Using Natural Biomacromolecules for Adsorptive and Enzymatic Removal of Aniline Blue from Water
Abstract
1. Introduction
2. Materials and Methods
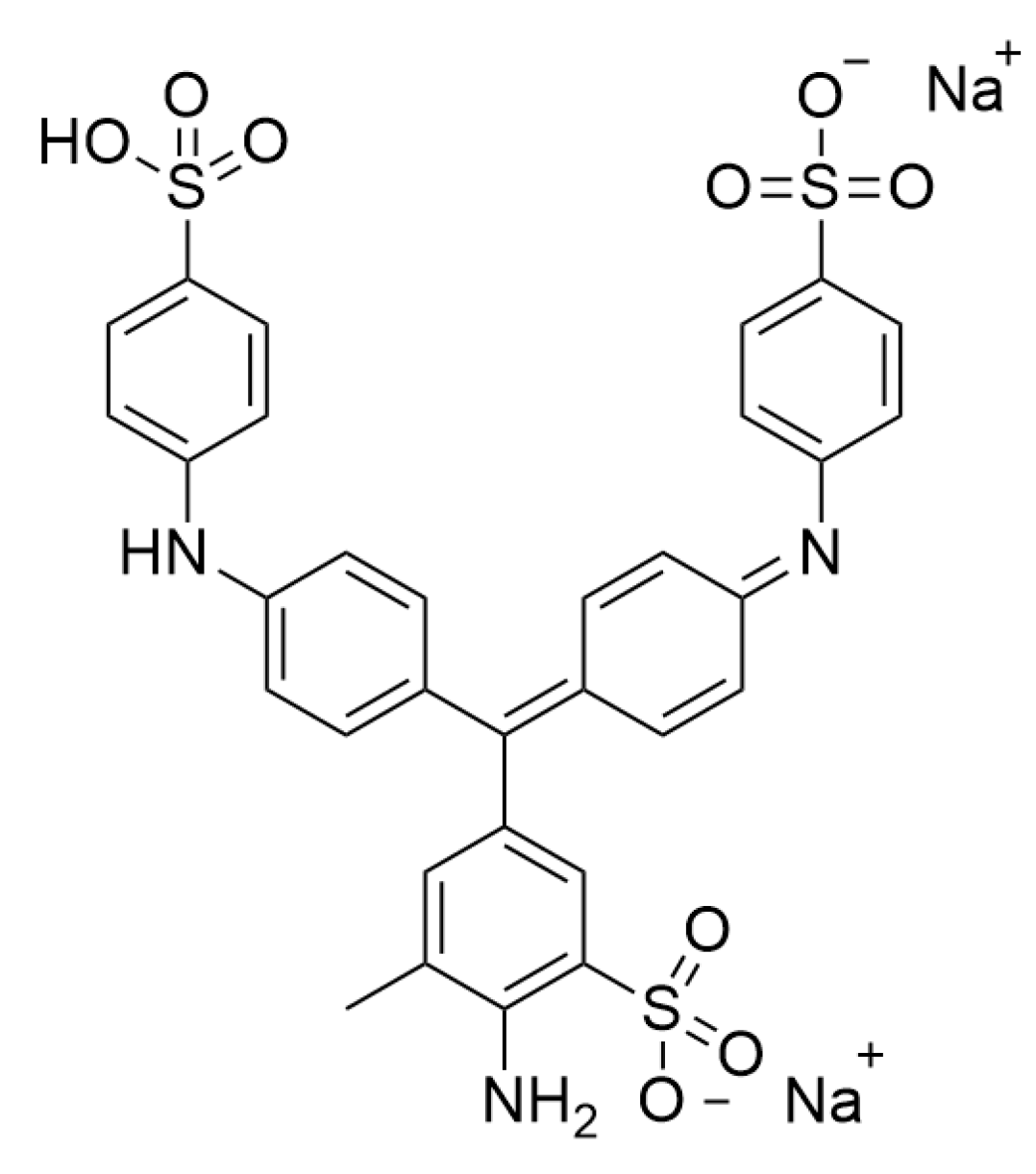
2.1. RW, PS, MW, AC, Lac, and Aniline Blue
2.2. Adsorption Experiments
2.3. The Effect of Influencing Factors on Dye Removal
2.4. Adsorption Kinetic Studies
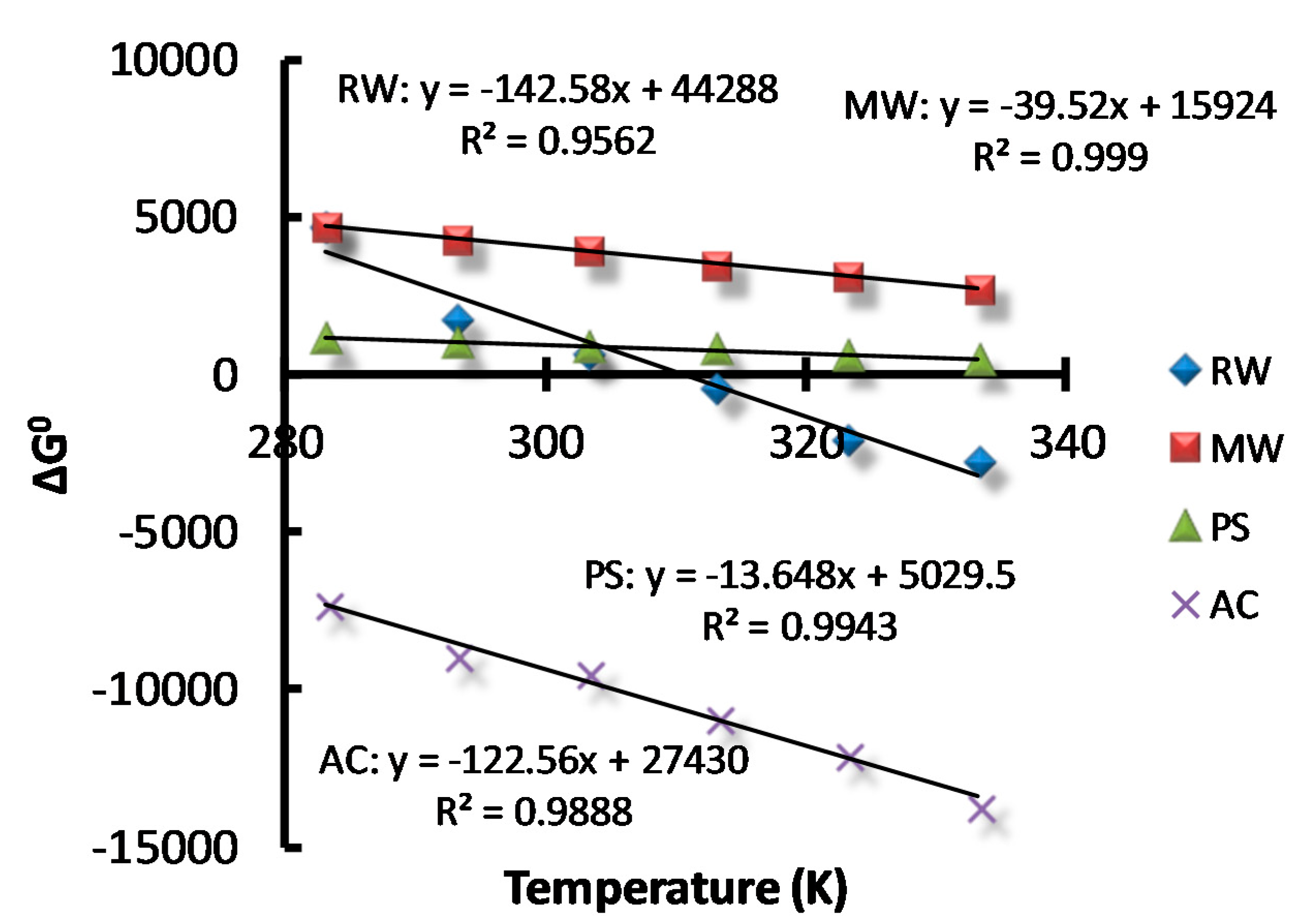
2.5. Adsorption Thermodynamics
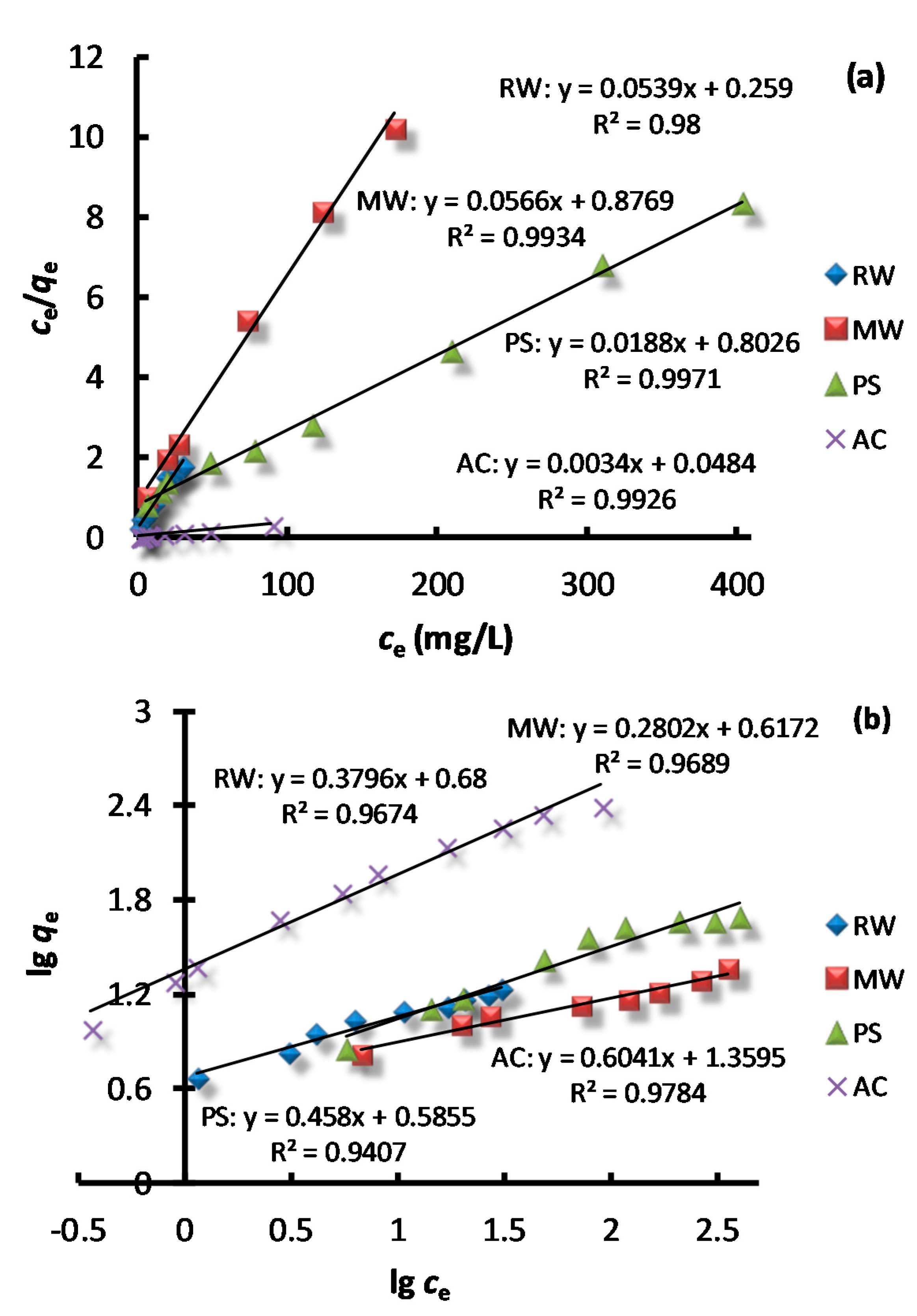
2.6. Adsorption Isotherms
3. Results and Discussions
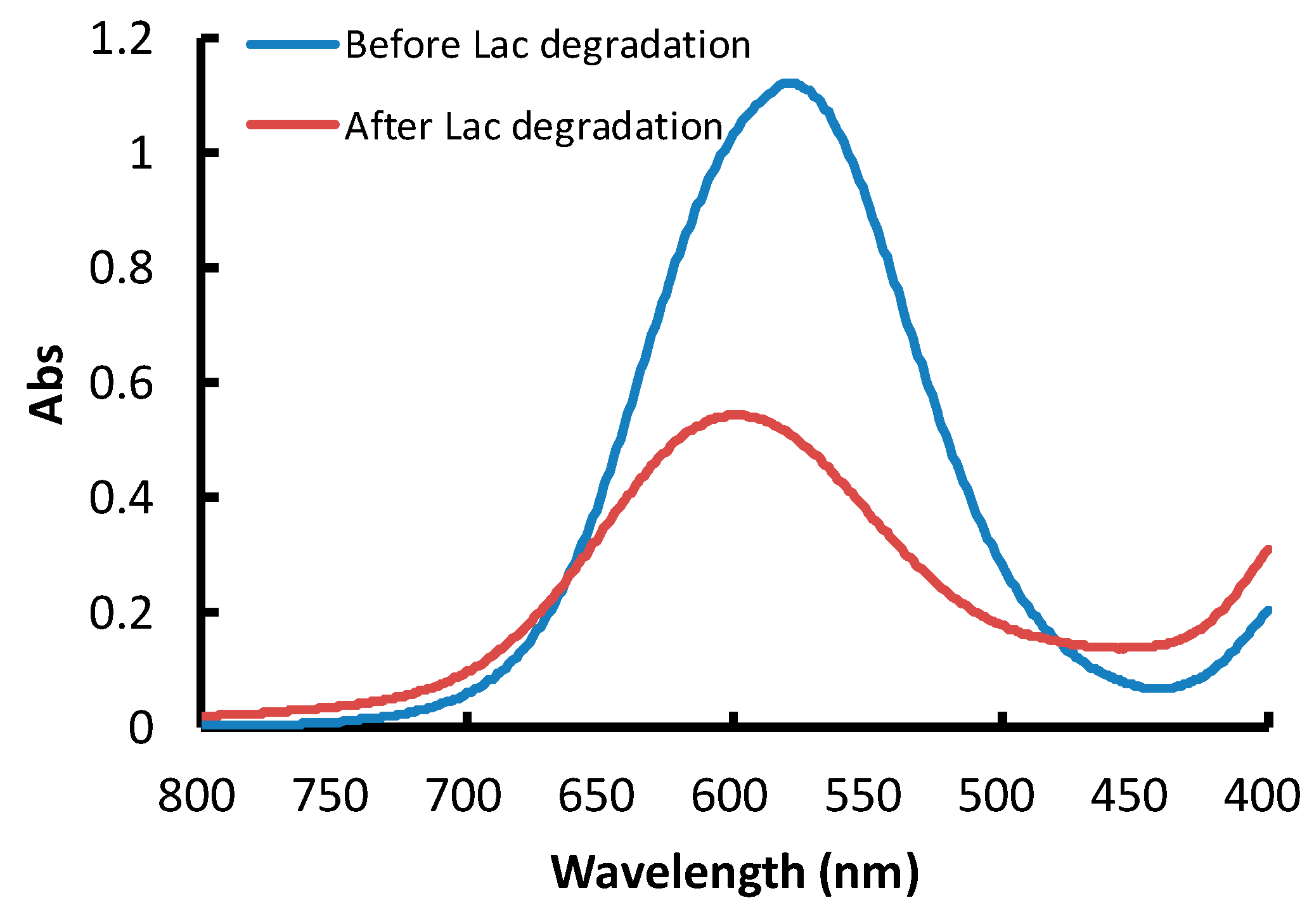
3.1. Spectrophotometric Calibration
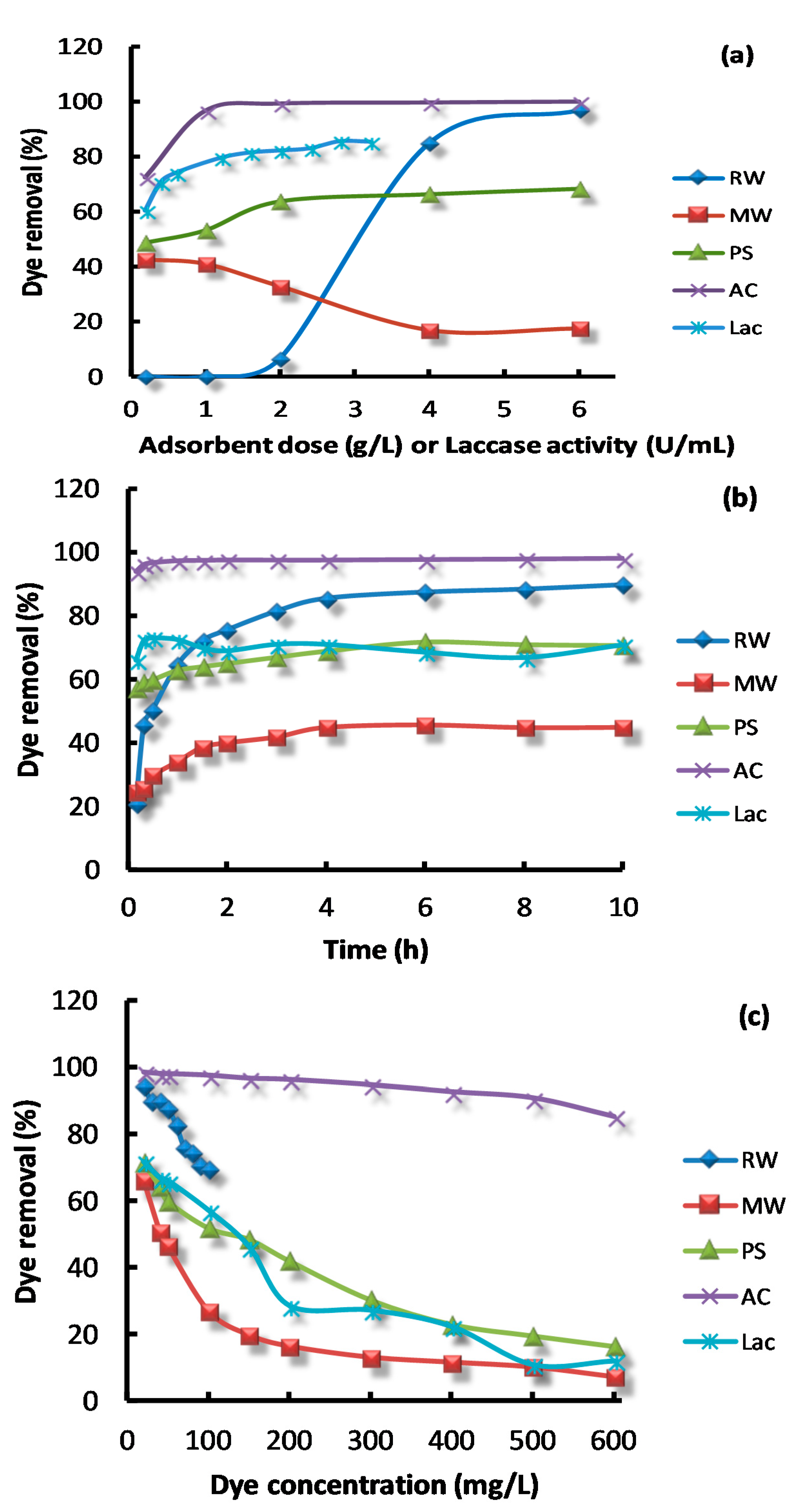
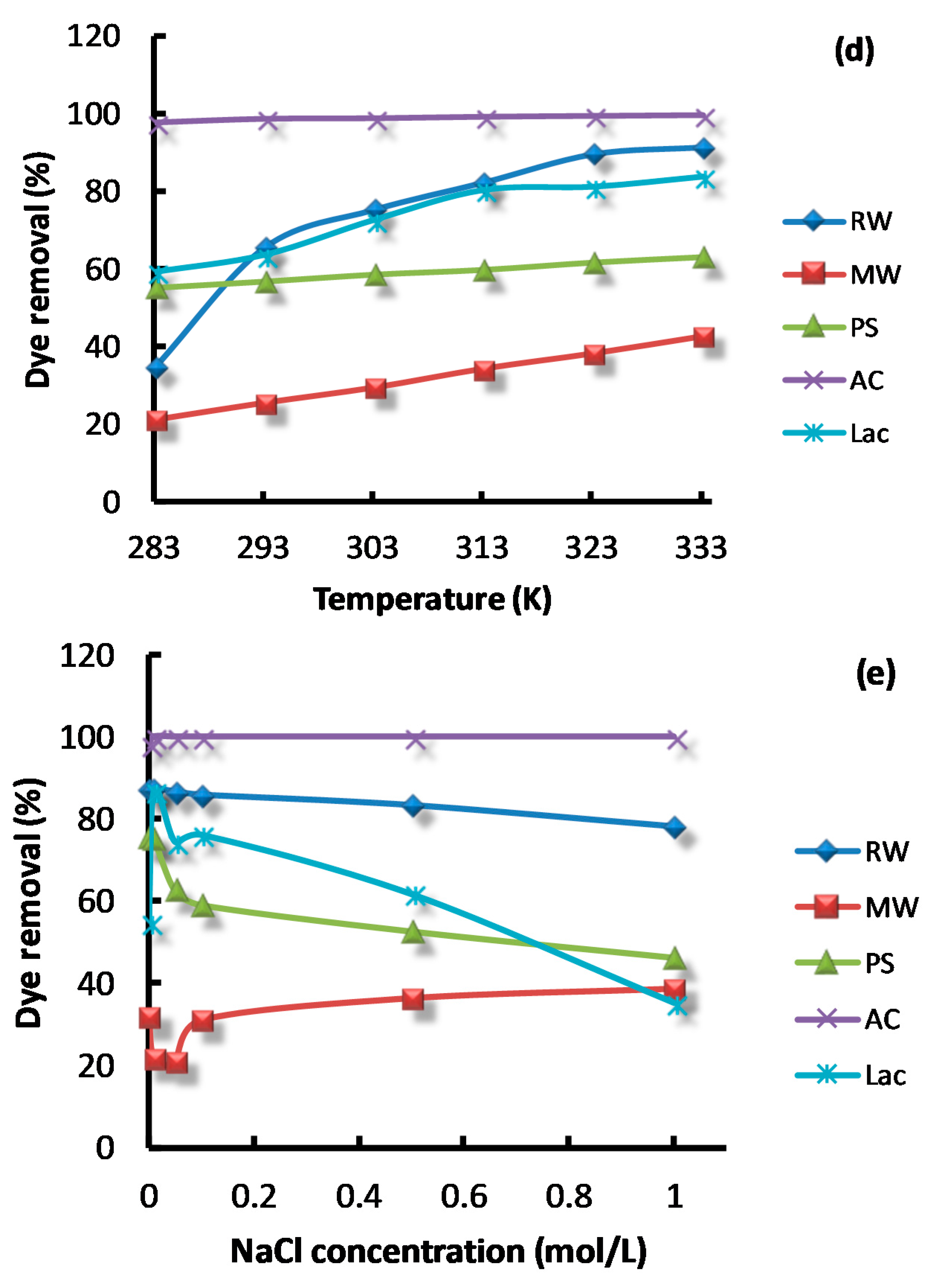
3.2. The Effect of Influencing Parameters on Dye Removal
3.3. Adsorption Kinetics
3.4. Adsorption Isotherms
3.5. Adsorption thermodynamics
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Afroze, S.; Sen, T.K.; Ang, M.; Nishioka, H. Adsorption of methylene blue dye from aqueous solution by novel biomass Eucalyptus sheathiana bark: Equilibrium, kinetics, thermodynamics and mechanism. Desalin. Water Treat. 2016, 57, 5858–5878. