Proteomic Analysis of Differentially-Expressed Proteins in the Liver of Streptozotocin-Induced Diabetic Rats Treated with Parkia biglobosa Protein Isolate
Abstract
1. Introduction
2. Results
3. Discussion and Conclusions
4. Materials and Methods
4.1. Chemicals
4.2. Plant Material and Extract Preparation
4.3. Experimental Animals and Experimental Design
4.4. Induction of Diabetes and Experimental Design
4.5. Processing of Tissue Samples and Protein Extraction
4.6. 1D SDS-PAGE, In-Gel Digestion and Peptide Extraction
4.7. nLC Procedure
4.8. Mass Spectrometry
4.9. Data Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Singh, R.; Bhardwaj, P.; Sharma, P. Antioxidant and toxicological evaluation of Cassia sopherain streptozotocin-induced diabetic Wistar rats. Pharmacogn. Res. 2013, 5, 225–232. [Google Scholar]
- Ogunyinka, B.I.; Oyinloye, B.E.; Adenowo, A.F.; Kappo, A.P. Potentials of some plant-derived foods in the management of diabetes and associated Complications. Afr. J. Tradit. Complement. Altern. Med. 2015, 12, 12–20. [Google Scholar] [CrossRef]
- Chang, M.H.; Valdez, R.; Ned, R.M.; Liu, T.; Yang, Q.; Yesupriya, A.; Dowling, N.F.; Meigs, J.B.; Bowen, M.S.; Khoury, M.J. Influence of familial risk on diabetes risk–reducing behaviours among US adults without diabetes. Diabetes Care 2011, 34, 2393–2399. [Google Scholar] [CrossRef] [PubMed]
- Surya, S.; Salam, A.D.; Tomy, D.V.; Carla, B.; Kumar, R.A.; Sunil, C. Diabetes mellitus and medicinal plants-a review. Asian Pac. J. Trop. Dis. 2014, 4, 337–347. [Google Scholar] [CrossRef]
- Arumugam, G.; Manjula, P.; Paari, N. A review: Anti diabetic medicinal plants used for diabetes mellitus. J. Acute Dis. 2013, 2, 196–200. [Google Scholar] [CrossRef]
- Ramachandran, S.; Rajasekaran, A.; Manisenthilkumar, K.T. Investigation of hypoglycemic, hypolipidemic and antioxidant activities of aqueous extract of Terminalia paniculata bark in diabetic rats. Asian Pac. J. Trop. Biomed. 2012, 2, 262–268. [Google Scholar] [CrossRef]
- Karthik, D.; Ilavenil, S.; Kaleeswaran, B.; Ravikumar, S. Analysis of Modification of Liver Proteome in Diabetic Rats by 2D Electrophoresis and MALDI-TOF-MS. Ind. J. Clin. Biochem. 2012, 27, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Colette, C.; Monnier, L. Acute glucose fluctuations and chronic sustained hyperglycemia as risk factors for cardiovascular diseases in patients with type 2 diabetes. Horm. Metab. Res. 2007, 39, 683–686. [Google Scholar] [CrossRef] [PubMed]
- Monnier, L.; Mas, E.; Ginet, C.; Michel, F.; Villon, L.; Cristol, J.P.; Colette, C. Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycaemia in patients with type 2 diabetes. J. Am. Med. Assoc. 2006, 295, 1681–1687. [Google Scholar] [CrossRef] [PubMed]
