10-HDA, A Major Fatty Acid of Royal Jelly, Exhibits pH Dependent Growth-Inhibitory Activity Against Different Strains of Paenibacillus larvae
Abstract
1. Introduction
2. Results
2.1. ERIC Genotypes of Bacterial Strains
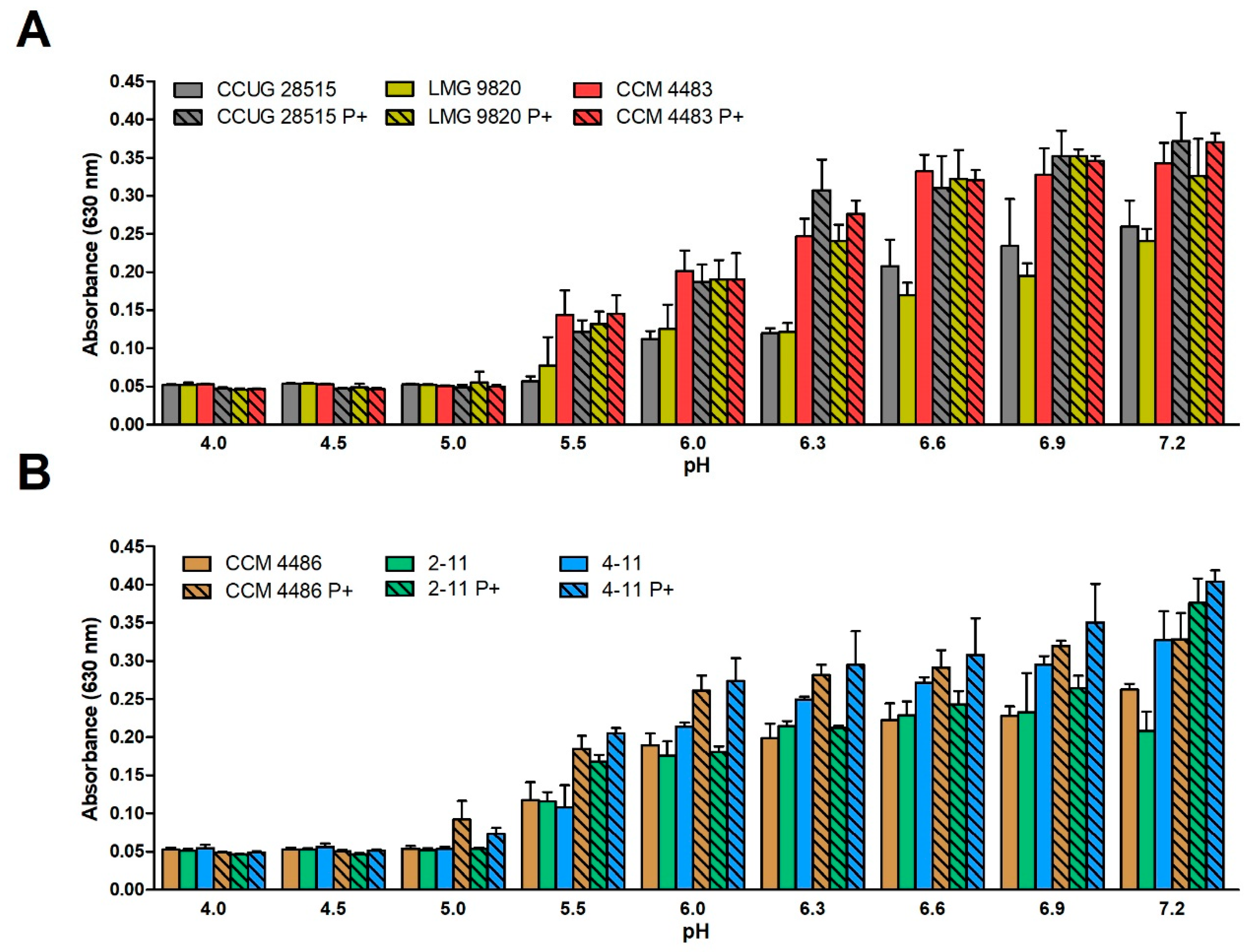
2.2. Effect of pH and Substance PIPES on Growth of P. larvae
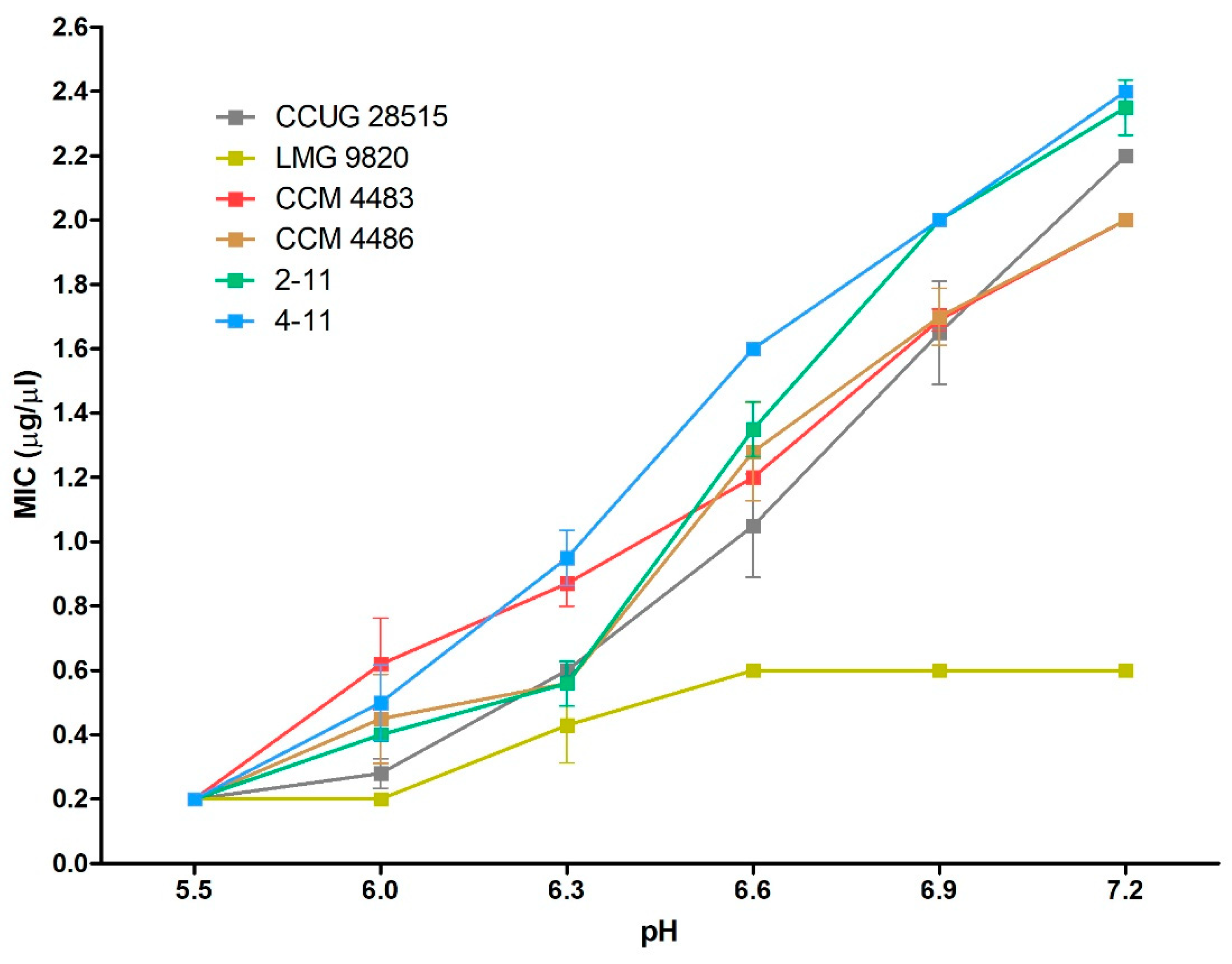
2.3. Efficacy of 10-HDA Against P. larvae at Various pH Levels
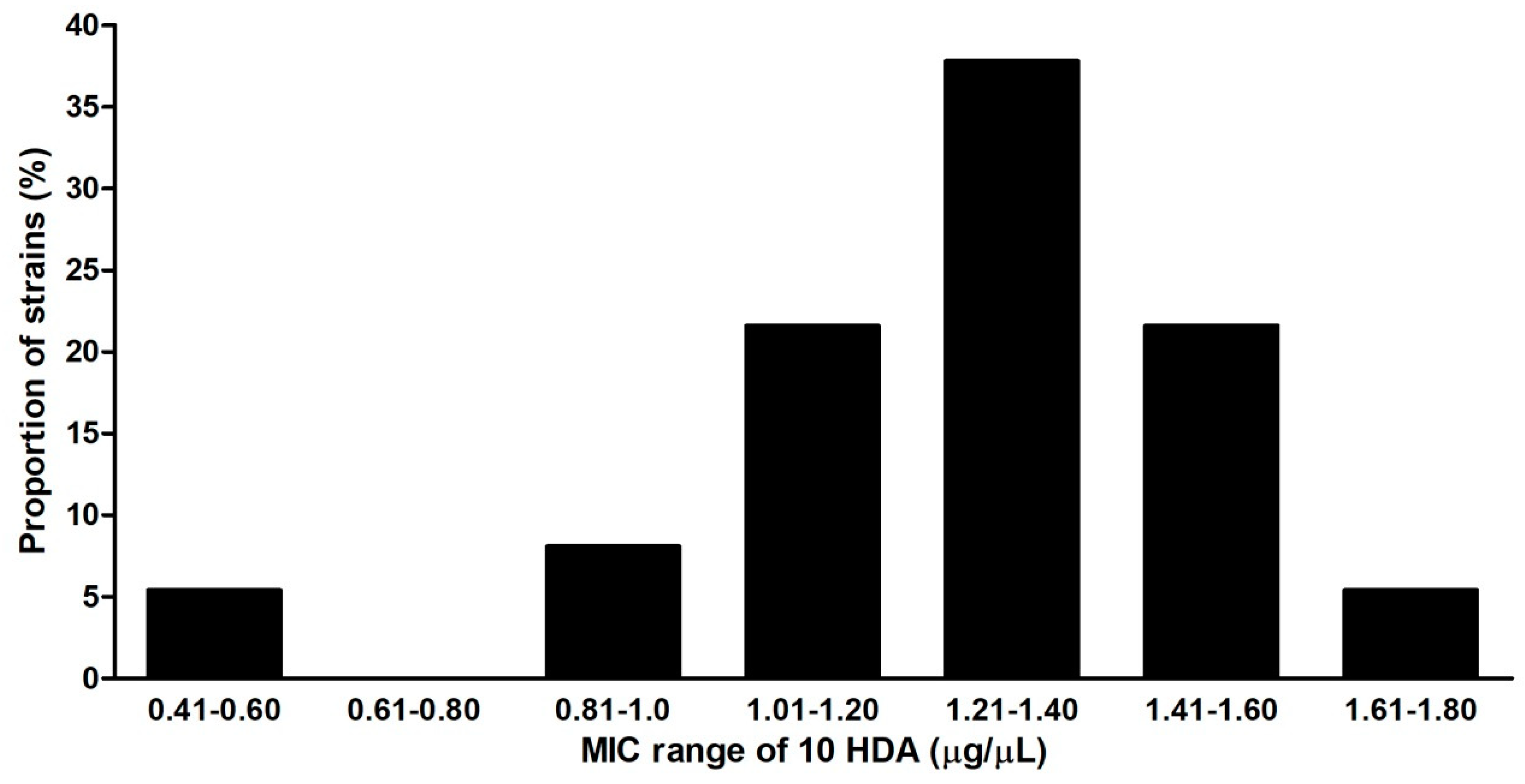
2.4. Susceptibility of Culture Strains, Field Isolates, Vegetative Cells and Spores of P. larvae to 10-HDA
3. Discussion
4. Materials and Methods
4.1. Fatty Acid
4.2. P. larvae Strains
4.3. ERIC-PCR Genotyping
4.4. Cultivation of P. larvae
4.5. Preparation of Spores
4.6. Minimum Inhibitory Concentration Assay
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bailey, L.; Ball, B.V. Honey Bee Pathology; Academic Press: London, UK, 1991; pp. 53–62. [Google Scholar]
- Hansen, H.; Brødsgaard, C.J. American foulbrood: A review of its biology, diagnosis and control. Bee World 1999, 80, 5–23. [Google Scholar] [CrossRef]
- Yue, D.; Nordhoff, M.; Wieler, L.H.; Genersch, E. Fluorescence in situ hybridization (FISH) analysis of the interactions between honeybee larvae and Paenibacillus larvae, the causative agent of American foulbrood of honeybees (Apis mellifera). Environ. Microbiol. 2008, 10, 1612–1620. [Google Scholar] [CrossRef] [PubMed]
- Antúnez, K.; Anido, M.; Schlapp, G.; Evans, J.D.; Zunino, P. Characterization of secreted proteases of Paenibacillus larvae, potential virulence factors involved in honeybee larval infection. J. Invertebr. Pathol. 2009, 102, 129–132. [Google Scholar] [CrossRef]
- Garcia-Gonzalez, E.; Poppinga, L.; Fünfhaus, A.; Hertlein, G.; Hedtke, K.; Jakubowska, A.; Genersch, E. Paenibacillus larvae chitin-degrading protein PlCBP49 is a key virulence factor in American foulbrood of honey bees. PLoS Path 2014, 10, e1004284. [Google Scholar] [CrossRef]
- Ebeling, J.; Knispel, H.; Hertlein, G.; Fünfhaus, A.; Genersch, E. Biology of Paenibacillus larvae, a deadly pathogen of honey bee larvae. Appl. Microbiol. Biotechnol. 2016, 100, 7387–7395. [Google Scholar] [CrossRef] [PubMed]
- Lindström, A.; Korpela, S.; Fries, I. The distribution of Paenibacillus larvae spores in adult bees and honey and larval mortality, following the addition of American foulbrood disease brood or spore-contaminated honey in honey bee (Apis mellifera) colonies. J. Invertebr. Pathol. 2008, 99, 82–86. [Google Scholar] [CrossRef]
