Polyphenol-Rich Extracts from Cotoneaster Leaves Inhibit Pro-Inflammatory Enzymes and Protect Human Plasma Components against Oxidative Stress In Vitro
Abstract
1. Introduction
2. Results
2.1. Phytochemical Standardization of Cotoneaster Leaf Extracts
2.2. Antioxidant Activity of Leaf Extracts in Chemical Models
2.3. Antioxidant Activity of Leaf Extracts in Human Plasma Model
2.4. Inhibitory Effect of Leaf Extracts on Pro-Inflammatory Enzymes
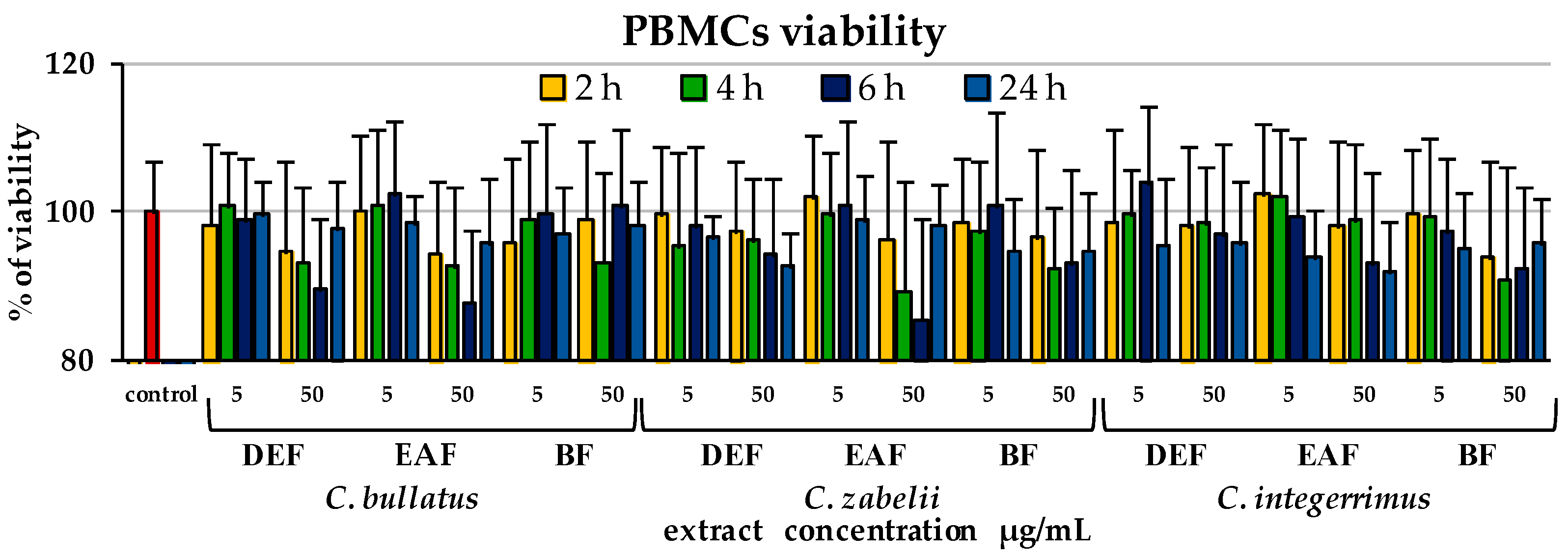
2.5. Influence of the Leaf Extracts on Cell Viability
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Material and Preparation of Dry Extracts
4.3. Phytochemical Profiling
4.4. Antioxidant Activity in Chemical Models
4.5. Antioxidant Activity in Human Plasma Models
4.5.1. Isolation of Blood Plasma and Sample Preparation
4.5.2. Evaluation of Oxidative and Nitrative Damage to Blood Plasma Proteins and Lipids
4.5.3. Determination of the Non-Enzymatic Antioxidant Capacity of Blood Plasma (NEAC)
4.5.4. Inhibition of Pro-Inflammatory Enzymes
4.5.5. Cellular Safety Testing
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, Y.J.; Gan, R.Y.; Li, S.; Zhou, Y.; Li, A.N.; Xu, D.P.; Li, H.B. Antioxidant phytochemicals for the prevention and treatment of chronic diseases. Molecules 2015, 20, 21138–21156. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Xia, S.; Kalionis, B.; Wan, W.; Sun, T. The role of oxidative stress and inflammation in CVD aging. Biomed. Res. Int. 2014, 2014, 1–13. [Google Scholar]
- Nimse, S.B.; Pal, D. Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv. 2015, 5, 27986–28006. [Google Scholar] [CrossRef]
- Farías, J.G.; Molina, V.M.; Carrasco, R.A.; Zepeda, A.B.; Figueroa, E.; Letelier, P.; Castillo, R.L. Antioxidant therapeutic strategies for cardiovascular conditions associated with oxidative stress. Nutrients 2017, 9, 966. [Google Scholar] [CrossRef] [PubMed]
- Les, F.; López, V.; Caprioli, G.; Iannarelli, R.; Fiorini, D.; Innocenti, M.; Bellumori, M.; Maggi, F. Chemical constituents, radical scavenging activity and enzyme inhibitory capacity of fruits from Cotoneaster pannosus Franch. Food Funct. 2017, 8, 1775–1784. [Google Scholar] [CrossRef] [PubMed]
