Preparative Purification of Polyphenols from Aronia melanocarpa (Chokeberry) with Cellular Antioxidant and Antiproliferative Activity
Abstract
:1. Introduction
2. Results
2.1. Purification of Polyphenols by Adsorption
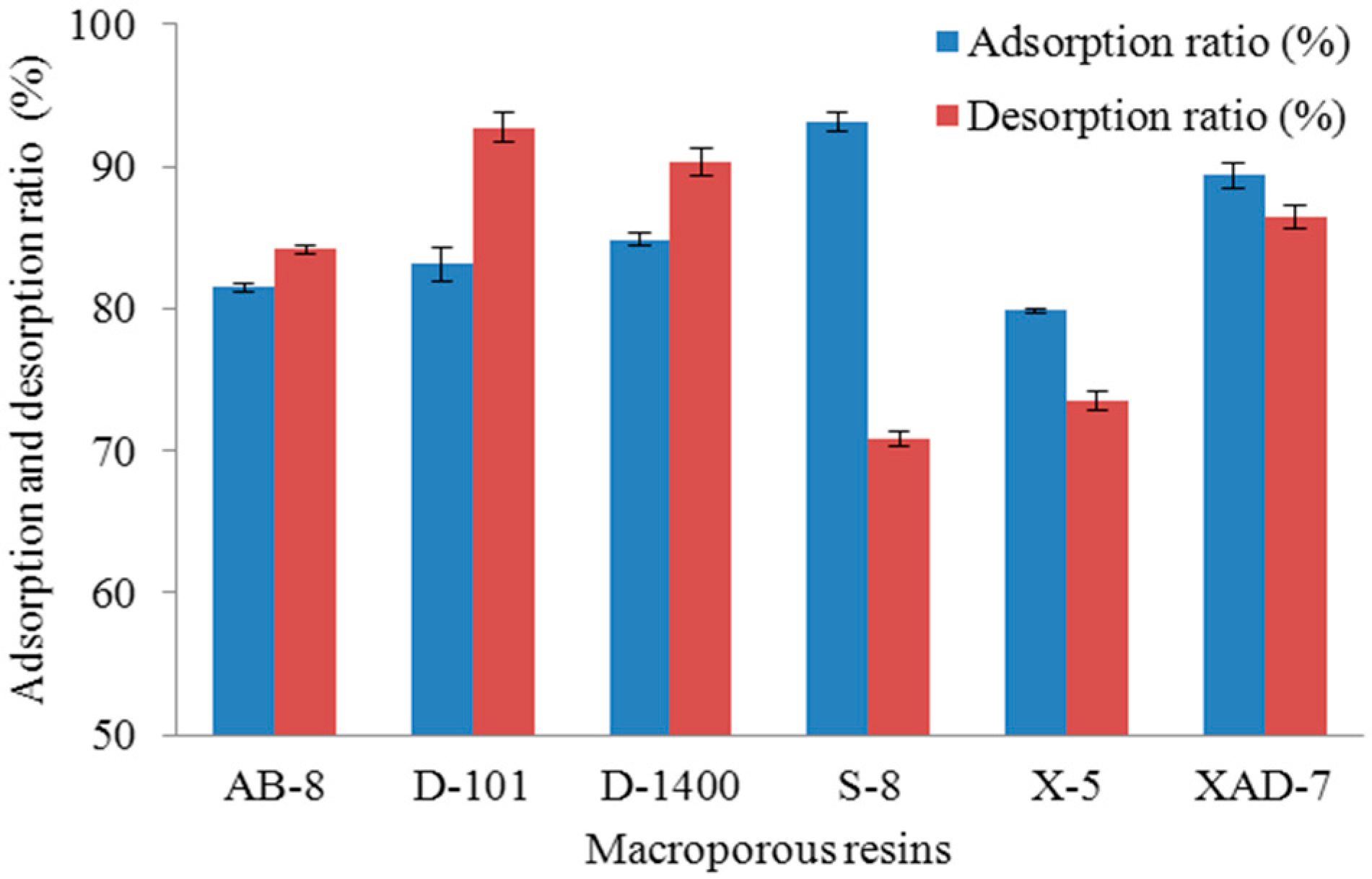
2.1.1. Screening of Suitable Adsorbents
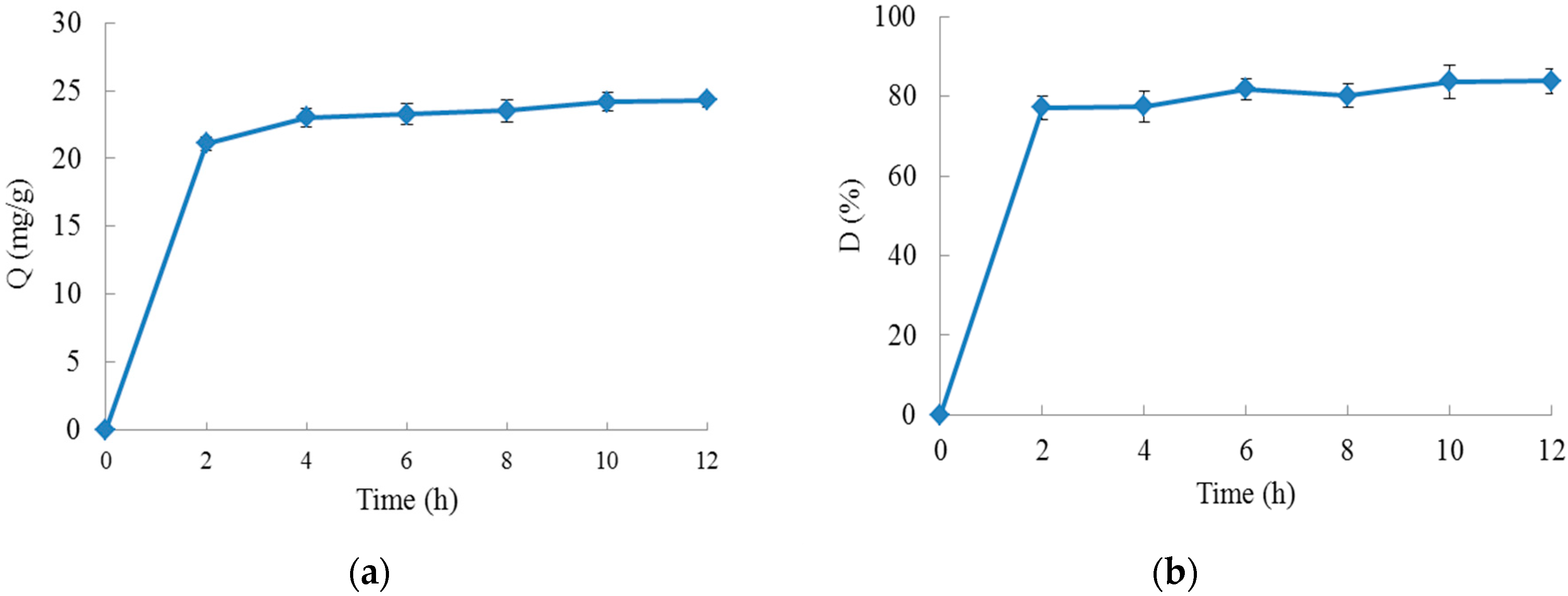
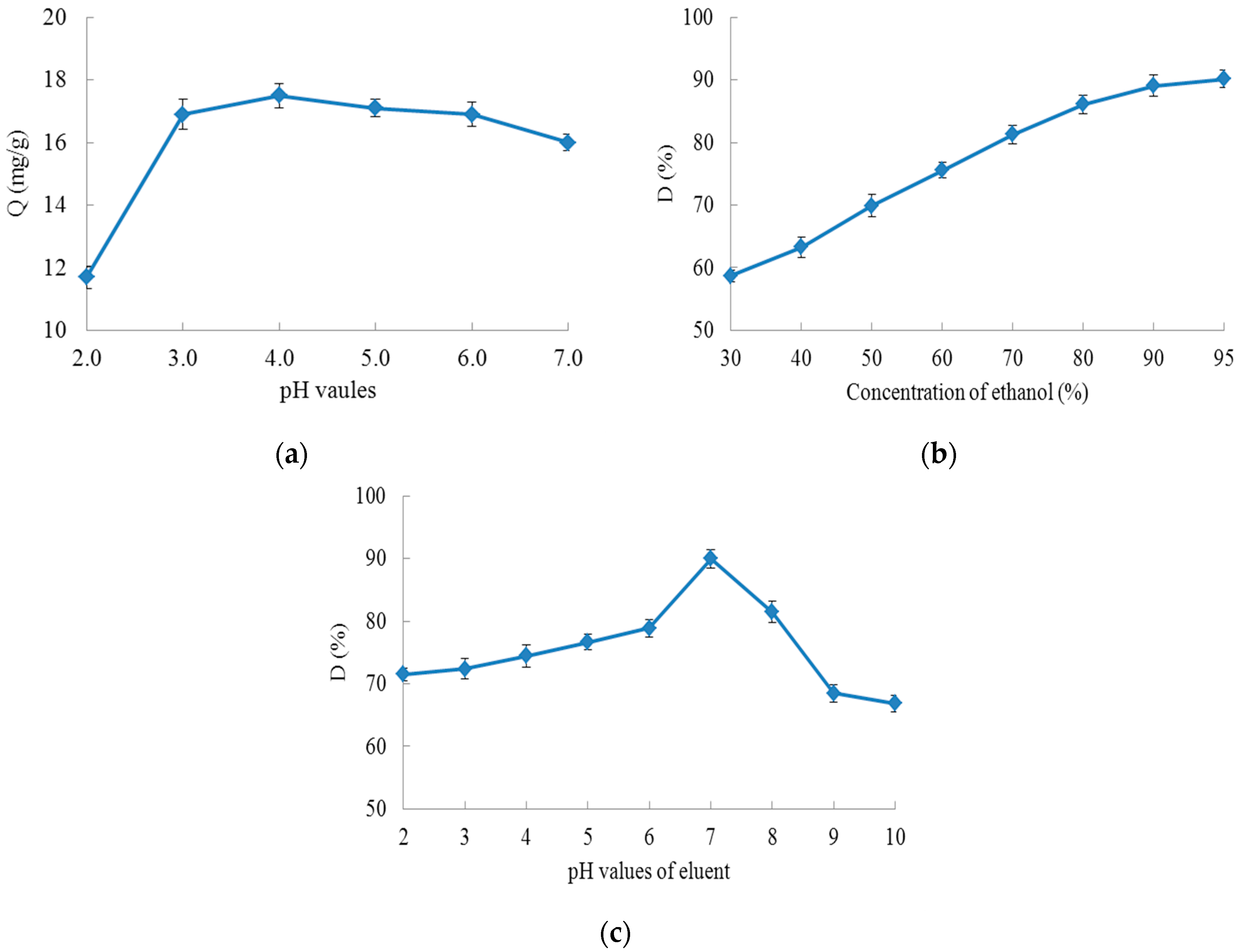
2.1.2. Static Adsorption and Desorption
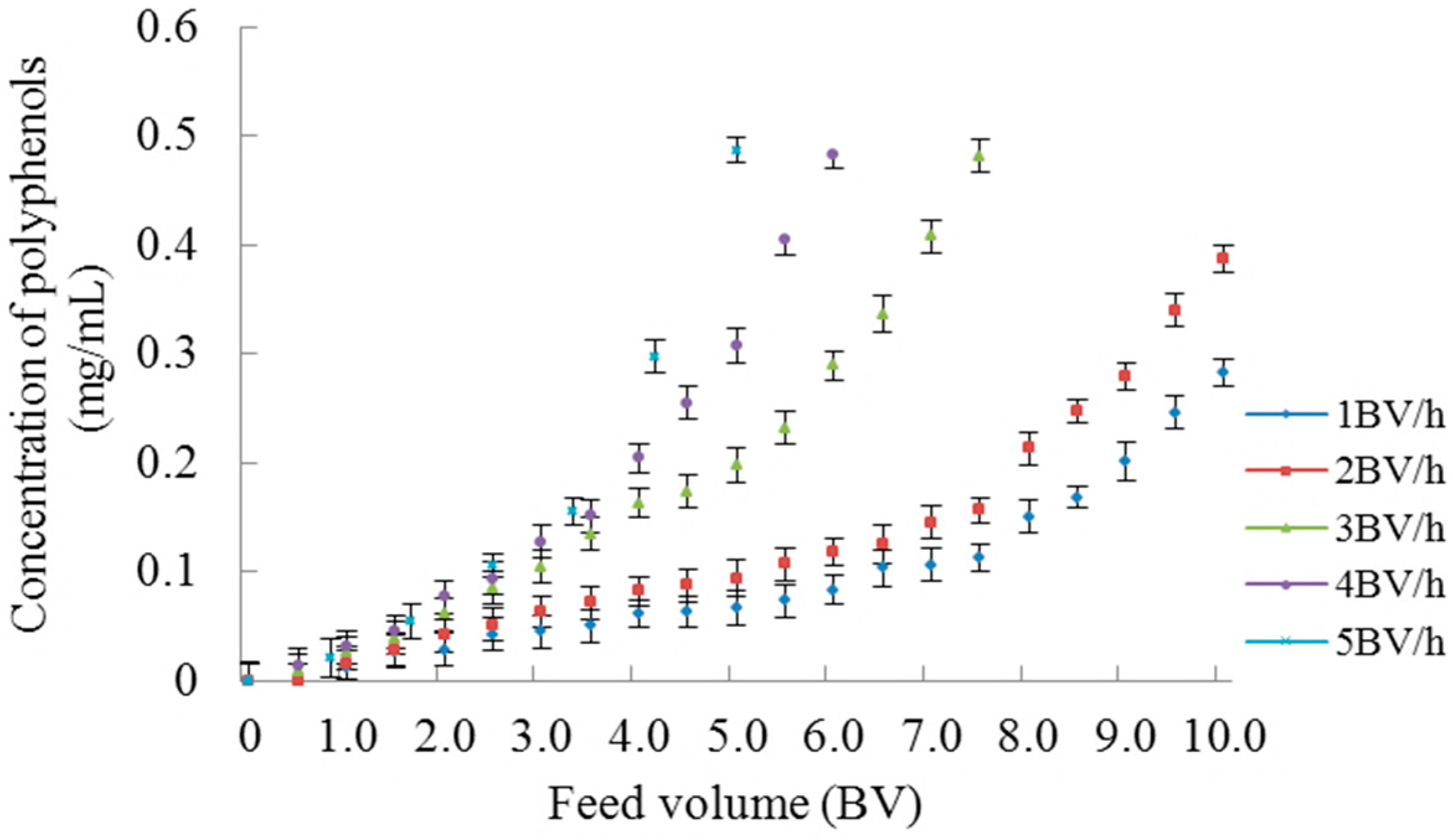
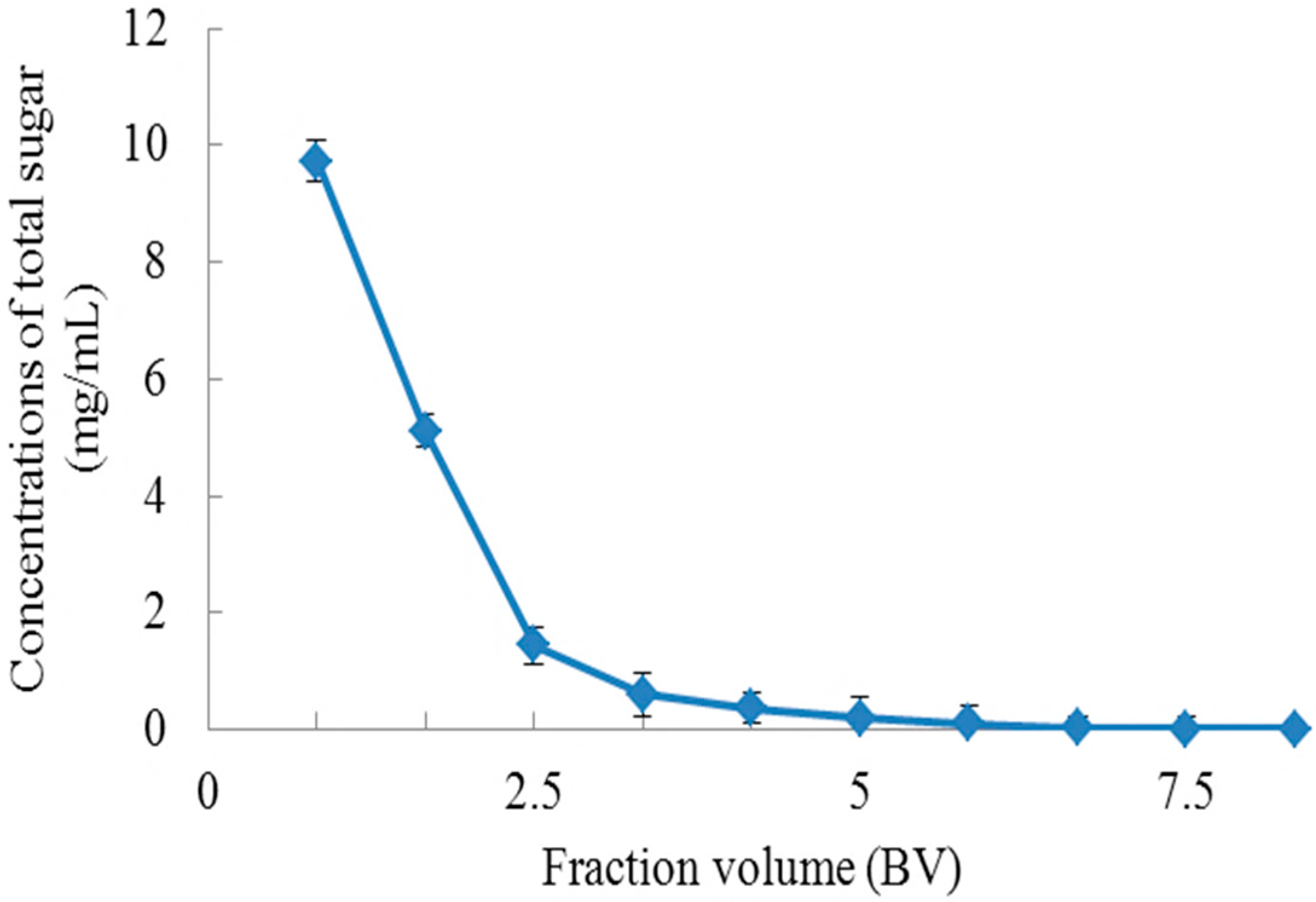
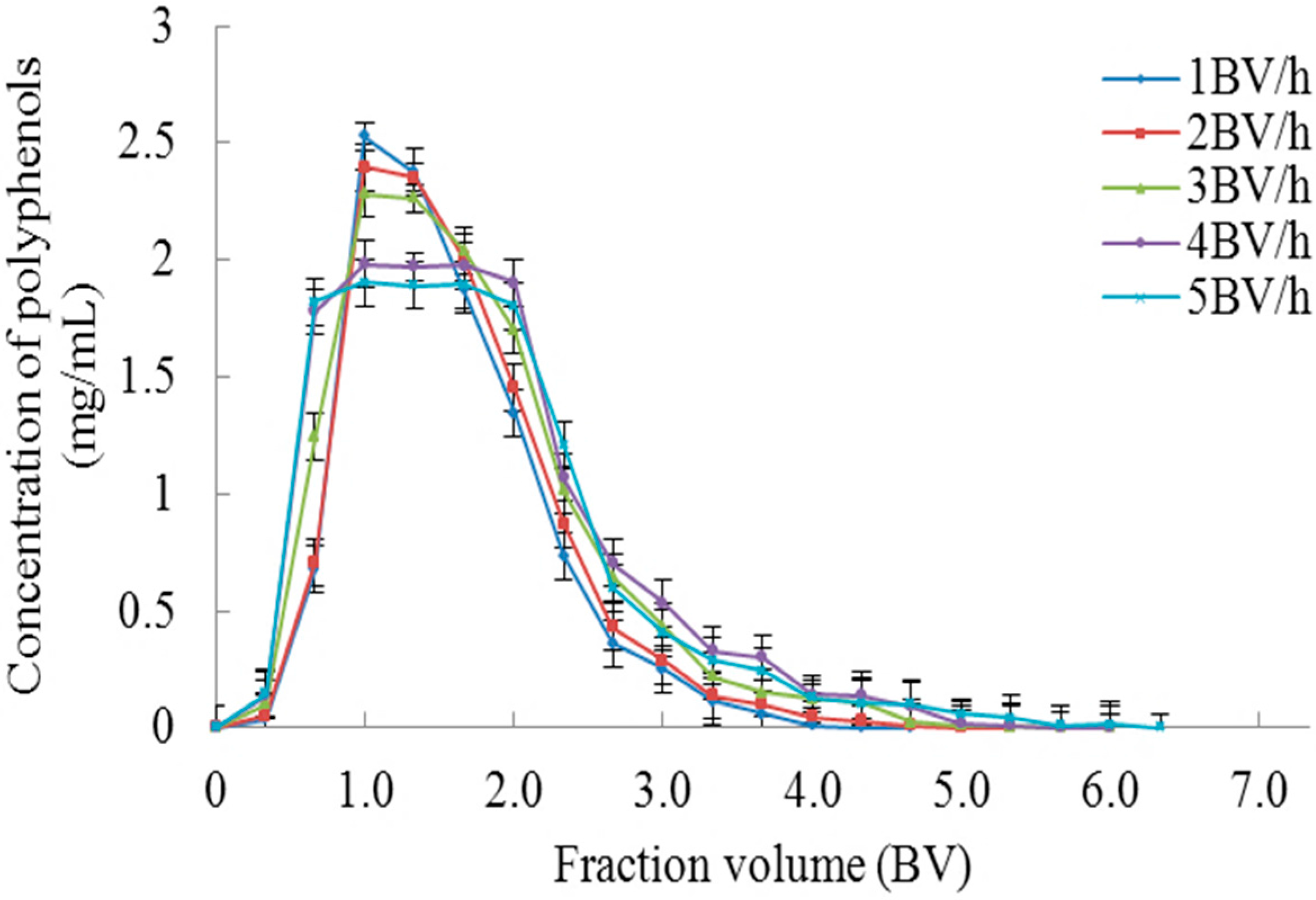
2.1.3. Dynamic Adsorption and Desorption
2.2. Cellular Antioxidant Activity
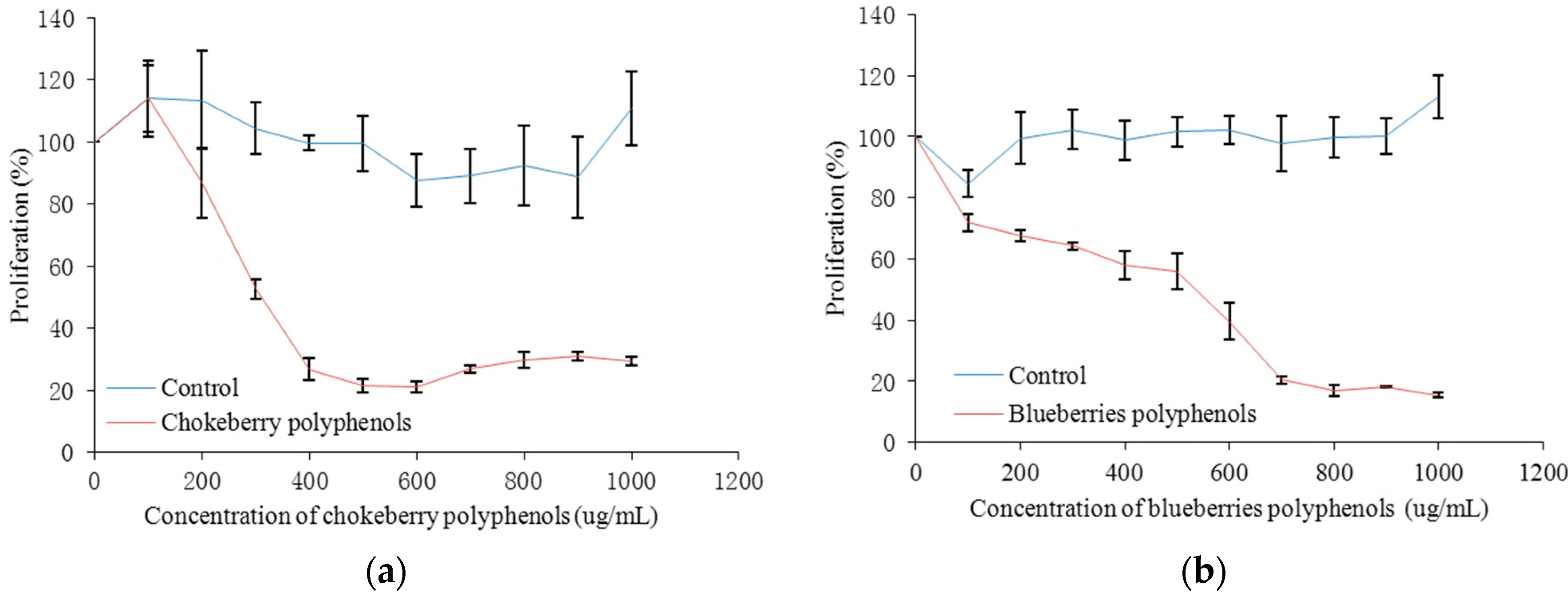
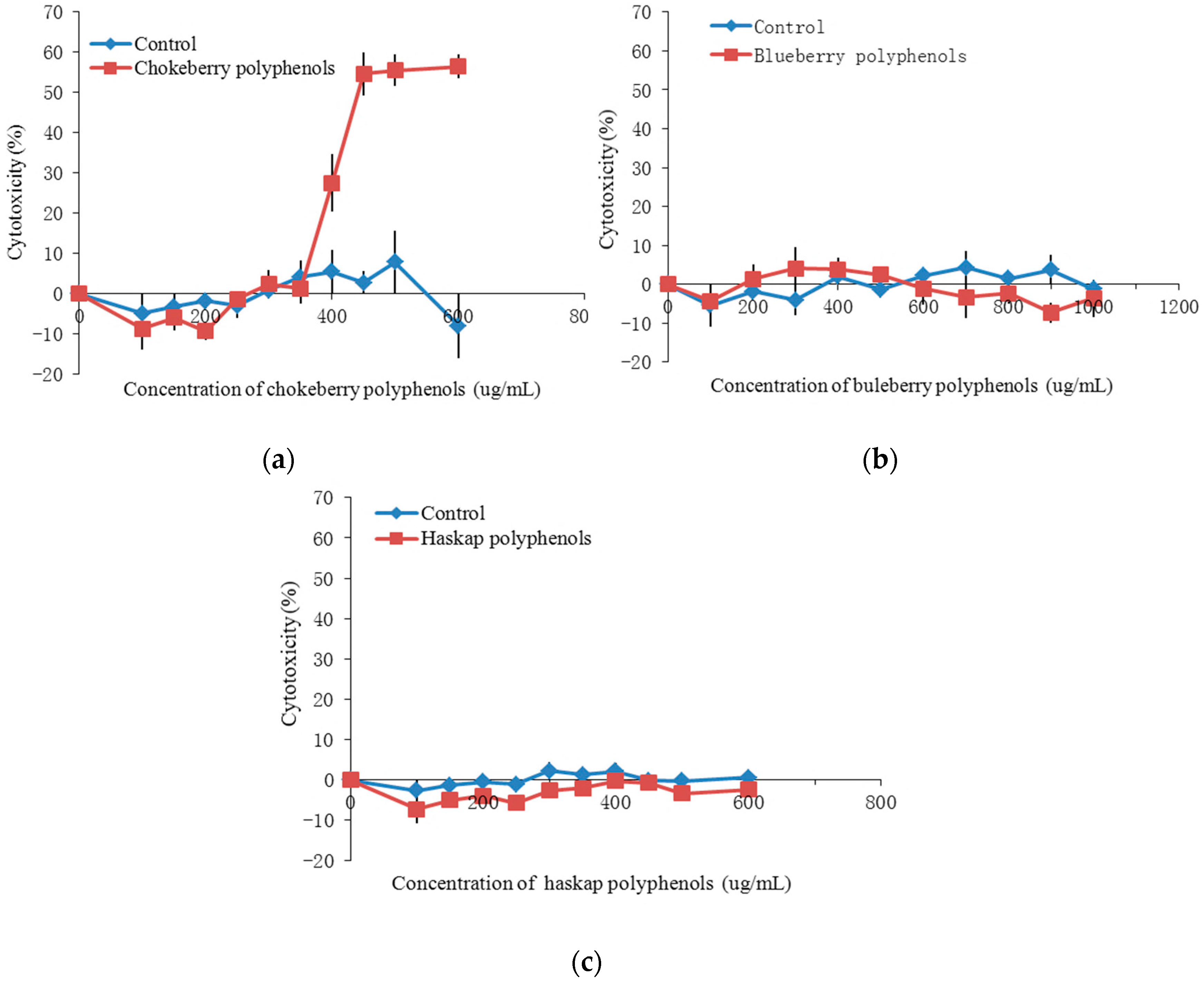
2.3. Antiproliferation Activity and Cytotoxicity
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Extraction of Polyphenols from Chokeberry
4.3. Determination of Total Polyphenols
4.4. Purification of Polyphenols by Adsorption
4.4.1. Adsorbents Preparation
4.4.2. Screening of Suitable Adsorbents
4.4.3. Static Adsorption and Desorption Tests
4.4.4. Dynamic Adsorption and Desorption Tests
4.5. Cytotoxicity Measurement
4.6. Cellular Antioxidant Activity
4.7. Cellular of Antiproliferative Activity
