Equilibrium and Kinetic Study of Lead and Copper Ion Adsorption on Chitosan-Grafted-Polyacrylic Acid Synthesized by Surface Initiated Atomic Transfer Polymerization
Abstract
:1. Introduction
2. Results and Discussion
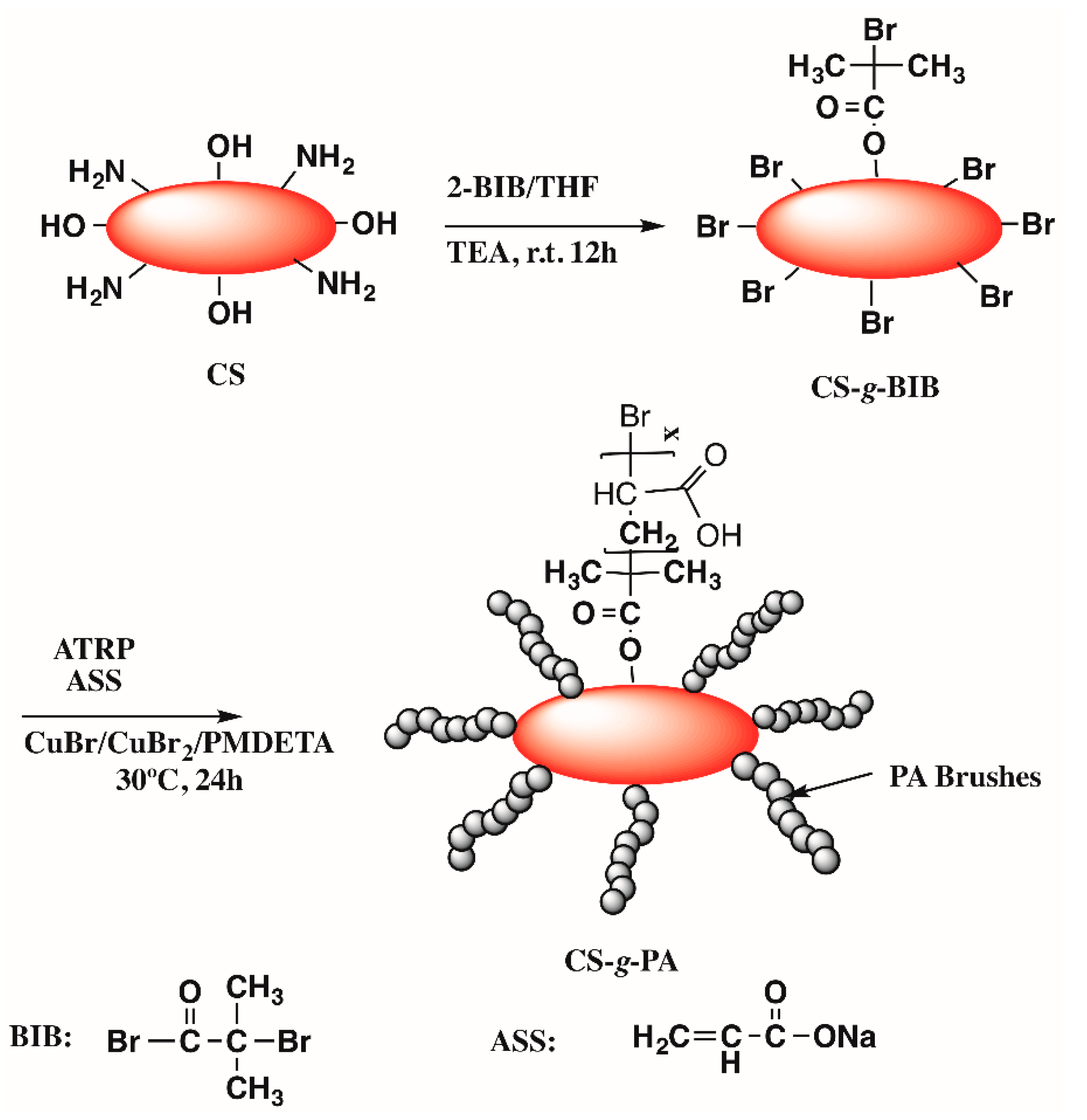
2.1. Synthesis
2.2. Chitosan Characterization
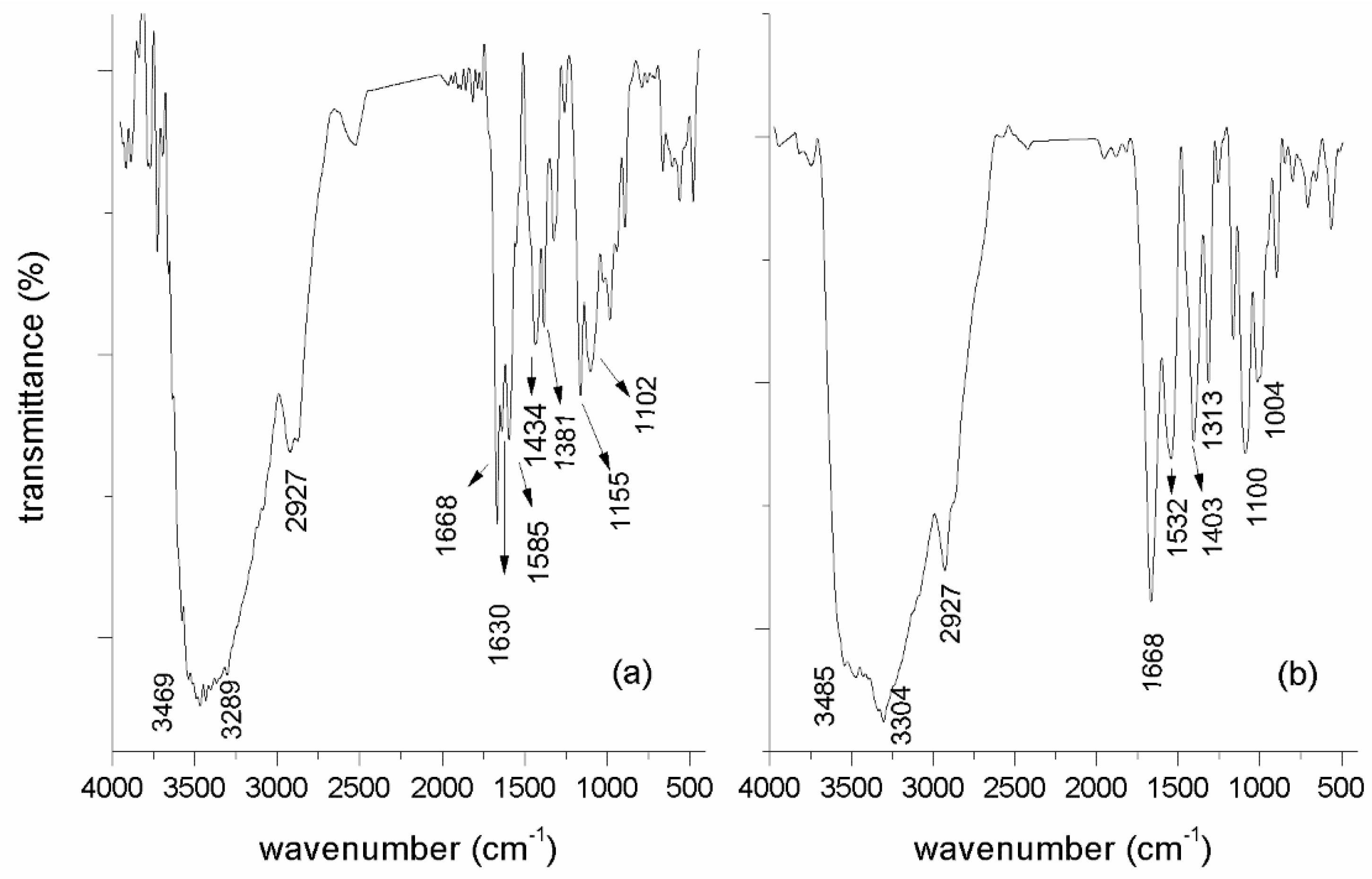
2.2.1. FT-IR Characterization
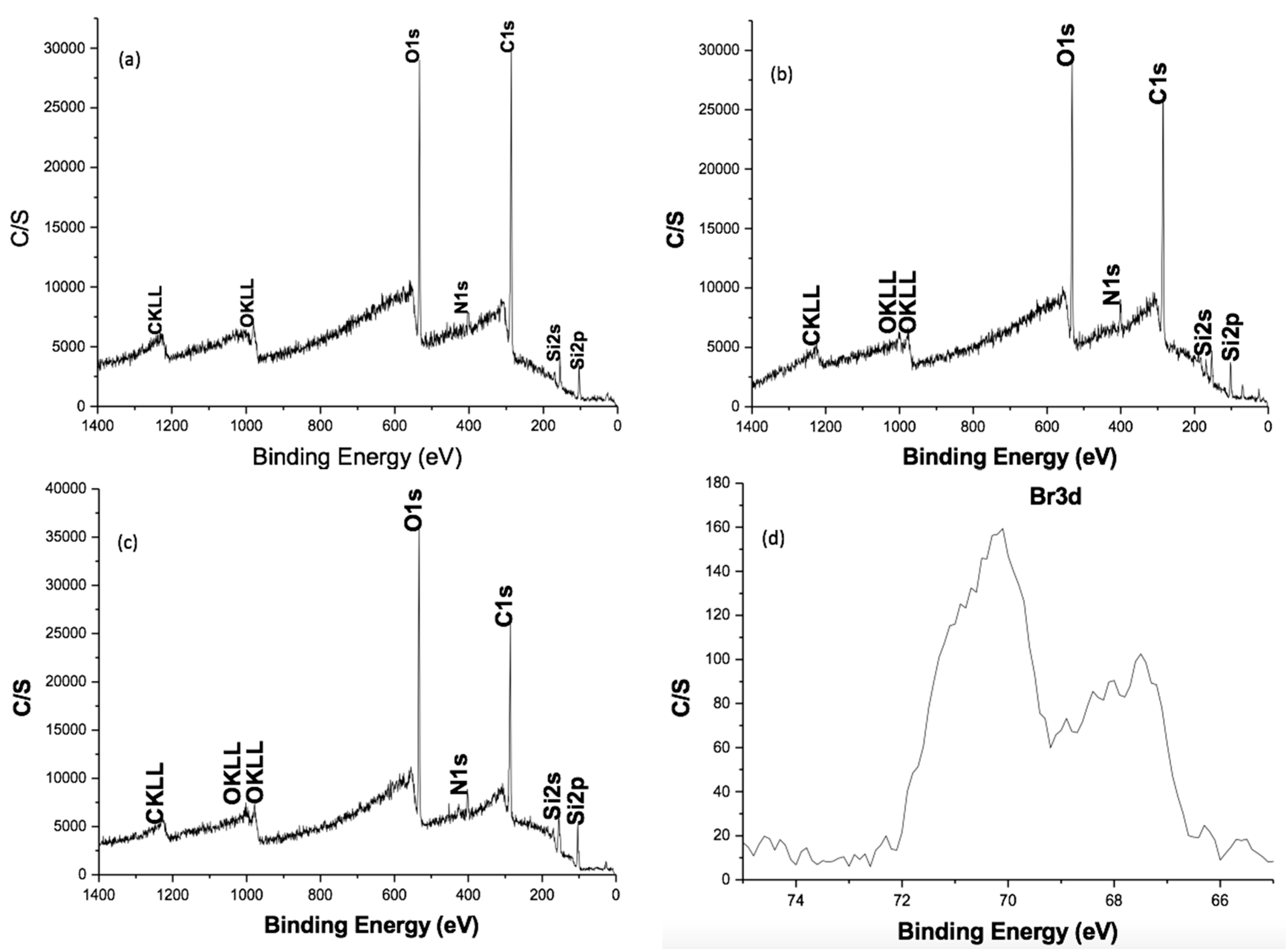
2.2.2. XPS Characterization
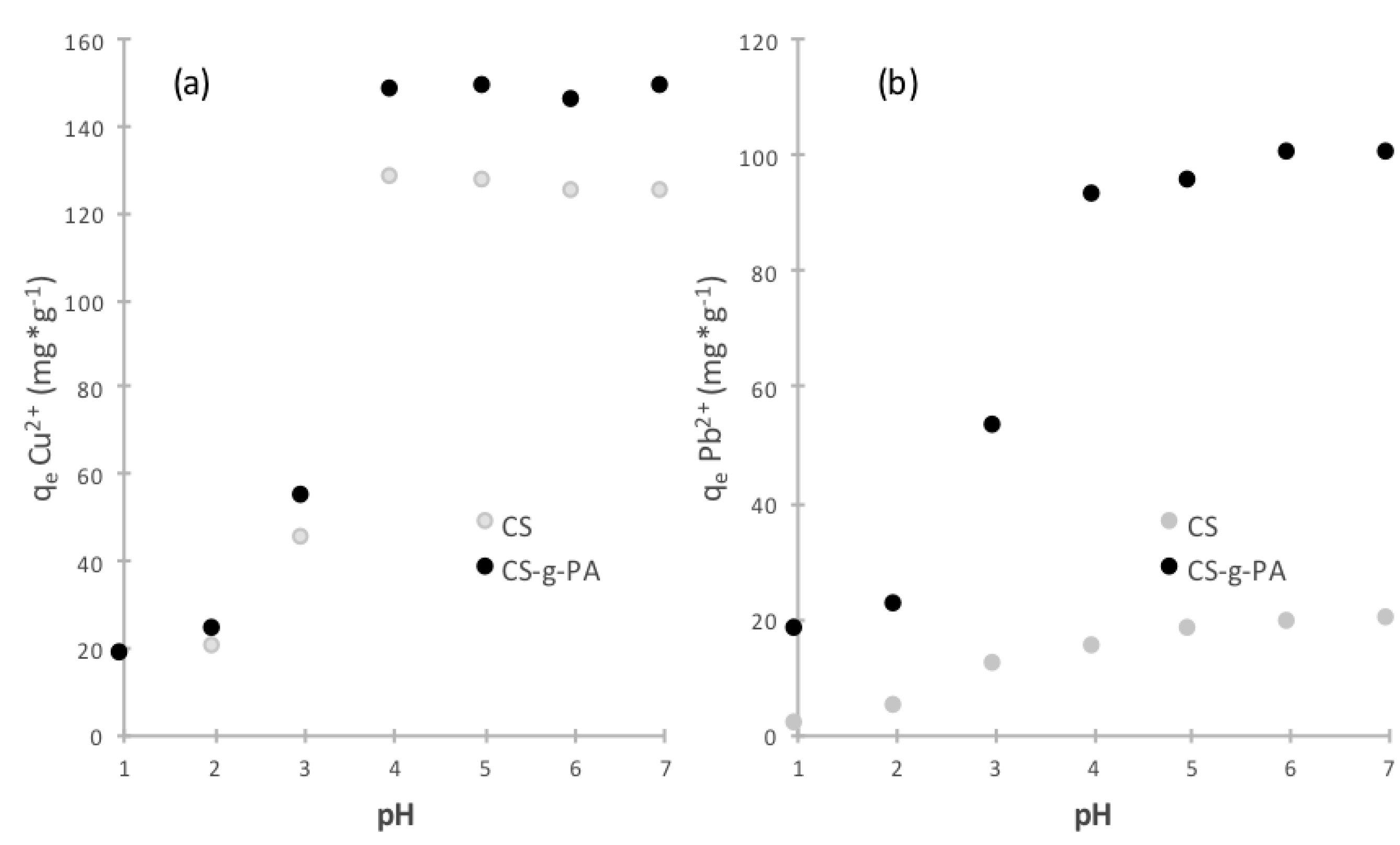
2.3. Initial pH Effect on Adsorption Process
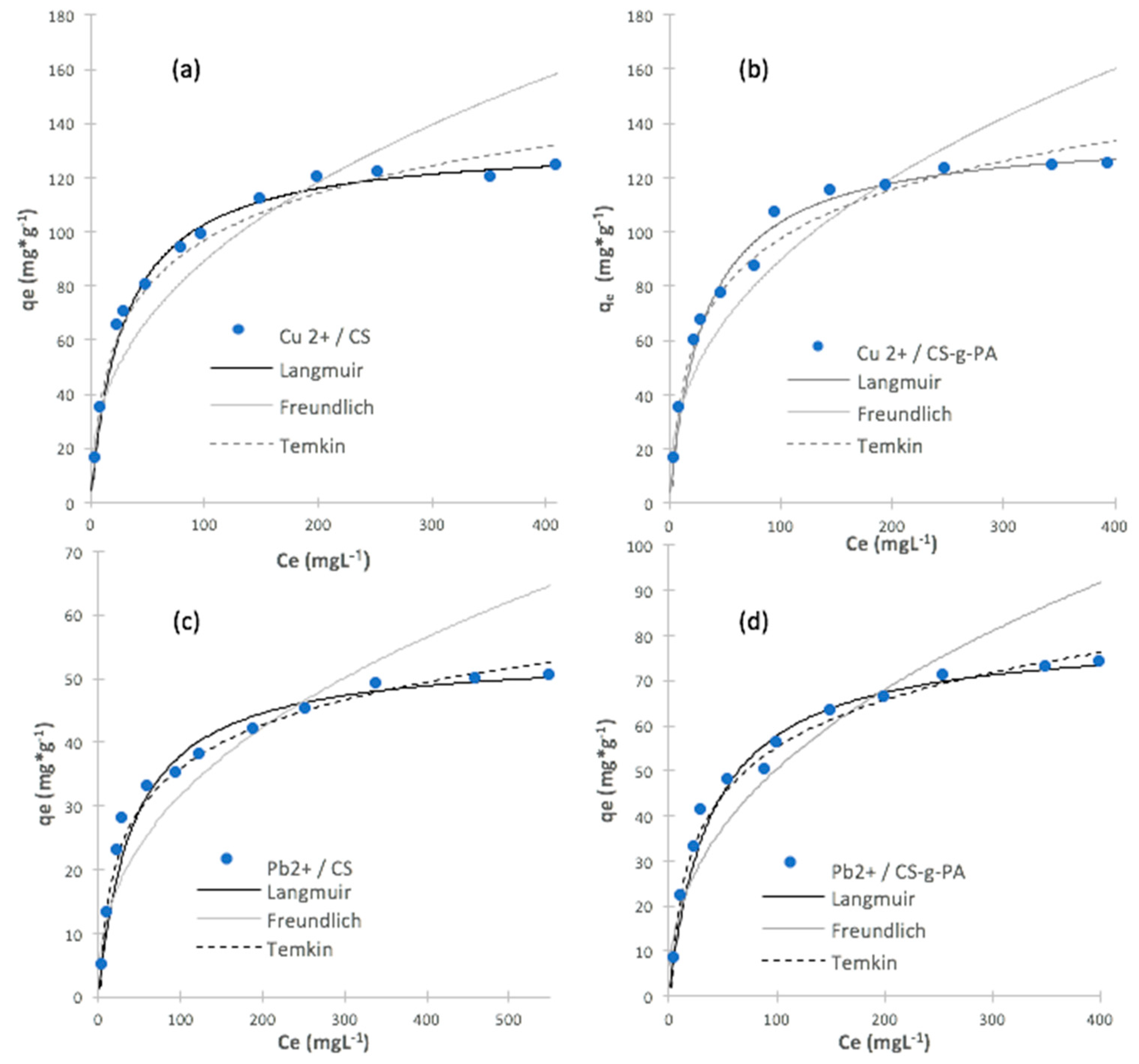
2.4. Adsorption Isotherms
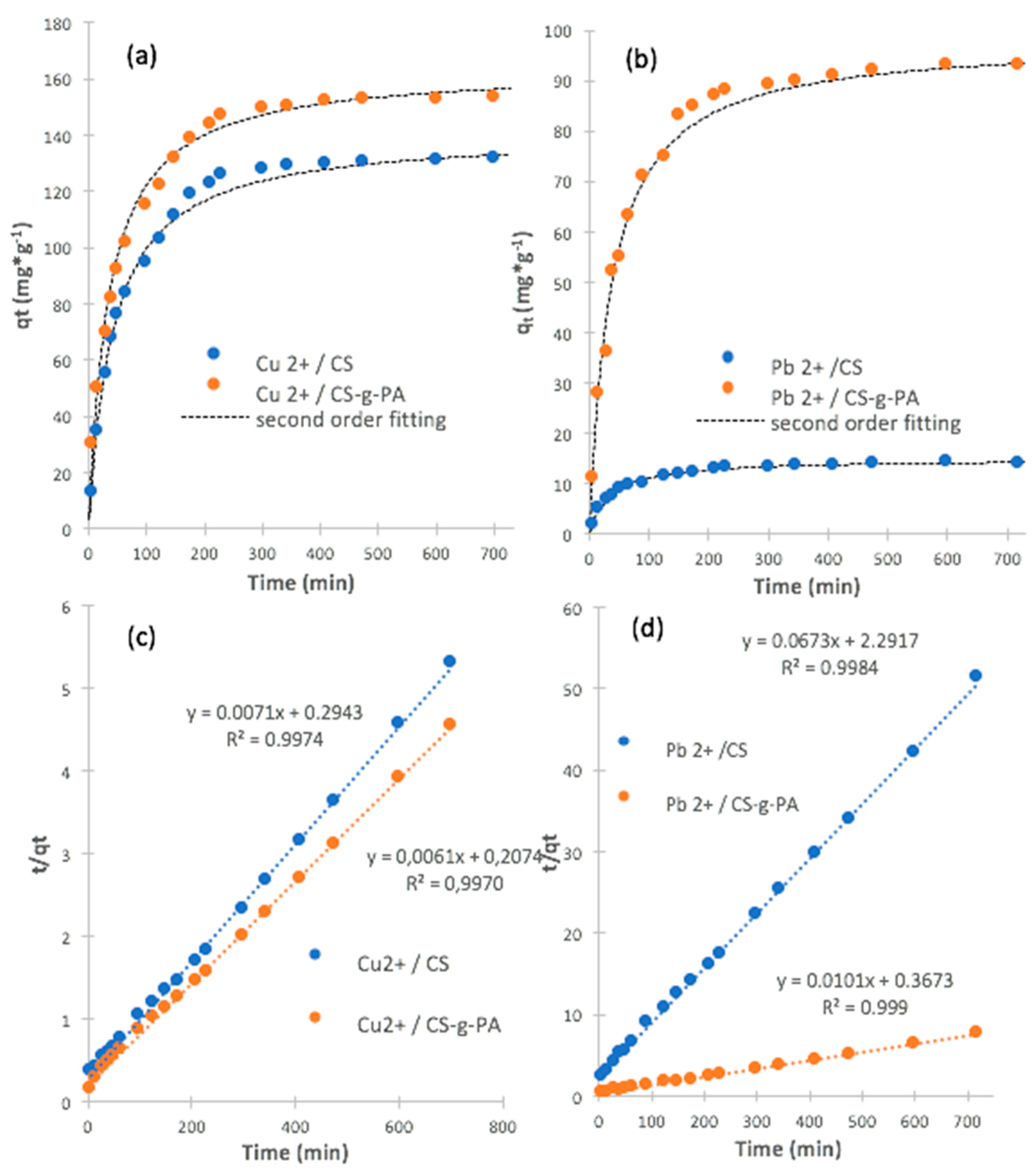
2.5. Adsorption Kinetics for Copper and Lead Ions
3. Experimental
3.1. Materials
3.2. Synthesis
3.3. Characterization
3.4. Adsorption Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Lee, C.-G.; Jeon, J.-W.; Hwang, M.-J.; Ahn, K.-H.; Park, C.; Choi, J.-W.; Lee, S.-H. Lead and copper removal from aqueous solutions using carbon foam derived from phenol resin. Chemosphere 2015, 130, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Environmental Protection Agency (EPA). Use of Lead Free Pipes, Fittings, Fixtures, Solder and Flux for Drinking Water; Extension of Comment Period; Environmental Protection Agency: Washington, WA, USA, 2017. [Google Scholar]
- Saleh, T.A.; Gupta, V.K. Column with CNT/magnesium oxide composite for lead(II) removal from water. Environ. Sci. Pollut. Res. 2012, 19, 1224–1228. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Thomas, K.M. Competitive Adsorption of Aqueous Metal Ions on an Oxidized Nanoporous Activated Carbon. Langmuir 2004, 20, 4566–4578. [Google Scholar] [CrossRef] [PubMed]
- Demiral, H.; Güngör, C. Adsorption of copper(II) from aqueous solutions on activated carbon prepared from grape bagasse. J. Clean. Prod. 2016, 124, 103–113. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Copper in Drinking-Water Background Document for Development of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Hua, M.; Zhang, S.; Pan, B.; Zhang, W.; Lv, L.; Zhang, Q. Heavy metal removal from water/wastewater by nanosized metal oxides: A review. J. Hazard. Mater. 2012, 211–212, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Karami, H. Heavy metal removal from water by magnetite nanorods. Chem. Eng. J. 2013, 219, 209–216. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Ku, Y.; Jung, I.L. Photocatalytic reduction of Cr(VI) in aqueous solutions by UV irradiation with the presence of titanium dioxide. Water Res. 2001, 35, 135–142. [Google Scholar] [CrossRef]
