Extraction, Characterization and Antioxidant Activity in vitro of Proteins from Semen Allii Fistulosi
Abstract
1. Introduction
2. Results and Discussion
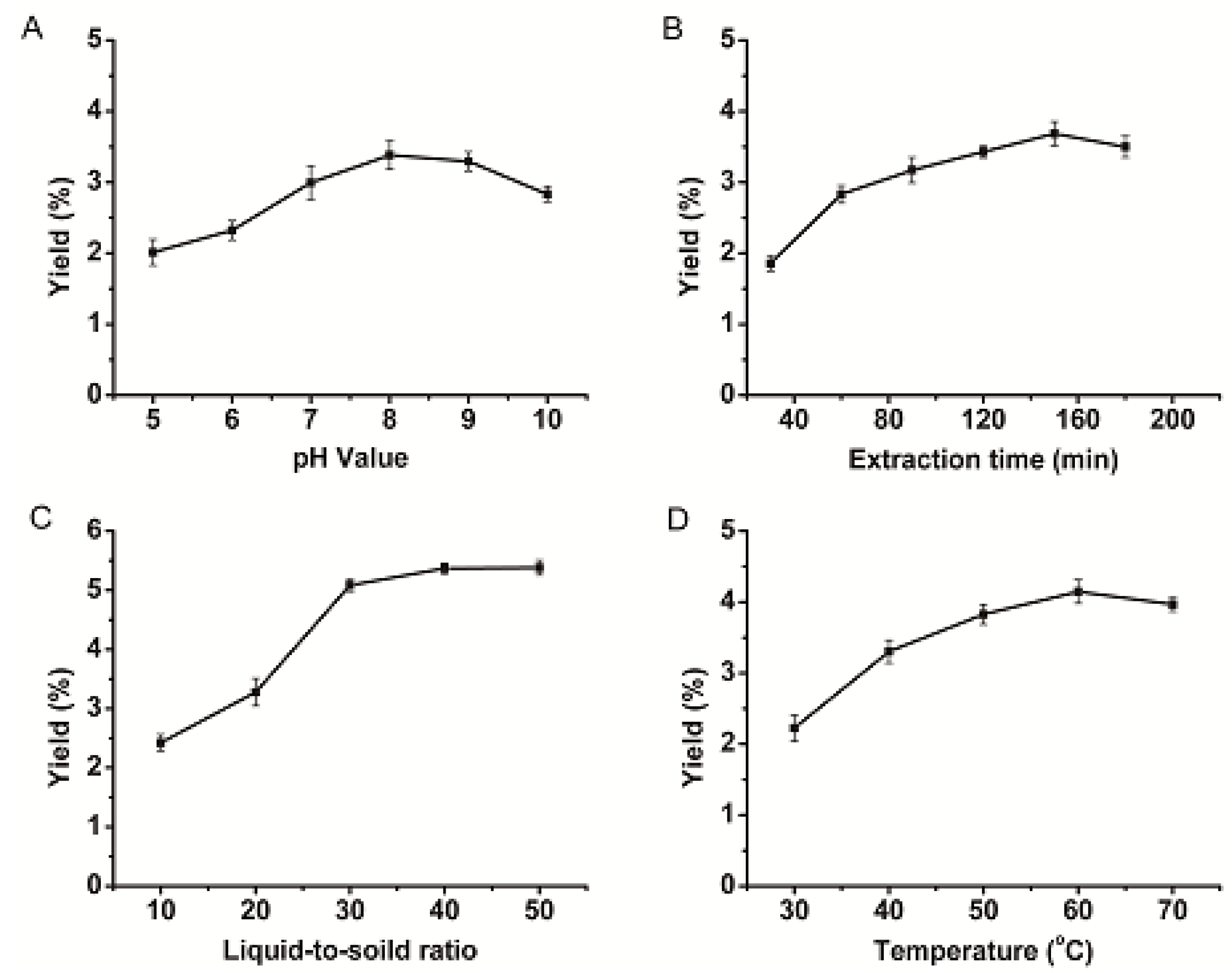
2.1. PSAF Extraction Process Parameters
2.2. Optimization for PSAF Extraction
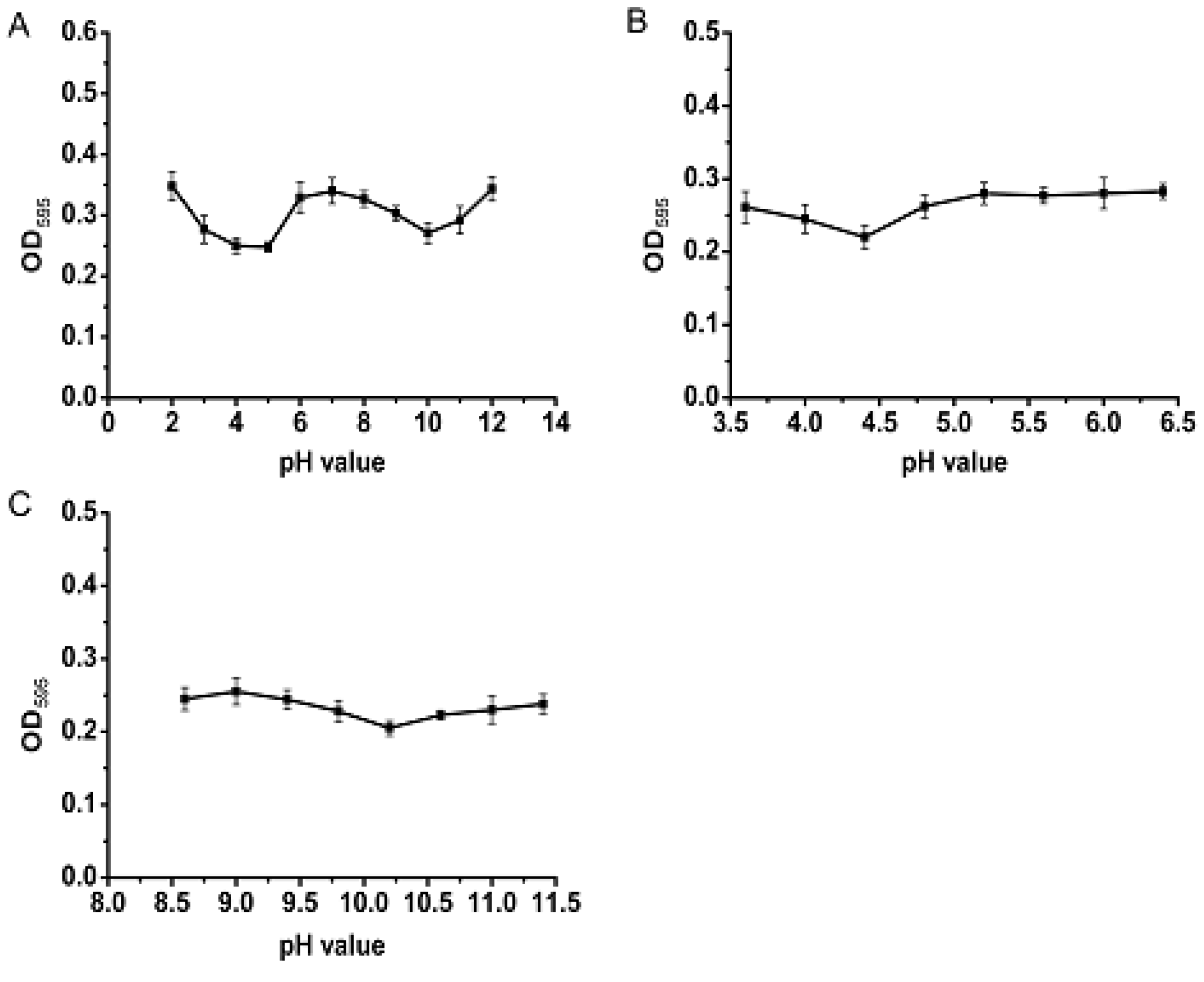
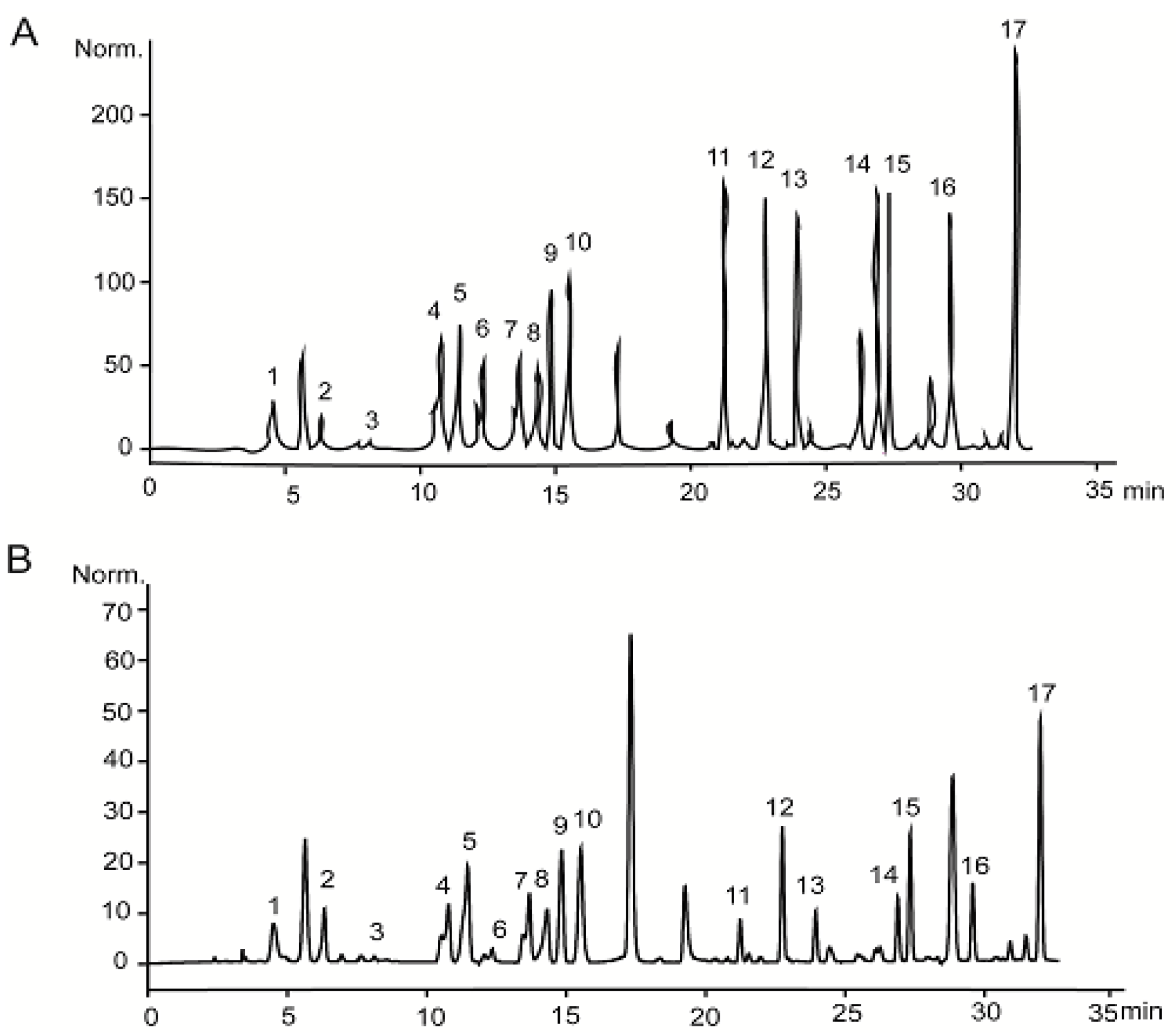
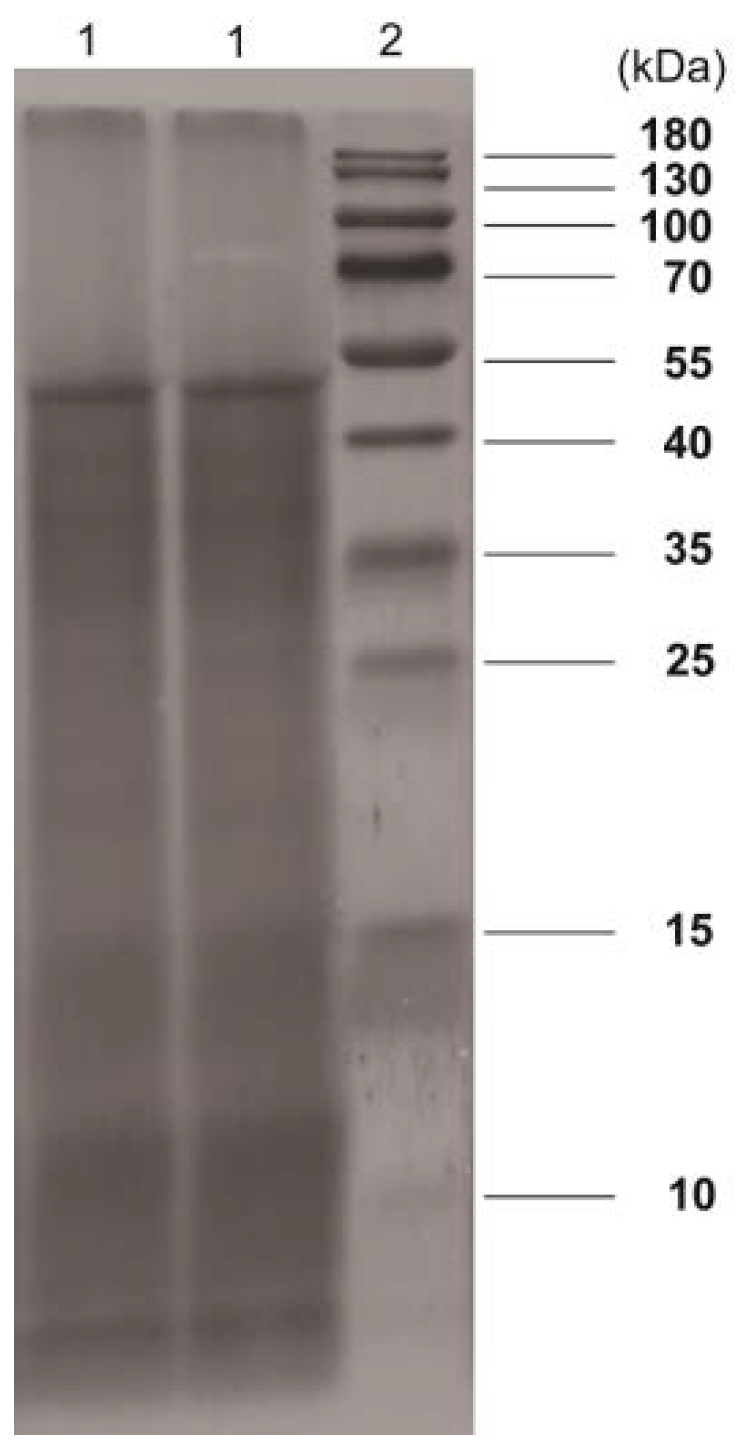
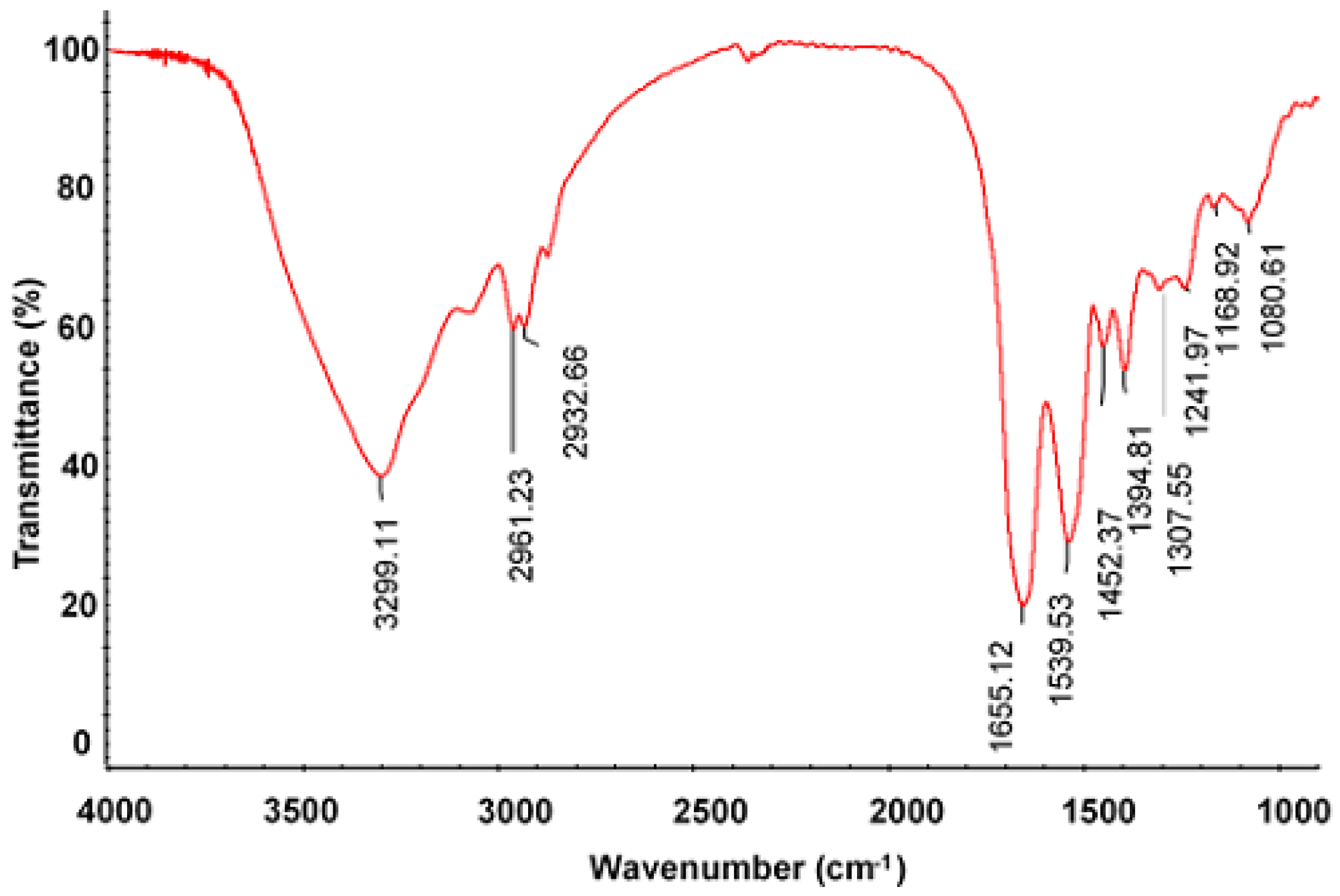
2.3. Characterization of PSAF
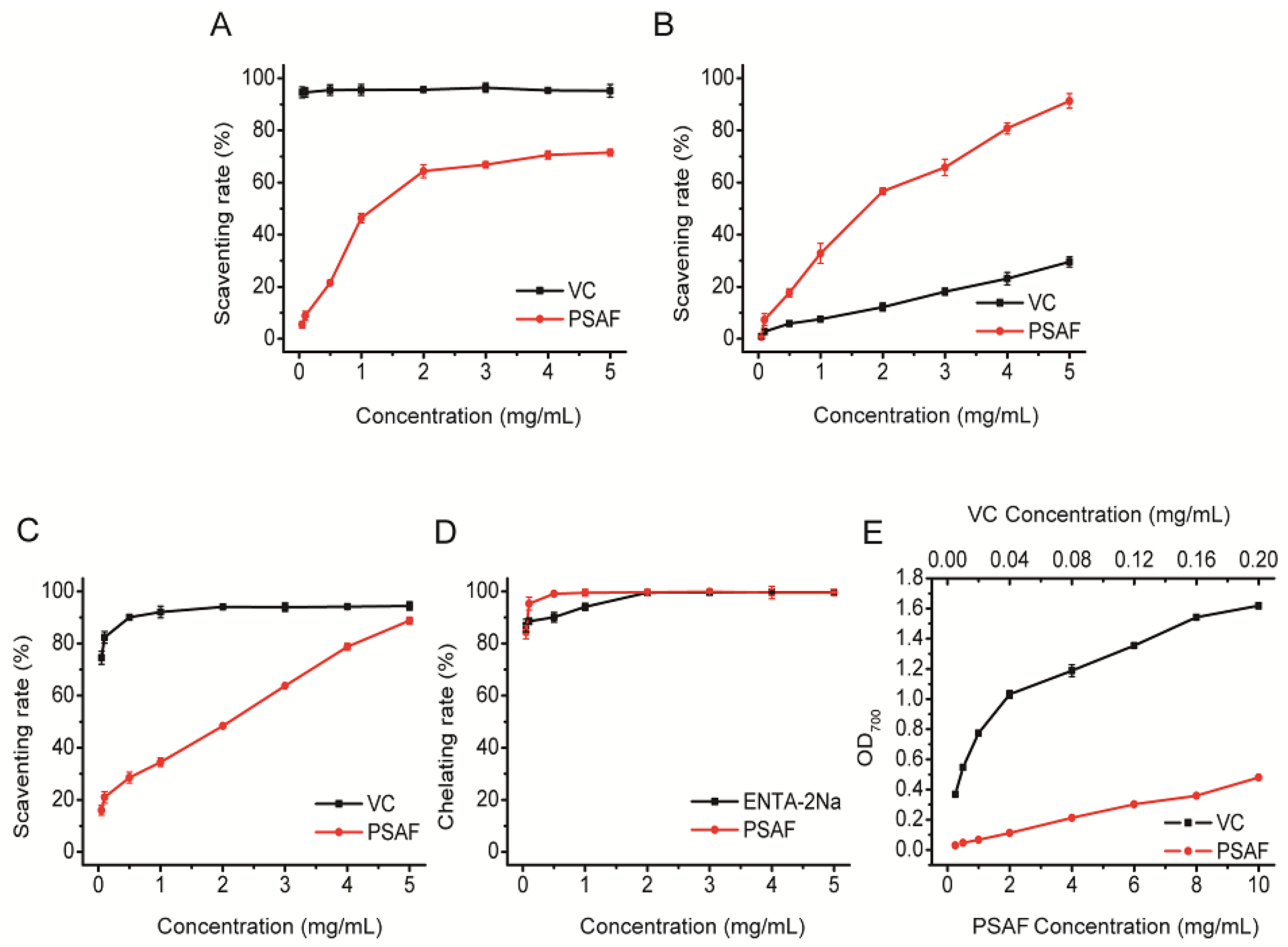
2.4. Antioxidant Activities in vitro
3. Materials and Methods
3.1. Materials and Equipment
3.2. Sample Pretreatment
3.3. Protein Content Determination
3.4. Extraction Optimization
3.5. Characterization of PSAF Isoelectric Point and Amino Acid Composition
3.6. SDS-PAGE and FT-IR Spectra Analysis
3.7. DPPH Radical Scavenging Assay
3.8. Hydroxyl and Superoxide Radical Scavenging Activity
3.9. Fe2+ Capacity Activity and Reducing Power Determination
3.10. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yuan, Y.; Lai, W.; Yang, Q.; Sun, L.N.; Chen, W.S. Study on chemica constituents of Semen Allii Fistulosi (II). J. Pharm. Pract. 2010, 28, 426–428. [Google Scholar] [CrossRef]
- Liu, J.T.; Wang, S.; Zhang, W.M.; Su, W.; Zhao, L. Review on Research Progress of Bioactive Constituents in Allium Species. Food Sci. 2007, 28, 348–350. [Google Scholar] [CrossRef]
- Wei, Y.; Shao, A.J.; Cheng, M.; Fu, G.F.; Chen, M.L.; Lin, S.F. Study on the Characters and Microscopic Identification of Semen Allii Fistulosi and chinese chive seed. Mod. Chin. Med. 2012, 14, 20–21. [Google Scholar] [CrossRef]
