Glycosylation of Methoxylated Flavonoids in the Cultures of Isaria fumosorosea KCH J2
Abstract
1. Introduction
2. Results and Discussion
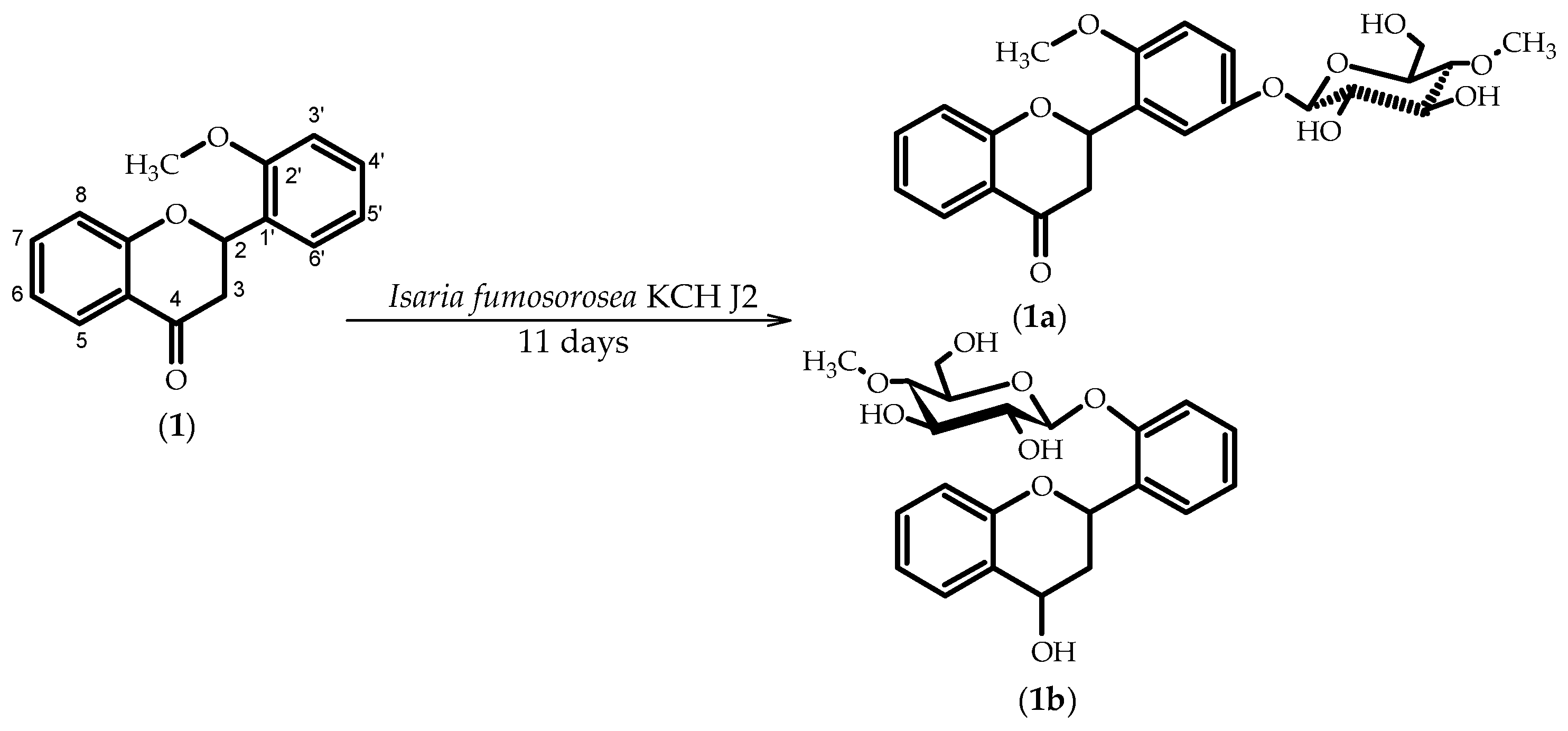
2.1. Biotransformations of 2′-Methoxyflavanone (1)
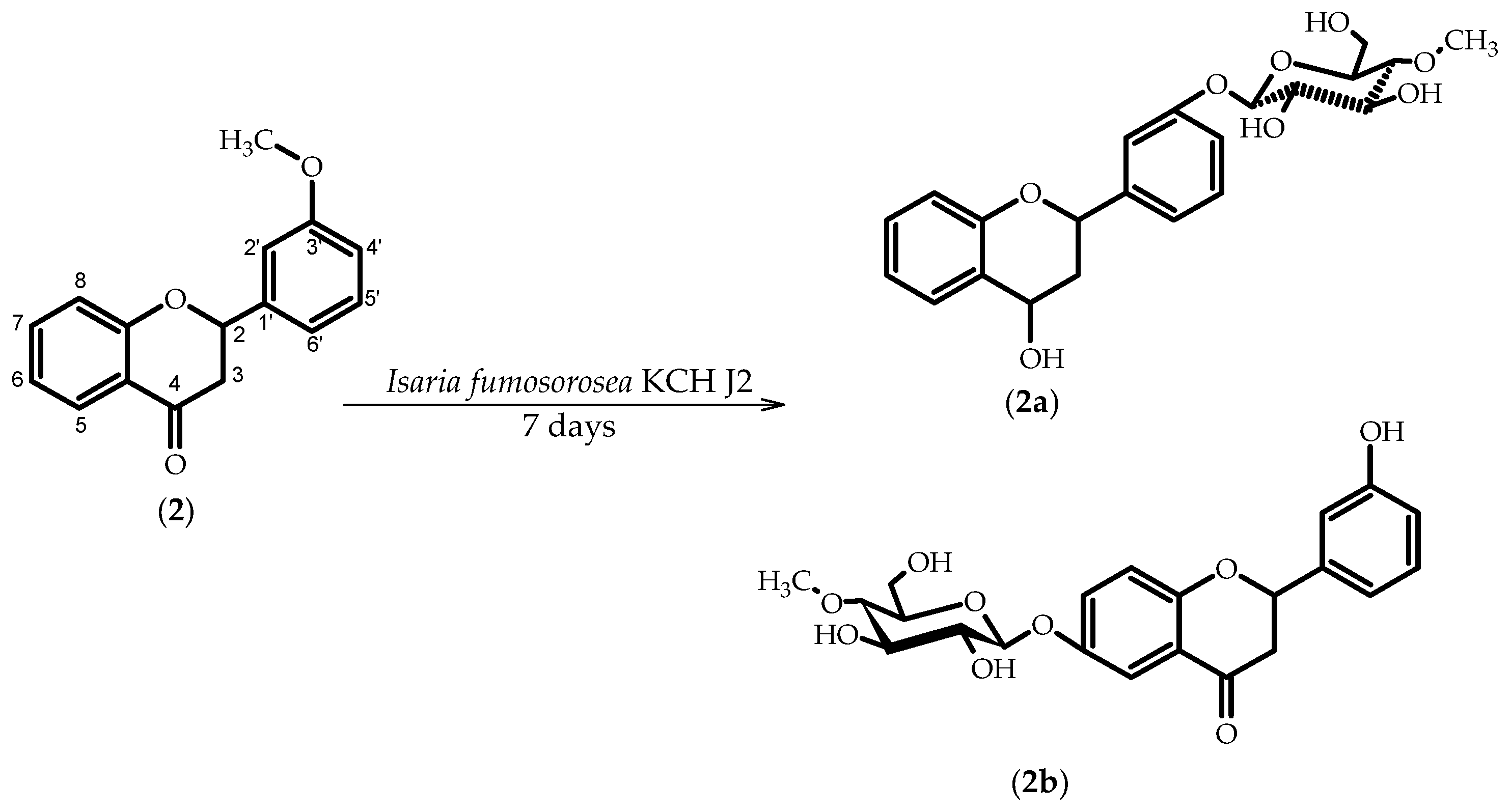
2.2. Biotransformations of 3′-Methoxyflavanone (2)
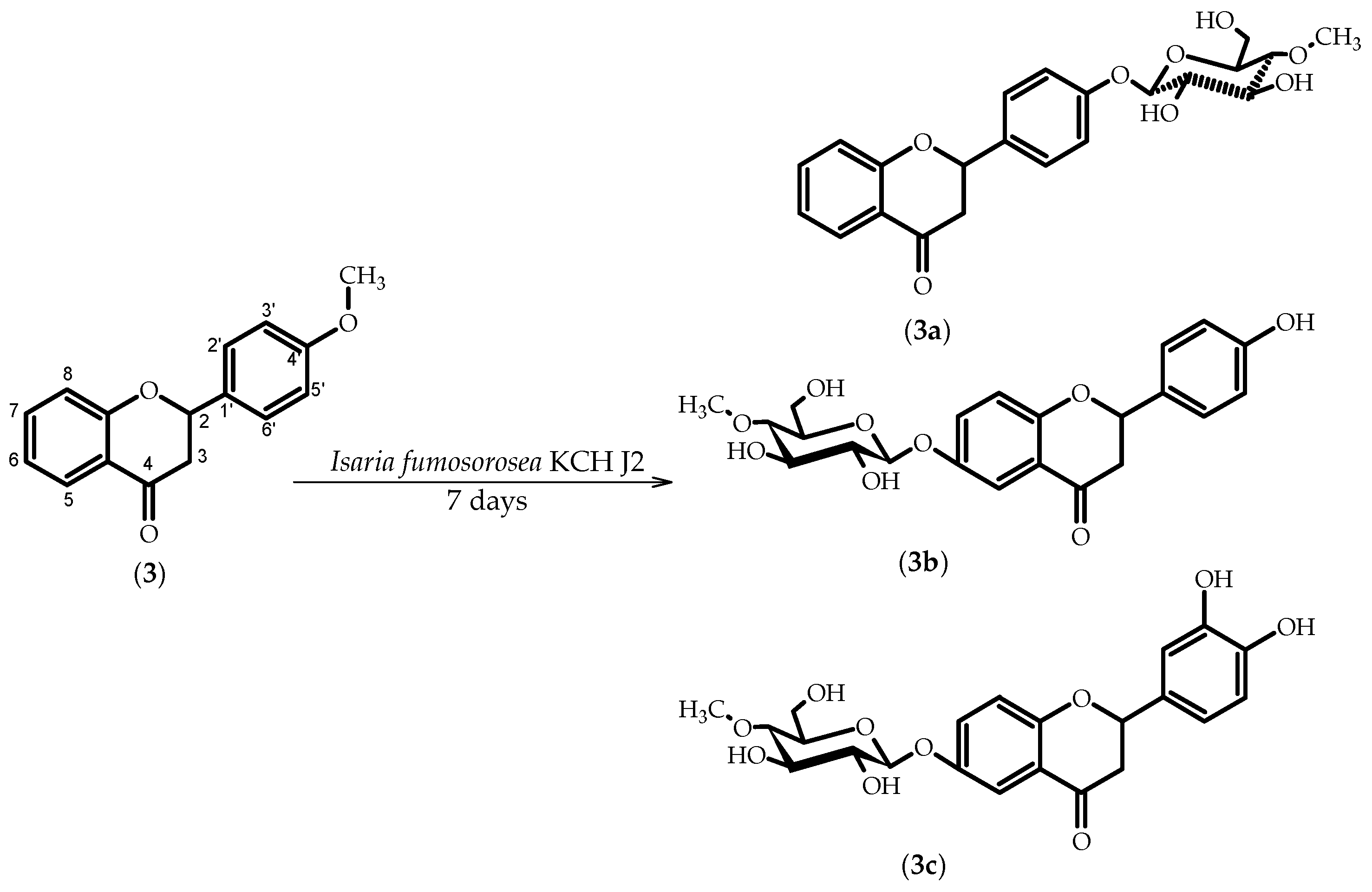
2.3. Biotransformations of 4′-Methoxyflavanone (3)
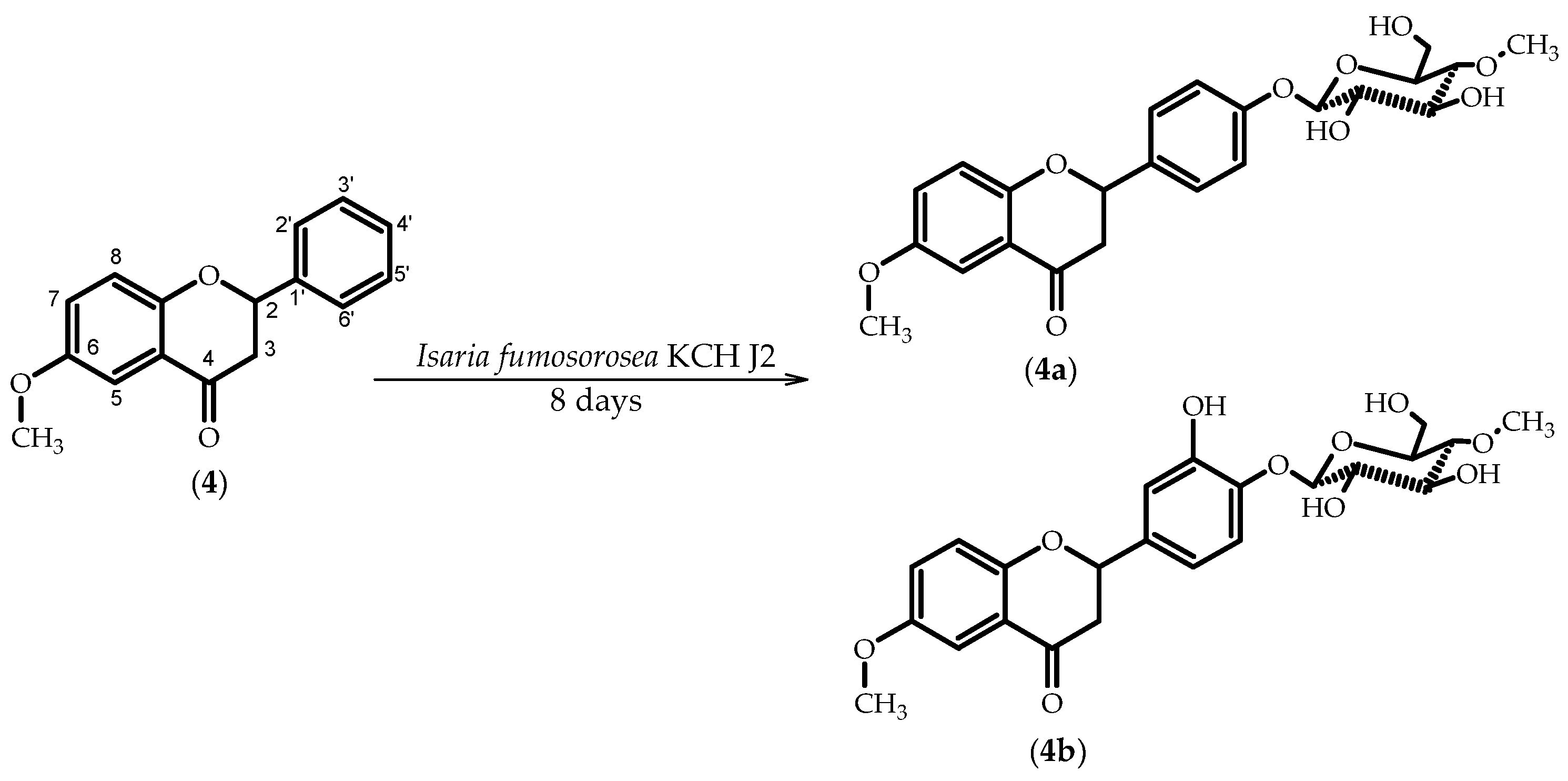
2.4. Biotransformations of 6-Methoxyflavanone (4)
2.5. Biotransformations of 6-Methoxyflavone (5)
3. Materials and Methods
3.1. Chemicals
3.1.1. 2′-Methoxyflavanone (1)
3.1.2. 3′-Methoxyflavanone (2)
3.1.3. 4′-Methoxyflavanone (3)
3.1.4. 6-Methoxyflavanone (4)
3.1.5. 6-Methoxyflavone (5)
3.1.6. Chemical Synthesis of 2′-Methoxyflavanone (1), 3′-Methoxyflavanone (2), 4′-Methoxyflavanone (3)
3.2. Microorganism
3.3. Analysis
3.4. Screening Procedure
3.5. Scale-Up Biotransformations
3.5.1. 2′-Methoxyflavanone 5′-O-β-d-(4″-O-methyl)-glucopyranoside (1a)
3.5.2. Flavan-4-ol 2′-O-β-d-(4″-O-methyl)-glucopyranoside (1b)
3.5.3. Flavan-4-ol 3′-O-β-d-(4″-O-methyl)-glucopyranoside (2a)
3.5.4. 3′-Hydroxyflavanone 6-O-β-d-(4″-O-methyl)-glucopyranoside (2b)
3.5.5. Flavanone 4′-O-β-d-(4″-O-methyl)-glucopyranoside (3a)
3.5.6. 4′-Hydroxyflavanone 6-O-β-d-(4″-O-methyl)-glucopyranoside (3b)
3.5.7. 3′,4′-Dihydroxyflavanone 6-O-β-d-(4″-O-methyl)-glucopyranoside (3c)
3.5.8. 6-Methoxyflavanone 4′-O-β-d-(4″-O-methyl)-glucopyranoside (4a)
3.5.9. 3′-Hydroxy-6-methoxyflavanone 4′-O-β-d-(4″-O-methyl)-glucopyranoside (4b)
3.5.10. 6-Methoxyflavone 3′-O-β-d-(4″-O-methyl)-glucopyranoside (5a)
3.5.11. 6-Methoxyflavone 4′-O-β-d-(4″-O-methyl)-glucopyranoside (5b)
3.5.12. 3′-Hydroxy-6-methoxyflavone 4′-O-β-d-(4″-O-methyl)-glucopyranoside (5c)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cassidy, A. Berry anthocyanin intake and cardiovascular health. Mol. Aspects Med. 2018, 61, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Zhang, J.; Chen, X. Bioactive flavonoids in Moringa oleifera and their health-promoting properties. J. Funct. Foods 2018, 47, 469–479. [Google Scholar] [CrossRef]
