Abstract
Schisandra rubriflora is a dioecious plant of increasing importance due to its lignan composition, and therefore, possible therapeutic properties. The aim of the work was lignan profiling of fruits, leaves and shoots of female (F) and male (M) plants using UHPLC-MS/MS. Additionally, the anti-inflammatory activity of plant extracts and individual lignans was tested in vitro for the inhibition of 15-lipooxygenase (15-LOX), phospholipases A2 (sPLA2), cyclooxygenase 1 and 2 (COX-1; COX-2) enzyme activities. The extracts of fruits, leaves and shoots of the pharmacopoeial species, S. chinensis, were tested for comparison. Twenty-four lignans were monitored. Lignan contents in S. rubriflora fruit extracts amounted to 1055.65 mg/100 g DW and the dominant compounds included schisanhenol, aneloylgomisin H, schisantherin B, schisandrin A, gomisin O, angeloylgomisin O and gomisin G. The content of lignan in leaf extracts was 853.33 (F) and 1106.80 (M) mg/100 g DW. Shoot extracts were poorer in lignans—559.97 (F) and 384.80 (M) mg/100 g DW. Schisantherin B, schisantherin A, 6-O-benzoylgomisin O and angeloylgomisin H were the dominant compounds in leaf and shoot extracts. The total content of detected lignans in S. chinensis fruit, leaf and shoot extracts was: 1686.95, 433.59 and 313.83 mg/100 g DW, respectively. Gomisin N, schisandrin A, schisandrin, gomisin D, schisantherin B, gomisin A, angeloylgomisin H and gomisin J were the dominant lignans in S. chinensis fruit extracts were. The results of anti-inflammatory assays revealed higher activity of S. rubriflora extracts. Individual lignans showed significant inhibitory activity against 15-LOX, COX-1 and COX-2 enzymes.
1. Introduction
Schisandra rubriflora (Franch.) Rehd. et Wils, is a rare and little-known plant species of the genus Schisandra beyond East Asian phytotherapy. S. rubriflora occurs at natural sites in the western Sichuan province of China. It is an endemic species that occurs only in this region [1,2]. S. rubriflora cultivations outside the East Asian region are rare, but attempts have recently been made to grow this species in Europe, including Poland [2,3].
S. rubriflora is a dioecious vine reaching about 3–4 meters in height [1]. S. rubriflora leaves are characterized by elliptical to obovate-elliptical shape, 7–11.5 cm long and 2.5–5.5 cm wide. The leaves are sharp-edged, rarely blunt-edged, and leaf blade edges are finely serrated. Mature berry-shaped fruits of S. rubriflora, collected in the hanging ears, are dark red in color, the size of peas, sitting on about 5–8 cm long stalks [1].
Schisandra chinensis (Turcz.) Baill. is a related species, much better known in terms of medicinal properties, for which cultivation methods have been developed (with cultivations in Europe and America) [1,4,5]. The description of the raw material, i.e., the fruit of the Chinese magnolia vine—Schisandrae chinensis fructus—appeared for the first time in 2008 in the European Pharmacopoeia 6 [6]. The raw material has been used for many years in the official health care of Asian countries [7,8,9,10]. It is a pharmacopoeial species also known in the USA [11]. A World Health Organization (WHO) monograph is also devoted to this plant [12]. Schisandrae chinensis fruit extracts show valuable, proven, therapeutic properties. These include: anti-inflammatory, anti-tumor, and anti-ulcer properties, anti-bacterial and anti-fungal activity; additionally, they can act hepatoprotectively, adaptogenically and ergogenically; these extract also have antioxidant and detoxification properties [4,13,14].
Scientific information about therapeutic properties of S. rubriflora fruits is less available, and its monograph is not listed in any of the world pharmacopoeias [2]. This species is known in the traditional Chinese medicine as a sedative and toning agent, and its fruits are still consumed locally. There are indications regarding the use of this species in the treatment of hepatitis, chronic gastroenteritis and neurasthenia [2,15]. The biological activity of compounds contained in the fruit of this species, described only by Chinese research groups, is limited mainly to the anti-HIV-1 properties, resulting from the inhibition of HIV-1 replication in H9 lymphocytes [16,17]. According to available sources, compounds belonging to the group of dibenzocyclooctadiene lignans as well as nortriterpenoids and bisnortriterpenoids are both responsible for anti-HIV-1 activity [16,18,19]. Furthermore, extracts from S. rubriflora shoots have been shown to effectively reduce the level of GPT (glutamin-pyruvate transaminase) in the blood, which may be useful in the treatment of liver and bile duct diseases [2,20].
Valuable biological properties and therapeutic applications resulting from them are conditioned by the unique chemical composition of S. chinensis [4,21]. Lignans are the main group of secondary metabolites specific to this genus, among which the main role is played by dibenzocyclooctadiene lignans [21,22]. The majority of scientific research has focused on this group of metabolites. They are referred to as “schisandra lignans” due to the characteristic, complicated chemical structure of these compounds as well as the occurrence limited only to this genus. Schisandrin, gomisin A, deoxyschisandrin and schisantherin A and B are listed as the most important from the group of dibenzocyclooctadiene lignans (Figure 1). Recent studies have reported the identification of ever new structures from the group of lignans and their derivatives [22,23,24]. The available data show that dibenzocyclooctadiene lignans, and their derivatives, specific only for S. rubriflora, such as schirubrin A-D, rubrilignans A and B or rubrisandrin A and B are also present in S. rubriflora [16,18,25].
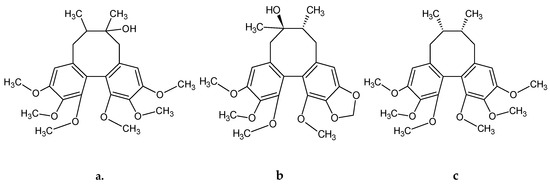
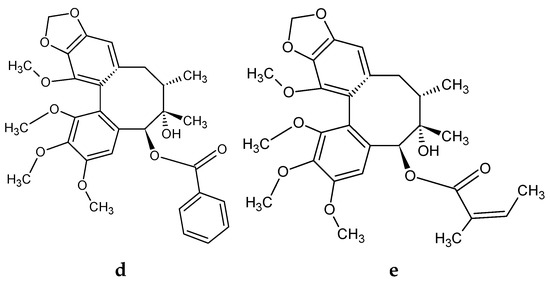
Figure 1.