- Lai, W. Studies on the Active Constituents and Quality Control of the seeds of Allium fistulosum. Ph.D. Thesis, The Second Military Medical University, Shang Hai, China, 2010. [Google Scholar]
- Devappa, R.K.; Makkar, H.P.; Becker, K. Nutritional, biochemical, and pharmaceutical potential of proteins and peptides from jatropha: Review. J. Agric. Food Chem. 2010, 58, 6543–6555. [Google Scholar] [CrossRef]
- Sun, Y. Chemical composition activities of antioxidation and anticancer of Tetrastigma hemsleyanum Diels et Gilg. Ph.D. Thesis, Nanchang University, Jiang Xi, China, 2018. [Google Scholar]
- Han, C.H.; Lin, Y.F.; Lin, Y.S.; Lee, T.L.; Huang, W.J.; Lin, S.Y.; Hou, W.C. Effects of yam tuber protein, dioscorin, on attenuating oxidative status and learning dysfunction in D-galactose-induced BALB/c mice. Food Chem. Toxicol. 2014, 65, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Chuethong, J.; Oda, K.; Sakurai, H.; Saiki, I.; Leelamanit, W. Cochinin B, a novel ribosome-inactivating protein from the seeds of Momordica cochinchinensis. Biol. Pharm. Bull. 2007, 30, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Chu, K.T.; Ng, T.B. Smilaxin, a novel protein with immunostimulatory, antiproliferative, and HIV-1-reverse transcriptase inhibitory activities from fresh Smilax glabra rhizomes. Biochem. Biophys. Res. Commun. 2006, 340, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Zhang, D.; Wu, J.; Chen, C.; Xu, Z.; Yang, H.; Zhou, P. Antidiabetic, antihyperlipidemic and antioxidant activities of a novel proteoglycan from Ganoderma lucidum fruiting bodies on db/db mice and the possible mechanism. PLoS ONE 2013, 8, e68332. