Physicochemical Characterization, Antioxidant and Immunostimulatory Activities of Sulfated Polysaccharides Extracted from Ascophyllum nodosum
Abstract
1. Introduction
2. Results and Discussion
2.1. Optimization of AnPS Extraction Conditions
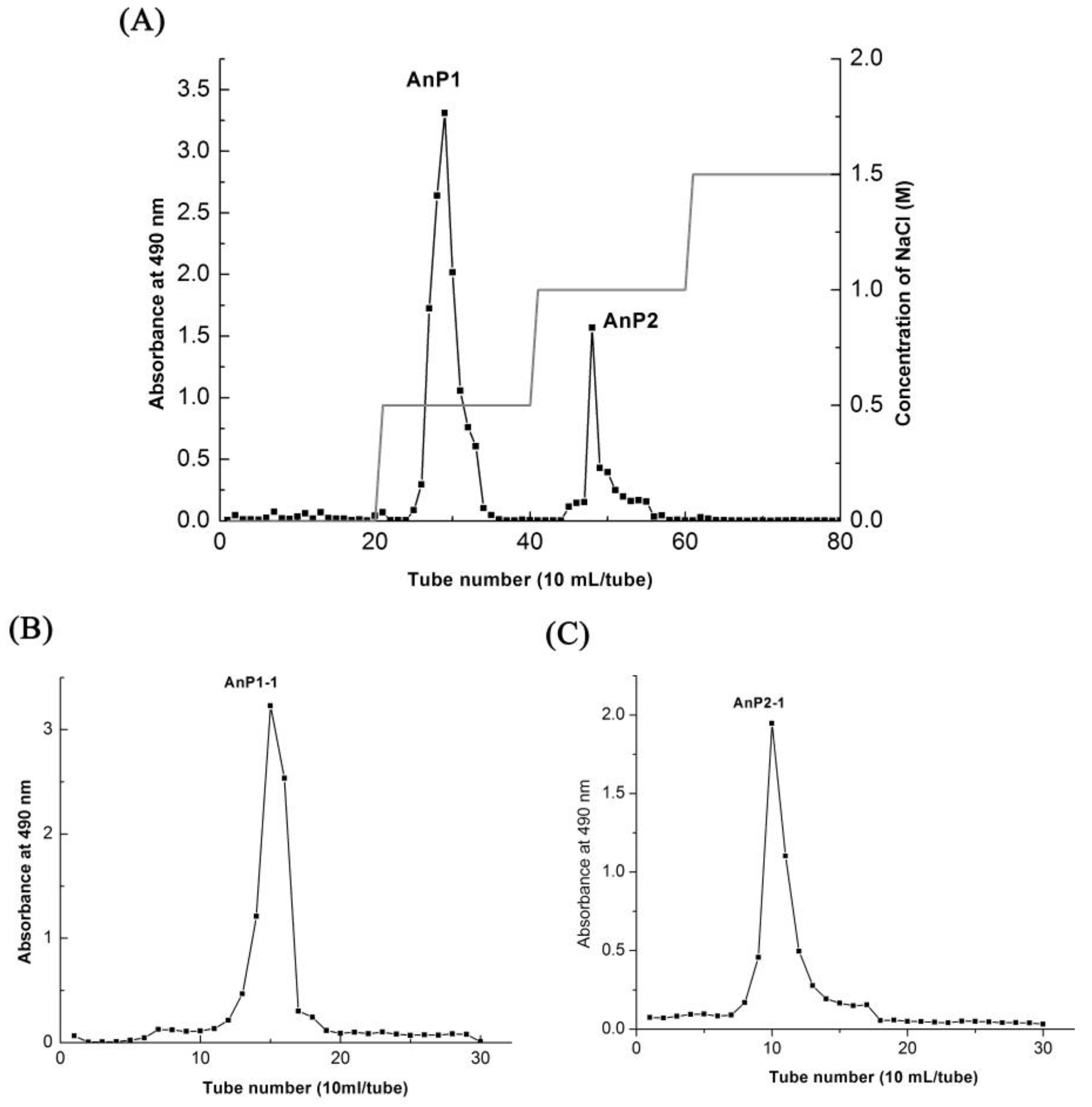
2.2. Isolation and Purification of Polysaccharide Fraction AnP1-1 and AnP2-1
2.3. Characterization, Molecular Weight and Monosaccharide Composition of AnP1-1 and AnP2-1
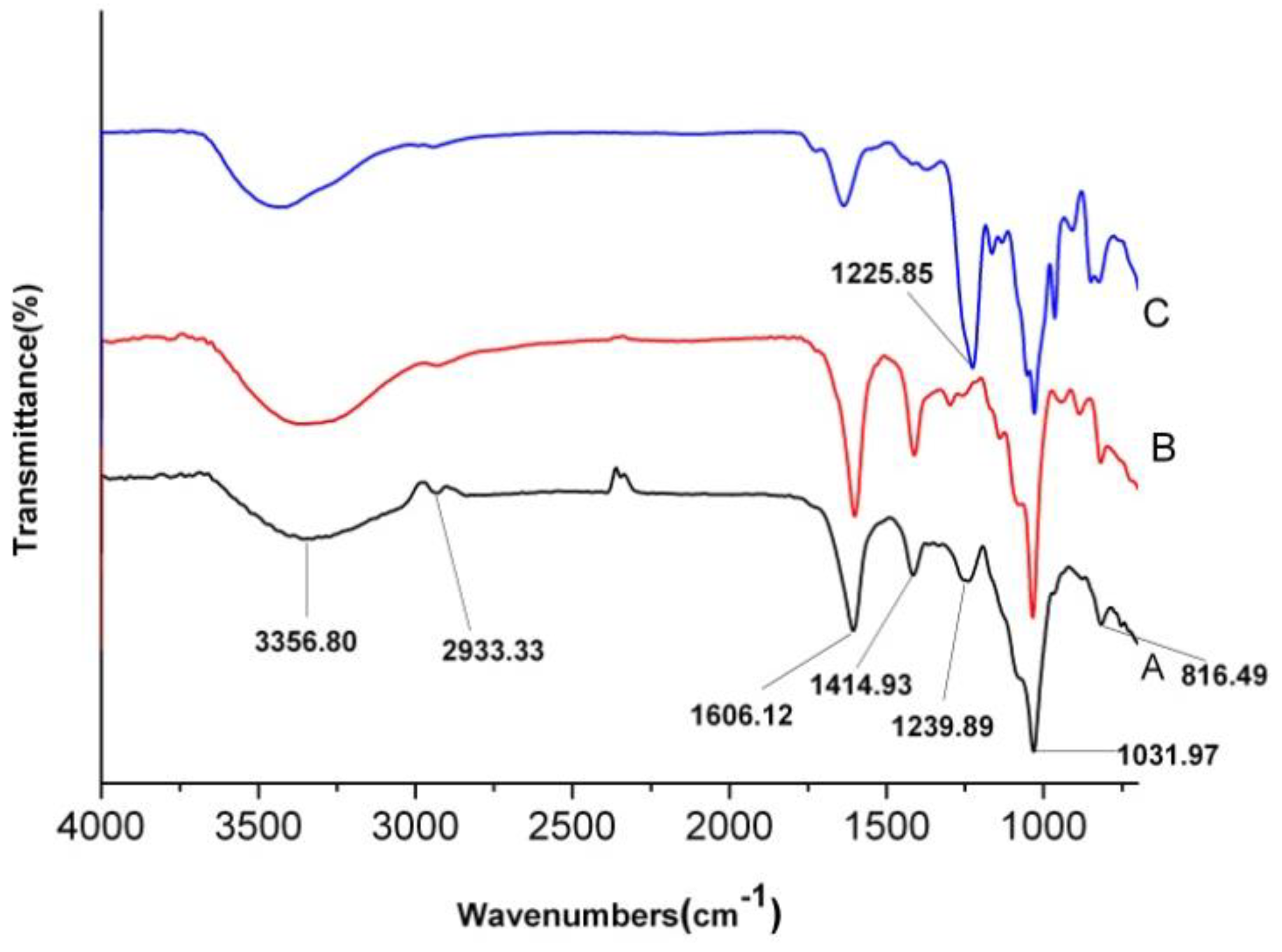
2.4. FT-IR Analysis
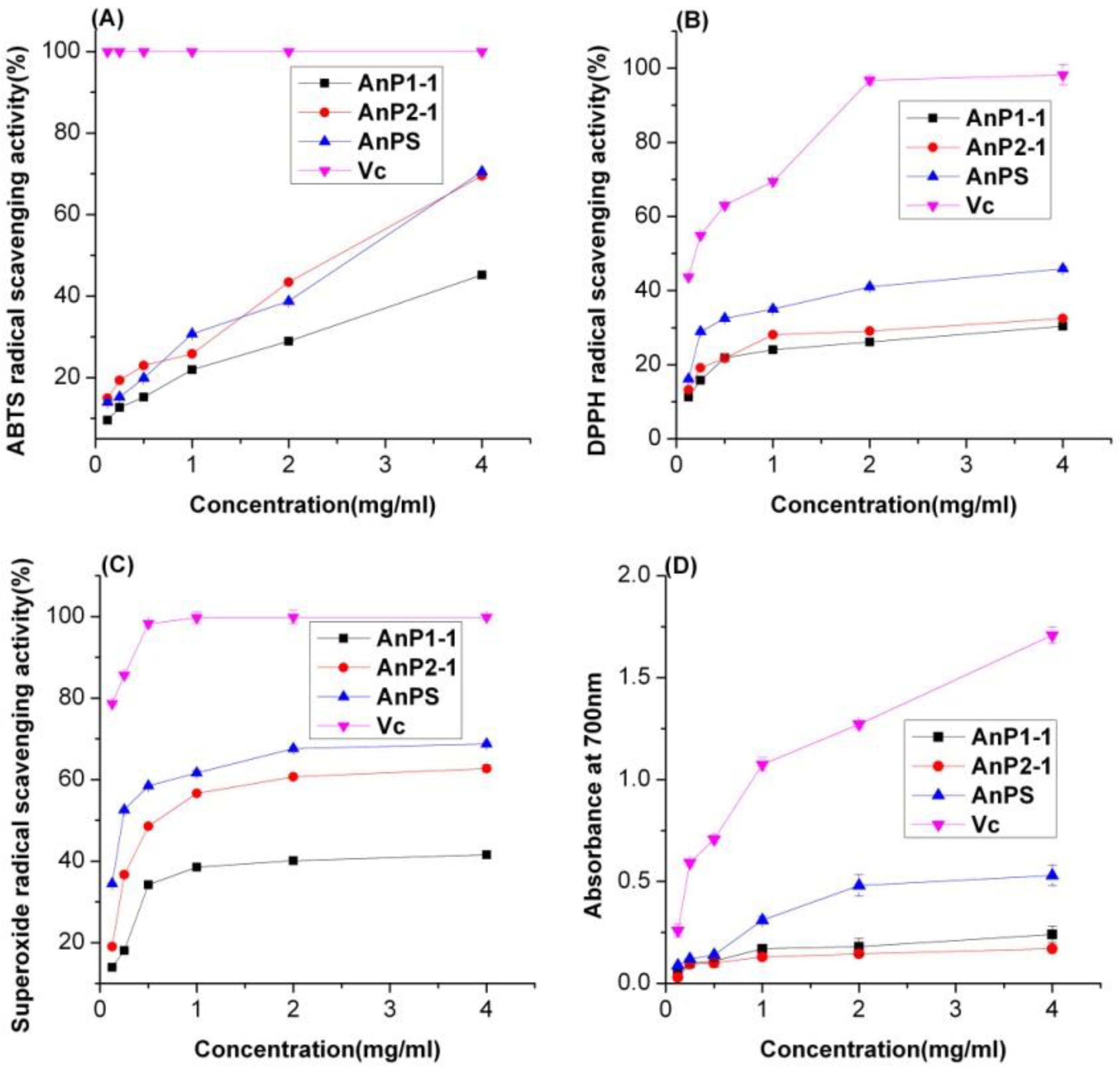
2.5. Antioxidant Activity of A. nodosum Polysaccharides
2.6. Protective Effect on H2O2-Induced Oxidative Injury
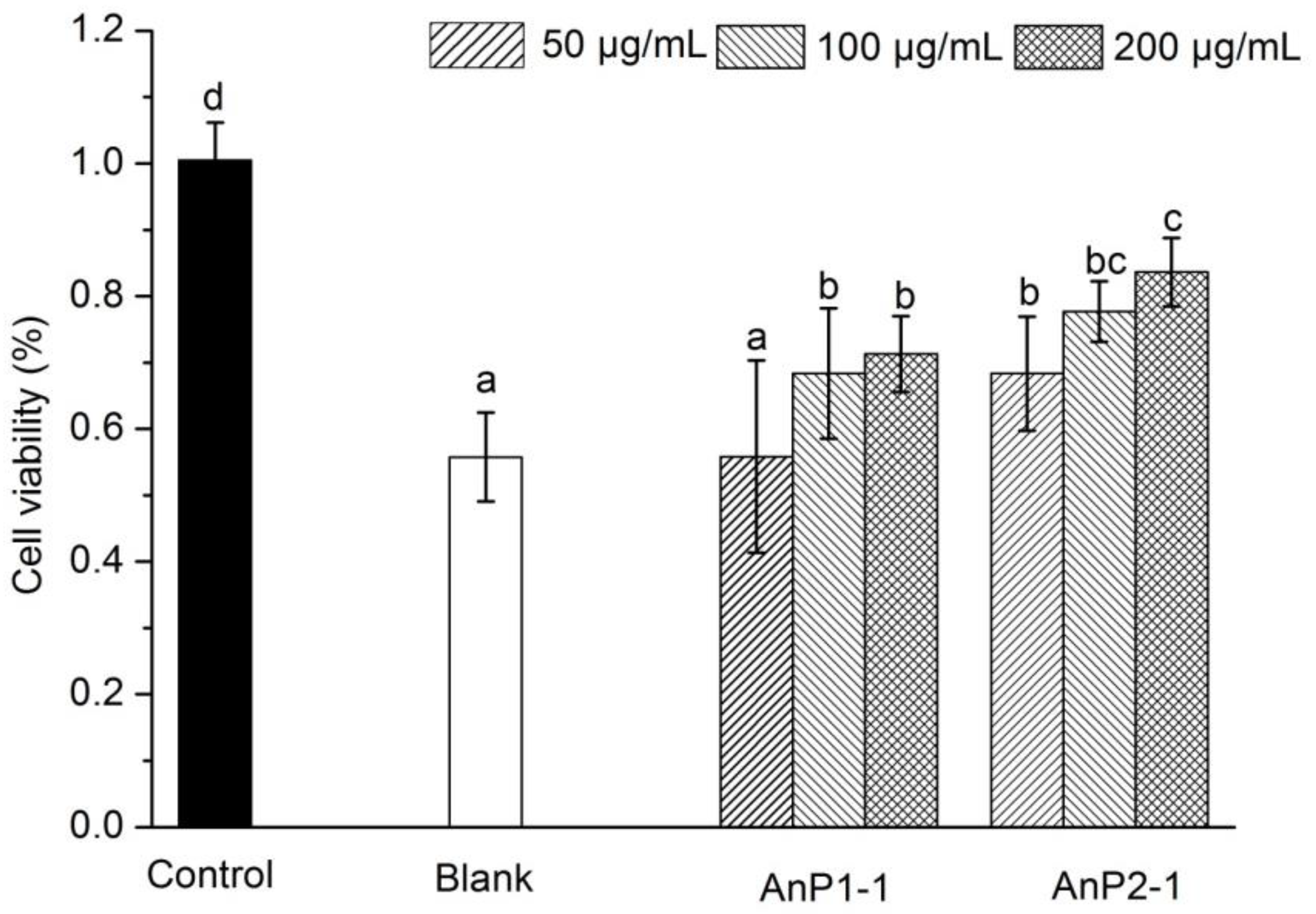
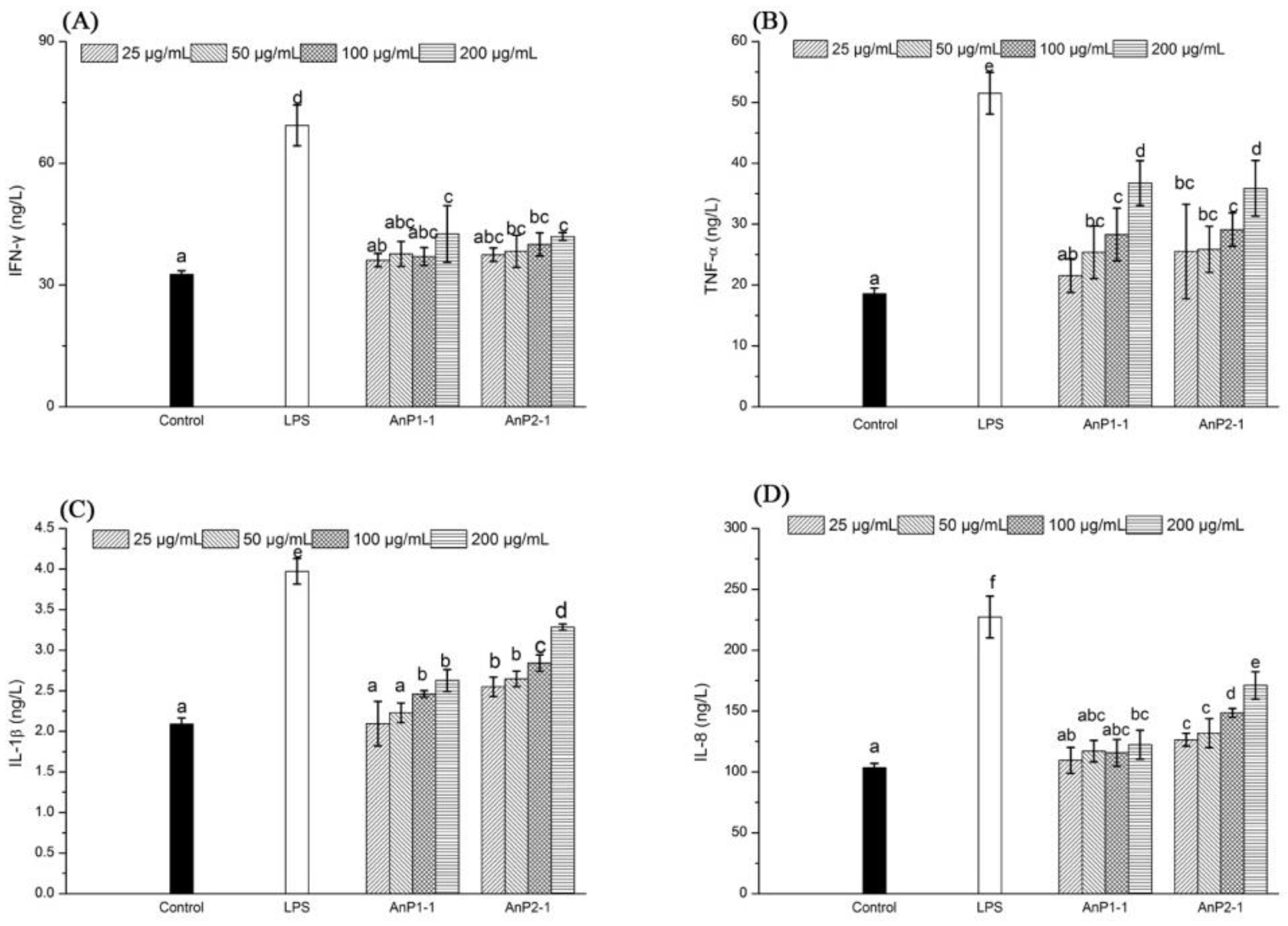
2.7. Immunostimulatory Activity on Caco-2 Cells
3. Materials and Methods
3.1. Materials and Chemicals
3.2. Extraction of Crude AnPS
3.3. Design of Experimental (DoE) in Response Surface Methodology (RSM)
3.4. Purification of A. nodosum Polysaccharides (AnPS)
3.5. Analysis and Characterization of the Polysaccharide Fractions
3.6. Antioxidant Activity Assays
3.6.1. ABTS Assay
3.6.2. DPPH Radical Scavenging Activity
3.6.3. Superoxide Anion Radical Scavenging Activity
3.6.4. Reducing Power Assay
3.7. Protective Effect on H2O2-Induced Oxidative Injury
3.8. Assay of Immunostimulatory Activity In Vitro
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chan, C.X.; Ho, C.L.; Phang, S.M. Trends in seaweed research. Trends Plant Sci. 2006, 11, 165–166. [Google Scholar] [CrossRef] [PubMed]
