Antifungal and Cytotoxic Activities of Sixty Commercially-Available Essential Oils
Abstract
1. Introduction
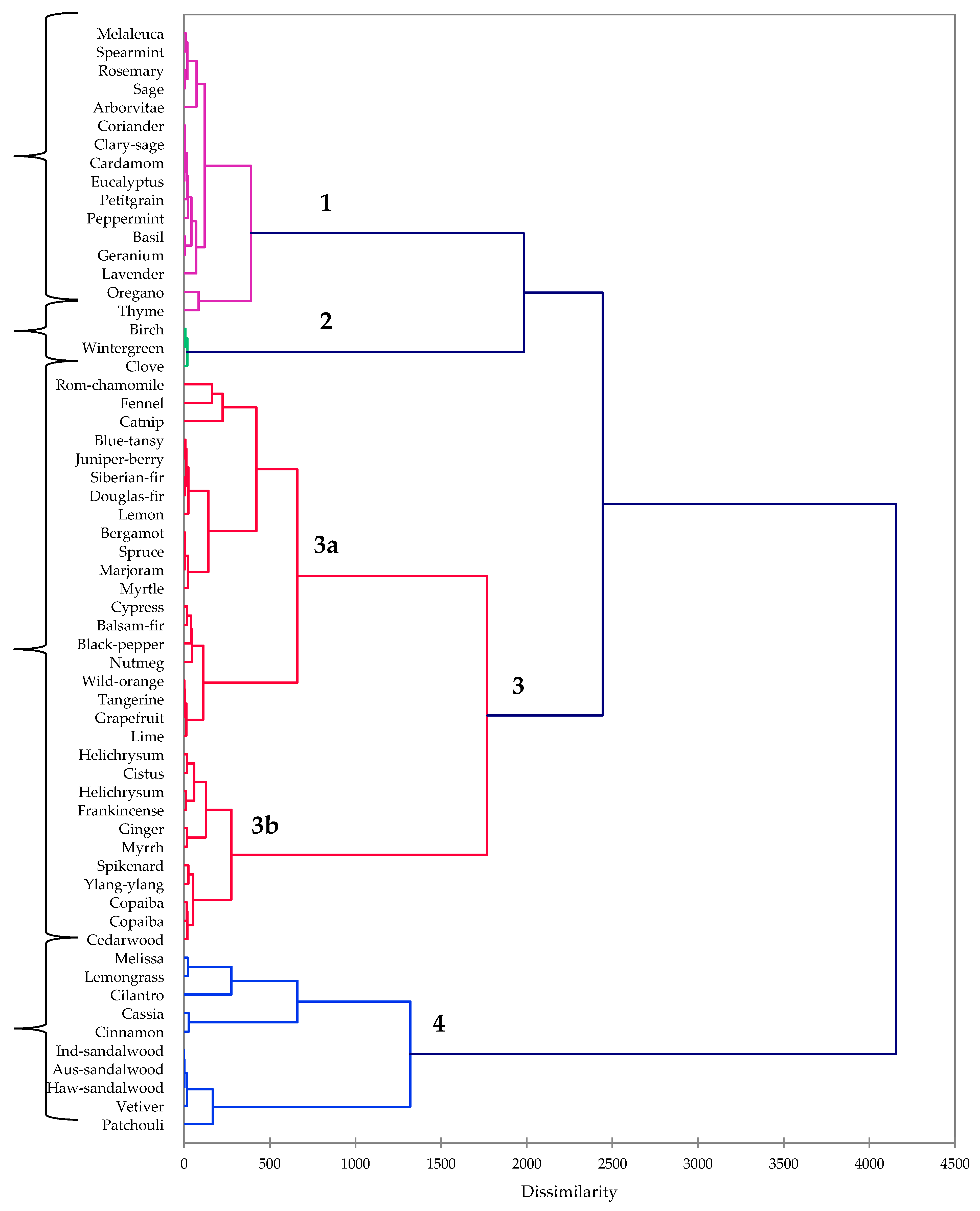
2. Results
3. Discussion
4. Materials and Methods
4.1. Essential Oils
4.2. Gas Chromatography-Mass Spectrometry
4.3. Antifungal Screening
4.4. Cytotoxicity Screening
4.5. Hierarchical Cluster Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hawksworth, D.L. The magnitude of fungal diversity: The 1.5 million species estimate revisited. Mycol. Res. 2001, 105, 1422–1432. [Google Scholar] [CrossRef]
- Horn, F.; Heinekamp, T.; Kniemeyer, O.; Pollmächer, J.; Valiante, V.; Brakhage, A.A. Systems biology of fungal infection. Front. Microbiol. 2012, 3, 108. [Google Scholar] [CrossRef] [PubMed]
- Enoch, D.A.; Ludlam, H.A.; Brown, N.M. Invasive fungal infections: A review of epidemiology and management options. J. Med. Microbiol. 2006, 55, 809–818. [Google Scholar] [CrossRef] [PubMed]
- Armstrong-James, D.; Meintjes, G.; Brown, G.D. A neglected epidemic: Fungal infections in HIV/AIDS. Trends Microbiol. 2014, 22, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Kosmidis, C.; Denning, D.W. The clinical spectrum of pulmonary aspergillosis. Thorax 2015, 70, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Antinori, S.; Milazzo, L.; Sollima, S.; Galli, M.; Corbellino, M. Candidemia and invasive candidiasis in adults: A narrative review. Eur. J. Intern. Med. 2016, 34, 21–28. [Google Scholar] [CrossRef] [PubMed]
- May, R.C.; Stone, N.R.H.; Wiesner, D.L.; Bicanic, T.; Nielsen, K. Cryptococcus: From environmental saprophyte to global pathogen. Nat. Rev. Microbiol. 2016, 14, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Girois, S.B.; Chapuis, F.; Decullier, E.; Revol, B.G.P. Adverse effects of antifungal therapies in invasive fungal infections: Review and meta-analysis. Eur. J. Clin. Microbiol. Infect. Dis. 2006, 25, 138–149. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A. Antifungal drug resistance: Mechanisms, epidemiology, and consequences for treatment. Am. J. Med. 2012, 125, S3–S13. [Google Scholar] [CrossRef] [PubMed]
- Başer, K.H.C.; Buchbauer, G. Handbook of Essential Oils: Science, Technology, and Applications; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Schmidt, J.M.; Noletto, J.A.; Vogler, B.; Setzer, W.N. Abaco bush medicine: Chemical composition of the essential oils of four aromatic medicinal plants from Abaco Island, Bahamas. J. Herbs Spices Med. Plants 2006, 12, 43–65. [Google Scholar] [CrossRef]
- Setzer, W.N.; Vogler, B.; Schmidt, J.M.; Leahy, J.G.; Rives, R. Antimicrobial activity of Artemisia douglasiana leaf essential oil. Fitoterapia 2004, 75, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Wright, B.S.; Bansal, A.; Moriarity, D.M.; Takaku, S.; Setzer, W.N. Cytotoxic leaf essential oils from Neotropical Lauraceae: Synergistic effects of essential oil components. Nat. Prod. Commun. 2007, 2, 1241–1244. [Google Scholar]