- Deng, J.; Yang, H.; Capanoglu, E.; Cao, H.; Xiao, J. Technological Aspects and Stability of Polyphenols; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Wang, W.; Sun, C.; Mao, L.; Ma, P.; Liu, F.; Yang, J.; Gao, Y. The biological activities, chemical stability, metabolism and delivery systems of quercetin: A review. Trends Food Sci. Technol. 2016, 56, 21–38. [Google Scholar] [CrossRef]
- Aklaghi, M.; Ghobadi, S.; Mohammad Hosseini, M.; Gholami, Z.; Mohammadian, F. Flavanols are potential anti-obesity agents, a systematic review and meta-analysis of controlled clinical trials. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 675–690. [Google Scholar] [CrossRef] [PubMed]
- Belščak-Cvitanović, A.; Durgo, K.; Huđek, A.; Bačun-Družina, V.; Komes, D. Overview of Polyphenols and Their Properties; Galanakis, C., Ed.; Woodhead Publishing: Sawston, Cambridge, UK, 2018. [Google Scholar]
- Perez-Vizcaino, F.; Fraga, C.G. Research trends in flavonoids and health. Arch. Biochem. Biophys. 2018, 646, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.-Y.; Li, Q.; Bi, K.-S. Bioactive flavonoids in medicinal plants: Structure, activity and biological fate. Asian J. Pharm. Sci. 2018, 13, 12–23. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013, 162750. [Google Scholar] [CrossRef] [PubMed]
- Treml, J.; Smejkal, K. Flavonoids as Potent Scavengers of Hydroxyl Radicals. Compr. Rev. Food Sci. Food Saf. 2016, 15, 720–738. [Google Scholar] [CrossRef]
- Symonowicz, M.; Kolanek, M. Flavonoids and their properties to form chelate complexes. Biotechnol. Food Sci. 2012, 76, 35–41. [Google Scholar]
- Gao, Z.; Gao, W.; Zeng, S.; Li, P.; Liu, E. Chemical structures, bioactivities and molecular mechanisms of citrus polymethoxy flavones. J. Funct. Foods 2018, 40, 498–509. [Google Scholar] [CrossRef]
- Hung, W.; Chang, W.; Lu, W.; Wei, G.; Wang, Y.; Ho, C.-T.; Hwang, L.S. Pharmacokinetics, bioavailability, tissue distribution and excretion of tangeretin in rat. J. Food Drug Anal. 2018, 26, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Akbar, S.; Subhan, F.; Karim, N.; Shahid, M.; Ahmad, N.; Ali, G.; Mahmood, W.; Fawad, K. 6-Methoxyflavanone attenuates mechanical allodynia and vulvodynia in the streptozotocin-induced diabetic neuropathic pain. Biomed. Pharmacother. 2016, 84, 962–971. [Google Scholar] [CrossRef] [PubMed]
