New Sample Preparation Method for Honey Volatiles Fingerprinting Based on Dehydration Homogeneous Liquid–Liquid Extraction (DHLLE)
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Samples
2.2. Dehydration Homogenous Liquid–Liquid Extraction Method
2.3. Dehydration Homogenous Liquid–Liquid Extraction Method Evaluation
2.4. Ultrasonic Solvent Extraction
2.5. Chromatographic Conditions
3. Results and Discussion
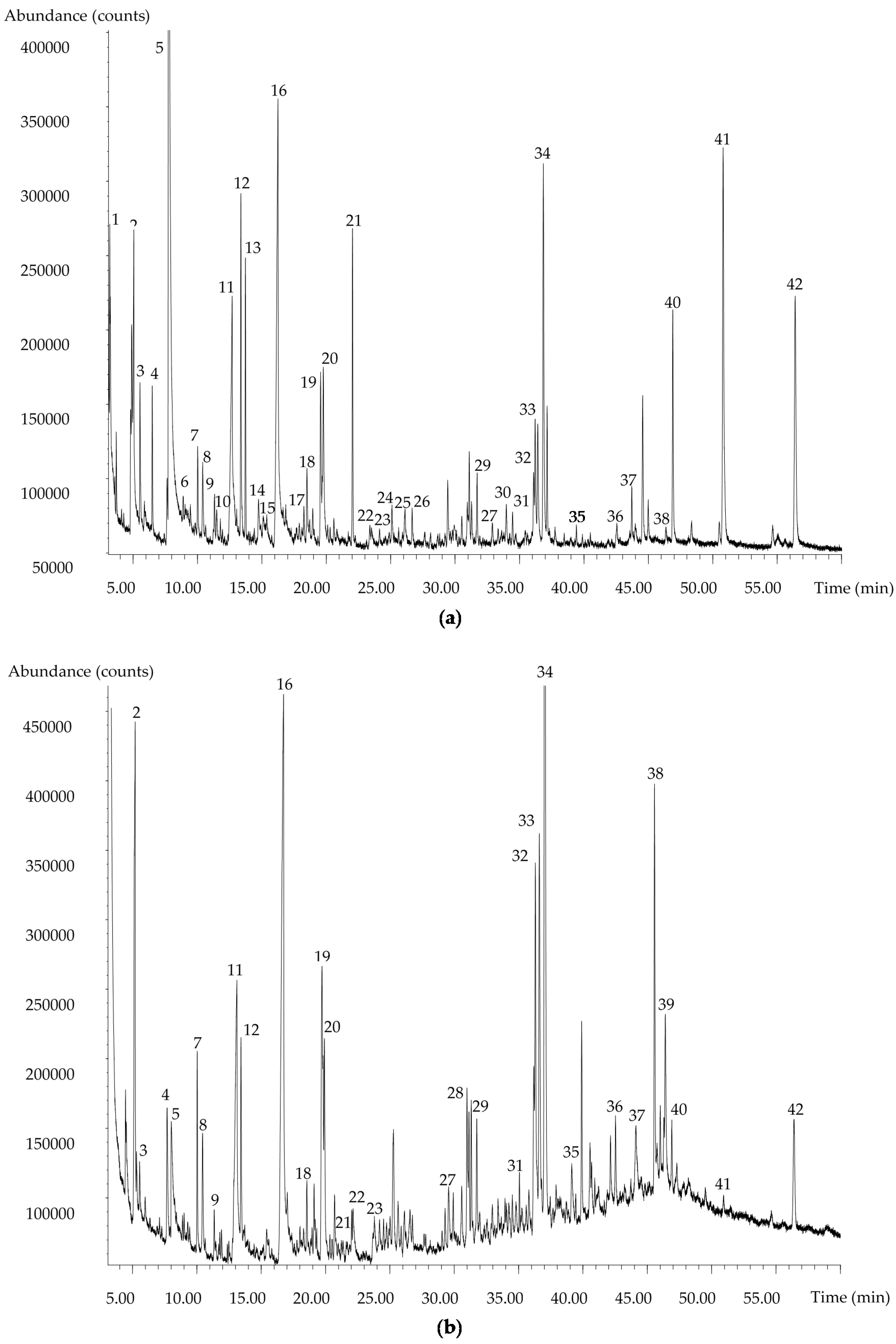
3.1. Performance of the Extraction Method
3.2. Comparison with Literature Data on Ultrasonic Solvent Extraction and Solid-Phase Extraction Methods
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- European Comission. Report from the Commission to the European Parliament and the Council on the Implementation of the Measures Concerning the Apiculture Sector of Regulation (EU) No 1308/2013 of the European Parliament and of the Council; European Comission: Brussels, Belgium, 2016. [Google Scholar]
- Mayer, A.; Slezak, V.; Takac, P.; Olejnik, J.; Majtan, J. Treatment of non-healing leg ulcers with honeydew honey. J. Tissue Viability 2014, 23, 94–97. [Google Scholar] [CrossRef] [PubMed]
- Kuś, P.M.; Szweda, P.; Jerković, I.; Tuberoso, C.I.G. Activity of Polish unifloral honeys against pathogenic bacteria and its correlation with colour, phenolic content, antioxidant capacity and other parameters. Lett. Appl. Microbiol. 2016, 62, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Schramm, D.D.; Karim, M.; Schrader, H.R.; Holt, R.R.; Cardetti, M.; Keen, C.L. Honey with high levels of antioxidants can provide protection to healthy human subjects. J. Agric. Food Chem. 2003, 51, 1732–1735. [Google Scholar] [CrossRef] [PubMed]
- Gheldof, N.; Wang, X.-H.; Engeseth, N.J. Buckwheat honey increases serum antioxidant capacity in humans. J. Agric. Food Chem. 2003, 51, 1500–1505. [Google Scholar] [CrossRef] [PubMed]
- Council of the European Union. Council Directive 2001/110/EC of 20 December 2001 Relating to Honey; European Comission: Brussels, Belgium, 2002. [Google Scholar]
- Aries, E.; Burton, J.; Carrasco, L.; De Rudder, O.; Maquet, A. Scientific Support to the Implementation of a Coordinated Control Plan with a View to Establishing the Prevalence of Fraudulent Practices in the Marketing of Honey; JRC Technical Report JRC104749; European Comission: Brussels, Belgium, 2016. [Google Scholar]
- Kaškonienė, V.; Venskutonis, P.R. Floral markers in honey of various botanical and geographic origins: A review. Compr. Rev. Food Sci. Food Saf. 2010, 9, 620–634. [Google Scholar] [CrossRef]
- Jerković, I. Volatile Benzene Derivatives as Honey Biomarkers. Synlett 2013, 24, 2331–2334. [Google Scholar] [CrossRef]