- Alvarez, M.T.; Crespo, C.; Mattiasson, B. Precipitation of Zn(II), Cu(II) and Pb(II) at bench-scale using biogenic hydrogen sulfide from the utilization of volatile fatty acids. Chemosphere 2007, 66, 1677–1683. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.; Wu, J.; Zhang, W.; Li, H.; Hen, Y.C.; Pan, B. Selective Removal of Cu(II) Ions by Using Cation-exchange Resin-Supported Polyethyleneimine (PEI) Nanoclusters. Environ. Sci. Technol. 2010, 44, 3508–3513. [Google Scholar] [CrossRef]
- Dermentzis, K.; Davidis, A.; Papadopoulou, D.; Christoforidis, A.; Ouzounis, K. Copper removal from industrial wastewaters by means of electrostatic shielding driven electrodeionization. J. Eng. Sci. Technol. Rev. 2009, 2, 131–136. [Google Scholar] [CrossRef]
- Mouni, L.; Merabet, D.; Bouzaza, A.; Belkhiri, L. Adsorption of Pb(II) from aqueous solutions using activated carbon developed from Apricot stone. Desalination 2011, 276, 148–153. [Google Scholar] [CrossRef]
- Ngah, W.S.W.; Fatinathan, S. Pb(II) biosorption using chitosan and chitosan derivatives beads: Equilibrium, ion exchange and mechanism studies. J. Environ. Sci. 2010, 22, 338–346. [Google Scholar] [CrossRef]
- Ngah, W.S.W.; Fatinathan, S. Adsorption of Cu(II) ions in aqueous solution using chitosan beads, chitosan-GLA beads and chitosan-alginate beads. Chem. Eng. J. 2008, 143, 62–72. [Google Scholar] [CrossRef]
- Kumar Gupta, V.; Nayak, A.; Agarwal, S. Bioadsorbents for remediation of heavy metals: Current status and their future prospects. Environ. Eng. Res. 2015, 20, 1–18. [Google Scholar] [CrossRef]
- Elieh-Ali-Komi, D.; Hamblin, M.R. Chitin and Chitosan: Production and Application of Versatile Biomedical Nanomaterials HHS Public Access. Int. J. Adv. Res. 2016, 4, 411–427. [Google Scholar]
- Zhang, L.; Zeng, Y.; Cheng, Z. Removal of heavy metal ions using chitosan and modified chitosan: A review. J. Mol. Liq. 2016, 214, 175–191. [Google Scholar] [CrossRef]
- Aliabadi, M.; Irani, M.; Ismaeili, J.; Najafzadeh, S. Design and evaluation of chitosan/hydroxyapatite composite nanofiber membrane for the removal of heavy metal ions from aqueous solution. J. Taiwan Inst. Chem. Eng. 2014, 45, 518–526. [Google Scholar] [CrossRef]
- Karthik, R.; Meenakshi, S. Synthesis, characterization and Cr(VI) uptake study of polyaniline coated chitin. Int. J. Biol. Macromol. 2015, 72, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Maleki, A.; Pajootan, E.; Hayati, B. Ethyl acrylate grafted chitosan for heavy metal removal from wastewater: Equilibrium, kinetic and thermodynamic studies. J. Taiwan Inst. Chem. Eng. 2015, 51, 127–134. [Google Scholar] [CrossRef]
