Epitaxially Grown Ultra-Flat Self-Assembling Monolayers with Dendrimers
Abstract
:1. Introduction
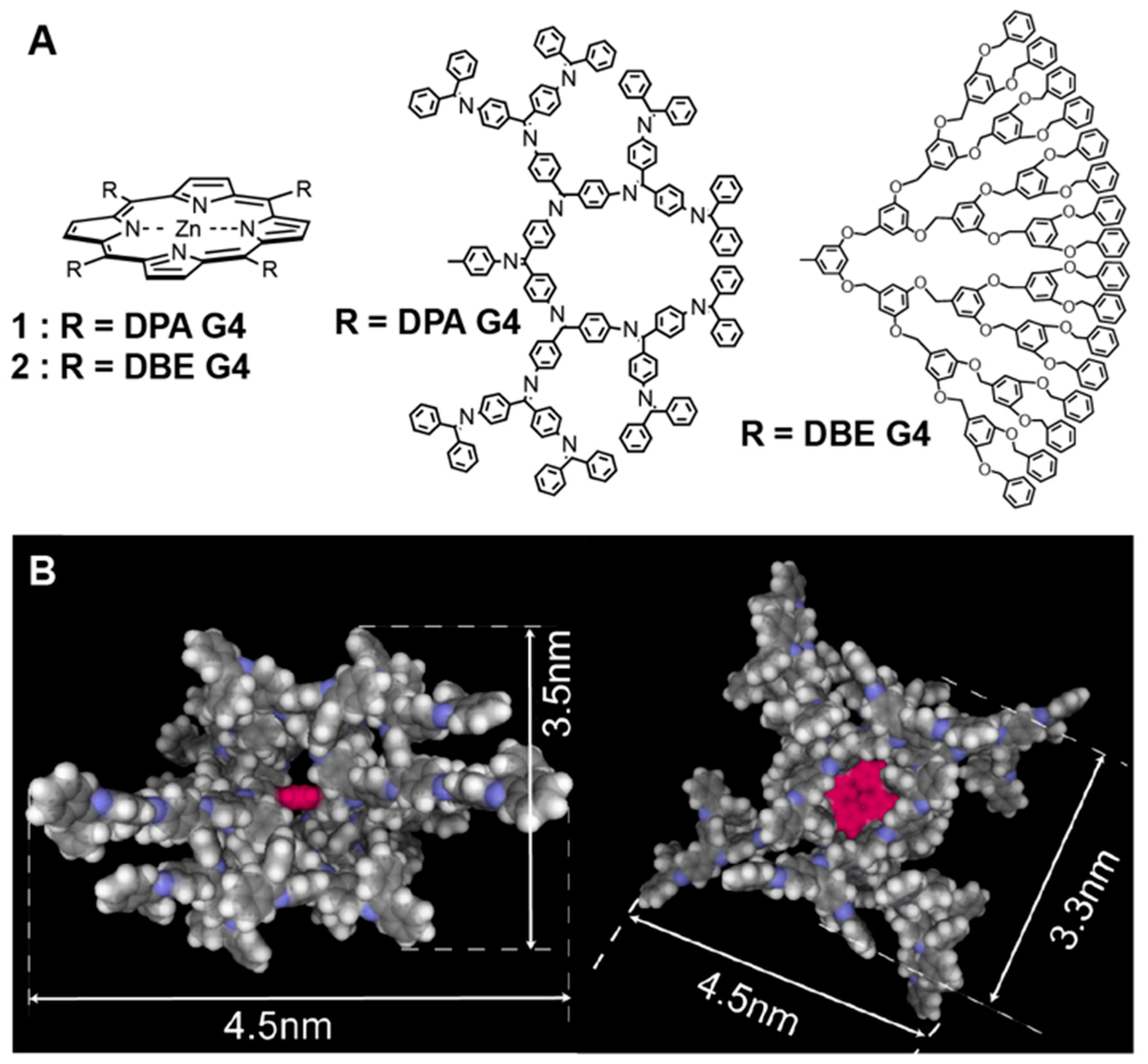
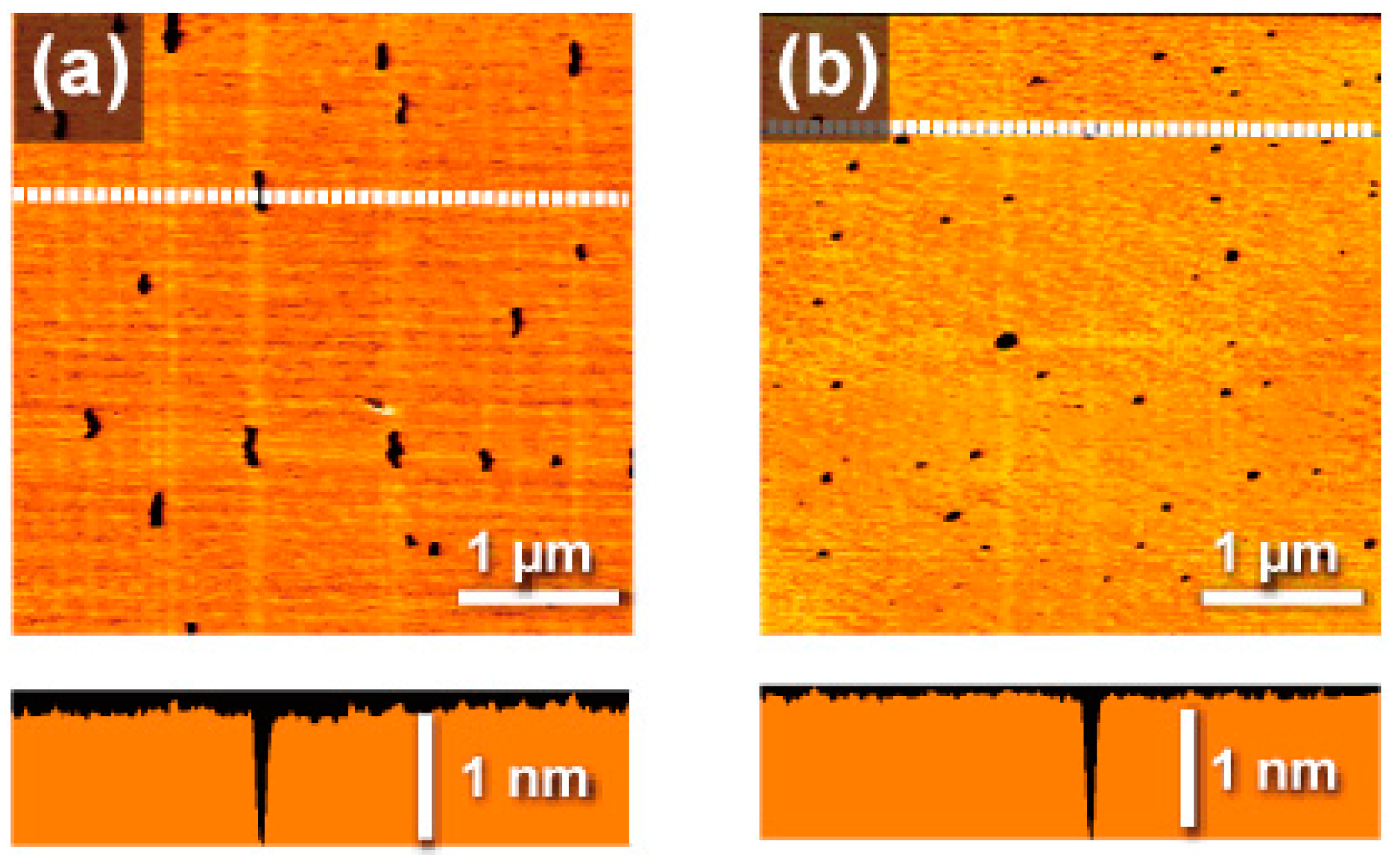
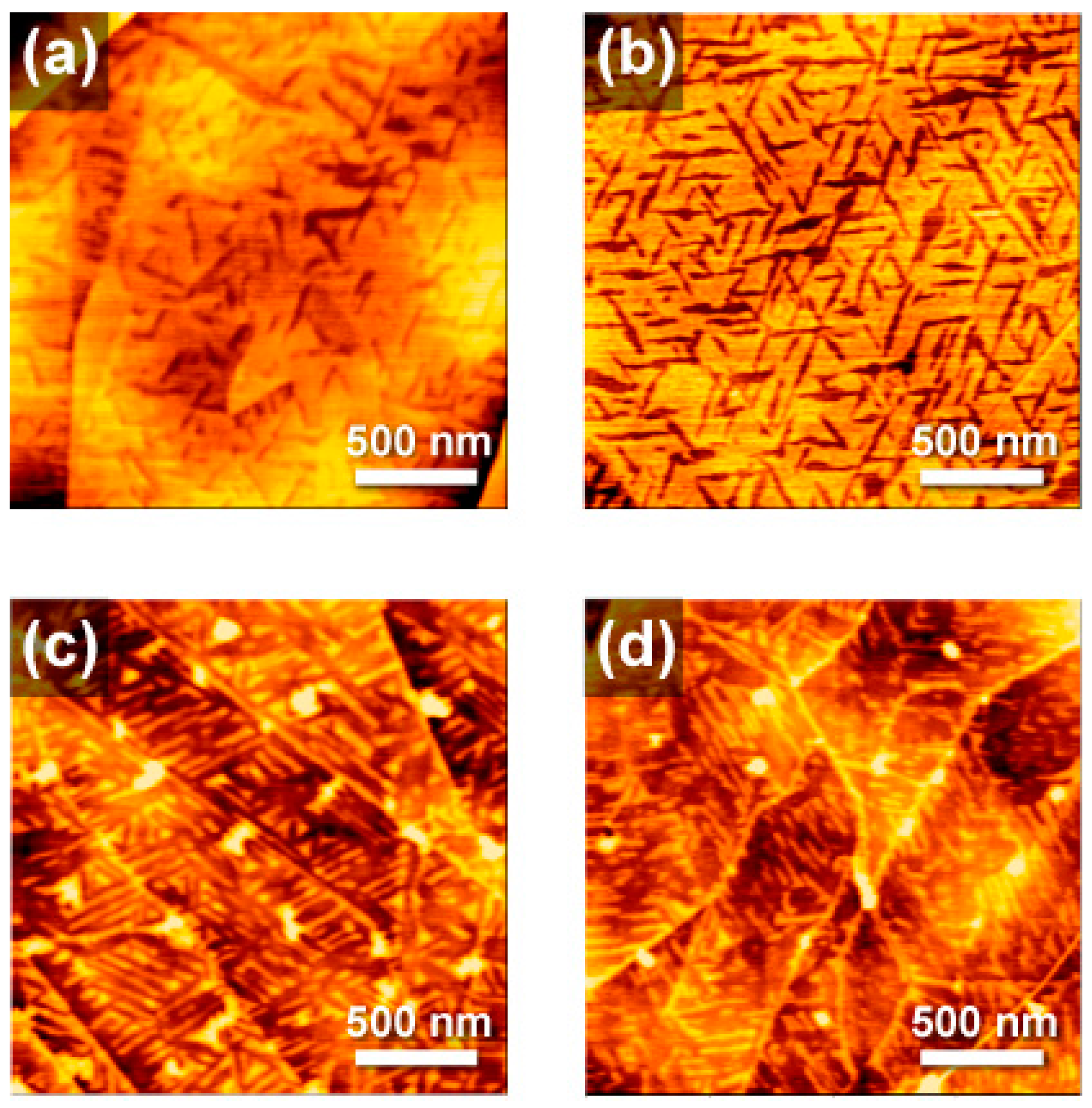
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Love, J.C.; Estroff, L.A.; Kriebel, J.K.; Nuzzo, R.G.; Whitesides, G.M. Self-Assembled Monolayers of Thiolates on Metals as a Form of Nanotechnology. Chem. Rev. 2005, 105, 1103–1170. [Google Scholar] [CrossRef] [PubMed]
- Poirier, G.E. Characterization of Organosulfur Molecular Monolayers on Au(111) using Scanning Tunneling Microscopy. Chem. Rev. 1997, 97, 1117–1128. [Google Scholar] [CrossRef] [PubMed]
- Yamada, R.; Sakai, H.; Uosaki, K. Solvent Effect on the Structure of the Self-Assembled Monolayer of Alkanethiol. Chem. Lett. 1999, 28, 667–668. [Google Scholar] [CrossRef]
- Tomalia, D.A.; Naylor, A.M.; Goddard, W.A. Starburst Dendrimers: Molecular-Level Control of Size, Shape, Surface Chemistry, Topology, and Flexibility from Atoms to Macroscopic Matter. Angew. Chem. Int. Ed. 1990, 29, 138–175. [Google Scholar] [CrossRef]
- Tomalia, D.A.; Khanna, S.N. A Systematic Framework and Nanoperiodic Concept for Unifying Nanoscience: Hard/Soft Nanoelements, Superatoms, Meta-Atoms, New Emerging Properties, Periodic Property Patterns, and Predictive Mendeleev-like Nanoperiodic Tables. Chem. Rev. 2016, 116, 2705–2774. [Google Scholar] [CrossRef] [PubMed]
- Tomalia, D.A. Dendrons/dendrimers: Quantized, nano-element like building blocks for soft-soft and soft-hard nano-compound synthesis. Soft Matter 2010, 6, 456–474. [Google Scholar] [CrossRef]
