Comparison of Physicochemical Properties of Starches from Nine Chinese Chestnut Varieties
Abstract
1. Introduction
2. Results and Discussion
2.1. Weight and Starch Content of Kernels
2.2. Morphology and Size Distribution of Starch Granule
2.3. Iodine Absorption Spectrum and Amylose Content of Starch
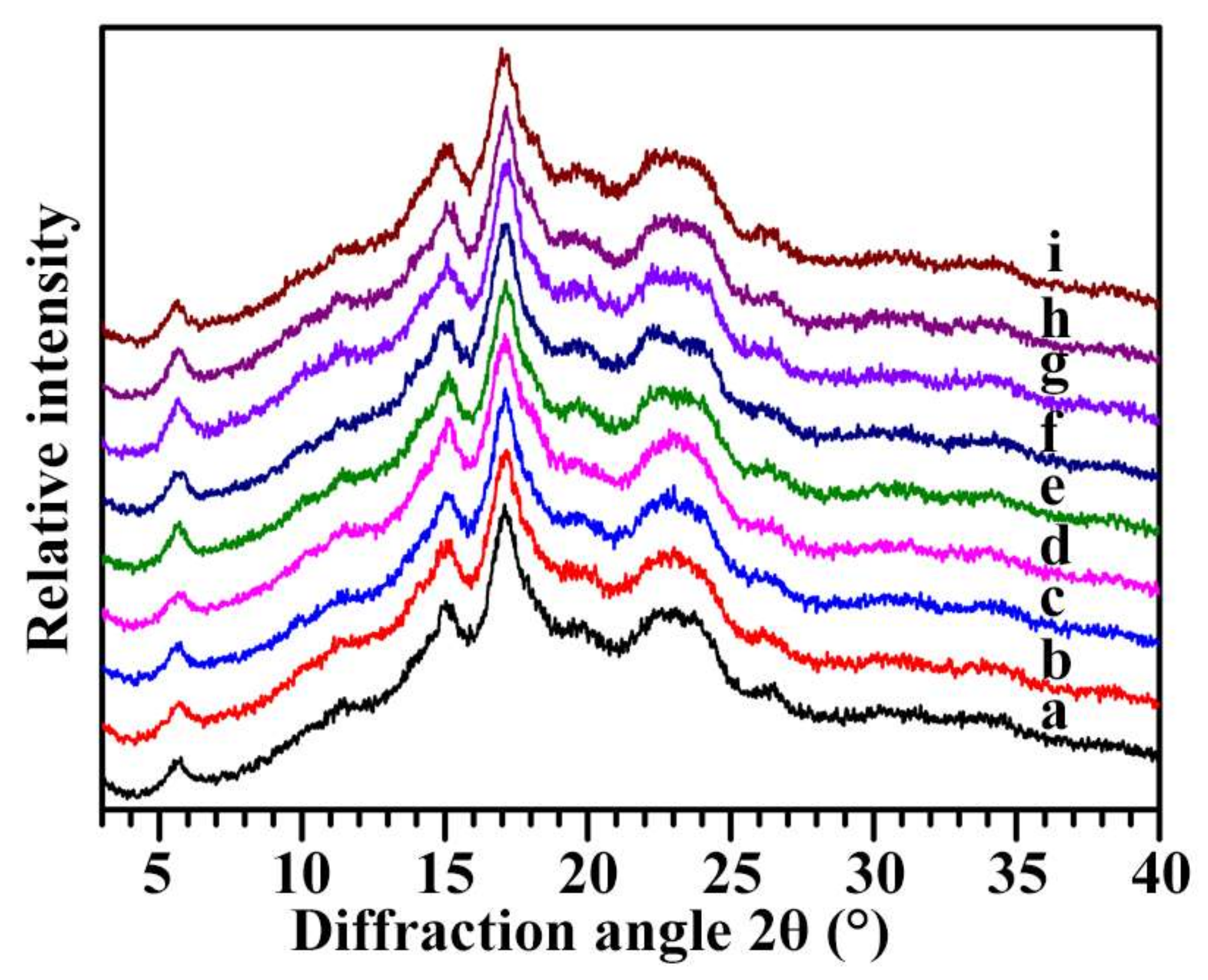
2.4. Crystalline Structure of Starch
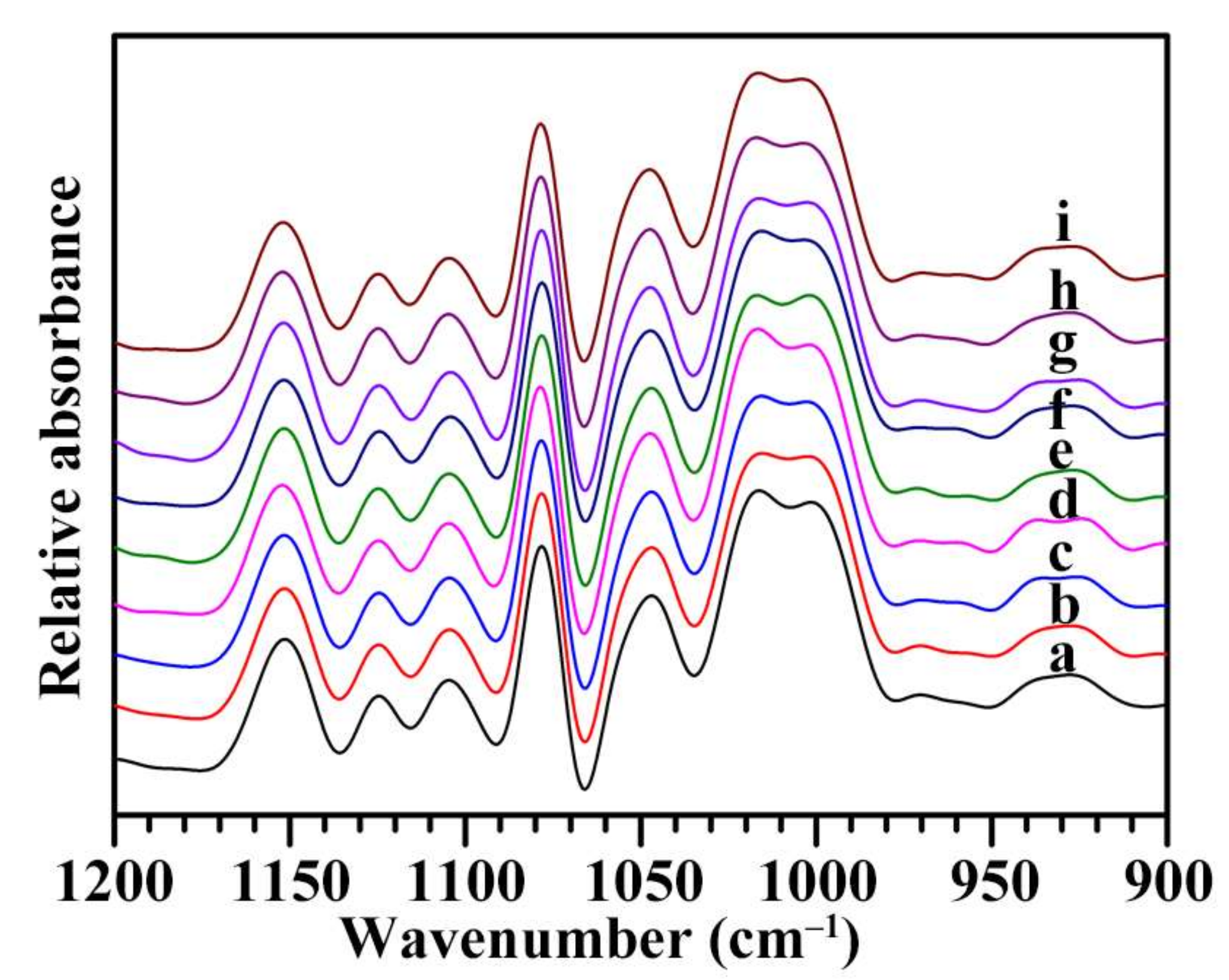
2.5. Short-Ranged Ordered Structure of Starch
2.6. Lamellar Structure of Starch
2.7. Thermal Properties of Starch
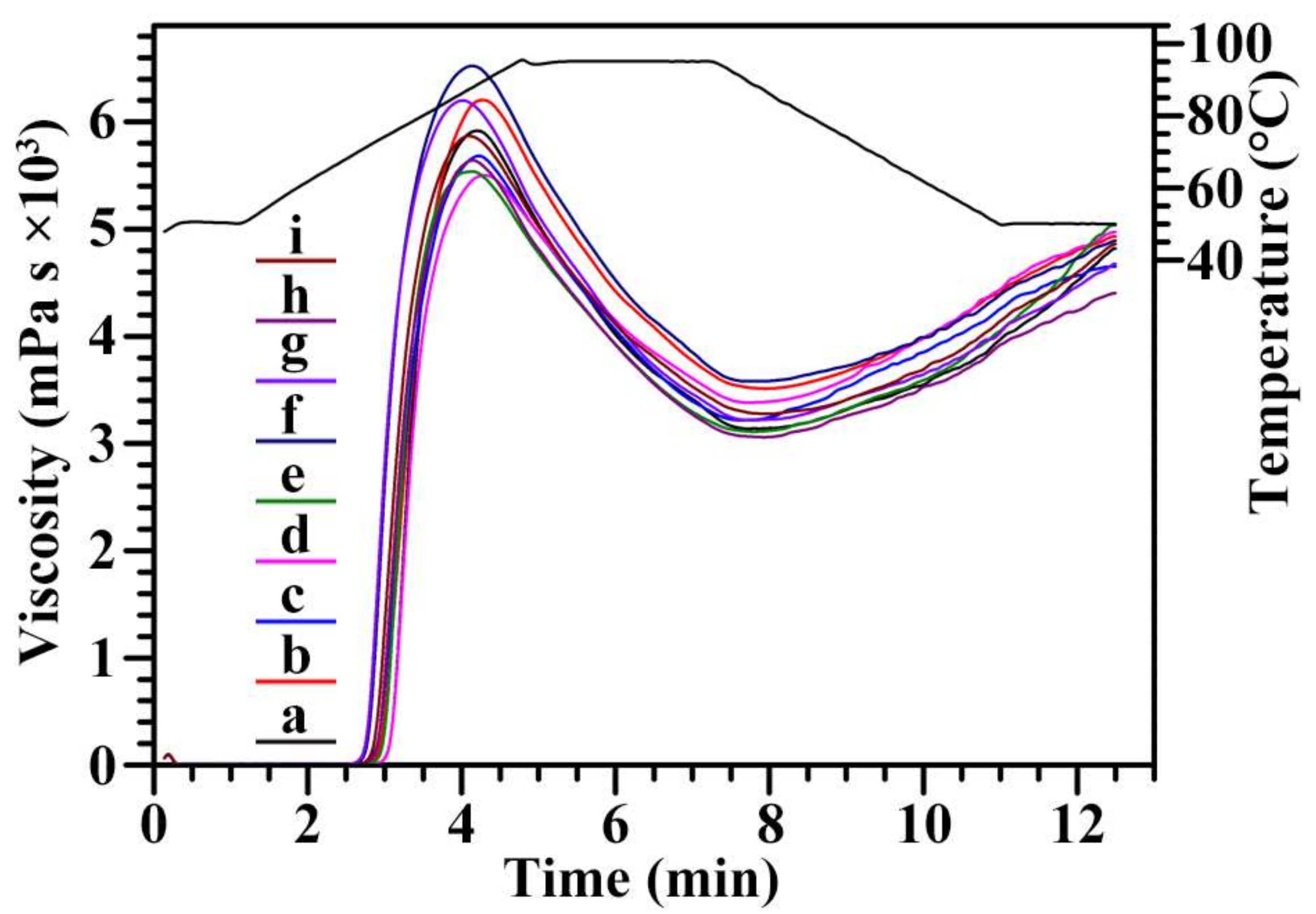
2.8. Pasting Properties of Starch
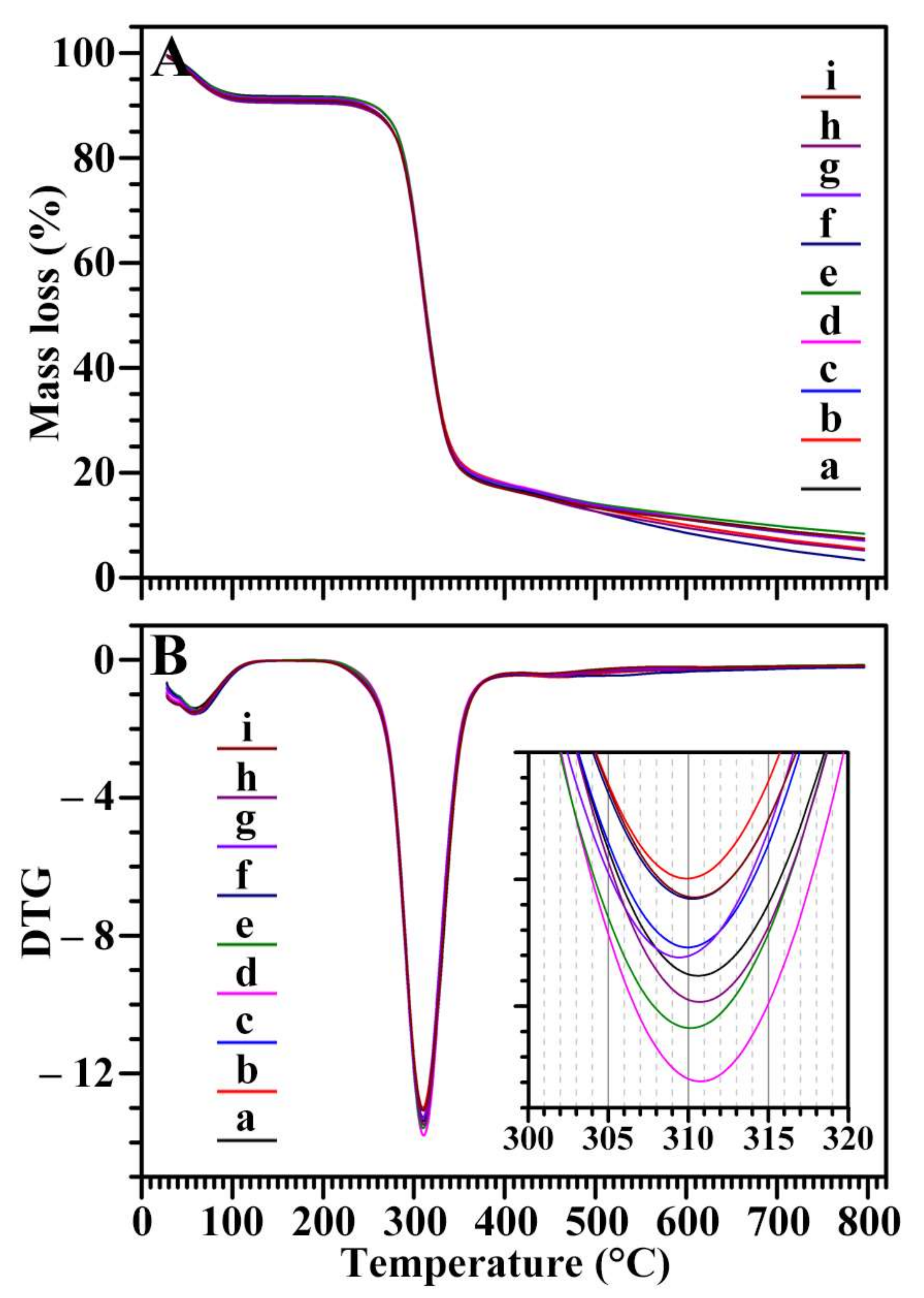
2.9. Thermal Stability of Starch
2.10. Digestion Properties of Starch
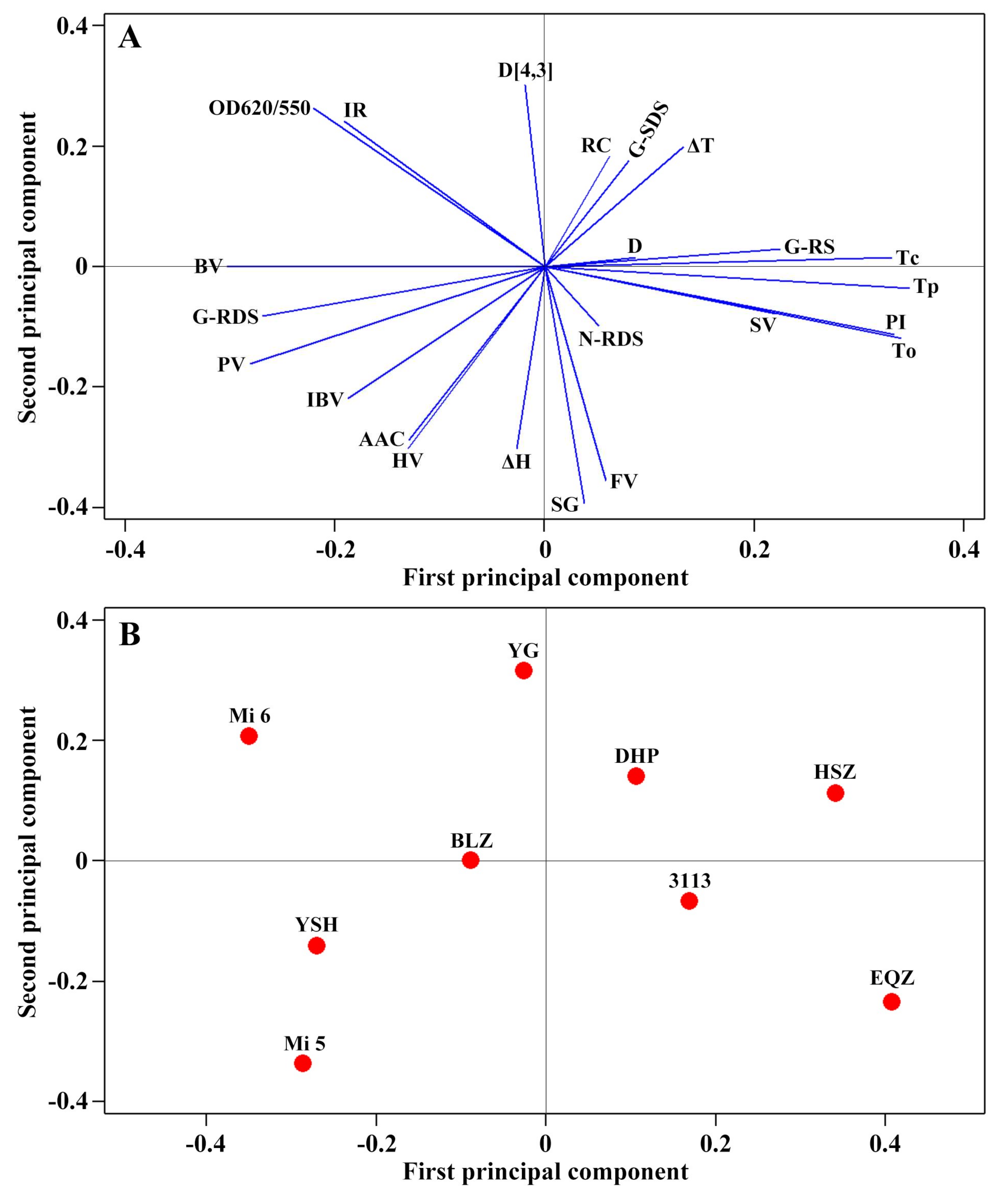
2.11. Principal Component Analysis
3. Materials and Methods
3.1. Plant Materials
3.2. Morphology, Weight, and Starch Content of Nut and Kernel
3.3. Starch Isolation