- Alissandrakis, E.; Tarantilis, P.A.; Harizanis, P.C.; Polissiou, M. Evaluation of four isolation techniques for honey aroma compounds. J. Sci. Food Agric. 2005, 85, 91–97. [Google Scholar] [CrossRef]
- Castro-Vázquez, L.; Pérez-Coello, M.S.; Cabezudo, M.D. Analysis of volatile compounds of rosemary honey. Comparison of different extraction techniques. Chromatographia 2003, 57, 227–233. [Google Scholar] [CrossRef]
- Castro-Vázquez, L.; Díaz-Maroto, M.C.; Guchu, E.; Pérez-Coello, M.S. Analysis of volatile compounds of eucalyptus honey by solid phase extraction followed by gas chromatography coupled to mass spectrometry. Eur. Food Res. Technol. 2006, 224, 27–31. [Google Scholar] [CrossRef]
- Armenta, S.; Garrigues, S.; de la Guardia, M. The role of green extraction techniques in Green Analytical Chemistry. TrAC, Trends Anal. Chem. 2015, 71, 2–8. [Google Scholar] [CrossRef]
- Alissandrakis, E.; Daferera, D.; Tarantilis, P.A.; Polissiou, M.; Harizanis, P.C. Ultrasound-assisted extraction of volatile compounds from citrus flowers and citrus honey. Food Chem. 2003, 82, 575–582. [Google Scholar] [CrossRef]
- Jerković, I.; Kuś, P.M.; Tuberoso, C.I.G.; Šarolić, M. Phytochemical and physical-chemical analysis of Polish willow (Salix spp.) honey: Identification of the marker compounds. Food Chem. 2014, 145, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Prat, D.; Wells, A.; Hayler, J.; Sneddon, H.; McElroy, C.R.; Abou-Shehada, S.; Dunn, P.J. CHEM21 selection guide of classical- and less classical-solvents. Green Chem. 2016, 18, 288–296. [Google Scholar] [CrossRef]
- Anastassiades, M.; Lehotay, S.J.; Stajnbaher, D.; Schenck, F. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “Dispersive Solid-Phase Extraction” for the determination of pesticide residues in Produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [PubMed]
- Angioni, A.; Pintore, G.A.M.; Caboni, P. Determination of wine aroma compounds by dehydration followed by GC/MS. J. AOAC Int. 2012, 95, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Jerković, I.; Kuś, P.M. Terpenes in honey: Occurrence, origin and their role as chemical biomarkers. RSC Adv. 2014, 4, 31710–31728. [Google Scholar] [CrossRef]
- Jerković, I.; Tuberoso, C.I.G.; Kuś, P.M.; Marijanović, Z.; Kranjac, M. Screening of Coffea spp. honey by different methodologies: Theobromine and caffeine as chemical markers. RSC Adv. 2014, 4, 60557–60562. [Google Scholar] [CrossRef]
