Dynamic Changes in Phenolics and Antioxidant Capacity during Pecan (Carya illinoinensis) Kernel Ripening and Its Phenolics Profiles
Abstract
:1. Introduction
2. Results and Discussion
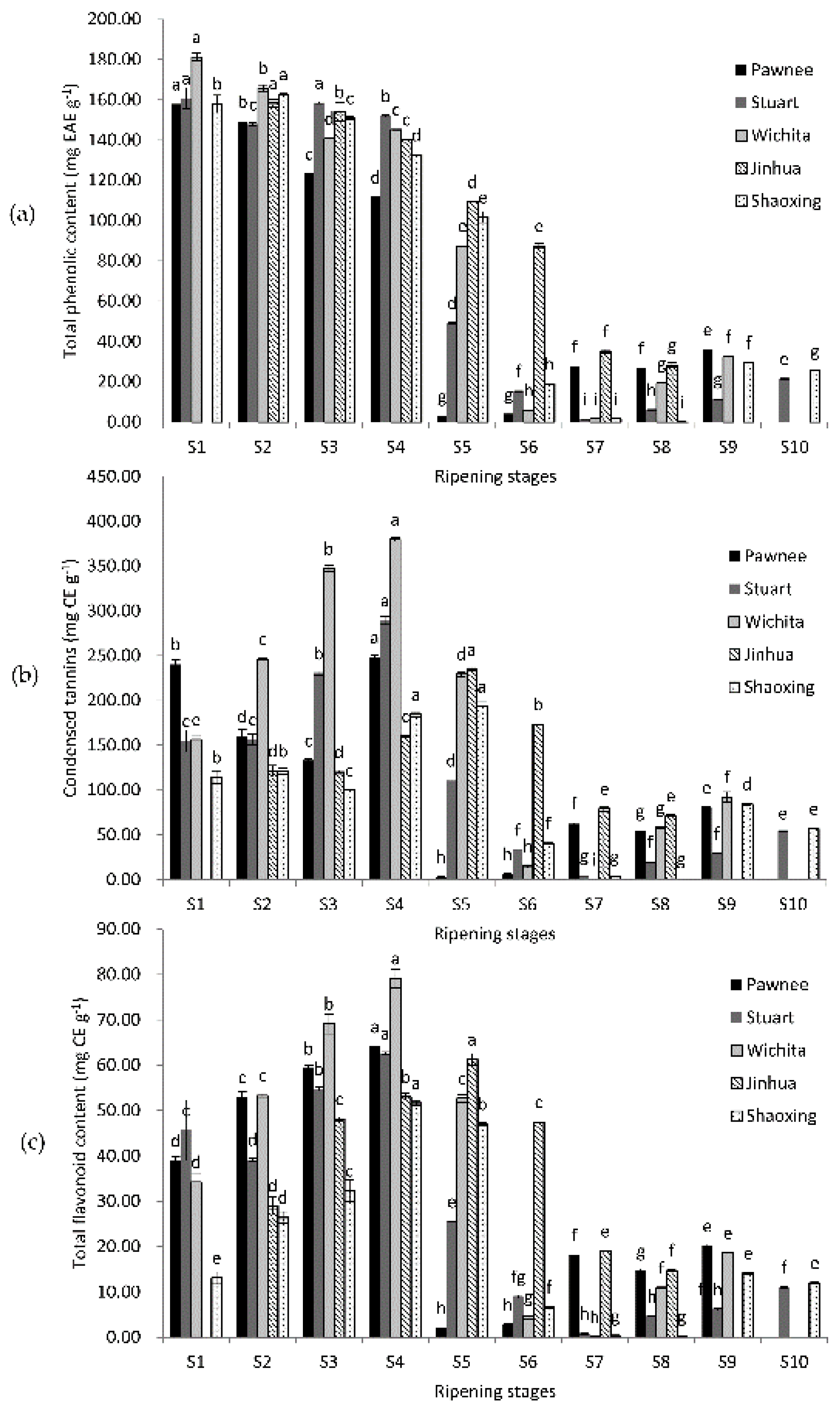
2.1. Changes of Total Phenolic Content, Condensed Tannins, and Total Flavonoid Content
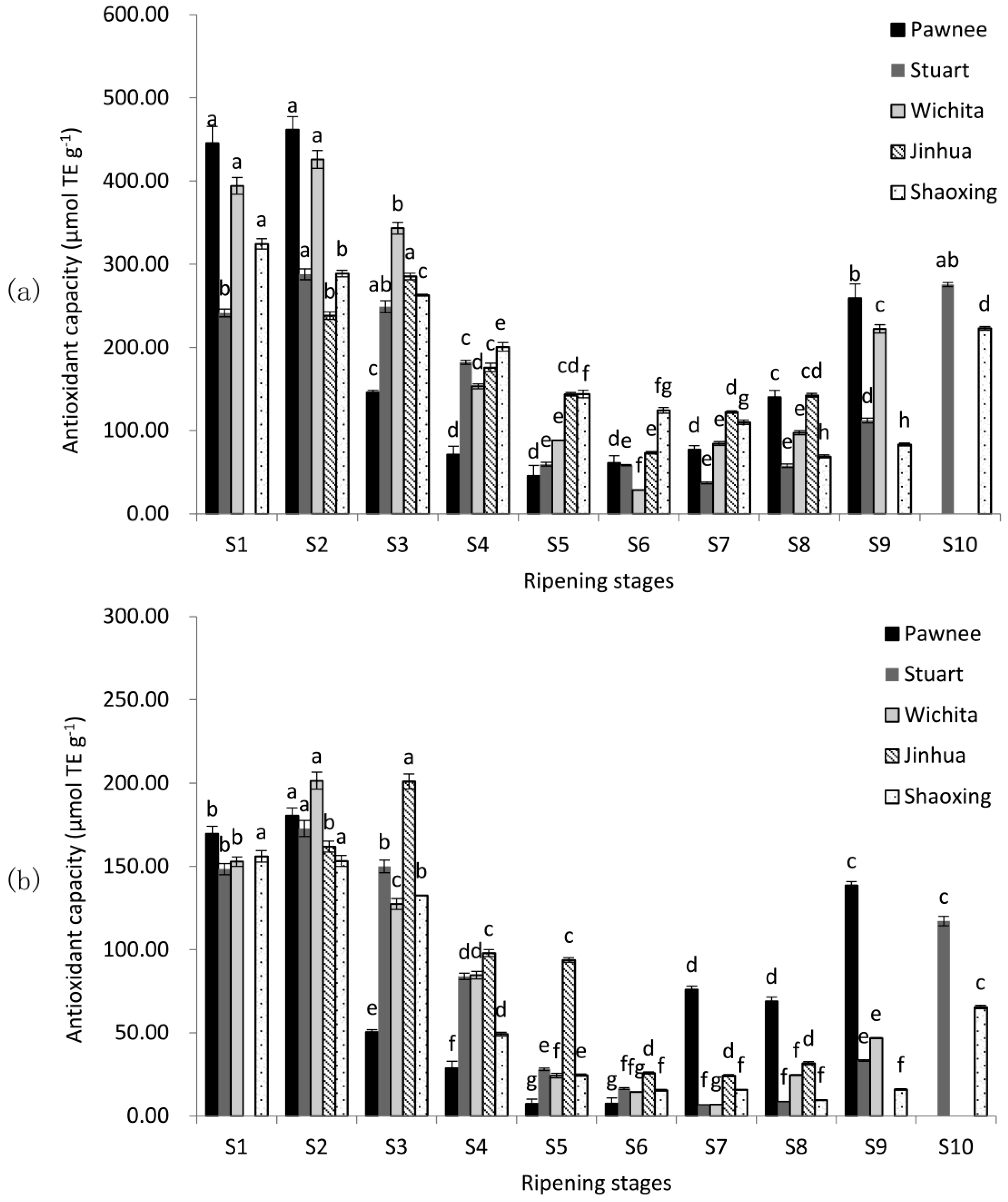
2.2. Antioxidant Capacity
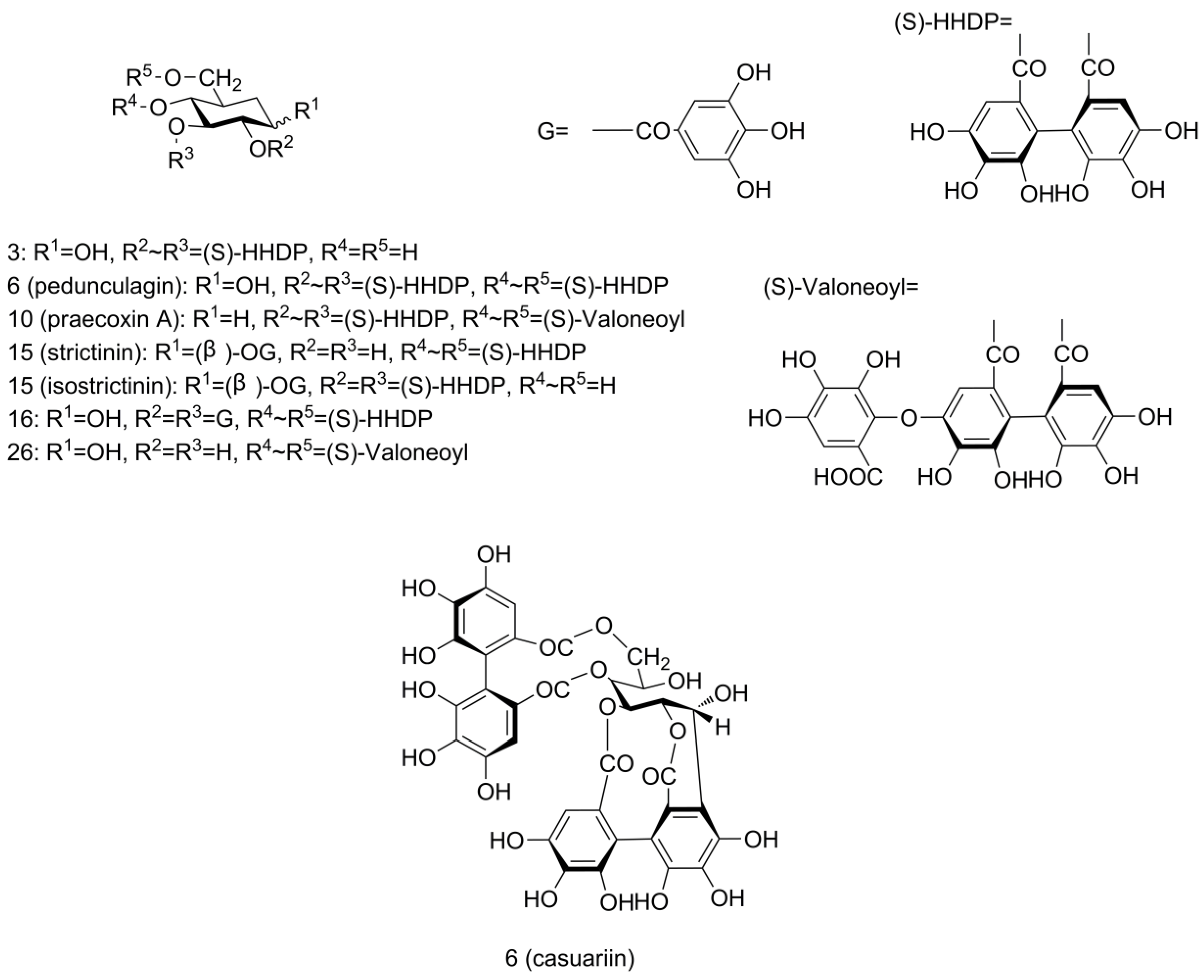
2.3. Identification of Phenolics in Pecan Kernels Using UPLC-Q/TOF-MS
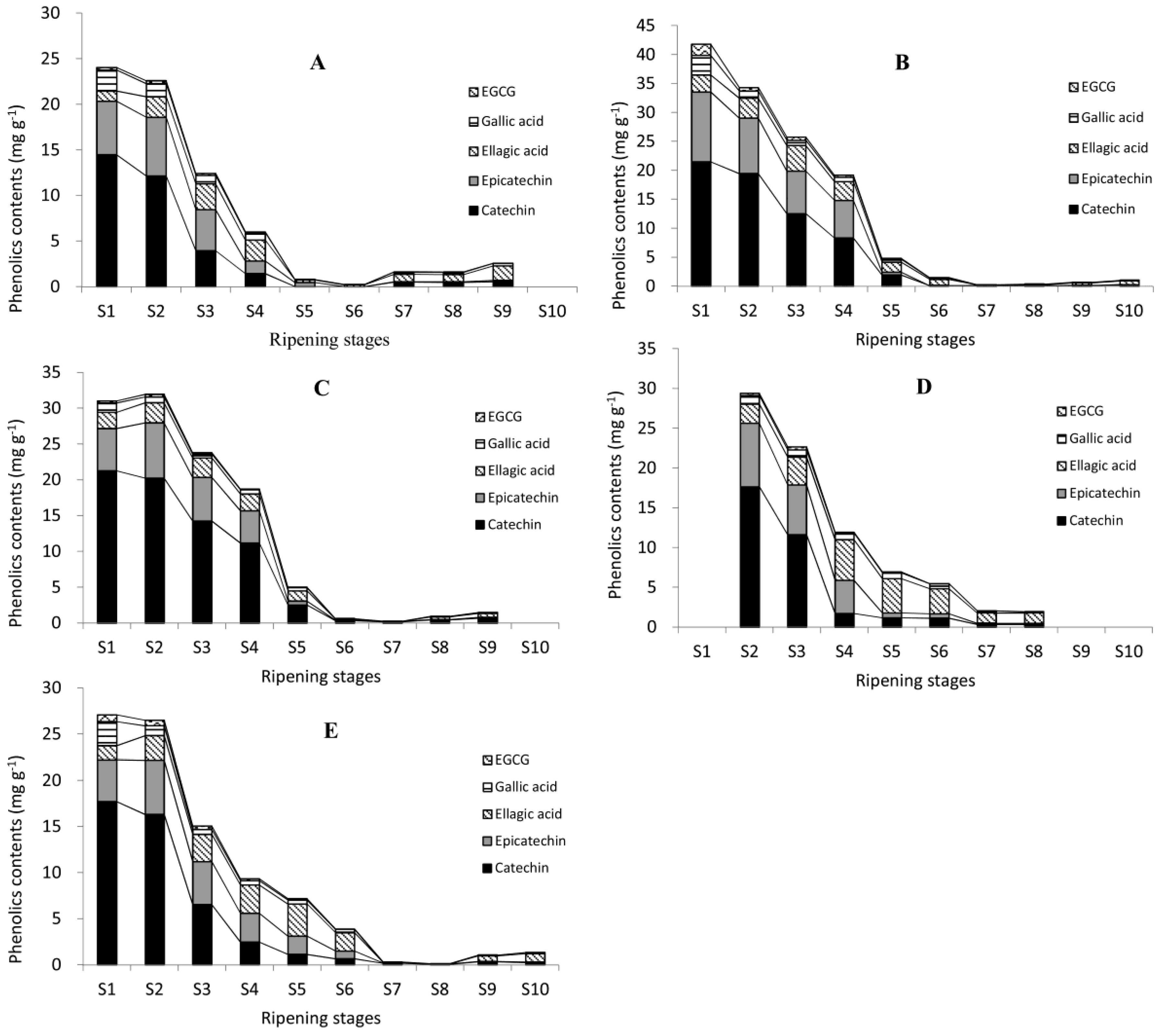
2.4. Quantification of Phenolics in Pecan Kernels Using HPLC
2.5. The Correlation of Antioxidant Capacity and Phenolics
3. Materials and Methods
3.1. Pecan Samples
3.2. Chemicals
3.3. Sample Extraction
3.4. Total Phenolic Content
3.5. Condensed Tannins
3.6. Total Flavonoid Content
3.7. Antioxidant Capacity
3.8. UPLC-Q/TOF-MS
3.9. HPLC
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yao, X.H.; Chang, J.; Wang, K.L. The Research Proceeding of Pecan in China, 1st ed.; Science Press: Beijing, China, 2014; pp. 70–84. ISBN 978-7-03-041754-1. [Google Scholar]
- Zhang, R.; Peng, F.; Li, Y. Pecan production in China. Sci. Hortic. (Amsterdam) 2015, 197, 719–727. [Google Scholar] [CrossRef]
- Wu, X.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Lipophilic and hydrophilic antioxidant capacities of common foods in the United States. J. Agric. Food Chem. 2004, 52, 4026–4037. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Kelm, M.A.; Hammerstone, J.F.; Beecher, G.; Holden, J.; Haytowitz, D.; Gebhardt, S.; Prior, R.L. Concentrations of proanthocyanidins in common foods and estimations of normal consumption. J. Nutr. 2004, 134, 613–617. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Quezada, A.G.; Lombardini, L.; Cisneros-Zevallos, L. Antioxidants in pecan nut cultivars [Carya illinoinensis (Wangenh.) K. Koch]. In Pecan Nut Cultivars and Antioxidants; Elsevier Inc.: Amsterdam, The Netherlands, 2011; pp. 881–889. [Google Scholar]
- Villarreal-Lozoya, J.E.; Lombardini, L.; Cisneros-Zevallos, L. Phytochemical constituents and antioxidant capacity of different pecan [Carya illinoinensis (Wangenh.) K. Koch] cultivars. Food Chem. 2007, 102, 1241–1249. [Google Scholar] [CrossRef]
- Harnly, J.M.; Doherty, R.F.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Bhagwat, S.; Gebhardt, S. Flavonoid content of U.S. fruits, vegetables, and nuts. J. Agric. Food Chem. 2006, 54, 9966–9977. [Google Scholar] [CrossRef] [PubMed]