Example structural formulas of abundant S. rubriflora dibenzocyclooctadiene lignans: (a) schisandrin; (b) gomisin A; (c) deoxyschisandrin; (d) schisantherin A; (e) schisantherin B. Structural formulas drown in: ACD/ChemSkech (Freeware), version 12.00, Advanced Chemistry Development, Inc., Toronto, ON, Canada, www.acdlabs.com, 2010.
There are several studies on the anti-inflammatory activity of S. chinensis fruit extracts [26,27] and some individual lignans [28,29,30,31], but there are no studies on S. chinensis leaf and shoot extracts. importantly, these investigations have not yet been performed in S. rubriflora species. Moreover, there are no studies comparing the results obtained for complex plant material to the results obtained for pure lignans. In this work, we attempt to assess the anti-inflammatory potential of plant extracts and compare it with anti-inflammatory properties of pure lignan samples.
The present study introduces phytochemical characteristics of lignan contents using the UHPLC-MS/MS method in S. rubriflora, including the division of the material into female (F) and male (M) specimens of soil-grown plants. The results were compared to the analyses of pharmacopoeial species–S. chinensis—performed for comparison purposes.
Moreover, the anti-inflammatory potential of S. rubriflora fruits, leaves and shoots of F and M specimens was studied for the first time using estimations based on the inhibition of eicosanoid-generating enzymes; these included cyclooxygenases (COX-1 and COX-2), lipoxygenase (LOX) and secretory phospholipase A2 (sPLA2), reducing the concentrations of prostanoids and leukotrienes. Additionally, the analyses involved individual lignans as well as an artificially created “average sample of S. rubriflora lignan composition”. Comparative studies with S. chinensis shoot, leaf and fruit extracts were also conducted in this study.
2. Results
2.1. Schisandra Rubriflora Lignan Profile
The UHPLC-MS/MS method was used for both qualitative and quantitative analyses of the extracts tested (Tables S1 and S2, Figure S1). Twenty-four lignans were quantified in all analyzed samples, representing four lignan groups: dibenzocyclooctadiene lignans (schisantherin A and B, schisandrin, schisandrin C, gomisin A, D, G, J, N, O, 6-O-benzoylgomisin O, schisandrin A, rubrisandrin A, epigomisin O, schisanhenol, rubriflorin A, angeloylgomisin H and O), aryltetralin lignan (wulignan A1), dibenzylbutane lignans (pregomisin, mesodihydroguaiaretic acid), tetrahydrofuran lignan (fragransin A2) and dihydrobenzofuran neolignans (licarin A and B) [32,33,34].
The total lignan content in the analyzed fruit extracts of S. rubriflora was 1055.65 mg/100 g DW. Quantitatively dominant compounds were: Schisanhenol (268.02 mg/100 g DW), aneloylgomisin H (185.10 mg/100 g DW), schisantherin B (118.07 mg/100 g DW), schisandrin A (104.32 mg/100 g DW), gomisin O (103.64 mg/100 g DW), angeloylgomisin O (76.88 mg/100 g DW) and gomisin G (66.39 mg/100 g DW) (Table 1).

Table 1.
The lignan contents [mg/100g DW] ± SD (n =5) in fruit and leaf and shoot female (F) and male (M) extracts of Schisandra rubriflora.
The presence of twenty-four and twenty-three lignans was found in the analyzed extracts of female (F) and male (M) S. rubriflora specimens, respectively (Table 1). No fragransin A2 was found in leaf extracts of male specimens. The total contents of the tested group of compounds in leaf extracts were: F—853.33 mg/100 g DW and M—1106.80 mg/100 g DW.
Qualitatively dominant compounds in F leaf extracts were: Schisantherin B (291.47 mg/100 g DW), schisantherin A (226.80 mg/100 g DW), 6-O-benzoylgomisin O (134.51 mg/100 g DW) and angeloylgomisin H (100.83 mg/100 g DW). Qualitatively dominant in M leaf extracts were: 6-O-benzoylgomisin O (564.62 mg/100 g DW), angeloylgomisin H (129.28 mg/100 g DW), gomisin D (116.51 mg/100 g DW), schisantherin A (107.17 mg/100 g DW), schisantherin B (104.28 mg/100 g DW) and angeloylgomisin O (48.80 mg/100 g DW) (Table 1).
The presence of twenty-four and twenty-three lignans were found in the analyzed shoot extracts of female (F) and male (M) S. rubriflora specimens, respectively. These were the same compounds that were identified in fruit and leaf extracts (Table 1). The total contents of the tested group of compounds in shoot extracts were: F—559.97 mg/100 g DW and M—384.80 mg/100 g DW.
Qualitatively the dominant compounds in F shoot extracts were: Schisantherin B (239.11 mg/100 g DW), angeloylgomisin H (105.80 mg/100 g DW), schisantherin A (84.35 mg/100 g DW) and 6-O-benzoylgomisin O (72.38 mg/100 g DW). Fragransin A2 was not found in these extracts. Qualitatively dominant compounds in M shoot extracts were: schisantherin B (169.04 mg/100 g DW), angeloylgomisin H (74.73 mg/100 g DW) and 6-O-benzoylgomisin O (52.18 mg/100 g DW) (Table 1).
2.2. Schisandra Chinensis Lignan Profile
The UHPLC-MS/MS analysis of lignans in fruit, leaf and shoot extracts of Schisandra chinensis was performed for comparative purposes. When comparing the results, qualitative similarities and quantitative differences were found between the extracts tested (Table 2). In all analyzed samples, twenty-four lignans were quantified, representing four lignan groups: dibenzocyclooctadiene lignans (schisantherin A and B, schisandrin, schisandrin C, gomisin A, D, G, J, N, O, 6-O-benzoylgomisin O, schisandrin A, rubrisandrin A, epigomisin O, schisanhenol, rubriflorin A, angeloylgomisin H and O), aryltetralin lignan (wulignan A1), dibenzylbutane lignans (pregomisin, mesodihydroguaiaretic acid) and tetrahydrofuran lignan (fragransin A2). In addition, dihydrobenzofuran neolignans (licarin A and B) were also found in the analyzed extracts (Table 2).

Table 2.
The lignan contents (mg/100g DW) ± SD (n =5) in fruit, leaf and shoot extracts of Schisandra chinensis.