- Kuś, P.M.; Jerković, I.; Marijanović, Z.; Tuberoso, C.I.G. Screening of Polish Fir Honeydew Honey Using GC/MS, HPLC-DAD, and Physical-Chemical Parameters: Benzene Derivatives and Terpenes as Chemical Markers. Chem. Biodivers. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Jerković, I.; Kranjac, M.; Šuste, M.; Kuś, P.M.; Svečnjak, L. Rhamnus frangula Honey: Screening of Volatile Organic Compounds and Their Composition after Short-Term Heating. Chem. Nat. Compd. 2015, 51. [Google Scholar] [CrossRef]
- Jerković, I.; Marijanović, Z.; Ljubičić, I.; Gugić, M. Contribution of the bees and combs to honey volatiles: Blank-trial probe for chemical profiling of honey biodiversity. Chem. Biodivers. 2010, 7, 1217–1230. [Google Scholar] [CrossRef] [PubMed]
- Hristea, E.N.; Covaci-Cîmpeanu, I.C.; Ionita, G.; Ionita, P.; Draghici, C.; Caproiu, M.T.; Hillebrand, M.; Constantinescu, T.; Balaban, A.T. Reactions of 2, 2-diphenyl-1-picrylhydrazyl ( DPPH ) with two syringylic phenols or one aroxide derivative. Eur. J. Org. Chem. 2009, 626–634. [Google Scholar] [CrossRef]
- Kuś, P.M.; Jerković, I.; Tuberoso, C.I.G.; Marijanović, Z.; Šarolić, M. GC-MS Fingerprints and other physico-chemical characteristics of rare unifloral Prunus cerasus L. honey. Nat. Prod. Commun. 2013, 8, 651–654. [Google Scholar]
- Jerković, I.; Prđun, S.; Marijanović, Z.; Zekić, M.; Bubalo, D.; Svečnjak, L.; Tuberoso, C.I.G. Traceability of Satsuma mandarin (Citrus unshiu Marc.) honey through nectar/honey-sac/honey pathways of the headspace, volatiles, and semi-volatiles: Chemical markers. Molecules 2016, 21, 1302. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, A.M. The Pherobase: Database of Pheromones and Semiochemicals. Available online: http://www.pherobase.com/ (accessed on 10 January 2018).
- Adams, R.P. Identification of Essential oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Pub Corp: Carol Stream, IL, USA, 2007; ISBN 1581152833. [Google Scholar]
- Byrne, F.P.; Jin, S.; Paggiola, G.; Petchey, T.H.M.; Clark, J.H.; Farmer, T.J.; Hunt, A.J.; McElroy, R.C.; Sherwood, J. Tools and techniques for solvent selection: Green solvent selection guides. Sustain. Chem. Process. 2016, 4, 1–24. [Google Scholar] [CrossRef]
- Constable, D.J.C.; Jimenez-Gonzalez, C.; Henderson, R.K. Perspective on Solvent Use in the Pharmaceutical Industry. Org. Process Res. Dev. 2007, 11, 133–137. [Google Scholar] [CrossRef]
- Snyder, L.R. Classification of the solvent properties of common liquids. J. Chromatogr. 1974, 92, 223–230. [Google Scholar] [CrossRef]
- Barton, A.F.M. (Ed.) IUPAC Solubility Data Series: Alcohol with Water; Pergamon: New York, NY, USA, 1984; Volume 15, ISBN 0080239277. [Google Scholar]
- European Commission Directoriate General for Health and Food Safety. Guidance Document on Analytical Quality Control and Method Validation Procedures for Pesticide Residues Analysis in Food and Feed; SANTE/11813/2017; European Commission: Brussels, Belgium, 2017. [Google Scholar]