- Genersch, E. American Foulbrood in honeybees and its causative agent, Paenibacillus larvae. J. Invert. Pathol. 2010, 87, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Genersch, E.; Ashiralieva, A.; Fries, I. Strain- and genotype-specific differences in virulence of Paenibacillus larvae subsp. larvae, the causative agent of American foulbrood disease in honey bees. Appl. Environ. Microbiol. 2005, 71, 7551–7555. [Google Scholar] [CrossRef] [PubMed]
- Genersch, E.; Forsgren, E.; Pentikäinen, J.; Ashiralieva, A.; Rauch, S.; Kilwinski, J.; Fries, I. Reclassification of Paenibacillus larvae subsp. pulvifaciens and Paenibacillus larvae subsp. larvae as Paenibacillus larvae without subspecies differentiation. Int. J. Syst. Evol. Microbiol. 2006, 56, 501–511. [Google Scholar] [CrossRef]
- Spivak, M.; Gilliam, M. Hygienic behaviour of honey bees and its application for control of brood diseases and varroa. Part I. Hygienic behaviour and resistance to American foulbrood. Bee World 1998, 79, 124–134. [Google Scholar] [CrossRef]
- Rauch, S.; Ashiralieva, A.; Hedtke, K.; Genersch, E. Negative correlation between individual-insect-level virulence and colony-level virulence of Paenibacillus larvae, the ethiological agent of American foulbrood of honeybees. Appl. Environ. Microbiol. 2009, 75, 3344–3347. [Google Scholar] [CrossRef] [PubMed]
- Alippi, A.M.; Albo, G.N.; Leniz, D.; Rivera, I.; Zanelli, M.; Roca, A.E. Comparative study of tylosin, erythromycin and oxytetracycline to control of American foulbrood in honey bees. J. Apic. Res. 1999, 38, 149–158. [Google Scholar] [CrossRef]
- Dobbelaere, W.; de Graaf, D.C.; Peeters, J.E.; Jacobs, F.J. Development of a fast and reliable diagnostic method for American foulbrood disease (Paenibacillus larvae subsp. larvae) using a 16S rDNA gene based PCR. Apidologie 2001, 32, 363–370. [Google Scholar] [CrossRef]
- Wilson-Rich, N.; Spivak, M.; Fefferman, N.H.; Starks, P.T. Genetic, individual, and group facilitation of disease resistance in insect societies. Annu. Rev. Entomol. 2009, 54, 405–423. [Google Scholar] [CrossRef]
- Buczek, K. Range of susceptibility of Paenibacillus larvae to antibacterial compounds. Med. Weter. 2011, 67, 87–90. [Google Scholar]
- Evans, J.D.; Pettis, J.S. Colony-level impacts of immune responsiveness in honey bees, Apis mellifera. Evolution 2005, 59, 2270–2274. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.D.; Spivak, M. Socialized Medicine: Individual and communal disease barriers in honey bees. J. Invertebr. Pathol. 2010, 103, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Spivak, M.; Reuter, G.D. Resistance to American foulbrood disease by honey bee colonies Apis mellifera bred for hygienic behavior. Apidologie 2001, 32, 555–565. [Google Scholar] [CrossRef]
