Novel Topologically Complex Scaffold Derived from Alkaloid Haemanthamine
Abstract
1. Introduction
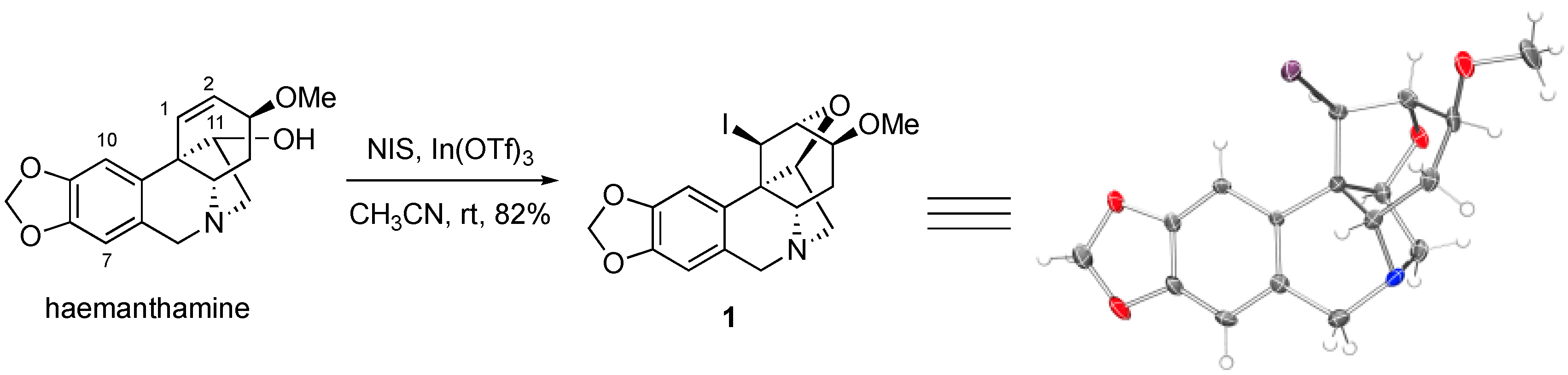
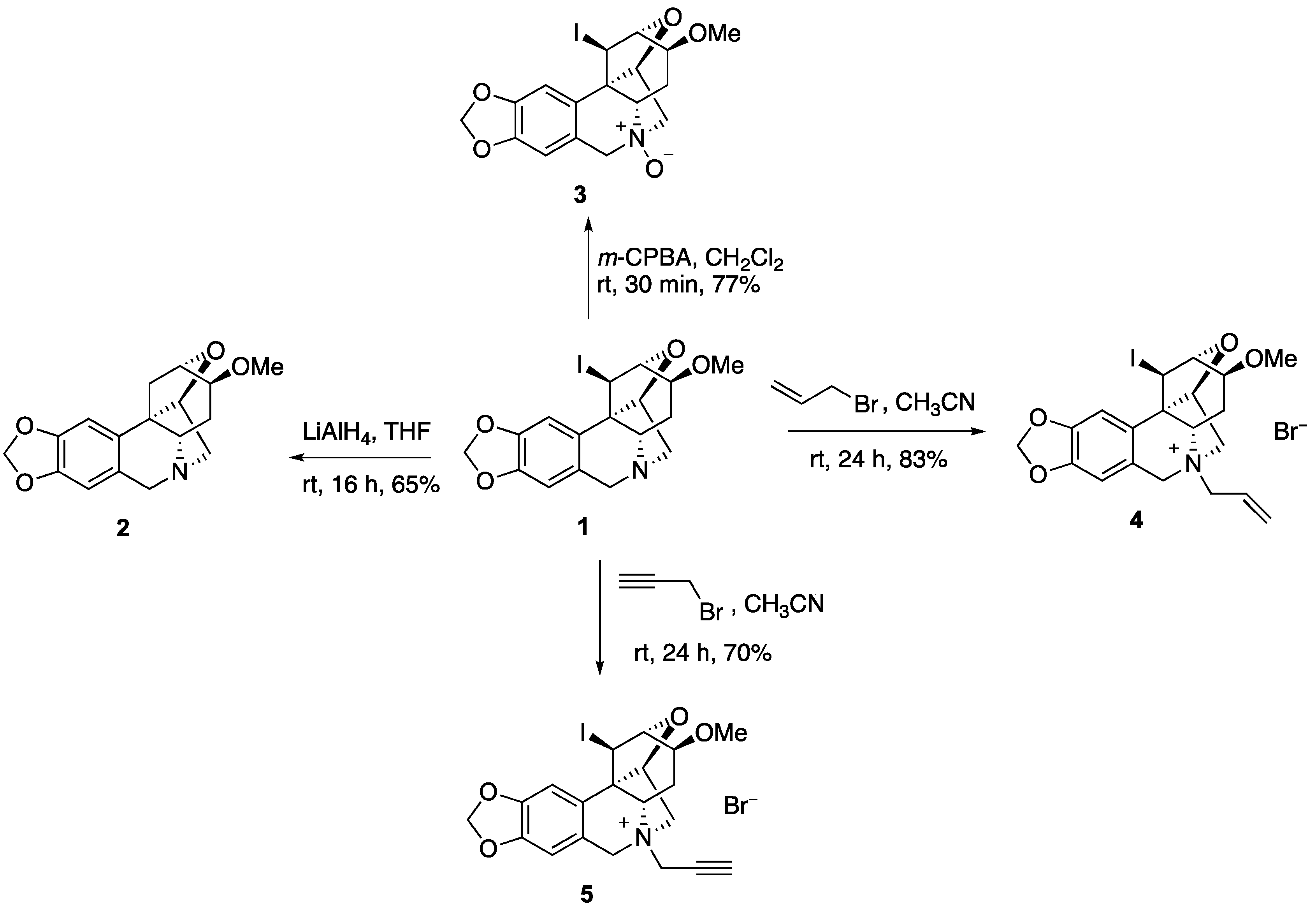
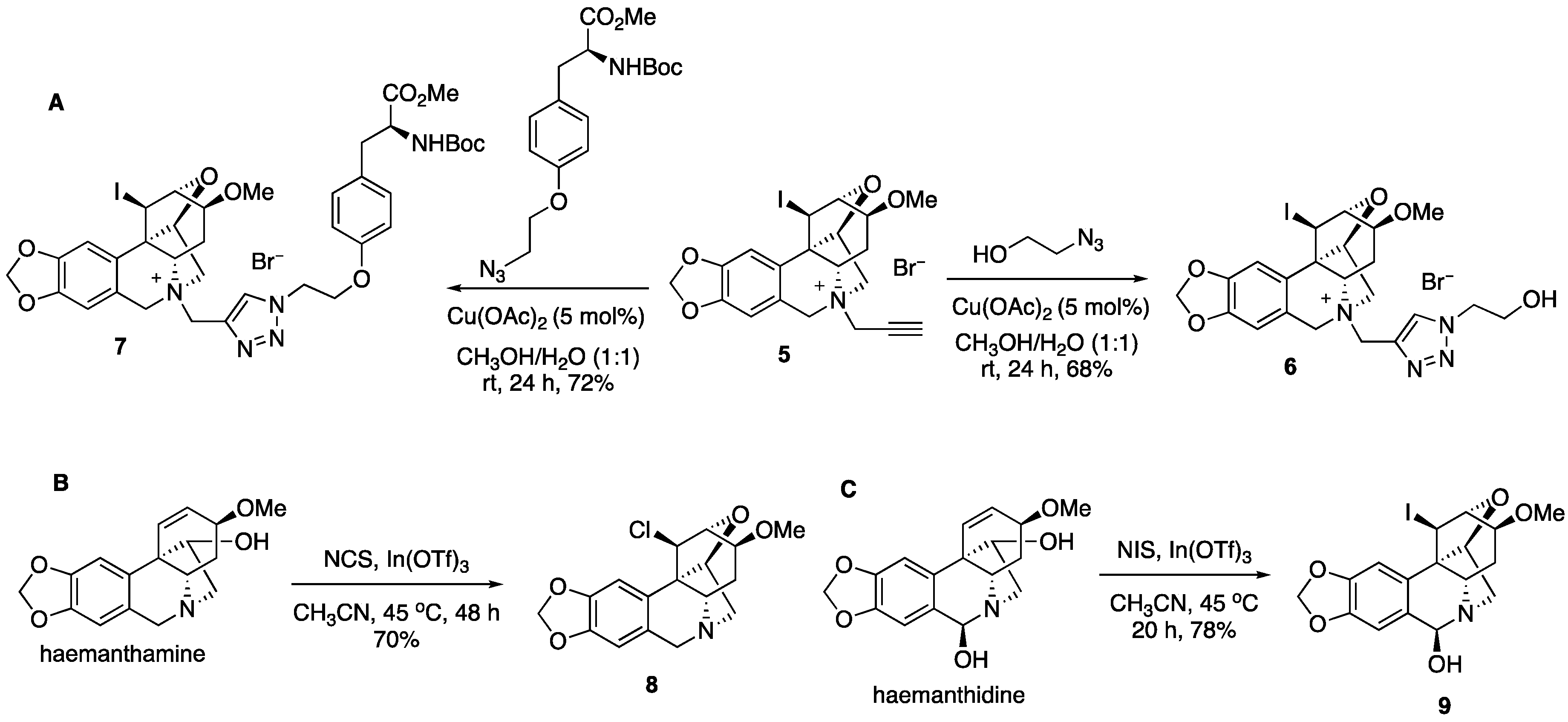
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. Crystallographic Information
3.3. Experimental Procedures
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- Galloway, W.R.; Isidro-Llobet, A.; Spring, D.R. Diversity-oriented synthesis as a tool for the discovery of novel biologically active small molecules. Nat. Commun. 2010, 1, 80. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Castro, M.; Zimmerman, S.; Sankar, M.G.; Kumar, K. Scaffold diversity synthesis and its application in probe and drug discovery. Angew. Chem. Int. Ed. 2016, 55, 7586–7605. [Google Scholar] [CrossRef] [PubMed]
- McLeod, M.C.; Singh, G.; Plampin, J.N., III; Rane, D.; Wang, J.L.; Day, V.W.; Aube, J. Probing chemical space with alkaloid-inspired libraries. Nat. Chem. 2014, 6, 133–140. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cui, J.; Hao, J.; Ulanovkaya, O.A.; Dundas, J.; Liang, J.; Kozmin, S.A. Creation and manipulation of common functional groups en route to a skeletally diverse chemical library. PNAS 2011, 108, 6763–6768. [Google Scholar] [CrossRef] [PubMed]
- Borzilleri, R.M.; Vite, G.D. Case history: Discovery of ixabepilone (IXEMPRA™), a first-in-class epothilone analogue for treatment of metastatic breast cancer. Annu. Rep. Med. Chem. 2009, 44, 301–322. [Google Scholar]
