Geographic Variation in the Chemical Composition and Antioxidant Properties of Phenolic Compounds from Cyclocarya paliurus (Batal) Iljinskaja Leaves
Abstract
1. Introduction
2. Results and Discussion
2.1. Phytochemical Composition
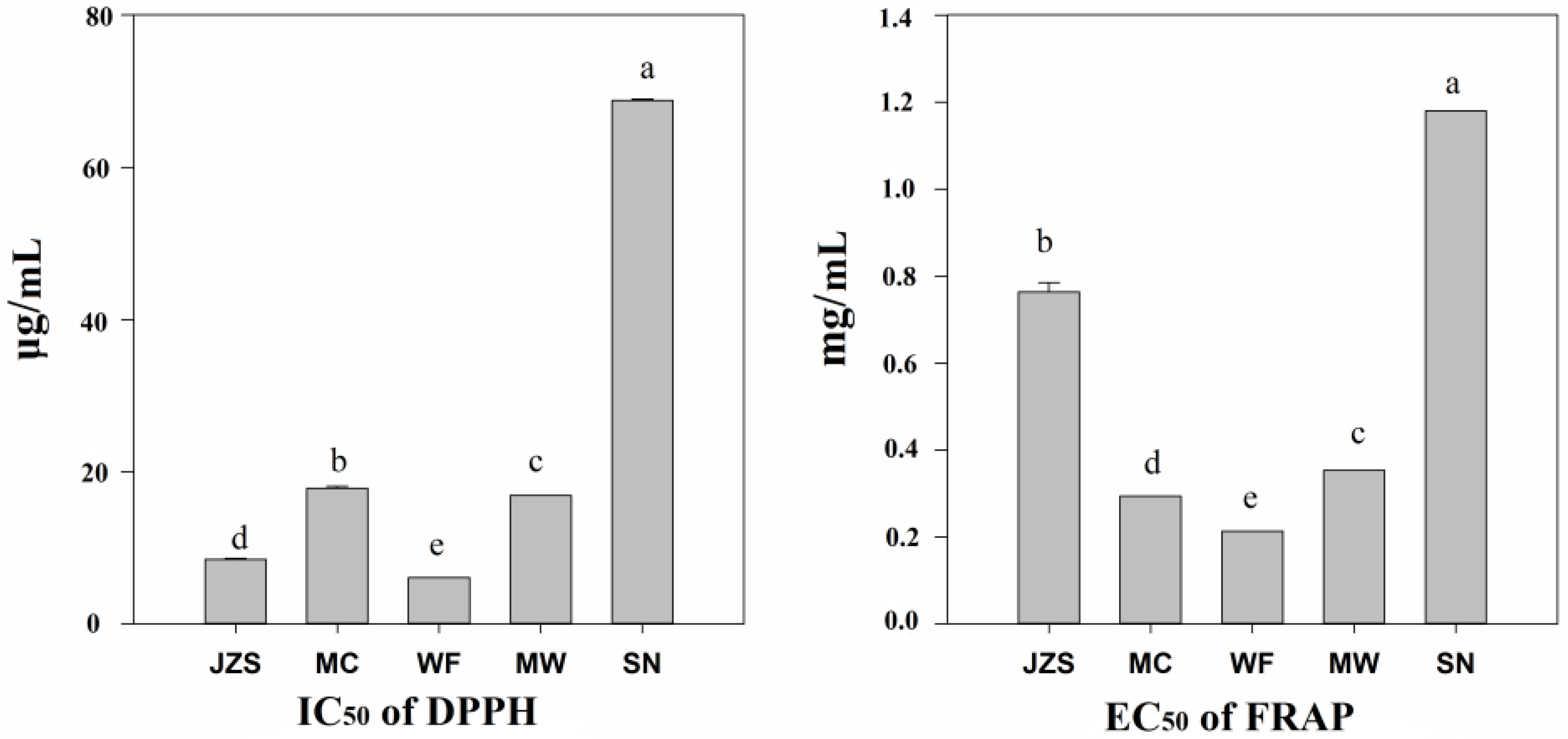
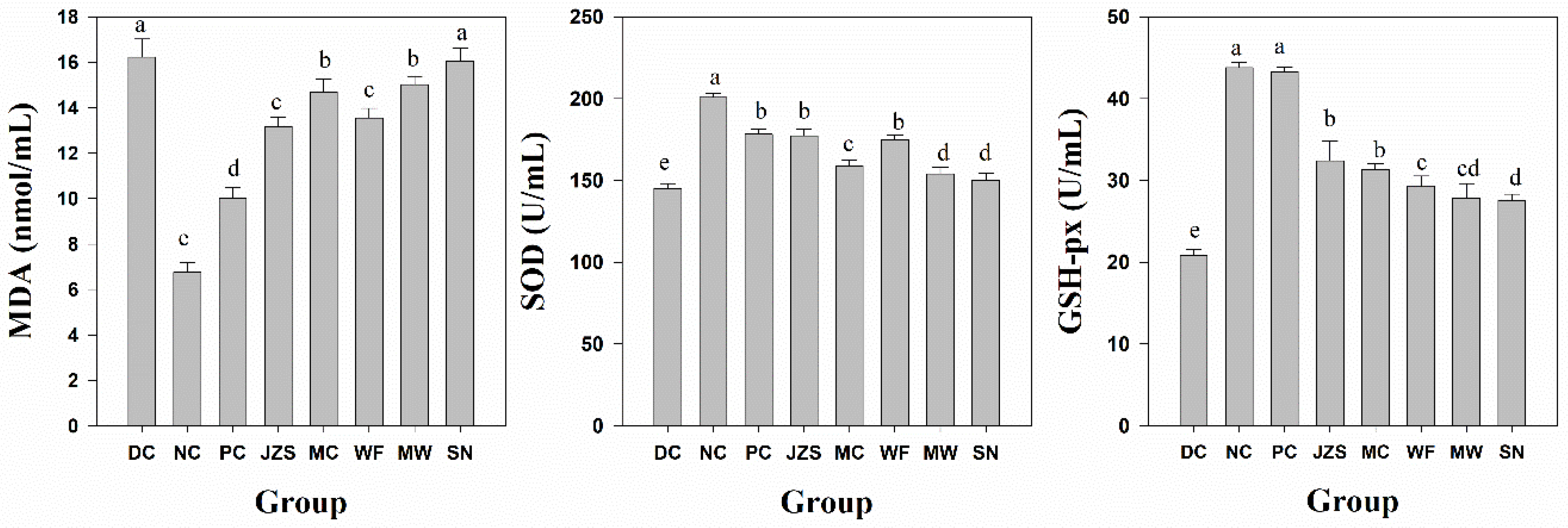
2.2. Variation in Antioxidant Activity
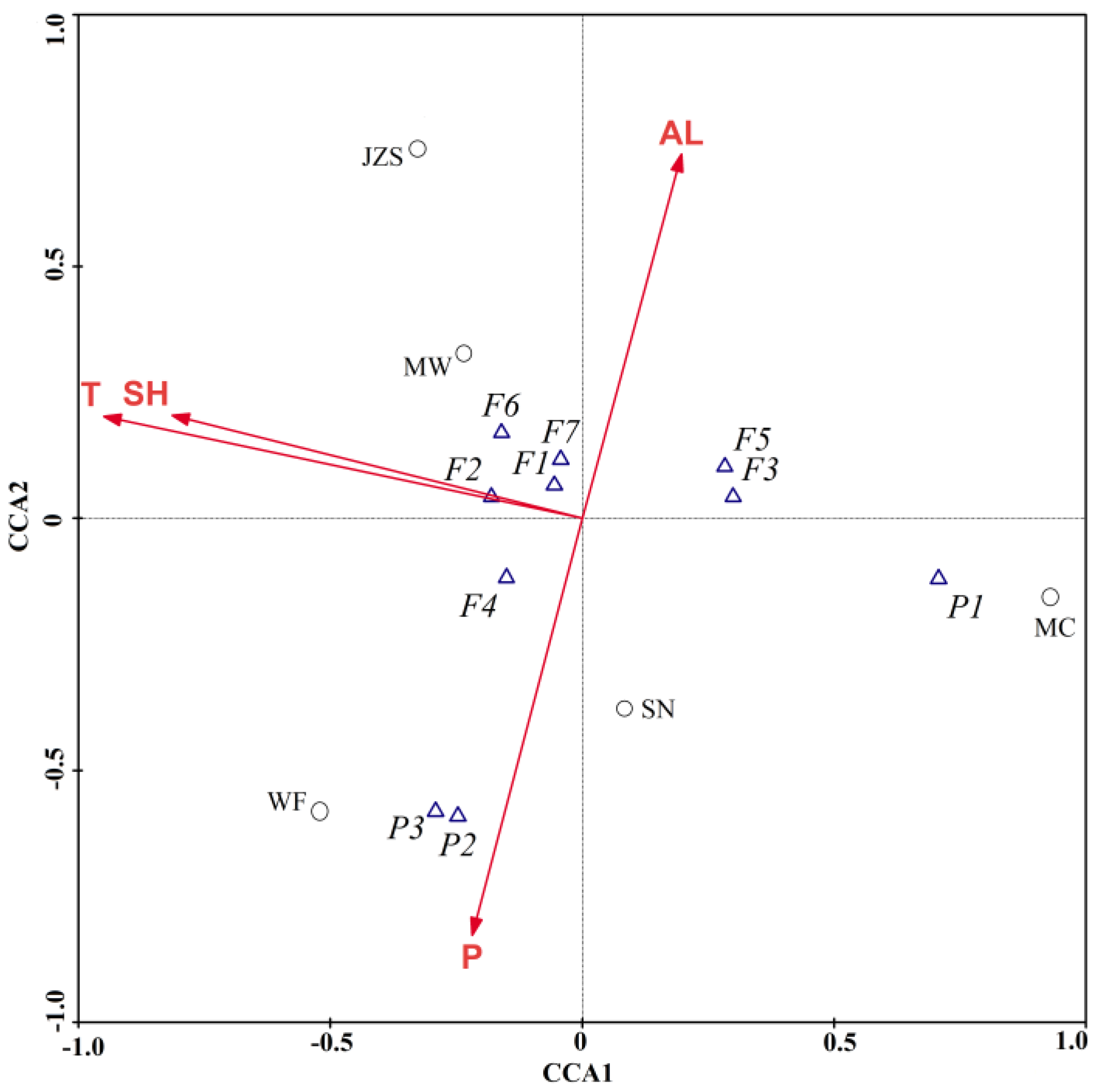
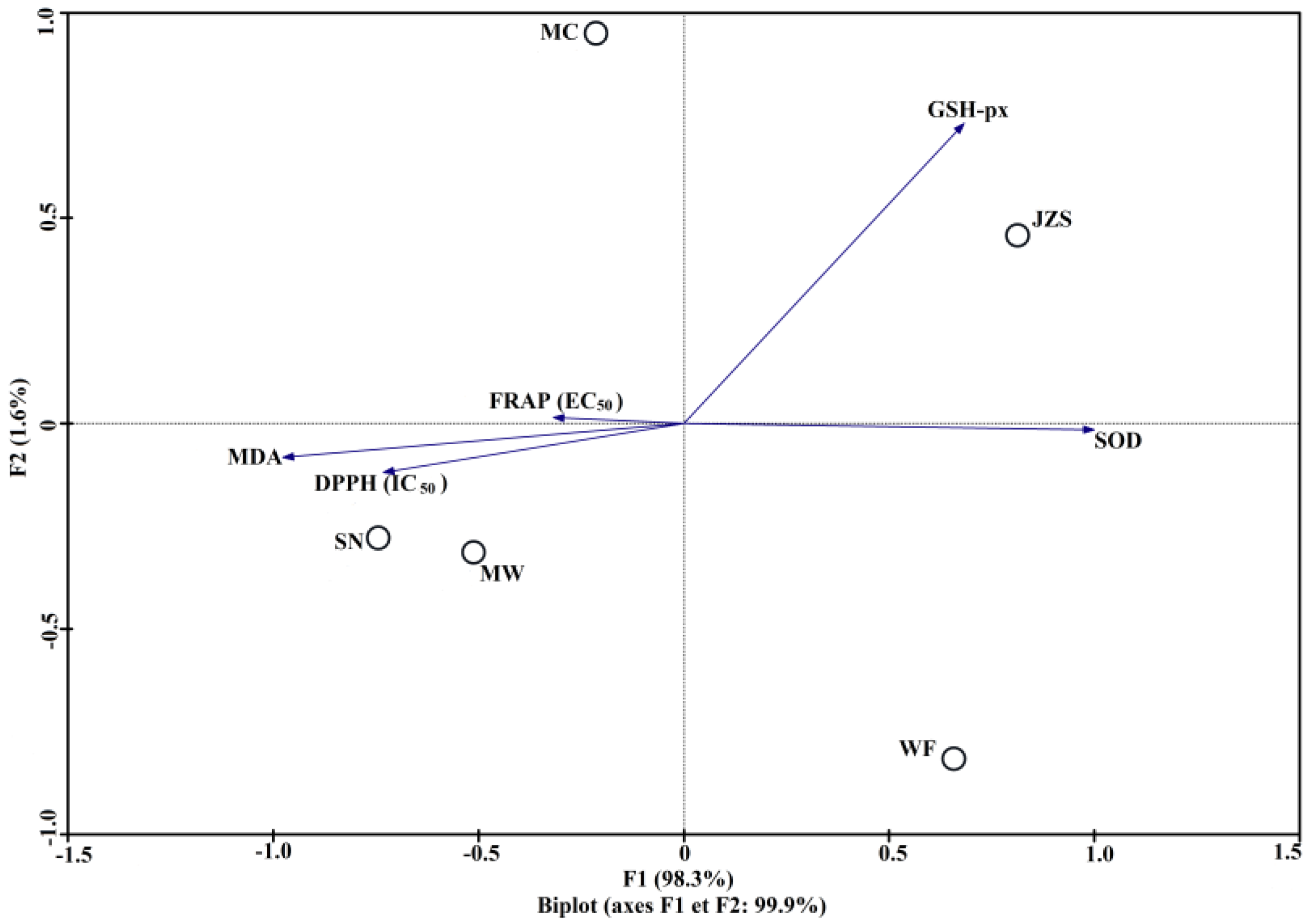
2.3. Correlation of Phenolic Compounds and Antioxidant Activity
3. Materials and Methods
3.1. Plant Material
3.2. Preparation of C. paliurus Extracts
3.3. HPLC-Q-TOF-MS Confirmation
3.4. Determination of Phenolic Compounds by HPLC
3.5. Antioxidant Activity In Vitro
3.5.1. DPPH Radical-Scavenging Activity
3.5.2. FRAP
3.6. Antioxidant Activity In Vivo
3.6.1. Animals and Experimental Design
3.6.2. Biochemical Assay
3.7. Data Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Delfanian, M.; Esmaeilzadeh Kenari, R.; Sahari, M.A. Utilization of Jujube fruit (Ziziphus mauritiana Lam.) extracts as natural antioxidants in stability of frying oil. Int. J. Food Prop. 2016, 19, 789–801. [Google Scholar] [CrossRef]
- Kahl, R.; Kappus, H. Toxicology of the synthetic antioxidants BHA and BHT in comparison with the natural antioxidant vitamin E. Z. Lebensm. Unters. Forsch. 1993, 196, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.D.; Ma, Y.J.; Parry, J.; Gao, J.M.; Yu, L.L.; Wang, M. Phenolics content and antioxidant activity of tartary buckwheat from different locations. Molecules 2011, 16, 9850–9867. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cao, Y.N.; Fang, S.Z.; Wang, T.L.; Fu, X.X. Antidiabetic effect of Cyclocarya paliurus leaves depends on the contents of antihyperglycemic flavonoids and antihyperlipidemic triterpenoids. Molecules 2018, 23, 1042. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Luo, H.; Xu, M.; Zhai, M.; Guo, Z.; Qiao, Y.; Wang, L. Dynamic changes in phenolics and antioxidant capacity during Pecan (Carya illinoinensis) kernel ripening and its phenolics profiles. Molecules 2018, 23, 435. [Google Scholar] [CrossRef] [PubMed]
