Validation of Internal Control Genes for Quantitative Real-Time PCR Gene Expression Analysis in Morchella
Abstract
:1. Introduction
2. Results
2.1. Primer Design and Verification
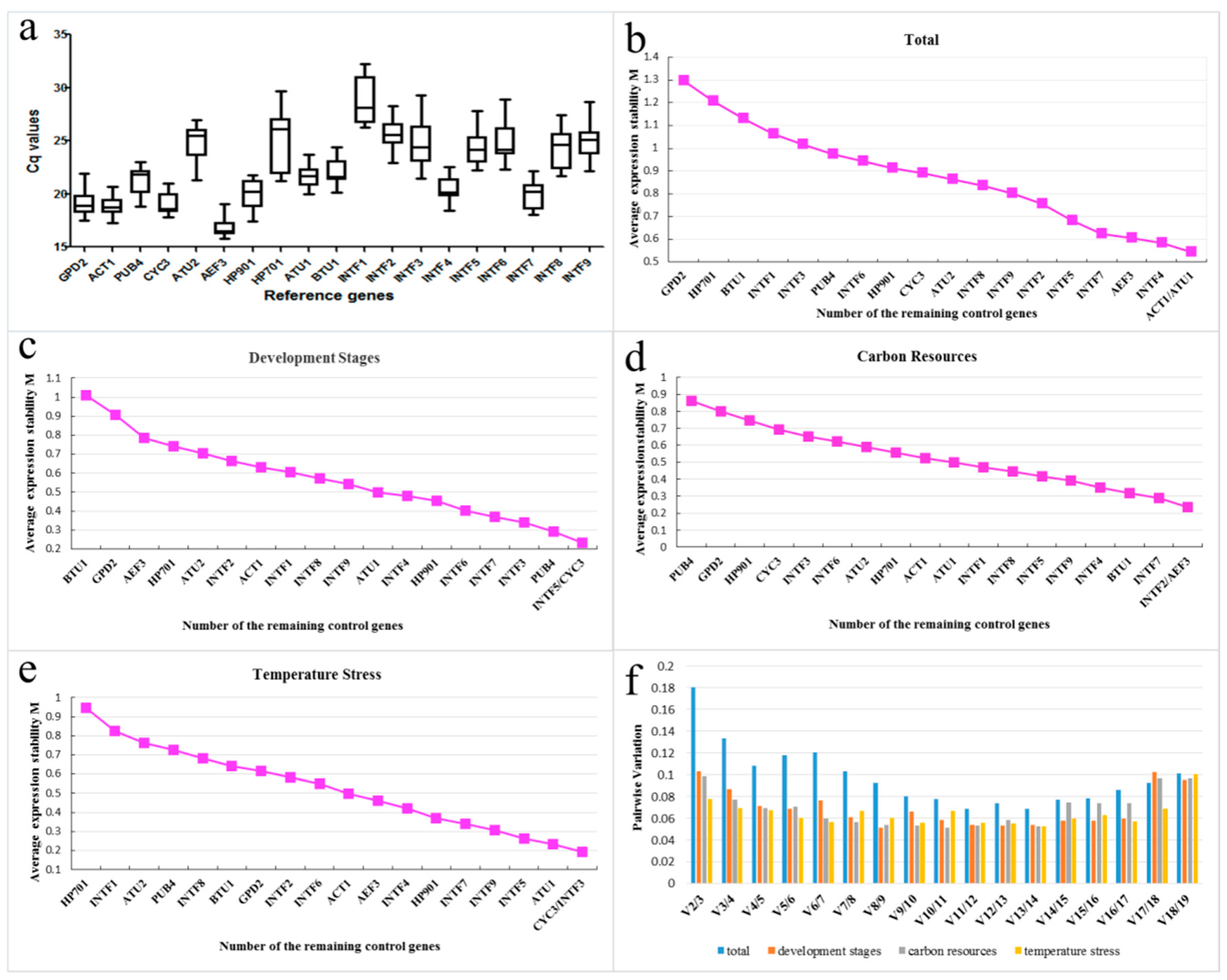
2.2. Verification and Expression Stability of Candidate Reference Genes
2.3. Validation of the Selected Reference Genes in Morchella
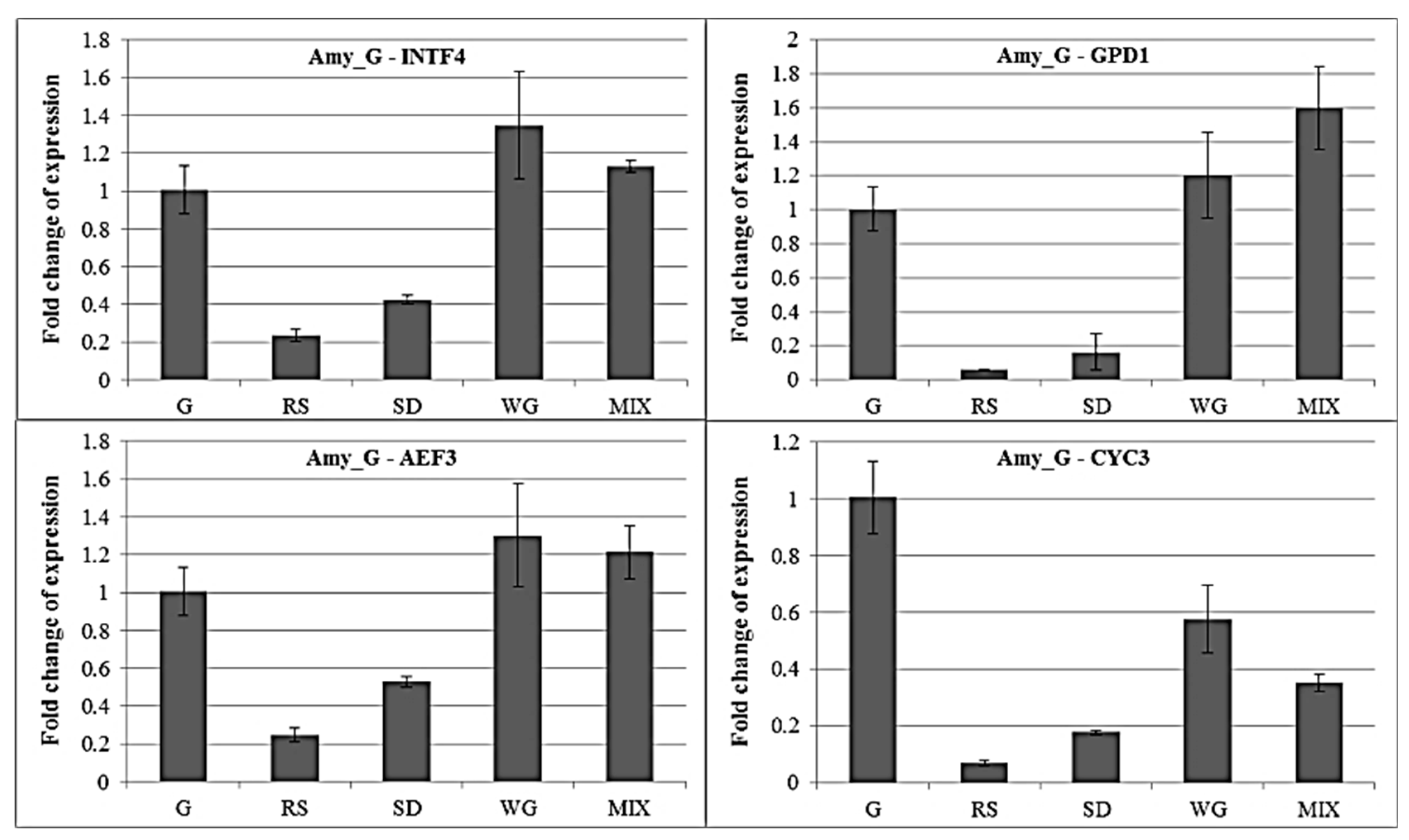
2.4. qRT-PCR of Amylase Gene under Different Carbon Sources with Different Internal Control Genes
3. Discussion
4. Materials and Methods
4.1. Materials and Experimental Treatments
4.2. Selection of Reference Genes and Primer Design
4.3. Total RNA Isolation and cDNA Synthesis
4.4. Quantitative Real-Time PCR Analysis
4.5. Validation of Reference Genes
Author Contributions
Funding
Conflicts of Interest
Ethical Approval
References
- Rosa, A.; Tuberoso, C.I.; Atzeri, A.; Melis, M.P.; Bifulco, E.; Dessi, M.A. Antioxidant profile of strawberry tree honey and its marker homogentisic acid in several models of oxidative stress. Food Chem. 2011, 129, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- He, P.X.; Geng, L.J.; Mao, D.B.; Xu, C.P. Production, characterization and antioxidant activity of exopolysaccharides from submerged culture of Morchella crassipes. Bioproc. Biosyst. Eng. 2012, 35, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Nitha, B.; Meera, C.R.; Janardhanan, K.K. Anti-inflammatory and antitumour activities of cultured mycelium of morel mushroom, Morchella esculenta. Curr. Sci. India 2007, 92, 235–239. [Google Scholar]
- Turkoglu, A.; Kivrak, I.; Mercan, N.; Duru, M.E.; Gezer, K.; Turkoglu, H. Antioxidant and antimicrobial activities of Morchella conica Pers. Afr. J. Biotechnol. 2006, 5, 1146–1150. [Google Scholar]
- Li, S.; Gao, A.; Dong, S.; Chen, Y.; Sun, S.; Lei, Z.; Zhang, Z. Purification, antitumor and immunomodulatory activity of polysaccharides from soybean residue fermented with Morchella esculenta. Int. J. Biol. Macromol. 2017, 96, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Heleno, S.A.; Stojković, D.; Barros, L.; Glamočlija, J.; Soković, M.; Martins, A.; Queiroz, M.; Ferreira, I.C. A comparative study of chemical composition, antioxidant and antimicrobial properties of Morchella esculenta (L.) Pers. from Portugal and Serbia. Food Res. Int. 2013, 51, 236–243. [Google Scholar] [CrossRef]
- Liu, C.; Sun, Y.H.; Mao, Q.; Guo, X.L.; Li, P.; Liu, Y.; Xu, N. Characteristics and Antitumor Activity of Morchella esculenta Polysaccharide Extracted by Pulsed Electric Field. Int. J. Mol. Sci. 2016, 17, 6. [Google Scholar] [CrossRef] [PubMed]
- Amir, R.; Levanon, D.; Hadar, Y.; Chet, I. Morphology and physiology of Morchella esculenta during sclerotial formation. Mycol. Res. 1993, 97, 683–689. [Google Scholar] [CrossRef]
- Bunyard, B.A.; Nicholson, M.S.; Royse, D.J. A Systematic Assessment of Morchella Using RFLP Analysis of the 28S ribosomal RNA gene. Mycologia 1994, 86, 762–772. [Google Scholar] [CrossRef]
- Ower, R.D.; Mills, G.L.; Malachowski, J.A. Cultivation of Morchella. U.S. Patent 4,866,878, 19 September 1986. [Google Scholar]
- Volk, T.J.; Leonard, T.J. Physiological and Environmental Studies of Sclerotium Formation and Maturation in Isolates of Morchella crassipes. Appl. Environ. Microbiol. 1989, 55, 3095–3100. [Google Scholar] [PubMed]
