Antiviral Mechanism of Action of Epigallocatechin-3-O-gallate and Its Fatty Acid Esters
Abstract
1. Introduction
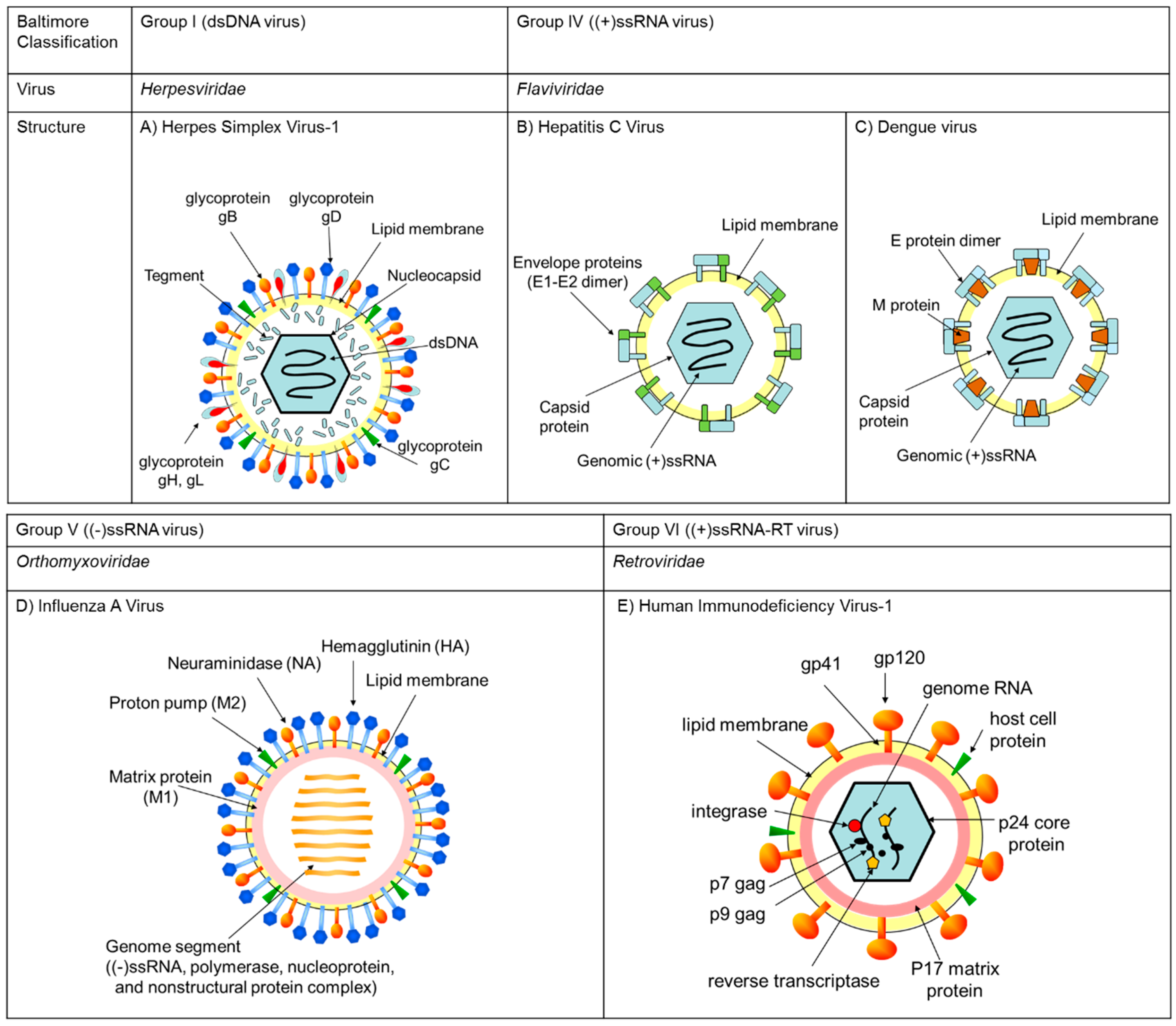
2. Classifications, Structures, and Life Cycles of Viruses Discussed in This Review
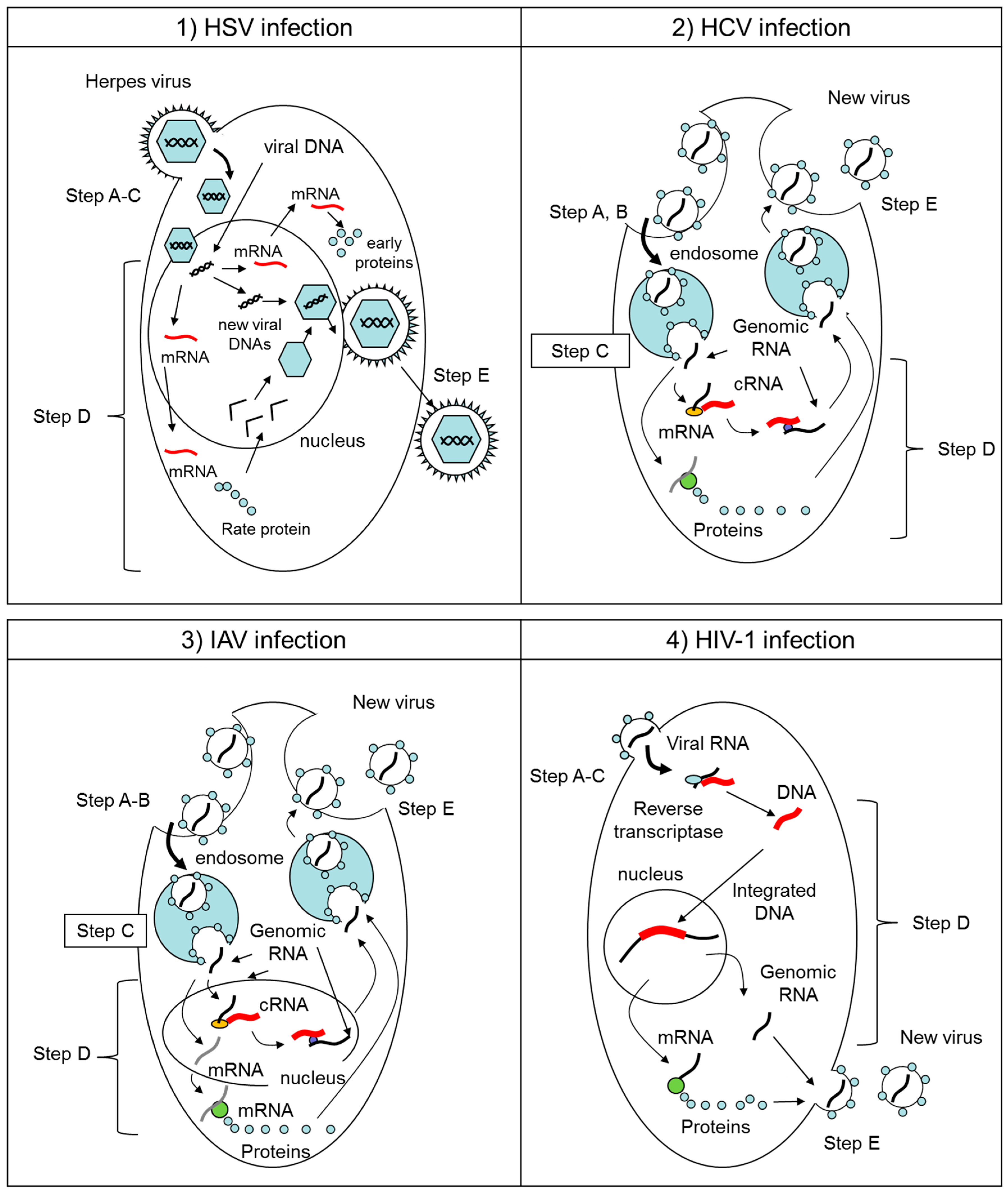
2.1. Herpes Simplex Virus (HSV)
2.2. Hepatitis C Virus (HCV), Zika Virus (ZIKV), West Nile Virus (WNV), Dengue Virus (DENV), and Chikungunya Virus (CHIKV)
2.3. Influenza A Virus (IAV)
2.4. Human Immunodeficiency Virus-1 (HIV-1)
3. Antiviral Activity of Catechins on Enveloped DNA Viruses
4. Antiviral Activity of Catechins on Enveloped (+) ssRNA Viruses
5. Antiviral Activity of Catechins on Enveloped (−) RNA Virus
5.1. Antiviral Mechanism of Action of EGCG on HIV-1
5.2. Antiviral Mechanism of EGCG on Influenza Virus
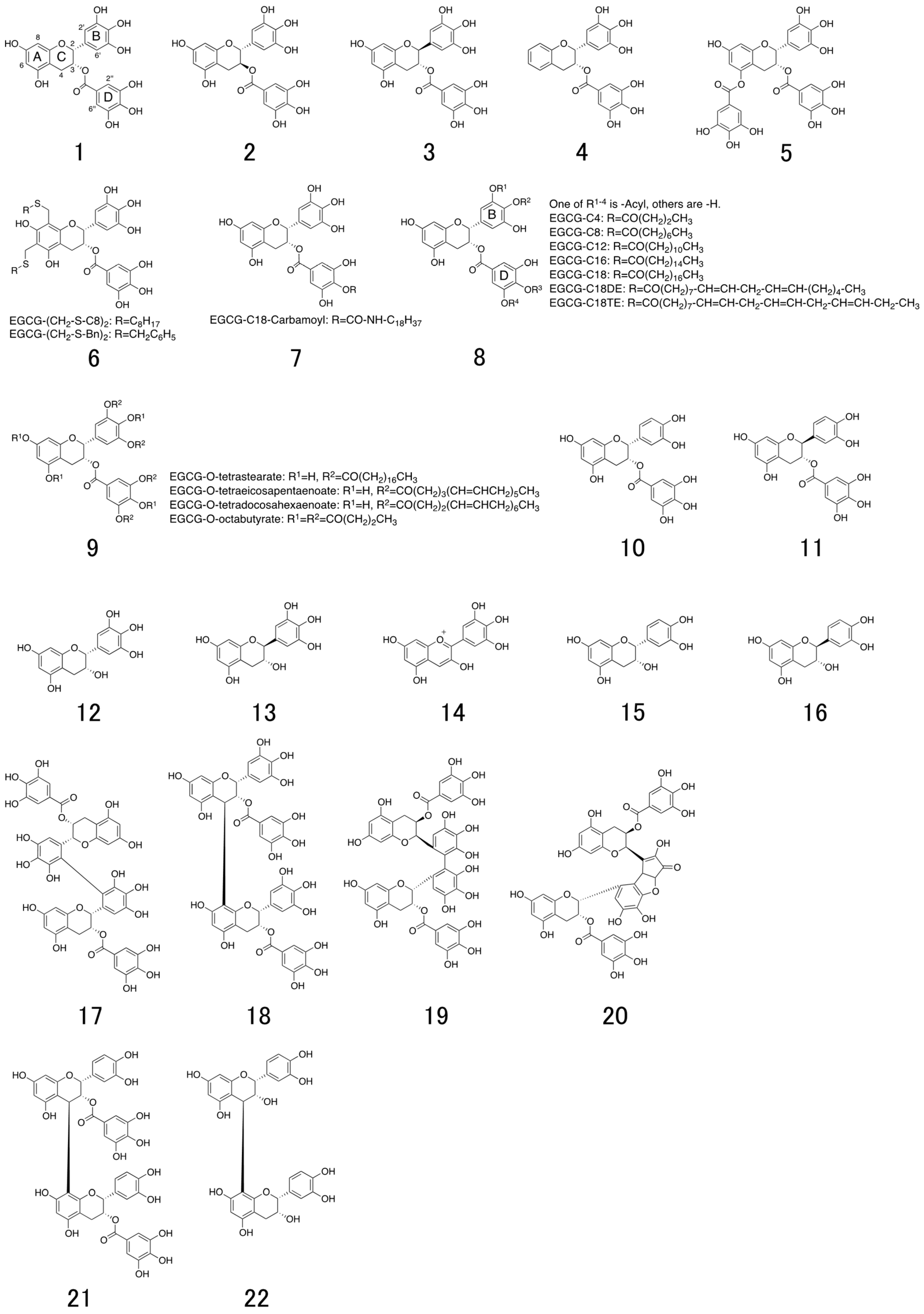
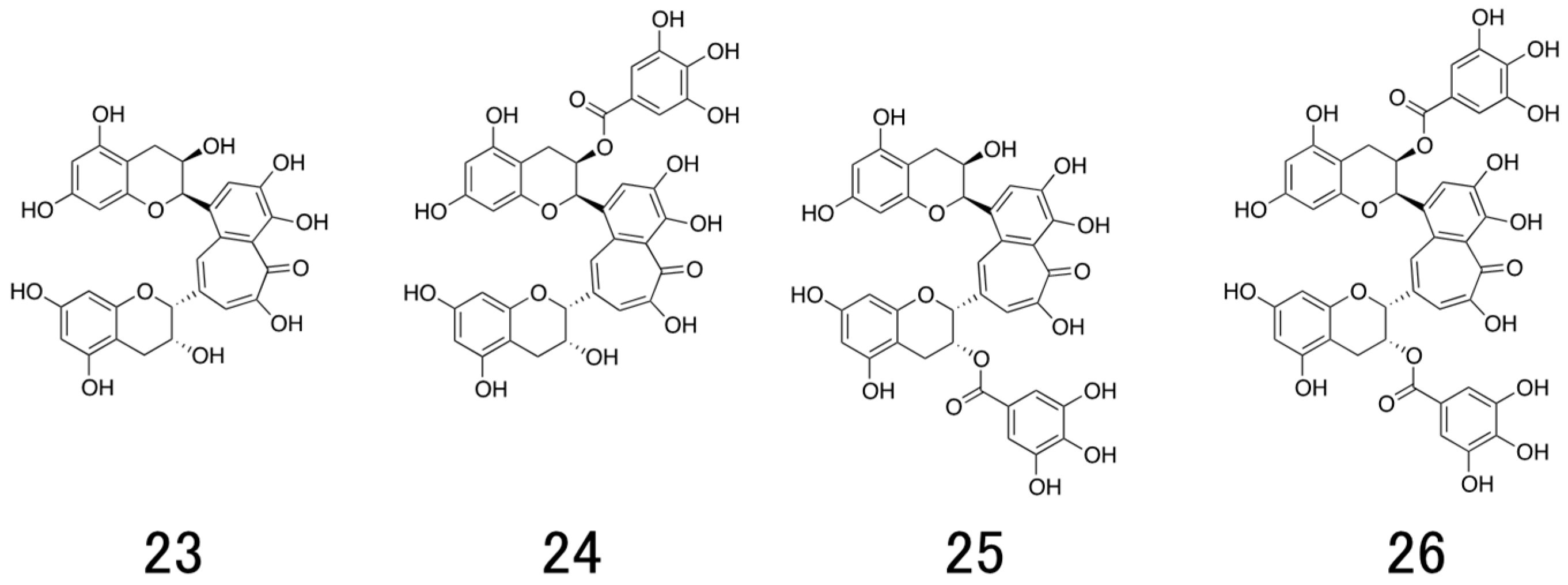
6. Structure and Antiviral Activity of Catechin Derivatives
7. Lipid Bilayer Affinity of EGCG-Alkyl Ether Derivatives
8. EGCG-Fatty Acid Derivatives
8.1. Synthesis of EGCG-Fatty Acid Derivatives
8.2. Cytotoxicity and Influenza Virus Inhibitory Effect of EGCG and EGCG-Fatty Acid Derivatives
8.3. Anti-Influenza Virus Activity of EGCG-C16 in Chicken Embryonated Eggs
8.4. Antiviral Activity of EGCG-Fatty Acid Derivatives for Other Viruses
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AAPH | 2,2′-azobis(2-amidinopropane) dihydrocholoride |
| AMVN | 2,2′-azobis (2,4-dimethylvaleronitrile) |
| bEGCdG | 2′,2′-bisepigallocatechin digallate |
| BVDV | bovine viral diarrhea virus |
| C | (−)-catechin |
| CC50 | 50% cytotoxic concentration |
| CG | (−) catechin-3-O-gallate |
| CHIKV | chikungunya virus |
| DENV | dengue virus |
| DO-EGCG | 5,7-dideoxy-EGCG |
| E | envelope protein |
| EBOV | Ebola virus |
| EC | (−) epicatechin |
| EC50 | 50% effective concentration |
| ECG | (−) epicatechin-3-O-gallate |
| EGC | (−)-epigallocatechin |
| EGCDG | epigallocatechin 3,5-digallate |
| EGCG | (−)-epigallocatechin-3-O-gallate |
| ELISA | enzyme-linked immunosorbent assay |
| EM | electron microscopy |
| GC | (−)-gallocatechin |
| GCG | gallocatechin-3-O-gallate |
| HA | hemagglutinin |
| HBV | hepatitis B virus |
| HCV | hepatitis C virus |
| HCVcc | cell-culture-derived HCV |
| HPV | human papilloma virus |
| HSV | herpes simplex virus |
| HVEM | herpes virus entry mediator |
| IAV | influenza A virus |
| IC50 | 50% inhibitory concentration |
| JEV | Japanese encephalitis |
| M | matrix protein |
| MALDI | matrix assisted laser desorption/ionization |
| MDCK | Madin-Darby canine kidney |
| MUNANA | 2′-(4-methylumbelliferyl)-α-d-N-acetylneuraminic acid |
| NA | neuraminidase |
| NS | nonstructural protein |
| NMR | nuclear magnetic resonance |
| PA | polymerase subunit A |
| PB1 | polymerase subunit B1 |
| PB2 | polymerase subunit B2 |
| PRRSV | porcine reproductive and respiratory syndrome virus |
| RNP | ribonucleoprotein |
| RT | reverse transcription |
| SINV | sindbis virus |
| SEVI | semen-derived enhancer of virus infection |