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Yin, G.Y.; Ding, M.; Tao, Z.X. Study on extraction and antioxidant activity of protein from Chinese chive seed. Sci. Technol. Food Ind. 2014, 35, 291–294. [Google Scholar] [CrossRef]
- Cao, W.; Pan, C.L.; Tang, W.; Sun, P.D. Extraction and antioxidant activity of camellia seed protein by different methods. Chin. Oils Fats 2014, 8, 26–30. [Google Scholar] [CrossRef]
- Wang, S.R.; Meng, C.; Zhang, S.J.; Lu, Y.; Li, W.Z. Rapid Purification of Antioxidant Proteins from Toona sinensis Seeds Using Affinity Chromatography. Nat. Prod. Res. Dev. 2017, 1, 96–100. [Google Scholar] [CrossRef]
- Pan, Y.; Lv, C.J.; X, C.L.; Pei, H.Y.; He, C.F.; Dong, Y.M.; Wang, C.T. Preliminary study on enzymatic extraction method and the effect of mung bean protein. Sci. Technol. Food Ind. 2010, 31, 238–241. [Google Scholar]
- Shang, H.L.; Meng, X.; Zhang, T. Extraction and SDS-PAGE Analysis of Proteins from Job’s Tears Seed. Chin. Agric. Sci. Bull. 2012, 28, 260–265. [Google Scholar] [CrossRef]
- Fan, S.H.; Liu, Y.R.; Yuan, C. Preparation and Functional Properties of Protein Isolates from Pumpkin Seeds. Food Sci. 2010, 31, 97–100. [Google Scholar] [CrossRef]
- Tan, S.H.; Mailer, R.J.; Blanchard, C.L.; Agboola, S.O. Canola proteins for human consumption: Extraction, profile, and functional properties. J. Food Sci. 2011, 76, R16–R28. [Google Scholar] [CrossRef] [PubMed]