- Alamoudi, O.A.; Mutawie, H.H.; Patel, A.V.; Blunden, G. Chemical composition and antioxidant activities of Jeddah corniche algae, Saudi Arabia. Saudi J. Biol. Sci. 2009, 16, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Macquarrie, D.C. Microwave assisted extraction of sulfated polysaccharides (fucoidan) from Ascophyllum nodosum and its antioxidant activity. Carbohydr. Polym. 2015, 129, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Kindleysides, S.; Quek, S.Y.; Miller, M.R. Inhibition of fish oil oxidation and the radical scavenging activity of New Zealand seaweed extracts. Food Chem. 2012, 133, 1624–1631. [Google Scholar] [CrossRef]
- Balboa, E.M.; Conde, E.; Moure, A.; Falqué, E.; Domínguez, H. In vitro antioxidant properties of crude extracts and compounds from brown algae. Food Chem. 2013, 138, 1764–1785. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.S.; Wang, F.; Wang, X.M.; Liu, X.L.; Hou, Y.; Zhang, Q.B. Extraction of the polysaccharides from five algae and their potential antioxidant activity in vitro. Carbohydr. Polym. 2010, 82, 118–121. [Google Scholar] [CrossRef]
- Yang, W.N.; Chen, P.W.; Huang, C.Y. Compositional characteristics and in vitro evaluations of antioxidant and neuroprotective properties of crude extracts of fucoidan prepared from compressional puffing-pretreated Sargassum crassifolium. Mar. Drugs 2017, 15, 183. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.; Sun, H. Purification of a fucoidan from kelp polysaccharide and its inhibitory kinetics for tyrosinase. Carbohydr. Polym. 2014, 99, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Wu, J.; Liu, T.T.; Hu, Y.D.; Zheng, Q.S.; Wang, B.S.; Lin, H.Y.; Li, X. Separation, characterization and anticancer activities of a sulfated polysaccharide from Undaria pinnatifida. Int. J. Biol. Macromol. 