- Marei, G.I.K.; Abdel Rasoul, M.A.; Abdelgaleil, S.A.M. Comparative antifungal activities and biochemical effects of monoterpenes on plant pathogenic fungi. Pestic. Biochem. Physiol. 2012, 103, 56–61. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antifungal activity of the components of Melaleuca alternifolia (tea tree) oil. J. Appl. Microbiol. 2003, 95, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Aumeeruddy-Elalfi, Z.; Gurib-Fakim, A.; Mahomoodally, F. Antimicrobial, antibiotic potentiating activity and phytochemical profile of essential oils from exotic and endemic medicinal plants of Mauritius. Ind. Crops Prod. 2015, 71, 197–204. [Google Scholar] [CrossRef]
- Ahmad, A.; Khan, A.; Akhtar, F.; Yousuf, S.; Xess, I.; Khan, L.A.; Manzoor, N. Fungicidal activity of thymol and carvacrol by disrupting ergosterol biosynthesis and membrane integrity against Candida. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Marchese, A.; Orhan, I.E.; Daglia, M.; Barbieri, R.; Di Lorenzo, A.; Nabavi, S.F.; Gortzi, O.; Izadi, M.; Nabavi, S.M. Antibacterial and antifungal activities of thymol: A brief review of the literature. Food Chem. 2016, 210, 402–414. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Varoni, E.M.; Iriti, M.; Martorell, M.; Setzer, W.N.; Contreras, M.D.M.; Salehi, B.; Soltani-Nejad, A.; Rajabi, S.; Tajbakhsh, M.; et al. Carvacrol and human health: A comprehensive review. Phyther. Res. 2018, in press. [Google Scholar] [CrossRef] [PubMed]
- Satyal, P.; Murray, B.L.; McFeeters, R.L.; Setzer, W.N. Essential oil characterization of Thymus vulgaris from various geographical locations. Foods 2016, 5, 70. [Google Scholar] [CrossRef] [PubMed]
- Pawar, V.C.; Thaker, V.S. In vitro efficacy of 75 essential oils against Aspergillus niger. Mycoses 2006, 49, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, V.; Lal, P.; Pruthi, V. Effect of plant oils on Candida albicans. J. Microbiol. Immunol. Infect. 2010, 43, 447–451. [Google Scholar] [CrossRef]
- Gayoso, C.W.; Lima, E.O.; Oliveira, V.T.; Pereira, F.O.; Souza, E.L.; Lima, I.O.; Navarro, D.F. Sensitivity of fungi isolated from onychomycosis to Eugenia cariophyllata essential oil and eugenol. Fitoterapia 2005, 76, 247–249. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.-C.; Hsu, K.-P.; Ho, C.-L. Composition, in vitro cytotoxicity, and anti-mildew activities of the leaf essential oil of Machilus thunbergii from Taiwan. Nat. Prod. Commun. 2015, 10, 2013–2016. [Google Scholar] [PubMed]
- Yen, H.; Wang, S.-Y.; Wu, C.-C.; Lin, W.-Y.; Wu, T.-Y.; Chang, F.-R.; Wang, C.-K. Cytotoxicity, anti-platelet aggregation assay and chemical components analysis of thirty-eight kinds of essential oils. J. Food Drug Anal. 2012, 20, 478–483. [Google Scholar]