3.4. Morphology Observation and Size Distribution Analysis of Starch
3.5. Analysis of Iodine Absorption Spectrum and Determination of Apparent Amylose Content
3.6. XRD Analysis
3.7. ATR-FTIR Analysis
3.8. SAXS Analysis
3.9. DSC Analysis
3.10. RVA Analysis
3.11. TGA Analysis
3.12. Determination of Digestion Properties
3.13. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Emmambux, M.N.; Taylor, J.R.N. Morphology, physical, chemical, and functional properties of starches from cereals, legumes, and tubers cultivated in Afria: A review. Starch 2013, 65, 715–729. [Google Scholar] [CrossRef]
- García, N.L.; Famá, L.; Dufresne, A.; Aranguren, M.; Goyanes, S. A comparison between the physic-chemical properties of tuber and cereal starches. Food Res. Int. 2009, 42, 976–982. [Google Scholar] [CrossRef]
- Liu, Q.; Donner, E.; Yin, Y.; Huang, R.L.; Fan, M.Z. The physicochemical properties and in vitro digestibility of selected cereals, tubers and legumes grown in China. Food Chem. 2006, 99, 470–477. [Google Scholar] [CrossRef]
- Hoover, R. Composition, molecular structure, and physicochemical properties of tuber and root starches: A review. Carbohydr. Polym. 2001, 45, 253–267. [Google Scholar] [CrossRef]
- Wani, I.A.; Sogi, D.S.; Hamdani, A.M.; Gani, A.; Bhat, N.A.; Shah, A. Isolation, composition, and physicochemical properties of starch from legumes: A review. Starch 2016, 68, 1–12. [Google Scholar] [CrossRef]
- Guo, K.; Lin, L.; Fan, X.; Zhang, L.; Wei, C. Comparison of structural and functional properties of starches from five fruit kernels. Food Chem. 2018, 257, 75–82. [Google Scholar] [CrossRef]
- Fan, X.; Zhang, S.; Lin, L.; Zhao, L.; Liu, A.; Wei, C. Properties of new starches from tubers of Arisaema elephas, yunnanense and erubescens. Food Hydrocolloid. 2016, 61, 183–190. [Google Scholar] [CrossRef]
- Huang, J.; Zhao, L.; Man, J.; Wang, J.; Zhou, W.; Huai, H.; Wei, C. Comparison of physicochemical properties of B-type nontraditional starches from different sources. Int. J. Biol. Macromol. 2015, 78, 165–172. [Google Scholar] [CrossRef]
- Gao, H.; Cai, J.; Han, W.; Huai, H.; Chen, Y.; Wei, C. Comparison of starches isolated from three different Trapa species. Food Hydrocolloid. 2014, 37, 174–181. [Google Scholar] [CrossRef]
- Huang, J.; Zhao, L.; Huai, H.; Li, E.; Zhang, F.; Wei, C. Structural and functional properties of starches from wild Trapa quadrispinosa, japonica, mammillifera and incise. Food Hydrocolloid. 2015, 48, 117–126. [Google Scholar] [CrossRef]
- Zhu, F. Properties and food uses of chestnut flour and starch. Food Bioprocess. Technol. 2017, 10, 1173–1191. [Google Scholar] [CrossRef]
- FAOSTAT. Available online: http://www.fao.org/faostat/en/#data/QC/visualize (accessed on 12 October 2018).