- Zengin, G.; Uysal, A.; Gunes, E.; Aktumsek, A. Survey of phytochemical composition and biological effects of three extracts from a wild plant (Cotoneaster nummularia Fisch. et Mey.): A potential source for functional food ingredients and drug formulations. PLoS ONE 2014, 9, e113527. [Google Scholar] [CrossRef] [PubMed]
- Holzer, V.M.; Lower-Nedza, A.D.; Nandintsetseg, M.; BatHokhuu, J.; Brantner, A.H. Antioxidant constituents of Cotoneaster melanocarpus Lodd. Antioxidants 2013, 2, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, S.; Ghiaee, A.; Naghibi, F.; Mosaddegh, M. Antiplasmodial activity and cytotoxicity of plants used in traditional medicine of Iran for the treatment of fever. Iran J. Pharm. Res. 2015, 14, 103–107. [Google Scholar] [PubMed]
- Uysal, A.; Zengin, G.; Mollica, A.; Gunes, E.; Locatelli, M.; Yilmaz, T.; Aktumsek, A. Chemical and biological insights on Cotoneaster integerrimus: A new (−)-epicatechin source for food and medicinal applications. Phytomedicine 2016, 15, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Kicel, A.; Michel, P.; Owczarek, A.; Marchelak, A.; Żyżelewicz, D.; Budryn, G.; Oracz, J.; Olszewska, M.A. Phenolic profile and antioxidant potential of leaves from selected Cotoneaster Medik. species. Molecules 2016, 21, 688. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N.; Knight, S.; Kuhnert, N. Discriminating between the six isomers of dicaffeoylquinic acid by LC-MSn. J. Agric. Food Chem. 2005, 53, 3821–3832. [Google Scholar] [CrossRef] [PubMed]
- Hamed, A.I.; Al-Ayed, A.S.; Moldoch, J.; Piacenta, S.; Oleszek, W.; Stochmal, A. Profiles analysis of proanthocyanidins in the argun nut (Medemia argun—An ancien Egyptian palm) by LC-ESI-MS/MS. Int. J. Mass Spectrom. 2014, 49, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Karar, M.G.E.; Kuhnert, N. UPLC-ESI-Q-TOF-MS/MS characterization of phenolics from Crataegus monogyna and Crataegus laevigata (Hawthorn) leaves, fruits and their herbal derived drops (Crataegutt Tropfen). J. Chem. Biol. Ther. 2015, 1, 1–23. [Google Scholar]
- Amarowicz, R.; Pegg, R.B. Content of proanthocyanidins in selected plant extracts as determined via n-butanol/HCl hydrolysis and a colorimetric assay or by HPLC—A short report. Pol. J. Food Nutr. Sci. 2006, 15, 319–322. [Google Scholar]
- Beecher, G.R. Proanthocyanidins: Biological activities associated with human health. Pharm. Biol. 2004, 42, 2–20. [Google Scholar] [CrossRef]