- Laus, R.; Tadeu De Fávere, V. Competitive adsorption of Cu(II) and Cd(II) ions by chitosan crosslinked with epichlorohydrin-triphosphate. Bioresour. Technol. 2011, 102, 8769–8776. [Google Scholar] [CrossRef] [PubMed]
- Morales Futalan, C.; Kan, C.-C.; Lourdes Dalida, M.; Hsien, K.-J.; Pascua, C.; Wan, M.-W. Comparative and competitive adsorption of copper, lead, and nickel using chitosan immobilized on bentonite. Carbohydr. Polym. 2010, 83, 528–536. [Google Scholar] [CrossRef]
- Zoppe, J.O.; Ataman, N.C.; Mocny, P.; Wang, J.; Moraes, J.; Klok, H.-A. Surface-Initiated Controlled Radical Polymerization: State-of-the-Art, Opportunities, and Challenges in Surface and Interface Engineering with Polymer Brushes. Chem. Rev. 2017, 117, 1105–1318. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Yun, Y.-S.; Cho, H.Y.; Park, J.M. Chromium Biosorption by Thermally Treated Biomass of the Brown Seaweed, Ecklonia sp. Ind. Eng. Chem. Res. 2004, 43, 8226–8232. [Google Scholar] [CrossRef]
- Tugulu, S.; Barbey, R.; Harms, M.; Fricke, M.; Volkmer, D.; Rossi, A.A.; Klok, H.-A. Synthesis of Poly(methacrylic acid) Brushes via Surface-Initiated Atom Transfer Radical Polymerization of Sodium Methacrylate and Their Use as Substrates for the Mineralization of Calcium Carbonate. Macromolecules 2006, 40, 168–177. [Google Scholar] [CrossRef]
- Stephen, G.B.; Akgun, B.; William, J.B.; Foster, M.D. Synthesis, Characterization, and Properties of Polyelectrolyte Block Copolymer Brushes Prepared by Atom Transfer Radical Polymerization and Their Use in the Synthesis of Metal Nanoparticles. Macromolecules 2003, 36, 9539–9548. [Google Scholar] [CrossRef]
- Sheng, P.X.; Ting, Y.-P.; Chen, J.P.; Hong, L. Sorption of lead, copper, cadmium, zinc, and nickel by marine algal biomass: Characterization of biosorptive capacity and investigation of mechanisms. J. Colloid Interface Sci. 2004, 275, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Colombani, O.; Ruppel, M.; Schubert, F.; Zettl, H.; Dmitry, V.P.; Axel, H.E.M. Synthesis of Poly(n-butyl acrylate)-block-poly(acrylic acid) Diblock Copolymers by ATRP and Their Micellization in Water. Macromolecules 2007, 40, 4338–4350. [Google Scholar] [CrossRef]
- Grande, C.D.; Alí, F.; Zuluaga, F. Síntesis de quitosano modificado con poli (ácido acrílico) vía polimerización por transferencia atómica iniciada desde la superficie (SIP-ATRP). Iberoam. Polím. 2010, 11, 505–519. [Google Scholar]