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Benvenuti, S.; Pellati, F.; Melegari, M.; Bertelli, D. Polyphenols, anthocyanins, ascorbic acid, and radical scavenging activity of Rubus, Ribes, and Aronia. J. Food Sci. 2004, 69, FCT164–FCT169. [Google Scholar] [CrossRef]
- Šavikin, K.; Zdunić, G.; Janković, T.; Gođevac, D.; Stanojković, T.; Pljevljakušić, D. Berry fruit teas: Phenolic composition and cytotoxic activity. Food Res. Int. 2014, 62, 677–683. [Google Scholar] [CrossRef]
- Mirosława, T.; Aneta, W. Comparison of phenolic compounds and antioxidant potential between selected edible fruits and their leaves. J. Funct. Foods 2015, 14, 736–746. [Google Scholar] [CrossRef]
- Stanisavljević, N.; Samardžić, J.; Janković, T.; Šavikin, K.; Mojsin, M.; Topalović, V.; Stevanović, M. Antioxidant and antiproliferative activity of chokeberry juice phenolics during in vitro simulated digestion in the presence of food matrix. Food Chem. 2015, 175, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Kosmala, M.; Zduńczyk, Z.; Karlińska, E.; Juśkiewicz, J. The effects of strawberry, black currant, and chokeberry extracts in a grain dietary fiber matrix on intestinal fermentation in rats. Food Res. Int. 2014, 64, 752–761. [Google Scholar] [CrossRef]