- Volk, T.J.; Leonard, T.J. Cytology of the life-cycle of Morchella. Mycol. Res. 1990, 94, 399–406. [Google Scholar] [CrossRef]
- Pilz, D.; Rebecca, M.L.; Susan, A.; Luis, V.R.; Shannon, B.; Tricia, W.; Parks, C.G.; Erika, M.F.; Blaze, B. Ecology and management of morels harvested from the forests of Western North America. Gen. Tech. Rep. 2007, 710. [Google Scholar]
- Wei, L.; Ya, Z.; Peixin, H. Morel Biology and Cultivation; Jilin Science and Technology Press: Jilin, China, 2017. [Google Scholar]
- Ower, R. Notes on the Development of the Morel Ascocarp: Morchella esculenta. Mycologia 1982, 74, 142–144. [Google Scholar] [CrossRef]
- Masaphy, S. External ultrastructure of fruit body initiation in Morchella. Mycol. Res. 2005, 109, 508–512. [Google Scholar] [CrossRef] [PubMed]
- Masaphy, S. Biotechnology of morel mushrooms: Successful fruiting body formation and development in a soilless system. Biotechnol. Lett. 2010, 32, 1523–1527. [Google Scholar] [CrossRef] [PubMed]
- He, P.X.; Cai, Y.L.; Liu, S.M.; Han, L.; Huang, L.N.; Liu, W. Morphological and ultrastructural examination of senescence in Morchella elata. Micron 2015, 78, 79–84. [Google Scholar] [CrossRef] [PubMed]
- He, P.-X.; Liu, W.; Cai, Y.-L.; He, X.S. Strain identification and phylogenetic analysis of cultivated and wild strains of Morchella belonging to Elata Clade in China. J. Zhengzhou Univ. Light Ind. (Nat. Sci.) 2015, 30, 26–29. [Google Scholar]
- He, P.; Wang, K.; Cai, Y.; Hu, X.; Zheng, Y.; Zhang, J.; Liu, W. Involvement of autophagy and apoptosis and lipid accumulation in sclerotial morphogenesis of Morchella importuna. Micron 2018, 109, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Yingli, C.; Ya, Z. The resolution of key technologies for rapid development of Morchella cultivation in China. Edible Med. Mushrooms 2018, 26, 142–147. [Google Scholar]
- Du, X.-H.; Zhao, Q.; Xia, E.-H.; Gao, L.-Z.; Richard, F.; Yang, Z.L. Mixed-reproductive strategies, competitive mating-type distribution and life cycle of fourteen black morel species. Sci. Rep. 2017, 7, 1493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, X.-H.; Zhao, Q.; Xu, J.; Yang, Z.L. High inbreeding, limited recombination and divergent evolutionary patterns between two sympatric morel species in China. Sci. Rep. 2016, 6, 22434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chai, H.; Chen, L.; Chen, W.; Zhao, Q.; Zhang, X.; Su, K.; Zhao, Y. Characterization of mating-type idiomorphs suggests that Morchella importuna, Mel-20 and M. sextelata are heterothallic. Mycol. Prog. 2017, 16, 743–752. [Google Scholar] [CrossRef]
- He, P.X.; Wang, K.; Cai, Y.L.; Liu, W. Live cell confocal laser imaging studies on the nuclear behavior during meiosis and ascosporogenesis in Morchella importuna under artificial cultivation. Micron 2017, 101, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Chen, L.F.; Cai, Y.L.; Zhang, Q.Q.; Bian, Y.B. Opposite polarity monospore genome de Novo sequencing and comparative analysis reveal the possible heterothallic life cycle of Morchella importuna. Int. J. Mol. Sci. 2018, 19, 2525. