Wasabia koreana Nakai: A Preliminary Study on Nutrients and Chemical Compounds That May Impact Sensory Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Proximate Composition
2.3. Constituents and Free amino Acid Composition
2.4. Fatty Acid Composition
2.5. Mineral Composition
2.6. Vitamin C Content
2.7. Total Phenolic Compounds
2.8. Antioxidant Capacity
2.9. Sinigrin and AITC Content
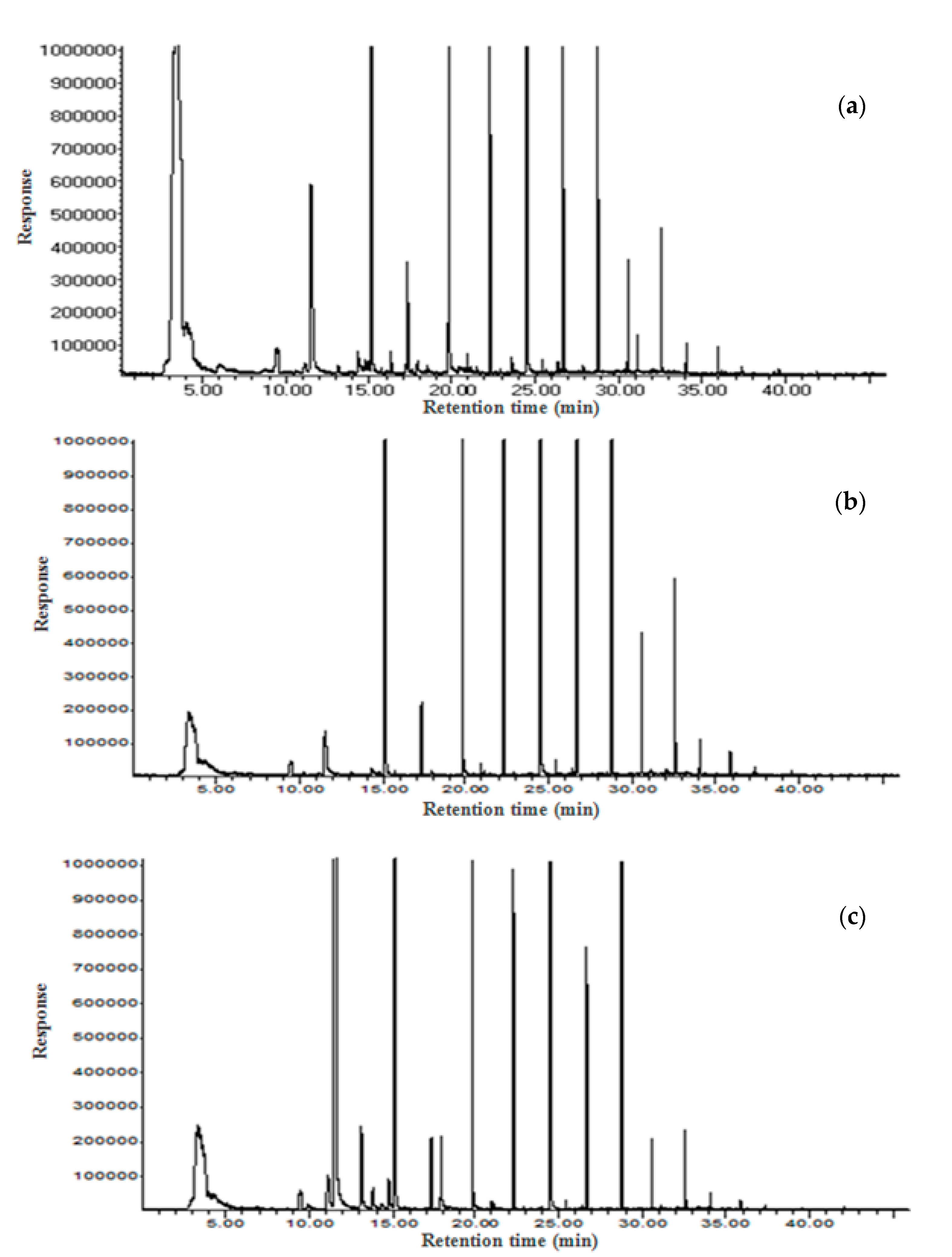
2.10. Volatile Compounds and Sniffing Test
2.11. Electronic Nose
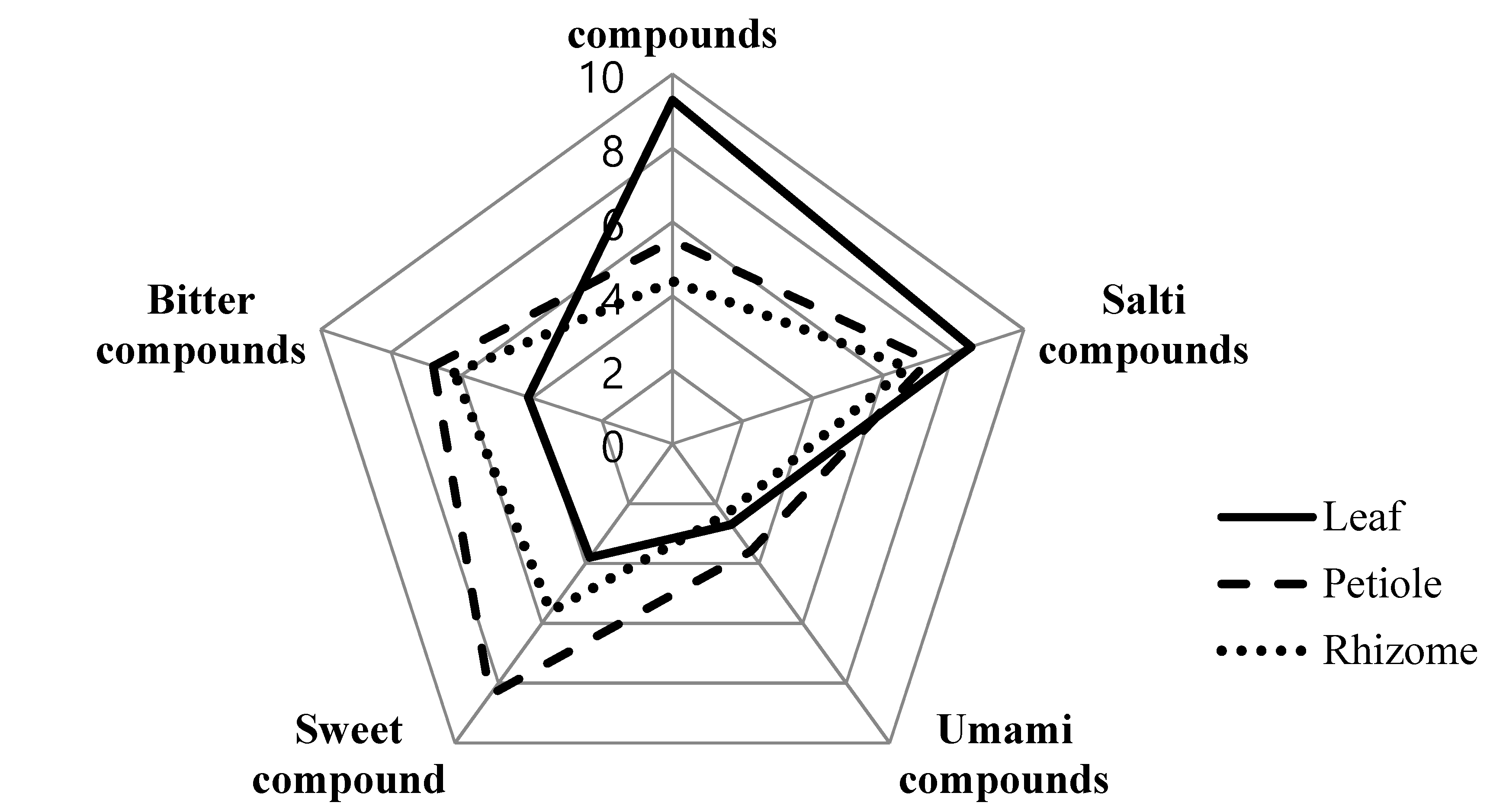
2.12. Electronic Tongue
2.13. Statistical Analysis
3. Results and Discussion
3.1. Proximate Composition
3.2. Constituents and Free Amino Acid Compositions
3.3. Fatty Acid Composition
3.4. Mineral Composition
3.5. Vitamin C, Total Phenolic Contents and Antioxidant Capacity
3.6. Sinigrin and AITC Contents
3.7. Volatile Compounds and Sniffing Test
3.8. Electronic Nose (E-Nose)
3.9. Electronic Tongue
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shin, E.H. Quality characteristics of vinegar using Wasabi japonica Matsum leaves. Korean J. Food Cook. Sci. 2015, 21, 255–263. [Google Scholar]
- Park, K.I.; Yoon, M.J.; Kwon, S.W.; Choi, S.Y.; Lee, K.S. Effects of colchicine and EMS on induction of variants Wasabia japonica Matsu. Korean J. Med. Crop Sci. 1996, 4, 12–18. [Google Scholar]
- Chin, H.W.; Lindsay, R.C. Volatile sulfur compounds formed in disrupted tissues of different cabbage cultivars. J. Food Sci. 1993, 58, 835–839. [Google Scholar] [CrossRef]
- Amelia, S.; Lubis, N.D.A.; Siregar, M.A.; Rozi, M.F. Antimicrobial effect of wasabia japonica or wasabi on raw salmon in served sashimi at Japanese restaurants in Medan. Int. J. Med. Sci. Clin. Interv. 2018, 5, 3944–3947. [Google Scholar]
- Shuichi, M.; Hideki, M.; Yuko, S.; Chitose, S.; Fumiyo, T. Improvement effects of wasabi (Wasabia japonica) leaves and allylisothiocyanate on stomach lesions of Mongolian gerbils infected with helicobacter pylori. Nat. Prod. Commun. 2017, 12, 595–598. [Google Scholar]
- Subedi, L.; Venkatesan, R.; Kim, S.Y. Neuroprotective and anti-inflammatory activities of allyl isothiocyanate through attenuation of JNK/NF-B/TNF-Signaling. Int. J. Mol. Sci. 2017, 18, 1423. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, I.; Miyamoto, T.; Kleinwachter, M.; Selmar, D.; Hara, M. Wasabi (wasabia japonica Matsum.) rhizome possesses potent amylolytic activity. Food Sci. Technol. Res. 2013, 19, 133–138. [Google Scholar] [CrossRef]