- Jun, H.; Hua, X.; Liang, L.I.; Ting, L.I.; Zhen, L.C. Enzymolysis of residue of rice and determination of its protein isoelectric point. Sci. Technol. Food Ind. 2008, 29, 174–176. [Google Scholar] [CrossRef]
- Liu, G.; Wang, H.; Zhou, B.H. Analysis of 17 amion acids in Tricholoma matsutakes from Yunnan Province and its nutritional evaluation. Chin. J. Hosp. Pharm. 2008, 28, 552–554. [Google Scholar] [CrossRef]
- Tan, E.S.; Ying, Y.N.; Gan, C.Y. A comparative study of physicochemical characteristics and functionalities of pinto bean protein isolate (PBPI) against the soybean protein isolate (SPI) after the extraction optimisation. Food Chem. 2014, 152, 447–455. [Google Scholar] [CrossRef]
- Wang, F.; Qiao, L.; Zhang, Q.Q.; Shen, B. Amino Acid Composition and Nutritional Evaluation of Mulberry Leaves. Food Sci. 2015, 36, 225–228. [Google Scholar] [CrossRef]
- Hébert, E.M.; Raya, R.R.; Giori, G.S.D. Use of SDS-PAGE of cell-wall proteins for rapid differentiation of Lactobacillus delbrueckii subsp. lactis and Lactobacillus helveticus. Biotechnol. Lett. 2000, 22, 1003–1006. [Google Scholar] [CrossRef]
- Jamin, N.; Dumas, P.; Moncuit, J.; Fridman, W.H.; Teillaud, J.L.; Williams, C.G.P. Highly resolved chemical imaging of living cells by using synchrotron infrared microspectrometry. Proc. Natl. Acad. Sci. USA 1998, 95, 4837–4840. [Google Scholar] [CrossRef] [PubMed]