- Akbar, S.; Subhan, F.; Karim, N.; Aman, U.; Ullah, S.; Shahid, M.; Ahmad, N.; Fawad, K.; Sewell, R.D.E. Characterization of 6-methoxyflavanone as a novel anxiolytic agent: A behavioral and pharmacokinetic approach. Eur. J. Pharmacol. 2017, 801, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Thilakarathna, S.H.; Vasantha Rupasinghe, H.P. Flavonoid bioavailability and attempts for bioavailability enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Liu, H.; Yang, J.; Jiang, Y. New insights on bioactivities and biosynthesis of flavonoid glycosides. Trends Food Sci. Technol. 2018, 79, 116–124. [Google Scholar] [CrossRef]
- Mojzer, E.B.; Hrncic, M.K.; Škerget, M.; Knez, Ž.; Bren, U. Polyphenols: Extraction methods, antioxidative action, bioavailability and anticarcinogenic effects. Molecules 2016, 21, 901. [Google Scholar] [CrossRef] [PubMed]
- Archivio, M.D.; Filesi, C.; Varì, R.; Scazzocchio, B. Bioavailability of the polyphenols: Status and controversies. Int. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar] [CrossRef] [PubMed]
- Dymarska, M.; Grzeszczuk, J.; Urbaniak, M.; Janeczko, T.; Pląskowska, E.; Stępień, Ł.; Kostrzewa-Susłow, E. Glycosylation of 6-methylflavone by the strain Isaria fumosorosea KCH J2. PLoS ONE 2017, 12, e0184885. [Google Scholar] [CrossRef] [PubMed]
- Dymarska, M.; Janeczko, T.; Kostrzewa-Susłow, E. Biotransformations of flavones and an isoflavone (daidzein) in cultures of entomopathogenic filamentous fungi. Molecules 2018, 23, 1358. [Google Scholar] [CrossRef] [PubMed]
- Dymarska, M.; Janeczko, T.; Kostrzewa-Susłow, E. Glycosylation of 3-hydroxyflavone, 3-methoxyflavone, quercetin and baicalein in fungal cultures of the genus Isaria. Molecules 2018, 23, 2477. [Google Scholar] [CrossRef] [PubMed]
- Kostrzewa-Susłow, E.; Dymarska, M.; Białońska, A.; Janeczko, T. Enantioselective conversion of certain derivatives of 6-hydroxyflavanone. J. Mol. Catal. B Enzym. 2014, 102, 59–65. [Google Scholar] [CrossRef]
- Kostrzewa-Susłow, E.; Dmochowska-Gładysz, J.; Janeczko, T.; Środa, K.; Michalak, K.; Palko, A. Microbial transformations of 6- and 7-methoxyflavones in Aspergillus niger and Penicillium chermesinum cultures. Zeitschrift fur Naturforsch. Sect. C J. Biosci. 2012, 67C, 411–417. [Google Scholar] [CrossRef]