- Viollon, C.; Chaumont, J.-P. Antifungal properties of essential oils and their main components upon Cryptococcus neoformans. Mycopathologia 1994, 128, 151–152. [Google Scholar] [CrossRef] [PubMed]
- Tampieri, M.P.; Galuppi, R.; Macchioni, F.; Carelle, M.S.; Falcioni, L.; Cioni, P.L.; Morelli, I. The inhibition of Candida albicans by selected essential oils and their major components. Mycopathologia 2005, 159, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Jirovetz, L.; Buchbauer, G.; Denkova, Z.; Stoyanova, A.; Murgov, I.; Gearon, V.; Birkbeck, S.; Schmidt, E.; Geissler, M. Comparative study on the antimicrobial activities of different sandalwood essential oils of various origin. Flavour Fragr. J. 2006, 21, 465–468. [Google Scholar] [CrossRef]
- Ortiz, C.; Morales, L.; Sastre, M.; Haskins, W.E.; Matta, J. Cytotoxicity and genotoxicity assessment of sandalwood essential oil in human breast cell lines MCF-7 and MCF-10A. Evid.-Based Complement. Altern. Med. 2016, 2016, 3696232. [Google Scholar] [CrossRef] [PubMed]
- Santha, S.; Dwivedi, C. Anticancer effects of sandalwood (Santalum album). Anticancer Res. 2015, 35, 3137–3145. [Google Scholar] [PubMed]
- Santha, S.; Bommareddy, A.; Rule, B.; Guillermo, R.; Kaushik, R.S.; Young, A.; Dwivedi, C. Antineoplastic effects of α-santalol on estrogen receptor-positive and estrogen receptor-negative breast cancer cells through cell cycle arrest at G2/M phase and induction of apoptosis. PLoS ONE 2013, 8, e56982. [Google Scholar] [CrossRef]
- Bommareddy, A.; Crisamore, K.; Fillman, S.; Brozena, S.; Steigerwalt, J.; Landis, T.; Vanwert, A.L.; Dwivedi, C. Survivin down-regulation by α-santalol is not mediated through PI3K–AKT pathway in human breast cancer cells. Anticancer Res. 2015, 35, 5353–5357. [Google Scholar] [PubMed]
- Carmo, E.S.; Lima, E.D.O.; Souza, E.L.D.; Sousa, F.B.D. Effect of Cinnamomum zeylanicum Blume essential oil on the growth and morphogenesis of some potentially pathogenic Aspergillus species. Braz. J. Microbiol. 2008, 39, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Giordani, R.; Regli, P.; Kaloustian, J.; Portugal, H. Potentiation of antifungal activity of amphotericin B by essential oil from Cinnamomum cassia. Phyther. Res. 2006, 20, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Unlu, M.; Ergene, E.; Vardar, G.; Sivas, H.; Vural, N. Composition, antimicrobial activity and in vitro cytotoxicity of essential oil from Cinnamomum zeylanicum Blume (Lauraceae). Food Chem. Toxicol. 2010, 48, 3274–3280. [Google Scholar] [CrossRef] [PubMed]
- Bin Jantan, I.; Moharam, B.A.K.; Santhanam, J.; Abdul, B.; Moharam, K.; Santhanam, J.; Jamal, J.A. Correlation between chemical composition and antifungal activity of the essential oils of eight Cinnamomum species. Pharm. Biol. 2008, 46, 406–412. [Google Scholar] [CrossRef]