- De Vasconcelos, M.C.B.M.; Bennett, R.N.; Rosa, E.A.S.; Ferreira-Cardoso, J.V. Composition of European chestnut (Castanea sativa Mill.) and association with health effects: Fresh and processed products. J. Sci. Food Agric. 2010, 90, 1578–1589. [Google Scholar] [CrossRef] [PubMed]
- Frati, A.; Landi, D.; Marinelli, C.; Gianni, G.; Fontana, L.; Migliorini, M.; Pierucci, F.; Garcia-Gil, M.; Meacci, E. Nutraceutical properties of chestnut flours: Beneficial effects on skeletal muscle atrophy. Food Funct. 2014, 5, 2870–2882. [Google Scholar] [CrossRef] [PubMed]
- Demiate, I.M.; Oetterer, M.; Wosiacki, G. Characterization of chestnut (Castanea sativa, Mill) starch for industrial utilization. Braz. Arch. Biol. Techn. 2001, 44, 69–78. [Google Scholar] [CrossRef]
- Liu, C.; Wang, S.; Chang, X.; Wang, S. Structural and functional properties of starches from Chinese chestnuts. Food Hydrocolloid. 2015, 43, 568–576. [Google Scholar] [CrossRef]
- Silva, A.P.; Oliveira, I.; Silva, M.E.; Guedes, C.M.; Borges, O.; Magalhães, B. Starch characterization in seven raw, boiled and roasted chestnuts (Castanea sativa Mill.) cultivars from Portugal. J. Food Sci. Technol. 2016, 53, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.; Li, Q.; Bao, W.; Wu, Y.; Ouyang, J. Relationship between physicochemical characteristics and in vitro digestibility of chestnut (Castanea mollissima) starch. Food Hydrocolloid. 2018, 84, 193–194. [Google Scholar] [CrossRef]
- Correia, P.R.; Nunes, M.C.; Beirão-da-Costa, M.L. The effect of starch isolation method on physical and functional properties of Portuguese nuts starches. I. Chestnuts (Castanea sativa Mill. var. Martainha and Longal) fruits. Food Hydrocolloid. 2012, 27, 256–263. [Google Scholar] [CrossRef]
- Cruz, B.R.; Abraão, A.S.; Lemos, A.M.; Nunes, F.M. Chemical composition and functional properties of native chestnut starch (Castanea sativa Mill). Carbohyd. Polym. 2013, 94, 594–602. [Google Scholar] [CrossRef]
- Lemos, A.M.; Abraão, A.S.; Cruz, B.R.; Morgado, M.L.; Rebelo, M.; Nunes, F.M. Effect of granular characteristics on the viscoelastic and mechanical properties of native chestnut starch (Castanea sativa Mill). Food Hydrocolloid. 2015, 51, 305–317. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, H.; Zhang, Y. Physicochemical, thermal, and pasting properties of Chinese chestnut (Castanea mollissima Bl.) starches as affected by different drying methods. Starch 2011, 63, 260–267. [Google Scholar] [CrossRef]
- Yang, B.; Jiang, G.; Prasad, K.N.; Gu, C.; Jiang, Y. Crystalline, thermal and textural characteristics of starches isolated from chestnut (Castanea mollissima Bl.) seeds at different degrees of hardness. Food Chem. 2010, 119, 995–999. [Google Scholar] [CrossRef]
- Wang, S.; Liu, C.; Wang, S. Drying methods used in starch isolation change properties of C-type chestnut (Castanea mollissima) starches. LWT-Food Sci. Technol. 2016, 73, 663–669. [Google Scholar] [CrossRef]
- Kan, L.; Li, Q.; Xie, S.; Hu, J.; Wu, Y.; Ouyang, J. Effect of thermal processing on the physicochemical properties of chestnut starch and textural profile of chestnut kernel. Carbohyd. Polym. 2016, 151, 614–623. [Google Scholar] [CrossRef]
- Chen, L.; Lu, D.; Wang, T.; Li, Z.; Zhao, Y.; Jiang, Y.; Zhang, Q.; Cao, Q.; Fang, K.; Xing, Y.; et al. Identification and expression analysis of starch branching enzymes involved in starch synthesis during the development of chestnut (Castanea mollissima Blume) cotyledons. PLoS ONE 2017, 12, e0177792. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, Y.; Wu, Y.; Liu, L.; Ouyang, J. Physicochemical properties and in vitro digestibility of starch from naturally air-dried chestnut. Int. J. Biol. Macromol. 2018, 117, 1074–1080. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Lin, Q.; Feng, Y.; Fan, X.; Zou, F.; Yuan, D.Y.; Zeng, X.; Cao, H. Transcriptomic identification and expression of starch and sucrose metabolism genes in the seeds of Chinese chestnut (Castanea mollissima). J. Agric. Food Chem. 2015, 63, 929–942. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.H.; Lee, C.S.; Kim, B.S.; Shin, M. The properties and molecular structures of gusiljatbam starch compared to those of acorn and chestnut starches. Starch 2012, 64, 339–347. [Google Scholar] [CrossRef]