- Nalwa, H.S. Handbook of Organic Conductive Molecules and Polymers, 3rd ed.; John Wiley & Sons: New York, NY, USA, 1997; Volume 3, ISBN 9780471965954. [Google Scholar]
- Beamson, G.; Briggs, D. High resolution XPS of organic polymers: The Scienta ESCA 300 database. In Surface and Interface Analysis; Wiley-Blackwell: New York, NY, USA, 1992; ISBN 0471 935921. [Google Scholar]
- Save, M.; Granvorka, G.; Bernard, J.; Charleux, B.; Boissière, C.; Grosso, D.; Sanchez, C. Atom Transfer Radical Polymerization of Styrene and Methyl Methacrylate from Mesoporous Ordered Silica Particles. Macromol. Rapid Commun. 2006, 27, 393–398. [Google Scholar] [CrossRef]
- Wang, X.; Kim, Y.-G.; Drew, C.; Ku, B.-C.; Kumar, J.; Samuelson, L.A. Electrostatic Assembly of Conjugated Polymer Thin Layers on Electrospun Nanofibrous Membranes for Biosensors. Nano Lett. 2004, 4, 331–334. [Google Scholar] [CrossRef]
- Xu, C.; Wang, J.; Yang, T.; Chen, X.; Liu, X.; Ding, X. Adsorption of uranium by amidoximated chitosan-grafted polyacrylonitrile, using response surface methodology. Carbohydr. Polym. 2015, 121, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.L.; Bai, R. Adsorption of lead and humic acid on chitosan hydrogel beads. Water Res. 2005, 39, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Claesson, P.M.; Ninham, B.W. pH-dependent interactions between adsorbed chitosan layers. Langmuir 1992, 8, 1406–1412. [Google Scholar] [CrossRef]
- Zhang, X.; Bai, R. Mechanisms and kinetics of humic acid adsorption onto chitosan-coated granules. J. Colloid Interface Sci. 2003, 264, 30–38. [Google Scholar] [CrossRef]
- And, N.L.; Bai, R. Highly Enhanced Adsorption of Lead Ions on Chitosan Granules Functionalized with Poly(acrylic acid). Ind. Eng. Chem. Res. 2006, 45, 7897–7904. [Google Scholar] [CrossRef]
- Langmuir, I. The Constitution and Fundamental Properties of Solids and Liquids. Part I. Solids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- James, E.H. Principles of Chemical Kinetics, 2nd ed.; Academic Press: Suite 400, Burlington, MA, USA, 2007; ISBN 978-0-12-356787-1. [Google Scholar]
- Inyinbor, A.A.; Adekola, F.A.; Olatunji, G.A. Kinetics, isotherms and thermodynamic modeling of liquid phase adsorption of Rhodamine B dye onto Raphia hookerie fruit epicarp. Water Resour. Ind. 2016, 15, 14–27. [Google Scholar] [CrossRef]
- Saha, P.; Chowdhury, S.; Gupta, S.; Kumar, I. Insight into adsorption equilibrium, kinetics and thermodynamics of Malachite Green onto clayey soil of Indian origin. Chem. Eng. J. 2010, 165, 874–882. [Google Scholar] [CrossRef]