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.J.; Chai, H.M.; Chen, W.M.; Huang, X.Q.; Zhao, Y.C. Gene Expressing Difference in Sclerotial Formation of Morchella conica. Indian J. Microbiol. 2014, 54, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhu, X.; Gong, Y.; Xu, L.; Wang, Y.; Liu, L. Evaluation of reference genes for gene expression studies in radish (Raphanus sativus L.) using quantitative real-time PCR. Biochem. Biophy. Res. Commun. 2012, 424, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A. Absolute quantification of mRNA using real-time reverse transcription polymerase chain reaction assays. J. Mol. Endocrinol. 2000, 25, 169–193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, J.S.; Reed, A.; Feng, C.; Stewart, C.N. Statistical analysis of real-time PCR data. BMC Bioinform. 2006, 7, 85. [Google Scholar] [CrossRef] [PubMed]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative c(t) method. Nat. Protoc. 2008, 3, 1101. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Q.; Li, J.; Qin, P.; He, M.; Yu, X.; Zhao, K.; Zhang, X.; Ma, M.; Chen, Q.; Chen, X.; et al. Identification and evaluation of reference genes for qRT-PCR studies in Lentinula edodes. PLoS ONE 2018, 13, e0190226. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Lu, M.; Chen, Y.; Zhan, Z.; Cui, Q.; Wang, Y. Selection of reliable reference genes for gene expression studies using real-time PCR in tung tree during seed development. PLoS ONE 2012, 7, e43084. [Google Scholar] [CrossRef] [PubMed]
- Dekkers, B.J.; Willems, L.; Bassel, G.W.; van Bolderen-Veldkamp, R.P.; Ligterink, W.; Hilhorst, H.W.; Bentsink, L. Identification of reference genes for RT-qPCR expression analysis in Arabidopsis and tomato seeds. Plant Cell Physiol. 2012, 53, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Wan, H.; Yuan, W.; Ruan, M.; Ye, Q.; Wang, R.; Li, Z.; Zhou, G.; Yao, Z.; Zhao, J.; Liu, S. Identification of reference genes for reverse transcription quantitative real-time PCR normalization in pepper (Capsicum annuum L.). Biochem. Biophy. Res. Commun. 2011, 416, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Nolan, T. Pitfalls of quantitative real-time reverse-transcription polymerase chain reaction. J. Biomol. Tech. 2004, 15, 155–166. [Google Scholar] [PubMed]
- Schmittgen, T.D.; Zakrajsek, B.A. Effect of experimental treatment on housekeeping gene expression: Validation by real-time, quantitative RT-PCR. J. Biochem. Biophys. Methods 2000, 46, 69–81. [Google Scholar] [CrossRef]
- Thellin, O.; Zorzi, W.; Lakaye, B.; Borman, B.D.; Coumans, B.; Hennen, G.; Grisar, T.; Igout, A.; Heinen, E. Housekeeping genes as internal standards: Use and limits. J. Biotechnol. 1999, 75, 291–295. [Google Scholar] [CrossRef]
- Kim, H.-K.; Yun, S.-H. Evaluation of potential reference genes for quantitative RT-PCR analysis in Fusarium graminearum under different culture conditions. Plant. Pathol. J. 2011, 27, 301–309. [Google Scholar] [CrossRef]
- Lord, J.C.; Hartzer, K.; Toutges, M.; Oppert, B. Evaluation of quantitative PCR reference genes for gene expression studies in Tribolium castaneum after fungal challenge. J. Microbiol. Methods 2010, 80, 219–221. [Google Scholar] [CrossRef] [PubMed]