- Kong, J.; Yu, S. Fourier transform infrared spectroscopic analysis of protein secondary structures. Acta Biochim. Biophys. Sin. 2007, 39, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Ru, Y.; Chen, F.; Wang, X.; Zhao, X.; Ao, Q.l. FTIR spectroscopic characterization of soy proteins obtained through AOT reverse micelles. Food Hydrocoll. 2013, 31, 435–437. [Google Scholar] [CrossRef]
- Carpenter, J.F.; Crowe, J.H. An infrared spectroscopic study of the interactions of carbohydrates with dried proteins. Biochemistry 1989, 28, 3916. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.L.; Kong, L.C.; Yin, C.P.; Jiao, J.Q.; Xiao, W.X. Extraction optimization by response surface methodology, purification and principal antioxidant metabolites of red pigments extracted from bayberry (Myrica rubra) pomace. LWT--Food Sci. Technol. 2013, 51, 343–347. [Google Scholar] [CrossRef]
- Spencer, J.P.; Jenner, A.; Aruoma, O.I.; Evans, P.J.; Kaur, H.; Dexter, D.T.; Jenner, P.; Lees, A.J.; Marsden, D.C.; Halliwell, B. Intense oxidative DNA damage promoted by L-dopa and its metabolites Implications for neurodegenerative disease. FEBS Lett. 1994, 353, 246–250. [Google Scholar] [CrossRef]
- Li, S.; Shah, N.P. Antioxidant and antibacterial activities of sulphated polysaccharides from Pleurotus eryngii and Streptococcus thermophilus ASCC 1275. Food Chem. 2014, 165, 262–270. [Google Scholar] [CrossRef]
- Liu, C.H.; Wang, C.H.; Xu, Z.L.; Wang, Y. Isolation, chemical characterization and antioxidant activities of two polysaccharides from the gel and the skin of Aloe barbadensis Miller irrigated with sea water. Process. Biochem. 2007, 42, 961–970. [Google Scholar] [CrossRef]