- Oyinloye, B.E.; Adenowo, A.F.; Kappo, A.P. Reactive Oxygen Species, Apoptosis, Antimicrobial Peptides and Human Inflammatory Diseases. Pharmaceuticals 2015, 8, 151–175. [Google Scholar] [CrossRef] [PubMed]
- Maiti, R.; Jana, D.; Das, U.K.; Ghosh, D. Antidiabetic effect of aqueous extract of seed of Tamarindus indica in streptozotocin-induced diabetic rats. J. Ethnopharmacol. 2004, 92, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Grover, J.K.; Yadav, S.; Vats, V. Medicinal plants of India with anti-diabetic potential. J. Ethnopharmacol. 2002, 81, 81–100. [Google Scholar] [CrossRef]
- Adi, K.; Metowogo, K.; Mouzou, A.; Lawson-Evi, P.; Eklu-Gadegbeku, K.; Agbonon, A.; Lamboni, C.; Essien, K.; Aklikokou, K.; Gbeassor, M.; et al. Evaluation of cardioprotective effects of Parkia biglobosa (jacq. benth) Mimosaceae stem bark. J. App. Pharm. Sci. 2013, 3, 60–64. [Google Scholar] [CrossRef]
- Odetola, A.A.; Akinloye, O.; Egunjobi, C.; Adekunle, W.A.; Ayoola, A.O. Possible antidiabetic and antihyperlipidaemic effect of fermented Parkia biglobosa (JACQ) extract in alloxan-induced diabetic rats. Clin. Exp. Pharmacol. Physiol. 2006, 33, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Ogunyinka, B.I.; Oyinloye, B.E.; Osunsanmi, F.O.; Kappo, A.P.; Opoku, A.R. Protective effects of Parkia biglobosa protein isolate on streptozotocin-induced hepatic damage and oxidative stress in diabetic male rats. Molecules 2017, 22, 1654. [Google Scholar] [CrossRef] [PubMed]
- Visweswara Rao, P.; Madhavi, K.; Dhananjaya Naidu, M.; Gan, S.H. Rhinacanthus nasutus Improves the Levels of Liver Carbohydrate, Protein, Glycogen, and Liver Markers in Streptozotocin-Induced Diabetic Rats. Evid. Based Complement. Altern. Med. 2013, 2013, 102901. [Google Scholar]
- Karthik, D.; Ravikumar, S. Characterization of the brain proteome of rats with diabetes mellitus through two-dimensional electrophoresis and mass spectrometry. Brain Res. 2011, 1371, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Nagappan, A.; Park, H.S.; Park, K.I.; Kim, J.A.; Hong, G.E.; Kang, S.R.; Zhang, J.; Kim, E.H.; Lee, W.S.; Won, C.K.; et al. Proteomic analysis of differentially expressed proteins in vitamin C-treated AGS cells. BMC Biochem. 2013, 14, 24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, L.; Ma, Z.; Cheng, J.; Liu, J. Anti-Diabetic, Anti-Oxidant and Anti-Hyperlipidemic Activities of Flavonoids from Corn Silk on STZ-Induced Diabetic Mice. Molecules 2015, 21, 7. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Tripathi, P.; Pandey, R.; Srivatava, R.; Goswami, S. Alternative therapies useful in the management of diabetes: A systematic review. J. Pharm. Bioallied. Sci. 2011, 3, 504. [Google Scholar] [PubMed]
- Dinić, S.; Uskoković, A.; Mihailović, M.; Grdović, N.; Arambašić, J.; Marković, J.; Poznanović, G.; Vidaković, M. Ameliorating effects of antioxidative compounds from four plant extracts in experimental models of diabetes. J. Serb. Chem. Soc. 2013, 78, 365–380. [Google Scholar] [CrossRef]