- Fasolo, A.; Sessa, C. Current and future directions in mammalian target of rapamycin inhibitor development. Expert Opin. Investig. Drugs. 2011, 20, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Morrison, K.C.; Hergenrother, P.J. Natural products as starting points for the synthesis of complex and diverse compounds. Nat. Prod. Rep. 2014, 31, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Pelish, H.E.; Westwood, N.J.; Feng, Y.; Kirchhausen, T.; Shair, M.D. Use of biomimetic diversity-oriented synthesis to discover galanthamine-like molecules with biological properties beyond those of natural product. J. Am. Chem. Soc. 2001, 123, 6740–6741. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Kiuchi, M.; Tallarico, J.A.; Schreiber, S.L. Small-molecule diversity using a skeletal transformation strategy. Org. Lett. 2005, 7, 2535–2538. [Google Scholar] [CrossRef] [PubMed]
- Balthaser, B.R.; Maloney, M.C.; Beeler, A.B.; Porco, J.A., Jr.; Snyder, J.K. Remodeling of the natural product fumagillol employing a reaction discovery approach. Nat. Chem. 2011, 3, 969–973. [Google Scholar] [CrossRef] [PubMed]
- Appendino, G.C.; Tron, G.C.; Jarevang, T.; Sterner, O. Unnatural natural products from the transannular cyclization of lathyrane diterpenes. Org. Lett. 2001, 3, 1609–1612. [Google Scholar] [CrossRef] [PubMed]
- de la Torre, M.C.; Garcia, I.; Sierra, M.A. Diversity oriented synthesis of hispanane-like terpene derivatives from (R)-(+)-scareolide. Chem. Eur. J. 2005, 11, 3659–3667. [Google Scholar] [CrossRef] [PubMed]