- Mensor, L.L.; Menezes, F.S.; Leitão, G.G.; Reis, A.S.; Santos, T.C.D.; Coube, C.S.; Leitão, S.G. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother. Res. 2001, 15, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Čopra-Janićijević, A.; Čulum, D.; Vidic, D.; Tahirović, A.; Klepo, L.; Bašić, N. Chemical composition and antioxidant activity of the endemic Crataegus microphylla Koch subsp malyana KI Chr. and Janjić from Bosnia. Ind. Crops Prod. 2018, 113, 75–79. [Google Scholar] [CrossRef]
- Xie, J.H.; Dong, C.J.; Nie, S.P.; Li, F.; Wang, Z.J.; Shen, M.Y.; Xie, M.Y. Extraction, chemical composition and antioxidant activity of flavonoids from Cyclocarya paliurus (Batal.) Iljinskaja leaves. Food Chem. 2015, 186, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.; Wang, J.; Wei, Z.; Zhu, Z. Methods to break seed dormancy in Cyclocarya paliurus (Batal) Iljinskaja. Sci. Hortic. 2006, 110, 305–309. [Google Scholar] [CrossRef]
- Kurihara, H.; Asami, S.; Shibata, H.; Fukami, H.; Tanaka, T. Hypolipemic effect of Cyclocarya paliurus (Batal) Iljinskaja in lipid-loaded mice. Biol. Pharm. Bull. 2003, 26, 383–385. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.; Yang, W.; Chu, X.; Shang, X.; She, C.; Fu, X. Provenance and temporal variations in selected flavonoids in leaves of Cyclocarya paliurus. Food Chem. 2011, 124, 1382–1386. [Google Scholar] [CrossRef]
- Deng, B.; Shang, X.; Fang, S.; Li, Q.; Fu, X.; Su, J. Integrated effects of light intensity and fertilization on growth and flavonoid accumulation in Cyclocarya paliurus. J. Agric. Food Chem. 2012, 60, 6286–6292. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Fang, S.; Yin, Z.; Fu, X.; Shang, X.; Yang, W.; Yang, H. Chemical Fingerprint and Multicomponent Quantitative Analysis for the Quality Evaluation of Cyclocarya paliurus Leaves by HPLC-Q-TOF-MS. Molecules 2017, 22, 1927. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Gao, W.; Tang, L. Antidiabetic herbal drugs officially approved in China. Phytother. Res. 2003, 17, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Ahmad Aufa, Z.; Hassan, F.A.; Ismail, A.; Mohd Yusof, B.N.; Hamid, M. Chemical compositions and antioxidative and antidiabetic properties of underutilized vegetable palm hearts from Plectocomiopsis geminiflora and Eugeissona insignis. J. Agric. Food Chem. 2014, 62, 2077–2084. [Google Scholar] [CrossRef] [PubMed]
- Altiparmak, I.H.; Erkus, M.E.; Gunebakmaz, O. Oxidative stress is associated with not only coronary artery disease on statin therapy but also diabetes mellitus and hypertension. Indian Heart J. 2016, 68, 194–195. [Google Scholar] [CrossRef] [PubMed]
- Bandeira, S.D.M.; Guedes, G.D.S.; Fonseca, L.J.S.D.; Pires, A.S.; Gelain, D.P.; Moreira, J.C.F.; Goulart, M.O.F. Characterization of blood oxidative stress in type 2 diabetes mellitus patients: Increase in lipid peroxidation and SOD activity. Oxid. Med. Cell. Longev. 2012, 2012, 819310. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Jin, X.; Lam, C.W.K.; Yan, S.K. Oxidative stress and diabetes mellitus. Clin. Chem. Lab. Med. 2011, 49, 1773–1782. [Google Scholar] [CrossRef] [PubMed]
- Kowald, A.; Hamann, A.; Zintel, S.; Ullrich, S.; Klipp, E.; Osiewacz, H.D. A systems biological analysis links ROS metabolism to mitochondrial protein quality control. Mech. Ageing Dev. 2012, 133, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Yu, M.; Li, M.; Zhao, R.; Zhu, Q.; Zhou, W.; Zhao, W. Polymorphic variations in manganese superoxide dismutase (MnSOD), glutathione peroxidase-1 (GPX1), and catalase (CAT) contribute to elevated plasma triglyceride levels in Chinese patients with type 2 diabetes or diabetic cardiovascular disease. Mol. Cell. Biochem. 2012, 363, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Xi, W.; Zhang, Y.; Sun, Y.; Shen, Y.; Ye, X.; Zhou, Z. Phenolic composition of Chinese wild mandarin (Citrus reticulata Balnco.) pulps and their antioxidant properties. Ind. Crops Prod. 2014, 52, 466–474. [Google Scholar] [CrossRef]