- Kuś, P.M.; Jerković, I.; Tuberoso, C.I.G.; Šarolić, M. The Volatile Profiles of a Rare Apple (Malus domestica Borkh.) Honey: Shikimic Acid-Pathway Derivatives, Terpenes, and Others. Chem. Biodivers. 2013, 10, 1638–1652. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Compound | RI 1 | Concentration Level A (n = 5) | Concentration Level B (n = 5) | ||||
|---|---|---|---|---|---|---|---|
| Recovery 2 (%) | SD | RSD% | Recovery 2 (%) | SD | RSD% | ||
| Benzyl alcohol | 1039 | 63.9 | 3.0 | 4.8 | 77.7 | 2.6 | 3.4 |
| Linalool | 1100 | 83.7 | 3.4 | 4.0 | 89.0 | 4.9 | 5.5 |
| Borneol | 1174 | 76.9 | 3.8 | 5.0 | 82.0 | 4.1 | 5.0 |
| Terpinen-4-ol | 1180 | 76.9 | 3.5 | 4.6 | 79.2 | 2.9 | 3.7 |
| α-Terpineol | 1193 | 85.7 | 4.7 | 5.5 | 85.9 | 2.1 | 2.4 |
| p-Anisaldehyde | 1256 | 91.7 | 4.8 | 5.3 | 93.5 | 5.3 | 5.6 |
| Eugenol | 1361 | 78.7 | 4.3 | 5.5 | 75.2 | 3.2 | 4.3 |
| Vanillin | 1403 | 87.6 | 0.7 | 0.8 | 77.9 | 3.8 | 4.9 |
| Methyl syringate | 1773 | 54.3 | 3.8 | 7.1 | 75.2 | 1.3 | 1.7 |
| Caffeine | 1843 | 57.0 | 5.1 | 8.9 | 67.3 | 3.7 | 5.5 |
| No. | Compound | RI 1 | DHLLE | USE | ||||
|---|---|---|---|---|---|---|---|---|
| Av. (Area %) 2 | ±SD | ±RSD% | Av. (Area %) 2 | ±SD | ±RSD% | |||
| 1 | 3-Methylbutanoic acid (Isovaleric acid) | <900 | 7.9 | 0.8 | 10.4 | nd | - | - |
| 2 | 3-Methylpentanoic acid | 942 | 3.9 | 0.2 | 5.2 | 8.1 | 0.5 | 6.3 |
| 3 | Benzaldehyde | 966 | 1.2 | 0.0 | 3.8 | 0.8 | 0.1 | 9.9 |
| 4 | Benzyl alcohol | 1039 | 0.6 | 0.0 | 4.9 | 2.1 | 0.1 | 4.3 |
| 5 | Vinyl caproate 3 | 1048 | 22.4 | 1.6 | 7.2 | 4.0 | 0.2 | 4.2 |
| 6 | Phenylacetaldehyde | 1049 | 0.5 | 0.0 | 6.0 | nd | - | - |
| 7 | Hotrienol | 1106 | 0.8 | 0.1 | 8.9 | 1.9 | 0.1 | 3.1 |
| 8 | 2-Phenylethanol | 1115 | 0.9 | 0.1 | 5.6 | 1.8 | 0.2 | 8.9 |
| 9 | 2-Phenylacetonitrile | 1140 | 0.4 | 0.0 | 6.5 | 0.7 | 0.1 | 20.2 |
| 10 | 2,3-Dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one | 1142 | 0.5 | 0.0 | 5.0 | nd | - | - |
| 11 | Benzoic acid | 1162 | 6.8 | 0.2 | 3.4 | 8.6 | 0.4 | 4.8 |
| 12 | 3,7-Dimethylocta-1,5-diene-3,7-diol (Terpendiol I) | 1191 | 3.4 | 0.1 | 1.7 | 2.3 | 0.3 | 13.9 |
| 13 | Dodecane | 1200 | 1.8 | 0.1 | 1.1 | nd | - | - |
| 14 | 2,3-Dihydrobenzofuran | 1222 | 0.5 | 0.1 | 10.6 | nd | - | - |
| 15 | HMF | 1230 | 0.5 | 0.0 | 6.3 | nd | - | - |
| 16 | Phenylacetic acid | 1251 | 13.8 | 1.3 | 9.2 | 18.2 | 0.5 | 2.7 |
| 17 | 1H-Indole | 1295 | 0.5 | 0.0 | 7.0 | nd | - | - |
| 18 | 4-Vinyl-2-methoxyphenol | 1314 | 0.6 | 0.1 | 11.0 | 0.8 | 0.0 | 3.7 |
| 19 | 3-Hydroxy-4-phenylbutan-2-one | 1352 | 2.1 | 0.1 | 2.4 | 1.0 | 0.1 | 8.9 |
| 20 | (E)-8-Hydroxylinalool | 1367 | 0.4 | 0.0 | 6.1 | 1.0 | 0.1 | 11.4 |