2016, 83, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.J.; Yuan, Q.X.; Saeeduddin, M.; Ou, S.; Zeng, X.X.; Ye, H. Recent advances in tea polysaccharides: Extraction, purification, physicochemical characterization and bioactivities. Carbohydr. Polym. 2016, 153, 663–678. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.J.; Xie, M.H.; Wan, P.; Chen, D.; Ye, H.; Chen, L.G.; Zeng, X.X.; Liu, Z.H. Digestion under saliva, simulated gastric and small intestinal conditions and fermentation in vitro by human intestinal microbiota of polysaccharides from Fuzhuan brick tea. Food Chem. 2018, 244, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.D.; Song, G.; Liu, J.; Wang, J.Y.; Zhang, P.Y.; Chen, K.S. Beneficial effects of extracellular polysaccharide from Rhizopus nigricans on the intestinal immunity of colorectal cancer mice. Int. J. Biol. Macromol. 2018, 115, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Yu, K.W.; Hong, H.D.; Shin, K.S. Effect of arabinoxylan- and rhamnogalacturonan I-rich polysaccharides isolated from young barley leaf on intestinal immunostimulatory activity. Funct. Foods 2017, 35, 384–390. [Google Scholar] [CrossRef]
- Brito, T.V.; Barros, F.C.N.; Silva, R.O.; Dias Júnior, G.J.; Júnior, J.S.C.; Franco, Á.X.; Soares, P.M.G.; Chaves, L.S.; Abreu, C.M.W.S.; de Paula, R.C.M.; et al. Sulfated polysaccharide from the marine algae Hypnea musciformis inhibits TNBS-induced intestinal damage in rats. Carbohydr. Polym. 2016, 151, 957–964. [Google Scholar] [CrossRef] [PubMed]
- Hammi, K.M.; Hammami, M.; Rihouey, C.; Cerf, D.L.; Ksouri, R.; Majdoub, H. Optimization extraction of polysaccharide from Tunisian Zizyphus lotus fruit by response surface methodology: Composition and antioxidant activity. Food Chem. 2016, 212, 476–484. [Google Scholar] [CrossRef] [PubMed]