| TCID50 | 50% tissue culture infectious dose |
| TF | theaflavin |
| TFDG | theaflavin-3,3′-O-digallate |
| TF-3-G | theaflavin-3-gallate |
| TF-3′-G | theaflavin-3′-gallate |
| WNV | West Nile viruses |
| YFV | yellow fever virus |
| ZIKV | zika virus |
References
- Wang, Y.; Ho, C.-T. Polyphenolic Chemistry of Tea and Coffee: A Century of Progress. J. Agric. Food Chem. 2009, 57, 8109–8114. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, M.; Suzuki, K.; Toda, M.; Okubo, S.; Hara, Y.; Shimamura, T. Inhibition of the infectivity of influenza virus by tea polyphenols. Antivir. Res. 1993, 21, 289–299. [Google Scholar] [CrossRef]
- Taguri, T.; Tanaka, T.; Kouno, I. Antibacterial spectrum of plant polyphenols and extracts depending upon hydroxyphenyl structure. Biol. Pharm. Bull. 2006, 29, 2226–2235. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Daniel, K.G.; Kuhn, D.J.; Kazi, A.; Bhuiyan, M.; Li, L.; Wang, Z.; Wan, S.B.; Lam, W.H.; Chan, T.H.; et al. Green tea and tea polyphenols in cancer prevention. Front. Biosci. 2004, 9, 2618–2631. [Google Scholar] [CrossRef] [PubMed]
- Lyu, S.-Y.; Rhim, J.-Y.; Park, W.-B. Antiherpetic Activities of Flavonoids against Herpes Simplex Virus Type 1 (HSV-1) and Type 2 (HSV-2) In Vitro. Arch. Pharm. Res. 2005, 28, 1293–1301. [Google Scholar] [CrossRef] [PubMed]
- Savi, L.A.; Barardi, C.R.A.; Simoes, C.M.O. Evaluation of Antiherpetic Activity and Genotoxic Effects of Tea Catechin Derivatives. J. Agric. Food Chem. 2006, 54, 2552–2557. [Google Scholar] [CrossRef] [PubMed]
- Isaacs, C.E.; Wen, G.Y.; Xu, W.; Jia, J.H.; Rohan, L.; Corbo, C.; Di Maggio, V.; Jenkins, E.C., Jr.; Hillier, S. Epigallocatechin Gallate Inactivates Clinical Isolates of Herpes Simplex Virus. Antimicrob. Agents Chemother. 2008, 52, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Gescher, K.; Hensel, A.; Hafezi, W.; Derksen, A.; Kuhn, J. Oligomeric proanthocyanidins from Rumex acetosa L. inhibit the attachment of herpes simplex virus type-1. Antivir. Res. 2011, 89, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Isaacs, C.E.; Xu, W.; Merz, G.; Hillier, S.; Rohan, L.; Wen, G.Y. Digallate Dimers of (−)-Epigallocatechin Gallate Inactivate Herpes Simplex Virus. Antimicrob. Agents Chemother. 2011, 55, 5646–5653. [Google Scholar] [CrossRef] [PubMed]
- Colpitts, C.C.; Schang, L.M. A Small Molecule Inhibits Virion Attachment to Heparan Sulfate- or Sialic Acid-Containing Glycans. J. Virol. 2014, 88, 7806–7817. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, P.; Nguyen, M.L. Herpes simplex virus virucidal activity of MST-312 and epigallocatechin gallate. Virus Res. 2018, 249, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.M.; Ruzindana-Umunyana, A.; Imbeault, L.; Sircar, S. Inhibition of adenovirus infection and adenain by green tea catechins. Antivir. Res. 2003, 58, 167–173. [Google Scholar] [CrossRef]