- López-Villar, E.; Martos-Moreno, G.Á.; Chowen, J.A.; Okada, S.; Kopchick, J.J.; Argente, J. A proteomic approach to obesity and type 2 diabetes. J. Cell. Mol. Med. 2015, 19, 1455–1470. [Google Scholar] [CrossRef] [PubMed]
- Awdeh, Z.L.; Yunis, E.J.; Audeh, M.J.; Fici, D.; Pugliese, A.; Larsen, C.E.; Alper, C.A. A genetic explanation for the rising incidence of type 1 diabetes, a polygenic disease. J. Autoimmun. 2006, 27, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Gerich, J.E. The genetic basis of type 2 diabetes mellitus: Impaired insulin secretion versus impaired insulin sensitivity. Endocr. Rev. 1998, 19, 491–503. [Google Scholar] [CrossRef] [PubMed]
- Lao, Y.; Wang, X.; Xu, N.; Zhang, H.; Xu, H. Application of proteomics to determine the mechanism of action of traditional Chinese medicine remedies. J. Ethnopharmacol. 2014, 155, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.B.; Vijayakumar, M. Anti-Hyperglycemic and Antioxidant Effect of Saraca asoca (Roxb. De Wilde) Flowers in Streptozotocin-Nicotinamide Induced Diabetic Rats: A Therapeutic Study. J. Bioanal. Biomed. 2014, S12, 3. [Google Scholar]
- Maulik, N.; Das, D.K. Emerging potential of thioredoxin and thioredoxin interacting proteins in various disease conditions. Biochim. Biophys. Acta 2008, 1780, 1368–1382. [Google Scholar] [CrossRef] [PubMed]
- Schulze, P.C.; Yoshioka, J.; Takahashi, T.; He, Z.; King, G.L.; Lee, R.T. Hyperglycemia promotes oxidative stress through inhibition of thioredoxin function by thioredoxin-interacting protein. J. Biol. Chem. 2004, 279, 30369–30374. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhong, Q.; Yang, T.; Xie, X.; Chen, M. The role of profilin-1 in endothelial cell injury induced by advanced glycation end products (AGEs). Cardiovasc. Diabetol. 2013, 12, 141. [Google Scholar] [CrossRef] [PubMed]
- Pae, M.; Romeo, G.R. The multifaceted role of profilin-1 in adipose tissue inflammation and glucose homeostasis. Adipocyte 2014, 3, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Romeo, G.R.; Kazlauskas, A. Oxysterol and diabetes activate STAT3 and control endothelial expression of profilin-1 via OSBP1. J. Biol. Chem. 2008, 283, 9595–9605. [Google Scholar] [CrossRef] [PubMed]
- Romeo, G.; Frangioni, J.V.; Kazlauskas, A. Profilin acts downstream of LDL to mediate diabetic endothelial cell dysfunction. FASEB J. 2004, 18, 725–727. [Google Scholar] [CrossRef] [PubMed]
- Timón-Gómez, A.; Proft, M.; Pascual-Ahuir, A. Differential regulation of mitochondrial pyruvate carrier genes modulates respiratory capacity and stress tolerance in yeast. PLoS ONE 2013, 8, e79405. [Google Scholar] [CrossRef] [PubMed]
