 | 85.77 | <0.0001 | 2145.15 | 73.07 | <0.0001 |
| Temp. (°C) | Time (min) | Conc. (v/v%) | TPC (mg GAE/g DW) | TIA (%) | DPPH (IC50, μg/mL) | |
|---|---|---|---|---|---|---|
| Run #15 | 56.5 | 40.0 | 61.3 | 1.68 | 54.7 | 394.3 |
| Run #8 | 90.0 | 50.0 | 82.5 | 5.09 | 61.2 | 161.8 |
| Run #14 | 98.5 | 40.0 | 61.3 | 4.55 | 76.3 | 188.5 |
| Optimized condition | 88.1 | 43.7 | 72.0 | 5.41 | 82.4 | - |
| Validation experiment | 88.1 | 43.7 | 72.0 | 5.6 ± 0.2 | 79.6 ± 3.3 (4.4 ± 0.3 μg/mL) * | 91.8 ± 5.1 |
| Soxhlet extraction | 88.1 | 43.7 | 72.0 | 2.3 ± 0.2 | 53.6 ± 2.2 | 190.6 ± 8.2 |
| Ascorbic acid | - | - | - | - | 88.9 | 40.2 ± 1.8 |
| −1.68 | 1.0 | 0 | 1.0 | 1.68 | |
|---|---|---|---|---|---|
| Extraction Temperature (°C) | 56.5 | 65.0 | 77.5 | 90.0 | 98.5 |
| Extraction Time (min) | 23.2 | 30.0 | 40.0 | 50.0 | 56.8 |
| Ethanol Concentration (v/v%) | 25.5 | 40.0 | 61.3 | 82.5 | 97.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.J.; Kim, K.S.; Yu, B.J. Optimization of Antioxidant and Skin-Whitening Compounds Extraction Condition from Tenebrio molitor Larvae (Mealworm). Molecules 2018, 23, 2340. https://doi.org/10.3390/molecules23092340
Kim JJ, Kim KS, Yu BJ. Optimization of Antioxidant and Skin-Whitening Compounds Extraction Condition from Tenebrio molitor Larvae (Mealworm). Molecules. 2018; 23(9):2340. https://doi.org/10.3390/molecules23092340
Chicago/Turabian StyleKim, Julie J., Kyoung Seob Kim, and Byung Jo Yu. 2018. "Optimization of Antioxidant and Skin-Whitening Compounds Extraction Condition from Tenebrio molitor Larvae (Mealworm)" Molecules 23, no. 9: 2340. https://doi.org/10.3390/molecules23092340
APA StyleKim, J. J., Kim, K. S., & Yu, B. J. (2018). Optimization of Antioxidant and Skin-Whitening Compounds Extraction Condition from Tenebrio molitor Larvae (Mealworm). Molecules, 23(9), 2340. https://doi.org/10.3390/molecules23092340