- Pérez-Sato, J.A.; Châlin, N.; Martin, S.J.; Hughes, W.O.H.; Ratnieks, F.L.W. Multi-level selection for hygienic behaviour in honeybee. Heredity 2009, 102, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Brouwers, E.V.M.; Ebert, R.; Beetsma, J. Behavioural and physiological aspects of nurse bees in relation to the composition of larval food during caste differentiation in the honeybee. J. Apic. Res. 1987, 26, 11–23. [Google Scholar] [CrossRef]
- Fujiwara, S.; Imai, J.; Fujiwara, M.; Yaeshima, T.; Kawashima, T.; Kobayashi, K. A potent antibacterial protein in royal jelly. J. Biol. Chem. 1990, 265, 11333–11337. [Google Scholar]
- Fontana, R.; Mendes, M.A.; de Souza, B.M.; Konno, K.; César, L.M.M.; Malaspina, O.; Palma, M.S. Jelleines: A family of antibacterial peptides from the royal jelly of honeybees (Apis mellifera). Peptides 2004, 25, 919–928. [Google Scholar] [CrossRef]
- Klaudiny, J.; Albert, Š.; Bachanová, K.; Kopernický, J.; Šimúth, J. Two structurally different defensin genes, one of them encoding a novel defensin isoform, are expressed in honeybee Apis mellifera. Insect Biochem. Mol. Biol. 2005, 35, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Bíliková, K.; Mirgorodskaya, E.; Bukovská, G.; Gobom, J.; Lehrach, H.; Šimúth, J. Towards functional proteomics of minority component of honeybee royal jelly: The effect of post-translational modifications on the antimicrobial activity of apalbumin2. Proteomics 2009, 9, 2131–2138. [Google Scholar] [CrossRef] [PubMed]
- Blum, M.S.; Novak, A.F.; Taber, S. 10-hydroxy-Δ2-decenoic acid, an antibiotic found in royal jelly. Science 1959, 130, 452–453. [Google Scholar] [CrossRef]
- Yatsunami, K.; Echigo, T. Antibacterial action of royal jelly. Bull. Fac. Agr. Tamagawa Univ. 1985, 25, 13–22. [Google Scholar]
- Melliou, E.; Chinou, I. Chemistry and bioactivity of royal jelly from Greece. J. Agric. Food Chem. 2005, 53, 8987–8992. [Google Scholar] [CrossRef]
- Alreshoodi, F.; Sultanbawa, Y. Antimicrobial activity of royal jelly. Anti-Infective Agents 2015, 13, 50–59. [Google Scholar] [CrossRef]
- Rose, R.I.; Briggs, J.D. Resistance to American foulbrood in honey bees. Effects of honey bee larval food on the growth and viability of Bacillus larvae. J. Invertebr. Pathol 1969, 13, 74–80. [Google Scholar] [CrossRef]
- Hornitzky, M.A.Z. The pathogenicity of Paenibacillus larvae subsp. larvae spores and vegetative cells to honey bee (Apis mellifera) colonies and their susceptibility to royal jelly. J. Apic. Res. 1998, 37, 267–271. [Google Scholar] [CrossRef]
- Crailsheim, K.; Riessberger-Gallé, U. Honey bee age-dependent resistance against American foulbrood. Apidologie 2001, 32, 91–103. [Google Scholar] [CrossRef]
- Bíliková, K.; Gusui, W.; Šimúth, J. Isolation of a peptide fraction from honeybee royal jelly as a potential antifoulbrood factor. Apidologie 2001, 32, 275–283. [Google Scholar] [CrossRef]