- Liu, Y.; Qian, C.; Ding, S.; Shang, X.; Yang, W.; Fang, S. Effect of light regime and provenance on leaf characteristics, growth and flavonoid accumulation in Cyclocarya paliurus (Batal) Iljinskaja coppices. Bot. Stud. 2016, 57, 28. [Google Scholar] [CrossRef] [PubMed]
- Bessada, S.M.; Barreira, J.C.; Barros, L.; Ferreira, I.C.; Oliveira, M.B.P. Phenolic profile and antioxidant activity of Coleostephus myconis (L.) Rchb. f.: An underexploited and highly disseminated species. Ind. Crops Prod. 2016, 89, 45–51. [Google Scholar] [CrossRef]
- Li, X.C.; Fu, X.X.; Shang, X.L.; Yang, W.X.; Fang, S.Z. Natural population structure and genetic differentiation for heterodicogamous plant: Cyclocarya paliurus (Batal.) Iljinskaja (Juglandaceae). Tree Genet. Genomes 2017, 13, 80. [Google Scholar] [CrossRef]
- Sosa, T.; Alías, J.C.; Escudero, J.C.; Chaves, N. Interpopulational variation in the flavonoid composition of Cistus ladanifer L. exudate. Biochem. Syst. Ecol. 2005, 33, 353–364. [Google Scholar] [CrossRef]
- Contreras-Calderón, J.; Calderón-Jaimes, L.; Guerra-Hernández, E.; García-Villanova, B. Antioxidant capacity, phenolic content and vitamin C in pulp, peel and seed from 24 exotic fruits from Colombia. Food Res. Int. 2011, 44, 2047–2053. [Google Scholar] [CrossRef]
- Fatiha, B.; Didier, H.; Naima, G.; Khodir, M.; Martin, K.; Léocadie, K.; Pierre, D. Phenolic composition, in vitro antioxidant effects and tyrosinase inhibitory activity of three Algerian Mentha species: M. spicata (L.), M. pulegium (L.) and M. rotundifolia (L.) Huds (Lamiaceae). Ind. Crops Prod. 2015, 74, 722–730. [Google Scholar] [CrossRef]
- Huang, W.; Xue, A.; Niu, H.; Jia, Z.; Wang, J. Optimised ultrasonic-assisted extraction of flavonoids from Folium eucommiae and evaluation of antioxidant activity in multi-test systems in vitro. Food Chem. 2009, 114, 1147–1154. [Google Scholar] [CrossRef]
- Wang, L.; Yang, X.; Qin, P.; Shan, F.; Ren, G. Flavonoid composition, antibacterial and antioxidant properties of tartary buckwheat bran extract. Ind. Crops Prod. 2013, 49, 312–317. [Google Scholar] [CrossRef]