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Sillanpää, M.; Witek-Krowiak, A. Agricultural waste peels as versatile biomass for water purification—A review. Chem. Eng. J. 2015, 270, 244–271. [Google Scholar] [CrossRef]
- Daâssi, D.; Frikha, F.; Zouari-Mechichi, H.; Belbahri, L.; Woodward, S.; Mechichi, T. Application of response surface methodology to optimize decolourization of dyes by the laccase-mediator system. J. Environ. Manag. 2012, 108, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Danish, M.; Ahmad, T. A review on utilization of wood biomass as a sustainable precursor for activated carbon production and application. Renew. Sustain. Energy Rev. 2018, 87, 1–21. [Google Scholar] [CrossRef]
- Goh, P.S.; Ismail, A.F. A review on inorganic membranes for desalination and wastewater treatment. Desalination 2018, 434, 60–80. [Google Scholar] [CrossRef]
- Grandclément, C.; Seyssiecq, I.; Piram, A.; Wong-Wah-Chung, P.; Vanot, G.; Tiliacos, N.; Roche, N.; Doumenq, P. From the conventional biological wastewater treatment to hybrid processes, the evaluation of organic micropollutant removal: A review. Water Res. 2017, 111, 297–317. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Tamilarasan, R. Modeling studies: Adsorption of aniline blue by using Prosopis Juliflora carbon/Ca/alginate polymer composite beads. Carbohydr. Polym. 2013, 92, 2171–2180. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Cai, Y.; Liao, X.; Liaoa, X.; Huang, Q.; Hao, Z.; Hu, M.; Zhang, D. Purification and characterisation of a novel thermal stable laccase from Pycnoporus sp. SYBC-L3 and its use in dye decolorisation. Biol. Environ. Proc. R. Irish Acad. 2013, 113, 27–39. [Google Scholar] [CrossRef]
- Liu, J.; Li, E.; You, X.; Hu, C.; Huang, Q. Adsorption of methylene blue on an agro-waste oiltea shell with and without fungal treatment. Sci. Rep. 2016, 6, 38450. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, Z.; Li, H.; Hu, C.; Raymer, P.; Huang, Q. Effect of solid state fermentation of peanut shell on its dye adsorption performance. Bioresour. Technol. 