Propolis Induces AQP3 Expression: A Possible Way of Action in Wound Healing
Abstract
1. Introduction
2. Results
2.1. Cell Proliferation and Metabolism
2.2. Scratch Wound Repair of HaCaT in the Presence of Propolis
2.3. Propolis Chemoattractant Effect
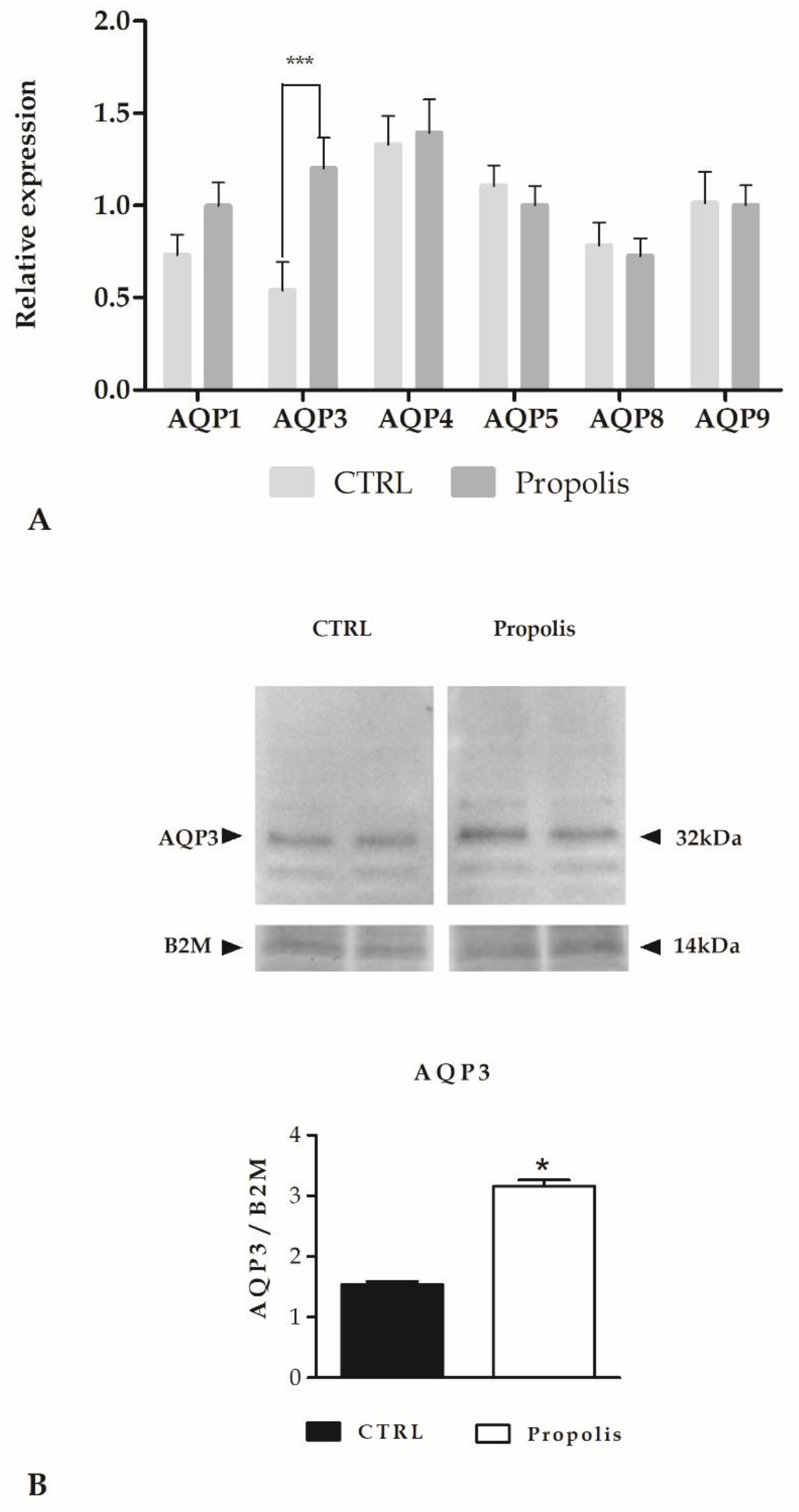
2.4. Aquaporins (AQPs) Expression upon Propolis Exposure
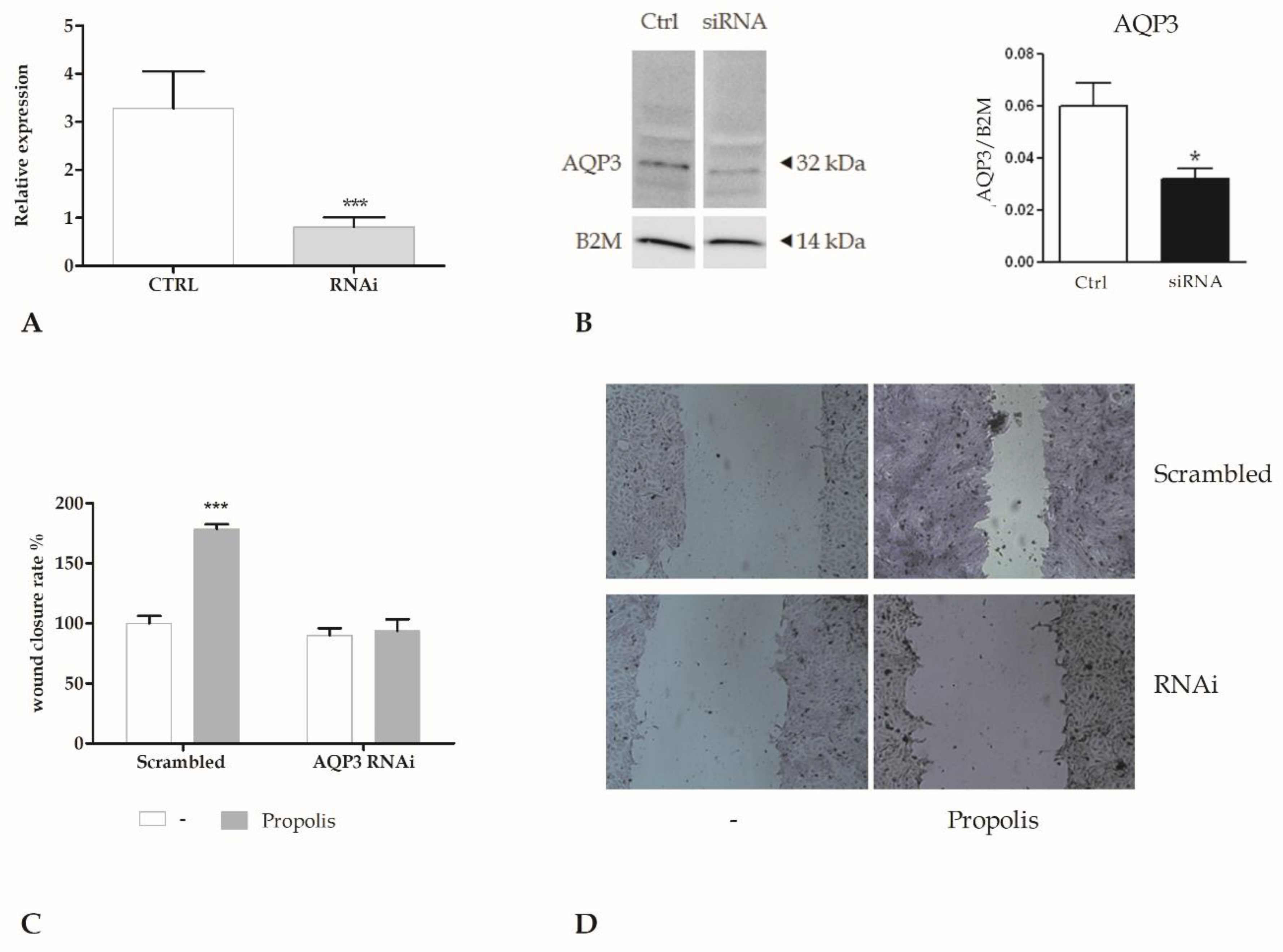
2.5. Role of AQP3 in H2O2 Influx
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Reagents
4.2. Calcein-AM Assay
4.3. Scratch Wound Assay
4.4. Platelet Lysate Preparation
4.5. Cell Migration Assay
4.6. Quantitative Reverse Transcriptase PCR (qRT-PCR)
| β-actin | 5′-TCCCTGGAGAAGAGCTACGA-3′ |
| 5′-AGCACTGTGTTGGCGTACAG-3′ | |
| GADPH | 5′-AATCCCATCACCATCTTCCA-3′ |
| 5′-TGGACTCCACGACGTACTCA-3′ | |
| AQP1 | 5′-TAAGGAGAGGAAAGTTCCAG-3′ |
| 5′-AAAGGCAGACATACACATAC-3′ | |
| AQP3 | 5′-CTGTGTATGTGTATGTCTGC-3′ |
| 5′-TTATGACCTGACTTCACTCC-3′ | |
| AQP4 | 5’-GCTGTGATTCCAAACGGACTGATC-3’ |
| 5′-CTGACTCCTGTTGTCCTCCACCTC-3′ | |
| AQP5 | 5′-GCTGGCACTCTGCATCTTCGC-3′ |
| 5′-AGGTAGAAGTAAAGGATGGCAGC-3′ | |
| AQP8 | 5′- GGAGATAAGAGTATCTTGCAC-3′ |
| 5′- CTTGTCATTGCCAAATTCAG-3′ | |
| AQP9 | 5′-GTATTGGTAGAAACAGGAGTC-3′ |
| 5′-GGACAATCAAGATGAACGTG-3′ |
4.7. RNA Interference
4.8. Immunoblotting
4.9. Measurement of Intracellular ROS
4.10. Hydrogen Peroxide Assay
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Martinotti, S.; Ranzato, E. Propolis: A new frontier for wound healing? Burns Trauma 2015, 3, 9. [Google Scholar] [CrossRef]
- Pietta, P.G.; Gardana, C.; Pietta, A.M. Analytical methods for quality control of propolis. Fitoterapia 2002, 73, S7–S20. [Google Scholar] [CrossRef]
- Oryan, A.; Alemzadeh, E.; Moshiri, A. Potential role of propolis in wound healing: Biological properties and therapeutic activities. Biomed. Pharmacother. 2018, 98, 469–483. [Google Scholar] [CrossRef] [PubMed]