- Tohidi, B.; Rahimmalek, M.; Arzani, A. Essential oil composition, total phenolic, flavonoid contents, and antioxidant activity of Thymus species collected from different regions of Iran. Food Chem. 2017, 220, 153–161. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Huang, B.; Ban, X.; Tian, J.; Zhu, L.; Wang, Y. In vitro and in vivo antioxidant activity of the ethanolic extract from Meconopsis quintuplinervia. J. Ethnopharmacol. 2012, 141, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S. Effects of soy protein and genistein on blood glucose, antioxidant enzyme activities, and lipid profile in streptozotocin-induced diabetic rats. Life Sci. 2006, 79, 1578–1584. [Google Scholar] [CrossRef] [PubMed]
- Boots, A.W.; Haenen, G.R.; Bast, A. Health effects of quercetin: From antioxidant to nutraceutical. Eur. J. Pharmacol. 2008, 585, 325–337. [Google Scholar] [CrossRef] [PubMed]
- Calderon-Montano, J.M.; Burgos-Morón, E.; Pérez-Guerrero, C.; López-Lázaro, M. A review on the dietary flavonoid kaempferol. Mini-Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Muzashvili, T.S.; Georgiev, M.I. Advances in the biotechnological glycosylation of valuable flavonoids. Biotechnol. Adv. 2014, 32, 1145–1156. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Fang, S.Z.; Zhou, M.M.; Shang, X.L.; Yang, W.X.; Fu, X.X. Geographic variation in water-soluble polysaccharide content and antioxidant activities of Cyclocarya paliurus leaves. Ind. Crops Prod. 2018, 121, 180–186. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Oyaizu, M. Antioxidative activity of browning products of glucosamine fractionated by organic solvent and thin layer chromatography. Nippon Shokuhin Kogyo Gakkaishi 1986, 35, 771–775. [Google Scholar] [CrossRef]