- Romeo, L.; Iori, R.; Rollin, P.; Bramanti, P.; Mazzon, E. Isothiocyanates: An overview of their antimicrobial activity against human infections. Molecules 2018, 23, 624. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S. Anti-hypercholesterolemic effects of Wasabia japonica in high cholesterol-fed SD strain rats. J. Life Sci. 2008, 18, 747–751. [Google Scholar] [CrossRef]
- Sultana, T.; McNeil, D.L.; Porter, N.G.; Savage, G.P. Investigation of isothiocyanate yield from flowering and non-flowering tissues of wasabi grown in a flooded system. J. Food Compost. Anal. 2003, 16, 637–646. [Google Scholar] [CrossRef]
- Dai, R.; Lim, L.T. Release of allyl isothiocyanate from mustard seed meal powder entrapped in electrospun PLA-PEO. J. Food Sci. 2015, 77, 467–475. [Google Scholar] [CrossRef]
- Park, Y.Y.; Cho, M.S.; Park, S.; Lee, Y.D.; Jeong, B.R.; Chung, J.B. Sinigrin contents in different tissues of wasabi and antimicrobial activity of their water extracts. Korean J. Hort. Sci. Technol. 2006, 24, 480–487. [Google Scholar]
- Sung, E.H.; Shin, S.M.; Kang, Y.H. Physicochemical quality characteristics and antioxidant activity of Wasabi (Wasabia japonica) leaf and petiole extracts. J. Korean Soc. Food Sci. Nutr. 2017, 46, 335–342. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis, 16th ed.; Method 932.06, 925.09, and 923.03; Association of Official Analytical Communities: Arlington, VA, USA, 1995. [Google Scholar]
- Kim, D.S.; Kim, H.S.; Hong, S.J.; Cho, J.J.; Shin, E.C. Changes in physicochemical and antioxidative properties of Cynanchi wilfordii Radix after a roasting treatment. J. Korean Soc. Food Sci. Nutr. 2018, 47, 363–372. [Google Scholar] [CrossRef]
- Kim, D.S.; Kim, H.S.; Lee, K.T.; Hong, D.L.; Cho, S.R.; Pan, J.H.; Park, Y.B.; Lee, Y.B.; Kim, J.K.; Shin, E.C. Chemical characterization and oxidative stability of medium- and long-chain fatty acid profiles in tree-borne seed oils. J. Anal. Methods Chem. 2018, 2018, 2178684. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.J.; Kim, J.K.; Cho, K.M.; Joo, O.S.; Nam, S.H.; Lee, S.W.; Kim, H.J.; Shin, E.C. Analysis of nutritional components, volatile properties, and sensory attributes of Cynanchi wilfordii Radix: Characterization study. J. Korean Soc. Food Sci. Nutr. 2015, 44, 564–572. [Google Scholar] [CrossRef]