- Kitayama, T.; Iwabuchi, R.; Minagawa, S.; Shiomi, F.; Cappiello, J.; Sawada, S.; Utsumi, R.; Okamoto, T. Unprecedented olefin-dependent histidine-kinase inhibitory of zerumbone ring-opening material. Bioorg. Med. Chem. Lett. 2004, 14, 5943–5946. [Google Scholar] [CrossRef] [PubMed]
- Huisgens, R.W., III; Morrison, K.C.; Hicklin, R.W.; Flood, T.A., Jr.; Richter, M.F.; Hergenrother, P.J. A ring-distortion strategy to construct stereochemically complex and structurally diverse compounds from natural products. Nat. Chem. 2013, 5, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Van Goietsenoven, G.; Andolfi, A.; Lallemand, B.; Cimmino, A.; Lamoral-Theys, D.; Gras, T.; Abou-Donia, A.; Dubois, J.; Lefranc, F.; Mathieu, V.; et al. Amaryllidaceae alkaloids belonging to different structural subgroups display activity against apoptosis-resistant cancer cells. J. Nat. Prod. 2010, 73, 1223–1227. [Google Scholar] [CrossRef] [PubMed]
- Cedron, J.C.; Gutierrez, D.; Flores, N.; Ravelo, A.G.; Estevez-Braun, A. Synthesis and antimalarial activity of new haemanthamine-type derivatives. Bioorg. Med. Chem. 2012, 20, 5464–5472. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.-Y.; Li, J.; Peddibholta, S.; Romo, D. Mild arming and derivatization of natural products via an In(OTf)3-catalyzed arene iodination. Org. Lett. 2010, 12, 2104–2107. [Google Scholar] [CrossRef] [PubMed]
- Cedron, J.C.; Estevez-Braun, A.; Ravelo, A.G.; Guiterez, D.; Flores, N.; Bucio, M.A.; Perez-Hernandez, N.; Joseph-Nathan, P. Bioactive montanine derivatives from halide-induced rearrangements of haemanthamine-type Alkaloids. Absolute configuration by VCD. Org. Lett. 2009, 11, 1491–1494. [Google Scholar] [CrossRef] [PubMed]