- Chen, X.Q.; Hu, T.; Han, Y.; Huang, W.; Yuan, H.B.; Zhang, Y.T.; Du, Y.; Jiang, Y.W. Preventive effects of catechins on cardiovascular disease. Molecules 2016, 21, 1759. [Google Scholar] [CrossRef] [PubMed]
- Gallol, L.C.; Bucalá, V.; Rigo, M.V.R.; Piña, J. Herbal Medicine: Dry Extracts Production and Applications. In Plant Extracts: Role in Agriculture, Health Effects and Medical Applications; Giordano, A., Costs, A., Eds.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2013; pp. 171–198. [Google Scholar]
- Frejnagel, S. Comparison of polyphenolic composition of extracts from honeysuckle, chokeberries and green tea—A short report. Pol. J. Food Nutr. Sci. 2007, 57, 83–86. [Google Scholar]
- Chacko, S.M.; Thambi, P.T.; Kuttan, R.; Nishigaki, I. Beneficial effects of green tea: A literature review. Chin. Med. 2010, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.K.; Mehra, N.K.; Swarnakar, N.K. Role of antioxidants for the treatment of cardiovascular diseases: Challenges and opportunities. Curr. Pharm. Des. 2015, 21, 4441–4455. [Google Scholar] [CrossRef] [PubMed]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef] [PubMed]
- Szabó, C.; Ischiropoulos, H.; Radi, R. Peroxynitrite: Biochemistry, pathophysiology and development of therapeutics. Nat. Rev. Drug Discov. 2007, 6, 662–680. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.; Margaritis, M.; Channon, K.M.; Antoniades, C. Evaluating oxidative stress in human cardiovascular disease: Methodological aspects and considerations. Curr. Med. Chem. 2012, 19, 2504–2520. [Google Scholar] [CrossRef] [PubMed]
- Ho, E.; Karimi Galougahi, K.; Liu, C.C.; Bhindi, R.; Figtree, G.A. Biological markers of oxidative stress: Applications to cardiovascular research and practice. Redox Biol. 2013, 1, 483–4891. [Google Scholar] [CrossRef] [PubMed]
- Hollands, W.J.; Hart, D.J.; Dainty, J.R.; Hasselwander, O.; Tiihonen, K.; Wood, R.; Kroon, P.A. Bioavailability of epicatechin and effects on nitric oxide metabolites of an apple flavanol-rich extract supplemented beverage compared to a whole apple puree: a randomized, placebo-controlled, crossover trial. Mol. Nutr. Food Res. 2013, 57, 1209–1217. [Google Scholar] [CrossRef]