- Valcheva-Kuzmanova, S.; Kuzmanov, K.; Tsanova-Savova, S.; Mihova, V.; Krasnaliev, I.; Borisova, P.; Belcheva, A. Lipid-lowering effects of Aronia melanocarpa fruit juice in rats fed cholesterol-containing diets. J. Food Biochem. 2007, 31, 589–602. [Google Scholar] [CrossRef]
- Denev, P.; Kratchanova, M.; Ciz, M.; Lojek, A.; Vasicek, O.; Nedelcheva, P.; Blazheva, D.; Toshkova, R.; Gardeva, E.; Yossifova, L. Biological activities of selected polyphenol-rich fruits related to immunity and gastrointestinal health. Food Chem. 2014, 157, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Oszmiański, J.; Wojdylo, A. Aronia melanocarpa phenolics and their antioxidant activity. Eur. Food Res. Technol. 2005, 221, 809–813. [Google Scholar] [CrossRef]
- Grunovaitė, L.; Pukalskienė, M.; Pukalskas, A.; Venskutonis, P.R. Fractionation of black chokeberry pomace into functional ingredients using high pressure extraction methods and evaluation of their antioxidant capacity and chemical composition. J. Funct. Foods 2016, 24, 85–96. [Google Scholar] [CrossRef]
- Wangensteen, H.; Bräunlich, M.; Nikolic, V.; Malterud, K.E.; Slimestad, R.; Barsett, H. Anthocyanins, proanthocyanidins and total phenolics in four cultivars of aronia: Antioxidant and enzyme inhibitory effects. J. Funct. Foods 2014, 7, 746–752. [Google Scholar] [CrossRef]