- De la Rosa, L.A.; Alvarez-Parrilla, E.; Shahidi, F. Phenolic compounds and antioxidant activity of kernels and shells of Mexican pecan (Carya illinoinensis). J. Agric. Food Chem. 2011, 59, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Robbins, K.S.; Ma, Y.; Wells, M.L.; Greenspan, P.; Pegg, R.B. Separation and characterization of phenolic compounds from U.S. pecans by liquid chromatography-tandem mass spectrometry. J. Agric. Food Chem. 2014, 62, 4332–4341. [Google Scholar] [CrossRef] [PubMed]
- Hudthagosol, C.; Haddad, E.H.; McCarthy, K.; Wang, P.; Oda, K.; Sabaté, J. Pecans acutely increase plasma postprandial antioxidant capacity and catechins and decrease LDL oxidation in humans. J. Nutr. 2011, 141, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Han, J.; Hu, F.B.; Giovannucci, E.L.; Stampfer, M.J.; Willett, W.C.; Fuchs, C.S. Association of nut consumption with total and cause-specific mortality. N. Engl. J. Med. 2013, 369, 2001–2011. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Flores, A.A.; Wong-Paz, J.E.; Lerma-Herrera, M.A.; Martinez-Gonzalez, A.I.; Olivas-Aguirre, F.J.; Aguilar, C.N.; Wall-Medrano, A.; Gonzalez-Aguilar, G.A.; Alvarez-Parrilla, E.; de la Rosa, L.A. Proanthocyanidins from the kernel and shell of pecan (Carya illinoinensis): Average degree of polymerization and effects on carbohydrate, lipid, and peptide hydrolysis in a simulated human digestive system. J. Funct. Foods 2017, 28, 227–234. [Google Scholar] [CrossRef]
- Okuda, T. Systematics and health effects of chemically distinct tannins in medicinal plants. Phytochemistry 2005, 66, 2012–2031. [Google Scholar] [CrossRef] [PubMed]
- Engler Ribeiro, P.C.; de Britto Policarpi, P.; Dal Bo, A.; Barbetta, P.A.; Block, J.M. Impact of pecan nut shell aqueous extract on the oxidative properties of margarines during storage. J. Sci. Food Agric. 2017, 97, 3005–3012. [Google Scholar] [CrossRef] [PubMed]
- Corthout, J.; Pieters, L.A.; Claeys, M.; Vanden Berghe, D.A.; Vlietinck, A.J. Antiviral ellagitannins from Spondias mombin. Phytochemistry 1991, 30, 1129–1130. [Google Scholar] [CrossRef]
- Singanusong, R.; Mason, R.L.; D’Arcy, B.R.; Nottingham, S.M. Compositional changes of Australia-grown Western Schley pecans [Carya illinoinensis (Wangenh.) K. Koch] during maturation. J. Agric. Food Chem. 2003, 51, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Bouali, I.; Trabelsi, H.; Abdallah, I.B.; Albouchi, A.; Martine, L.; Grégoire, S.; Bouzaien, G.; Gandour, M.; Boukhchina, S.; Berdeaux, O. Changes in fatty acid, tocopherol and xanthophyll contents during the development of Tunisian-grown pecan nuts. J. Am. Oil Chem. Soc. 2013, 90, 1869–1876. [Google Scholar] [CrossRef]