- Torres, M.D.; Moreira, R.; Chenlo, F.; Morel, M.H.; Barron, C. Physicochemical and structural properties of starch isolated from fresh and dried chestnuts and chestnut flour. Food Technol. Biotechnol. 2014, 52, 135–139. [Google Scholar]
- Lindeboom, N.; Chang, P.R.; Tyler, R.T. Analytical, biochemical and physicochemical aspects of starch granule size, with emphasis on small granule starches: A review. Starch 2004, 56, 89–99. [Google Scholar] [CrossRef]
- Wei, C.; Zhang, J.; Chen, Y.; Zhou, W.; Xu, B.; Wang, Y.; Chen, J. Physicochemical properties and development of wheat large and small starch granules during endosperm development. Acta Physiol. Plant. 2010, 32, 905–916. [Google Scholar] [CrossRef]
- Naguleswaran, S.; Li, J.; Vasanthan, T.; Bressler, D.; Hoover, R. Amylolysis of large and small granules of native triticale, wheat and corn starches using a mixture of α-amylase and glucoamylase. Carbohydr. Polym. 2012, 88, 864–874. [Google Scholar] [CrossRef]
- He, W.; Wei, C. Progress in C-type starches from different plant sources. Food Hydrocolloid. 2017, 73, 162–175. [Google Scholar] [CrossRef]
- Atichokudomchai, N.; Varavinit, S.; Chinachoti, P. A study of ordered structure in acid-modified tapioca starch by 13C CP/MAS solid-state NMR. Carbohydr. Polym. 2004, 58, 383–389. [Google Scholar] [CrossRef]
- Sevenou, O.; Hill, S.E.; Farhat, I.A.; Mitchell, J.R. Organisation of the external region of the starch granule as determined by infrared spectroscopy. Int. J. Biol. Macromol. 2002, 31, 79–85. [Google Scholar] [CrossRef]
- Blazek, J.; Gilbert, E.P. Application of small-angle X-ray and neutron scattering techniques to the characterisation of starch structure: A review. Carbohydr. Polym. 2011, 85, 281–293. [Google Scholar] [CrossRef]
- Yuryev, V.P.; Krivandin, A.V.; Kiseleva, V.I.; Wasserman, L.A.; Genkina, N.K.; Fornal, J.; Blaszczak, W.; Schiraldi, A. Structural parameters of amylopectin clusters and semi-crystalline growth rings in wheat starches with different amylose content. Carbohydr. Res. 2004, 339, 2683–2691. [Google Scholar] [CrossRef]
- Lee, J.W.; Choi, H.W.; Seo, D.H.; Park, J.D.; Kum, J.S.; Kim, B.Y.; Baik, M.Y. Isolation and characterization of starches from chestnuts cultivated in three regions in Korea. Starch 2015, 67, 585–594. [Google Scholar] [CrossRef]
- Wang, S.; Copeland, L. Phase transitions of pea starch over a wide range of water content. J. Agric. Food Chem. 2012, 60, 6439–6446. [Google Scholar] [CrossRef] [PubMed]
- Rincón-Londoño, N.; Vega-Rojas, L.J.; Contreras-Padilla, M.; Acosta-Osorio, A.A.; Rodríguez-García, M.E. Analysis of the pasting profile in corn starch: Structural, morphological, and thermal transformations, Part I. Int. J. Biol. Macromol. 2016, 91, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Rincón-Londoño, N.; Millan-Malo, B.; Rodríguez-García, M.E. Analysis of thermal pasting profile in corn starch rich in amylose and amylopectin: Physicochemical transformations, Part II. Int. J. Biol. Macromol. 2016, 89, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Abegunde, O.K.; Mu, T.H.; Chen, J.W.; Deng, F.M. Physicochemical characterization of sweet potato starches popularly used in Chinese starch industry. Food Hydrocolloid. 2013, 33, 169–177. [Google Scholar] [CrossRef]
- Singh, N.; Kaur, L.; Ezekiel, R.; Guraya, H.S. Microstructural, cooking and textural characteristics of potato (Solanum tuberosum L.) tubers in relation to physicochemical and functional properties of their flours. J. Sci. Food Agric. 2005, 85, 1275–1284. [Google Scholar] [CrossRef]
- Wang, L.; Xie, B.; Xiong, G.; Du, X.; Qiao, Y.; Liao, L. Study on the granular characteristics of starches separated from Chinese rice cultivars. Carbohydr. Polym. 2012, 87, 1038–1044. [Google Scholar] [CrossRef]
- Englyst, H.N.; Kingman, S.M.; Cummings, J.H. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 45, S33–S50. [Google Scholar]
- Lin, L.; Cai, C.; Gilbert, R.G.; Li, E.; Wang, J.; Wei, C. Relationships between amylopectin molecular structures and functional properties of different-sized fractions of normal and high-amylose maize starches. Food Hydrocolloid. 2016, 52, 359–368. [Google Scholar] [CrossRef]
- Lin, L.; Guo, D.; Huang, J.; Zhang, X.; Zhang, L.; Wei, C. Molecular structure and enzymatic hydrolysis properties of starches from high-amylose maize inbred lines and their hybrids. Food Hydrocolloid. 2016, 58, 246–254. [Google Scholar] [CrossRef]