- Gironés-Vilaplana, A.; Valentão, P. Phytochemical profile of a blend of black chokeberry and lemon juice with cholinesterase inhibitory effect and antioxidant potential. Food Chem. 2012, 134, 2090–2096. [Google Scholar] [CrossRef] [PubMed]
- Mezadri, T.; Villano, D. Antioxidant compounds and antioxidant activity in acerola (Malpighia emarginata DC.) fruits and derivatives. J. Food Compos. Anal. 2008, 21, 282–290. [Google Scholar] [CrossRef]
- Hwang, S.J.; Yoon, W.B.; Lee, O.-H.; Cha, S.J.; Kim, J.D. Radical-scavenging-linked antioxidant activities of extracts from black chokeberry and blueberry cultivated in Korea. Food Chem. 2014, 146, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Ku, C.S.; Pham, T.X.; Park, Y.-K.; Martin, D.A.; Xie, L.-Y.; Taheri, R.; Lee, J.-Y.; Bolling, B.W. Aronia melanocarpa (chokeberry) polyphenol–rich extract improves antioxidant function and reduces total plasma cholesterol in apolipoprotein E knockout mice. Nutr. Res. 2013, 33, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Al-Hallaq, E.K.; Kasabri, V.; Abdalla, S.S.; Bustanji, Y.K.; Afifi, F.U. Anti-Obesity and Antihyperglycemic Effects of Crataegus aronia Extracts: In Vitro and in Vivo Evaluations. Food Nutr. Sci. 2013, 4, 972–983. [Google Scholar] [CrossRef]