- Folin, O.; Denis, W. On phosphotungstic-phosphomolybdic compounds as color reagents. J. Biol. Chem. 1912, 12, 239–243. [Google Scholar]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Tsao, R.; Yu, Q.; Potter, J.; Chiba, M. Direct and simultaneous analysis of sinigrin and allyl isothiocyanate in mustard samples by high-performance liquid chromatography. J. Agric. Food Chem. 2002, 50, 4749–4753. [Google Scholar] [CrossRef] [PubMed]
- McLafferty, F.W.; Turecek, F. Interpretation of Mass Spectra; University Science Books: Sausalito, CA, USA, 1993; pp. 225–282. [Google Scholar]
- Jo, Y.H.; Gu, S.Y.; Chung, N.H.; Gao, Y.; Kim, H.J.; Jeong, M.H.; Jeong, Y.J.; Kwon, J.H. Comparative analysis of sensory profiles of commercial cider vinegars from Korea, China, Japan, and US by SPME/GC-MS, E-nose, and E-tongue. Korean J. Food Sci. Technol. 2016, 48, 430–436. [Google Scholar] [CrossRef] [Green Version]
- Lim, H.J.; Kim, D.S.; Pan, J.H.; Pae, S.B.; Kim, H.S.; Shin, E.C.; Kim, J.K. Characterization of physicochemical and sensory attributes of a novel high-oleic peanut oil cultivar (Arachis hypogaea ssp. Fastigiata L.). Appl. Biol. Chem. 2017, 60, 653–657. [Google Scholar] [CrossRef]
- Shelp, B.J.; Brown, A.W.; McLean, M.D. Metabolism and functions of gamma-aminobutyric acid. Trends Plant Sci. 1999, 4, 446–452. [Google Scholar] [CrossRef]
- Jung, Y.S.; Park, S.J.; Kim, J.E.; Yang, S.A.; Park, J.H.; Kim, J.H.; Jhee, K.H.; Lee, S.P.; Lee, I.S. A comparative study of GABA, glutamate contents, acetylcholinesterase inhibition and antiradical activity of the methanolic extracts from 10 edible plants. J. Korean Soc. Food Sci. Nutr. 2012, 44, 447–451. [Google Scholar]
- Kato, H.; Rhue, M.R.; Nishimura, T. Role of free amino acids and peptides in food taste. In Flavor Chemistry: Trends and Developments; American Chemical Society: Washington DC, USA, 1989; pp. 158–174. [Google Scholar]
- Kang, S.I.; Heu, M.S.; Choi, B.D.; Kim, K.H.; Kim, Y.J.; Kim, J.S. Investigation of food quality characterization of processing by-products (frame muscle) from the sea rainbow trout Oncorhychus mykiss. Korean J. Fish. Aquat. Sci. 2015, 48, 26–35. [Google Scholar]