- Ranzato, E.; Patrone, M.; Mazzucco, L.; Burlando, B. Platelet lysate stimulates wound repair of HaCaT keratinocytes. Br. J. Dermatol. 2008, 159, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Ranzato, E.; Martinotti, S.; Burlando, B. Wound healing properties of jojoba liquid wax: An in vitro study. J. Ethnopharmacol. 2011, 134, 443–449. [Google Scholar] [CrossRef]
- Verkman, A.S. Aquaporins at a glance. J. Cell Sci. 2011, 124, 2107–2112. [Google Scholar] [CrossRef]
- Hara-Chikuma, M.; Verkman, A.S. Roles of aquaporin-3 in the epidermis. J. Invest. Dermatol. 2008, 128, 2145–2151. [Google Scholar] [CrossRef] [PubMed]
- Martinotti, S.; Laforenza, U.; Patrone, M.; Moccia, F.; Ranzato, E. Honey-Mediated Wound Healing: H(2)O(2) Entry through AQP3 Determines Extracellular Ca(2+) Influx. Int. J. Mol. Sci. 2019, 20. [Google Scholar]
- Ranzato, E.; Mazzucco, L.; Patrone, M.; Burlando, B. Platelet lysate promotes in vitro wound scratch closure of human dermal fibroblasts: Different roles of cell calcium, P38, ERK and PI3K/AKT. J. Cell Mol. Med. 2009, 13, 2030–2038. [Google Scholar] [CrossRef]
- Hara-Chikuma, M.; Verkman, A.S. Aquaporin-3 facilitates epidermal cell migration and proliferation during wound healing. J. Mol. Med. (Berl) 2008, 86, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Hara-Chikuma, M.; Verkman, A.S. Prevention of skin tumorigenesis and impairment of epidermal cell proliferation by targeted aquaporin-3 gene disruption. Mol. Cell Biol. 2008, 28, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Stojko, A.; Scheller, S.; Szwarnowiecka, I.; Tustanowski, J.; Ostach, H.; Obuszko, Z. Biological properties and clinical application of propolis. VIII. Experimental observation on the influence of ethanol extract of propolis (EEP) on the regeneration of bone tissue. Arzneimittelforschung 1978, 28, 35–37. [Google Scholar]