- Samavati, V.; Yarmand, M.S. Statistical modeling of process parameters for the recovery of polysaccharide from Morus alba leaf. Carbohydr. Polym. 2013, 98, 793–806. [Google Scholar] [CrossRef] [PubMed]
- Ren, B.B.; Chen, C.; Li, C.; Fu, X.; You, L.J.; Liu, R.H. Optimization of microwave-assisted extraction of Sargassum thunbergii polysaccharides and its antioxidant and hypoglycemic activities. Carbohydr. Polym. 2017, 173, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.L.; Huang, Q.; Zhang, B.; Li, C.; Fu, X. Physicochemical characterization and in vitro hypoglycemic activities of polysaccharides from Sargassum pallidum by microwave-assisted aqueous two-phase extraction. Int. J. Biol. Macromol. 2018, 109, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Thambiraj, S.R.; Phillips, M.; Koyyalamudi, S.R.; Reddy, N. Antioxidant activities and characterisation of polysaccharides isolated from the seeds of Lupinus angustifolius. Ind. Crop. Prod. 2015, 74, 950–956. [Google Scholar] [CrossRef]
- Souza, B.W.; Cerqueira, M.A.; Bourbon, A.I.; Pinheiro, A.C.; Martins, J.T.; Teixeira, J.A.; Coimbra, M.A.; Vicente, A.A. Chemical characterization and antioxidant activity of sulfated polysaccharide from the red seaweed Gracilaria birdiae. Food Hydrocol. 2012, 27, 287–292. [Google Scholar] [CrossRef]
- Floegel, A.; Kim, D.O.; Chung, S.J.; Koo, S.I.; Chun, O.K. Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods. J. Food Compd. Anal. 2011, 24, 1043–1048. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Q.B.; Zhang, Z.S.; Li, Z.E. Antioxidant activity of sulfated polysaccharide fractions extracted from Laminaria japonica. Int. J. Biol. Macromol. 