- Nelson, G.J. Dietary fatty acids and lipid metabolism. In Fatty Acids in Foods and Their Health Implications; Chow, C.K., Ed.; Marcel Dekker Inc.: New York, NY, USA, 1992; pp. 437–471. [Google Scholar]
- Belitz, H.D.; Grosch, W.; Schieberle, P. Food Chemistry, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2004; pp. 427–433. [Google Scholar]
- Kim, S.J.; Lee, M.K.; Back, S.S.; Chun, B.S. Extraction and identification of volatile isocyanates from wasabi using supercritical carbon dioxide. Korean J. Biotechnol. Bioeng. 2007, 22, 174–178. [Google Scholar]
- Kumagai, H.; Kishima, N.; Seki, T.; Sakurai, H.; Ishii, K.; Ariga, T. Analysis of volatile components in essential oil of upland wasabi and their inhibitory effects on platelet aggregation. Biosci. Biotechnol. Biochem. 1994, 58, 2131–2135. [Google Scholar] [CrossRef]
- Ina, K.; Ina, H.; Ueda, M.; Yagi, A.; Kishima, I. ω-Methylthioalkyl isothiocyanates in wasabi. Agric. Biol. Chem. 1989, 53, 537–538. [Google Scholar]
- Depree, J.A.; Howard, T.M.; Savage, G.P. Flavor and pharmaceutical properties of the volatile sulfur compounds of wasabi. Food Res. Int. 1999, 31, 329–337. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Parts | Proximates (g/100 g) | ||||
|---|---|---|---|---|---|
| Moisture | Crude Protein | Crude Lipid | Crude Ash | Carbohydrate (1) | |
| Leaf | 92.50 ± 0.24 b(2) | 3.21 ± 0.36 a | 0.32 ± 0.01 a | 1.72 ± 0.01 b | 2.26 ± 0.58 b |
| Petiole | 94.37 ± 0.5 a | 2.45 ± 0.14 b | 0.13 ± 0.01 b | 2.07 ± 0.35 a | 0.98 ± 0.36 b |
| Rhizome | 81.35 ± 0.23 c | 2.33 ± 0.35 b | 0.10 ± 0.01 c | 0.98 ± 0.36 c | 15.48 ± 0.28 a |
| Amino Acid | Composition (%) | ||
|---|---|---|---|
| Leaf | Petiole | Rhizome | |
| Essential amino acids | |||
| Lysine | 7.17 ± 1.24 a(1) | 8.51 ± 1.07 a | 6.15 ± 1.36 b |
| Methionine | 1.50 ± 0.12 a | 0.17 ± 0.05 b | 1.61 ± 0.09 a |
| Leucine | 7.79 ± 1.47 a | 6.60 ± 1.26 a | 4.56 ± 0.95 b |
| Phenylalanine | 7.27 ± 0.87 a | 6.80 ± 1.05 a | 5.03 ± 0.74 a |
| Valine | 4.06 ± 0.58 a | 4.56 ± 0.69 a | 5.04 ± 0.77 a |
| Histidine | 3.73 ± 0.64 a | 4.05 ± 0.45 a | 3.05 ± 0.38 a |
| Threonine | 4.96 ± 0.48 a | 5.19 ± 0.57 a | 5.57 ± 0.74 a |