- Cao, H.; Chen, X.; Jassbi, A.R.; Xiao, J. Microbial biotransformation of bioactive flavonoids. Biotechnol. Adv. 2015, 33, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Janeczko, T.; Dymarska, M.; Siepka, M.; Gniłka, R.; Leśniak, A.; Popłoński, J.; Kostrzewa-Susłow, E. Enantioselective reduction of flavanone and oxidation of cis- and trans-flavan-4-ol by selected yeast cultures. J. Mol. Catal. B Enzym. 2014, 109, 47–52. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1, 1a, 1b, 2, 2a, 2b, 3, 3a, 3b, 3c, 4, 4a, 4b, 5, 5a, 5b, 5c are available from the authors. |

| Proton | Compound | |||||
|---|---|---|---|---|---|---|
| 1 | 1a | 1b | 2 | 2a | 2b | |
| H-2 | 5.89 (dd) J2,3a = 13.3, J2,3e = 2.8 | 5.85 (dd) J2,3a = 13.0, J2,3e = 2.8 | 5.84 (d) J = 11.4 | 5.65 (dd) J2,3a = 13.0, J2,3e = 2.9 | 5.31 (dd) J2,3a = 11.7, J2,3e = 2.1 | 5.57 (dd) J2,3a = 12.7, J2,3e = 3.0 |
| H-3a | 3.05 (dd) J3a,2 = 13.3, J3a,3e = 16.7 | 3.01 (m) | 2.49 (d) J = 14.0 | 3.19 (dd) J3a,2 = 13.1, J3a,3e = 16.8 | 2.26 (m) | 3.12 (dd) J3a,2 = 12.7, J3a,3e = 16.8 |
| H-3e | 2.88 (m) | 2.88 (m) | 2.01 (m) | 2.90 (dd) J3e,2 = 2.9, J3e,3a = 16.8 | 2.15 (m) | 2.87 (m) |
| H-4 | - | - | 4.83 (m) | - | 4.81 (m) | - |
| H-5 | 7.90 (dd) J5,6 = 8.2, J5,7 = 1.8 | 7.89 (dd) J5,6 = 7.8, J5,7 = 1.6 | 7.60 (d) J5,6 = 7.0 | 7.88 (dd) J5,6 = 8.0, J5,7 = 1.8 | 7.39 (dd) J5,6 = 7.6, J5,7 = 1.6 | 7.49 (t) J = 3.1 |
| H-6 | 7.12 (m) | 7.13 (m) | 7.19 (t) J = 7.4 | 7.13 (m) | 6.96(dt) J = 7.4, J = 1.1 | - |
| H-7 | 7.62 (m) | 7.63 (m) | 7.33 (m) | 7.63 (ddd) J7,5 = 1.7, J7,6 = 7.2, J7,8 = 8.3 | 7.25 (m) | 7.37 (dd) J7,5 = 3.1, J7,8 = 9.0 |
| H-8 | 7.12 (m) | 7.13 (m) | 7.33 (m) | 7.13 (m) | 6.91 (dd) J8,6 = 0.9, J8,7 = 8.1 | 7.07 (m) |
| H-2′ | 7.22 (m) | 7.23 (m) | 7.07 (m) | |||
| H-3′ | 7.12 (m) | 7.03 (d) J3′,4′ = 8.9 | 6.92 (d) J3′,4′ = 8.2 | - | - | - |
| H-4′ | 7.41 (m) | 7.11 (dd) J4′,3′ = 9.1, J4′,6′ = 2.9 | 4.25 (m) | 7.00 (ddd) J4′,2′ = 0.8, J4′,5′ = 8.3, J4′,6′ = 2.6 | 7.08 (ddd) J4′,2′ = 0.7, J4′,5′ = 8.5, J4′,6′ = 2.6 | 6.89 (ddd) J4′,2′ = 0.8, J4′,5′ = 8.1, J4′,6′ = 2.3 |
| H-5′ | 7.12 (m) | - | 6.96 (t) J = 7.4 | 7.40 (m) | 7.36 (t) J = 8.0 | 7.29 (t) J = 7.9 |
| H-6′ | 7.68 (dd) J6′,4′ = 1.5, J6′,5′ = 7.6 | 7.41 (d) J6′,4′ = 2.8 | 7.42 (d) J = 6.5 | 7.19 (m) | 7.17 (d) J6′,5′ = 7.6 | 7.07 (m) |
| H-1″ | - | 4.90 (d) J = 7.8 | 4.85 (d) J = 7.8 | - | 5.03 (d) J = 7.8 | 4.93 (d) J = 7.8 |
| H-2″ | - | 3.52 (m) | 3.53 (m) | - | 3.51 (m) | 3.49 (m) |