- Bachanová, K.; Klaudiny, J.; Kopernický, J.; Šimúth, J. Identification of honeybee peptide active against Paenibacillus larvae larvae through bacterial growth-inhibition assay on polyacrylamide gel. Apidologie 2002, 33, 259–269. [Google Scholar] [CrossRef]
- Bíliková, K.; Huang, S.C.; Lin, I.P.; Simuth, J.; Peng, Ch.Ch. Structure and antimicrobial activity relationship of royalisin, an antimicrobial peptide from royal jelly of Apis mellifera. Peptides 2015, 68, 190–196. [Google Scholar] [CrossRef]
- Lercker, G.; Capella, P.; Conte, L.S.; Ruini, F.; Giordani, G. Components of royal jelly: I. Identification of the organic acids. Lipids 1981, 16, 912–919. [Google Scholar] [CrossRef]
- Sugiyama, T.; Takahashi, K.; Mori, H. Royal jelly acid, 10-hydroxy-trans-2-decenoic acid, as a modulator of the innate immune responses. Endocr. Metab. Immune Disord. Drug Targets. 2012, 12, 368–376. [Google Scholar] [CrossRef]
- Li, X.; Huang, C.; Xue, Y. Contribution of lipids in honeybee (Apis mellifera) royal jelly to health. J. Med. Food 2013, 16, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.F.; Wang, K.; Zhang, Y.Z.; Zheng, Y.F.; Hu, F.L. In vitro anti-inflammatory effects of three fatty acids from royal jelly. Mediators Inflamm. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Sabatini, A.G.; Marcazzan, G.L.; Caboni, M.F.; Bogdanov, S.; de Almeida-Muradian, L.B. Quality and standardization of royal jelly. J. ApiProd. Apimed. Sci. 2009, 1, 16–21. [Google Scholar] [CrossRef]
- Garcia, M.C.; Finola, M.S.; Marioli, J.M. Bioassay direct identification of royal jelly’s active compounds against the growth of bacteria capable of infecting cutaneous wounds. Adv. Microbiol. 2013, 3, 138–144. [Google Scholar] [CrossRef]
- Alippi, A.M.; Reynaldi, F.J.; López, A.C.; Aguilar, O.M.; de Giusti, M.R. Molecular epidemiology of Paenibacillus larvae larvae and incidence of American foulbrood in Argentinean honeys from Buenos Aires province. J. Apic. Res. 2004, 43, 135–143. [Google Scholar] [CrossRef]
- Antunez, K.; D’Alessandro, B.; Piccini, C.; Corbella, E.; Zunino, P. Paenibacillus larvae larvae spores in honey samples from Uruguay; a nationwide survey. J. Invertebr. Pathol. 2004, 86, 56–58. [Google Scholar] [CrossRef]
- Sturtevant, A.P.; Revell, I.L. Reduction of Bacillus larvae spores in liquid food of honeybees by action of the honey stopper, and its relation to the development of American foulbrood. J. Econ. Entomol. 1953, 46, 855–860. [Google Scholar] [CrossRef]
- Thompson, V.C.; Rothenbuhler, W.C. Resistance to American foulbrood in honey bees. I. Differential protection of larvae by adults of different genetic lines. J. Econ. Entomol. 1957, 50, 731–737. [Google Scholar] [CrossRef]