2018, 249, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yu, Z.; Liao, X.; Liu, J.; Mao, F.; Huang, Q. Scalable production, fast purification, and spray drying of native Pycnoporus laccase and circular dichroism characterization. J. Clean. Prod. 2016, 127, 600–609. [Google Scholar] [CrossRef]
- Majeau, J.-A.; Brar, S.K.; Tyagi, R.D. Laccases for removal of recalcitrant and emerging pollutants. Bioresour. Technol. 2010, 101, 2331–2350. [Google Scholar] [CrossRef] [PubMed]
- Miklos, D.B.; Remy, C.; Jekel, M.; Linden, K.G.; Drewes, J.E.; Hübner, U. Evaluation of advanced oxidation processes for water and wastewater treatment—A critical review. Water Res. 2018, 139, 118–131. [Google Scholar] [CrossRef] [PubMed]
- Moawed, E.A. Effect of heating processes on Salvadora persica (Miswak) and its application for removal and determination of aniline blue from wastewater. J. Taibah Univ. Sci. 2013, 7, 26–34. [Google Scholar] [CrossRef]
- Moawed, E.A.; Abulkibash, A.B.; El-Shahat, M.F. Synthesis and characterization of iodo polyurethane foam and its application in removing of aniline blue and crystal violet from laundry wastewater. J. Taibah Univ. Sci. 2015, 9, 80–88. [Google Scholar] [CrossRef]