| Isoleucine | 2.02 ± 0.09 ab | 2.39 ± 0.13 a | 1.79 ± 0.14 b |
| Non-essential amino acids | |||
| Arginine | 7.16 ± 1.05 b | 4.84 ± 0.69 c | 10.24 ± 1.36 a |
| Glutamic acid | 18.61 ± 2.35 b | 23.27 ± 2.98 a | 20.47 ± 3.05 ab |
| Aspartic acid | 12.97 ± 3.24 b | 12.95 ± 3.14 b | 17.72 ± 2.99 a |
| Alanine | 7.33 ± 1.21 ab | 7.95 ± 1.36 a | 6.71 ± 1.45 b |
| Glycine | 6.53 ± 2.05 a | 6.13 ± 1.22 a | 4.90 ± 1.07 b |
| Serine | 5.96 ± 0.94 a | 6.58 ± 0.87 a | 5.87 ± 1.08 a |
| Tyrosine | 2.93 ± 0.15 a | 0.00 ± 0.00 | 1.29 ± 0.08 b |
| %Essential amino acids | 38.51 ± 4.17 a | 38.27 ± 5.02 a | 38.27 ± 5.02 a |
| %Non-essential amino acids | 61.49 ± 6.62 a | 61.73 ± 8.54 a | 67.20 ± 7.13 a |
| %Total amino acids | 100.00 | 100.00 | 100.00 |
| Amino Acid | Composition (%) | ||
|---|---|---|---|
| Leaf | Petiole | Rhizome | |
| Aspartic acid | 1.27 ± 0.05 a(1) | 0.76 ± 0.08 b | 0.74 ± 0.09 b |
| ρ-Serine | 0.00 ± 0.00 | 0.33 ± 0.05 | 0.00 ± 0.00 |
| Threonine | 6.37 ± 0.14 c | 7.38 ± 0.21 b | 11.02 ± 0.32 a |
| Serine | 4.20 ± 0.11 b | 4.77 ± 0.13 a | 3.18 ± 0.14 c |
| Glutamic acid | 6.90 ± 0.14 b | 7.64 ± 0.24 a | 3.58 ± 0.18 c |
| Glycine | 0.38 ± 0.07 b | 0.41 ± 0.09 b | 1.70 ± 0.08 a |
| Alanine | 12.49 ± 0.43 b | 19.55 ± 0.61 a | 8.75 ± 0.25 c |
| Valine | 8.81 ± 0.17 b | 9.25 ± 0.31 b | 10.08 ± 0.33 a |
| Cysteine | 0.00 ± 0.00 | 0.47 ± 0.08 b | 0.68 ± 0.09 a |
| Methionine | 0.11 ± 0.02 b | 0.24 ± 0.03 a | 0.26 ± 0.04 a |
| Isoleucine | 3.67 ± 0.25 b | 3.41 ± 0.12 b | 3.95 ± 0.15 a |
| Leucine | 3.73 ± 0.19 a | 2.28 ± 0.09 b | 3.75 ± 0.18 a |
| Tyrosine | 2.64 ± 0.15 a | 1.03 ± 0.05 b | 2.71 ± 0.10 a |
| Phenylalanine | 5.64 ± 0.24 a | 2.65 ± 0.07 b | 2.14 ± 0.06 b |
| β-alanine | 2.08 ± 0.11 a | 1.16 ± 0.08 b | 1.00 ± 0.07 b |
| γ-Aminobutyric acid | 33.29 ± 0.97 a | 33.37 ± 1.06 a | 26.82 ± 1.15 b |
| Lysine | 1.30 ± 0.07 a | 0.55 ± 0.10 b | 0.76 ± 0.06 b |
| Histidine | 1.15 ± 0.04 c | 3.85 ± 0.11 b | 5.77 ± 0.15 a |
| Arginine | 5.97 ± 0.22 b | 0.90 ± 0.07 c | 13.12 ± 0.47 a |
| Total | 100.00 | 100.00 | 100.00 |
| Fatty Acid | Composition (%Weight) | ||
|---|---|---|---|
| Leaf | Petiole | Rhizome | |
| Palmitic acid | 11.68 ± 0.01 c(1) | 17.56 ± 0.02 b | 19.37 ± 0.05 a |
| Stearic acid | 3.68 ± 0.01 a | 2.25 ± 0.03 c | 3.04 ± 0.09 b |