- Bamrick, J.F.; Rothenbuhler, W.C. Resistance to American foulbrood in honey bees. IV. The relationship between larval age at inoculation and mortality in a resistant and in susceptible line. J. Insect Pathol. 1961, 3, 381–390. [Google Scholar]
- Davidson, E.W. Ultrastructure of American foulbrood disease pathogenesis in larvae of the worker honey bee, Apis mellifera. J. Invertebr. Pathol. 1973, 21, 53–61. [Google Scholar] [CrossRef]
- Rinderer, T.E.; Rothenbuhler, W.C.; Gochnauer, T.A. The influence of pollen on the susceptibility honey bee larvae to Bacillus larvae. J. Invertebr. Pathol. 1974, 23, 347–350. [Google Scholar] [CrossRef]
- Reiche, R.; Martin, K.; Mollmann, J.; Hentschel, E. Beitrag zur Klärung der Resistenz der Bienenlarven gegen den Erreger der Amerikanischen Faulbrut Paenibacillus (voher Bacillus) larvae. Apidologie 1996, 27, 296–297. [Google Scholar]
- Evans, J.D.; Armstrong, T.N. Inhibition of the American foulbrood bacterium, Paenibacillus larvae, by bacteria isolated form honey bees. J. Apic. Res. 2005, 44, 168–171. [Google Scholar] [CrossRef]
- Evans, J.D. Transcriptional immune responses by honey bee larvae during invasion by the bacterial pathogen, Paenibacillus larvae. J. Invertebr. Pathol. 2004, 85, 105–111. [Google Scholar] [CrossRef]
- Evans, J.D.; Aronstein, K.; Chen, Y.P.; Hetru, C.; Imler, J.L.; Jiang, H.; Kanost, M.; Thompson, G.J.; Zou, Z.; Hultmark, D. Immune pathways and defence mechanisms in honey bees Apis mellifera. Insect Mol. Biol. 2006, 15, 645–656. [Google Scholar] [CrossRef] [PubMed]
- Decanini, L.I.; Collins, A.M.; Evans, J.D. Variation and heritability in immune gene expression by diseased honeybees. J. Hered. 2007, 98, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Chan, Q.W.T.; Melathopoulos, A.P.; Pernal, S.F.; Foster, L.J. The innate immune and systemic response in honey bees to a bacterial pathogen, Paenibacillus larvae. BMC Genomics 2009, 10, 387. [Google Scholar] [CrossRef] [PubMed]
- Wedenig, M.; Riessberger-Gallé, U.; Crailsheim, K. A substance in honey bee larvae inhibits the growth of Paenibacillus larvae larvae. Apidologie 2003, 34, 43–51. [Google Scholar] [CrossRef]
- Rothenbuhler, W.C. Behaviour genetics of nest cleaning in honeybees. I. Responses of four inbred lines to disease-killed brood. Anim. Behav. 1964, 12, 578–583. [Google Scholar] [CrossRef]
- Klaudiny, J.; Bachanová, K.; Kohútová, L.; Dzúrová, M.; Kopernický, J.; Majtán, J. Expression of larval jelly antimicrobial peptide defensin1 in Apis mellifera colonies. Biologia 2012, 67, 200–211. [Google Scholar] [CrossRef]