- Wang, Q.; Jiang, C.; Fang, S.; Wang, J.; Ji, Y.; Shang, X.; Zhang, J. Antihyperglycemic, antihyperlipidemic and antioxidant effects of ethanol and aqueous extracts of Cyclocarya paliurus leaves in type 2 diabetic rats. J. Ethnopharmacol. 2013, 150, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 4-O-caffeoylquinic acid, 3-O-caffeoylquinic acid, quercetin-3-O-glucuronide, isoquercitrin, quercetin-3-O-galactoside, kaempferol-3-O-glucuronide, kaempferol-3-O-glucoside, kaempferol-3-O-rhamnoside, quercetin-3-O-rhamnoside, and 4, 5-di-O-caffeoylquinic acid are available from the authors. |
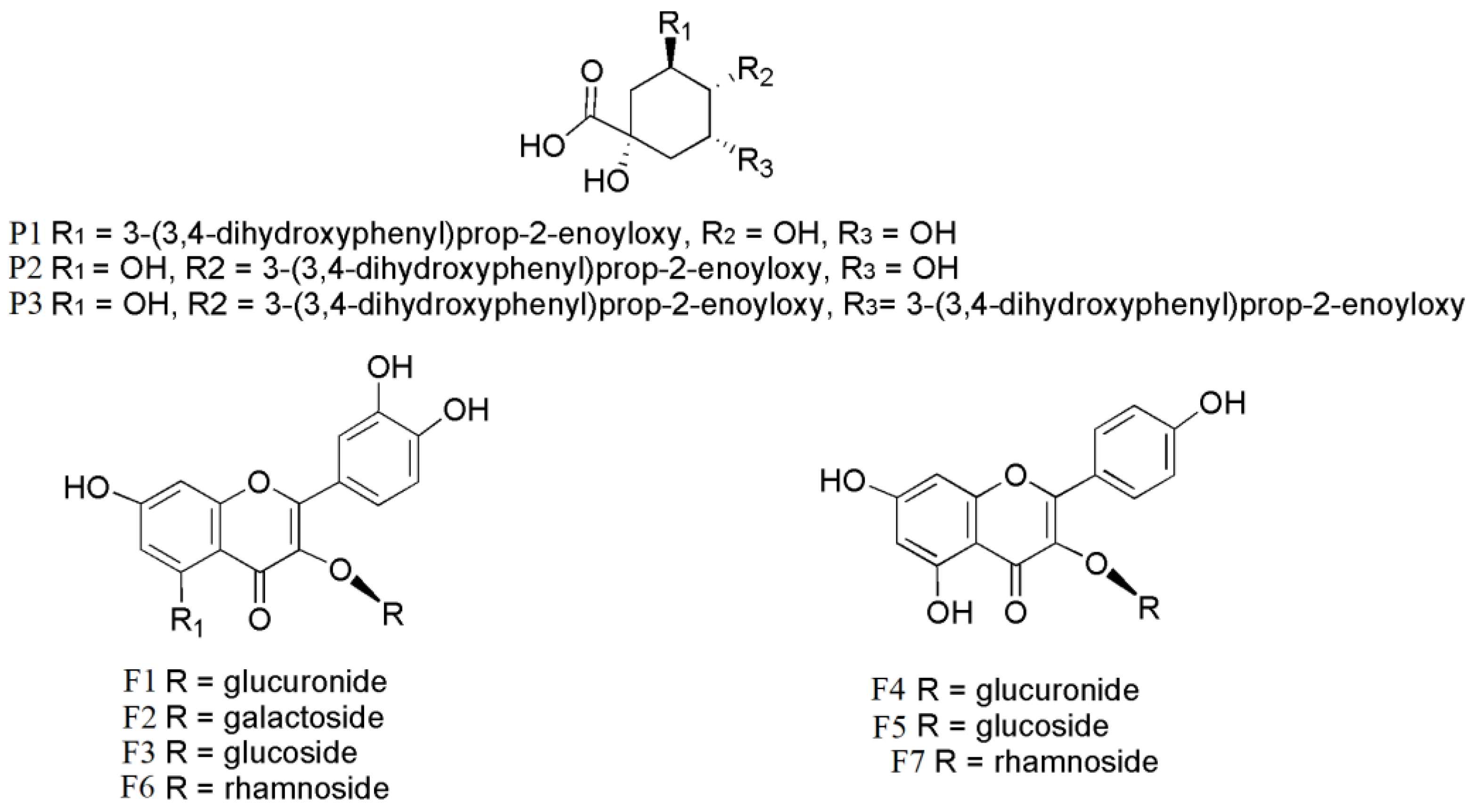
| Compounds | tR (min) | [M − H]− | MS/MS Fragment Ion (m/z) | Formula |
|---|---|---|---|---|
| 3-O-caffeoylquinic acid (P1) | 7.4 | 353.9874 | 191.0554 | C16H8O9 |
| 4-O-caffeoylquinic acid (P2) | 7.8 | 353.0875 | 191.0552; 179.0339; 135.0444 | C16H8O9 |
| quercetin-3-O-glucuronide (F1) | 14.9 | 477.0673 | 301.0351; 178.9979; 151.003 | C21H18O13 |
| quercetin-3-O-galactoside (F2) | 15.4 | 463.0888 | 301.0343; 271.0244; 178.9979; 151.0029 | C21H20O12 |
| isoquercitrin (F3) | 15.8 | 463.0888 | 301.0338; 271.0239; 178.9981; 151.0029 | C21H20O12 |
| kaempferol-3-O-glucuronide (F4) | 17.6 | 461.9727 | 286.0431; 285.0401; 113.0235; 85.0296 | C21H18O12 |
| kaempferol-3-O-glucoside (F5) | 18.5 | 447.0935 | 285.0388; 284.0319; 255.0295 | C21H20O11 |
| quercetin-3-O-rhamnoside (F6) | 18.8 | 447.0973 | 301.0337; 285.0388; 255.0294; 217.0133 | C21H20O11 |
| 4,5-di-O-caffeoylquinic acid (P3) | 19.8 | 515.1199 | 353.0864; 191.0553; 179.0342; 135.0443 | C25H24O12 |
| kaempferol-3-O-rhamnoside (F7) | 23.4 | 431.0981 | 285.0394; 255.0295; 227.0342 | C21H20O10 |
| Compounds | LOD (ng/mL) | LOQ (ng/mL) | JZS (mg/g) | MC (mg/g) | WF (mg/g) | MW (mg/g) | SN (mg/g) |
|---|---|---|---|---|---|---|---|
| 3-O-caffeoylquinic acid (P1) | 64.25 | 214.21 | 0.35 ± 0.011b | 1.38 ± 0.153a | 0.39 ± 0.003b | 0.45 ± 0.028b | 0.31 ± 0.003b |
| 4-O-caffeoylquinic acid (P2) | 57.93 | 197.58 | 0.03 ± 0.001d | 0.08 ± 0.003c | 0.28 ± 0.003a | 0.11 ± 0.003b | 0.08 ± 0.002c |
| 4,5-di-O-caffeoylquinic acid (P3) | 58.97 | 201.22 | 0.05 ± 0.003d | 0.10 ± 0.001c | 0.42 ± 0.028a | 0.16 ± 0.003b | 0.08 ± 0.003cd |
| quercetin-3-O-glucuronide (F1) | 40.28 | 128.74 | 2.15 ± 0.010b | 1.57 ± 0.006d | 2.11 ± 0.003c | 2.38 ± 0.005a | 0.78 ± 0.003e |