- Rosen, B.M.; Wilson, C.J.; Wilson, D.A.; Peterca, M.; Imam, M.R.; Percec, V. Dendron-Mediated Self-Assembly, Disassembly, and Self-Organization of Complex Systems. Chem. Rev. 2009, 109, 6275–6540. [Google Scholar] [CrossRef] [PubMed]
- Percec, V.; Wilson, D.A.; Leowanawat, P.; Wilson, C.J.; Hughes, A.D.; Kaucher, M.S.; Hammer, D.A.; Levine, D.H.; Kim, A.J.; Bates, F.S.; et al. Self-Assembly of Janus Dendrimers into Uniform Dendrimersomes and Other Complex Architectures. Science 2010, 328, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Tanaka, R.; Arimoto, S.; Sakai, M.; Fujii, M.; Yamamoto, K. Probing Stepwise Complexation in Phenylazomethine Dendrimers by a Metallo-Porphyrin Core. J. Am. Chem. Soc. 2005, 127, 13896–13905. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Ueda, H.; Yamamoto, K. Enhancing the photoelectric effect with a potential-programmed molecular rectifier. J. Am. Chem. Soc. 2012, 134, 8412–8415. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Kawana, Y.; Kurokawa, T.; Yamamoto, K. Macromolecular semi-rigid nanocavities for cooperative recognition of specific large molecular shapes. Nat. Commun. 2013, 4, 2581. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Imaoka, T. Dendrimer complexes based on fine-controlled metal assembling. Bull. Chem. Soc. Jpn. 2006, 79, 511–526. [Google Scholar] [CrossRef]
- Ochi, Y.; Fujii, A.; Nakajima, R.; Yamamoto, K. Stepwise Radial Complexation of Triphenylmethyliums on a Phenylazomethine Dendrimer for Organic–Metal Hybrid Assembly. Macromolecules 2010, 43, 6570–6576. [Google Scholar] [CrossRef]