- Bouali, I.; Trabelsi, H.; Herchi, W.; Martine, L.; Albouchi, A.; Bouzaien, G.; Sifi, S.; Boukhchina, S.; Berdeaux, O. Analysis of pecan nut (Carya illinoinensis) unsaponifiable fraction. Effect of ripening stage on phytosterols and phytostanols composition. Food Chem. 2014, 164, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Robbins, K.S.; Gong, Y.; Wells, M.L.; Greenspan, P.; Pegg, R.B. Investigation of the antioxidant capacity and phenolic constituents of U.S. pecans. J. Funct. Foods 2015, 15, 11–22. [Google Scholar] [CrossRef]
- De la Rosa, L.A.; Vazquez-Flores, A.A.; Alvarez-Parrilla, E.; Rodrigo-García, J.; Medina-Campos, O.N.; Ávila-Nava, A.; González-Reyes, S.; Pedraza-Chaverri, J. Content of major classes of polyphenolic compounds, antioxidant, antiproliferative, and cell protective activity of pecan crude extracts and their fractions. J. Funct. Foods 2014, 7, 219–228. [Google Scholar] [CrossRef]
- Salvador, A.A.; Podestá, R.; Block, J.M.; Ferreira, S.R.S. Increasing the value of pecan nut [Carya illinoinensis (Wangenh) C. Koch] cake by means of oil extraction and antioxidant activity evaluation. J. Supercrit. Fluids 2016, 116, 215–222. [Google Scholar] [CrossRef]
- Yang, J.; Liu, R.H.; Linna, H. Antioxidant and antiproliferative activities of common edible nut seeds. LWT Food Sci. Technol. 2009, 42, 1–8. [Google Scholar] [CrossRef]
- Kornsteiner, M.; Wagner, K.H.; Elmadfa, I. Tocopherols and total phenolics in 10 different nut types. Food Chem. 2006, 98, 381–387. [Google Scholar] [CrossRef]
- Flores-Córdova, M.A.; Sánchez, E.; Muñoz-Márquez, E.; Ojeda-Barrios, D.L.; Soto-Parra, J.M.; Preciado-Rangel, P. Phytochemical composition and antioxidant capacity in Mexican pecan nut. Emir. J. Food Agric. 2017, 29, 346–350. [Google Scholar]
- Alasalvar, C.; Bolling, B.W. Review of nut phytochemicals, fat-soluble bioactives, antioxidant components and health effects. Br. J. Nutr. 2015, 113, S68–S78. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, C.J.; Odell, G.V.; Hinrichs, H.A.; Hopfer, D.A.; Kays, S.J. Genetic, environmental, and maturity effects on pecan kernel lipid, fatty acid, tocopherol, and protein composition. J. Food Qual. 1992, 15, 263–278. [Google Scholar] [CrossRef]
- Awad, M.A.; Al-Qurashi, A.D.; Alrashdi, A.M.A.; Mohamed, S.A.; Faidi, F. Developmental changes in phenolic compounds, antioxidant capacity and enzymes activity in skin of ‘El-Bayadi’ table grapes. Sci. Hortic. (Amsterdam) 2017, 224, 219–225. [Google Scholar] [CrossRef]