| H-3″ | - | 3.65 (m) | 3.63 (t) J = 8.5 | - | 3.66 (m) | 3.67 (dt) J = 9.0, J = 3.7 |
| H-4″ | - | 3.23 (m) | 3.28 (t) J = 9.3 | - | 3.26 (m) | 3.27 (t) J = 9.3 |
| H-5″ | - | 3.48 (m) | 3.42 (ddd) J = 9.4, J = 4.0, J = 2.0 | - | 3.51 (m) | 3.49 (m) |
| H-6″ | - | 3.72 (m) 3.85 (m) | 3.84 (dd) J = 5.2, J = 2.4 3.74 (dd) J = 11.0, J = 3.8 | - | 3.85 (m) 3.72 (m) | 3.87 (m) 3.76 (m) |
| C-4″-OCH3 | - | 3.59 (s) | 3.59 (s) | - | 3.60 (s) | 3.60 (s) |
| C-2′-OCH3 | 3.92 (s) | 3.89 (s) | - | - | - | - |
| C-3′-OCH3 | - | - | - | 3.88 (s) | - | - |
| C-3′-OH | - | - | - | - | - | 8.54 (s) |
| Carbon | Compound | |||||
|---|---|---|---|---|---|---|
| 1 | 1a | 1b | 2 | 2a | 2b | |
| C-2 | 75.5 | 75.0 | 68.9 | 80.3 | 73.8 | 80.3 |
| C-3 | 44.1 | 44.0 | 38.4 | 45.0 | 39.6 | 44.9 |
| C-4 | 192.1 | 192.1 | 64.3 | 191.8 | 63.6 | 191.7 |
| C-4a | 121.9 | 121.9 | 133.2 | 122.0 | 126.1 | 122.1 |
| C-5 | 127.4 | 127.4 | 127.2 | 127.4 | 131.5 | 113.7 |
| C-6 | 122.2 | 122.3 | 124.1 | 122.3 | 121.1 | 153.1 |
| C-7 | 136.8 | 136.8 | 129.5 | 136.8 | 129.9 | 126.8 |
| C-8 | 118.9 | 119.0 | 118.5 | 118.9 | 117.5 | 118.2 |
| C-8a | 162.8 | 162.7 | 156.1 | 162.4 | 155.6 | 157.8 |
| C-1′ | 128.4 | 129.2 | 125.7 | 141.9 | 144.2 | 141.9 |
| C-2′ | 157.0 | 152.0 | 155.4 | 112.9 | 115.4 | 114.2 |
| C-3′ | 111.8 | 112.7 | 117.6 | 160.9 | 158.9 | 158.5 |
| C-4′ | 130.4 | 117.8 | 130.0 | 114.7 | 116.5 | 116.2 |
| C-5′ | 121.6 | 152.8 | 121.1 | 130.6 | 130.3 | 130.6 |
| C-6′ | 127.4 | 116.3 | 131.7 | 119.3 | 120.7 | 119.9 |
| C-1″ | - | 102.4 | 104.3 | - | 101.5 | 102.6 |
| C-2″ | - | 75.3 | 75.1 | - | 75.0 | 75.0 |
| C-3″ | - | 78.0 | 77.6 | - | 78.0 | 77.8 |
| C-4″ | - | 80.2 | 79.8 | - | 80.1 | 79.9 |
| C-5″ | - | 77.0 | 76.9 | - | 77.0 | 77.0 |
| C-6″ | - | 62.2 | 61.8 | - | 62.1 | 62.0 |
| C-4″-OCH3 | - | 60.5 | 60.5 | - | 60.5 | 60.5 |
| C-2′-OCH3 | 56.0 | 56.4 | - | - | - | - |
| C-3′-OCH3 | - | - | - | 55.6 | - | - |
| Proton | Compound | |||
|---|---|---|---|---|
| 3 | 3a | 3b | 3c | |
| H-2 | 5.60 (dd) J2,3a = 13.1, J2,3e = 2.8 | 5.63 (dd) J2,3a = 13.0, J2,3e = 2.8 | 5.51 (ddd) J2,3a = 13.1, J2,3e = 2.8 J = 4.8 | 5.45 (ddd) J2,3a = 12.9, J2,3e = 2.9 J = 5.2 |
| H-3a | 3.22 (dd) J3a,2 = 13.1, J3a,3e = 16.7 | 3.21 (m) | 3.17 (ddd) J3a,2 = 13.2, J3a,3e = 16.8, J = 0.9 | 3.13 (ddd) J3a,2 = 12.9, J3a,3e = 16.8, J = 1.3 |