- Ferhout, H.; Bohatier, J.; Guillot, J.; Chalchat, J.C. Antifungal activity of selected essential oils, cinnamaldehyde and carvacrol against Malassezia furfur and Candida albicans. J. Essent. Oil Res. 1999, 11, 119–129. [Google Scholar] [CrossRef]
- Siegfried, S.A.; Schroeder, J.R. Toxicity of thieves oils to MCF-7 and MDA-MB-231 breast cancer cells. Am. J. Essent. Oils Nat. Prod. 2018, 6, 1–8. [Google Scholar]
- Kumari, P.; Mishra, R.; Arora, N.; Chatrath, A.; Gangwar, R.; Roy, P.; Prasad, R. Antifungal and anti-biofilm activity of essential oil active components against Cryptococcus neoformans and Cryptococcus laurentii. Front. Microbiol. 2017, 8, 2161. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.; Rao, Y.K.; Tzeng, Y.-M. Cytotoxic effect of trans-cinnamaldehyde from Cinnamomum osmophloeum leaves on human cancer cell lines. Int. J. Appl. Sci. Eng. 2004, 2, 136–147. [Google Scholar]
- Pattnaik, S.; Subramanyam, V.R.; Bapaji, M.; Kole, C.R. Antibacterial and antifungal activity of aromatic constituents of essential oils. Microbios 1997, 89, 39–46. [Google Scholar] [PubMed]
- Setzer, W.N.; Schmidt, J.M.; Eiter, L.C.; Haber, W.A. The leaf oil composition of Zanthoxylum fagara (L.) Sarg. from Monteverde, Costa Rica, and its biological activities. J. Essent. Oil Res. 2005, 17, 333–335. [Google Scholar] [CrossRef]
- O’Brien, P.J.; Siraki, A.G.; Shangari, N. Aldehyde sources, metabolism, molecular toxicity mechanisms, and possible effects on human health. Crit. Rev. Toxicol. 2005, 35, 609–662. [Google Scholar] [CrossRef] [PubMed]
- Sharopov, F.S.; Wink, M.; Khalifaev, D.R.; Zhang, H.; Dosoky, N.S.; Setzer, W.N. Composition and bioactivity of the essential oil of Melissa officinalis L. growing wild in Tajikistan. Int. J. Tradit. Nat. Med. 2013, 2, 86–96. [Google Scholar]
- Sharopov, F.S.; Valiev, A.K.; Satyal, P.; Setzer, W.N.; Wink, M. Chemical composition and anti-proliferative activity of the essential oil of Coriandrum sativum L. Am. J. Essent. Oils Nat. Prod. 2017, 5, 11–14. [Google Scholar]
- Kocevski, D.; Du, M.; Kan, J.; Jing, C.; Lačanin, I.; Pavlović, H. Antifungal effect of Allium tuberosum, Cinnamomum cassia, and Pogostemon cablin essential oils and their components against population of Aspergillus species. J. Food Sci. 2013, 78, M731–M737. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007. [Google Scholar]
- Satyal, P. Development of GC-MS Database of Essential Oil Components by the Analysis of Natural Essential Oils and Synthetic Compounds and Discovery of Biologically Active Novel Chemotypes in Essential Oils. Ph.D. Thesis, University of Alabama in Huntsville, Huntsville, AL, USA, 2015. [Google Scholar]
- Dosoky, N.S. Isolation and Identification of Bioactive Compounds from Conradina canescens Gray. Ph.D. Thesis, University of Alabama in Huntsville, Huntsville, AL, USA, 2015. [Google Scholar]