- Olivera, S.; Muralidhara, H.B.; Venkatesh, K.; Guna, V.K.; Gopalakrishna, K.; Kumar, K.Y. Potential applications of cellulose and chitosan nanoparticles/composites in wastewater treatment: A review. Carbohydr. Polym. 2016, 153, 600–618. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Zhang, J.; Esquivel-Elizondo, S.; Ma, L.; Wu, Y. Uncovering the flocculating potential of extracellular polymeric substances produced by periphytic biofilms. Bioresour. Technol. 2018, 248, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.B.; Vakili, M.; Horri, B.A.; Poh, P.E.; Abdullah, A.Z.; Salamatinia, B. Adsorption of dyes by nanomaterials: Recent developments and adsorption mechanisms. Separ. Purif. Technol. 2015, 150, 229–242. [Google Scholar] [CrossRef]
- Unuabonah, E.I.; Adebowale, K.O.; Dawodu, F.A. Equilibrium, kinetic and soe clay adsorbrber design studies on the adsorption of Aniline blue dye by sodium tetraborate-modified Kaolinitent. J. Hazard. Mater. 2008, 157, 397–409. [Google Scholar] [CrossRef] [PubMed]
- Wan Ngah, W.S.; Teong, L.C.; Hanafiah, M.A.K.M. Adsorption of dyes and heavy metal ions by chitosan composites: A review. Carbohydr. Polym. 2011, 83, 1446–1456. [Google Scholar] [CrossRef]
- Wong, S.; Ngadi, N.; Inuwa, I.M.; Hassan, O. Recent advances in applications of activated carbon from biowaste for wastewater treatment: A short review. J. Clean. Prod. 2018, 175, 361–375. [Google Scholar] [CrossRef]
- Yang, Y.; Wei, X.; Sun, P.; Wan, J. Preparation, characterization and adsorption performance of a novel anionic starch microsphere. Molecules 2010, 15, 2872–2885. [Google Scholar] [CrossRef] [PubMed]
- Zaaba, N.; Ismail, H.; Jaafar, M. Effect of Peanut Shell Powder Content on the Properties of Recycled Polypropylene (RPP)/Peanut Shell Powder (PSP) Composites. BioResources 2013, 8, 5826–5841. [Google Scholar] [CrossRef]
- Zhang, C.; Diao, H.; Lu, F.; Bie, X.; Wang, Y.; Lu, Z. Degradation of triphenylmethane dyes using a temperature and pH stable spore laccase from a novel strain of Bacillus vallismortis. Bioresour. Technol. 2012, 126, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Dong, X.; Yang, X.; Kattel, G.; Zhao, Y.; Wang, R. Ecological shift and resilience in China’s lake systems during the last two centuries. Glob. Planet. Chang. 