- Ramos, I.F.; Biz, M.T.; Paulino, N.; Scremin, A.; Della Bona, A.; Barletta, F.B.; Figueiredo, J.A. Histopathological analysis of corticosteroid-antibiotic preparation and propolis paste formulation as intracanal medication after pulpectomy: An in vivo study. J. Appl. Oral Sci. 2012, 20, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Daleprane, J.B.; Abdalla, D.S. Emerging roles of propolis: Antioxidant, cardioprotective, and antiangiogenic actions. Evid. Based Complement. Alternat Med. 2013, 2013, 175135. [Google Scholar] [CrossRef] [PubMed]
- Sforcin, J.M. Propolis and the immune system: A review. J. Ethnopharmacol. 2007, 113, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bankova, V.; Christov, R.; Kujumgiev, A.; Marcucci, M.C.; Popov, S. Chemical composition and antibacterial activity of Brazilian propolis. Z Naturforsch C 1995, 50, 167–172. [Google Scholar] [CrossRef]
- Teixeira, E.W.; Message, D.; Negri, G.; Salatino, A.; Stringheta, P.C. Seasonal variation, chemical composition and antioxidant activity of Brazilian propolis samples. Evid. Based Complement. Alternat. Med. 2010, 7, 307–315. [Google Scholar] [CrossRef]
- Hashemi, J.M. Biological Effect of Bee Propolis: A Review. Eur. J. Appl. Sci. 2016, 8, 311–318. [Google Scholar]
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef]
- Sforcin, J.M.; Orsi, R.O.; Bankova, V. Effect of propolis, some isolated compounds and its source plant on antibody production. J. Ethnopharmacol. 2005, 98, 301–305. [Google Scholar] [CrossRef]
- Orsolic, N.; Knezevic, A.H.; Sver, L.; Terzic, S.; Basic, I. Immunomodulatory and antimetastatic action of propolis and related polyphenolic compounds. J. Ethnopharmacol. 2004, 94, 307–315. [Google Scholar] [CrossRef]