| Oleic acid (ω-9) | 16.33 ± 0.09 a | 14.39 ± 0.04 b | 14.22 ± 0.13 b |
| Linoleic acid (ω-6) | 8.12 ± 0.07 c | 12.19 ± 0.10 b | 18.19 ± 0.12 a |
| Linolenic acid (ω-3) | 59.57 ± 0.12 a | 52.87 ± 0.14 b | 44.25 ± 0.23 a |
| Arachidic acid | 0.26 ± 0.01 b | 0.27 ± 0.01 b | 0.49 ± 0.04 a |
| Gondoic acid (ω-9) | 0.20 ± 0.02 b | 0.27 ± 0.03 a | 0.26 ± 0.02 a |
| Behenic acid | 0.15 ± 0.02 b | 0.21 ± 0.02 a | 0.19 ± 0.09 a |
| %Saturated fatty acids | 15.78 ± 0.21 c | 20.29 ± 0.14 b | 23.09 ± 0.16 a |
| %Monounsaturated fatty acids | 16.53 ± 0.15 a | 14.66 ± 0.09 b | 14.48 ± 0.13 b |
| %Polyunsaturated fatty acids | 67.99 ± 0.25 a | 65.06 ± 0.31 b | 62.44 ± 0.24 c |
| Mineral | Composition (%) | ||
|---|---|---|---|
| Leaf | Petiole | Rhizome | |
| P (phosphorus) | 9.11 ± 0.87 a(1) | 5.34 ± 0.56 b | 6.75 ± 0.69 b |
| S (Sulfur) | 20.44 ± 1.97 a | 6.89 ± 0.74 c | 14.92 ± 1.22 b |
| K (potassium) | 43.75 ± 4.35 c | 65.02 ± 4.21 a | 57.93 ± 3.23 b |
| Ca (calcium) | 19.66 ± 1.21 a | 18.99 ± 2.09 a | 16.15 ± 1.43 b |
| Mg (magnesium) | 6.61 ± 0.43 a | 3.24 ± 0.33 b | 4.02 ± 0.29 b |
| Fe (iron) | 0.15 ± 0.04 a | 0.08 ± 0.02 b | 0.06 ± 0.02 b |
| As (arsenic) | 0.05 ± 0.03 a | 0.06 ± 0.03 a | 0.04 ± 0.03 a |
| Zn (zinc) | 0.08 ± 0.02 a | 0.04 ± 0.01 b | 0.08 ± 0.03 a |
| Mn (manganese) | 0.15 ± 0.02 a | 0.14 ± 0.03 a | 0.04 ± 0.02 b |
| Al (aluminum) | 0.01 ± 0.00 b | 0.19 ± 0.03 a | 0.01 ± 0.00 b |
| Wasabia koreana Nakai | |||
|---|---|---|---|
| Leaf | Petiole | Rhizome | |
| Vitamin C (mg/g) | 108.09 ± 7.43 a | 64.72 ± 4.21 c | 94.17 ± 6.97 b |
| Total phenolic content (mg GAE/100 g) | 12.75 ± 1.70 a(1) | 0.42 ± 0.81 b | 3.82 ± 0.60 b |
| IC50 (mg) | 7.64 ± 0.54 b | 17.24 ± 1.17 a | 16.95 ± 0.61 a |
| Wasabia koreana Nakai | |||
|---|---|---|---|
| Leaf | Petiole | Rhizome | |
| Sinigrin (mg/g) | 23.45 ± 0.33 b(1) | 22.80 ± 0.41 b | 92.79 ± 1.81 a |
| AITC (mg/g) | 0.25 ± 0.01 b | 0.17 ± 0.02 c | 0.89 ± 0.01 a |
| Compounds | RT (min) a | RI b | Content (μg/g) | Odor Intensity | Odor Description | ||
|---|---|---|---|---|---|---|---|
| Leaf | Petiole | Rhizome | |||||
| Acids | |||||||
| 2-Hydroxybenzene methanol | 5.43 | <800 | 0.34 ± 0.49 | n.d. c | n.d. | ||
| Dimethyl silanediol | 5.81 | <800 | n.d. | 7.71 ± 10.90 | n.d. | ||