The total contents of detected lignans in fruit, leaf and shoot extracts were equal to: 1686.95, 433.59 and 313.83 mg/100 g DW, respectively. Qualitatively the dominant compounds in S. chinensis fruits were: gomisin N (259.05 mg/100 g DW), schisandrin A (212.50 mg/100 g DW), schisandrin (206.08 mg/100 g DW), gomisin D (195.22 mg/100 g DW), schisantherin B (195.82 mg/100 g DW), gomisin A (177.94 mg/100 g DW), angeloylgomisin H (161.90 mg/100 g DW) and gomisin J (142.35 mg/100 g DW). Fragrasin A2 was not detected in the fruit extract (Table 2).
The amounts of individual compounds were lower in leaf and shoot extracts than in fruit extracts. Rubriflorin A was detected only in trace amounts in the leaf extract. The dominant lignans in both leaf and shoot extracts were: Schisantherin B, gomisin A, gomisin N and angeloylgomisin H, and their quantities were equal to 102.47 and 35.27; 73.82 and 36.29; 55.06 and 62.69; and 47.34 and 44.84 mg/100 g DW, respectively (Table 2).
2.3. Anti-Inflammatory Activity
The following plant material extracts were tested for anti-inflammatory activity: Fruits and leaves of Schisandra rubriflora and Schisandra chinensis as well as selected most abundant lignans present in plant samples: 6-O-benzoylgomisin O, schisandrin, gomisin D, gomisin N and schisantherin A. Extracts from the shoots were not assayed for their anti-inflammatory activity, due to the relatively low lignan contents, determined in phytochemical studies, compared to leaf and fruit extracts (Table 1 and Table 2).
The tests were based on the in vitro inhibition of 15-lipooxygenase (15-LOX), phospholipase A2 (sPLA2), cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2) enzymes.
Plant material extracts showed moderate inhibition of 15-LOX and relatively high inhibitory activity against COX-1, COX-2 and sPLA2 (Table 3). Evaluation of 15-LOX inhibition showed that S. rubriflora fruit and leaf extracts moderately inhibited this enzyme: 22%—fruits, 38%—F leaves, 42%—M leaves at 17.5 μg/mL. For S. chinensis, the activity was lower: 25%—fruits (17.5 μg/mL) and 31%—leaves (175.0 μg/mL) (Table 3).

Table 3.
In vitro inhibition activity of studied S. rubriflora and S. chinensis extracts solutions against 15-LOX, COX-1, COX-2, and sPLA2.
The sPLA2 enzyme inhibition assay showed that fruit and leaf extracts of S. rubriflora inhibited its activity to about 62–65% at 175.0 μg/mL. Inhibition percentage for fruit and leaf extracts of S. chinensis was lower: 25% and 49%, respectively (at 175.0 μg/mL) (Table 3).
The most promising results were obtained for in vitro inhibitory COX-1 and COX-2 enzyme activities. S. rubriflora fruit extracts (at 17.4 μg/mL) inhibited COX-1 and COX-2 activities in 71% and 48%, respectively. Leaf extracts showed higher activity at 175.0 μg/mL, and the inhibition was 86% and 82% (F), and 96% and 90% (M), respectively (Table 3). S. chinensis extracts exhibited lower activity. The percentage of COX-1 and COX-2 inhibition was 59% and 66% for fruits, and 69% and 77% for leaves, respectively (Table 3).
Evaluation of anti-inflammatory properties of individual lignan solutions and the average sample of lignan composition (MIX) (Table S3) showed that they were not active against the sPLA2 enzyme (Table 4). All studied lignans, i.e., 6-O-benzoylgomisin O, schisandrin, gomisin D, gomisin N and schisantherin A, as well as their MIX sample, showed from 49% to 57% 15-LOX inhibitory activity at 0.175 μg/mL (Table 4). The highest inhibition for COX-1 was estimated for schisandrin—62% at 1.75 μg/mL, schisantherin A—74% at 0.175 μg/mL, and for the average sample of lignan composition—61% at 1.75 μg/mL (Table 4). The highest inhibition for COX-2 was detected for schisandrin—54% at 1.75 μg/mL, gomisin D—62% at 1.75 μg/mL, gomisin N—70% at 0.175 μg/mL and for the MIX sample—56% at 0.175 μg/mL (Table 4).

Table 4.
In vitro inhibition activity of selected pure lignan solutions against 15-LOX, COX-1, COX-2, and sPLA2.
3. Discussion
Twenty-four lignans were identified from four chemical lignan groups in all analyzed samples of both plant species: Dibenzocyclooctadiene lignans (schisantherin A and B, schisandrin, schisandrin C, gomisin A, D, G, J, N, O, 6-O-benzoylgomisin O, schisandrin A, rubrisandrin A, epigomisin O, schisanhenol, rubriflorin A, angeloylgomisin H and O), aryltetralin lignan (wulignan A1), dibenzylbutane lignans (pregomisin, mesodihydroguaiaretic acid), and tetrahydrofuran lignan (fragransin A2). In addition, the presence of dihydrobenzofuran neolignans (licarin A and B) was also found in the analyzed extracts. Until now, there have been no reports on the detection of so many lignan compounds in S. rubriflora fruit, leaf and shoot extracts, including the differentiation on male and female specimens (Table 1, Tables S1 and S2, Figure S1).
Schisanhenol was quantitatively predominant in the analyzed S. rubriflora fruit extracts (268.02 mg/100 g DW), and its content was: 1.45-, 2.27-, 2.57-, 2.59-, 3.48- and 4.04-fold higher, respectively, than the content of the remaining dominant compounds: Angeloylgomisin H, schisantherin B, schisandrin A, gomisin O, angeloylgomisin O and gomisin G (Table 1).
Twenty-four and twenty-three lignans were found in both leaf and shoot extracts of F and M S. rubriflora specimens, respectively. Fragransin A2 was found in the extracts from leaves and shoots of F specimens, while it was not detected in analogous extracts from M specimens. The total content of lignans in leaf extracts of F specimens was 1.30-fold lower compared to the content in leaf extracts of M specimens (Table 1). The following compounds were predominant in F specimen leaf extracts: schisantherin A and B, 6-O-benzoylgomisin O, and angeloylgomisin H. The contents of schisantherin A and B were: 2.12- and 2.80-fold higher, respectively, compared to their contents in leaf extracts of M. specimens. The contents of 6-O-benzoylgomisin O and angeloylgomisin H in leaf extracts of M specimens were: 4.20-, 1.28-fold higher, respectively, than in leaf extracts of F specimens (Table 1). The most dominant compounds in leaf extracts of M specimens were: 6-O-benzoylgomisin O, angeloylgomisin H, gomisin D, angeloylgomisin O, schisantherin A and B. Gomisin D and angeloylgomisin O contents in leaf extracts of M. specimens were: 7.08-, 2.84-fold higher, respectively, than in leaf extracts of F specimens (Table 1).