- Nassar, S.A.; Mohamed, A.H.; Soufy, H.; Nasr, S.M.; Mahran, K.M. Immunostimulant effect of Egyptian propolis in rabbits. Sci. World J. 2012, 2012, 901516. [Google Scholar] [CrossRef]
- Lisicic, D.; Benkovic, V.; Ethikic, D.; Blazevic, A.S.; Mihaljevic, J.; Orsolic, N.; Knezevic, A.H. Addition of propolis to irinotecan therapy prolongs survival in ehrlich ascites tumor-bearing mice. Cancer Biother. Radiopharm. 2014, 29, 62–69. [Google Scholar] [CrossRef]
- Mazzucco, L.; Medici, D.; Serra, M.; Panizza, R.; Rivara, G.; Orecchia, S.; Libener, R.; Cattana, E.; Levis, A.; Betta, P.G.; et al. The use of autologous platelet gel to treat difficult-to-heal wounds: A pilot study. Transfusion 2004, 44, 1013–1018. [Google Scholar] [CrossRef]
- Tsai, Y.C.; Wang, Y.H.; Liou, C.C.; Lin, Y.C.; Huang, H.; Liu, Y.C. Induction of oxidative DNA damage by flavonoids of propolis: Its mechanism and implication about antioxidant capacity. Chem. Res. Toxicol. 2012, 25, 191–196. [Google Scholar] [CrossRef]
- Boury-Jamot, M.; Daraspe, J.; Bonte, F.; Perrier, E.; Schnebert, S.; Dumas, M.; Verbavatz, J.M. Skin aquaporins: Function in hydration, wound healing, and skin epidermis homeostasis. Handb. Exp. Pharmacol. 2009, 190, 205–217. [Google Scholar]
- Boury-Jamot, M.; Sougrat, R.; Tailhardat, M.; Le Varlet, B.; Bonte, F.; Dumas, M.; Verbavatz, J.M. Expression and function of aquaporins in human skin: Is aquaporin-3 just a glycerol transporter? Biochim. Biophys. Acta 2006, 1758, 1034–1042. [Google Scholar] [CrossRef]
- Cipriani, V.; Ranzato, E.; Balbo, V.; Mazzucco, L.; Cavaletto, M.; Patrone, M. Long-term effect of platelet lysate on primary fibroblasts highlighted with a proteomic approach. J. Tissue Eng. Regen Med. 2009, 3, 531–538. [Google Scholar] [CrossRef]