2008, 42, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Q.; Zhang, B.; Zhang, Z.S.; Song, H.F.; Li, P.C. Potential antioxidant and anticoagulant capacity of low molecular weight fucoidan fractions extracted from Laminaria japonica. Int. J. Biol. Macromol. 2010, 46, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.J.; Yuan, Q.X.; Zhang, Y.X.; Li, J.L.; Zhu, X.J.; Zhao, L.L.; Wen, J.; Liu, J.K.; Zhao, L.Y.; Zhao, J.H. Enzyme-Assisted Extraction Optimization, Characterization and Antioxidant Activity of Polysaccharides from Sea Cucumber Phyllophorus proteus. Molecules 2018, 23, 590. [Google Scholar] [CrossRef] [PubMed]
- Di, T.; Chen, G.J.; Sun, Y.; Ou, S.; Zeng, X.X.; Ye, H. Antioxidant and immunostimulating activities in vitro of sulfated polysaccharides isolated from Gracilaria rubra. J. Funct. Foods 2017, 28, 64–75. [Google Scholar] [CrossRef]
- Shim, W.B.; Kim, K.Y.; Chung, D.H. Development and Validation of a Gold Nanoparticle Immunochromatographic Assay (ICG) for the Detection of Zearalenone. J. Agric. Food Chem. 2009, 57, 4035–4041. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.F.; Yang, J.G.; Luo, Z.; Zhang, X.W. Lentinula edodes-derived polysaccharide enhances systemic and mucosal immunity by spatial modulation of intestinal gene expression in mice. Food Funct. 2015, 6, 2068–2080. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Q.X.; Xie, Y.F.; Wang, W.; Yan, Y.H.; Ye, H.; Jabbar, S.; Zeng, X.X. Extraction optimization, characterization and antioxidant activity in vitro of polysaccharides from mulberry (Morus alba L.) leaves. Carbohydr. Polym. 