Biosynthesis of Silver Nanoparticles on Orthodontic Elastomeric Modules: Evaluation of Mechanical and Antibacterial Properties
Abstract
:1. Introduction
2. Results
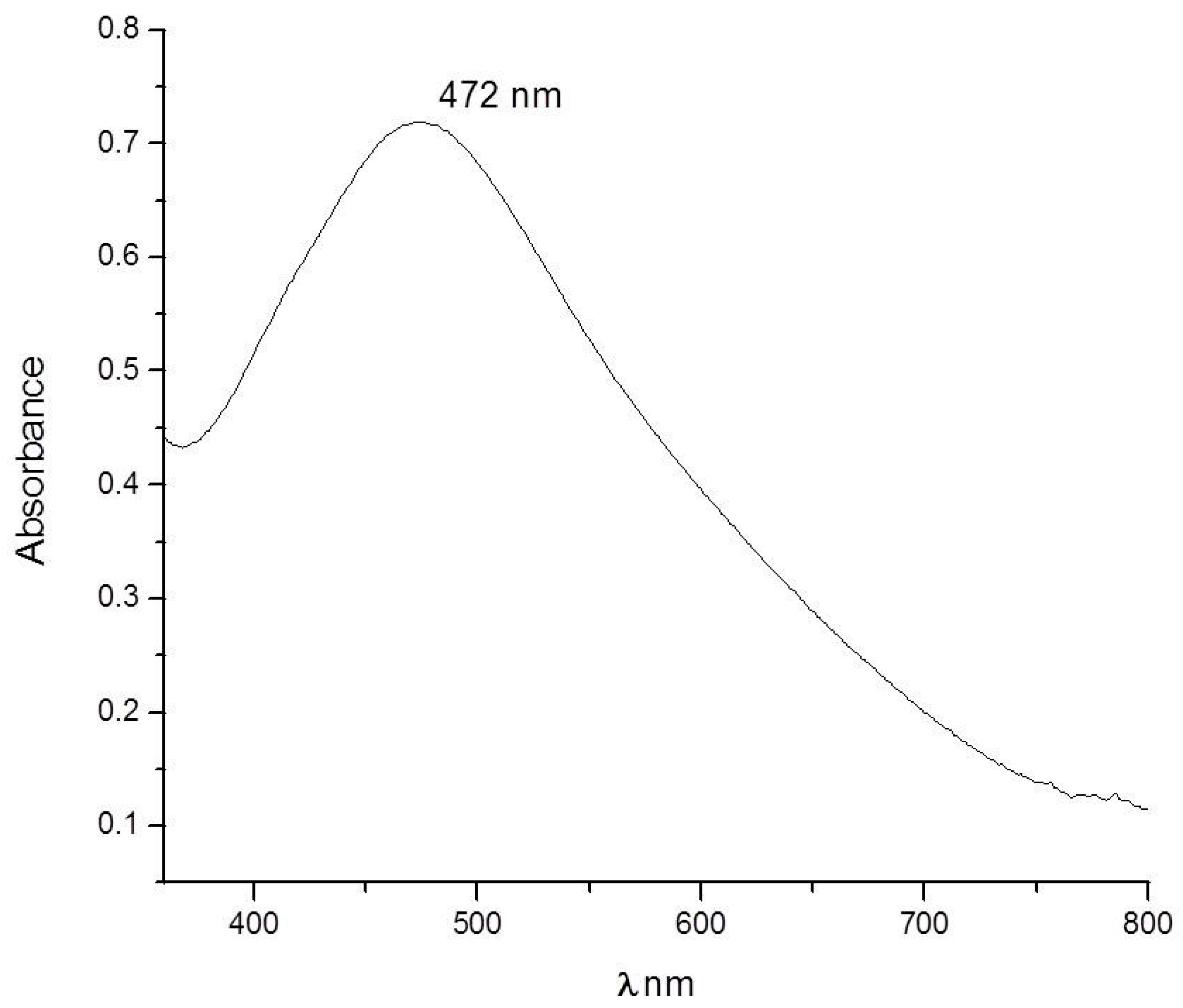
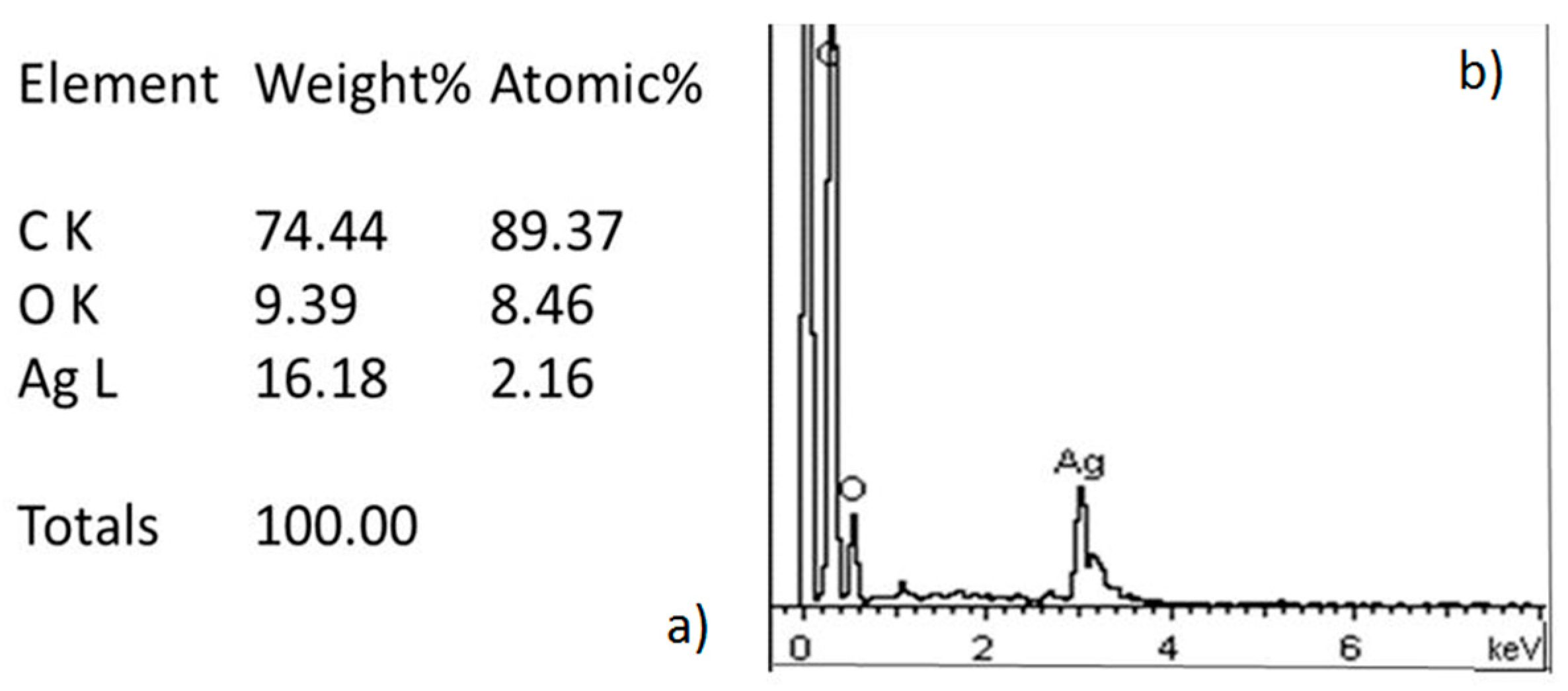
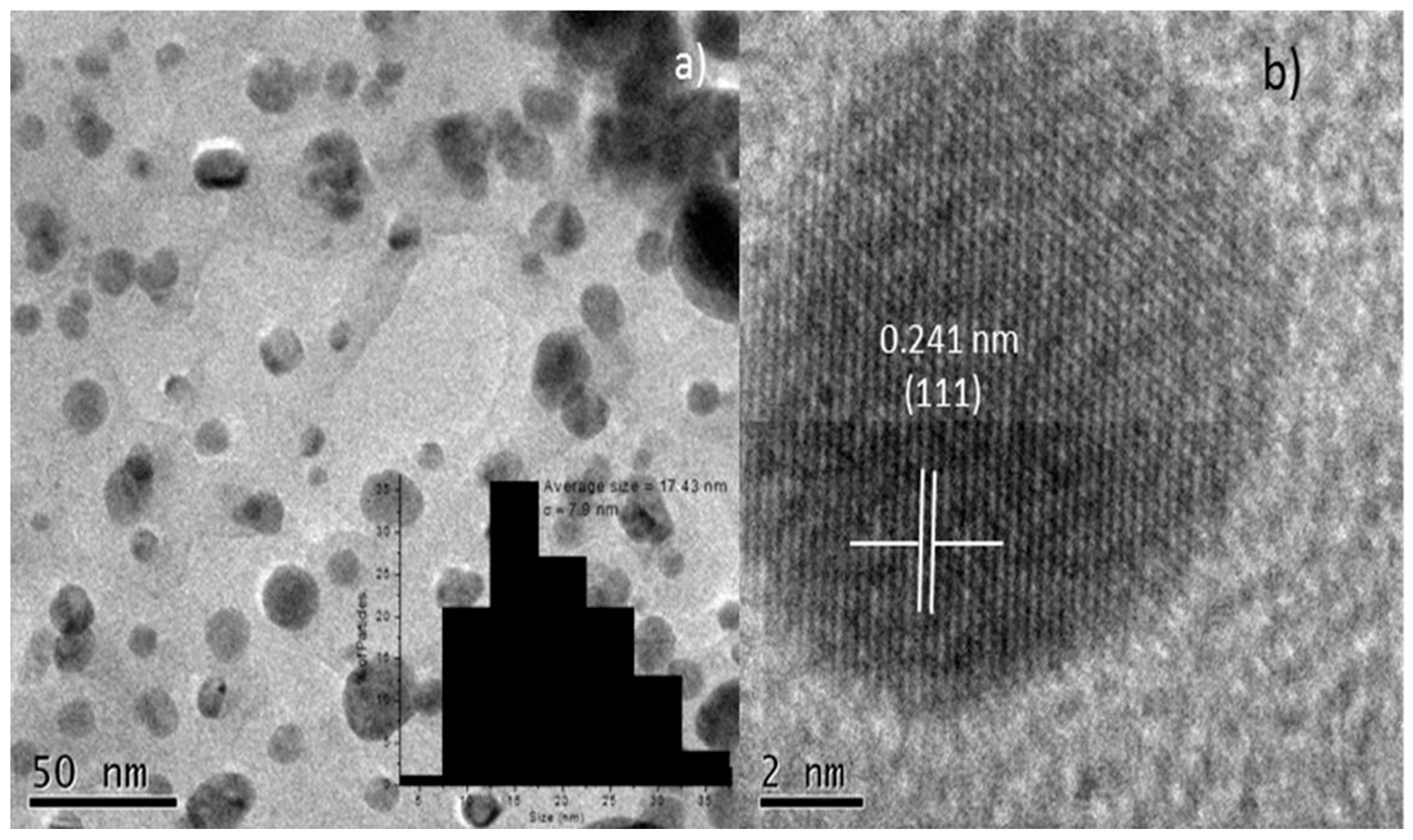
2.1. Characterization of the Silver Nanoparticles Biosynthesized
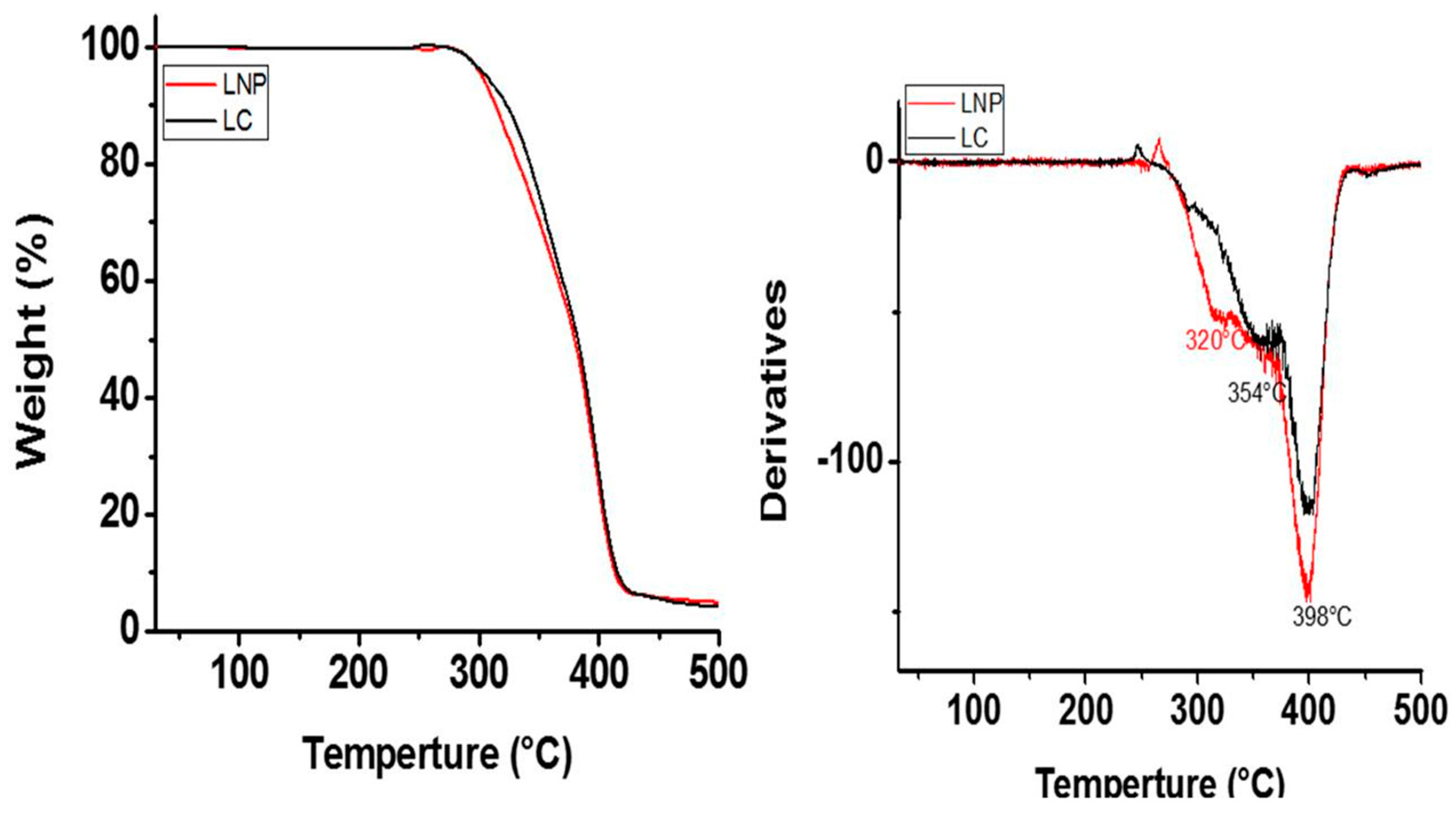
2.2. Thermogravimetric Analysis
2.3. Antibacterial Activity
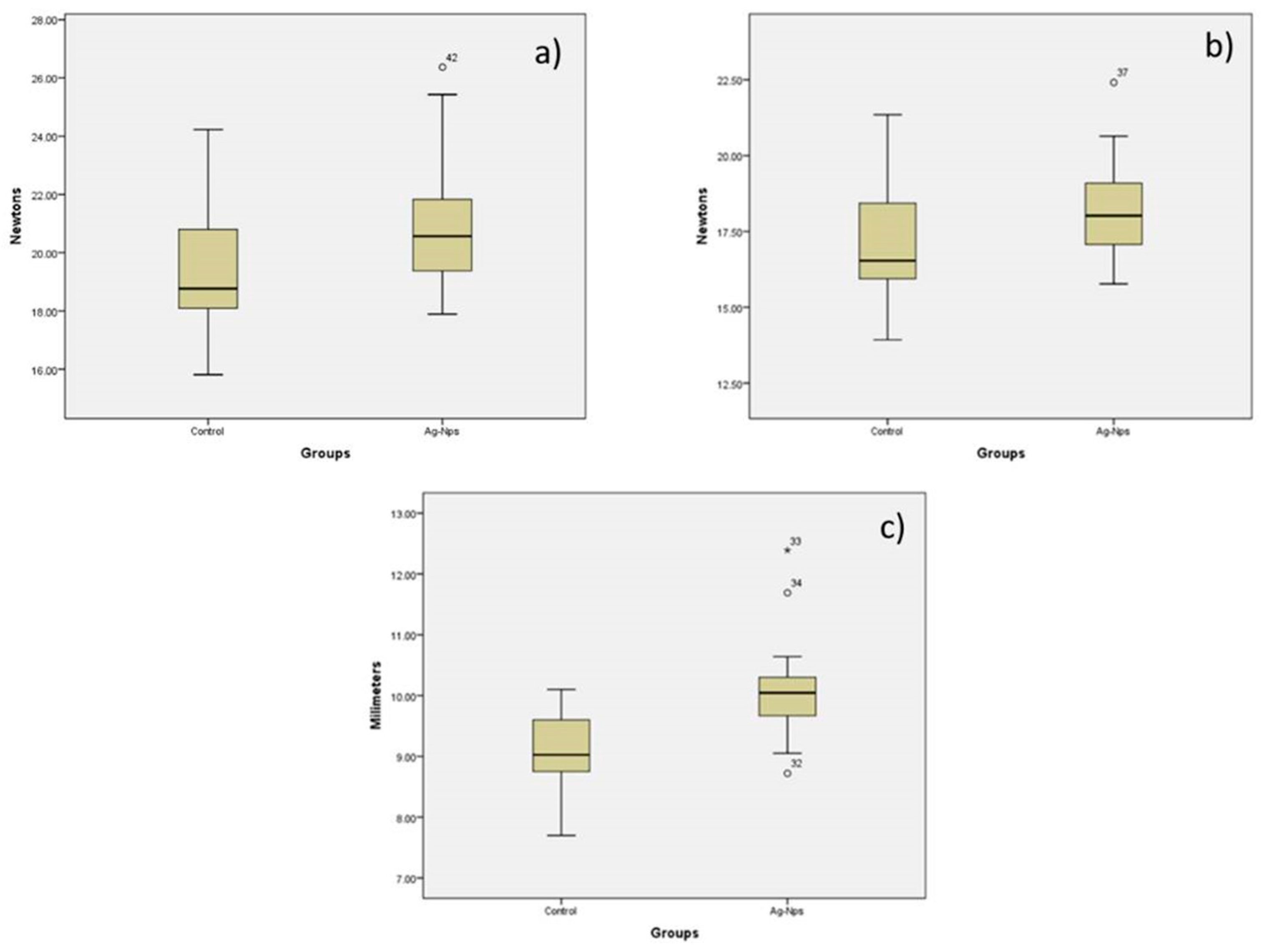
2.4. Physical Properties
3. Discussion
4. Materials and Methods