- Ferreyra, R.M.; Viña, S.Z.; Mugridge, A.; Chaves, A.R. Growth and ripening season effects on antioxidant capacity of strawberry cultivar Selva. Sci. Hortic. (Amsterdam) 2007, 112, 27–32. [Google Scholar] [CrossRef]
- Price, M.L.; Scoyoc, S.V.; Butler, L.G. A critical evaluation of the vanillin reaction as an assay for tannin in sorghum grain. J. Agric. Food Chem. 1978, 26, 1214–1218. [Google Scholar] [CrossRef]
- Regueiro, J.; Sánchez-González, C.; Vallverdú-Queralt, A.; Simal-Gándara, J.; Lamuela-Raventós, R.; Izquierdo-Pulido, M. Comprehensive identification of walnut polyphenols by liquid chromatography coupled to linear ion trap–Orbitrap mass spectrometry. Food Chem. 2014, 152, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Haidy, A.G.; Nahla, A.A.; Mohammed, M.A. Phenolic constituents with promising antioxidant and hepatoprotective activities from the leaves extract of Carya illinoinensis. Nat. Prod. Indian J. 2007, 3, 151–158. [Google Scholar]
- Gong, Y.; Pegg, R.B. Separation of ellagitannin-rich phenolics from U.S. pecans and Chinese hickory nuts using fused-core HPLC columns and their characterization. J. Agric. Food Chem. 2017, 65, 5810–5820. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Caravaca, A.M.; Verardo, V.; Segura-Carretero, A.; Caboni, M.F.; Fernández-Gutiérrez, A. Development of a rapid method to determine phenolic and other polar compounds in walnut by capillary electrophoresis–electrospray ionization time-of-flight mass spectrometry. J. Chromatogr. A 2008, 1209, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, S.; Misra, K. Ellagic acid 4-O-rutinoside from pods of Prosopis juliflora. Phytochemistry 1981, 20, 2439–2440. [Google Scholar] [CrossRef]
- Wang, K.J.; Du, M.; Hu, X.S.; Qi, J.X.; Hao, Y.B. Identification of phenolic compounds in Walnut (Juglans regia L.) seeds by high performance liquid chromatography-electrospray ionization mass spectrometry. Chin. J. Anal. Chem. 2009, 37, 867–872. [Google Scholar]
- Santos, S.A.O.; Villaverde, J.J.; Freire, C.S.R.; Domingues, M.R.M.; Neto, C.P.; Silvestre, A.J.D. Phenolic composition and antioxidant activity of Eucalyptus grandis, E. urograndis (E. grandis × E. urophylla) and E. maidenii bark extracts. Ind. Crops Prod. 2012, 39, 120–127. [Google Scholar] [CrossRef]
- Sarnoski, P.J.; Johnson, J.V.; Reed, K.A.; Tanko, J.M.; O’Keefe, S.F. Separation and characterisation of proanthocyanidins in Virginia type peanut skins by LC–MSn. Food Chem. 2012, 131, 927–939. [Google Scholar] [CrossRef]
