Aspergoterpenins A–D: Four New Antimicrobial Bisabolane Sesquiterpenoid Derivatives from an Endophytic Fungus Aspergillus versicolor
Abstract
:1. Introduction
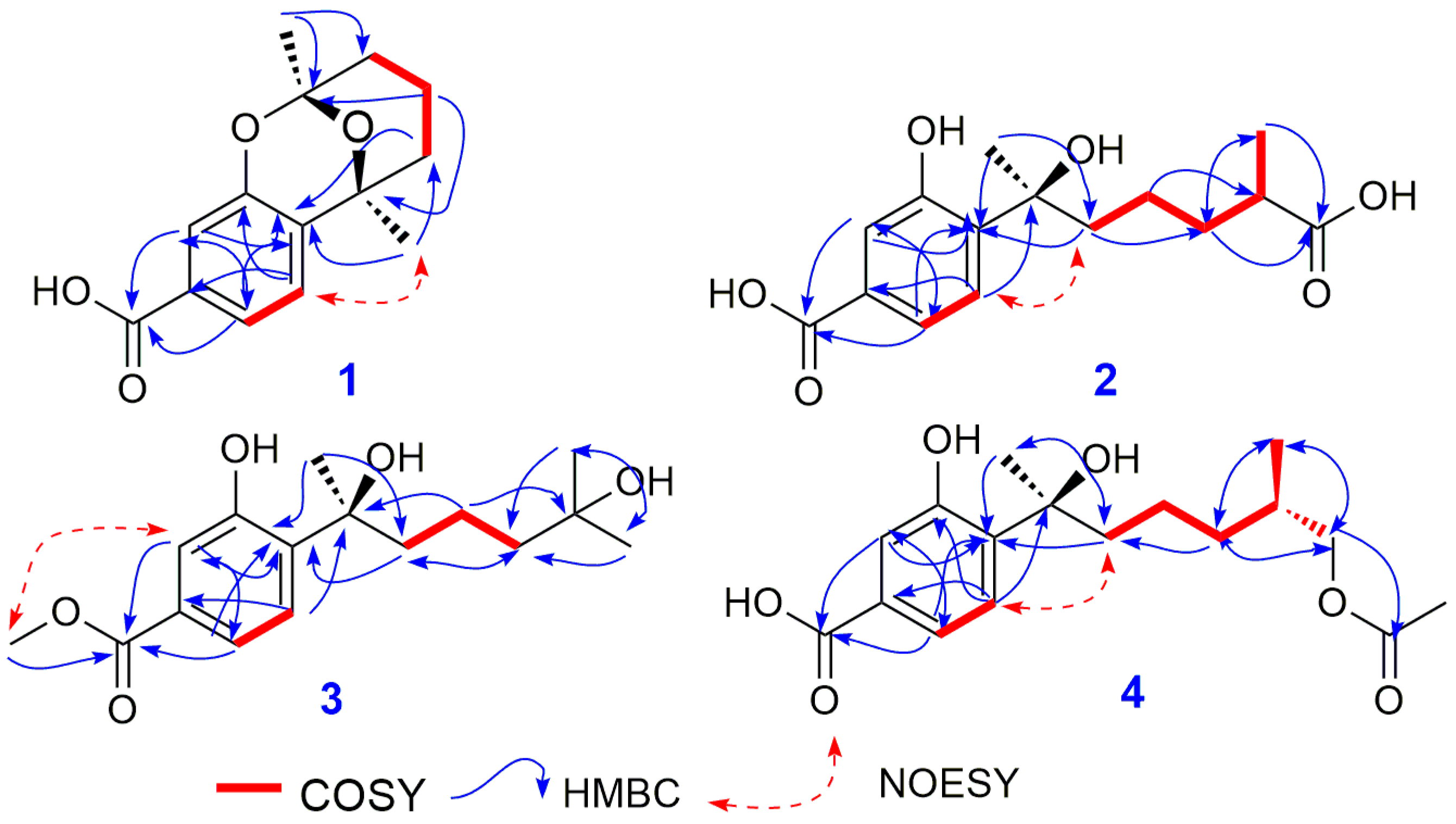
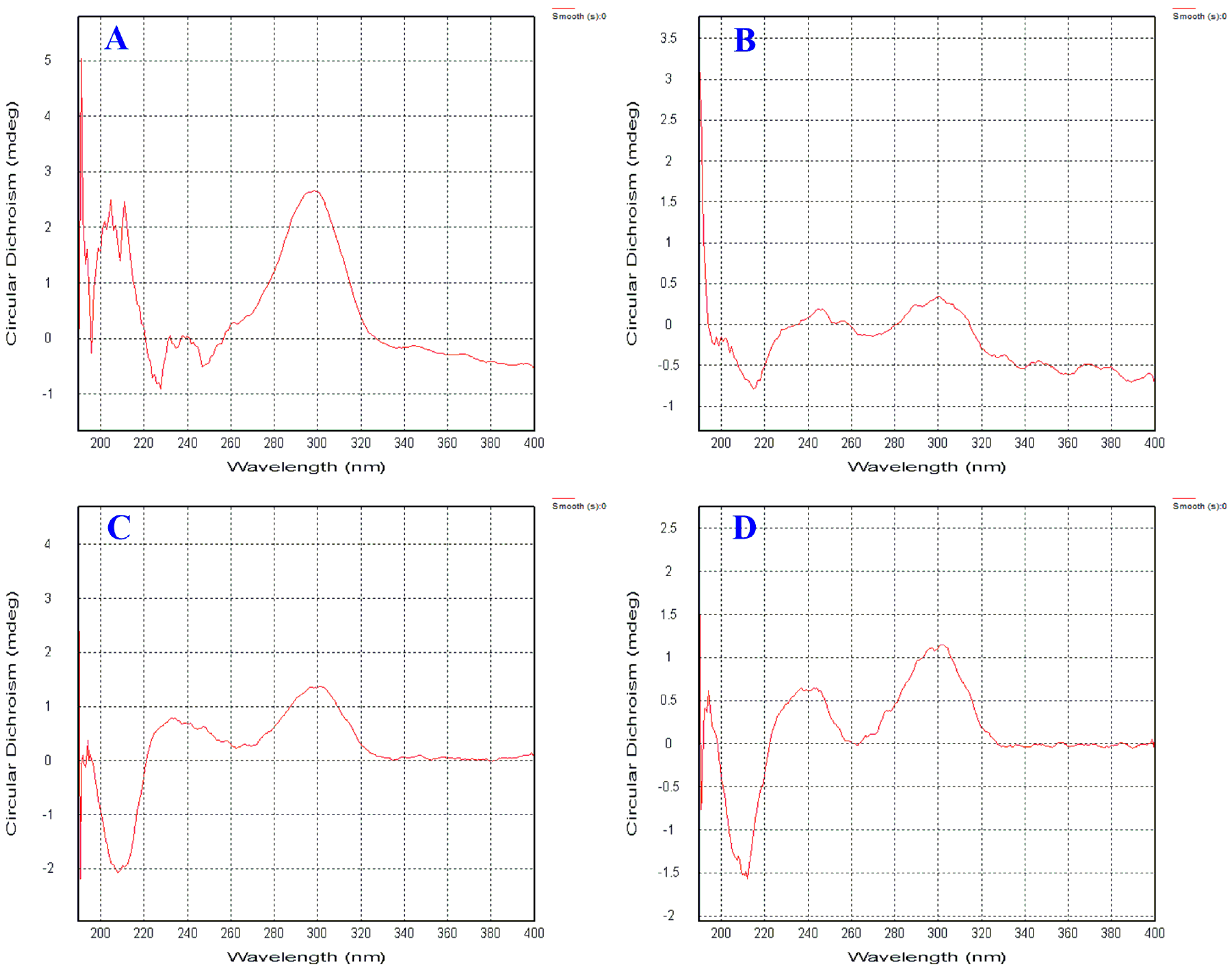
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Isolation and Identification of the Endophytic Fungus
3.3. Extraction and Isolation of Compounds
3.4. Computational Methods
3.5. Biological Activity Assay
3.5.1. Cytotoxic Activity to Four Cancer Cells In Vitro
3.5.2. Antimicrobial Activity
3.6. Spectroscopic Data of the Compounds
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Wu, Z.Y.; Chen, X.Q. The Flora of China Plants, 1st ed.; Science Publication: Beijing, China, 1979; p. 96. [Google Scholar]
- Xiu, J.; Ji, G.; Wang, M.; Yang, Y.; Li, C. Molecular identification and genetic diversity in Konnyaku’s soft rot bacteria. Acta Microbiol. Sin. 2006, 46, 522–525. [Google Scholar]
- Ding, Z.; Wan, Z.; Jiao, Z.; Zhou, R.; Tan, X.; Zhang, K.; Wu, J. Progress and Countermeasures for Soft Rot Disease of Amorphophallus konjac. Chin. Agric. Sci. Bull. 2014, 30, 238–241. [Google Scholar]
- Chua, M.; Baldwin, T.C.; Hocking, T.J.; Chan, K. Traditional uses and potential health benefits of Amorphophallus konjac K. Koch ex N.E.Br. J. Ethnopharmacol. 2010, 128, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Jiao, P.; Mudur, S.V.; Gloer, J.B.; Wicklow, D.T. Kipukasins, Nucleoside Derivatives from Aspergillus versicolor. J. Nat. Prod. 2007, 70, 1308–1311. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, J.; Park, J.; Ghisalberti, E.L.; Sivasithamparam, K.; Skelton, B.W.; White, A.H. New Chlorinated Diphenyl Ethers from an Aspergillus Species. J. Nat. Prod. 2002, 65, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.; You, M.; Chung, B.K.; Oh, D.C.; Oh, K.B.; Shin, J. Alkaloidal Metabolites from a Marine-Derived Aspergillus sp. Fungus. J. Nat. Prod. 