| quercetin-3-O-galactoside (F2) | 52.94 | 174.17 | 0.47 ± 0.005c | 0.24 ± 0.003d | 0.52 ± 0.003b | 0.57 ± 0.005a | 0.20 ± 0.003e |
| isoquercitrin (F3) | 58.42 | 192.52 | 0.22 ± 0.005b | 0.32 ± 0.006a | 0.18 ± 0.006c | 0.21 ± 0.013b | 0.08 ± 0.001d |
| kaempferol-3-O-glucuronide (F4) | 43.98 | 153.14 | 1.33 ± 0.001b | 0.86 ± 0.001d | 1.88 ± 0.001a | 0.95 ± 0.001c | 0.51 ± 0.001e |
| kaempferol-3-O-glucoside (F5) | 53.85 | 187.37 | 0.18 ± 0.001b | 0.20 ± 0.001a | 0.12 ± 0.001c | 0.08 ± 0.001d | 0.05 ± 0.001e |
| quercetin-3-O-rhamnoside (F6) | 62.48 | 199.32 | 0.23 ± 0.001a | 0.11 ± 0.001d | 0.20 ± 0.001b | 0.19 ± 0.001c | nd |
| kaempferol-3-O-rhamnoside (F7) | 64.13 | 211.81 | 2.30 ± 0.001a | 1.39 ± 0.001d | 1.78 ± 0.001b | 1.50 ± 0.006c | 0.68 ± 0.001e |
| total phenolic acids (TPA) | - | - | 0.43 ± 0.014c | 1.57 ± 0.154a | 1.09 ± 0.023b | 0.71 ± 0.034c | 0.47 ± 0.007c |
| total flavonoids (TF) | - | - | 6.88 ± 0.011a | 4.70 ± 0.003d | 6.80 ± 0.006b | 5.90 ± 0.008c | 2.30 ± 0.003e |
| total phenolics (TPC) | - | - | 7.31 ± 0.025b | 6.27 ± 0.152d | 7.89 ± 0.017a | 6.61 ± 0.042c | 2.77 ± 0.008e |
| Assays | DPPH (IC50) | FRAP (EC50) | SOD | GSH-px | MDA |
|---|---|---|---|---|---|
| total phenolic compounds (TPC) | −0.991 ** | −0.796 | 0.775 | 0.528 | −0.878 * |
| total phenolic acids (TPA) | −0.373 | −0.732 | 0.010 | 0.249 | −0.090 |
| total flavonoids (TF) | −0.950 * | −0.655 | 0.814 | 0.493 | −0.903 * |
| 3-O-caffeoylquinic acid (P1) | −0.195 | −0.452 | −0.182 | 0.419 | 0.070 |
| 4-O-caffeoylquinic acid (P2) | −0.312 | −0.553 | 0.314 | −0.323 | −0.256 |
| 4,5-di-O-caffeoylquinic acid (P3) | −0.392 | −0.597 | 0.378 | −0.266 | −0.331 |
| quercetin-3-O-glucuronide (F1) | −0.898 * | −0.700 | 0.559 | 0.287 | −0.698 |
| quercetin-3-O-galactoside (F2) | −;0.716 | −0.530 | 0.500 | −0.004 | −0.584 |
| isoquercitrin (F3) | −0.687 | −0.698 | 0.242 | 0.684 | −0.413 |
| kaempferol-3-O-glucuronide (F4) | −0.768 | −0.584 | 0.871 | 0.312 | −0.855 |