- Kapoor, A.; Yang, R.T. Surface diffusion on energetically heterogeneous surfaces. AIChE J. 1989, 35, 1735–1738. [Google Scholar] [CrossRef]
- Piccin, J.S.; Dotto, G.L.; Pinto, L.A.A. Adsorption Isotherms and Thermochemical data of FD&C RED N° 40 Binding by Qhitosan. Braz. J. Chem. Eng. 2011, 28, 295–304. [Google Scholar] [CrossRef]
- Wong, Y.C.; Szeto, Y.S.; Cheung, W.H.; Mckay, G. Adsorption of acid dyes on chitosan-equilibrium isotherm analyses. Process Biochem. 2004, 39, 693–702. [Google Scholar] [CrossRef]
- Darder, M.; Montserrat Colilla, A.; Ruiz-Hitzky, E. Biopolymer−Clay Nanocomposites Based on Chitosan Intercalated in Montmorillonite. Chem. Mater. 2003, 15, 3774–3780. [Google Scholar] [CrossRef]
- Wan, M.-W.; Kan, C.-C.; Rogel, B.D.; Lourdes, M.; Dalida, P. Adsorption of copper (II) and lead (II) ions from aqueous solution on chitosan-coated sand. Carbohydr. Polym. 2010, 80, 891–899. [Google Scholar] [CrossRef]
- Aydın, H.; Bulut, Y.; Yerlikaya, Ç. Removal of copper (II) from aqueous solution by adsorption onto low-cost adsorbents. J. Environ. Manag. 2008, 87, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Özer, A.; Özer, D.; Özer, A. The adsorption of copper(II) ions on to dehydrated wheat bran (DWB): Determination of the equilibrium and thermodynamic parameters. Process Biochem. 2004, 39, 2183–2191. [Google Scholar] [CrossRef]
- Liang, S.; Guo, X.; Feng, N.; Tian, Q. Adsorption of Cu2+ and Cd2+ from aqueous solution by mercapto-acetic acid modified orange peel. Colloids Surf. B Biointerfaces 2009, 73, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.; Saeed, A.; Kalim, I. Characterization of Adsorptive Capacity and Investigation of Mechanism of Cu2+, Ni2+ and Zn2+ Adsorption on Mango Peel Waste from Constituted Metal Solution and Genuine Electroplating Effluent. Sep. Sci. Technol. 2009, 44, 3770–3791. [Google Scholar] [CrossRef]
- Çay, S.; Uyanık, A.; Özaşık, A. Single and binary component adsorption of copper(II) and cadmium(II) from aqueous solutions using tea-industry waste. Sep. Purif. Technol. 2004, 38, 273–280. [Google Scholar] [CrossRef]
- Zheng, G.H.; Wang, L.; Zhou, Q.; Li, F.T. Optimisation of cell surface and structural components for improving adsorption capacity of Pseudomonas putida 5-x to Cu2+. Int. J. Environ. Pollut. 2008, 34, 285. [Google Scholar] [CrossRef]