2015, 78, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Reategui, R.; Rhea, J.; Adolphson, J.; Waikins, K.; Newell, R.; Rabenstein, J.; Mocek, U.; Luche, M.; Carr, G. Leporizines A–C: Epithiodiketopiperazines Isolated from an Aspergillus Species. J. Nat. Prod. 2013, 76, 1523–1527. [Google Scholar] [CrossRef] [PubMed]
- Aparicio-Cuevas, M.A.; Rivero-Cruz, I.; Sánchez-Castellanos, M.; Menéndez, D.; Raja, H.A.; Joseph-Nathan, P.; González, M.C.; Figueroa, M. Dioxomorpholines and Derivatives from a Marine-Facultative Aspergillus Species. J. Nat. Prod. 2017, 80, 2311–2318. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.K.; Yan, T.; Guo, Y.; Song, Y.C.; Jiao, R.H.; Tan, R.X.; Ge, H.M. p-Terphenyl and Diterpenoid Metabolites from Endophytic Aspergillus sp. YXf3. J. Nat. Prod. 2012, 75, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wei, X.; Qin, X.; Tian, X.; Liao, L.; Li, K.; Zhou, X.; Yang, X.; Wang, F.; Zhang, T.; et al. Antiviral Merosesquiterpenoids Produced by the Antarctic Fungus Aspergillus ochraceopetaliformis SCSIO 05702. J. Nat. Prod. 2016, 79, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Guo, L.; Hao, J.; Wang, L.; Zhu, W. α-Glucosidase Inhibitors from the Marine-Derived Fungus Aspergillus flavipes HN4-13. J. Nat. Prod. 2016, 79, 2977–2981. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Chen, R.; Luo, Z.; Wang, W.; Chen, J. Antimicrobial activity and molecular docking studies of a novel anthraquinone from a marine-derived fungus Aspergillus versicolor. Nat. Prod. Res. 2018, 32, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.X.; Ding, L.; He, S. Discovery of a new biphenyl derivative by epigenetic manipulation of marine-derived fungus Aspergillus versicolor. Nat. Prod. Res. 2018, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Hamasaki, T.; Nagayama, K.; Hatsuda, Y. Two new metabolites, Sydonic acid and Hydroxysydonic Acid from Aspergillus sydowi. Agric. Biol. Chem. 1978, 42, 37–40. [Google Scholar] [CrossRef]