2015, 128, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Li, J.E.; Nie, S.P.; Xie, M.Y.; Li, C. Isolation and partial characterizationof a neutral polysaccharide from Mosla chinensis Maxim. cv. Jiangxiangru and its antioxidant and immunomodulatory activities. J. Funct. Foods 2014, 6, 410–418. [Google Scholar] [CrossRef]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Doigson, K.S.; Price, R.G. A note on the determination of the ester sulfate content of sulfated polysaccharides. Biochem. J. 1962, 84, 106–110. [Google Scholar] [CrossRef]
- Xie, M.H.; Hu, B.; Wang, Y.; Zeng, X.X. Grafting of gallic acid onto chitosan enhances antioxidant activities and alters rheological properties of the copolymer. J. Agric. Food Chem. 2014, 62, 9128–9136. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.B.; Huo, J.L.; Jiang, H.X.; Yang, F. Chemical compositions and bioactivites of crude polysaccharides from tea leaves beyond their useful date. Int. J. Biol. Macromol. 2011, 49, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.G.; Xia, X.; Chen, X.F.; Xiao, J.B.; Wang, Q.X. Charactetization of flavonoids from Dryopteris erythrosora and evaluation of their antioxidant, anticancer and acetylcholinesterase inhibition activities. Food Chem. Toxicol. 2013, 51, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Zhou, H.; Qian, H. Antioxidant and free radical-scavenging activities of wheat germ zsprotein hydrolysates (WGPH) prepared with alcalase. Process Biochem. 2006, 411, 296–302. [Google Scholar]
Sample Availability: Samples of the compounds AnPS are available from the authors. |
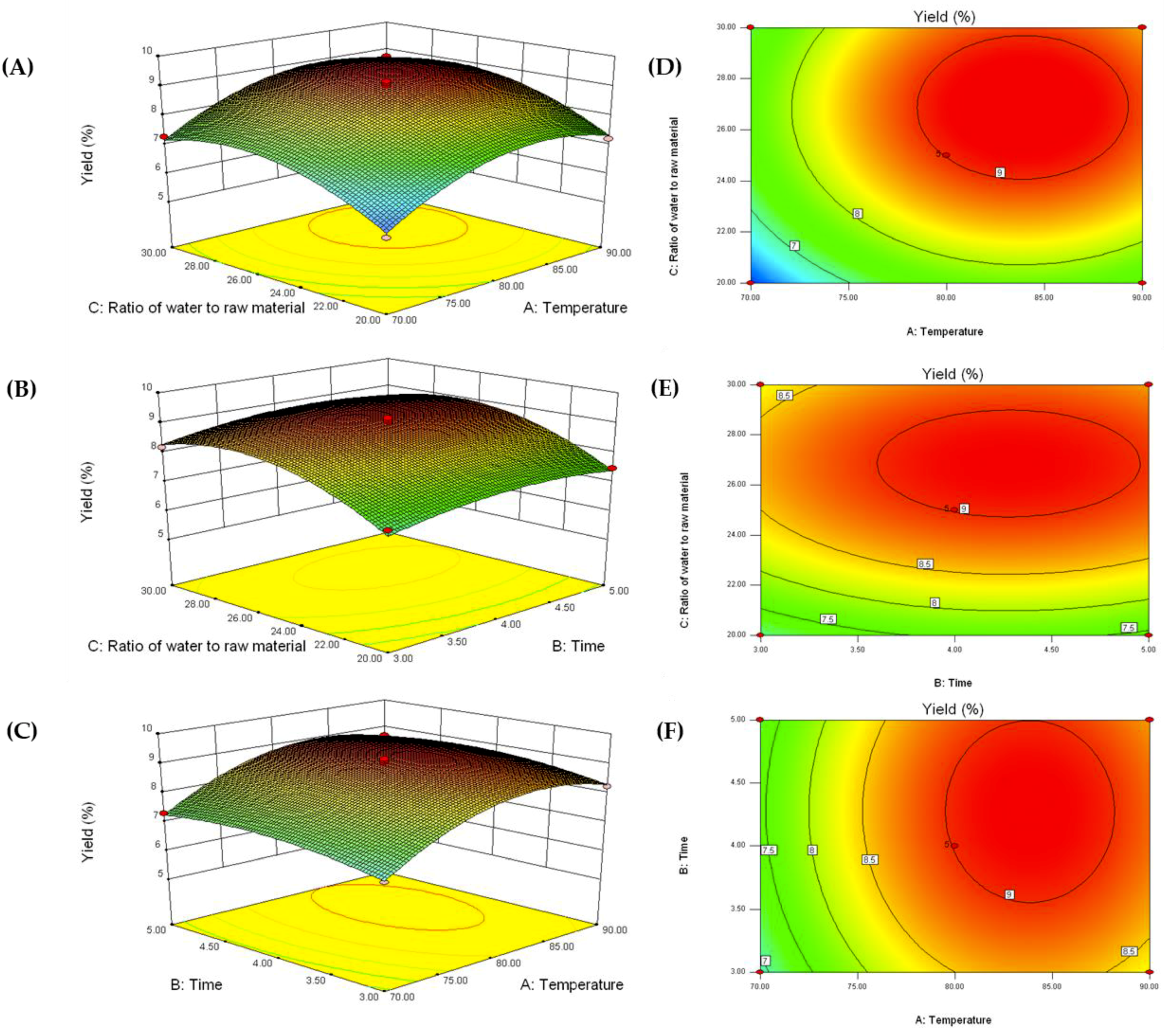
| Run | Temperature (°C) | Time (h) | Ratio of Water to Raw Material (mL/g) | Predicted Value (%) | Experimental Value (%) | |||