- He, L.; Zhang, E.; Shi, J.; Li, X.; Zhou, K.; Zhang, Q.; Le, A.D.; Tang, X. (−)-Epigallocatechin-3-gallate inhibits human papillomavirus (HPV)-16 oncoprotein-induced angiogenesis in non-small cell lung cancer cells by targeting HIF-1a. Cancer Chemother. Pharmacol. 2013, 71, 713–725. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Li, L.-X.; Liao, Q.-J.; Liu, C.-L.; Chen, X.-L. Epigallocatechin gallate inhibits HBV DNA synthesis in a viral replication-inducible cell line. World J. Gastroenterol. 2011, 17, 1507–1514. [Google Scholar] [CrossRef] [PubMed]
- Zuo, G.; Li, Z.; Chen, L.; Xu, X. Activity of compounds from Chinese herbal medicine Rhodiola kirilowii (Regel) Maxim against HCV NS3 serine protease. Antivir. Res. 2007, 76, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Ciesek, S.; von Hahn, T.; Colpitts, C.C.; Schang, L.M.; Friesland, M.; Steinmann, J.; Manns, M.P.; Ott, M.; Wedemeyer, H.; Meuleman, P.; et al. The Green Tea Polyphenol, Epigallocatechin-3-Gallate, Inhibits Hepatitis C Virus Entry. Hepatology 2011, 54, 1947–1955. [Google Scholar] [CrossRef] [PubMed]
- Calland, N.; Albecka, A.; Belouzard, S.; Wychowski, C.; Duverlie, G.; Descamps, V.; Hober, D.; Dubuisson, J.; Rouillé, Y.; Séron, K. (−)-Epigallocatechin-3-Gallate Is a New Inhibitor of Hepatitis C Virus Entry. Hepatology 2012, 55, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Bhat, R.; Adam, A.T.; Lee, J.J.; Deloison, G.; Rouillé, Y.; Séron, K.; Rotella, D.P. Structure-activity studies of (−)-epigallocatechin gallate derivatives as HCV entry inhibitors. Bioorg. Med. Chem. Lett. 2014, 24, 4162–4165. [Google Scholar] [CrossRef] [PubMed]
- Calland, N.; Sahuc, M.E.; Belouzard, S.; Pène, V.; Bonnafous, P.; Mesalam, A.A.; Deloison, G.; Descamps, V.; Sahpaz, S.; Wychowski, C.; et al. Polyphenols Inhibit Hepatitis C Virus Entry by a New Mechanism of Action. J. Virol. 2015, 89, 10053–10063. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, B.M.; Batista, M.N.; Braga, A.C.S.; Nogueira, M.L.; Rahal, P. The green tea molecule EGCG inhibits Zika virus entry. Virology 2016, 496, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Gupta, D.; Pathan, J.K.; Malviya, S.; Kharia, A. The recent allopathic and herbal approaches for Zika Virus. Int. J. Pharm. Life Sci. 2016, 7, 5271–5280. [Google Scholar]
- Sharma, N.; Murali, A.; Singh, S.K.; Giri, R. Epigallocatechin gallate, an active green tea compound inhibits the Zika virus entry into host cells via binding the envelope protein. Int. J. Biol. Macromol. 2017, 104, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Calvo, Á.; Jiménez de Oya, N.; Martín-Acebes, M.A.; Garcia-Moruno, E.; Saiz, J.C. Antiviral Properties of the Natural Polyphenols Delphinidin and Epigallocatechin Gallate against the Flaviviruses West Nile Virus, Zika Virus, and Dengue Virus. Front. Microbiol. 2017, 8, 1314. [Google Scholar] [CrossRef] [PubMed]