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
| Essential Oil | Source | Antifungal Activity (MIC, μg/mL) | Cytotoxicity (IC50, μg/mL, Standard Deviations in Parentheses) | Major Components (>5%) | |||||
|---|---|---|---|---|---|---|---|---|---|
| A. niger | C. albicans | C. neoformans | MCF-7 | MDA-MB-231 | Others | ||||
| Abies balsamea | Balsam fir | Ameo | 1250 | 625 | 313 | 50.5 (15.0) | 86.7 (7.4) | β-pinene (26.4%), δ-3-carene (18.3%), α-pinene (16.0%), sylvestrene (15.0%), bornyl acetate (9.7%), camphene (5.7%), | |
| Abies sibirica | Siberian fir | doTERRA | 625 | 625 | 156 | >100 | >100 | camphene (24.8%), bornyl acetate (21.1%), α-pinene (15.2%), δ-3-carene (14.6%), limonene (5.7%) | |
| Anthemis nobilis | Roman chamomile | doTERRA | 625 | 625 | 313 | >100 | >100 | α-pinene (15.5%), isobutyl angelate (12.6%), methallyl angelate (10.9%), 3-methylpentyl angelate (5.4%) | |
| Betula lenta | Birch | doTERRA | 625 | 625 | 625 | >100 | >100 | methyl salicylate (99.9%) | |
| Boswellia carteri | Frankincense | Ameo | 625 | 1250 | 313 | 39.8 (4.1) | 50.6 (1.0) | limonene (22.4%), β-caryophyllene (22.2%), p-cymene (10.0%), δ-cadinene (9.4%), α-copaene (4.8%) | |
| Cananga odorata | Ylang ylang | Ameo | 1250 | 625 | 78 | 36.8 (2.3) | 61.6 (4.4) | 50.4 (7.2) (Hs-578T) | germacrene D (25.0%), β-caryophyllene (15.8%), (E,E)-α-farnesene (11.0%), benzyl benzoate (8.5%), geranyl acetate (5.2%) |
| Cinnamomum cassia | Cassia | doTERRA | 78 | 78 | 20 | 14.0 (1.4) | 16.9 (1.0) | 16.4 (0.9) (Hep-G2) | (E)-cinnamaldehyde (79.9%), (E)-o-methoxycinnamaldehyde (12.0%) |
| Cinnamomum zeylanicum | Cinnamon | doTERRA | 78 | 78 | 20 | 13.3 (1.6) | 24.2 (1.5) | 25.2 (2.2) (Hep-G2) | (E)-cinnamaldehyde (63.9%), eugenol (7.0%), (E)-cinnamyl acetate (5.1%) |
| Cistus ladanifer | Cistus | Albert Vielle | 625 | 625 | 156 | 36.6 (3.0) | 71.1 (5.3) | 46.3 (4.0) (Hs-578T) | α-pinene (20.8%), viridiflorene (10.9%),bornyl acetate (6.3%), viridoflorol (5.2%) |
| Citrus aurantifolia | Lime | doTERRA | 625 | 625 | 313 | 67.4 (5.9) | 40.3 (7.0) | limonene (51.9%), β-pinene (18.8%), γ-terpinene (8.1%) | |
| Citrus aurantium | Petitgrain | doTERRA | 625 | 625 | 313 | >100 | >100 | linalyl acetate (51.5%), linalool (25.4%) | |
| Citrus bergamia | Bergamot | Ameo | 625 | 625 | 313 | >100 | >100 | limonene (34.6%), linalyl acetate (34.3%), linalool (12.7%), γ-terpinene (6.6%), β-pinene (5.6%) | |
| Citrus limon | Lemon | doTERRA | 625 | 625 | 313 | 94.8 (8.1) | >100 | limonene (56.1%), β-pinene (15.8%), γ-terpinene (10.5%) | |
| Citrus reticulata | Tangerine | doTERRA | 625 | 625 | 156 | 99.8 (10.0) | 54.8 (10.7) | limonene (91.3%) | |
| Citrus sinensis | Wild orange | doTERRA | 625 | 625 | 156 | 87.4 (3.0) | 50.4 (11.0) | limonene (94.8%) | |