| H-3e | 2.85 (dd) J3e,2 = 2.9, J3e,3a = 16.8 | 2.86 (m) | 2.81 (dt) J = 16.8, J = 3.0 | 2.81 (m) |
| H-5 | 7.88 (dd) J5,6 = 7.7, J5,7 = 1.8 | 7.88 (dd) J5,6 = 7.8, J5,7 = 1.7 | 5.49 (t) J = 3.0 | 7.49 (t) J = 3.0 |
| H-6 | 7.12 (m) | 7.11 (m) | - | - |
| H-7 | 7.61 (m) | 7.61 (m) | 7.35 (dd) J7,5 = 3.1, J7,8 = 9.0 | 7.35 (dd) J7,5 = 3.1, J7,8 = 9.0 |
| H-8 | 7.09 (d) J8,7 = 8.7 | 7.11 (m) | 7.03 (dd) J8,7 = 8.9, J = 1.4 | 7.02 (dd) J8,7 = 9.0, J = 1.4 |
| H-2′ | 7.56 (m) | 7.55 (m) | 6.44 (m) | 7.08 (t) J = 1.9 |
| H-3′ | 7.04 (m) | 7.16 (m) | 6.93 (m) | - |
| H-5′ | 7.04 (m) | 7.16 (m) | 6.93 (m) | 6.91 (m) |
| H-6′ | 7.56 (m) | 7.55 (m) | 6.44 (m) | 6.91 (m) |
| H-1″ | - | 5.02 (dd) J = 7.8, J = 1.9 | 4.93 (d) J = 7.7 | 4.92 (d) J = 6.3 |
| H-2″ | - | 3.52 (m) | 3.49 (m) | 3.49 (m) |
| H-3″ | - | 3.67 (dt) J = 9.1, J = 3.9 | 3.67 (m) | 3.67 (t) J = 9.0 |
| H-4″ | - | 3.25 (m) | 3.27 (t) J = 9.3 | 3.27 (t) J = 9.3 |
| H-5″ | - | 3.52 (m) | 3.49 (m) | 3.49 (m) |
| H-6″ | - | 3.87 (m) 3.74 (m) | 3.87 (m) 3.75 (m) | 3.87 (d) J = 11.4 3.75 (dd) J = 10.9, J = 5.5 |
| C-4″-OCH3 | - | 3.60 (s) | 3.60 (s) | 3.60 (s) |
| C-4′-OCH3 | 3.87 (s) | - | - | - |
| C-4′-OH | - | - | 8.57 (s) | - |
| Carbon | Compound | |||
|---|---|---|---|---|
| 3 | 3a | 3b | 3c | |
| C-2 | 80.2 | 80.1 | 80.4 | 80.4 |
| C-3 | 44.8 | 44.8 | 44.8 | 44.8 |
| C-4 | 192.1 | 192.0 | 192.1 | 192.1 |
| C-4a | 121.9 | 121.9 | 122.0 | 122.0 |
| C-5 | 127.4 | 127.4 | 113.7 | 113.7 |
| C-6 | 122.1 | 122.2 | 153.0 | 153.0 |
| C-7 | 136.8 | 136.8 | 126.8 | 126.8 |
| C-8 | 118.9 | 118.9 | 119.9 | 119.9 |
| C-8a | 162.5 | 162.5 | 158.0 | 157.9 |
| C-1′ | 132.2 | 133.8 | 131.1 | 131.9 |
| C-2′ | 128.9 | 128.7 | 129.0 | 114.7 |
| C-3′ | 114.8 | 117.4 | 116.2 | 146.0 |
| C-4′ | 160.9 | 158.9 | 158.6 | 146.3 |
| C-5′ | 114.8 | 117.4 | 116.2 | 119.2 |
| C-6′ | 128.9 | 128.7 | 129.0 | 116.0 |
| C-1″ | - | 101.6 | 102.7 | 102.6 |
| C-2″ | - | 74.0 | 75.0 | 75.0 |
| C-3″ | - | 78.0 | 77.9 | 77.8 |
| C-4″ | - | 80.1 | 79.9 | 79.9 |
| C-5″ | - | 77.1 | 77.0 | 76.9 |
| C-6″ | - | 62.1 | 62.0 | 62.0 |
| C-4″-OCH3 | - | 60.6 | 60.5 | 60.5 |
| C-4′-OCH3 | 55.6 | - | - | - |
| Proton | Compound | ||||||
|---|---|---|---|---|---|---|---|
| 4 | 4a | 4b | 5 | 5a | 5b | 5c | |