- Slimestad, R.; Torskangerpoll, K. Flavonoids from black chokeberries, Aronia melanocarpa. J. Compos. Anal. 2005, 18, 61–68. [Google Scholar] [CrossRef]
- Sun, L.-J.; Guo, Y.-R.; Fu, C.-C.; Li, J.-J.; Li, Z. Simultaneous separation and purification of total polyphenols, chlorogenic acid and phlorizin from thinned young apples. Food Chem. 2013, 136, 1022–1029. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, J.; Farahmandazad, H.; Vuorinen, A.; Kallio, H.; Yang, B.; Sainio, T. Extraction and purification of anthocyanins from purple-fleshed potato. Food Bioprod. Process. 2016, 99, 136–146. [Google Scholar] [CrossRef]
- Jampani, C.; Naik, A.; Raghavarao, K.S.M.S. Purification of anthocyanins from jamun (Syzygiu cumini L.) employing adsorption. Sep. Purif. Technol. 2014, 125, 170–178. [Google Scholar] [CrossRef]
- Worsztynowicz, P.; Napierała, M.; Białasa, W.; Grajek, W.; Olkowicz, M. Pancreatic α-amylase and lipase inhibitory activity of polyphenolic compounds present in the extract of black chokeberry (Aronia melanocarpa L.). Process Biochem. 2014, 49, 1457–1463. [Google Scholar] [CrossRef]