- Kaushik, D.; O’Fallon, K.; Clarkson, P.M.; Dunne, C.P.; Conca, K.R.; Michniak-Kohn, B. Comparison of quercetin pharmacokinetics following oral supplementation in humans. J. Food Sci. 2012, 77, H231–H238. [Google Scholar] [CrossRef]
- Graefe, E.U.; Wittig, J.; Mueller, S.; Riethling, A.K.; Uehleke, B.; Drewelow, B.; Pforte, H.; Jacobasch, G.; Derendorf, H.; Veit, M. Pharmacokinetics and bioavailability of quercetin glycosides in humans. J. Clin. Pharmacol. 2001, 41, 492–499. [Google Scholar] [CrossRef]
- Farah, A.; Monteiro, M.; Donangelo, C.M.; Lafay, S. Chlorogenic acids from green coffee extract are highly bioavailable in humans. J. Nutr. 2008, 138, 2309–2315. [Google Scholar] [CrossRef]
- Bijak, M.; Nowak, P.; Borowiecka, M.; Ponczek, M.B.; Żbikowska, H.M.; Wachowicz, B. Protective effects of (-)-epicatechin against nitrative modifications of fibrinogen. Thromb. Res. 2012, 130, 123–128. [Google Scholar] [CrossRef]
- Libby, P. Inflammatory mechanisms: The molecular basis of inflammation and disease. Nutr. Rev. 2007, 65, 140–146. [Google Scholar] [CrossRef]
- Haeggström, J.Z.; Funk, C.D. Lipoxygenase and leukotriene pathways: Biochemistry, biology, and roles in disease. Chem. Rev. 2011, 111, 5866–5898. [Google Scholar] [CrossRef]
- Spanbroek, R.; Grabner, R.; Lotzer, K.; Hildner, M.; Urbach, A.; Ruhling, K.; Moos, M.P.; Kaiser, B.; Cohnert, T.U.; Wahlers, T.; et al. Expanding expression of the 5-lipoxygenase pathway within the arterial wall during human atherogenesis. Proc. Natl. Acad. Sci. USA 2003, 100, 1238–1243. [Google Scholar] [CrossRef] [PubMed]
- Stern, R.; Jedrzejas, M.J. Hyaluronidases: Their genomics, structures, and mechanisms of action. Chem. Rev. 2006, 106, 818–839. [Google Scholar] [CrossRef] [PubMed]
- Kucur, M.; Karadag, B.; Isman, F.K.; Ataev, Y.; Duman, D.; Karadag, N.; Ongen, Z.; Vural, V.A. Plasma hyaluronidase activity as an indicator of atherosclerosis in patients with coronary artery disease. Bratisl Lek Listy 2009, 110, 21–26. [Google Scholar] [PubMed]
- Pryor, W.A.; Cueto, R.; Jin, X.; Koppenol, W.H.; Ngu-Schwemlein, M.; Squadrito, G.L.; Uppu, P.L.; Uppu, R.M. A practical method for preparing peroxynitrite solutions of low ionic strength and free of hydrogen peroxide. Free Radic. Biol. Med. 1995, 18, 75–83. [Google Scholar] [CrossRef]