- Raekiansyah, M.; Buerano, C.C.; Luz, M.A.D.; Morita, K. Inhibitory effect of the green tea molecule EGCG against dengue virus infection. Arch. Virol. 2018, 163, 1649–1655. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.-W.; Hsieh, P.-S.; Lin, C.-C.; Hu, M.-K.; Huang, S.-M.; Wang, Y.-M.; Liang, C.-Y.; Gong, Z.; Ho, Y.-J. Synergistic effects of combination treatment using EGCG and suramin against the chikungunya virus. Biochem. Biophys. Res. Commun. 2017, 491, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Liu, S.; Li, C.; Yang, L.; Zu, Y. In Vitro Evaluation of the Antiviral Activity of the Synthetic Epigallocatechin Gallate Analog-Epigallocatechin Gallate (EGCG) Palmitate against Porcine Reproductive and Respiratory Syndrome Virus. Viruses 2014, 6, 938–950. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.W.; Hsu, F.L.; Lin, J.Y. Inhibitory Effects of Polyphenolic Catechins from Chinese Green Tea on HIV Reverse Transcriptase Activity. J. Biomed. Sci. 1994, 1, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Tillekeratne, L.M.V.; Sherette, A.; Grossman, P.; Hupe, L.; Hupe, D.; Hudson, R.A. Simplified Catechin-Gallate Inhibitors of HIV-1 Reverse Transcriptase. Bioorg. Med. Chem. Lett. 2001, 11, 2763–2767. [Google Scholar] [CrossRef]
- Kawai, K.; Tsuno, N.H.; Kitayama, J.; Okaji, Y.; Yazawa, K.; Asakage, M.; Hori, N.; Watanabe, T.; Takahashi, K.; Nagawa, H. Epigallocatechin gallate, the main component of tea polyphenol, binds to CD4 and interferes with gp120 binding. J. Allergy Clin. Immunol. 2003, 112, 951–957. [Google Scholar] [CrossRef]
- Liu, S.; Lu, H.; Zhao, Q.; He, Y.; Niu, J.; Debnath, A.K.; Wu, S.; Jiang, S. Theaflavin derivatives in black tea and catechin derivatives in green tea inhibit HIV-1 entry by targeting gp41. Biochim. Biophys. Acta 2005, 1723, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Williamson, M.P.; McCormick, T.G.; Nance, C.L.; Shearer, W.T. Epigallocatechin gallate, the main polyphenol in green tea, binds to the T-cell receptor, CD4: Potential for HIV-1 therapy. J. Allergy Clin. Immunol. 2006, 118, 1369–1374. [Google Scholar] [CrossRef] [PubMed]
- Nance, C.L.; Siwak, E.B.; Shearer, W.T. Preclinical development of the green tea catechin, epigallocatechin gallate, as an HIV-1 therapy. J. Allergy Clin. Immunol. 2009, 123, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Chen, W.; Yi, K.; Wu, Z.; Si, Y.; Han, W.; Zhao, Y. The evaluation of catechins that contain a galloyl moiety as potential HIV-1 integrase inhibitors. Clin. Immunol. 2010, 137, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Hattori, T.; Kodama, E.N. Epigallocatechin gallate inhibits the HIV reverse transcription step. Antivir. Chem. Chemother. 2011, 21, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Hartjen, P.; Frerk, S.; Hauber, I.; Matzat, V.; Thomssen, A.; Holstermann, B.; Hohenberg, H.; Schulze, W.; Schulze zur Wiesch, J.; van Lunzen, J. Assessment of the range of the HIV-1 infectivity enhancing effect of individual human semen specimen and the range of inhibition by EGCG. AIDS Res. Ther. 2012, 9, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Castellano, L.M.; Hammond, R.M.; Holmes, V.M.; Weissman, D.; Shorter, J. Epigallocatechin-3-gallate rapidly remodels PAP85-120, SEM1(45-107), and SEM2(49-107) seminal amyloid fibrils. Biol. Open. 2015, 4, 1206–1212. [Google Scholar] [CrossRef] [PubMed]