- Jiang, C.; Li, X.; Jiao, Y.; Jiang, D.; Zhang, L.; Fan, B.; Zhang, Q. Optimization for ultrasound-assisted extraction of polysaccharides with antioxidant activity in vitro from the aerial root of Ficus microcarpa. Carbohydr. Polym. 2014, 110, 10–17. [Google Scholar] [CrossRef]
- Liu, J.; Luo, J.; Ye, H.; Sun, Y.; Liu, Z.; Zeng, X. In vitro and in vivo antioxidant activity of exopolysaccharides from endophytic bacterium Paenibacillus polymyxa EJS-3. Carbohydr. Polym. 2010, 82, 1278–1283. [Google Scholar] [CrossRef]
- Yang, Z.K.; Wang, X.L.; Long, S.H.; Hao, Z.B.; San, Z.Z.; Zhou, F.F. Determination of Protein in Soybean Stems and Leaves by Bradford Method. Hubei Agr. Sci. 2012, 51, 4610–4612. [Google Scholar] [CrossRef]
- Min, J.H.; Li, J.K.; Chen, T. Study on the optimization of extraction technology of silkwormpupa protein. J. Shaanxi Normal Univ. 2009, 37, 92–96. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Liu, X.; Hao, J.; Shan, X.; Zhang, X.; Zhao, X.; Li, Q.; Wang, X.; Cai, C.; Li, G.; Yu, G. Antithrombotic activities of fucosylated chondroitin sulfates and their depolymerized fragments from two sea cucumbers. Carbohydr. Polym. 2016, 152, 343–350. [Google Scholar] [CrossRef]
- Yuan, Q.; Xie, Y.; Wang, W.; Yan, Y.; Ye, H.; Jabbar, S.; Zeng, X. Extraction optimization, characterization and antioxidant activity in vitro of polysaccharides from mulberry (Morus alba L.) leaves. Carbohydr. Polym. 2015, 128, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Smirnoff, N.; Cumbes, Q.J. Hydroxyl radical scavenging activity of compatible solutes. Phytochemistry 1989, 28, 1057–1060. [Google Scholar] [CrossRef]