2018, 165, 147–159. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, L.; Cheng, Z. Removal of organic pollutants from aqueous solution using agricultural wastes: A review. J. Mol. Liq. 2015, 212, 739–762. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |

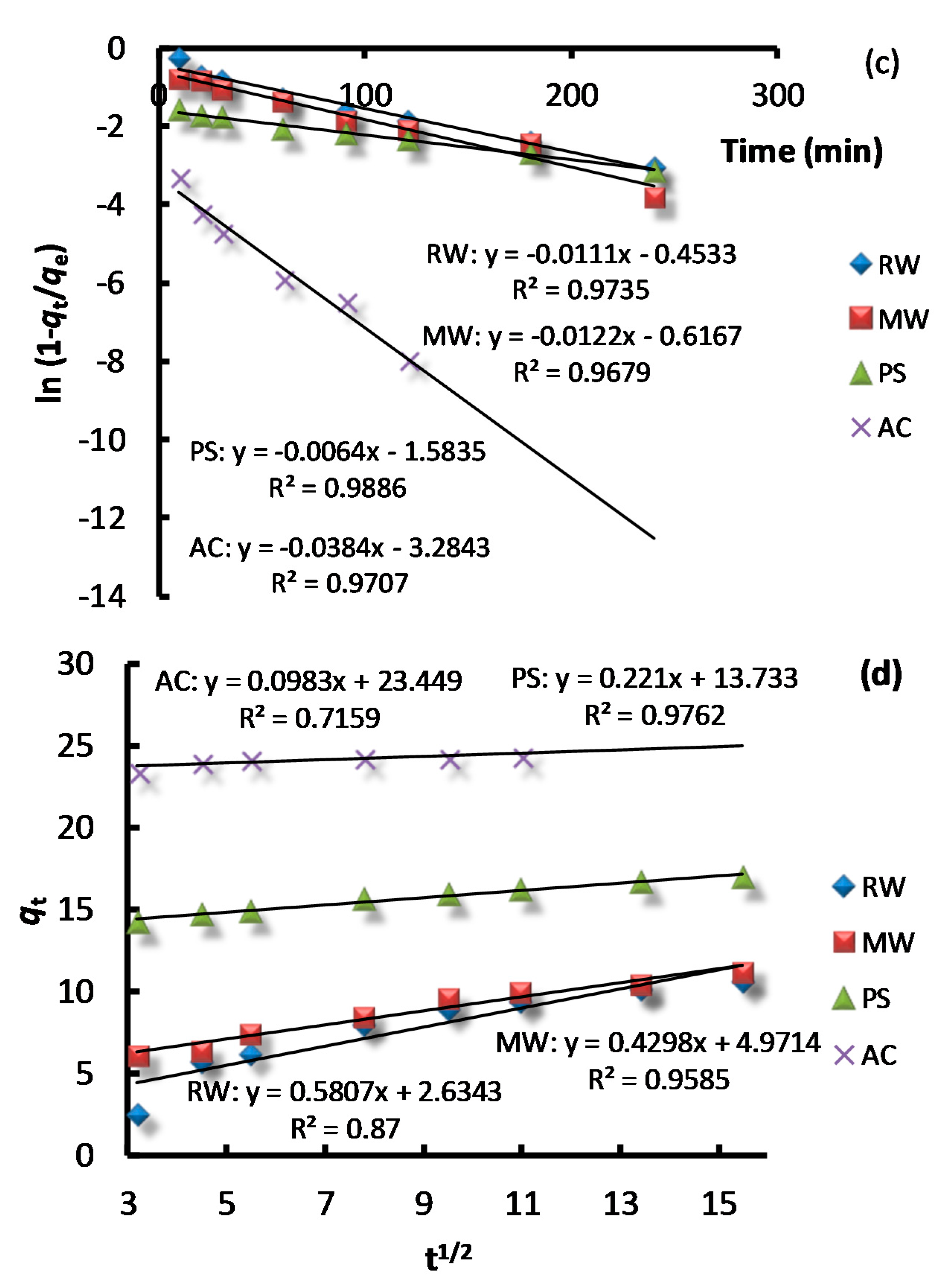
| Adsorbent | Pseudo First Order Kinetic Model | Pseudo Second Order Kinetic Model | Experimental | ||
|---|---|---|---|---|---|
| K1 (min−1) | qe(cal) (mg/g) | K2 (g/mg·min) | qe(cal) (mg/g) | qe(exp) (mg/g) | |
| RW | 0.001 | 7.12 | 0.003 | 11.88 | 11.08 |
| MW | 0.012 | 6.09 | 0.016 | 11.74 | 11.25 |
| PS | 0.007 | 4.03 | 0.014 | 17.21 | 17.91 |
| AC | 0.09 | 0.91 | 0.12 | 24.45 | 24.52 |
| Adsorbent | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| qm (mg/g) | KL | R2 | 1/n | KF | R2 | |
| RW | 18.55 | 0.21 | 0.98 | 0.38 | 4.79 | 0.967 |
| MW | 17.67 | 0.06 | 0.993 | 0.28 | 4.20 | 0.969 |
| PS | 52.91 | 0.023 | 0.997 | 0.46 | 3.89 | 0.941 |
| AC | 294.12 | 0.07 | 0.993 | 0.60 | 22.89 | 0.978 |