- Gray, L.R.; Rauckhorst, A.J.; Taylor, E.B. A Method for Multiplexed Measurement of Mitochondrial Pyruvate Carrier Activity. J. Biol. Chem. 2016, 291, 7409–7417. [Google Scholar] [CrossRef] [PubMed]
- Schell, J.C.; Rutter, J. The long and winding road to the mitochondrial pyruvate carrier. Cancer Metab. 2013, 1, 6. [Google Scholar] [PubMed]
- Divakaruni, A.S.; Wiley, S.E.; Rogers, G.W.; Andreyev, A.Y.; Petrosyan, S.; Loviscach, M.; Wall, E.A.; Yadava, N.; Heuck, A.P.; Ferrick, D.A.; et al. Thiazolidinediones are acute, specific inhibitors of the mitochondrial pyruvate carrier. Proc. Natl. Acad. Sci. USA 2013, 110, 5422–5427. [Google Scholar] [CrossRef] [PubMed]
- Patterson, J.N.; Cousteils, K.; Lou, J.W.; Fox, J.E.M.; MacDonald, P.E.; Joseph, J.W. Mitochondrial metabolism of pyruvate is essential for regulating glucose-stimulated insulin secretion. J. Biol. Chem. 2014, 289, 13335–13346. [Google Scholar] [CrossRef] [PubMed]
- Bricker, D.K.; Taylor, E.B.; Schell, J.C.; Orsak, T.; Boutron, A.; Chen, Y.C.; Cox, J.E.; Cardon, C.M.; Van Vranken, J.G.; Dephoure, N.; et al. A mitochondrial pyruvate carrier required for pyruvate uptake in yeast, Drosophila, and humans. Science 2012, 337, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Bender, T.; Martinou, J.C. The mitochondrial pyruvate carrier in health and disease: To carry or not to carry? Biochim. Biophys. Acta 2016, 1863, 2436–2442. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, M.; Hotamisligil, G.S. Fatty acid-binding proteins: Role in metabolic diseases and potential as drug targets. Nat. Rev. Drug Discov. 2008, 7, 489–503. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Cao, H.; Kono, K.; Gorgun, C.Z.; Furuhashi, M.; Uysal, K.T.; Cao, Q.; Atsumi, G.; Malone, H.; Krishnan, B.; et al. Adipocyte/macrophage fatty acid binding proteins control integrated metabolic responses in obesity and diabetes. Cell Metab. 2005, 1, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Melki, S.A.; Abumrad, N.A. Expression of the adipocyte fatty acid-binding protein in streptozotocin-diabetes: Effects of insulin deficiency and supplementation. J. Lip. Res. 1993, 34, 1527–1534. [Google Scholar]
- Nkosi, C.Z.; Opoku, A.R.; Terblanche, S.E. Effects of pumpkin seed (Cucurbita pepo) protein isolate on the activity levels of certain plasma enzymes in CCI4—Induced liver injury in low-protein fed rats. Phytother. Res. 2005, 19, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Hule, A.K.; Shah, A.S.; Gambhire, M.N.; Juvekar, A.R. An evaluation of the antidiabetic effects of Elaeocarpus ganitrus in experimental animals. Indian J. Pharmacol. 2011, 43, 56–59. [Google Scholar] [PubMed]
- Li, C.; Guo, Z.; Zhao, R.; Sun, W.; Xie, M. Proteomic Analysis of Liver Proteins in a Rat Model of Chronic Restraint Stress-Induced Depression. Biomed. Res. Int. 2017, 2017, 7508316. [Google Scholar] [CrossRef] [PubMed]