| H-2 | 5.62 (dd) J2,3a = 13.1, J2,3e = 2.9 | 7.56 (dd) J2,3a = 13.0, J2,3e = 2.8 | 5.52 (dd) J2,3a = 12.8, J2,3e = 2.9 | - | - | - | - |
| H-3a | 3.15 (dd) J3a,2 = 13.1, J3a,3e = 16.8 | 3.17 (dd) J3a,2 = 13.0, J3a,3e = 16.8 | 3.14 (m) | - | - | - | - |
| H-3e | 2.88 (m) | 2.87 (m) | 2.85 (m) | ||||
| H-3 (flavone) | - | - | - | 6.88 (s) | 6.88 (s) | 6.80 (s) | 6.79 (s) |
| H-5 | 7.33 (d) J5,7 = 3.2 | 7.32 (d) J5,7 = 3.1 | 7.31 (m) | 7.54 (d) J5,7 = 3.1 | 7.54 (m) | 7.54 (d) J5,7 = 3.1 | 7.54 (d) J5,7 = 3.1 |
| H-7 | 7.23 (dd) J7,5 = 3.2, J7,8 = 9.0 | 7.22 (dd) J7,5 = 3.2, J7,8 = 9.0 | 7.21 (m) | 7.43 (dd) J7,5 = 3.1, J7,8 = 9.1 | 7.43 (dd) J7,5 = 3.2, J7,8 = 9.1 | 7.41 (dd) J7,5 = 3.2, J7,8 = 9.1 | 7.42 (dd) J7,5 = 3.2, J7,8 = 9.1 |
| H-8 | 7.08 (d) J8,7 = 9.0 | 7.05 (d) J8,7 = 9.0 | 7.05 (m) | 7.73 (d) J8,7 = 9.1 | 7.78 (d) J8,7 = 9.1 | 7.72 (d) J8,7 = 9.1 | 7.74 (d) J8,7 = 9.1 |
| H-2′ | 7.62 (d) J2′,3′ = 7.4 | 7.54 (d) J2′,3′ = 8.6 | 7.14 (d) J2′,5′ = 2.2 | 8.13 (m) | 7.81 (m) | 8.08 (m) | 7.61 (m) |
| H-3′ | 7.49 (t) J = 7.5 | 7.15 (d) J3′,2′ = 8.6 | - | 7.64 (m) | - | 7.28 (m) | - |
| H-4′ | 7.43 (m) | - | - | 7.64 (m) | 7.31 (m) | - | - |
| H-5′ | 7.49 (t) J = 7.5 | 7.15 (d) J5′,6′ = 8.6 | 7.00 (dd) J5′,2′ = 2.1, J5′,6′ = 8.4 | 7.64 (m) | 7.54 (m) | 7.28 (m) | 7.37 (d) J5′,6′ = 8.5 |
| H-6′ | 7.62 (d) J6′,5′ = 7.4 | 7.54 (d) J6′,5′ = 8.6 | 7.24 (d) J6′,5′ = 8.3 | 8.13 (m) | 7.77 (m) | 8.08 (m) | 7.58 (dd) J6′,2′ = 2.3, J6′,5′ = 8.5 |
| H-1″ | - | 5.01 (d) J = 7.8 | 4.81 (d) J = 7.9 | - | 5.14 (d) J = 7.8 | 5.14 (d) J = 7.8 | 5.00 (d) J = 7.8 |
| H-2″ | - | 3.51 (m) | 3.53 (m) | - | 3.57 (m) | 3.55 (m) | 3.57 (m) |
| H-3″ | - | 3.67 (t) J = 8.9 | 3.67 (t) J = 9.0 | - | 3.71 (m) | 3.69 (m) | 3.72 (m) |
| H-4″ | - | 3.26 (t) J = 9.3 | 3.26 (m) | - | 3.25 (dd) J = 9.5, J = 9.1 | 3.28 (m) | 3.29 (m) |
| H-5″ | - | 3.53 (m) | 3.50 (m) | - | 3.65 (ddd) J = 9.7, J = 5.5, J = 1.9 | 3.59 (m) | 3.57 (m) |
| H-6″ | - | 3.86 (m) 3.72 (dd) J = 12.0, J = 4.8 | 3.88 (m) 3.73 (m) | - | 3.94 (m) 3.76 (m) | 3.90 (m) 3.75 (m) | 3.92 (m) 3.77 (m) |
| C-4″-OCH3 | - | 3.60 (s) | 3.60 (s) | - | 3.62 (s) | 3.61 (s) | 3.61 (s) |
| C-6-OCH3 | 3.86 (s) | 3.85 (s) | 3.85 (s) | 3.96 (s) | 3.97 (s) | 3.96 (s) | 3.96 (s) |
| Carbon | Compound | ||||||
|---|---|---|---|---|---|---|---|
| 4 | 4a | 4b | 5 | 5a | 5b | 5c | |