- Wang, S.; Copeland, L. Molecular disassembly of starch granules during gelatinization and its effect on starch digestibility: A review. Food Funct. 2013, 4, 1564–1580. [Google Scholar] [CrossRef]
- Cai, C.; Lin, L.; Man, J.; Zhao, L.; Wang, Z.; Wei, C. Different structural properties of high-amylose maize starch fractions varying in granule size. J. Agric. Food Chem. 2014, 62, 11711–11721. [Google Scholar] [CrossRef]
- Man, J.; Lin, L.; Wang, Z.; Wang, Y.; Liu, Q.; Wei, C. Different structure of heterogeneous starch granules from high-amylose rice. J. Agric. Food Chem. 2014, 62, 11254–11263. [Google Scholar] [CrossRef]
- Zhang, B.; Guo, K.; Lin, L.; Wei, C. Comparison of structural and functional properties of starches from the rhizome and bulbil of Chinese yam (Dioscorea opposite Thunb.). Molecules 2018, 23, 427. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Zhang, Q.; Zhang, L.; Wei, C. Evaluation of the molecular structural parameters of normal rice starch and their relationships with its thermal and digestion properties. Molecules 2017, 22, 1526. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the starches are available from the authors. |




| Varieties | Fresh Nut Weight (g/nut) | Fresh Kernel Weight (g/kernel) | Dry Kernel | |
|---|---|---|---|---|
| Kernel Weight (g/kernel) | Starch Content (%) | |||
| 3113 | 6.4 ± 0.1 a | 5.5 ± 0.1a | 2.51 ± 0.03 a | 43.3 ± 0.2 a,b |
| Benlizi | 11.9 ± 0.5 e | 9.9 ± 0.4 d,e | 4.95 ± 0.20 d | 46.7 ± 1.0 b,c |
| Dahongpao | 12.2 ± 0.7 e | 10.5 ± 0.5 e | 5.61 ± 0.29 e | 49.2 ± 0.9 c |
| Erqingzao | 6.7 ± 0.5 a,b | 5.5 ± 0.4 a | 3.29 ± 0.22 b | 42.7 ± 1.0 a |
| Heishanzhai 7 | 7.0 ± 0.2 a,b,c | 6.1 ± 0.2 a,b | 2.98 ± 0.11 b | 47.6 ± 1.5 c |
| Mi 5 | 7.9 ± 0.1 c | 6.8 ± 0.1 b | 3.90 ± 0.04 c | 43.6 ± 1.2 a,b |
| Mi 6 | 7.7 ± 0.6 b,c | 6.6 ± 0.5 b | 3.16 ± 0.22 b | 46.4 ± 1.0 a,b,c |
| Yangguang | 10.6 ± 0.1 d | 9.2 ± 0.2 c,d | 4.29 ± 0.08 c | 49.3 ± 1.2 c |
| Yanshanhong | 10.2 ± 0.3 d | 8.7 ± 0.3 c | 4.78 ± 0.17 d | 43.1 ± 0.6 a,b |
| Mean ± SD | 9.0 ± 2.3 | 7.6 ± 1.9 | 3.94 ± 1.04 | 45.8 ± 2.7 |
| Sig. | 0.160 | 0.205 | 0.802 | 0.137 |
| Varieties | Small Granule (%) | d(0.1) (μm) | d(0.5) (μm) | d(0.9) (μm) | D[4,3] (μm) |
|---|---|---|---|---|---|
| 3113 | 11.04 ± 0.05 f | 1.934 ± 0.012 c | 7.431 ± 0.004 e | 13.620 ± 0.008 f | 7.819 ± 0.003 f |
| Benlizi | 10.24 ± 0.04 b | 2.156 ± 0.015 f | 7.706 ± 0.001 g | 13.855 ± 0.004 h | 8.048 ± 0.001 g |
| Dahongpao | 10.70 ± 0.04 e | 1.988 ± 0.011 d | 7.291 ± 0.001 d | 13.073 ± 0.004 c | 7.607 ± 0.001 d |
| Erqingzao | 11.27 ± 0.06 g | 1.776 ± 0.016 b | 6.901 ± 0.002 a | 12.241 ± 0.001 a | 7.170 ± 0.003 a |
| Heishanzhai 7 | 10.60 ± 0.03 d | 2.264 ± 0.013 g | 7.911 ± 0.001 i | 14.047 ± 0.003 i | 8.226 ± 0.001 i |
| Mi 5 | 11.84 ± 0.05 h | 1.778 ± 0.009 b | 7.148 ± 0.003 c | 13.127 ± 0.008 d | 7.496 ± 0.002 c |
| Mi 6 | 9.76 ± 0.03 a | 2.337 ± 0.014 h | 7.764 ± 0.001 h | 13.835 ± 0.002 g | 8.099 ± 0.001 h |
| Yangguang | 10.43 ± 0.04 c | 2.091 ± 0.013 e | 7.506 ± 0.002 f | 13.350 ± 0.005 e | 7.807 ± 0.001 e |
| Yanshanhong | 11.29 ± 0.02 g | 1.737 ± 0.001 a | 6.954 ± 0.017 b | 12.983 ± 0.017 b | 7.329 ± 0.015 b |
| Mean ± SD | 10.80 ± 0.63 | 2.007 ± 0.220 | 7.401 ± 0.358 | 13.348 ± 0.563 | 7.733 ± 0.361 |
| Sig. | 0.993 | 0.485 | 0.744 | 0.564 | 0.819 |
| Varieties | AAC (%) | RC (%) | OD | Lamellar Structure Parameters | |
|---|---|---|---|---|---|
| PI (counts) | D (nm) | ||||
| 3113 | 26.1 ± 0.5 b,c | 20.0 ± 0.8 a,b | 0.61 ± 0.02 a | 143 ± 8 a,b | 9.46 ± 0.09 a |
| Benlizi | 24.2 ± 0.7 a,b | 18.5 ± 0.5 a | 0.63 ± 0.02 a | 132 ± 6 a,b | 9.49 ± 0.00 a |
| Dahongpao | 23.8 ± 1.1 a | 20.7 ± 0.8 a,b | 0.63 ± 0.02 a | 136 ± 16 a,b | 9.38 ± 0.01 a |
| Erqingzao | 24.5 ± 0.9 a,b | 20.4 ± 0.5 a,b | 0.62 ± 0.02 a | 150 ± 3 b | 9.44 ± 0.04 a |
| Heishanzhai 7 | 23.8 ± 0.9 a | 21.2 ± 0.9 b | 0.64 ± 0.02 a | 146 ± 7 a,b | 9.30 ± 0.01 a |
| Mi 5 | 26.2 ± 1.1 b,c | 20.0 ± 1.4 a,b | 0.63 ± 0.01 a | 133 ± 8 a,b | 9.27 ± 0.03 a |
| Mi 6 | 23.8 ± 0.9 a | 20.7 ± 0.4 a,b | 0.65 ± 0.02 a | 120 ± 4 a | 9.40 ± 0.01 a |
| Yangguang | 24.6 ± 0.6 a,b | 21.2 ± 0.9 b | 0.64 ± 0.02 a | 130 ± 1 a,b | 9.33 ± 0.14 a |
| Yanshanhong | 27.3 ± 0.5 c | 20.7 ± 0.6 a,b | 0.64 ± 0.02 a | 119 ± 3 a | 9.33 ± 0.01 a |