- Artico, S.; Nardeli, S.M.; Brilhante, O.; Grossi-De-Sa, M.F.; Alves-Ferreira, M. Identification and evaluation of new reference genes in Gossypium hirsutum for accurate normalization of real-time quantitative RT-PCR data. BMC Plant Biol. 2010, 10, 49. [Google Scholar] [CrossRef] [PubMed]
- Olsvik, P.A.; Lie, K.K.; Jordal, A.E.; Nilsen, T.O.; Hordvik, I. Evaluation of potential reference genes in real-time RT-PCR studies of Atlantic salmon. BMC Mol. Biol. 2005, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Vandesompele, J.; Preter, K.D.; Pattyn, F.; Poppe, B.; Roy, N.V.; Paepe, A.D.; Speleman, F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 3, 7. [Google Scholar] [CrossRef] [Green Version]
- Nicot, N.; Hausman, J.-F.; Hoffmann, L.; Evers, D. Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. J. Exp. Bot. 2005, 56, 2907–2914. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radonić, A.; Thulke, S.; Mackay, I.M.; Landt, O.; Siegert, W.; Nitsche, A. Guideline to reference gene selection for quantitative real-time PCR. Biochem. Biophy. Res. Commun. 2004, 313, 856. [Google Scholar] [CrossRef]
- Andersen, C.L.; Jensen, J.L.; Ørntoft, T.F. Normalization of real-time quantitative reverse transcription-PCR data: A model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res. 2004, 64, 5245. [Google Scholar] [CrossRef] [PubMed]
- Taskin, H.; Buyukalaca, S.; Hansen, K.; ODonnell, K. Multilocus phylogenetic analysis of true morels (Morchella) reveals high levels of endemics in Turkey relative to other regions of Europe. Mycologia 2012, 104, 446–461. [Google Scholar] [CrossRef] [PubMed]
- Kuo, M.; Dewsbury, D.R.; ODonnell, K.; Carter, M.C.; Rehner, S.A.; Moore, J.D.; Moncalvo, J.M.; Canfield, S.A.; Stephenson, S.L.; Methven, A.S.; et al. Taxonomic revision of true morels (Morchella) in Canada and the United States. Mycologia 2012, 104, 1159–1177. [Google Scholar] [CrossRef] [PubMed]
- Du, X.H.; Zhao, Q.; ODonnell, K.; Rooney, A.P.; Yang, Z.L. Multigene molecular phylogenetics reveals true morels (Morchella) are especially species-rich in China. Fungal Genet. Biol. 2012, 49, 455–469. [Google Scholar] [CrossRef] [PubMed]
- Svingen, T.; Jørgensen, A.; Rajpert-De Meyts, E. Validation of endogenous normalizing genes for expression analyses in adult human testis and germ cell neoplasms. Mol. Hum. Reprod. 2014, 20, 709–718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dheda, K.; Huggett, J.F.; Chang, J.S.; Kim, L.U.; Bustin, S.A.; Johnson, M.A.; Rook, G.A.W.; Zumla, A. The implications of using an inappropriate reference gene for real-time reverse transcription PCR data normalization. Anal. Biochem. 2005, 344, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Hellemans, J.; Mortier, G.; De, P.A.; Speleman, F.; Vandesompele, J. qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol. 2007, 8, R19. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Higgins, P.J.; Crawford, D.R. Control selection for RNA quantitation. Biotechniques 2000, 29, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Martins, P.K.; Mafra, V.; de Souza, W.R.; Ribeiro, A.P.; Vinecky, F.; Basso, M.F.; da Cunha, B.A.; Kobayashi, A.K.; Molinari, H.B. Selection of reliable reference genes for RT-qPCR analysis during developmental stages and abiotic stress in Setaria viridis. Sci. Rep. 2016, 6, 28348. [Google Scholar] [CrossRef] [PubMed]