| 21 | Tetradecane | 1400 | 3.1 | 0.1 | 3.7 | 0.3 | 0.1 | 25.8 |
| 22 | 4-Hydroxy-3-methoxybenzaldehyde (Vanillin) | 1403 | 0.6 | 0.1 | 11.0 | 1.3 | 0.3 | 24.4 |
| 23 | trans-Cinnamic acid | 1434 | 1.9 | 0.1 | 6.9 | 1.5 | 0.4 | 26.6 |
| 24 | Dodecan-1-ol | 1479 | 0.8 | 0.1 | 5.9 | nd | - | - |
| 25 | p-Hydroxybenzoic acid | 1517 | 0.2 | 0.0 | 7.7 | nd | - | - |
| 26 | 4-Hydroxy-3-methoxybenzoic acid (Vanillic acid) | 1567 | 0.4 | 0.0 | 1.4 | nd | - | - |
| 27 | 5-Amino indanone | 1594 | 1.1 | 0.1 | 4.6 | 1.1 | 0.0 | 2.6 |
| 28 | Hexadecane | 1600 | nd | - | - | 0.8 | 0.1 | 7.1 |
| 29 | 3-Hydroxy-β-damascone | 1617 | 0.7 | 0.0 | 5.2 | 1.1 | 0.2 | 19.2 |
| 30 | 3-Hydroxy-4-phenyl-2(5H)-furanone | 1695 | 0.7 | 0.1 | 9.0 | nd | - | - |
| 31 | 6,7-Dehydro-7,8-dihydro-3-oxo-α-ionol | 1733 | 0.9 | 0.0 | 2.4 | 0.7 | 0.1 | 18.8 |
| 32 | 9-Hydroxymegastigma-4,6-dien-3-one | 1769 | 1.0 | 0.1 | 5.8 | 2.1 | 0.4 | 21.7 |
| 33 | Methyl syringate | 1773 | 2.7 | 0.1 | 5.2 | 5.4 | 1.2 | 22.5 |
| 34 | 4-Hydroxy-3,5,5-trimethyl-4-(3-oxo-1-butenyl)-2-cyclohexen-1-one (Vomifoliol) | 1803 | 4.8 | 0.2 | 3.7 | 17.5 | 0.7 | 4.0 |
| 35 | Hexadecan-1-ol | 1882 | 0.4 | 0.0 | 10.9 | 2.4 | 0.1 | 3.9 |
| 36 | Hexadecanoic acid | 1966 | 1.0 | 0.1 | 4.7 | 1.1 | 0.0 | 2.7 |
| 37 | Methyl indole-3-acetate | 1980 | 1.1 | 0.1 | 7.2 | 2.0 | 0.5 | 23.3 |
| 38 | (Z)-Octadec-9-en-1-ol | 2059 | 0.5 | 0.0 | 7.9 | 4.9 | 0.1 | 1.1 |
| 39 | Octadecan-1-ol | 2084 | nd | - | - | 1.0 | 0.0 | 4.2 |
| 40 | Heneicosane | 2100 | 1.2 | 0.0 | 2.7 | 0.8 | 0.0 | 4.7 |
| 41 | Octadecanoic acid | 2181 | 4.0 | 0.2 | 5.9 | 0.6 | 0.1 | 21.5 |
| 42 | Tricosane | 2300 | 2.6 | 0.3 | 9.5 | 1.6 | 0.1 | 4.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuś, P.M.; Jerković, I. New Sample Preparation Method for Honey Volatiles Fingerprinting Based on Dehydration Homogeneous Liquid–Liquid Extraction (DHLLE). Molecules 2018, 23, 1769. https://doi.org/10.3390/molecules23071769
Kuś PM, Jerković I. New Sample Preparation Method for Honey Volatiles Fingerprinting Based on Dehydration Homogeneous Liquid–Liquid Extraction (DHLLE). Molecules. 2018; 23(7):1769. https://doi.org/10.3390/molecules23071769
Chicago/Turabian StyleKuś, Piotr M., and Igor Jerković. 2018. "New Sample Preparation Method for Honey Volatiles Fingerprinting Based on Dehydration Homogeneous Liquid–Liquid Extraction (DHLLE)" Molecules 23, no. 7: 1769. https://doi.org/10.3390/molecules23071769
APA StyleKuś, P. M., & Jerković, I. (2018). New Sample Preparation Method for Honey Volatiles Fingerprinting Based on Dehydration Homogeneous Liquid–Liquid Extraction (DHLLE). Molecules, 23(7), 1769. https://doi.org/10.3390/molecules23071769