- Reid, S.P.; Shurtleff, A.C.; Costantino, J.A.; Tritsch, S.R.; Retterer, C.; Spurgers, K.B.; Bavari, S. HSPA5 is an essential host factor for Ebola virus infection. Antivir. Res. 2014, 109, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Green, R.H. Inhibition of multiplication of influenza virus by extracts of tea. Proc. Soc. Exp. Biol. Med. 1949, 71, 84–85. [Google Scholar] [CrossRef] [PubMed]
- Imanishi, N.; Tuji, Y.; Katada, Y.; Maruhashi, M.; Konosu, S.; Mantani, N.; Terasawa, K.; Ochiai, H. Additional Inhibitory Effect of Tea Extract on the Growth of Influenza A and B Viruses in MDCK Cells. Microbiol. Immunol. 2002, 46, 491–494. [Google Scholar] [CrossRef] [PubMed]
- Song, J.-M.; Lee, K.-H.; Seong, B.-L. Antiviral effect of catechins in green tea on influenza virus. Antivir. Res. 2005, 68, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Furuta, T.; Hirooka, Y.; Abe, A.; Sugata, Y.; Ueda, M.; Murakami, K.; Suzuki, T.; Tanaka, K.; Kan, T. Concise synthesis of dideoxy-epigallocatechin gallate (DO-EGCG) and evaluation of its anti-influenza virus activity. Bioorg. Med. Chem. Lett. 2007, 17, 3095–3098. [Google Scholar] [CrossRef] [PubMed]
- Kuzuhara, T.; Iwai, Y.; Takahashi, H.; Hatakeyama, D.; Echigo, N. Green tea catechins inhibit the endonuclease activity of influenza A virus RNA polymerase. PLoS Curr. 2009, 1, RRN1052. [Google Scholar] [CrossRef]
- Zu, M.; Yang, F.; Zhou, W.; Liu, A.; Du, G.; Zheng, L. In vitro anti-influenza virus and anti-inflammatory activities of theaflavin derivatives. Antivir. Res. 2012, 94, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Ling, J.-X.; Wei, F.; Li, N.; Li, J.-L.; Chen, L.-J.; Liu, Y.-Y.; Luo, F.; Xiong, H.-R.; Hou, W.; Yang, Z.-Q. Amelioration of influenza virus-induced reactive oxygen species formation by epigallocatechin gallate derived from green tea. Acta Pharmacol. Sin. 2012, 33, 1533–1541. [Google Scholar] [CrossRef] [PubMed]
- Kowalinski, E.; Zubieta, C.; Wolkerstorfer, A.; Szolar, O.H.J.; Ruigrok, R.W.H.; Cusack, S. Structural Analysis of Specific Metal Chelating Inhibitor Binding to the Endonuclease Domain of Influenza pH1N1 (2009) Polymerase. PLoS Pathog. 2012, 8, e1002831. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Kim, S.-Y.; Lee, H.W.; Shin, J.S.; Kim, P.; Jung, Y.-S.; Jeong, H.-S.; Hyun, J.-K.; Lee, C.-K. Inhibition of influenza virus internalization by (−)-epigallocatechin-3-gallate. Antivir. Res. 2013, 100, 460–472. [Google Scholar] [CrossRef] [PubMed]
- Müller, P.; Downard, K.M. Catechin inhibition of influenza neuraminidase and its molecular basis with mass spectrometry. J. Pharm. Biomed. Anal. 2015, 111, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Quosdorf, S.; Schuetz, A.; Kolodziej, H. Different Inhibitory Potencies of Oseltamivir Carboxylate, Zanamivir, and Several Tannins on Bacterial and Viral Neuraminidases as Assessed in a Cell-Free Fluorescence-Based Enzyme Inhibition Assay. Molecules 2017, 22, 1989. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Kusano, R.; Kouno, I. Synthesis and antioxidant activity of novel amphipathic derivatives of tea polyphenol. Bioorg. Med. Chem. Lett. 1998, 8, 1801–1806. [Google Scholar] [CrossRef]