- Michel, P.; Dobrowolska, A.; Kicel, A.; Owczarek, A.; Bazylko, A.; Granica, S.; Piwowarski, J.P.; Olszewska, M.A. Polyphenolic profile, antioxidant and anti-inflammatory activity of Eastern Teaberry (Gaultheria procumbens L.) leaf extracts. Molecules 2014, 19, 20498–20520. [Google Scholar] [CrossRef] [PubMed]
- Oleszek, W.; Kapusta, I.; Stochmal, A. TLC of Triterpenes (Including Saponins). In Book Thin Layer Chromatography in Phytochemistry; Taylor & Francis Group: New York, NY, USA, 2007; pp. 519–541. [Google Scholar]
- Olszewska, M.A.; Michel, P. Antioxidant activity of inflorescences, leaves and fruits of three Sorbus species in relation to their polyphenolic composition. Nat. Prod. Res. 2009, 23, 1507–1521. [Google Scholar] [CrossRef] [PubMed]
- Olszewska, M.A.; Nowak, S.; Michel, P.; Banaszczak, P.; Kicel, A. Assessment of the content of phenolics and antioxidant action of inflorescences and leaves of selected species from the genus Sorbus sensu stricto. Molecules 2010, 15, 8769–8783. [Google Scholar] [CrossRef] [PubMed]
- Olszewska, M.A.; Presler, A.; Michel, P. Profiling of phenolic metabolites and antioxidant activity of dry extracts from the selected Sorbus species. Molecules 2012, 17, 3093–3113. [Google Scholar] [CrossRef] [PubMed]
- Kolodziejczyk-Czepas, J.; Nowak, P.; Wachowicz, B.; Piechocka, J.; Głowacki, R.; Moniuszko-Szajwaj, B.; Stochmal, A. Antioxidant efficacy of Kalanchoe daigremontiana bufadienolide-rich fraction in blood plasma in vitro. Pharm. Biol. 2016, 54, 3182–3188. [Google Scholar] [CrossRef] [PubMed]
- Marchelak, A.; Owczarek, A.; Matczak, M.; Pawlak, A.; Kolodziejczyk-Czepas, J.; Nowak, P.; Olszewska, M.A. Bioactivity potential of Prunus spinosa L. flower extracts: Phytochemical profiling, cellular safety, pro-inflammatory enzymes inhibition and protective effects against oxidative stress in vitro. Front. Pharmacol. 2017, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kolodziejczyk-Czepas, J.; Nowak, P.; Moniuszko-Szajwaj, B.; Kowalska, I.; Stochmal, A. Free radical scavenging actions of three Trifolium species in the protection of blood plasma antioxidant capacity in vitro. Pharm. Biol. 2015, 53, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Krzyzanowska-Kowalczyk, J.; Kolodziejczyk-Czepas, J.; Kowalczyk, M.; Pecio, Ł.; Nowak, P.; Stochmal, A. Yunnaneic Acid B, a component of Pulmonaria officinalis extract, prevents peroxynitrite-induced oxidative stress in vitro. J. Agric. Food Chem. 2017, 65, 3827–3834. [Google Scholar] [CrossRef] [PubMed]