|---|---|---|---|---|---|---|---|---|
| X1 | Code X1 | X2 | Code X2 | X3 | Code X3 | |||
| 1 | 80 | 0 | 4 | 0 | 25 | 0 | 9.01 | 9.09 |
| 2 | 90 | 1 | 4 | 0 | 20 | −1 | 7.34 | 7.22 |
| 3 | 70 | −1 | 5 | 1 | 25 | 0 | 7.26 | 7.32 |
| 4 | 70 | −1 | 3 | −1 | 25 | 0 | 6.89 | 6.85 |
| 5 | 80 | 0 | 4 | 0 | 25 | 0 | 9.01 | 8.90 |
| 6 | 80 | 0 | 5 | 1 | 30 | 1 | 8.65 | 8.47 |
| 7 | 70 | −1 | 4 | 0 | 30 | 1 | 7.17 | 7.29 |
| 8 | 80 | 0 | 3 | −1 | 30 | 1 | 8.28 | 8.20 |
| 9 | 80 | 0 | 3 | −1 | 20 | −1 | 7.05 | 7.23 |
| 10 | 90 | 1 | 4 | 0 | 30 | 1 | 8.61 | 8.75 |
| 11 | 70 | −1 | 4 | 0 | 20 | −1 | 5.96 | 5.82 |
| 12 | 80 | 0 | 4 | 0 | 25 | 0 | 9.01 | 8.85 |
| 13 | 80 | 0 | 4 | 0 | 25 | 0 | 9.01 | 9.04 |
| 14 | 80 | 0 | 4 | 0 | 25 | 0 | 9.01 | 9.17 |
| 15 | 80 | 0 | 5 | 1 | 20 | −1 | 7.41 | 7.49 |
| 16 | 90 | 1 | 5 | 1 | 25 | 0 | 8.65 | 8.69 |
| 17 | 90 | 1 | 3 | −1 | 25 | 0 | 8.29 | 8.23 |
| Source | Sum of Squares | DF | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 14.77 | 9 | 1.64 | 49.98 | <0.0001 |
| X1 | 3.93 | 1 | 3.93 | 119.77 | <0.0001 |
| X2 | 0.27 | 1 | 0.27 | 8.11 | 0.0248 |
| X3 | 3.06 | 1 | 3.06 | 93.25 | <0.0001 |
| X1X2 | 0 | 1 | 0 | 0 | 0.9788 |
| X1X3 | 0 | 1 | 0 | 0.027 | 0.8732 |
| X2X3 | 0 | 1 | 0 | 0 | 0.9788 |
| X12 | 3.47 | 1 | 3.47 | 105.57 | <0.0001 |
| X22 | 0.46 | 1 | 0.46 | 13.96 | 0.0073 |
| X32 | 2.92 | 1 | 2.92 | 88.84 | <0.0001 |
| Residual | 0.23 | 7 | 0.033 | ||
| Lack of fit | 0.16 | 3 | 0.053 | 3.01 | 0.1575 |
| Pure error | 0.071 | 4 | 0.018 | ||
| Cor. total | 15 | 16 |
| Item | AnPS | AnP1-1 | AnP2-1 |
|---|---|---|---|
| Carbohydrate (%) | 42.31 ± 1.12 | 26.55 ± 1.12 | 36.69 ± 0.03 |
| Protein (%) | 1.43 ± 0.02 | 0.53 ± 0.01 | 0.42 ± 0.04 |