- Masilamani, T.J.; Loiselle, J.J.; Sutherland, L.C. Assessment of reference genes for real-time quantitative PCR gene expression normalization during C2C12 and H9c2 skeletal muscle differentiation. Mol. Biotechnol. 2014, 56, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.; Dobersch, S.; Dammann, R.H.; Mehta, A.; Dobersch, S.; Dammann, R.H.; Bellusci, S.; Ilinskaya, O.N.; Braun, T. Validation of Tuba1a as appropriate internal control for normalization of gene expression analysis during mouse lung development. Int. J. Mol. Sci. 2015, 16, 4492–4511. [Google Scholar] [CrossRef] [PubMed]
- Han, P.-P.; Qin, L.; Li, Y.-S.; Liao, X.-S.; Xu, Z.-X.; Hu, X.-J.; Xie, L.-H.; Yu, C.-B.; Wu, Y.-F.; Liao, X. Identification of suitable reference genes in leaves and roots of rapeseed (Brassica napus L.) under different nutrient deficiencies. J. Integr. Agric. 2017, 16, 809–819. [Google Scholar] [CrossRef]
- Xu, J.; Xu, Z.C.; Zhu, Y.J.; Luo, H.M.; Qian, J.; Ji, A.J.; Hu, Y.L.; Sun, W.; Wang, B.; Song, J.Y. Identification and evaluation of reference genes for qRT-PCR normalization in Ganoderma lucidum. Curr. Microbiol. 2014, 68, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, K.D.C.; Melo, D.F.D.; Morais, V.D.; Vasconcelos, I.M.; Costa, J.H. Selection of suitable soybean EF1α genes as internal controls for real-time PCR analyses of tissues during plant development and under stress conditions. Plant Cell Rep. 2014, 33, 1453. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, G.W.; Delaney, S.K. Stable internal reference genes for normalization of real-time RT-PCR in tobacco (Nicotiana tabacum) during development and abiotic stress. Mol. Genet. Genom. 2010, 283, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Nillegoda, N.B.; Bukau, B. Metazoan Hsp70-based protein disaggregases: Emergence and mechanisms. Front. Mol. Biosci. 2015, 2, 57. [Google Scholar] [CrossRef] [PubMed]
- Balakirev, M.Y.; Mullally, J.E.; Favier, A.; Assard, N.; Sulpice, E.; Lindsey, D.F.; Rulina, A.V.; Gidrol, X.; Wilkinson, K.D. Wss1 metalloprotease partners with Cdc48/Doa1 in processing genotoxic SUMO conjugates. eLife 2015, 4, e06763. [Google Scholar] [CrossRef] [PubMed]
- Aghazadeh, Y.; Papadopoulos, V. The role of the 14-3-3 protein family in health, disease, and drug development. Drug Discov. Today 2016, 21, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Parente-Rocha, J.A.; Parente, A.F.A.; Baeza, L.C.; Bonfim, S.M.R.C.; Hernandez, O.; McEwen, J.G.; Bailão, A.M.; Taborda, C.P.; Borges, C.L.; Soares, C.M.D.A. Macrophage Interaction with Paracoccidioides brasiliensis Yeast Cells Modulates Fungal Metabolism and Generates a Response to Oxidative Stress. PLoS ONE 2015, 10, e0137619. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Shu, S.; Zhu, W.; Xiong, Y.; Peng, F. The Kinome of Edible and Medicinal Fungus Wolfiporia cocos. Front. Microbiol. 2016, 7, 1495. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Cai, F.; Zhang, S.; Zhang, S.; Song, W. Overexpression of ubiquitin carboxyl-terminal hydrolase L1 (UCHL1) delays Alzheimer’s progression in vivo. Sci. Rep. 2014, 4, 7298. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.; Hamilton, G.; Wilkes, J.M.; Hutchinson, S.; Barrett, M.P.; Horn, D. Vacuolar ATPase depletion affects mitochondrial ATPase function, kinetoplast dependency, and drug sensitivity in trypanosomes. Proc. Nat. Acad. Sci. USA 2015, 112, 9112. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Wang, Y.; Liu, F.; Zhou, K.; Ding, Y.; Zhou, F.; Wang, Y.; Liu, K.; Gan, L.; Ma, W.; et al. Encodes a Regulator of Post-Golgi Vesicular Traffic Essential for Vacuolar Protein Sorting in Rice Endosperm. Plant Cell 2014, 26, 410–425. [Google Scholar] [CrossRef] [PubMed]
- Bray, N.L.; Pimentel, H.; Melsted, P.; Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 2016, 34, 525. [Google Scholar] [CrossRef] [PubMed]
- NCBI Resource Coordinators. Database Resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2017, 45, D12–D17. [Google Scholar] [CrossRef] [PubMed]
- Walker, N.J. A technique whose time has come. Science 2002, 296, 557–559. [Google Scholar] [CrossRef] [PubMed]
| Primer Name | Gene Number a | Gene Description b | p-Value c | Primer Sequence | Amplicon Size (bp) | Tm (°C) | E (%) | R2 | TMP Average d | TMP CV e |
|---|---|---|---|---|---|---|---|---|---|---|
| GPD2F | TRINITY_DN12699_c0_g1 | Glyceraldehyde-3-phosphate dehydrogenase | 3.00 × 10−167 | GGTACGACAACGAATGGGGT | 197 | 60.04 | 93.8 | 0.997 | 405.63 | 0.2815 |
| GPD2R | CCGCAAAAATCTCTTCCCGC | 60.18 | ||||||||
| ACT1F | TRINITY_DN11687_c0_g1 | Actin, gamma | 0 | GTACCCTGGTATTGCCGACC | 197 | 60.18 | 98.8 | 0.999 | 746.06 | 0.1969 |
| ACT1R | GGACGATGGAAGGACCACTC | 59.82 | ||||||||
| PUB4F | TRINITY_DN12852_c0_g2 | Polyubiquitin | 9.00 × 10−158 | GTCCACCCTTCACTTGGTTCT | 163 | 59.86 | 105.8 | 0.998 | 1232.22 | 0.546 |
| PUB4R | ACGTTGTTGATCTGGGGGAAT | 59.65 | ||||||||
| CYC3F | TRINITY_DN9811_c0_g1 | Peptidyl-prolyl cis-trans isomerase | 3.00 × 10−86 | TCCTCCATCCTCCGAACACA | 208 | 60.25 | 117.4 | 0.997 | 3505.12 | 0.3792 |
| CYC3R | TCTCAGCGGTTCTAGGGACA | 59.96 | ||||||||
| ATU2F | TRINITY_DN12747_c0_g4 | Alpha-tubulin 2 | 0 | TTCGACGGGCATCTGAATGT | 209 | 59.75 | 93.3 | 0.995 | 17.69 | 0.4123 |
| ATU2R | ATGTATTTCCCCTTCGCGGG | 60.18 | ||||||||
| AEF3F | TRINITY_DN10564_c0_g2 | Elongation factor 1-alpha | 0 | CTGTCATTGATGCCCCTGGT | 169 | 60.03 | 106.7 | 0.992 | 6267.16 | 0.4861 |
| AEF3R | CCAAGGGTGTAGGCGAGAAG | 60.11 | ||||||||
| HP901F | TRINITY_DN10080_c0_g1 | HSP90-domain-containing protein | 0 | GAGAAGGTTGTCGTCTCGCA | 182 | 60.04 | 97.8 | 0.996 | 418.67 | 0.1946 |
| HP901R | TTGACGATGGGGGACTTTGG | 59.96 | ||||||||
| HP701F | TRINITY_DN9777_c0_g1 | Hsp70 protein-like protein | 1.00 × 10−119 | TTTGGTGCGTCAGCGTATCT | 206 | 60.04 | 99.7 | 0.995 | 194.33 | 0.3632 |
| HP701R | GCGGTGCCATTGTGTTTCAT | 60.04 | ||||||||
| ATU1F | TRINITY_DN5830_c0_g1 | Alpha-tubulin | 0 | TTTCGCTGTGGTGTGGGTAG | 169 | 60.25 | 91 | 0.998 | 422.43 | 0.335 |
| ATU1R | ATGGAAAACGGAAATGGGAACC | 59.43 | ||||||||
| BTU1F | TRINITY_DN10333_c0_g2 | Beta-tubulin | 0 | CAGTCGTTCCTTCGCCCAAA | 229 | 60.88 | 97 | 0.997 | 368.32 | 0.2409 |
| BTU1R | CCAGGGAAACGAAGGGATGT | 59.67 | ||||||||
| INTF1F | TRINITY_DN10662_c0_g1 | Similar to DNA damage response protein wss1 | 5.00 × 10−104 | CCGCCAGTCCTAACTCACTC | 179 | 59.83 | 104.5 | 0.987 | 10.35 | 0.0313 |