The total lignan content in shoot extracts of F specimens was 1.46-fold higher than in the extracts from M specimens (Table 1). The quantitatively dominant compounds in shoot extracts of F specimens were: Schisantherin B, angeloylgomisin H, schisantherin A, and 6-O-benzoylgomisin O: 1.41-, 1.42-, 3.48- and 1,39-fold higher, respectively, compared to shoot extracts of M specimens. Schisantherin B, angeloylgomisin H, and 6-O-benzoylgomisin O were predominant in the extracts of M specimens (Table 1).
This work presents for the first time such a wide determination of lignan contents in the extracts of S. rubriflora, taking into account the division into the material originating from female (F) and male (M) specimens (leaves and shoots and fruits). Studies on lignan composition of S. rubriflora were performed before only by Chinese teams [16,17,18,25,35]. Importantly, these studies were only qualitative, no quantitative data were found, and the authors did not distinguish extracts in terms of gender. In 2006, Chen et al. [18] isolated fruit extracts of S. rubriflora and detected following compounds by the 1H NMR method: schisanhenol, schisandrin, deoxyschisandrin, schisantherin B, angeloylgomisin P, tigloylgomisin P, gomisin M1, M2, O and J as well as specific rubrisandrins A and B. In 2010, Xiao et al. [17] identified the following lignans in fruit extracts based on the 1H and 2H-NMR spectroscopy: Gomisin G and O, angeloylgomisin P, wulignan A2, epiwulignan A1 and rubrisandrin C. Mu et al. [16] conducted in 2011 an isolation and structure elucidation of the following lignans from S. rubriflora fruit extracts using preparative HPLC and 13C-NMR: Schisandrin, schisandrin A and C, rubschisantherin, angeloygomisin Q, benzoylgomisin Q, gomisin J, Q, C, B, K, N, S, T, isogomisin O, wilsonilignangomisin G, marlignan L and G. Moreover, the latter authors detected for the first time two new lignans in the fruit extract, i.e., rubrilignans A and B. In comparison to those results, the current study identified eight additional new compounds in fruit extracts: Gomisin A, 6-O-benzoylgomisin O, angeloylgomisin H and O, pregomisin, mesodihydroguaiaretic acid and licarin A and B.
Extracts from aerial parts of S. rubriflora were studied by Li et al. [25] using 1H- and 13C-NMR and they found the following compounds: gomisin K, M1 and R, dimethylgomisin J, angeloylgomisin K3 and R, interiotherin B, schisantherin D, mesodihydroguaiaretic acid, dihydroguaiaretic acid and pregomisin. Li et al. [35] detected rubriflorin A and B in stem extracts. In the present study, additional nineteen compounds were detected both in shoot and leaf extracts: Schisantherin A and B, schisandrin, schisandrin C, gomisin A, D, G, J, N, O, 6-O-benzoylgomisin O, schisandrin A, rubrisandrin A, epigomisin O, schisanhenol, angeloylgomisin H and O, wulignan A1 and fragransin A2. Moreover, our study included quantitative estimation and division on female and male plants, which had not been done before (Table 1).
A comparative lignan profiling of extracts from fruits, leaves and shoots of S. chinensis using the UHPLC-MS MS method (Table 2 and Table 5) was carried out in the present study. The total content of the tested group of compounds in S. rubriflora fruit extracts was 1.60-fold lower than in S. chinensis fruit extracts. The total lignan contents in S. rubriflora fruit extracts of F specimen was: 1.97-, 2.55-fold higher, respectively, than in S. chinensis leaf extracts (Table 1). The total lignan content in extracts from F and M shoots of S. rubriflora was: 1.78- and 1.23-fold higher, respectively, than in S. chinensis shoot extracts (Table 1).

Table 5.
The comparison of the amounts (mg/100g DW) of the dominant lignans in the studied S. rubriflora and S. chinensis extracts.
Schisanhenol, schisantherin B, schisandrin A, gomisin O and angeloylgomisin H were predominant in S. rubriflora fruit extracts in terms of quantity (Table 3). The contents of schisanhenol, gomisin O and angeloylgomisin H in S. rubriflora fruit extracts was: 27.92- and 1.14-fold higher, respectively, than in S. chinensis fruit extracts (Table 1). The total content of schisantherin B and schisandrin A in S. rubriflora fruit extracts was: 1.57- and 2.04-fold lower, respectively, than in S. chinensis fruit extracts (Table 1).
Schisantherin A and schisantherin B, 6-O-benzoylgomisin O and angeloylgomisin H were among the quantitatively predominant compounds in S. rubriflora leaf extracts (Table 2). The quantities of these compounds in F and M leaf extracts of S. rubriflora were: 58.76- and 27.76-; 2.84- and 1.02-; 12.42- and 52.13-; and 2.13- and 2.73-fold higher, respectively than in S. chinensis leaf extracts (Table 5).
Similarly as in leaf extracts, schisantherin A and schisantherin B, 6-O-benzoylgomisin O and angeloylgomisin H were among the quantitatively predominant compounds in S. rubriflora shoot extracts (Table 2). The quantities of these compounds in F and M leaf extracts of S. rubriflora were: 38.00- and 10.93-; 6.78- and 9.68-; 6.98- and 52.13-; and 2.36- and 1.67-fold higher, respectively, than in S. chinensis shoot extracts (Table 5).
Different compounds were proved to be dominant in S. chinensis extracts. The quantities of schisandrin, gomisin D, gomisin J, gomisin A, schisandrin A, gomisin N and schisandrin C in S. chinensis fruit extracts were: 31.37-; 55.46-; 26.36-; 157.25-; 2.04-; 13.49- and 3.47-fold higher, respectively, than in S. rubriflora fruit extracts (Table 5). The dominant compounds in S. chinensis leaf and shoot extracts included schisandrin, gomisin A, gomisin N and schisandrin C. The amount of these lignans in leaf extracts, in comparison to their quantities in F and M leaf extracts, were: 3.99- and 5.71-; 11.53- and 17.58-; 32.95- and 24.91-; and 84.55- and 195.79-fold higher, respectively. Correspondingly, the quantities of these compounds in shoot extracts, compared to S. rubriflora F and M shoot extracts, were: 8.20- and 14.61-; 12.14- and 22.00-; 77.40- and 58.05; and 106.93- and 332.67-fold higher, respectively (Table 5).