| Citrus × paradisi | Grapefruit | doTERRA | 313 | 625 | 78 | 79.7 (3.6) | 50.6 (8.7) | limonene (91.3%) | |
| Commiphora myrrha | Myrrh | Ameo | 625 | 1250 | 313 | >100 | 86.4 (8.5) | furanoeudesma-1,3-diene (18.1%), curzerene (16.1%), lindestrene (6.9%), α-pinene (6.8%), neryl acetate (6.3%) | |
| Copaifera officinalis | Copaiba | Ameo | 1250 | 1250 | 313 | 22.7 (1.5) | 67.2 (2.2) | β-caryophyllene (87.3%) | |
| Copaifera spp. | Copaiba | doTERRA | 625 | 1250 | 625 | 60.4 (1.9) | 59.8 (6.1) | β-caryophyllene (50.0%), trans-α-bergamotene (8.5%), α-copaene (6.8%), α-humulene (6.0%) | |
| Coriandrum sativum | Cilantro | doTERRA | 313 | 313 | 20 | 42.8 (2.3) | 43.1 (3.9) | linalool (29.8%), (2E)-decenal (25.9%), (2E)-decen-1-ol (10.6%), n-decanal (7.9%) | |
| Coriandrum sativum | Coriander | doTERRA | 625 | 1250 | 625 | 98.6 (4.4) | >100 | linalool (73.5%), α-pinene (5.3%) | |
| Cupressus sempervirens | Cypress | Ameo | 1250 | 625 | 313 | 34.5 (2.6) | 65.2 (1.5) | α-pinene (49.7%), δ-3-carene (27.0%) | |
| Cymbopogon flexuosus | Lemongrass | doTERRA | 313 | 313 | 78 | 23.1 (1.4) | 30.7 (2.1) | geranial (49.9%), neral (23.4%), geraniol (7.6%), geranyl acetate (6.4%) | |
| Elettaria cardamomum | Cardamom | doTERRA | 625 | 625 | 156 | >100 | >100 | α-terpinyl acetate (37.2%), 1,8-cineole (35.3%), linalyl acetate (5.0%) | |
| Eucalyptus radiata | Eucalyptus | doTERRA | 313 | 625 | 156 | >100 | >100 | 1,8-cineole (78.8%), α-terpineol (8.6%) | |
| Eugenia caryophyllata | Clove | doTERRA | 156 | 313 | 156 | >100 | >100 | eugenol (80.6%), eugenyl acetate (10.5%), β-caryophyllene (6.5%) | |
| Foeniculum vulgare | Fennel | doTERRA | 625 | 625 | 313 | 95.9 (2.6) | >100 | (E)-anethole (75.1%), limonene (11.5%), fenchone (6.5%) | |
| Gualtheria fragrantissima | Wintergreen | doTERRA | 625 | 625 | 625 | >100 | >100 | methyl salicylate (99.7%) | |
| Helichrysum italicum | Helichrysum | Ameo | 1250 | 625 | 313 | 44.8 (1.4) | 39.5 (5.7) | neryl acetate (18.3%), α-pinene (18.0%), γ-curcumene (11.6%), β-selinene (10.3%), β-caryophyllene (6.1%), italicene (5.5%), valencene (5.1%) | |
| Helichrysum italicum | Helichrysum | doTERRA | 625 | 625 | 313 | 81.8 (10.0) | >100 | neryl acetate (33.9%), γ-curcumene (14.7%), α-pinene (13.4%) | |
| Juniperus communis | Juniper berry | Ameo | 625 | 1250 | 625 | >100 | >100 | α-pinene (34.9%), myrcene (11.9%), sabinene (11.4%), β-pinene (7.9%), β-caryophyllene (5.1%) | |
| Juniperus virginiana | Cedarwood | doTERRA | 625 | 625 | 313 | 37.2 (2.2) | 35.7 (1.8) | α-cedrene (41.4%), cis-thujopsene (20.0%), cedrol (13.4%), β-cedrene (7.5%) | |
| Lavandula angustifolia | Lavender | Ameo | 625 | 625 | 156 | 94.7 (4.7) | 60.3 (17.3) | linalyl acetate (41.5%), linalool (34.4%) | |
| Melaleuca alternifolia | Melaleuca | doTERRA | 625 | 625 | 625 | >100 | >100 | terpinen-4-ol (47.5%), γ-terpinene (20.2%), α-terpinene (8.6%) | |