| kaempferol-3-O-glucoside (F5) | −0.654 | −0.420 | 0.560 | 0.955 * | −0.655 |
| quercetin-3-O-rhamnoside (F6) | −0.939 * | −0.639 | 0.735 | 0.467 | −0.848 |
| kaempferol-3-O-rhamnoside (F7) | −0.874 | −0.426 | 0.883 * | 0.716 | −0.954 * |
| Sample ID | Location | Latitude (N) | Longitude (E) | Mean Annual Temperature (°C) | Altitude (m) | Mean Annual Sunshine Hours (h) | Mean Annual Precipitation (mm) |
|---|---|---|---|---|---|---|---|
| JZS | Jinzhongshan, Guangxi | 24°36′36″ | 104°57′00″ | 17.1 | 1798 | 1475 | 1200 |
| MC | Muchuan, Sichuan | 28°58′00″ | 103°47′00″ | 12.9 | 1200 | 965.3 | 1533 |
| WF | Wufeng, Hubei | 30°17′00″ | 110°80′00″ | 16.7 | 688 | 1533 | 1893 |
| MW | Meiwu, Zhejiang | 27°46′00″ | 119°17′00″ | 16.5 | 678 | 1862 | 1600 |
| SN | Suining, Hunan | 26°22′00″ | 110°07′00″ | 16.7 | 862 | 1348 | 1320 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Chen, P.; Zhou, M.; Wang, T.; Fang, S.; Shang, X.; Fu, X. Geographic Variation in the Chemical Composition and Antioxidant Properties of Phenolic Compounds from Cyclocarya paliurus (Batal) Iljinskaja Leaves. Molecules 2018, 23, 2440. https://doi.org/10.3390/molecules23102440
Liu Y, Chen P, Zhou M, Wang T, Fang S, Shang X, Fu X. Geographic Variation in the Chemical Composition and Antioxidant Properties of Phenolic Compounds from Cyclocarya paliurus (Batal) Iljinskaja Leaves. Molecules. 2018; 23(10):2440. https://doi.org/10.3390/molecules23102440
Chicago/Turabian StyleLiu, Yang, Pei Chen, Mingming Zhou, Tongli Wang, Shengzuo Fang, Xulan Shang, and Xiangxiang Fu. 2018. "Geographic Variation in the Chemical Composition and Antioxidant Properties of Phenolic Compounds from Cyclocarya paliurus (Batal) Iljinskaja Leaves" Molecules 23, no. 10: 2440. https://doi.org/10.3390/molecules23102440
APA StyleLiu, Y., Chen, P., Zhou, M., Wang, T., Fang, S., Shang, X., & Fu, X. (2018). Geographic Variation in the Chemical Composition and Antioxidant Properties of Phenolic Compounds from Cyclocarya paliurus (Batal) Iljinskaja Leaves. Molecules, 23(10), 2440. https://doi.org/10.3390/molecules23102440