- Öztürk, A.; Artan, T.; Ayar, A. Biosorption of nickel(II) and copper(II) ions from aqueous solution by Streptomyces coelicolor A3(2). Colloids Surf. B Biointerfaces 2004, 34, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, M.; Noronha, S.B.; Suraishkumar, G.K. Kinetic modeling for the biosorption of copper by pretreated Aspergillus niger biomass. Bioresour. Technol. 2006. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.P.; Yang, L. Chemical Modification of Sargassum sp. for Prevention of Organic Leaching and Enhancement of Uptake during Metal Biosorption. Ind. Eng. Chem. Res. 2005, 44, 9931–9942. [Google Scholar] [CrossRef]
- Gupta, V.K.; Rastogi, A.; Saini, V.K.; Jain, N. Biosorption of copper(II) from aqueous solutions by Spirogyra species. J. Colloid Interface Sci. 2006, 296, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Bulut, Y.; Baysal, Z. Removal of Pb(II) from wastewater using wheat bran. J. Environ. Manag. 2006, 78, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Naiya, T.K.; Bhattacharya, A.K.; Mandal, S.; Das, S.K. The sorption of lead(II) ions on rice husk ash. J. Hazard. Mater. 2009, 163, 1254–1264. [Google Scholar] [CrossRef] [PubMed]
- Anwar, J.; Shafique, U. Waheed-uz-Zaman; Salman, M.; Dar, A.; Anwar, S. Removal of Pb(II) and Cd(II) from water by adsorption on peels of banana. Bioresour. Technol. 2010, 101, 1752–1755. [Google Scholar] [CrossRef] [PubMed]
- Uslu, G.; Tanyol, M. Equilibrium and thermodynamic parameters of single and binary mixture biosorption of lead (II) and copper (II) ions onto Pseudomonas putida: Effect of temperature. J. Hazard. Mater. 2006, 135, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Selatnia, A.; Bakhti, M.Z.; Madani, A.; Kertous, L.; Mansouri, Y. Biosorption of Cd2+ from aqueous solution by a NaOH-treated bacterial dead Streptomyces rimosus biomass. Hydrometallurgy 2004, 75, 11–24. [Google Scholar] [CrossRef]
- Chen, C.; Wang, J. Influence of metal ionic characteristics on their biosorption capacity by Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 2007, 74, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Ting, Y.-P. Characterization of PEI-modified biomass and biosorption of Cu(II), Pb(II) and Ni(II). Water Res. 2005, 39, 2167–2177. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.; Cheng, P. Biosorption of Metal Ions with Penicillium chrysogenum. Appl. Biochem. Biotechnol. 2003, 104, 119–128. [Google Scholar] [CrossRef]
- Martins, B.L.; Cruz, C.C.V.; Luna, A.S.; Henriques, C.A. Sorption and desorption of Pb2+ ions by dead Sargassum sp. biomass. Biochem. Eng. J. 2006, 27, 310–314. [Google Scholar] [CrossRef]