| INTF1R | CGATAGCTTTGTCGGGTCCA | 59.83 | ||||||||
| INTF2F | TRINITY_DN13390_c6_g2 | Vesicle transport protein, Got1/SFT2-like | 1.00 × 10−66 | AGATTCCGTAAAAGCGGGGG | 238 | 60.11 | 104.7 | 0.994 | 35.04 | 0.0355 |
| INTF2R | GCGCATTAACCGAGTGCAAA | 60.11 | ||||||||
| INTF3F | TRINITY_DN9447_c0_g1 | Vacuolar protein sorting/targeting protein 10 | 0 | CTGTTGATGGGTTGGCTGGT | 229 | 60.54 | 94.4 | 0.995 | 25.38 | 0.0528 |
| INTF3R | GGGTTCATAGGCTTCGTCGT | 59.82 | ||||||||
| INTF4F | TRINITY_DN8632_c0_g1 | Vacuolar ATP synthase 16 kDa proteolipid subunit | 2.00 × 10−57 | CTGCTTTGGTGCCAGTTACG | 217 | 59.76 | 107.9 | 0.999 | 869.67 | 0.0528 |
| INTF4R | GGAAAGACCAGCACCGAGTT | 60.25 | ||||||||
| INTF5F | TRINITY_DN12928_c0_g1 | Similar to Ubiquitin carboxyl-terminal hydrolase 4 | 0 | TCAGTGTCCCACCCGTATCT | 211 | 59.96 | 103.3 | 0.999 | 16.53 | 0.0544 |
| INTF5R | CCGCGTGGAGTTATTGGAGT | 60.11 | ||||||||
| INTF6F | TRINITY_DN12591_c0_g2 | Similar to GTPase-activating protein BEM2/IPL2 | 0 | AGAATGGGTTGACGCTGGTT | 173 | 59.89 | 101.3 | 0.998 | 12.15 | 0.0671 |
| INTF6R | CAGTTGAGGAGGGTTCGCAT | 60.04 | ||||||||
| INTF7F | TRINITY_DN13203_c1_g1 | 14-3-3 protein | 3.00 × 10−148 | GCACGTTGGAAAGATCCGTG | 170 | 59.83 | 99.2 | 0.995 | 766.99 | 0.0725 |
| INTF7R | CGGCGAGATAACGGTGGTAG | 60.32 | ||||||||
| INTF8F | TRINITY_DN7177_c0_g1 | Hypothetical protein | / | GATGCCCGATCCGATGCTC | 274 | 60.74 | 92 | 0.989 | 37.43 | 0.0713 |
| INTF8R | TGGGCGAGGGTATTTTGGAG | 59.75 | ||||||||
| INTF9F | TRINITY_DN13335_c6_g2 | Select seq ESZ90392.1 splicing factor 3b | 0 | ACGACACCGACTACGAATCC | 239 | 59.55 | 92.2 | 0.987 | 25.29 | 0.0733 |
| INTF9R | CGCCTCCAGATACGCACTAA | 59.62 |
| Ranking | Total | Development Stages | Carbon Resources | Temperature Stress | ||||
|---|---|---|---|---|---|---|---|---|
| geNorm | Stability Value | geNorm | Stability Value | geNorm | Stability Value | geNorm | Stability Value | |
| 1 | ACT1 | 0.54 | INTF5 | 0.23 | INTF2 | 0.23 | CYC3 | 0.19 |
| 2 | ATU1 | 0.54 | CYC3 | 0.23 | AEF3 | 0.23 | INTF3 | 0.19 |
| 3 | INTF4 | 0.58 | PUB4 | 0.29 | INTF7 | 0.29 | ATU1 | 0.23 |
| 4 | AEF3 | 0.61 | INTF3 | 0.34 | BTU1 | 0.32 | INTF5 | 0.26 |
| 5 | INTF7 | 0.63 | INTF7 | 0.37 | INTF4 | 0.35 | INTF9 | 0.31 |
| 6 | INTF5 | 0.68 | INTF6 | 0.4 | INTF9 | 0.39 | INTF7 | 0.34 |
| 7 | INTF2 | 0.76 | HP901 | 0.45 | INTF5 | 0.42 | HP901 | 0.37 |
| 8 | INTF9 | 0.8 | INTF4 | 0.48 | INTF8 | 0.44 | INTF4 | 0.42 |
| 9 | INTF8 | 0.84 | ATU1 | 0.5 | INTF1 | 0.47 | AEF3 | 0.46 |
| 10 | ATU2 | 0.86 | INTF9 | 0.54 | ATU1 | 0.5 | ACT1 | 0.5 |
| 11 | CYC3 | 0.89 | INTF8 | 0.57 | ACT1 | 0.52 | INTF6 | 0.55 |
| 12 | HP901 | 0.91 | INTF1 | 0.6 | HP701 | 0.56 | INTF2 | 0.58 |
| 13 | INTF6 | 0.94 | ACT1 | 0.63 | INTF6 | 0.6 | GPD2 | 0.61 |
| 14 | PUB4 | 0.97 | INTF2 | 0.66 | INTF3 | 0.63 | BTU1 | 0.64 |
| 15 | INTF3 | 1.02 | ATU2 | 0.7 | HP901 | 0.7 | INTF8 | 0.68 |
| 16 | INTF1 | 1.06 | HP701 | 0.74 | GPD2 | 0.77 | PUB4 | 0.73 |
| 17 | BTU1 | 1.13 | AEF3 | 0.78 | PUB4 | 0.84 | ATU2 | 0.76 |
| 18 | HP701 | 1.21 | GPD2 | 0.91 | CYC3 | 0.95 | INTF1 | 0.82 |
| 19 | GPD2 | 1.29 | BTU1 | 1.01 | ATU2 | 1.06 | HP701 | 0.94 |
| Best pair | ACT1/ATU1 | INTF5/CYC3 | INTF2/AEF3 | CYC3/INTF3 | ||||