| C-2 | 80.4 | 80.1 | 80.1 | 163.6 | 163.2 | 163.5 | 163.4 |
| C-3 | 45.0 | 44.8 | 44.9 | 107.2 | 107.4 | 106.1 | 106.4 |
| C-4 | 191.8 | 192.0 | 191.9 | 177.7 | 177.7 | 177.6 | 177.6 |
| C-4a | 121.9 | 121.9 | 121.9 | 125.5 | 125.4 | 125.5 | 125.5 |
| C-5 | 108.3 | 108.3 | 108.3 | 105.8 | 105.7 | 105.8 | 105.8 |
| C-6 | 155.2 | 155.1 | 155.1 | 158.0 | 158.1 | 158.0 | 158.0 |
| C-7 | 125.3 | 125.2 | 125.2 | 124.0 | 124.0 | 123.8 | 123.8 |
| C-8 | 120.2 | 120.2 | 120.2 | 120.8 | 120.8 | 120.7 | 120.7 |
| C-8a | 156.9 | 157.0 | 156.9 | 151.8 | 151.8 | 151.7 | 151.7 |
| C-1′ | 140.5 | 133.9 | 136.3 | 132.9 | 134.2 | 126.3 | 128.0 |
| C-2′ | 127.3 | 128.7 | 115.0 | 127.1 | 115.0 | 128.7 | 114.5 |
| C-3′ | 129.5 | 117.4 | 148.8 | 130.0 | 159.3 | 117.6 | 148.8 |
| C-4′ | 129.3 | 158.9 | 146.2 | 132.4 | 120.6 | 161.3 | 148.9 |
| C-5′ | 129.5 | 117.4 | 118.6 | 130.0 | 131.0 | 117.6 | 118.3 |
| C-6′ | 127.3 | 128.7 | 119.4 | 127.1 | 120.9 | 128.7 | 119.2 |
| C-1″ | - | 101.6 | 104.3 | - | 101.7 | 101.2 | 103.2 |
| C-2″ | - | 74.8 | 74.8 | - | 75.0 | 74.9 | 74.9 |
| C-3″ | - | 77.9 | 78.7 | - | 78.1 | 78.0 | 77.4 |
| C-4″ | - | 80.1 | 80.0 | - | 80.3 | 80.1 | 80.0 |
| C-5″ | - | 77.1 | 77.3 | - | 77.3 | 77.2 | 77.4 |
| C-6″ | - | 62.0 | 61.9 | - | 62.2 | 62.1 | 62.0 |
| C-4″-OCH3 | - | 60.6 | 60.6 | - | 60.6 | 60.6 | 60.6 |
| C-6-OCH3 | 56.0 | 56.1 | 56.0 | 56.2 | 56.2 | 56.2 | 56.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dymarska, M.; Janeczko, T.; Kostrzewa-Susłow, E. Glycosylation of Methoxylated Flavonoids in the Cultures of Isaria fumosorosea KCH J2. Molecules 2018, 23, 2578. https://doi.org/10.3390/molecules23102578
Dymarska M, Janeczko T, Kostrzewa-Susłow E. Glycosylation of Methoxylated Flavonoids in the Cultures of Isaria fumosorosea KCH J2. Molecules. 2018; 23(10):2578. https://doi.org/10.3390/molecules23102578
Chicago/Turabian StyleDymarska, Monika, Tomasz Janeczko, and Edyta Kostrzewa-Susłow. 2018. "Glycosylation of Methoxylated Flavonoids in the Cultures of Isaria fumosorosea KCH J2" Molecules 23, no. 10: 2578. https://doi.org/10.3390/molecules23102578
APA StyleDymarska, M., Janeczko, T., & Kostrzewa-Susłow, E. (2018). Glycosylation of Methoxylated Flavonoids in the Cultures of Isaria fumosorosea KCH J2. Molecules, 23(10), 2578. https://doi.org/10.3390/molecules23102578