- Mori, S.; Miyake, S.; Kobe, T.; Nakaya, T.; Fuller, S.D.; Kato, N.; Kaihatsu, K. Enhanced anti-influenza A virus activity of (−)-epigallocatechin-3-O-gallate fatty acid monoester derivatives: Effect of alkyl chain length. Bioorg. Med. Chem. Lett. 2008, 18, 4249–4252. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.L.; He, W.Y.; Yao, L.; Zhang, H.P.; Liu, Z.G.; Wang, W.P.; Ye, Y.; Cao, J.J. Characterization of Binding Interactions of (−)-Epigallocatechin-3-gallate from Green Tea and Lipase. J. Agric. Food Chem. 2013, 61, 8829–8835. [Google Scholar] [CrossRef] [PubMed]
- Kaihatsu, K.; Mori, S.; Matsumura, H.; Daidoji, T.; Kawakami, C.; Kurata, H.; Nakaya, T.; Kato, N. Broad and potent anti-influenza virus spectrum of epigallocatechin-3-O-gallate-monopalmitate. J. Mol. Genet. Med. 2009, 3, 195–197. [Google Scholar] [PubMed]
- Daidoji, T.; Kaihatsu, K.; Nakaya, T. The Role of Apoptosis in Influenza Virus Pathogenesis and the Mechanisms Involved in Anti-Influenza Therapies. Curr. Chem. Biol. 2010, 4, 208–218. [Google Scholar]
- Kaihatsu, K.; Barnard, D.L. Recent Developments in Anti-influenza A Virus Drugs and Use in Combination Therapies. Mini Rev. Org. Chem. 2012, 9, 3–10. [Google Scholar] [CrossRef]
- Zhong, Y.; Ma, C.-M.; Shahidi, F. Antioxidant and antiviral activities of lipophilic epigallocatechin gallate (EGCG) derivatives. J. Funct. Foods 2012, 4, 87–93. [Google Scholar] [CrossRef]
- De Oliveira, A.; Adams, S.D.; Lee, L.H.; Murray, S.R.; Hsu, S.D.; Hammond, J.R.; Dickinson, D.; Chen, P.; Chu, T.-C. Inhibition of herpes simplex virus type 1 with the modified green tea polyphenol palmitoyl-epigallocatechin gallate. Food Chem. Toxicol. 2013, 52, 207–215. [Google Scholar] [CrossRef] [PubMed]
| Compound | Virus | Assay | Activity | Ref. |
|---|---|---|---|---|
| ECG | HSV-1 F1/HSV-2 333 | Cytopathic effect inhibition | EC50 = 4.0/63.0 μM | [5] |
| EC | EC50 = 2.5/35.0 μM | |||
| EGC | HSV-1 KOS/HSV-1 29R | Virus replication inhibition | EC50 = 173.6/70.4 μM | [6] |
| GC | EC50 = 103.3/140.1 μM | |||
| C | EC50 = 630.0/629.4 μM | |||
| EC | EC50 = 458.6/107.1 μM | |||
| EGCG | HSV-1 F1/HSV-2 333 | Titer reduction | 102.0/104.4 reduction at 100 μM | [7,9] |
| Theasinensin A | 104.0–5.0/104.0 reduction at 100 μM | |||
| EGCG | HSV-1 17 syn+ | Cytotoxicity inhibition | 98% at 2.0 μM | [8] |
| ECG | 36% at 2.0 μM | |||
| EC | 16% at 2.0 μM | |||
| GC | 3% at 2.0 μM | |||
| EGCG | HSV-1 KOS | Plaque formation inhibition | 60% inhibition (2.0 μM at 4 °C) 98% inhibition (2.0 μM at RT) 80% inhibition (2.0 μM at 37 °C) | [11] |
| Compound | Target | Assay | Activity | Ref. |
|---|---|---|---|---|
| EGCG | HCV serine protease (NS3-SP) | Serine protease inhibition | IC50 = 8.51 μM | [15] |
| ECG | IC50 = 18.55 μM | |||