- Strober, W. Trypan blue exclusion test of cell viability. Curr. Protoc. Immunol. 2001. [Google Scholar] [CrossRef]
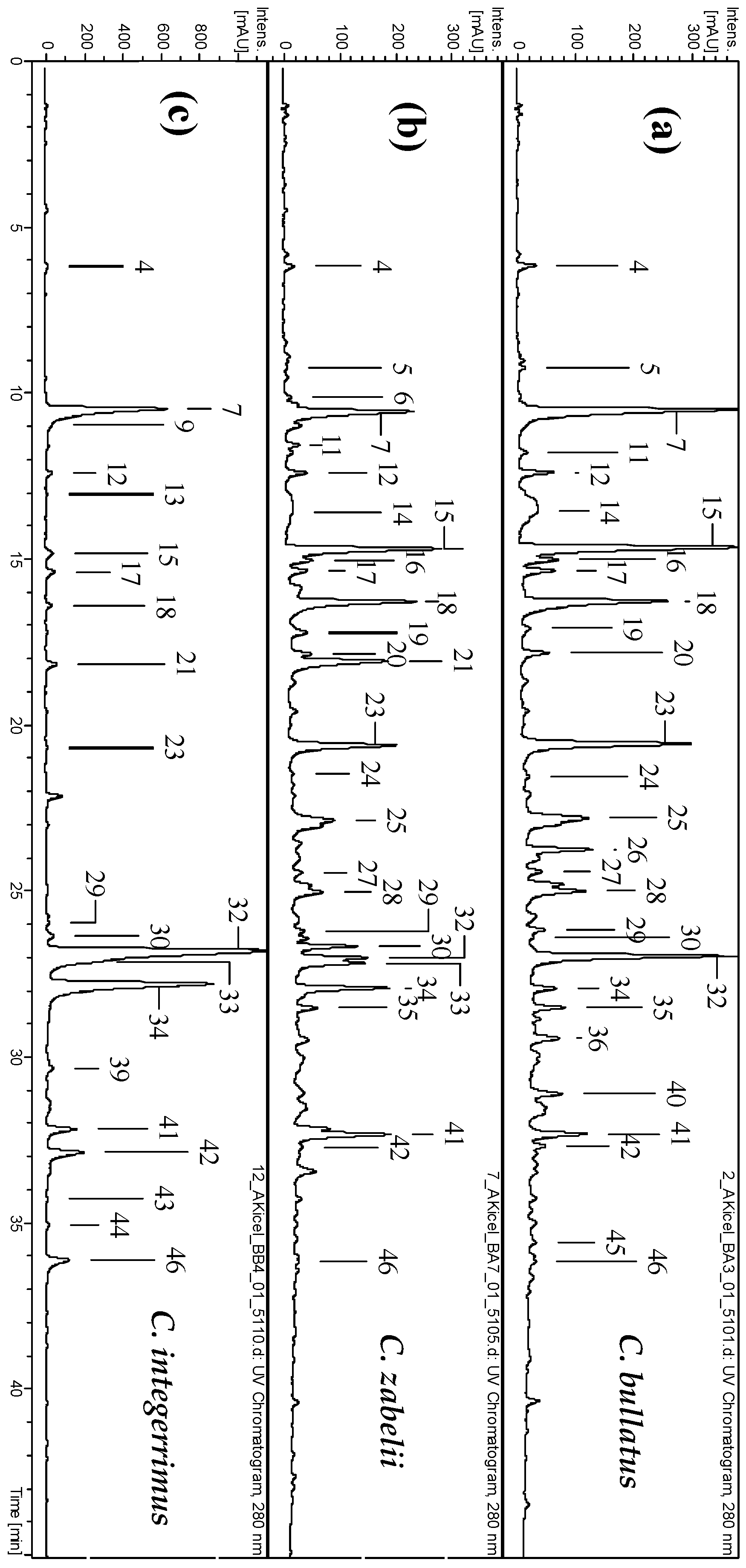
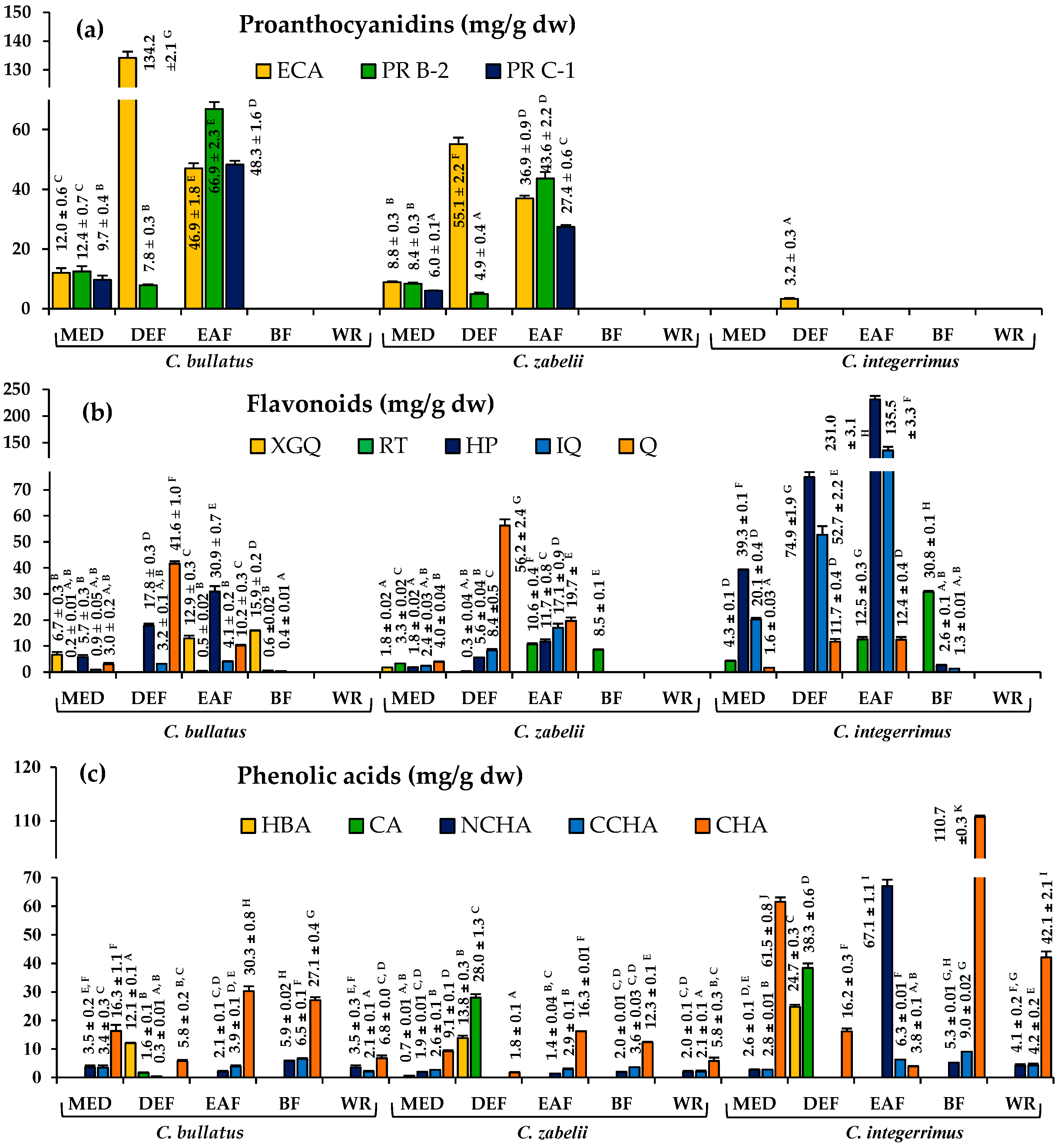

| Extracts a | Extraction yield b (% dw) | Total Phenolics c TPC (mg GAE/g dw) | Total Flavonoids d TFC (mg/g dw) | Total Phenolic Acids e TAC (mg/g dw) | Total Proanthocyanidins | |
| TPA mg CYE/g dw f | TLPA mg/g dw g | |||||
| C. bullatus | ||||||
| MED | 25.60 | 332.94 ± 4.78 E | 18.12 ± 0.71 A | 23.37 ± 1.08 B | 239.62 ± 12.36 G | 43.25 ± 2.01 B |
| DEF | 0.74 | 546.89 ± 25.63 H | 66.55 ± 0.05 C | 30.71 ± 0.90 C | 33.04 ± 1.47 B,C | 278.66 ± 3.21 E |
| EAF | 4.35 | 650.75 ± 7.97 I | 62.56 ± 1.96 C | 37.05 ± 1.04 D,E | 358.22 ± 3.86 I | 252.47 ± 6.64 D |
| BF | 7.21 | 502.93 ± 7.98 G | 20.08 ± 0.27 A | 39.50 ± 0.42 E | 434.88 ± 14.57 J | 9.11 ± 0.09 A |
| WR | 14.72 | 101.32 ± 1.79 A | nd | 12.37 ± 0.67 A | 83.26 ± 2.04 D | nd |
| C. zabelii | ||||||
| MED | 28.20 | 311.48 ± 8.18 D,E | 14.70 ± 0.05 A | 21.87 ± 0.29 B | 241.84 ± 7.27 G | 33.12 ± 0.40 B |
| DEF | 0.54 | 502.69 ± 25.12 G | 81.77 ± 1.05 E | 76.62 ± 0.78 H | 17.63 ± 0.89 A,B | 145.15 ± 7.03 C |
| EAF | 2.55 | 568.25 ± 10.36 H | 76.31 ± 1.60 D,E | 33.39 ± 0.54 C,D | 264.23 ± 13.28 H | 135.03 ± 2.92 C |
| BF | 4.61 | 439.07 ± 4.88 F | 12.66 ± 0.07 A | 20.93 ± 0.14 B | 354.06 ± 1.50 I | 31.42 ± 0.17 B |
| WR | 12.64 | 152.93 ± 1.45 B | nd | 16.12 ± 0.92 A | 122.10 ± 2.10 E | 4.17 ± 0.21 A |
| C. integerrimus | ||||||
| MED | 23.33 | 241.25 ± 7.09 C | 67.71 ± 0.80 C,D | 71.32 ± 0.87 G | 128.62 ± 2.18 E | nd |
| DEF | 0.47 | 453.12 ± 3.36 F | 144.80 ± 2.62 F | 147.77 ± 1.06 K | 9.72 ± 0.26 A | 32.19 ± 0.84 B |
| EAF | 3.35 | 470.93 ± 6.19 F | 403.56 ± 7.48 G | 102.17 ± 1.79 I | 78.47 ± 2.26 D | nd |
| BF | 3.45 | 295.19 ± 1.34 D | 47.25 ± 0.31 B | 127.76 ± 0.06 J | 163.19 ± 5.51 F | nd |
| WR | 20.36 | 107.32 ± 3.54 A | nd | 53.35 ± 2.71 F | 45.60 ± 0.86 C | nd |
| Extracts | DPPH a | FRAP b | ||
|---|---|---|---|---|
| EC50 (µg/mL) | TEAA (mmol TX/g) | (mmol Fe2+/g dw) | TERP (mmol TX/g) | |
| C. bullatus | ||||
| MED | 7.19 ± 0.32 H | 3.06 ± 0.14 D | 10.74 ± 0.07 D | 3.76 ± 0.03 D |
| DEF | 4.84 ± 0.03 D,E,F,G | 4.55 ± 0.03 G,H | 16.99 ± 0.78 I,J | 6.62 ± 0.36 J |