| Ranking | Total | Development Stages | Carbon Resources | Temperature Stress | ||||
|---|---|---|---|---|---|---|---|---|
| Norm Finder | Stability Value | Norm Finder | Stability Value | Norm Finder | Stability Value | Norm Finder | Stability Value | |
| 1 | INTF7 | 0.174 | INTF5 | 0.105 | INTF4 | 0.123 | ATU1 | 0.058 |
| 2 | INTF4 | 0.254 | ATU1 | 0.178 | AEF3 | 0.157 | INTF3 | 0.058 |
| 3 | INTF5 | 0.287 | CYC3 | 0.205 | INTF7 | 0.207 | INTF5 | 0.064 |
| 4 | AEF3 | 0.439 | INTF7 | 0.208 | INTF2 | 0.211 | CYC3 | 0.093 |
| 5 | ATU1 | 0.439 | INTF6 | 0.222 | ACT1 | 0.247 | INTF9 | 0.185 |
| 6 | ACT1 | 0.472 | HP901 | 0.292 | ATU1 | 0.257 | INTF7 | 0.226 |
| 7 | INTF9 | 0.472 | PUB4 | 0.32 | BTU1 | 0.26 | HP901 | 0.299 |
| 8 | INTF2 | 0.511 | INTF3 | 0.331 | INTF8 | 0.347 | INTF4 | 0.313 |
| 9 | HP901 | 0.514 | INTF4 | 0.335 | INTF9 | 0.381 | AEF3 | 0.35 |
| 10 | INTF8 | 0.523 | INTF1 | 0.357 | HP701 | 0.426 | ACT1 | 0.42 |
| 11 | ATU2 | 0.562 | ACT1 | 0.378 | INTF1 | 0.448 | INTF8 | 0.485 |
| 12 | INTF6 | 0.615 | INTF9 | 0.475 | INTF5 | 0.449 | INTF2 | 0.521 |
| 13 | CYC3 | 0.632 | INTF8 | 0.524 | INTF6 | 0.631 | INTF6 | 0.536 |
| 14 | PUB4 | 0.672 | AEF3 | 0.57 | HP901 | 0.64 | GPD2 | 0.591 |
| 15 | INTF3 | 0.787 | INTF2 | 0.614 | INTF3 | 0.671 | PUB4 | 0.603 |
| 16 | INTF1 | 0.861 | ATU2 | 0.626 | GPD2 | 0.726 | BTU1 | 0.622 |
| 17 | BTU1 | 0.987 | HP701 | 0.776 | PUB4 | 0.85 | ATU2 | 0.659 |
| 18 | HP701 | 1.196 | GPD2 | 1.208 | CYC3 | 1.129 | INTF1 | 0.864 |
| 19 | GPD2 | 1.325 | BTU1 | 1.248 | ATU2 | 1.264 | HP701 | 1.317 |
| Best pair | INTF7 | INTF5 | INTF4 | ATU1 | ||||
| Test Number | Strains Number | Species Name | Phylogenetics Number | Clade |
|---|---|---|---|---|
| 1 | Ma_2 | Morchella importuna | Mel-10 | Elata |
| 2 | 16-9 | Morchella sextelata | Mel-6 | Elata |
| 3 | 16-41 | Morchella sp. Mel-21 | Mel-21 | Elata |
| 4 | 16-57 | Morchella spongiola | Mel-21/33 | Elata |
| 5 | 16-50 | Morchella costata | Mel-13/26 | Elata |
| 6 | 16-72 | Morchella sp. Mes-23/24 | Mes-23/24 | Esculenta |
| 7 | 16-103 | Morchella Mes-6 | Mes-6 | Esculenta |
| 8 | Win8 | Morchella sp. Mes-21 | Mes-21 | Esculenta |
| 9 | shanyang | Morchella Mes-19 | Mes-19 | Esculenta |
| 10 | M10 | Morchella sp. | Mes-24 | Esculenta |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Q.; Liu, W.; Cai, Y.; Lan, A.-F.; Bian, Y. Validation of Internal Control Genes for Quantitative Real-Time PCR Gene Expression Analysis in Morchella. Molecules 2018, 23, 2331. https://doi.org/10.3390/molecules23092331
Zhang Q, Liu W, Cai Y, Lan A-F, Bian Y. Validation of Internal Control Genes for Quantitative Real-Time PCR Gene Expression Analysis in Morchella. Molecules. 2018; 23(9):2331. https://doi.org/10.3390/molecules23092331
Chicago/Turabian StyleZhang, Qianqian, Wei Liu, Yingli Cai, A-Feng Lan, and Yinbing Bian. 2018. "Validation of Internal Control Genes for Quantitative Real-Time PCR Gene Expression Analysis in Morchella" Molecules 23, no. 9: 2331. https://doi.org/10.3390/molecules23092331
APA StyleZhang, Q., Liu, W., Cai, Y., Lan, A.-F., & Bian, Y. (2018). Validation of Internal Control Genes for Quantitative Real-Time PCR Gene Expression Analysis in Morchella. Molecules, 23(9), 2331. https://doi.org/10.3390/molecules23092331