| EGCG | HCVcc | Luciferase reporter | IC50 = 5.5 μM | [16] |
| EGCG | HCVcc | Luciferase reporter | IC50 = 5.0 μM | [17] |
| EGCG | ZIKV BR/ZIKV MR766 | Focus forming inhibition | >90% inhibition at 100 μM | [20] |
| EGCG | WNV-NY99 | Plaque forming inhibition | >104.0 reduction at 10 μM | [23] |
| ZIKV MR766 | >104.0 at 10 μM | |||
| ZIKV PA259459 | >103.0 at 10 μM | |||
| DENV-2 | >103.0 at 10 μM | |||
| EGCG | CHIKV S27 | Microneutralization | IC50 = 1.99 μg/mL (4.34 μM) | [25] |
| EGCG | DENV-1 | Antigen reduction | EC50 = 14.8 μM | [24] |
| DENV-2 | EC50 = 18.0 μM | |||
| DENV-3 | EC50 = 11.2 μM | |||
| DENV-4 | EC50 = 13.6 μM |
| Compound | Target | Assay | Activity | Ref. |
|---|---|---|---|---|
| EGCG | HIV-1 purified reverse transcriptase | Reverse transcriptase inhibition | IC50 = 0.68 μM | [27] |
| ECG | IC50 = 0.32 μM | |||
| EGC | IC50 = 7.80 μM | |||
| EGCG | HIV-1 purified reverse transcriptase | Reverse transcriptase inhibition | IC50 = 0.73 μM | [28] |
| ECG | IC50 = 0.76 μM | |||
| EGCG | HIV-1 CD4 protein | Anti-CD4 binding to CD4 protein inhibition | Approx. 70% inhibition at 100 μM | [29] |
| EGCG/GCG | HIV-1IIIB | p24 antigen production | IC50 = 9.89 μM/4.61 μM | [30] |
| HIV-1IIIB | Cell-cell fusion | IC50 = no inhibition/7.55 μM | ||
| HIV-1NL4-3-luc pseudotyped with HIV-1HXB2 | Virus-cell fusion | IC50 = 3.44 μM/2.45 μM | ||
| EGCG | HIV-1 gp120 | Inhibition of gp120 binding to CD4+ cells | 40% inhibition at 0.2 μM | [31] |
| EGCG | HIV-1SF162 (R5) | p24 antigen production | IC50 = 4.5 μM | [32] |
| HIV-189.6 (X4/R5) | IC50 = 8.0 μM | |||
| HIV-192UG038 (X4) | IC50 = 9.0 μM | |||
| HIV-1JV1083 (R5) | IC50 = 9.0 μM | |||
| EGCG/EGC | HIV-1 IIIB | Multinuclear activation of galactosidase inhibition | EC50 = 1.6 μM/3.4 μM | [34] |
| HIV-2 EHO | EC50 = 2.0 μM/7.9 μM | |||
| EGCG | HIV-1BAL(X5) | Semen-derived enhancer of virus infection monitored by luciferase reporter expression | Inhibited > 70.6% at 0.4 mM | [35] |
| HIV-1NL4/3 (X4) | ||||
| HIV-1B (isolate) | ||||
| EGCG | HIV-1BL2 | Semen-derived enhancer of virus infection monitored by luciferase reporter expression | ~61% inhibition at 0.25 μM | [36] |
| HIV-1BAL | ~35% inhibition at 0.25 μM | |||
| HIV-89.6 | ~11% inhibition at 0.25 μM |
| Compound | Target | Assay | Activity | Ref. |
|---|---|---|---|---|
| EGCG/ TFDG | A/Yamagata/120/86(H1N1) | Plaque forming inhibition | 100% inhibition at 1.5 μM/ 100% inhibition at 1.5 μM | [2] |
| B/USSR/100/83 | 100% inhibition at 1.5 μM/ 100% inhibition at 1.5 μM | |||
| EGCG/ ECG/EGC | A/Chile/1/83(H1N1) | Plaque forming inhibition | EC50 = 28.4/26.4/318 μM | [40] |
| A/Sydney/5/97(H3N2) | EC50 = 22.8/22.2/309 μM | |||
| B/Yamagata/16/88 | EC50 = 26.1/40.4/311.1 μM | |||
| EGCG/ DO-EGCG | A/Memphis/1/71(H3N2) | Focus forming inhibition | IC50 = 41.25/11.92 μM | [41] |
| EGCG/ ECG | N-terminal endonuclease domain protein of A/PR/8/34(H1N1) RNA polymerase PA | Endonuclease inhibition | 100% inhibition at 10 μM/ 100% inhibition at 10 μM | [42] |