- Chen, B.J.; Shi, M.J.; Cui, S.; Hao, S.X.; Hider, R.C.; Zhou, T. Improved antioxidant and anti-tyrosinase activity of polysaccharide from Sargassum fusiforme by degradation. Int. J. Biol. Macromol. 2016, 92, 715–722. [Google Scholar] [CrossRef]
- Decker, E.A.; Welch, B. Role of ferritin as a lipid oxidation catalyst in muscle food. J. Agric. Food Chem. 1990, 38, 674–677. [Google Scholar] [CrossRef]
- Xu, Y.; Niu, X.; Liu, N.; Gao, Y.; Wang, L.; Xu, G.; Li, X.; Yang, Y. Characterization, antioxidant and hypoglycemic activities of degraded polysaccharides from blackcurrant (Ribes nigrum L.) fruits. Food Chem. 2018, 243, 26–35. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| No. | pH (A) | Extraction Time (B) | Liquid-to-Solid Ratio (C) | Temperature (D) | Yield of PSAF (%) |
|---|---|---|---|---|---|
| 1 | 1 | 1 | 1 | 1 | 4.41 |
| 2 | 1 | 2 | 2 | 2 | 4.86 |
| 3 | 1 | 3 | 3 | 3 | 6.29 |
| 4 | 2 | 1 | 3 | 3 | 4.67 |
| 5 | 2 | 2 | 1 | 1 | 4.94 |
| 6 | 2 | 3 | 2 | 2 | 4.63 |
| 7 | 3 | 1 | 2 | 2 | 6.11 |
| 8 | 3 | 2 | 3 | 3 | 4.75 |
| 9 | 3 | 3 | 1 | 1 | 4.52 |
| K1 | 5.19 | 5.06 | 4.60 | 4.62 | |
| K2 | 4.74 | 4.85 | 4.68 | 5.20 | |
| K3 | 5.13 | 5.15 | 5.78 | 5.24 | |
| R | 0.44 | 0.30 | 1.18 | 0.62 |
| Amino Acid | PSAF | FAO/WHO | Amino Acid | PSAF | FAO/WHO |
|---|---|---|---|---|---|
| Aspartic acid | 6.33 | - | Valine | 4.49 | 4.96 |
| Serine | 4.47 | - | Cysteine & methionine | 5.44 | 3.52 |
| Glutamic acid | 15.27 | - | Isoleucine | 2.01 | 4.0 |