- Schäfer, M.O.; Genersch, E.; Fünfhaus, A.; Poppinga, L.; Formella, N.; Bettin, B.; Karger, A. Rapid identification of differentially virulent genotypes of Paenibacillus larvae, the causative organism of American foulbrood of honey bees, by whole cell MALDI-TOF mass spectrometry. Vet. Microbiol. 2014, 170, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.Q.; O’Connor, Ch.J.; Roberton, A.M. Antibacterial actions of fatty acids and monoglycerides against Helicobacter pylori. FEMS Immunol. Microbiol. 2003, 36, 9–17. [Google Scholar] [CrossRef]
- Skřivanová, E.; Marounek, M. Influence of pH on antimicrobial activity of organic acids against rabbit enteropathogenic strain of Escherichia coli. Folia Microbiol. 2007, 52, 70–72. [Google Scholar] [CrossRef]
- Chauvin, R. Nutrition de I´abeille (Bee nutrition). Ann. Nutr. Aliment. 1962, 16, 41–63. [Google Scholar]
- Wardell, G. Honeybee nutrition and European foulbrood. In Proceedings of the International Symposium of European Foulbrood, Quebec, Canada, 18–20 October 1981; pp. 111–126. [Google Scholar]
- Bonvehí, J.S.; Jordá, R.E. Studie über die mikrobiologische Qualität und bakteriostatische Aktivität des Weiselfuttersaft (Gelée Royale): Beeinflussung durch organische Säuren. Deutsche Lebensmittel-Rundschau 1991, 87, 256–259. [Google Scholar]
- Bloodworth, B.C.; Harn, C.S.; Hock, C.T.; Boon, Y.O. Liquid chromatographic determination of trans-10-hydroxy-2-decenoic acid content of commercial products containing royal jelly. J. AOAC. Int. 1995, 78, 1019–1023. [Google Scholar] [PubMed]
- Genc, M.; Aslan, A. Determination of trans-10-hydroxy-2-decenoic acid content in pure royal jelly and royal jelly products by column liquid chromatography. J. Chromatogr. A 1999, 839, 265–268. [Google Scholar] [CrossRef]
- Ferioli, F.; Marcazzan, G.L.; Caboni, M.F. Determination of (E)-10-hydroxy-2-decenoic acid content in pure royal jelly: A comparison between a new CZE method and HPLC. J. Sep. Sci. 2007, 30, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Amoedo, L.H.; Almeida-Muradian, L.B. Physicochemical composition of pure and adulterated royal jelly. Quim. Nova 2007, 30, 257–259. [Google Scholar] [CrossRef]
- Zheng, H.Q.; Hu, F.L.; Dietemann, V. Changes in composition of royal jelly harvested at different times: Consequences for quality standards. Apidologie 2010, 42, 39–47. [Google Scholar] [CrossRef]
- Isidorov, V.A.; Bakier, S.; Grzech, I. Gas chromatographic-mass spectrometric investigation of volatile and extractable compounds of crude royal jelly. J. Chromatogr. B 2012, 885, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Wytrychowski, M.; Chenavas, S.; Daniele, G.; Casabianca, H.; Batteau, M.; Guibert, S.; Brion, B. Physicochemical characterization of French royal jelly: Comparison with commercial royal jellies and royal jellies produced through artificial bee-feeding. J. Food Compos. Anal. 2013, 29, 126–133. [Google Scholar] [CrossRef]