- Ochi, Y.; Suzuki, M.; Imaoka, T.; Murata, M.; Nishihara, H.; Einaga, Y.; Yamamoto, K. Controlled storage of ferrocene derivatives as redox-active molecules in dendrimers. J. Am. Chem. Soc. 2010, 132, 5061–5069. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Imaoka, T. Precision synthesis of subnanoparticles using dendrimers as a superatom synthesizer. Acc. Chem. Res. 2014, 47, 1127–1136. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Imaoka, T.; Chun, W.-J.; Enoki, O.; Katoh, H.; Takenaga, M.; Sonoi, A. Size-specific catalytic activity of platinum clusters enhances oxygen reduction reactions. Nat. Chem. 2009, 1, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Kitazawa, H.; Chun, W.-J.; Yamamoto, K. Finding the Most Catalytically Active Platinum Clusters with Low Atomicity. Angew. Chem. Int. Ed. 2015, 127, 9948–9953. [Google Scholar] [CrossRef]
- Takahashi, M.; Koizumi, H.; Chun, W.-J.; Kori, M.; Imaoka, T.; Yamamoto, K. Finely controlled multimetallic nanocluster catalysts for solvent-free aerobic oxidation of hydrocarbons. Sci. Adv. 2017, 3, e1700101. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Kitazawa, H.; Chun, W.-J.; Omura, S.; Albrecht, K.; Yamamoto, K. Magic Number Pt13 and Misshapen Pt12 Clusters: Which One is the Better Catalyst? J. Am. Chem. Soc. 2013, 135, 13089–13095. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Inoue, N.; Yamamoto, K. Electron-transfer through potential gradient based on a dendrimer architecture. Chem. Commun. 2012, 48, 7235–7237. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Kobayashi, H.; Katsurayama, M.; Yamamoto, K. A potential gradient along the layer-by-layer architecture for electron transfer rectification. Dalton Trans. 