The present study determined the complex in vitro anti-inflammatory activity of fruit and leaf extracts of S. rubriflora and S. chinensis. Moreover, the analyses were also performed on individual most abundant lignans (6-O-benzoylgomisin O, schisandrin, gomisin D, gomisin N and schisantherin A) and a synthetic average sample of lignan composition mixture (MIX) (representing mean concentrations of 16 most abundant lignans from fruit and leaf extracts of S. rubriflora and S. chinensis; see Table S3). In most cases, dose-dependent inhibition of selected enzymes by plant extracts or lignan solutions was observed. However, the most pronounced exceptions were found for 15-LOX inhibition (and partially COXs), especially by lignan solutions. An inverse dose-dependence in case of plant extracts can be contributed to a relatively wide confidence interval between dilutions (overlapping SDs), and thus the lack of significant difference. However, more interesting is an inverse dose-dependent inhibition of 15-LOX by lignan solutions. This is a clear deviation from the competitive inhibition mechanism. The observed dependencies allow us to suggest the occurrence of another mechanism, namely inhibitor acceleration of the enzyme by lignans. Known mechanisms of inhibitor acceleration rely on allostery and multiple active sites [36]. This assumption could be partially supported by the results for plant extracts, providing also some arguments for significant participation of lignans in their anti-inflammatory properties, albeit of low confidence (as mentioned earlier). This hypothesis should be confirmed in further studies. On the other hand, plant secondary metabolism is so rich and complicated that it is difficult to conclude that particular antioxidant properties, or anti-inflammatory in this case, are driven only by one compound group. Further, a parallel occurrence of components inhibiting as well as increasing the activity is possible due to the natural complexity of such a plant extract.
Previously, there were studies involving anti-inflammatory properties of certain schisandra lignans, but they were conducted on different models. Gomisin N, J and schisandrin C were proved to exert anti-inflammatory effect by reducing nitric oxide (NO) production from lipopolysaccharide-stimulated (LPS) RAW 264.7 cells [28]. Schisantherin A was shown to be an anti-inflammatory agent that down-regulated NF-κB and MAPK signaling pathways in LPS-treated RAW 264.7 cells [29]. Another study [30] demonstrated that schisandrin, deoxyschisandrin, schisandrin B and C and schisantherin A reduced LPS-induced NO production in RAW 264.7 cells. In addition, schisandrin was shown to exert a protective effect on LPS-induced sepsis [31]. In vitro studies performed by Guo et al. [31] showed that anti-inflammatory properties of schisandrin resulted from NO production inhibition, prostaglandin E2 (PGE2) release, COX-2 and inducible nitric oxide synthase (iNOS) expression, which in turn was caused by the inhibition of nuclear factor kappa B (NF-κB), c-Jun N-terminal kinase (JNK) and p38 mitogen-activated protein kinase (MAPK) activities in the RAW 264.7 macrophage cell line.
Moreover, extracts from S. chinensis fruits were tested for the anti-inflammatory activity by Huyke et al. [26]. Non-polar S. chinensis fruit extracts showed that the dose-dependent COX-2 inhibition (at 20 μg/mL) catalyzed prostaglandin production [26].
Lim et al. [27] conducted in vitro tests on such representative schisandra lignans as schisandrin, schisandrin A and C, gomisin B, C, G and N, as well as on methanolic extracts of S. chinensis fruits, for 5-lipoxygenase (5-LOX) inhibitory activity. The tested compounds inhibited 5-LOX-catalyzed leukotriene production by A23187-treated rat basophilic leukemia (RBL-1) cells at concentrations of 1–100 μM. Compounds, such as schisandrin and gomisins showed moderate inhibitory activity (IC50 < 10 μM) against 5-LOX-catalyzed leukotriene production, but they were significantly less active against COX-2-catalyzed PGE2 and inducible NO production [27].
We have also proved in our study the high anti-inflammatory potential of S. chinensis fruit extracts against COX-1 and COX-2 enzyme activities (Table 3). Positive results were also obtained for leaf extracts (Table 3). Moreover, the inhibitory activity against 15-LOX and sPLA2 enzymes has been demonstrated for the first time. To the best of our knowledge, the extracts from fruits and leaves of S. rubriflora have not yet been studied for anti-inflammatory activity. The obtained results from S. rubriflora plant materials showed a higher activity in comparison to S. chinensis (Table 3).
We have demonstrated, based on the results of individual lignan analyses, that 6-O-benzoylgomisin O, schisandrin, gomisin D, gomisin N and schisantherin A display significant 15-LOX, COX-1 and COX-2 inhibitory activities, and that they are virtually inactive against sPLA2 (Table 4). Our study also analyzed for the first the anti-inflammatory activity of 6-O-benzoylgomisin O and gomisin D.
4. Materials and Methods
4.1. Plant Material
Plant material was obtained as part of cooperation with Clematis—Źródło Dobrych Pnączy Spółka z o.o. spółka jawna with a registered office in Pruszków (address: ul. Duchnicka 27, 05-800 Pruszków, Poland) [37]. Plant species were identified by dr. eng. Szczepan Marczyński (owner of the Clematis arboretum). For the purpose of comparative phytochemical analysis, fruits and leaves and shoots (stems with leaves) of about 10 years old female (F) (100 individuals) and male (M) (50 individuals) S. rubriflora (Franch.) Rehd. et Wils specimens, and about 10 years old monoecious specimens of S. chinensis Turcz. Baill (100 individuals) were collected and dried. Leaves and shoots were harvested in May, fruits in September 2017. The fruits were lyophilized and the leaves and shoots were air-dried (about 25–30 °C). Dry plant material was pulverized in a mixing ball mill (MM 400, Retch, Haan, Germany).
4.2. Plant Sample Extraction
Methanol extracts were prepared from fruits, shoots and leaves of F and M S. rubriflora plants. The samples (0.3 g, 5 replicates) were extracted with 3 mL of methanol (grade-HPLC, Merck, Darmstadt, Germany). The extraction process was carried out twice in an ultrasonic bath (Sonic 2, POLSONIC Palczyński Sp.J., rsaw, Poland) for 20 min. The obtained extracts were centrifuged for 5 min (4000 rpm) in a centrifuge (Centrifuge MPW–223E, MPW Med. Instruments, Warsaw, Poland). The centrifuged extracts were filtered using sterilizing syringe filters (Millex®GP, 0.22 μm, Filter Unit, Millipore, Bedford, MA, USA).