- Ito, H.; Okuda, T.; Fukuda, T.; Hatano, T.; Yoshida, T. Two novel dicarboxylic Acid derivatives and a new dimeric hydrolyzable tannin from walnuts. J. Agric. Food Chem. 2007, 55, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Slatnar, A.; Mikulic-Petkovsek, M.; Stampar, F.; Veberic, R.; Solar, A. HPLC-MSn identification and quantification of phenolic compounds in hazelnut kernels, oil and bagasse pellets. Food Res. Int. 2014, 64, 783–789. [Google Scholar] [CrossRef]
- Gordon, A.; Friedrich, M.; Da Matta, V.M.; Herbster Moura, C.F.; Marx, F. Changes in phenolic composition, ascorbic acid and antioxidant capacity in cashew apple (Anacardium occidentale L.) during ripening. Fruits 2012, 67, 267–276. [Google Scholar] [CrossRef]
- Senter, S.D.; Forbus, W.R.; Smit, C.J.B. Leucoanthocyanidin oxidation in pecan kernels: Relation to discoloration and kernel quality. J. Food Sci. 1978, 43, 128–134. [Google Scholar] [CrossRef]
- Senter, S.D.; Horvat, R.J.; Forbus, W. Relation between phenolic acid content and stability of pecans in accelerated storage. J. Food Sci. 1980, 45, 1380–1382. [Google Scholar] [CrossRef]
- Natsume, M.; Osakabe, N.; Yamagishi, M.; Takizawa, T.; Nakamura, T.; Miyatake, H.; Hatano, T.; Yoshida, T. Analyses of polyphenols in cacao liquor, cocoa, and chocolate by normal-phase and reversed-phase HPLC. Biosci. Biotechnol. Biochem. 2000, 64, 2581–2587. [Google Scholar] [CrossRef] [PubMed]
- Trox, J.; Vadivel, V.; Vetter, W.; Stuetz, W.; Kammerer, D.R.; Carle, R.; Scherbaum, V.; Gola, U.; Nohr, D.; Biesalski, H.K. Catechin and epicatechin in testa and their association with bioactive compounds in kernels of cashew nut (Anacardium occidentale L.). Food Chem. 2011, 128, 1094–1099. [Google Scholar] [CrossRef]
- Holton, T.A.; Cornish, E.C. Genetics and biochemistry of anthocyanin biosynthesis. Plant Cell 1995, 7, 1071–1083. [Google Scholar] [CrossRef] [PubMed]
- Malik, N.S.; Perez, J.L.; Lombardini, L.; Cornacchia, R.; Cisneros-Zevallos, L. Phenolic compounds and fatty acid composition of organic and conventional grown pecan kernels. J. Sci. Food Agric. 2009, 89, 2207–2213. [Google Scholar] [CrossRef]
- Treutter, D. Biosynthesis of phenolic compounds and its regulation in apple. Plant Growth Regul. 2001, 34, 71–89. [Google Scholar] [CrossRef]
- Ghasemi, K.; Ghasemi, Y.; Ehteshamnia, A.; Nabavi, S.M.; Nabavi, S.F.; Ebrahimzadeh, M.A.; Pourmorad, F. Influence of environmental factors on antioxidant activity, phenol and flavonoids contents of walnut (Juglans regia L.) green husks. J. Med. Plant Res. 2011, 5, 1128–1133. [Google Scholar]