| TF, TF-3-G/ TF-3′-G/ TFDG | A/PR/8/34(H1N1) | NA inhibition | IC50 = 11.65 μg/mL (TF), IC50 = 31.91/35.23/26.25 μM | [43] |
| A/Sydney/5/97(H3N2) | IC50 = 25.72 μg/mL (TF), IC50 = 13.29/18.26/10.67 μM | |||
| B/Jiangsu/10/2003 | IC50 = 27.98 μg/mL (TF), IC50 = 49.60 /49.23 /42.07 μM | |||
| EGCG | A/Yamagata/120/86(H1N1) | Lethal murine infection model | Survival rate improved from 16.7% to 66.7% at 40 mg·kg−1·d−1 oral administration | [44] |
| EGCG | A/Puerto Rico/8/34 (H1N1) | NA activity inhibition | IC50 > 500 μM | [46] |
| Purified NA from A/California/04/2009 (H275Y) | IC50 = 233.7 μM | |||
| EGCG | A/PR/8/34(H1N1), A/USSR/90/77 (H1N1), A/Port Chalmers/1/73(H3N2), A/Aichi/2/68(H3N2) | Plaque forming inhibition | EC50 = 7.3~40.1 μM | [10] |
| EGCG | A/California/04/2009(H1N1) | NA inhibition | IC50 =1565 μM | [48] |
| (+)-GCG | IC50 = 396 μM |
| Target Virus | Virus Inhibitory Effect | Important Functional Group | Targets | Ref. |
|---|---|---|---|---|
| HSV | EGC > EC, GC > C | 5′-OH | Entire infection process | [6] |
| HCV | EGCG > ECG | 5′-OH | NS3-Serine Protease | [15] |
| HIV-1 | ECG, EGCG > EGC | 3-galloyl | Reverse transcriptase | [27] |
| HIV-1 | EGCG > EGC > ECG > C | 3-galloyl, 5′-OH | Glycoprotein(gp120) | [32] |
| Influenza | EGCG > ECG > EGC | 3-galloyl | Hemagglutinin, viral RNA synthesis, Neuraminidase (NA) | [40] |
| Influenza | EGCG > ECG > bEGCdG > EGC | 3-galloyl, 5′-OH | Viral envelope, NA surface glycoprotein | [46] |
| Compound | Assay | CC50 [μM] | EC50 [μM] | SI |
|---|---|---|---|---|
| EGCG | Plaque formation reduction | 275.5 ± 6.00 μM | 94.6 ± 11.1 μM | 2.91 |
| EGCG-C18 | 300.0 ± 25.0 μM | 64.0 ± 0.50 μM | 4.68 | |
| EGCG-C18DE | 250.0 ± 25.0 μM | 7.00 ± 0.50 μM | 35.7 | |
| EGCG-C18TE | 32.0 ± 3.50 μM | 3.00 ± 0.50 μM | 10.6 |
| Compound | Assay | CC50 [μM] | EC50 [μM] | SI |
|---|---|---|---|---|
| EGCG | Plaque formation reduction | 275.5 ± 6.00 μM | 0.391 ± 0.056 μM | 703 |
| EGCG-C18 | 300.0 ± 25.0 μM | 0.060 ± 0.010 μM | 5000 | |
| EGCG-C18DE | 250.0 ± 25.0 μM | 0.180 ± 0.050 μM | 1389 | |
| EGCG-C18TE | 32.0 ± 3.50 μM | 0.10 ± 0.208 μM | 320 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaihatsu, K.; Yamabe, M.; Ebara, Y. Antiviral Mechanism of Action of Epigallocatechin-3-O-gallate and Its Fatty Acid Esters. Molecules 2018, 23, 2475. https://doi.org/10.3390/molecules23102475
Kaihatsu K, Yamabe M, Ebara Y. Antiviral Mechanism of Action of Epigallocatechin-3-O-gallate and Its Fatty Acid Esters. Molecules. 2018; 23(10):2475. https://doi.org/10.3390/molecules23102475
Chicago/Turabian StyleKaihatsu, Kunihiro, Miyuki Yamabe, and Yasuhito Ebara. 2018. "Antiviral Mechanism of Action of Epigallocatechin-3-O-gallate and Its Fatty Acid Esters" Molecules 23, no. 10: 2475. https://doi.org/10.3390/molecules23102475
APA StyleKaihatsu, K., Yamabe, M., & Ebara, Y. (2018). Antiviral Mechanism of Action of Epigallocatechin-3-O-gallate and Its Fatty Acid Esters. Molecules, 23(10), 2475. https://doi.org/10.3390/molecules23102475