4.3. UHPLC–MS/MS Lignan Targeted Profiling
Lignan-targeted profiling was carried out in methanolic extracts of S. rubriflora and S. chinensis by means of ultra-high performance liquid chromatography coupled to a tandem mass spectrometer (UHPLC-MS/MS). An external standard addition method was used. Filtered plant extracts were aliquoted in two 45 µL portions. To the first, 5 µL of methanol was added, while to the second 5 µL of the standard lignan solution (all monitored compounds). Samples were analyzed on a UHPLC Infinity 1260 (Agilent, Wolbrom, Germany) coupled to a quadrupole tandem mass spectrometer 6410 QQQ LC/MS (Agilent, Santa Clara, CA, USA). Samples were separated on an analytical column (Kinetex C18 150 × 4.6 mm, 2.7 µm) in a gradient mode of 50% methanol in water (A) versus 100% methanol (B) with 0.1% of formic acid. A linear gradient was applied, 20% to 65% of B in 22 min at 0.5 mL/min at 60 °C; the injection volume was 2 µL. Standard lignan substances were purchased from ChemFaces Biochemical Co. Ltd. (Wuhan, China). The studied lignans and their structures and synonymous names are listed in Table S1. Lignans were analyzed in the MRM mode after ESI ionization (Table S2).
In addition to lignans, whose standards were commercially available (Tables S1 and S2, Figure S1), compounds from the dibenzocyclooctadiene lignan group (angeloylgomisins H and O) were also identified based on the UHPLC-MS/MS result analysis for the tested extracts. The identification was based on analyzing fragmentation ions of these compounds visible in mass spectra. The quantitative analysis of angeloylgomisyn H and O was based on their content conversion, according to schisandrin standard curve (UHPLC-MS/MS)—the main compound from the dibenzocyclooctadiene lignan group; according to pharmacopoeial requirements, the raw material should be standardized based on the content of this compound [3].
4.4. Anti-Inflammatory Activity
Plant material methanolic extracts of fruits and leaves of S. rubriflora (F and M) and S. chinensis were tested for anti-inflammatory activity. Additionally, the following most abundant lignans present in the plant samples were analyzed for anti-inflammatory activity: 6-O-benzoylgomisin O, schisandrin, gomisin D, gomisin N and schisantherin A (No: L10, L1, L16, L14, L6; Tables S1, S2 and Figure S1, respectively); in addition, the mixture of lignans, representing the average plant sample composition (MIX) (mean concentrations of 16 most abundant lignans of fruit and leaf extracts of S. rubriflora and S. chinensis; see Table S3), underwent analogous analysis.
The plant extracts (concentrations: 175.0 and 17.5 μg/mL, Table 3) and solutions of selected lignans (concentrations: 1.75 and 0.175 μg/mL, Table 4) were serially diluted in methanol. The tests were based on in vitro inhibition of 15-lipooxygenase (15-LOX), phospholipases A2 (sPLA2), cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2) enzymes.
4.4.1. Inhibitory Activity against 15-Lipooxygenase (15-LOX)
Samples were tested for their inhibitory activity against 15-LOX using an assay kit (760700, Cayman Chem. Co., Ann Arbor, MI, USA), according to the manufacturer’s instructions; arachidonic acid at 0.91 mM was the substrate; nordihydroguaiaretic acid (NDGA) at 100 µM served as a positive control inhibitor. The kit measures the concentration of hydroperoxides produced in the lipooxygenation reaction using purified soy 15-lipooxygenase standard at pH 7.4 in 10 mM Tris-HCl buffer. The reagent’s colorimetric composition is vendor proprietary. The measurements were carried out in 96-well plate using a Synergy II reader (Biotek, Winooski, VT, USA) at 490 nm. The end-point absorbance was recorded after 5-min incubation of enzyme and inhibitor followed by 15-min incubation after substrate addition and 5-min incubation after chromogen addition.
4.4.2. Inhibitory Activity against Cyclooxygenase-1 and Cyclooxygenase-2 (COX-1 and COX-2)
Samples were tested for their ability to inhibit COX-1 and COX-2 using the COX-1 (ovine) and COX-2 (human) inhibitor assay kit (701050, Cayman Chem. Co.), according to the manufacturer’s instructions; arachidonic acid at 1.1 mM was the substrate; ibuprofen at 10 µM served as a positive control inhibitor. The kit measures the peroxidase component of COXs. The appearance of oxidized N,N,N’,N’-tetramethyl-p-phenylenediamine (TMPD) was monitored kinetically for 5 min in a 96-well plate format at 590 nm using a Synergy II reader.
4.4.3. Inhibitory Activity against Phospholipases A2 (sPLA2)
Inhibition of sPLA2 activity was tested using an assay kit (10004883, Cayman Chem. Co.), according to the manufacturer’s instructions; diheptanoyl thio-PC at 1.44 mM was the substrate; thioetheramide-PC at 100 µM served as a positive control inhibitor. Human recombinant Type V sPLA2 was used. Free thiols released by cleavage of the diheptanoyl thio-PC ester bond were measured kinetically using DTNB (5-5’-dithio-bis-(2-nitrobenzoic acid), Ellman’s reagent) in a 96-well plate format at 420 nm using a Synergy II reader. The percent of inhibition was calculated according to Equation (1):
where: %Inh—percent of inhibition; IA—100% enzyme activity (without inhibitor); Inhibitor—enzyme activity with inhibitor added.
%Inh = [(IA−Inhibitor)\IA] × 100
All samples were assayed in triplicate, including 100% enzyme activity, positive control inhibitor and tested extracts and lignan solutions.
4.4.4. Statistical Analysis
Quantitative results are expressed in mg/100 g DW (dry weight) as the mean ± SD (standard deviation) of three or five samples (n = 3, n = 5) in the experiments that were repeated three times.