- Ranzato, E.; Martinotti, S.; Burlando, B. Epithelial mesenchymal transition traits in honey-driven keratinocyte wound healing: Comparison among different honeys. Wound Repair Regen. 2012, 20, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Xuan, H.; Li, Z.; Yan, H.; Sang, Q.; Wang, K.; He, Q.; Wang, Y.; Hu, F. Antitumor Activity of Chinese Propolis in Human Breast Cancer MCF-7 and MDA-MB-231 Cells. Evid. Based Complement. Alternat. Med. 2014, 2014, 280120. [Google Scholar] [CrossRef]
- Ranzato, E.; Martinotti, S.; Magnelli, V.; Murer, B.; Biffo, S.; Mutti, L.; Burlando, B. Epigallocatechin-3-gallate induces mesothelioma cell death via H2 O2 -dependent T-type Ca2+ channel opening. J. Cell Mol. Med. 2012, 16, 2667–2678. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |


| EC05 | EC50 |
|---|---|
| 0.015% (0.012–0.019%) | 0.048% (0.045–0.052%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinotti, S.; Pellavio, G.; Laforenza, U.; Ranzato, E. Propolis Induces AQP3 Expression: A Possible Way of Action in Wound Healing. Molecules 2019, 24, 1544. https://doi.org/10.3390/molecules24081544
Martinotti S, Pellavio G, Laforenza U, Ranzato E. Propolis Induces AQP3 Expression: A Possible Way of Action in Wound Healing. Molecules. 2019; 24(8):1544. https://doi.org/10.3390/molecules24081544
Chicago/Turabian StyleMartinotti, Simona, Giorgia Pellavio, Umberto Laforenza, and Elia Ranzato. 2019. "Propolis Induces AQP3 Expression: A Possible Way of Action in Wound Healing" Molecules 24, no. 8: 1544. https://doi.org/10.3390/molecules24081544
APA StyleMartinotti, S., Pellavio, G., Laforenza, U., & Ranzato, E. (2019). Propolis Induces AQP3 Expression: A Possible Way of Action in Wound Healing. Molecules, 24(8), 1544. https://doi.org/10.3390/molecules24081544