| Trifluorolactic acid | 18.81 | 1144 | n.d. | 0.08 ± 0.11 | n.d. | ||
| 3-Methyl-2-phenyl pentanoic acid | 27.26 | 1452 | 0.10 ± 0.14 | n.d. | n.d. | ||
| 2-Tridecyl ester methoxy acetic acid | 27.90 | 1481 | 0.69 ± 0.98 | n.d. | n.d. | ||
| Alcohols | |||||||
| 1,8-Cineole | 16.29 | 1061 | 1.81 ± 2.55 | n.d. | n.d. | ||
| 2,5-Dimethyl-2,5-hexanediol | 16.42 | 1065 | n.d. | 2.02 ± 2.86 | n.d. | ||
| Aldehydes | |||||||
| 2-1-Phenanthrylbenzaldehyde | 14.58 | 1005 | 0.27 ± 0.39 | n.d. | n.d. | ||
| 2-Hexenal | 19.26 | 1159 | n.d. | 0.08 ± 0.11 | n.d. | ||
| 3-Hexene-1,6-dialdehyde | 24.82 | 1361 | 1.17 ± 1.65 | 9.42 ± 3.32 | n.d. | ||
| Hydrocarbons | |||||||
| Ethyl-1,3-dithioisoindoline | 8.51 | 827 | 5.71 ± 8.08 | 0.64 ± 0.90 | n.d. | ||
| 2-Methyl-5-dibenzazepine | 9.52 | 858 | 0.64 ± 0.91 | n.d. | 4.47 ± 6.32 | ||
| 1-Heptadecene | 19.10 | 1154 | 0.05 ± 0.07 | n.d. | n.d. | ||
| Azulene | 20.13 | 1187 | n.d. | 1.22 ± 1.73 | n.d. | 1 | Wasabi |
| Nonadecane | 25.74 | 1396 | n.d. | 1.08 ± 1.52 | n.d. | ||
| Tridecane | 26.39 | 1411 | 0.86 ± 1.22 | n.d. | n.d. | ||
| Heterocyclic | |||||||
| 2-4′-Nitro-2′-thienylpyrimidine | 8.65 | 832 | n.d. | 1.73 ± 2.45 | n.d. | ||
| Isopropyl isothiocyanate | 9.91 | 869 | n.d. | n.d. | 1.37 ± 1.93 | ||
| 3-Methylisothiazole | 11.11 | 901 | 4.93 ± 1.32 | 2.84 ± 4.01 | 8.67 ± 2.26 | ||
| Allylisothiocyanate | 11.50 | 914 | 71.51 ± 8.14 | 8.99 ± 2.71 | 117.80 ± 66.60 | 2 | Wasabi |
| Diethyl-1-propanamine | 12.25 | 938 | 0.99 ± 1.39 | n.d. | n.d. | ||
| 4-Isothiocyanato-1-butene | 13.94 | 987 | 3.01 ± 1.74 | n.d. | 3.59 ± 5.08 | ||
| Dihydro-5,5-dimethyl-2-furanone | 14.12 | 991 | n.d. | 2.51 ± 3.55 | n.d. | ||
| 2-Ethyl-3-methyl-4-phenyl-6-chloroquinoline | 15.36 | 1032 | 0.32 ± 0.45 | n.d. | n.d. | ||
| Paromomycin | 18.84 | 1145 | n.d. | n.d. | 0.36 ± 0.52 | ||
| 2-Chloro-3,4-diphenylbenzofluoro pyridine | 20.26 | 1192 | n.d. | 0.09 ± 0.13 | n.d. | ||
| 3′,4′,5,5′,7-Pentamethoxyflavone | 21.19 | 1226 | n.d. | 0.39 ± 0.55 | n.d. | ||
| Tetramethylpyrazine | 22.50 | 1273 | 0.09 ± 0.13 | n.d. | n.d. | ||
| Ketone | |||||||
| 1,7,7-Trimethylbicyclo-2.2.1-heptan-2-one | 19.75 | 1175 | 46.63 ± 5.95 | n.d. | n.d. | ||
| No. | Compounds | Sensory Descriptors | RT (min) a | RI b | Leaf | Petiole | Rhizome |