- Gupta, V.K.; Rastogi, A. Biosorption of lead from aqueous solutions by green algae Spirogyra species: Kinetics and equilibrium studies. J. Hazard. Mater. 2008, 152, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Aljeboree, A.M.; Alshirifi, A.N.; Alkaim, A.F. Kinetics and equilibrium study for the adsorption of textile dyes on coconut shell activated carbon. Arab. J. Chem. 2017, 10, S3381–S3393. [Google Scholar] [CrossRef]
- Annadurai, G.; Juang, R.S.; Lee, D.J. Adsorption of heavy metals from water using banana and orange peels. Water Sci. Technol. 2003, 47, 185–190. [Google Scholar] [CrossRef]
- Robati, D. Pseudo-second-order kinetic equations for modeling adsorption systems for removal of lead ions using multi-walled carbon nanotube. J. Nanostruct. Chem. 2013, 3, 55. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, A.; Peng, F.; Yu, H.; Yang, J. Mechanism study on adsorption of acidified multiwalled carbon nanotubes to Pb(II). J. Colloid Interface Sci. 2007, 316, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Kovačevic, D.; Pohlmeier, A.; Özbas, G.; Narres, H.-D.; Kallay, M.J.N. The adsorption of lead species on goethite. Colloids Surf. A Physicochem. Eng. Asp. 2000, 166, 225–233. [Google Scholar] [CrossRef]
- Li, N.; Bai, R.; Liu, C. Enhanced and Selective Adsorption of Mercury Ions on Chitosan Beads Grafted with Polyacrylamide via Surface-Initiated Atom Transfer Radical Polymerization. Langmuir 2005, 21, 11780–11787. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available. |
| Ions | Method | Operation Condition | Removal Efficiency | Advantages/Disadvantages |
|---|---|---|---|---|
| Cu2+, Pb2+, Zn2+ | Chemical precipitation | Cu2+ = 0.018 mM, Pb2+ = 2.3 mM, Zn2+ = 1.34 mM; precipitant, H2S; pH = 3.0 | Cu2+ (100%), Pb2+ = (92%), Zn2+ = (94%) | Most widely used process in industry; it is relatively simple and inexpensive/it generates large volumes of relatively low density sludge; sulfide precipitants can result in the evolution of H2S [10,11]. |
| Cu2+, Cr3+, Pb2+, Zn2+ | [metal] = 100 mg·L−1 precipitant, CaO; pH = 3.0 | >99% | ||
| Cu2+ | Ion exchange | Cu2+ = 100 mg·L−1; Resin-supported polyethyleneimine; pH = 5.0 | >99% | High treatment capacity, high removal efficiency and fast kinetics/leaching during operation, highest costs for synthetic resins [9,11,12] |
| Pb2+; Cu2+ | Cu2+ = 25 mg·L−1; Resin-THQSA; pH = 4.5 | 60–90% | ||