- Soobrattee, M.A.; Neergheen, V.S.; Luximon-Ramma, A.; Aruoma, O.I.; Bahorun, T. Phenolics as potential antioxidant therapeutic agents: Mechanism and actions. Mutat. Res. 2005, 579, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Prado, A.C.P.D.; Manion, B.A.; Seetharaman, K.; Deschamps, F.C.; Barrera Arellano, D.; Block, J.M. Relationship between antioxidant properties and chemical composition of the oil and the shell of pecan nuts [Carya illinoinensis (Wangenh) C. Koch]. Ind. Crops Prod. 2013, 45, 64–73. [Google Scholar] [CrossRef]
- Antolovich, M.; Prenzler, P.D.; Patsalides, E.; McDonald, S.; Robards, K. Methods for testing antioxidant activity. Analyst 2002, 127, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Gil, M.I.; Tomás-Barberán, F.A.; Hess-Pierce, B.; Holcroft, D.M.; Kader, A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000, 48, 4581–4589. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds gallic acid, ellagic acid, (+)-catechin, (−)-epicatechin, rutin, and EGCG are available from the authors. |
| Peak No. | RT (min) | Molecular Ion (m/z) | Fragment Ions (m/z) | Neutral Loss (amu) | Tentative Structural Assignment | Reference | Molecular Formula | ∆m (ppm) |
|---|---|---|---|---|---|---|---|---|
| 1 | 0.65 | 341.0879 | 179 | 162 | Caffeic acid hexoside | [9] | C15H18O9 | −1.76 |
| 2 | 0.84 | 331.0684 | 169 | 162 | Gallic acid hexoside | [9] | C13H16O10 | −5.74 |
| 3 | 0.94 | 481.0643 | 301 | 180 | HHDP-glucose | [9] | C20H18O14 | −5.20 |
| 4 | 1.15 | 169.0146 | 125 | 44 | Gallic acid | Standard a | C7H6O5 | −5.33 |
| 5 | 1.30 | 137.0229 | 107 | 30 | p-Hydroxybenzoic acid | [9] | C7H6O3 | 7.30 |
| 6 | 2.05 | 783.0727 | 481-301 | 302-482 | Bis-HHDP-glucose (pedunculagin/casuariin isomer) * | [30] | C34H24O22 | −5.87 |
| 7 | 2.75 | 451.1263 | 289 | 162 | Catechin hexoside | [9] | C21H24O11 | −5.10 |
| 8 | 3.13 | 451.1242 | 289 | 162 | Catechin hexoside | [9] | C21H24O11 | −0.44 |
| 9 | 3.20 | 783.0684 | 481-301 | 302-482 | Bis-HHDP-glucose (pedunculagin/casuariin isomer) * | [30] | C34H24O22 | −0.38 |
| 10 | 3.27 | 951.0760 | 907-783-481-301 | 44-168-470-650 | HHDP-valoneoyl-glucose (praecoxin A/platycariin isomer) * | [30] | C41H28O27 | −2.10 |
| 11 | 3.40 | 577.1337 | 425 | 152 | Procyanidin dimer B-type [(E)C→B→(E)C] | [9] | C30H26O12 | 1.56 |
| 12 | 3.75 | 183.0310 | 124 | 59 | Methyl gallate ∆ | [31] | C8H8O5 | −8.74 |