| Melissa officinalis | Melissa | doTERRA | 313 | 313 | 78 | 32.4 (2.5) | 28.1 (1.5) | geranial (30.2%), neral (23.1%), β-caryophyllene (13.4%) | |
| Mentha piperita | Peppermint | doTERRA | 625 | 625 | 313 | >100 | >100 | menthol (43.8%), menthone (19.7%), menthyl acetate (6.5%), 1,8-cineole (5.0%) | |
| Mentha spicata | Spearmint | doTERRA | 313 | 625 | 313 | >100 | >100 | carvone (62.3%), limonene (20.1%) | |
| Myristica fragrans | Nutmeg | Ameo | 625 | 625 | 156 | 43.4 (0.3) | 32.6 (1.3) | sabinene (18.8%), myristicin (18.2%), α-pinene (17.1%), β-pinene (11.4%), sylvestrene (5.6%) | |
| Myrtis communis | Myrtle | Ameo | 1250 | 313 | 78 | >100 | >100 | α-pinene (46.1%), 1,8-cineole (27.5%), limonene (9.1%) | |
| Nardostachys jatamansi | Spikenard | doTERRA | 625 | 313 | 156 | 35.5 (2.2) | 65.2 (3.2) | viridiflorene (9.5%), 6,9-guaiadiene (8.8%), valeranone (7.8%), nardosina-7,9,11-triene (6.9%), β-gurjunene (6.7%), valerana-7,11-diene (6.2%), nardol (6.0%) | |
| Nepeta cataria | Catnip | Mountain Rose | 313 | 625 | 156 | >100 | >100 | 4aα,7α,7aβ-nepetalactone (58.1%), 4aα,7α,7aα- nepetalactone (20.6%), β-caryophyllene (6.8%) | |
| Ocimum basilicum | Basil | doTERRA | 313 | 625 | 313 | >100 | >100 | linalool (55.7%), 1,8-cineole (9.8%), trans-α-bergamotene (5.6%) | |
| Origanum majorana | Marjoram | doTERRA | 625 | 625 | 313 | >100 | >100 | terpinen-4-ol (28.9%), γ-terpinene (14.9%), trans-sabinene hydrate (9.5%), α-terpinene (8.7%), sabinene (7.2%) | |
| Origanum vulgare | Oregano | doTERRA | 156 | 313 | 78 | 35.3 (1.4) | 60.1 (17.3) | carvacrol (74.2%), γ-terpinene (5.2%) | |
| Pelargonium graveolens | Geranium | Ameo | 625 | 625 | 625 | >100 | >100 | citronellol (36.6%), iso-menthone (5.9%), geraniol (5.5%) | |
| Picea mariana | Spruce | Ameo | 625 | 625 | 313 | >100 | >100 | bornyl acetate (35.9%), camphene (14.5%), α-pinene (14.4%), δ-3-carene (8.2%) | |
| Piper nigrum | Black pepper | doTERRA | 625 | 1250 | 313 | 87.7 (4.1) | 74.0 (3.0) | β-caryophyllene (21.6%), limonene (15.1%), β-pinene (15.1%), sabinene (13.9%), α-pinene (11.1%), δ-3-carene (10.4%) | |
| Pogostemon cablin | Patchouli | Ameo | 156 | 625 | 20 | 25.0 (5.2) | 47.4 (1.1) | 22.6 (4.1) (Hep-G2) | patchouli alcohol (36.4%), α-bulnesene (16.3%), α-guaiene (12.4%), seychellene (8.7%), α-patchoulene (5.6%) |
| Pseudotsuga menziesii | Douglas fir | doTERRA | 625 | 313 | 156 | >100 | >100 | β-pinene (23.0%), sabinene (17.3%), terpinolene (13.5%), δ-3-carene (9.6%), α-pinene (8.1%) | |
| Rosmarinus officinalis | Rosemary | doTERRA | 625 | 625 | 313 | >100 | >100 | 1,8-cineole (45.9%), α-pinene (12.0%), camphor (10.9%), β-pinene (6.3%) | |
| Salvia officinalis | Sage | Mountain Rose | 1250 | 625 | 625 | >100 | >100 | cis-thujone (27.4%), camphor (21.4%), 1,8-cineole (11.9%), camphene (5.3%), α-pinene (5.2%) | |