- Lu, Z.; Zhu, H.; Fu, P.; Wang, Y.; Zhang, Z.; Lin, H.; Liu, P.; Zhuang, Y.; Hong, K.; Zhu, W. Cytotoxic Polyphenols from the Marine-Derived Fungus Penicillium expansum. J. Nat. Prod. 2010, 73, 911–914. [Google Scholar] [CrossRef] [PubMed]
- Kudo, S.; Murakami, T.; Miyanishi, J.; Tanaka, K.; Takada, N.; Hashimoto, M. Isolation and Absolute Stereochemistry of Optically Active Sydonic Acid from Glonium sp. (Hysteriales, Ascomycota). Biosci. Biothechnol. Biochem. 2009, 73, 203–204. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xu, Y.; Shao, C.L.; Yang, R.Y.; Zheng, C.J.; Chen, Y.Y.; Fu, X.M.; Qian, P.Y.; She, Z.G.; Voogd, N.J. Antibacterial Bisabolane-Type Sesquiterpenoids from the Sponge-Derived Fungus Aspergillus sp. Mar. Drugs 2012, 10, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Elnaggar, M.S.; Ebada, S.S.; Ashour, M.L.; Ebrahim, W.; Muller, W.E.G.; Mandi, A.; Kurtan, T.; Singa, A.; Lin, W.; Liu, Z. Xanthones and sesquiterpene derivatives from a marine-derived fungus Scopulariopsis sp. Tetrahedron 2016, 72, 2411–2419. [Google Scholar] [CrossRef]
- Li, X.B.; Zhou, Y.H.; Zhu, R.X.; Chang, W.Q.; Yuan, H.Q.; Gao, W.; Zhang, L.L.; Zhao, Z.T.; Lou, H.X. Identification and Biological Evaluation of Secondary Metabolites from the Endolichenic Fungus Aspergillus versicolor. Chem. Biodivers. 2015, 12, 575–592. [Google Scholar] [CrossRef] [PubMed]
- Yakushiji, F.; Miyamoto, Y.; Kunoh, Y.; Okamoto, R.; Nakaminami, H.; Yamazaki, Y.; Noguchi, N.; Hayashi, Y. Novel Hybrid-Type Antimicrobial Agents Targeting the Switch Region of Bacterial RNA Polymerase. ACS Med. Chem. Lett. 2013, 4, 220–224. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |


| No. | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 1 | 154.8 (C) | 156.8 (C) | 157.0 (C) | 156.8 (C) |
| 2 | 132.8 (C) | 137.2 (C) | 138.3 (C) | 137.6 (C) |