| 13 | 3.90 | 865.2006 | 577-289 | 288-576 | Procyanidin trimer (C1) [(E)C→B→(E)C→B→(E)C] | [9] | C45H38O18 | −3.01 |
| 14 | 3.97 | 483.0773 | 331-169 | 152-314 | Digalloyl-glucose | [30,32] | C20H20O14 | 0.41 |
| 15 | 4.04 | 289.0732 | 245-205-179 | 44-84-110 | (+)-Catechin | Standard | C15H14O6 | −6.92 |
| 16 | 4.59 | 633.0756 | 301 | 332 | Galloyl-HHDP-glucose (strictinin/isostrictinin) | [30,32] | C27H22O18 | −4.42 |
| 17 | 5.45 | 785.0768 | 483-301 | 302-484 | Digalloyl-HHDP-glucose (tellimagrandin I) | [30,32] | C34H26O22 | 8.92 |
| 18 | 5.64 | 289.0722 | 245-205-179 | 44-84-110 | (−)-Epicatechin | Standard | C15H14O6 | −3.46 |
| 19 | 5.78 | 463.0508 | 301 | 162 | Ellagic acid hexoside | [9] | C20H16O13 | 1.08 |
| 20 | 6.36 | 300.9961 | 217 | 84 | Ellagic acid | Standard | C14H6O8 | 7.64 |
| 21 | 6.51 | 433.0413 | 301 | 132 | Ellagic acid pentose | [9] | C19H14O12 | −1.39 |
| 22 | 6.64 | 565.2089 | 403 | 189 | Glansreginin B * | [33] | C24H38O15 | 7.78 |
| 23 | 6.70 | 615.1000 | 463-301 | 152-314 | Digalloyl ellagic acid | [9] | C28H24O16 | −2.28 |
| 24 | 6.79 | 477.0692 | 315-300 | 162-177 | Methyl ellagic acid hexoside | [9] | C21H18O13 | −4.82 |
| 25 | 6.90 | 609.1041 | 301 | 308 | Ellagic acid rutinoside # | [34] | C26H26O17 | 8.37 |
| 26 | 7.02 | 441.0782 | 289 | 152 | (Epi)catechin gallate | [9] | C22H18O10 | 9.07 |
| 27 | 7.21 | 491.0811 | 328 | 163 | Dimethyl ellagic acid hexoside | [9] | C22H20O13 | 3.05 |
| 28 | 7.37 | 649.0724 | 605-481-301 | 44-168-348 | Valoneoyl-glucose * | [35] | C27H22O19 | −7.24 |
| 29 | 7.44 | 447.0595 | 301 | 146 | Ellagic acid rhamnoside # | [36] | C20H16O12 | −6.93 |
| 30 | 7.48 | 315.0153 | 301 | 14 | Methyl ellagic acid | [19] | C15H8O8 | −3.81 |
| 31 | 7.51 | 447.0578 | 315 | 132 | Methyl ellagic acid pentose | [9] | C20H16O12 | −3.13 |
| 32 | 7.79 | 592.2066 | 403-343-241 | 189-249-351 | Glansreginin A * | [33] | C28H35NO13 | −6.08 |
| 33 | 7.86 | 585.0507 | 433-301 | 152-284 | Ellagic acid galloyl pentose | [9] | C26H18O16 | 1.71 |
| 34 | 8.09 | 457.0808 | 293-163 | 164-294 | Epigallocatechin gallate (EGCG) | Standard | C22H18O11 | −8.09 |
| 35 | 8.28 | 487.0124 | 469-425-301 | 18-62-186 | Valoneic acid dilactone hydrate | [9] | C21H12O14 | 5.13 |
| 36 | 8.44 | 599.0625 | 447-315 | 152-284 | Methyl ellagic acid galloyl pentose | [9] | C27H20O16 | 8.01 |
| 37 | 8.67 | 599.0670 | 447-315 | 152-284 | Methyl ellagic acid galloyl pentose | [9] | C27H20O16 | 0.50 |
| 38 | 8.86 | 599.0684 | 447-315 | 152-284 | Methyl ellagic acid galloyl pentose | [9] | C27H20O16 | −1.84 |