2015, 44, 15116–15120. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Inoue, N.; Yamamoto, K. Extended Potential-Gradient Architecture of a Phenylazomethine Dendrimer. Org. Lett. 2013, 15, 1810–1813. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, K.; Hirabayashi, Y.; Otake, M.; Mendori, S.; Tobari, Y.; Azuma, Y.; Majima, Y.; Yamamoto, K. Polymerization of a divalent/tetravalent metal-storing atom-mimicking dendrimer. Sci. Adv. 2016, 2, e1601414. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, T.; Bukeo, N.; Yamamoto, K. A Self-Assembling Dendritic Reactor: Versatile Formation of Characteristic Patterns with Nanoscale Dimension. Macromol. Rapid Commun. 2015, 36, 616–620. [Google Scholar] [CrossRef] [PubMed]
- Tsukruk, V.V.; Rinderspacher, F.; Bliznyuk, V.N. Self-Assembled Multilayer Films from Dendrimers. Langmuir 1997, 13, 2171–2176. [Google Scholar] [CrossRef]
- Hierlemann, A.; Campbell, J.; Baker, L.; Crooks, R.M.; Ricco, A.J. Structural Distortion of Dendrimers on Gold Surfaces: A Tapping-Mode AFM Investigation. J. Am. Chem. Soc. 1998, 120, 5323–5324. [Google Scholar] [CrossRef]
- Pollak, K.W.; Leon, J.W.; Frechet, J.M.J.; Maskus, M.; Abruña, H.D. Effects of Dendrimer Generation on Site Isolation of Core Moieties: Electrochemical and Fluorescence Quenching Studies with Metalloporphyrin Core Dendrimers. Chem. Mater. 1998, 10, 30–38. [Google Scholar] [CrossRef]
- Wan, L.; Itaya, K. In situ scanning tunnelling microscopy of benzene, naphthalene, and anthracene adsorbed on Cu(111) in solution. Langmuir 1997, 13, 7173–7179. [Google Scholar] [CrossRef]