- Kanelis, D.; Tananaki, C.; Liolios, V.; Dimou, M.; Goras, G.; Rodopoulou, M.A.; Karazafiris, E.; Thrasyvoulou, A. A suggestion for royal jelly specifications. Arh.Hig. Rada.Toksikol. 2015, 66. [Google Scholar] [CrossRef]
- Flanjak, I.; Jakovljevic, M.; Kenjeric, D.; Stokanovic, M.C.; Primorac, L.; Rajs, B.B. Determination of (2E)-10-hydroxydec-2-enoic acid in Croatian royal jelly by high-performance liquid chromatography. Croat. J. Food Sci. Technol. 2017, 9, 152–157. [Google Scholar] [CrossRef]
- Noda, N.; Umebayashi, K.; Nakatani, T.; Miyahara, K.; Ishiyama, K. Isolation and characterization of some hydroxy fatty and phosphoric acid esters of 10-hydroxy-2-decenoic acid from the royal jelly of honeybees (Apis mellifera). Lipids 2005, 40, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Jung-Hoffmann, I. Die Determination von Königin und Arbeiterin der Honigbiene. Zeitschrift für Bienenforschung 1966, 8, 296–322. [Google Scholar]
- Wang, Y.; Ma, L.; Zhang, W.; Cui, X.; Wang, H.; Xu, B. Comparison of the nutrient composition of royal jelly and worker jelly of honey bees (Apis mellifera). Apidologie 2016, 47, 48–56. [Google Scholar] [CrossRef]
- Loncaric, I.; Derakhshifar, I.; Oberlerchner, J.T.; Köglberger, H.; Moosbeckhofer, R. Genetic diversity among isolates of Paenibacillus larvae from Austria. J. Invertebr. Pathol. 2009, 100, 44–46. [Google Scholar] [CrossRef] [PubMed]
- Versalovic, J.; Schneider, M.; De Bruijn, F.J.; Lupski, J.R. Genomic fingerprinting of bacteria using repetitive sequence-based polymerase chain reaction. Methods Mol. Cell. Biol. 1994, 5, 25–40. [Google Scholar]
- Dingman, D.W.; Stahly, D.P. Medium promoting sporulation of Bacillus larvae and metabolism of medium components. Appl. Environ. Microbiol. 1983, 46, 860–869. [Google Scholar] [PubMed]
- Alvarado, I.; Phui, A.; Elekonich, M.M.; Abel-Santos, E. Requirements for in vitro germination of Paenibacillus larvae spores. J. Bacteriol. 2013, 195, 1005–1011. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
| Strain | Source | ERIC Genotype | MIC* (μg/μL) |
|---|---|---|---|
| CCUG 28515 | CCUG | I | 1.05 ± 0.16 |
| LMG 9820 | LMG | I | 0.60 ± 0.00 |
| CCM 4483 | CCM | I | 1.20 ± 0.00 |
| CCM 4486 | CCM | II | 1.28 ± 0.15 |
| CCM 38a | CCM | III | 2.80 ± 0.00 |
| CCM 38b | CCM | IV | 2.60 ± 0.00 |
| CCM 39 | CCM | III | 2.60 ± 0.00 |
| 1-99 | diseased brood | II | 1.47 ± 0.14 |
| 2-99 | diseased brood | II | 1.40 ± 0.00 |
| 3-99 | diseased brood | II | 1.02 ± 0.18 |
| 4-99 | diseased brood | II | 1.27 ± 0.14 |
| 1-09 | diseased brood | II | 1.20 ± 0.00 |
| 3-09 | diseased brood | II | 1.78 ± 0.04 |
| 4-09 | diseased brood | II | 1.11 ± 0.09 |
| 5-09 | diseased brood | II | 1.40 ± 0.00 |