| 39 | 9.17 | 329.0271 | 314-301 | 15-28 | Dimethyl ellagic acid | [9] | C16H10O8 | 7.90 |
| 40 | 9.34 | 329.0325 | 314-301 | 15-28 | Dimethyl ellagic acid | [9] | C16H10O8 | −8.51 |
| Antioxidant Capacity | TPC | CT | TFC | Individual Phenolics 1 | ||||
|---|---|---|---|---|---|---|---|---|
| GA | C | EC | EGCG | EA | ||||
| DPPH | 0.748 ** | 0.442 ** | 0.426 ** | 0.538 ** | 0.799 ** | 0.721 ** | 0.469 ** | 0.471 ** |
| ABTS | 0.841 ** | 0.497 ** | 0.506 ** | 0.662 ** | 0.871 ** | 0.851 ** | 0.597 ** | 0.606 ** |
| Sample Name | DAFB 1 | Sample Stage | Description 2 | Daily Mean Temperature of Sampling Interval (°C) 3 | Daily Mean Sunshine Duration of Sampling Interval (h) 3 |
|---|---|---|---|---|---|
| S1 | 85 | Water stage | Appearance of the kernel | 32.09 | 10.34 |
| S2 | 95 | Water stage | Quick expansion of the kernel | 30.38 | 8.38 |
| S3 | 105 | Water stage | Quick expansion of the kernel | 30.73 | 10.35 |
| S4 | 115 | Milk stage | Kernel became milky | 29.67 | 6.90 |
| S5 | 125 | Milk stage | Kernel became milky | 26.69 | 9.04 |
| S6 | 135 | Dough stage | Kernel became oily | 24.30 | 5.10 |
| S7 | 145 | Dough stage | Kernel became oily | 22.80 | 6.47 |
| S8 | 155 | Dough stage | Kernel ripening | 21.93 | 3.03 |
| S9 | 165 | Kernel stage | Kernel ripening | 18.27 | 2.42 |
| S10 | 175 | Kernel stage | Over ripeness | 19.12 | 1.00 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, X.; Luo, H.; Xu, M.; Zhai, M.; Guo, Z.; Qiao, Y.; Wang, L. Dynamic Changes in Phenolics and Antioxidant Capacity during Pecan (Carya illinoinensis) Kernel Ripening and Its Phenolics Profiles. Molecules 2018, 23, 435. https://doi.org/10.3390/molecules23020435
Jia X, Luo H, Xu M, Zhai M, Guo Z, Qiao Y, Wang L. Dynamic Changes in Phenolics and Antioxidant Capacity during Pecan (Carya illinoinensis) Kernel Ripening and Its Phenolics Profiles. Molecules. 2018; 23(2):435. https://doi.org/10.3390/molecules23020435
Chicago/Turabian StyleJia, Xiaodong, Huiting Luo, Mengyang Xu, Min Zhai, Zhongren Guo, Yushan Qiao, and Liangju Wang. 2018. "Dynamic Changes in Phenolics and Antioxidant Capacity during Pecan (Carya illinoinensis) Kernel Ripening and Its Phenolics Profiles" Molecules 23, no. 2: 435. https://doi.org/10.3390/molecules23020435
APA StyleJia, X., Luo, H., Xu, M., Zhai, M., Guo, Z., Qiao, Y., & Wang, L. (2018). Dynamic Changes in Phenolics and Antioxidant Capacity during Pecan (Carya illinoinensis) Kernel Ripening and Its Phenolics Profiles. Molecules, 23(2), 435. https://doi.org/10.3390/molecules23020435