|---|---|---|---|---|---|---|---|
| 1 | Hexadecafluoroheptane | - | 10.63 | 363 | 2.33 ± 0.16 | 3.05 ± 0.12 | 104.96 ± 13.9 |
| 2 | Ethanol | alcoholic, pungent, sweet | 13.18 | 427 | 2.15 ± 0.29 | 1.65 ± 0.08 | 479.39 ± 34.03 |
| 3 | 2-Propanol | alcoholic | 15.85 | 492 | 10.25 ± 7.8 | 11.15 ± 0.85 | 231.70 ± 11.30 |
| 4 | Ethyl 2-methylbutyrate | apple, green, sweet | 68.89 | 839 | 7.02 ± 1.12 | 22.69 ± 0.81 | 428.39 ± 58.59 |
| 5 | Allylisothiocyanate | garlic, pungent, sulfurous | 85.90 | 876 | 345.78 ± 48.90 | 1309.28 ± 160.62 | 13,426.24 ± 653.00 |
| 6 | Dimethyltrisulfide | onion, rotten food | 100.21 | 955 | n.d. c | n.d. c | 950.91 ± 99.54 |
| 7 | Amylpropanoate | apricot, fruity, very sweet | 103.04 | 971 | 2.14 ± 0.25 | 1.6 ± 0.34 | 235.05 ± 24.21 |
| 8 | 1-Octen-3-ol | garlic, herbaceous, spicy | 108.39 | 994 | 44.00 ± 6.20 | 105.01 ± 19.11 | 262.59 ± 32.82 |
| 9 | Linalool | green, muscat, parsley | 120.29 | 1,096 | 5.59 ± 2.11 | 1.5 ± 0.13 | 597.26 ± 64.23 |
| 10 | Decanal | green, herbaceous, peel | 131.60 | 1,206 | 0.53 ± 0.21 | 0.42 ± 0.03 | 74.82 ± 9.47 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, D.-S.; Kim, H.S.; Lee, J.; Pan, J.H.; Kim, Y.J.; Kim, J.K.; Woo, S.; Shin, E.-C. Wasabia koreana Nakai: A Preliminary Study on Nutrients and Chemical Compounds That May Impact Sensory Properties. Molecules 2018, 23, 2512. https://doi.org/10.3390/molecules23102512
Kim D-S, Kim HS, Lee J, Pan JH, Kim YJ, Kim JK, Woo S, Shin E-C. Wasabia koreana Nakai: A Preliminary Study on Nutrients and Chemical Compounds That May Impact Sensory Properties. Molecules. 2018; 23(10):2512. https://doi.org/10.3390/molecules23102512
Chicago/Turabian StyleKim, Da-Som, Hoe Sung Kim, Jookyeong Lee, Jeong Hoon Pan, Young Jun Kim, Jae Kyeom Kim, Seongmin Woo, and Eui-Cheol Shin. 2018. "Wasabia koreana Nakai: A Preliminary Study on Nutrients and Chemical Compounds That May Impact Sensory Properties" Molecules 23, no. 10: 2512. https://doi.org/10.3390/molecules23102512
APA StyleKim, D.-S., Kim, H. S., Lee, J., Pan, J. H., Kim, Y. J., Kim, J. K., Woo, S., & Shin, E.-C. (2018). Wasabia koreana Nakai: A Preliminary Study on Nutrients and Chemical Compounds That May Impact Sensory Properties. Molecules, 23(10), 2512. https://doi.org/10.3390/molecules23102512