| 6-09 | diseased brood | II | 1.58 ± 0.04 |
| 7-09 | diseased brood | II | 1.35 ± 0.09 |
| 1-11 | ground stock | II | 0.98 ± 0.04 |
| 2-11 | ground stock | II | 1.35 ± 0.09 |
| 4-11 | ground stock | II | 1.60 ± 0.00 |
| 7-11 | ground stock | II | 1.33 ± 0.11 |
| 8-11 | diseased brood | II | 1.38 ± 0.04 |
| 11-11 | ground stock | II | 1.50 ± 0.11 |
| 13-11 | ground stock | II | 1.18 ± 0.04 |
| 14-11 | ground stock | II | 1.20 ± 0.00 |
| 17-11 | ground stock | II | 0.93 ± 0.11 |
| 21-11 | ground stock | II | 1.78 ± 0.04 |
| 22-11 | ground stock | II | 1.50 ± 0.11 |
| 23-11 | ground stock | II | 1.38 ± 0.04 |
| 24-11 | ground stock | II | 1.40 ± 0.00 |
| 25-11 | ground stock | II | 1.20 ± 0.00 |
| 26-11 | ground stock | II | 1.43 ± 0.17 |
| 34-11 | ground stock | II | 1.58 ± 0.04 |
| 1-14 | diseased brood | II | 1.38 ± 0.04 |
| 2-14 | diseased brood | II | 1.58 ± 0.04 |
| 3-14 | diseased brood | II | 1.40 ± 0.00 |
| 4-14 | diseased brood | II | 0.60 ± 0.00 |
| 5-14 | diseased brood | II | 1.38 ± 0.04 |
| 6-14 | diseased brood | II | 1.00 ± 0.00 |
| 7-14 | diseased brood | II | 1.40 ± 0.00 |
| Strain | ERIC Genotype | MIC (μg/μL) * | |
|---|---|---|---|
| Vegetative Cells | Spores | ||
| CCUG 28515 | I | 1.05 ± 0.16 | 1.05 ± 0.16 |
| LMG 9820 | I | 0.60 ± 0.00 | 0.60 ± 0.00 |
| CCM 4483 | I | 1.20 ± 0.00 | 0.98 ± 0.04 |
| CCM 4486 | II | 1.28 ± 0.15 | 0.60 ± 0.00 |
| PL 2-11 | II | 1.35 ± 0.09 | 1.18 ± 0.04 |
| PL 4-11 | II | 1.60 ± 0.00 | 1.20 ± 0.00 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šedivá, M.; Laho, M.; Kohútová, L.; Mojžišová, A.; Majtán, J.; Klaudiny, J. 10-HDA, A Major Fatty Acid of Royal Jelly, Exhibits pH Dependent Growth-Inhibitory Activity Against Different Strains of Paenibacillus larvae. Molecules 2018, 23, 3236. https://doi.org/10.3390/molecules23123236
Šedivá M, Laho M, Kohútová L, Mojžišová A, Majtán J, Klaudiny J. 10-HDA, A Major Fatty Acid of Royal Jelly, Exhibits pH Dependent Growth-Inhibitory Activity Against Different Strains of Paenibacillus larvae. Molecules. 2018; 23(12):3236. https://doi.org/10.3390/molecules23123236
Chicago/Turabian StyleŠedivá, Mária, Maroš Laho, Lenka Kohútová, Andrea Mojžišová, Juraj Majtán, and Jaroslav Klaudiny. 2018. "10-HDA, A Major Fatty Acid of Royal Jelly, Exhibits pH Dependent Growth-Inhibitory Activity Against Different Strains of Paenibacillus larvae" Molecules 23, no. 12: 3236. https://doi.org/10.3390/molecules23123236
APA StyleŠedivá, M., Laho, M., Kohútová, L., Mojžišová, A., Majtán, J., & Klaudiny, J. (2018). 10-HDA, A Major Fatty Acid of Royal Jelly, Exhibits pH Dependent Growth-Inhibitory Activity Against Different Strains of Paenibacillus larvae. Molecules, 23(12), 3236. https://doi.org/10.3390/molecules23123236