- Chakarova-Kack, S.; Schroder, E.; Lundqvist, B.; Langreth, D. Application of van der Waals density functional to an extended system: Adsorption of benzene and naphthalene on graphite. Phys. Rev. Lett. 2006, 96, 146107. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Nemoto, T.; Yoshida, K.; Kurata, H.; Isoda, S. In situ atomic force microscopy observation of the desorption process from monomolecular organic layers of a naphthalene derivative. Jpn. J. Appl. Phys. 2004, 43, 4606–4609. [Google Scholar] [CrossRef]
- Elemans, J.A.A.W.; Lei, S.; de Feyter, S. Molecular and Supramolecular Networks on Surfaces: From Two-Dimensional Crystal Engineering to Reactivity. Angew. Chem. Int. Ed. 2009, 48, 7298–7332. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: All the reported data and samples are available from the corresponding authors on reasonable request. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imaoka, T.; Bukeo, N.; Yamamoto, K. Epitaxially Grown Ultra-Flat Self-Assembling Monolayers with Dendrimers. Molecules 2018, 23, 485. https://doi.org/10.3390/molecules23020485
Imaoka T, Bukeo N, Yamamoto K. Epitaxially Grown Ultra-Flat Self-Assembling Monolayers with Dendrimers. Molecules. 2018; 23(2):485. https://doi.org/10.3390/molecules23020485
Chicago/Turabian StyleImaoka, Takane, Noriko Bukeo, and Kimihisa Yamamoto. 2018. "Epitaxially Grown Ultra-Flat Self-Assembling Monolayers with Dendrimers" Molecules 23, no. 2: 485. https://doi.org/10.3390/molecules23020485
APA StyleImaoka, T., Bukeo, N., & Yamamoto, K. (2018). Epitaxially Grown Ultra-Flat Self-Assembling Monolayers with Dendrimers. Molecules, 23(2), 485. https://doi.org/10.3390/molecules23020485