| Mean ± SD | 24.9 ± 1.3 | 20.4 ± 0.8 | 0.63 ± 0.01 | 134 ± 11 | 9.38 ± 0.08 |
| Sig. | 0.055 | 0.059 | 0.586 | 0.674 | 0.780 |
| Varieties | To (°C) | Tp (°C) | Tc (°C) | ΔT (°C) | ΔH (J/g) |
|---|---|---|---|---|---|
| 3113 | 62.7 ± 0.3 c | 68.1 ± 0.1 f | 74.5 ± 0.3 e | 11.8 ± 0.0 d,e | 13.1 ± 0.1 a |
| Benlizi | 61.4 ± 0.1 b | 66.1 ± 0.1 c | 72.0 ± 0.1 c | 10.6 ± 0.0 c | 12.8 ± 0.2 a |
| Dahongpao | 61.4 ± 0.2 b | 66.7 ± 0.2 d | 73.3 ± 0.0 d | 12.0 ± 0.2 e | 13.1 ± 0.1 a |
| Erqingzao | 63.9 ± 0.1 d | 68.3 ± 0.1 f | 74.3 ± 0.0 e | 10.5 ± 0.1 b,c | 13.6 ± 0.1 a |
| Heishanzhai 7 | 62.9 ± 0.1 c | 67.6 ± 0.1 e | 73.2 ± 0.2 d | 10.3 ± 0.1 a,b | 12.8 ± 0.2 a |
| Mi 5 | 60.9 ± 0.0 b | 65.2 ± 0.0 b | 70.9 ± 0.0 a,b | 10.0 ± 0.0 a | 13.4 ± 0.2 a |
| Mi 6 | 60.4 ± 0.0 a | 64.8 ± 0.1 a | 70.5 ± 0.1 a | 10.1 ± 0.1 a | 13.3 ± 0.4 a |
| Yangguang | 61.3 ± 0.1 b | 66.4 ± 0.1 d | 72.9 ± 0.2 d | 11.6 ± 0.1 d | 12.8 ± 0.4 a |
| Yanshanhong | 60.9 ± 0.1 b | 65.4 ± 0.1 b | 71.1 ± 0.1 b | 10.2 ± 0.0 a,b | 13.2 ± 0.1 a |
| Mean ± SD | 61.8 ± 1.1 | 66.5 ± 1.3 | 72.5 ± 1.5 | 10.8 ± 0.8 | 13.1 ± 0.3 |
| Sig. | 0.226 | 0.564 | 0.467 | 0.053 | 0.345 |
| Varieties | PV (mPa s) | HV (mPa s) | BV (mPa s) | FV (mPa s) | SV (mPa s) |
|---|---|---|---|---|---|
| 3113 | 5866 ± 49 c | 3104 ± 31 a,b | 2762 ± 18 f | 4745 ± 65 b,c | 1641 ± 37 c |
| Benlizi | 6219 ± 38 d | 3487 ± 74 d | 2732 ± 48 e,f | 4954 ± 62 d | 1467 ± 41 d |
| Dahongpao | 5722 ± 51 b | 3260 ± 40 b,c | 2462 ± 56 b,c | 4650 ± 35 b | 1390 ± 42 a,b |
| Erqingzao | 5524 ± 21 a | 3319 ± 64 c | 2205 ± 84 a | 4942 ± 27 d | 1623 ± 50 c |
| Heishanzhai 7 | 5527 ± 23 a | 3125 ± 59 a,b | 2402 ± 46 b | 4913 ± 35 d | 1788 ± 79 d |
| Mi 5 | 6505 ± 41 e | 3616 ± 58 d | 2889 ± 51 g | 4942 ± 50 d | 1326 ± 27 a |
| Mi 6 | 6192 ± 23 d | 3238 ± 23 b,c | 2954 ± 29 g | 4663 ± 32 b | 1425 ± 53 a,b |
| Yangguang | 5609 ± 91 a | 3042 ± 98 a | 2567 ± 22 c,d | 4378 ± 68 a | 1336 ± 33 a |
| Yanshanhong | 5860 ± 27 c | 3220 ± 67 b,c | 2640 ± 45 d,e | 4829 ± 34 c,d | 1609 ± 33 c |
| Mean ± SD | 5892 ± 345 | 3268 ± 185 | 2624 ± 241 | 4780 ± 192 | 1512 ± 160 |
| Sig. | 0.320 | 0.486 | 0.954 | 0.098 | 0.417 |
| Varieties | Native Starch | Gelatinized Starch | ||||
|---|---|---|---|---|---|---|
| RDS (%) | SDS (%) | RS (%) | RDS (%) | SDS (%) | RS (%) | |
| 3113 | 2.6 ± 0.3 a | 12.6 ± 0.0 c | 84.8 ± 0.3 a | 86.5 ± 1.3 b | 3.5 ± 0.8 c,d | 10.0 ± 0.5 b |
| Benlizi | 3.7 ± 1.1 a | 11.3 ± 1.0 b,c | 85.0 ± 0.1 a | 84.8 ± 0.6 b | 3.1 ± 0.2 b,c,d | 12.1 ± 0.8 b,c |
| Dahongpao | 3.0 ± 0.0 a | 12.5 ± 0.3 c | 84.4 ± 0.3 a | 80.4 ± 0.2 a | 2.4 ± 1.1 a,b,c,d | 17.3 ± 1.3 d |
| Erqingzao | 3.3 ± 1.0 a | 10.0 ± 0.2 b | 86.7 ± 0.7 a | 80.7 ± 1.1 a | 1.9 ± 0.6 a,b,c | 17.4 ± 0.9 d |
| Heishanzhai 7 | 3.5 ± 1.3 a | 5.7 ± 0.9 a | 90.7 ± 0.4 b | 79.6 ± 1.4 a | 3.0 ± 0.6 b,c,d | 17.3 ± 1.3 d |
| Mi 5 | 3.4 ± 0.8 a | 11.7 ± 0.3 b,c | 84.9 ± 0.5 a | 84.8 ± 1.3 b | 1.3 ± 0.5 a | 13.9 ± 0.9 c |
| Mi 6 | 2.9 ± 0.1 a | 11.9 ± 0.8 b,c | 85.2 ± 0.9 a | 86.2 ± 1.2 b | 1.5 ± 0.5 a,b | 12.3 ± 1.6 b,c |
| Yangguang | 3.0 ± 0.0 a | 10.7 ± 0.6 b,c | 86.3 ± 0.6 a | 84.0 ± 1.4 b | 3.8 ± 0.5 d | 12.2 ± 1.5 b,c |
| Yanshanhong | 2.9 ± 0.1 a | 12.7 ± 0.9 c | 84.4 ± 1.0 a | 89.5 ± 1.3 c | 3.4 ± 0.6 c,d | 7.1 ± 0.8 a |
| Mean ± SD | 3.1 ± 0.4 | 11.0 ± 2.2 | 85.8 ± 2.0 | 84.1 ± 3.3 | 2.7 ± 0.9 | 13.3 ± 3.6 |
| Sig. | 0.770 | 0.006 | 0.002 | 0.562 | 0.398 | 0.292 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, L.; Liu, T.; Hu, G.; Guo, K.; Wei, C. Comparison of Physicochemical Properties of Starches from Nine Chinese Chestnut Varieties. Molecules 2018, 23, 3248. https://doi.org/10.3390/molecules23123248
Zhang L, Liu T, Hu G, Guo K, Wei C. Comparison of Physicochemical Properties of Starches from Nine Chinese Chestnut Varieties. Molecules. 2018; 23(12):3248. https://doi.org/10.3390/molecules23123248
Chicago/Turabian StyleZhang, Long, Tianxiang Liu, Guanglong Hu, Ke Guo, and Cunxu Wei. 2018. "Comparison of Physicochemical Properties of Starches from Nine Chinese Chestnut Varieties" Molecules 23, no. 12: 3248. https://doi.org/10.3390/molecules23123248
APA StyleZhang, L., Liu, T., Hu, G., Guo, K., & Wei, C. (2018). Comparison of Physicochemical Properties of Starches from Nine Chinese Chestnut Varieties. Molecules, 23(12), 3248. https://doi.org/10.3390/molecules23123248