- Bakala, H.; Ladouce, R.; Baraibar, M.A.; Friguet, B. Differential expression and glycative damage affect specific mitochondrial proteins with aging in rat liver. BBA-Mol. Basis Dis. 2013, 1832, 2057–2067. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compound PBPi are available from the authors. |
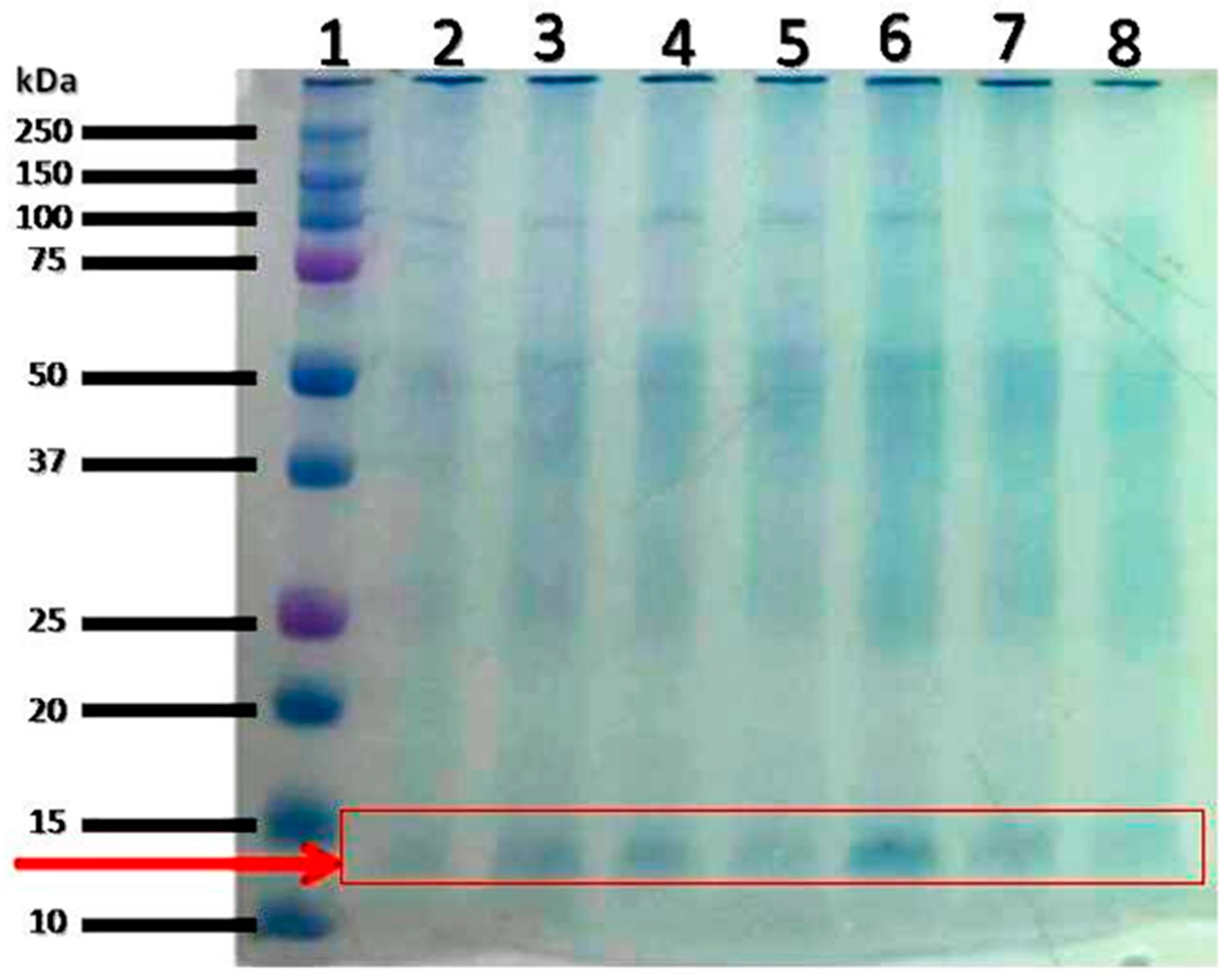
| S/N | Accession No. | Protein Name | Observed MW(kDa)/pI |
|---|---|---|---|
| 1 | THIO_RAT | Thioredoxin OS = Rattus norvegicus GN = Txn PE = 1 SV = 2 | 11.70/4.64 |
| 2 | PROF1_RAT | Profilin-1 OS = Rattus norvegicus GN = Pfn1 PE = 1 SV = 2 | 14.90/9.43 |
| 3 | MPC1_RAT | Mitochondrial pyruvate carrier 1 OS = Rattus norvegicus GN = Mpc1 PE = 2 SV=1 | 12.40/10.40 |
| 4 | FABPL_RAT | Fatty acid-binding protein, liver OS = Rattus norvegicus GN = Fabp1 PE = 1 SV = 1 | 14.30/8.96 |
| S/N | Accession No. | Control | STZ | STZ PI 200 | STZ PI 400 | ||||
|---|---|---|---|---|---|---|---|---|---|
| MOWSE Score | No. of Peptides/Seq. Coverage | MOWSE Score | No. of Peptides/Seq. Coverage | MOWSE Score | No. of Peptides/Seq. Coverage | MOWSE Score | No. of Peptides/Seq. Coverage | ||
| 1 | THIO_RAT | 65.90 | 1/8.60 (%) | - | _ | - | - | - | - |
| 2 | PROF1_RAT | 24.62 | 1/7.90 (%) | 266.82 | 6/36.70 (%) | - | - | - | - |
| 3 | MPC1_RAT | - | - | - | - | - | - | 23.47 | 1/7.30 (%) |
| 4 | FABPL_RAT | 275.54 | 7/33.10 (%) | 23.51 | 1/4.80 (%) | 68.58 | 1/14.20 (%) | 126.67 | 3/31.50 (%) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogunyinka, B.I.; Oyinloye, B.E.; Osunsanmi, F.O.; Opoku, A.R.; Kappo, A.P. Proteomic Analysis of Differentially-Expressed Proteins in the Liver of Streptozotocin-Induced Diabetic Rats Treated with Parkia biglobosa Protein Isolate. Molecules 2018, 23, 156. https://doi.org/10.3390/molecules23020156
Ogunyinka BI, Oyinloye BE, Osunsanmi FO, Opoku AR, Kappo AP. Proteomic Analysis of Differentially-Expressed Proteins in the Liver of Streptozotocin-Induced Diabetic Rats Treated with Parkia biglobosa Protein Isolate. Molecules. 2018; 23(2):156. https://doi.org/10.3390/molecules23020156
Chicago/Turabian StyleOgunyinka, Bolajoko Idiat, Babatunji Emmanuel Oyinloye, Foluso Oluwagbemiga Osunsanmi, Andrew Rowland Opoku, and Abidemi Paul Kappo. 2018. "Proteomic Analysis of Differentially-Expressed Proteins in the Liver of Streptozotocin-Induced Diabetic Rats Treated with Parkia biglobosa Protein Isolate" Molecules 23, no. 2: 156. https://doi.org/10.3390/molecules23020156
APA StyleOgunyinka, B. I., Oyinloye, B. E., Osunsanmi, F. O., Opoku, A. R., & Kappo, A. P. (2018). Proteomic Analysis of Differentially-Expressed Proteins in the Liver of Streptozotocin-Induced Diabetic Rats Treated with Parkia biglobosa Protein Isolate. Molecules, 23(2), 156. https://doi.org/10.3390/molecules23020156