| Adsorbent | ΔH° (kJ·mol−1) | ΔS° (J·mol−1·K−1) |
|---|---|---|
| RW | 44.29 | 142.58 |
| MW | 15.92 | 39.52 |
| PS | 5.03 | 13.65 |
| AC | 27.43 | 122.56 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
You, X.; Li, E.; Liu, J.; Li, S. Using Natural Biomacromolecules for Adsorptive and Enzymatic Removal of Aniline Blue from Water. Molecules 2018, 23, 1606. https://doi.org/10.3390/molecules23071606
You X, Li E, Liu J, Li S. Using Natural Biomacromolecules for Adsorptive and Enzymatic Removal of Aniline Blue from Water. Molecules. 2018; 23(7):1606. https://doi.org/10.3390/molecules23071606
Chicago/Turabian StyleYou, Xiaojuan, Enzhong Li, Jiayang Liu, and Songhua Li. 2018. "Using Natural Biomacromolecules for Adsorptive and Enzymatic Removal of Aniline Blue from Water" Molecules 23, no. 7: 1606. https://doi.org/10.3390/molecules23071606
APA StyleYou, X., Li, E., Liu, J., & Li, S. (2018). Using Natural Biomacromolecules for Adsorptive and Enzymatic Removal of Aniline Blue from Water. Molecules, 23(7), 1606. https://doi.org/10.3390/molecules23071606