- Ho, G.T.T.; Bräunlich, M.; Austarheim, I.; Wangensteen, H.; Malterud, K.E.; Slimestad, R.; Barsett, H. Immunomodulating Activity of Aronia melanocarpa Polyphenols. Int. J. Mol. Sci. 2014, 15, 11626–11636. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.A.; Taheri, R.; Brand, M.H.; Draghi, A., II; Sylvester, F.A.; Bolling, B.W. Anti-inflammatory activity of aronia berry extracts in murine splenocytes. J. Funct. Foods 2014, 8C, 68–75. [Google Scholar] [CrossRef]
- Selmeci, L.; Szekely, M.; Soos, P.; Seres, L.; Klinga, N.; Geiger, A.; Acsady, G. Human blood plasma advanced oxidation protein products (AOPP) correlates with fibrinogen levels. Free Radic. Res. 2006, 40, 952–958. [Google Scholar] [CrossRef] [PubMed]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric Oxide and Peroxynitrite in Health and Disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Saluk, J.; Antosik, A.; Ponczek, M.B.; Żbikowska, H.M. Aronia melanocarpa as a protector against nitration of fibrinogen. Int. J. Biol. Macromol. 2013, 55, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Li, B.; Liu, H.; Meng, X.; Jiao, X.; Lei, Y.; Zhang, Y.; Liu, Y.; Cheng, S.; Gao, F.; et al. Optimization of Extraction of Polyphenols from Aronia melanocarpa by Response Surface Methodology and Their Antioxidant Activity Evaluation. Sci. Technol. Food Ind. 2016, 19. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Ravento, R.M.; Lester, P. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Malta, L.G.; Tessaro, E.P.; Eberlin, M.; Pastore, G.M.; Liu, R.H. Assessment of antioxidant and antiproliferative activities and the identification of phenolic compounds of exotic Brazilian fruits. Food Res. Int. 2013, 53, 417–425. [Google Scholar] [CrossRef]
- Yang, J.; Martinson, T.E.; Liu, R.H. Phytochemical profiles and antioxidant activities of wine grapes. Food Chem. 2009, 116, 332–339. [Google Scholar] [CrossRef]
- Boaventura, B.C.B.; Amboni, R.D.M.C.; Silva, E.L.D.; Prudencio, E.S.; Pietro, P.F.D.; Malta, L.G.; Polinati, R.M.; Liu, R.H. Effect of in vitro digestion of yerba mate (Ilex paraguariensis A. St. Hil.) extract on the cellular antioxidant activity, antiproliferative activity and cytotoxicity toward HepG2 cells. Food Res. Int. 2015, 77, 257–263. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
| CAA (umol Q/100 ug) | EC50 (ug/mL) | IC50 (ug/mL) | |
|---|---|---|---|
| Chokeberry | 334.48 ± 29.57 | 0.069 ± 0.005 | 338.36 ± 23.17 |