| Total polyphenols (mg GAE/100 mg) | 2.11 ± 0.04 | 0.13 ± 0.01 | 0.11 ± 0.01 |
| Uronic acid (%) | 11.04 ± 0.23 | 13.53 ± 0.79 | 3.56 ± 0.80 |
| sulfate group (%) | 23.9 ± 0.07 | 5.4 ± 0.11 | 23.8 ± 0.02 |
| Molecular weight (kDa) | - | 165.92 | 370.68 |
| Monosaccharide composition (mol) | |||
| Man | 1 | 1 | 1 |
| Rib | - | 0.40 | - |
| GlcA | 1.22 | 0.45 | 1.50 |
| Glc | 0.10 | 0.32 | - |
| Gal | 0.58 | - | 1.39 |
| Xyl | 0.21 | ||
| Fuc | 2.90 | 3.44 | 8.32 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, L.; Wang, Y.; Yang, H.; Li, H.; Xu, W.; Chen, G.; Zhu, H. Physicochemical Characterization, Antioxidant and Immunostimulatory Activities of Sulfated Polysaccharides Extracted from Ascophyllum nodosum. Molecules 2018, 23, 1912. https://doi.org/10.3390/molecules23081912
Chen L, Wang Y, Yang H, Li H, Xu W, Chen G, Zhu H. Physicochemical Characterization, Antioxidant and Immunostimulatory Activities of Sulfated Polysaccharides Extracted from Ascophyllum nodosum. Molecules. 2018; 23(8):1912. https://doi.org/10.3390/molecules23081912
Chicago/Turabian StyleChen, Ligen, Yan Wang, Hui Yang, Han Li, Wei Xu, Guijie Chen, and Hongjun Zhu. 2018. "Physicochemical Characterization, Antioxidant and Immunostimulatory Activities of Sulfated Polysaccharides Extracted from Ascophyllum nodosum" Molecules 23, no. 8: 1912. https://doi.org/10.3390/molecules23081912
APA StyleChen, L., Wang, Y., Yang, H., Li, H., Xu, W., Chen, G., & Zhu, H. (2018). Physicochemical Characterization, Antioxidant and Immunostimulatory Activities of Sulfated Polysaccharides Extracted from Ascophyllum nodosum. Molecules, 23(8), 1912. https://doi.org/10.3390/molecules23081912