| Cu2 | membrane separation—Electrodyalisis | Cu2+ = 100 mg·L−1; membrane: packed beds of graphite powder; flow 1.29 × 10−4 Ls−1; current density 2 mAcm2; pH = 3.0 | >99% | It has high efficiency, it requires little space, it is not selective and is easy to operate/it generates a large amount of metal-containing sludge [13]. |
| Pb2+ | Adsorption | Pb2+ = 50 mg·L−1; adsorbent: active carbon; pH = 6.0 | >85% | Effective and economic method; it is flexible in design and operation and besides it can produce high-quality treated effluent; easy metal recovery [14,15,16] |
| Pb2+ | Pb2+ = 50 mg·L−1; adsorbent: chitosan; pH = 4.5 | >60% | ||
| Cu2+ | Pb2+ = 10 mg·L−1; adsorbent: chitosan; pH = 4.5 | >80% |
| Isothermal Model | Adsorbent | Metal Ion | Parameter (Unit) | Value |
|---|---|---|---|---|
| Langmuir | CS | Cu2+ | qm (mg·g−1) | 133 |
| kL (L·g−1) | 29.8 | |||
| 1 R2 | 0.998 | |||
| ARE | 3.30% | |||
| Pb2+ | qm (mg·g−1) | 54 | ||
| K (L·g−1) | 42.6 | |||
| R2 | 0.998 | |||
| ARE | 4.50% | |||
| CS-g-TA | Cu2+ | qm (mg·g−1) | 136 | |
| K(L·g−1) | 32.3 | |||
| R2 | 0.998 | |||
| ARE | 4.20% | |||
| Pb2+ | qm (mg·g−1) | 80.6 | ||
| K (L/g) | 39.6 | |||
| R2 | 0.997 | |||
| ARE | 3.50% |
| Adsorbent | Metal Ion | 1k2 (g·mg−1·min−1) × 10−4 | qe (mg·g−1) | R2 |
|---|---|---|---|---|
| CS | Cu2+ | 1.7 | 140 | 0.997 |
| Pb2+ | 19.8 | 14.8 | 0.998 | |
| CS-g-PA | Cu2+ | 1.8 | 164 | 0.997 |
| Pb2+ | 2.8 | 98.0 | 0.999 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grande-Tovar, C.D.; Vallejo, W.; Zuluaga, F. Equilibrium and Kinetic Study of Lead and Copper Ion Adsorption on Chitosan-Grafted-Polyacrylic Acid Synthesized by Surface Initiated Atomic Transfer Polymerization. Molecules 2018, 23, 2218. https://doi.org/10.3390/molecules23092218
Grande-Tovar CD, Vallejo W, Zuluaga F. Equilibrium and Kinetic Study of Lead and Copper Ion Adsorption on Chitosan-Grafted-Polyacrylic Acid Synthesized by Surface Initiated Atomic Transfer Polymerization. Molecules. 2018; 23(9):2218. https://doi.org/10.3390/molecules23092218
Chicago/Turabian StyleGrande-Tovar, Carlos David, William Vallejo, and Fabio Zuluaga. 2018. "Equilibrium and Kinetic Study of Lead and Copper Ion Adsorption on Chitosan-Grafted-Polyacrylic Acid Synthesized by Surface Initiated Atomic Transfer Polymerization" Molecules 23, no. 9: 2218. https://doi.org/10.3390/molecules23092218
APA StyleGrande-Tovar, C. D., Vallejo, W., & Zuluaga, F. (2018). Equilibrium and Kinetic Study of Lead and Copper Ion Adsorption on Chitosan-Grafted-Polyacrylic Acid Synthesized by Surface Initiated Atomic Transfer Polymerization. Molecules, 23(9), 2218. https://doi.org/10.3390/molecules23092218