| Blueberry | 357.64 ± 33.61 | 0.064 ± 0.006 | 359.74 ± 29.38 |
| Haskap | 252.87 ± 23.15 | 0.049 ± 0.003 | 552.07 ± 34.19 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, N.; Wang, Y.; Jiao, X.; Chou, S.; Li, E.; Li, B. Preparative Purification of Polyphenols from Aronia melanocarpa (Chokeberry) with Cellular Antioxidant and Antiproliferative Activity. Molecules 2018, 23, 139. https://doi.org/10.3390/molecules23010139
Gao N, Wang Y, Jiao X, Chou S, Li E, Li B. Preparative Purification of Polyphenols from Aronia melanocarpa (Chokeberry) with Cellular Antioxidant and Antiproliferative Activity. Molecules. 2018; 23(1):139. https://doi.org/10.3390/molecules23010139
Chicago/Turabian StyleGao, Ningxuan, Yuehua Wang, Xinyao Jiao, Shurui Chou, Enhui Li, and Bin Li. 2018. "Preparative Purification of Polyphenols from Aronia melanocarpa (Chokeberry) with Cellular Antioxidant and Antiproliferative Activity" Molecules 23, no. 1: 139. https://doi.org/10.3390/molecules23010139
APA StyleGao, N., Wang, Y., Jiao, X., Chou, S., Li, E., & Li, B. (2018). Preparative Purification of Polyphenols from Aronia melanocarpa (Chokeberry) with Cellular Antioxidant and Antiproliferative Activity. Molecules, 23(1), 139. https://doi.org/10.3390/molecules23010139