- Inubushi, Y.; Fales, H.M.; Warnhoff, E.W.; Wildman, W.C. Structures of montanine, coccinine, and manthine. J. Org. Chem. 1960, 25, 2153–2164. [Google Scholar] [CrossRef]
- Pellegrino, S.; Meyer, M.; Zorbas, C.; Bouchta, S.A.; Saraf, K.; Pelly, S.C.; Yusupova, G.; Evidente, A.; Mathieu, V.; Kornienko, A.; et al. The Amaryllidacae alkaloid haemanthamine binds the eukaryotic ribosome to repress cancer cell growh. Structure 2018, in press. [Google Scholar]
Sample Availability: Alkaloid haemanthamine is available from the authors. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Govindaraju, K.; Masi, M.; Colin, M.; Mathieu, V.; Evidente, A.; Hudnall, T.W.; Kornienko, A. Novel Topologically Complex Scaffold Derived from Alkaloid Haemanthamine. Molecules 2018, 23, 255. https://doi.org/10.3390/molecules23020255
Govindaraju K, Masi M, Colin M, Mathieu V, Evidente A, Hudnall TW, Kornienko A. Novel Topologically Complex Scaffold Derived from Alkaloid Haemanthamine. Molecules. 2018; 23(2):255. https://doi.org/10.3390/molecules23020255
Chicago/Turabian StyleGovindaraju, Karthik, Marco Masi, Margaux Colin, Veronique Mathieu, Antonio Evidente, Todd W. Hudnall, and Alexander Kornienko. 2018. "Novel Topologically Complex Scaffold Derived from Alkaloid Haemanthamine" Molecules 23, no. 2: 255. https://doi.org/10.3390/molecules23020255
APA StyleGovindaraju, K., Masi, M., Colin, M., Mathieu, V., Evidente, A., Hudnall, T. W., & Kornienko, A. (2018). Novel Topologically Complex Scaffold Derived from Alkaloid Haemanthamine. Molecules, 23(2), 255. https://doi.org/10.3390/molecules23020255