| Glycine | 4.2 | - | Leucine | 4.43 | 7.04 |
| Alanine | 4.88 | - | Phenylalanine & tyrosine | 6.74 | 6.08 |
| Histidine | 1.69 | - | Lysine | 6.22 | 5.44 |
| Arginine | 8.78 | - | FLAA | Ile | - |
| Proline | 2.26 | - | SLAA | Leu | - |
| Threonine | 2.81 | 4.0 |
| Factor Level | pH | Extraction Time (min) | Liquid-to-Solid Ratio | Temperature (°C) |
|---|---|---|---|---|
| 1 | 8.0 | 90 | 25:1 | 40 |
| 2 | 8.5 | 120 | 30:1 | 50 |
| 3 | 9.0 | 150 | 35:1 | 60 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuo, M.; Liu, X.-x.; Liu, D.; Zhao, H.-y.; Xuan, L.-l.; Jiang, W.-x.; Li, W.-z. Extraction, Characterization and Antioxidant Activity in vitro of Proteins from Semen Allii Fistulosi. Molecules 2018, 23, 3235. https://doi.org/10.3390/molecules23123235
Zuo M, Liu X-x, Liu D, Zhao H-y, Xuan L-l, Jiang W-x, Li W-z. Extraction, Characterization and Antioxidant Activity in vitro of Proteins from Semen Allii Fistulosi. Molecules. 2018; 23(12):3235. https://doi.org/10.3390/molecules23123235
Chicago/Turabian StyleZuo, Min, Xiao-xiao Liu, Di Liu, Hang-yun Zhao, Lu-lu Xuan, Wen-xian Jiang, and Wan-zhong Li. 2018. "Extraction, Characterization and Antioxidant Activity in vitro of Proteins from Semen Allii Fistulosi" Molecules 23, no. 12: 3235. https://doi.org/10.3390/molecules23123235
APA StyleZuo, M., Liu, X.-x., Liu, D., Zhao, H.-y., Xuan, L.-l., Jiang, W.-x., & Li, W.-z. (2018). Extraction, Characterization and Antioxidant Activity in vitro of Proteins from Semen Allii Fistulosi. Molecules, 23(12), 3235. https://doi.org/10.3390/molecules23123235