| EAF | 3.19 ± 0.10 B,C | 6.90 ±0.16 K | 17.66 ± 0.28 J | 6.93 ± 0.12 J |
| BF | 4.27 ± 0.24 D,E | 5.15 ± 0.29 H,I,J | 13.42 ± 0.15 E,F | 4.98 ± 0.07 E |
| WR | 22.41 ± 0.51 L | 0.98 ± 0.02 A,B | 3.99 ± 0.10 A | 1.25 ± 0.05 A |
| C. zabelii | ||||
| MED | 7.44 ± 0.03 H | 2.96 ± 0.01 D | 9.42 ± 0.13 C,D | 3.15 ± 0.06 C |
| DEF | 5.39 ± 0.09 F,G | 4.08 ± 0.07 E,F,G | 16.74 ± 0.14 H,I.J | 6.53 ± 0.08 I,J |
| EAF | 3.95 ± 0.20 C,D | 5.57 ± 0.28 J | 15.63 ± 0.16 G,H,I | 6.01 ± 0.07 G,H |
| BF | 4.78 ± 0.23 D,E,F | 4.60 ± 0.23 G,H,I | 10.81 ± 0.08 D | 3.80 ± 0.04 D |
| WR | 18.73 ± 0.33 K | 1.18 ± 0.02 A,B | 3.89 ± 0.15 A | 1.34 ± 0.07 A |
| C. integerrimus | ||||
| MED | 13.66 ± 0.47 J | 1.61 ± 0.05 B,C | 7.84 ± 0.25 B | 2.42 ± 0.11 B |
| DEF | 5.19 ± 0.22 E,F,G | 4.24 ± 0.18 F,G | 15.84 ± 0.12 G,H,I | 6.09 ± 0.05 H,I |
| EAF | 5.77 ± 0.06 G | 3.81 ± 0.04 E,F | 14.44 ± 0.08 F,G | 5.46 ± 0.04 F |
| BF | 10.51 ± 0.64 I | 2.09 ±0.13 C | 8.76 ± 0.33 B,C | 2.84 ± 0.15 B,C |
| WR | 27.88 ± 0.69 M | 0.79 ± 0.02 A | 3.82 ± 0.15 A | 1.17 ± 0.05 A |
| Standards | ||||
| QU | 1.70 ± 0.11 A | 9.00 ± 0.60 L | 31.20 ± 0.98 L | 11.88 ± 0.01 M |
| ECA | 2.35 ± 0.18 A,B | 6.98 ± 0.18 K | 35.79 ± 0.93 M | 8.22 ± 0.26 L |
| CHA | 4.60 ± 0.07 D,E,F | 3.52 ± 0.07 D,E | 25.68 ± 0.51 K | 5.59 ± 0.15 F,G |
| BHA | 2.90 ± 0.15 B | 5.20 ± 0.26 I,J | 16.14 ± 0.77 H,I | 7.73 ± 0.01 K |
| TX | 4.05 ± 0.10 C,D | - | 12.69 ± 0.42 E | - |
| Extracts | LOX | HYAL | ||
| IC50 a (µg/mL) | IC50 b (µg/U) | IC50 a (µg/mL) | IC50 b (µg/U) | |
| C. bullatus | ||||
| MED | 185.77 ± 9.08 E,F | 6.85 ± 0.33 E,F | 8.09 ± 0.67 B | 7.09 ± 0.59 B |
| DEF | 217.01 ± 2.60 F | 8.00 ± 0.10 F | 8.35 ± 0.53 B,C | 7.32 ± 0.46 B,C |
| EAF | 129.29 ± 7.38 C,D | 4.77 ± 0.27 C,D | 19.09 ± 0.62 G | 16.75 ± 0.55 G |
| BF | 95.19 ± 4.31 A,B | 3.51 ± 0.16 A,B | 2.81 ± 0.13 A | 2.46 ± 0.11 A |
| WR | 595.31 ± 27.09 I | 21.96 ± 1.00 I | 27.40 ± 1.14 I | 24.04 ± 1.00 I |
| C. zabelii | ||||
| MED | 217.80 ± 12.56 F | 8.03 ± 0.46 F | 7.92 ± 0.25 B | 6.94 ± 0.22 B |
| DEF | 202.21 ± 8.93 F | 7.46 ± 0.33 F | 7.82 ± 0.16 B | 6.86 ± 0.14 B |
| EAF | 136.39 ± 9.27 C,D | 5.03 ± 0.34 C,D | 15.53 ± 0.40 E,F | 13.63 ± 0.35 E,F |
| BF | 130.99 ± 3.78 C,D | 4.83 ± 0.14 C,D | 6.08 ± 0.19 B | 5.33 ± 0.17 B |