| 3 | 125.2 (CH) | 127.6 (CH) | 127.9 (CH) | 127.7 (CH) |
| 4 | 121.9 (CH) | 121.5 (CH) | 121.3 (CH) | 121.5 (CH) |
| 5 | 131.9 (C) | 133.5 (C) | 131.0 (C) | 132.5 (C) |
| 6 | 117.0 (CH) | 118.6 (CH) | 118.3 (CH) | 118.6 (CH) |
| 7 | 169.7 (C) | 171.3 (C) | 168.5 (C) | 169.9 (C) |
| 8 | 74.9 (C) | 77.8 (C) | 77.8 (C) | 77.7 (C) |
| 9 | 38.1 (CH2) | 43.3 (CH2) | 43.9 (CH2) | 43.4 (CH2) |
| 10 | 18.3 (CH2) | 23.0 (CH2) | 19.9 (CH2) | 22.4 (CH2) |
| 11 | 36.5 (CH2) | 35.4 (CH2) | 45.0 (CH2) | 34.7 (CH2) |
| 12 | 100.9 (C) | 40.9 (CH) | 71.4 (C) | 33.7 (CH) |
| 13 | 28.6 (CH3) | 181.6 (C) | 29.2 (CH3) | 17.1 (CH3) |
| 14 | 26.9 (CH3) | 17.7 (CH3) | 29.1 (CH3) | 70.5 (CH2) |
| 15 | - | 28.9 (CH3) | 28.8 (CH3) | 28.8 (CH3) |
| 16 | - | - | 52.5 (CH3)(COOCH3) | 173.1 (C)(COCH3) |
| COCH3 | - | - | - | 20.8 (CH3) |
| No. | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 3 | 7.16 (d, 8.0, 1H) | 7.24 (d, 8.2, 1H) | 7.30 (d, 8.2, 1H) | 7.27 (d, 8.0, 1H) |
| 4 | 7.50 (dd, 1.7, 8.0, 1H) | 7.43 (d, 8.4, 1H) | 7.44 (dd, 1.8, 8.2,1H) | 7.44 (d, 8.0, 1H) |
| 5 | - | - | - | - |
| 6 | 7.34 (d, 1.7, 1H) | 7.36 (br s, 1H) | 7.36 (d, 1.8, 1H) | 7.38 (br s, 1H) |
| 7 | - | - | - | - |
| 8 | - | - | - | - |
| 9 | 1.81 (td, 4.2, 13.3, 1H)1.70 (m, 1H) | 1.99 (m, 1H)1.81 (m, 1H) | 1.98 (m, 1H)1.80 (m, 1H) | 1.96(m, 1H)1.81(m, 1H) |
| 10 | 1.62 (m, 1H)1.45 (m, 1H) | 1.35 (m, 1H)1.21 (m, 1H) | 1.39 (m, 1H)1.25(m, 1H) | 1.29 (m,2H) |
| 11 | 1.97 (br d, 13.3, 1H)1.73 (m, 1H) | 1.58 (m, 1H)1.34 (m, 1H) | 1.39 (m, 2H) | 1.35 (m, 1H)1.11 (m, 1H) |
| 12 | - | 2.34 (m, 1H) | - | 1.72 (m, 1H) |
| 13 | 1.50 (s, 3H) | - | 1.01 (s, 3H) | 0.86 (d, 6.8, 3H) |
| 14 | 1.60 (s, 3H) | 1.07 (d, 7.0, 3H) | 1.01 (s, 3H) | 3.88 (m, 1H)3.80 (m, 1H) |
| 15 | - | 1.59 (s, 3H) | 1.60 (s, 3H) | 1.60 (s, 3H) |
| COOCH3 | - | - | 3.88 (s, 3H) | - |
| COCH3 | - | - | - | 2.00 (s, 3H) |