5. Conclusions
The present study is the first comparative, complex, qualitative and quantitative analyses of S. rubriflora and S. chinensis lignan composition derived from different groups. The contents of shoot and fruit extracts of both plant species were determined for the first time using the UHPLC-MS/MS method. The study identified and characterized twenty-four lignans representing four chemical groups: dibenzocyclooctadiene lignans (schisantherin A and B, schisandrin, schisandrin C, gomisin A, D, G, J, N, O, 6-O-benzoylgomisin O, schisandrin A, rubrisandrin A, epigomisin O, schisanhenol, rubriflorin A, angeloylgomisin H and O), aryltetralin lignan (wulignan A1), dibenzylbutane lignans (pregomisin, mesodihydroguaiaretic acid), tetrahydrofuran lignan (fragransin A2) and dihydrobenzofuran neolignans (licarin A and B). Qualitative and quantitative differences in lignan composition were recorded depending on the origin of samples (fruit, leaf and shoot) as well as plant species.
Additionally, the current work determined for the first time the anti-inflammatory activity, based on the in vitro inhibition of 15-lipooxygenase (15-LOX), phospholipases A2 (sPLA2), cyclooxygenases 1 and 2 (COX-1; COX-2) enzymes, of fruit and leaf extracts of the analyzed species as well as individual lignans: 6-O-benzoylgomisin O, schisandrin, gomisin D, gomisin N and schisantherin A; furthermore, a mixture of lignans representing an average plant sample composition was also tested. The results revealed a high competitiveness of S. rubriflora in relation to known, pharmacopoeial plant species—S. chinensis.
Based on our research, we suggest to consider the extracts of S. rubriflora (fruit, leaf and shoot), as a rich, valuable source of lignans with a promising anti-inflammatory potential. The objects of interest exhibited very interesting differences and showed new research directions involving these compounds, e.g., phenolic composition and other biological activities of S. rubriflora would be worth investigating.
Supplementary Materials
The following are available online, Figure S1: Exemplary UHPLC–MS/MS MRM chromatogram of lignan standard mixture at 100 ug/mL. Table S1: The standard lignan substances used in the performer studies. Table S2: The monitored fragmentation reactions (multiple reactions monitoring, MRM) for studied lignans. Table S3: Quantitative composition of “average sample lignan composition” (MIX) at 1.75 μg/mL.
Author Contributions
Conceptualization, A.S.; Methodology, A.S. and M.D.; Investigation, A.S., M.D., A.W., P.K. and M.K.-S.; Data Curation, A.S., M.D. and A.W.; Writing-Original Draft Preparation, A.S. and M.D.; Writing-Review & Editing, A.S., M.D., M.K.-S., H.E.; Project Administration, A.S.; Funding Acquisition, A.S.
Funding
This research was funded by National Science Centre, Poland, rant number: 2016/23/D/NZ7/01316.
Conflicts of Interest
All authors declare that they have no conflict of interest.
References
- Saunders, R.M.K. Monograph of Schisandra (Schisandraceae). In Systematic Botany Monographs; American Society of Plant Taxonomists: Laramie, WY, USA, 2000; Volume 58, pp. 1–146. ISBN 978-0912861586. [Google Scholar]
- Szopa, A.; Barnaś, M.; Ekiert, H. Phytochemical studies and biological activity of three Chinese Schisandra species (Schisandra sphenanthera, Schisandra henryi and Schisandra rubriflora): Current findings and future applications. Phytochem. Rev. 2018, 1–20. [Google Scholar] [CrossRef]
- European Directorate for the Quality of Medicines. Schisandrae chinensis fructus. In European Pharmacopoeia 9.0.; Council of Europe: Strasbourg Cedex, France, 2017. [Google Scholar]
- Szopa, A.; Ekiert, R.; Ekiert, H. Current knowledge of Schisandra chinensis (Turcz.) Baill. (Chinese magnolia vine) as a medicinal plant species: A review on the bioactive components, pharmacological properties, analytical and biotechnological studies. Phytochem. Rev. 2017, 16, 195–218. [Google Scholar] [CrossRef] [PubMed]
- Szopa, A.; Klimek, M.; Ekiert, H. Chinese magnolia vine (Schisandra chinensis)—therapeutic and cosmetic importance. Polish J. Cosmetol. 2016, 19, 274–284. [Google Scholar]
- European Directorate for the Quality of Medicines. Schisandrae chinensis fructus. In European Pharmacopoeia 6.0.; Council of Europe: Strasbourg Cedex, France, 2008. [Google Scholar]
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China; China Chemical Industry Press: Beijing, China, 2005. [Google Scholar]
- Committee of the Japanese Pharmacopoeia. Evaluation and Licensing Division Pharmaceuticals and Food Safety. In Japanese Pharmacopoeia; Bureau Ministry of Health, Labour and Welfare: Tokyo, Japan, 2006. [Google Scholar]
- Central Pharmaceutical Affairs Council of Korea. Korean Pharmacopoeia; Central Pharmaceutical Affairs Council of Korea: Seoul, Korea, 2002.