| Salvia sclarea | Clary sage | Ameo | 1250 | 1250 | 313 | 98.4 (3.6) | >100 | linalyl acetate (69.0%) | |
| Santalum album | Indian sandalwood | doTERRA | 313 | 625 | 20 | 9.39 (1.34) | 19.3 (0.2) | 14.2 (1.6) (Hep-G2) | (Z)-α-santalol (45.2%), (Z)-β-santalol (25.4%), (Z)-α-trans-bergamotol (7.8%) |
| Santalum austrocaledonicum | Australian sandalwood | Ameo | 313 | 625 | 20 | 9.52 (0.08) | 20.4 (1.0) | 22.2 (1.4) (Hep-G2) | (Z)-α-santalol (49.2%), (Z)-β-santalol (23.9%), (Z)-lanceol (6.4%) |
| Santalum paniculatum | Hawaiian sandalwood | doTERRA | 156 | 625 | 20 | 13.3 (2.4) | 23.7 (2.1) | 29.6 (1.7) (Hep-G2) | (Z)-α-santalol (49.9%), (Z)-β-santalol (15.9%), (Z)-lanceol (6.6%), (Z)-α-trans-bergamotol (5.1%) |
| Tanacetum annuum | Blue tansy | doTERRA | 625 | 625 | 156 | >100 | >100 | sabinene (21.5%), myrcene (14.3%), camphor (12.0%), α-phellandrene (7.4%), p-cymene (5.8%), chamazulene (5.0%) | |
| Thuja plicata | Arborvitae | doTERRA | 313 | 78 | 78 | 89.0 (6.3) | >100 | methyl thujate (51.2%), methyl myrtenate (6.6%) | |
| Thymus vulgaris | Thyme | doTERRA | 156 | 313 | 78 | >100 | >100 | thymol (43.9%), carvacrol (14.4%), p-cymene (10.5%), β-caryophyllene (7.0%), γ-terpinene (5.1%) | |
| Vetiveria zizanoides | Vetiver | doTERRA | 78 | 313 | 20 | 23.9 (1.1) | 36.2 (0.8) | 20.2 (4.4) (Hep-G2) | (E)-isovalencenol (13.5%), khusimol (12.1%), α-vetivone (5.4%) |
| Zingiber officinale | Ginger | doTERRA | 625 | 625 | 313 | >100 | 81.5 (5.9) | α-zingiberene (26.4%), camphene (12.6%), β-sesquiphellandrene (9.2%), ar-curcumene (6.5%), β-phellandrene (6.2%), β-bisabolene (5.1%) | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Powers, C.N.; Osier, J.L.; McFeeters, R.L.; Brazell, C.B.; Olsen, E.L.; Moriarity, D.M.; Satyal, P.; Setzer, W.N. Antifungal and Cytotoxic Activities of Sixty Commercially-Available Essential Oils. Molecules 2018, 23, 1549. https://doi.org/10.3390/molecules23071549
Powers CN, Osier JL, McFeeters RL, Brazell CB, Olsen EL, Moriarity DM, Satyal P, Setzer WN. Antifungal and Cytotoxic Activities of Sixty Commercially-Available Essential Oils. Molecules. 2018; 23(7):1549. https://doi.org/10.3390/molecules23071549
Chicago/Turabian StylePowers, Chelsea N., Jessica L. Osier, Robert L. McFeeters, Carolyn Brianne Brazell, Emily L. Olsen, Debra M. Moriarity, Prabodh Satyal, and William N. Setzer. 2018. "Antifungal and Cytotoxic Activities of Sixty Commercially-Available Essential Oils" Molecules 23, no. 7: 1549. https://doi.org/10.3390/molecules23071549
APA StylePowers, C. N., Osier, J. L., McFeeters, R. L., Brazell, C. B., Olsen, E. L., Moriarity, D. M., Satyal, P., & Setzer, W. N. (2018). Antifungal and Cytotoxic Activities of Sixty Commercially-Available Essential Oils. Molecules, 23(7), 1549. https://doi.org/10.3390/molecules23071549