| WR | 427.68 ± 12.47 H | 15.78 ± 0.46 H | 12.70 ± 0.66 D,E | 11.14 ± 0.58 D,E |
| C. integerrimus | ||||
| MED | 283.55 ± 14.12 G | 10.46 ± 0.52 G | 24.00 ± 0.93 H | 21.05 ± 0.82 H |
| DEF | 136.11 ± 8.10 C,D | 5.02 ± 0.30 C,D | 11.21 ± 0.07 C,D | 9.83 ± 0.06 C,D |
| EAF | 159.21 ± 3.97 D,E | 5.87 ± 0.15 D,E | 30.59 ± 2.18 J | 26.83 ± 1.91 J |
| BF | 199.42 ± 6.41 F | 7.36 ± 0.24 F | 11.09 ± 0.29 C,D | 9.73 ± 0.26 C,D |
| WR | 684.84 ± 18.86 J | 25.26 ± 0.70 J | 34.39 ± 0.61 K | 30.16 ± 0.53 K |
| Standards | ||||
| QU | 57.83 ± 1.47 A | 2.15 ± 0.04 A | 15.84 ± 0.87 F | 13.90 ± 0.76 F |
| ECA | 90.59 ± 0.50 A,B | 3.34 ± 0.02 A,B | 14.27 ± 0.20 D,E,F | 12.52 ± 0.18 D,E,F |
| CHA | 114.25 ± 2.87 B,C | 4.21 ± 0.11 B,C | 16.53 ±0.34 F,G | 14.50 ± 0.30 F,G |
| IND | 62.95 ± 2.19 A | 2.32 ± 0.08 A | 8.61 ± 0.22 B,C | 7.77 ± 019 B,C |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kicel, A.; Kolodziejczyk-Czepas, J.; Owczarek, A.; Marchelak, A.; Sopinska, M.; Ciszewski, P.; Nowak, P.; Olszewska, M.A. Polyphenol-Rich Extracts from Cotoneaster Leaves Inhibit Pro-Inflammatory Enzymes and Protect Human Plasma Components against Oxidative Stress In Vitro. Molecules 2018, 23, 2472. https://doi.org/10.3390/molecules23102472
Kicel A, Kolodziejczyk-Czepas J, Owczarek A, Marchelak A, Sopinska M, Ciszewski P, Nowak P, Olszewska MA. Polyphenol-Rich Extracts from Cotoneaster Leaves Inhibit Pro-Inflammatory Enzymes and Protect Human Plasma Components against Oxidative Stress In Vitro. Molecules. 2018; 23(10):2472. https://doi.org/10.3390/molecules23102472
Chicago/Turabian StyleKicel, Agnieszka, Joanna Kolodziejczyk-Czepas, Aleksandra Owczarek, Anna Marchelak, Malgorzata Sopinska, Pawel Ciszewski, Pawel Nowak, and Monika A. Olszewska. 2018. "Polyphenol-Rich Extracts from Cotoneaster Leaves Inhibit Pro-Inflammatory Enzymes and Protect Human Plasma Components against Oxidative Stress In Vitro" Molecules 23, no. 10: 2472. https://doi.org/10.3390/molecules23102472
APA StyleKicel, A., Kolodziejczyk-Czepas, J., Owczarek, A., Marchelak, A., Sopinska, M., Ciszewski, P., Nowak, P., & Olszewska, M. A. (2018). Polyphenol-Rich Extracts from Cotoneaster Leaves Inhibit Pro-Inflammatory Enzymes and Protect Human Plasma Components against Oxidative Stress In Vitro. Molecules, 23(10), 2472. https://doi.org/10.3390/molecules23102472