| No. | Anticanceractivity (IC50 in μg/mL) | Antimicobial Activity(MIC in μg/mL) | |||
|---|---|---|---|---|---|
| A549 | Caski | HepG2 | MCF-7 | Erwinia carotovora | |
| 1 | 54.2 | 58.3 | 80.3 | 90.6 | 15.2 |
| 2 | 65.4 | 75.3 | 90.8 | 57.4 | 35.6 |
| 3 | 67.3 | 73.5 | 69.5 | 64.4 | 40.5 |
| 4 | 85.6 | 85.5 | 74.4 | 88.7 | 50.8 |
| 5 | 72.2 | 92.2 | 75.7 | 65.3 | 64.3 |
| 6 | 90.6 | 78.2 | 75.8 | 80.4 | 68.9 |
| 7 | 82.5 | 90.4 | 60.5 | 77.5 | 75.2 |
| 8 | 64.4 | 65.6 | 59.2 | 64.1 | 85.2 |
| 9 | 89.7 | 83.5 | 76.2 | 79.1 | 68.8 |
| 10 | 94.2 | 90.4 | 88.2 | 96.8 | 75.3 |
| 11 | 100.1 | 95.3 | 100.5 | 78.9 | 65.4 |
| 12 | 55.6 | 85.2 | 78.8 | 65.4 | 50.9 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, Z.-Y.; Tan, M.-H.; Liu, C.-X.; Lv, M.-M.; Deng, Z.-S.; Cao, F.; Zou, K.; Proksch, P. Aspergoterpenins A–D: Four New Antimicrobial Bisabolane Sesquiterpenoid Derivatives from an Endophytic Fungus Aspergillus versicolor. Molecules 2018, 23, 1291. https://doi.org/10.3390/molecules23061291
Guo Z-Y, Tan M-H, Liu C-X, Lv M-M, Deng Z-S, Cao F, Zou K, Proksch P. Aspergoterpenins A–D: Four New Antimicrobial Bisabolane Sesquiterpenoid Derivatives from an Endophytic Fungus Aspergillus versicolor. Molecules. 2018; 23(6):1291. https://doi.org/10.3390/molecules23061291
Chicago/Turabian StyleGuo, Zhi-Yong, Ming-Hui Tan, Cheng-Xiong Liu, Meng-Meng Lv, Zhang-Shuang Deng, Fei Cao, Kun Zou, and Peter Proksch. 2018. "Aspergoterpenins A–D: Four New Antimicrobial Bisabolane Sesquiterpenoid Derivatives from an Endophytic Fungus Aspergillus versicolor" Molecules 23, no. 6: 1291. https://doi.org/10.3390/molecules23061291
APA StyleGuo, Z.-Y., Tan, M.-H., Liu, C.-X., Lv, M.-M., Deng, Z.-S., Cao, F., Zou, K., & Proksch, P. (2018). Aspergoterpenins A–D: Four New Antimicrobial Bisabolane Sesquiterpenoid Derivatives from an Endophytic Fungus Aspergillus versicolor. Molecules, 23(6), 1291. https://doi.org/10.3390/molecules23061291