- Xu, L.; Grandi, N.; Del Vecchio, C.; Mandas, D.; Corona, A.; Piano, D.; Esposito, F.; Parolin, C.; Tramontano, E. From the traditional Chinese medicine plant Schisandra chinensis new scaffolds effective on HIV-1 reverse transcriptase resistant to non-nucleoside inhibitors. J. Microbiol. 2015, 53, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Upton, R.; Graff, A.; Jolliffe, G.; Länger, R.; Williamson, E. American Herbal Pharmacopoeia: Botanical Pharmacognosy—Microscopic Characterization of Botanical Medicines; CRC Press: Boca Raton, FL, USA, 2011; ISBN 1420073281. [Google Scholar]
- World Health Organization. Fructus Schisandrae. WHO Monographs on Selected Medicinal Plants; WHO: Geneva, Switzerland, 2007; Volume 3. [Google Scholar]
- Panossian, A.; Wikman, G. Pharmacology of Schisandra chinensis Bail.: An overview of Russian research and uses in medicine. J. Ethnopharmacol. 2008, 118, 183–212. [Google Scholar] [CrossRef]
- Hancke, J.L.; Burgos, R.A.; Ahumada, F. Schisandra chinensis (Turcz.) Baill. Fitoterapia 1999, 70, 451–471. [Google Scholar] [CrossRef]
- Li, G.; Zhao, J.; Tu, Y.; Yang, X.; Zhang, H.; Li, L. Chemical constituents of Schisandra rubriflora Rehd. et Wils. J. Integr. Plant Biol. 2005, 47, 362–367. [Google Scholar] [CrossRef]
- Mu, H.X.; Li, X.S.; Fan, P.; Yang, G.Y.; Pu, J.X.; Sun, H.D.; Hu, Q.F.; Xiao, W.L. Dibenzocyclooctadiene lignans from the fruits of Schisandra rubriflora and their anti-HIV-1 activities. J. Asian Nat. Prod. Res. 2011, 13, 393–399. [Google Scholar] [CrossRef]
- Xiao, W.L.; Wang, R.R.; Zhao, W.; Tian, R.R.; Shang, S.Z.; Yang, L.M.; Yang, J.H.; Pu, J.X.; Zheng, Y.T.; Sun, H.D. Anti-HIV-1 activity of lignans from the fruits of Schisandra rubriflora. Arch Pharm. Res. 2010, 33, 697–701. [Google Scholar] [CrossRef]
- Chen, M.; Kilgore, N.; Lee, K.H.; Chen, D.F. Rubrisandrins A and B, lignans and related anti-HIV compounds from Schisandra rubriflora. J. Nat. Prod. 2006, 69, 1697–1701. [Google Scholar] [CrossRef]
- Xiao, W.L.; Li, X.; Wang, R.R.; Yang, L.M.; Li, M.; Huang, S.X.; Pu, J.X.; Zheng, Y.T.; Li, R.T.; Sun, H.D. Triterpenoids from Schisandra rubriflora. J. Nat. Prod. 2007, 70, 1056–1059. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Liu, G.T. Anti-oxidant activity of dibenzocyclooctene lignans isolated from Schisandraceae. Planta Med. 1992, 58, 311–313. [Google Scholar] [CrossRef] [PubMed]
- Opletal, L.; Sovová, H.; Bártlová, M. Dibenzo [a,c] cyclooctadiene lignans of the genus Schisandra: Importance, isolation and determination. J. Chromatogr. B 2004, 812, 357–371. [Google Scholar] [CrossRef]
- Chang, J.; Reiner, J.; Xie, J. Progress on the chemistry of dibenzocyclooctadiene lignans. Chem. Rev. 2005, 105, 4581–4609. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.; Han, N.; Yao, X.; Liu, Z.; Wang, Y.; Yang, J.; Yin, J. Structure-activity relationship study of dibenzocyclooctadiene lignans isolated from Schisandra chinensis on lipopolysaccharide-induced microglia activation. Planta Med. 2014, 80, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Xu, X.; Xu, B.; Yang, P.; Liao, Z.; Morris-Natschke, S.L.; Lee, K.H.; Chen, D. Neglschisandrins E-F: Two new lignans and related cytotoxic lignans from Schisandra neglecta. Molecules 2013, 18, 2297–2306. [Google Scholar] [CrossRef] [PubMed]
- Li, H.M.; Luo, Y.M.; Pu, J.X.; Li, X.N.; Lei, C.; Wang, R.R.; Zheng, Y.T.; Sun, H.D.; Li, R.T. Four new dibenzocyclooctadiene lignans from Schisandra rubriflora. Helv. Chim. Acta 2008, 91, 1053–1062. [Google Scholar] [CrossRef]
- Huyke, C.; Engel, K.; Simon-Haarhaus, B.; Quirin, K.-W.; Schempp, C. Composition and Biological Activity of Different Extracts from Schisandra sphenanthera and Schisandra chinensis. Planta Med. 2007, 73, 1116–1126. [Google Scholar] [CrossRef]
- Lim, H.; Son, K.H.; Bae, K.H.; Hung, T.M.; Kim, Y.S.; Kim, H.P. 5-Lipoxygenase-inhibitory constituents from Schisandra fructus and Magnolia flos. Phyther. Res. 2009, 23, 1489–1492. [Google Scholar] [CrossRef]
- Oh, S.-Y.; Kim, Y.H.; Bae, D.S.; Um, B.H.; Pan, C.-H.; Kim, C.Y.; Lee, H.J.; Lee, J.K. Anti-Inflammatory Effects of Gomisin N, Gomisin J, and Schisandrin C Isolated from the Fruit of Schisandra chinensis. Biosci. Biotechnol. Biochem. 2010, 74, 285–291. [Google Scholar] [CrossRef]
- Ci, X.; Ren, R.; Xu, K.; Li, H.; Yu, Q.; Song, Y.; Wang, D.; Li, R.; Deng, X. Schisantherin A exhibits anti-inflammatory properties by down-regulating NF-κB and MAPK signaling pathways in lipopolysaccharide-treated RAW 264.7 cells. Inflammation 2010, 33, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Zhao, X.; Li, Z.; Wang, L.; Wang, Y. Study on main pharmacodynamic effects for Schisandra lignans based upon network pharmacology. Chin. J. Chin. Mater. Med. 2015, 40, 522–527. [Google Scholar]
- Guo, L.Y.; Hung, T.M.; Bae, K.H.; Shin, E.M.; Zhou, H.Y.; Hong, Y.N.; Kang, S.S.; Kim, H.P.; Kim, Y.S. Anti-inflammatory effects of schisandrin isolated from the fruit of Schisandra chinensis Baill. Eur. J. Pharmacol. 2008, 591, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Whiting, D.A. Lignans and neolignans. Nat. Prod. Rep. 1985, 193, 191–211. [Google Scholar] [CrossRef]
- Whiting, D.A. Lignans, neolignans, and related compounds. Nat. Prod. Rep. 1990, 7, 349–364. [Google Scholar] [CrossRef]
- Gottlieb, O.R. Chemosystematics of the Lauraceae. Phytochemistry 1972, 11, 1537–1570. [Google Scholar] [CrossRef]
- Li, L.; Ren, H.Y.; Yang, X.D.; Zhao, J.F.; Li, G.P.; Zhang, H. Bin Rubriflorin A and B, two novel partially saturated dibenzocyclooctene lignans from Schisandra rubriflora. Helv. Chim. Acta 2004, 87, 2943–2947. [Google Scholar] [CrossRef]
- Wales, M.E.; Madison, L.L.; Glaser, S.S.; Wild, J.R. Divergent allosteric patterns verify the regulatory paradigm for aspartate transcarbamylase. J. Mol. Biol. 1999, 294, 1387–1400. [Google Scholar] [CrossRef]
- Clematis–Źródło Dobrych Pnączy Spółka z o.o. spółka jawna. Available online: http://www.clematis.com.pl/pl/ (accessed on 3 October 2018).
Sample Availability: Samples of the compounds (all tested lignans) are available from the authors. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).