4.1. Experimental and In Vitro Study
4.1.1. Pre-Treatment of Orthodontic Elastic Ligatures
4.1.2. Preparation of the Heterotheca Inuloides Extract
4.1.3. In Situ Synthesis of AgNPs in Orthodontic Elastic Ligatures
4.2. Characterization of AgNPs
4.3. Characterization of Orthodontic Elastic Ligatures Decorated with AgNPs
4.3.1. Thermogravimetric Analysis
4.3.2. Antibacterial Activity
4.3.3. Mechanical Properties
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Restrepo, M.; Bussaneli, D.G.; Jeremias, F.; Cordeiro, R.C.; Magalhães, A.C.; Palomari Spolidorio, D.M.; Santos-Pinto, L. Control of white spot lesion adjacent to orthodontic bracket with use of fluoride varnish or chlorhexidine gel. Sci. World J. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, P.; Sauerwein, R.; Finlayson, T.; McLeod, J.; Covell, D.A., Jr.; Maier, T.; Machida, C.A. Plaque retention by self-ligating vs elastomeric orthodontic brackets: Quantitative comparison of oral bacteria and detection with adenosine triphosphate-driven bioluminescence. Am. J. Orthod. Dentofac. Orthop. 2009, 135, 426.e1–426.e9. [Google Scholar] [CrossRef]
- Beyth, N.; Redlich, M.; Harari, D.; Friedman, M.; Steinberg, D. Effect of sustained-release chlorhexidine varnish on Streptococcus mutans and Actinomyces viscosus in orthodontic patients. Am. J. Orthod. Dentofac. Orthop. 2003, 123, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Lucchese, A.; Gherlone, E. Prevalence of White-spot lesions before and during orthodontic treatment with fixed appliances. Eur. J. Orthod. 2013, 35, 664–668. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Chen, C.; Weir, M.; Bai, Y.; Xu, H. Antibacterial and protein-repellent orthodontic cement to combat biofilm and white spot lesion. J. Dent. 2015, 43, 1529–1538. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, M.M.; Featherstone, J.D. Demineralization and remineralization around orthodontic appliances: An in vivo study. Am. J. Orthod. Dentofac. Orthop. 1987, 92, 33–40. [Google Scholar] [CrossRef]
- Øgaard, B. White spot lesions during orthodontic treatment: mechanisms and fluoride preventive aspects. Semin. Orthod. 2008, 14, 183–193. [Google Scholar] [CrossRef]
- Gorelick, L.; Geiger, A.M.; Gwinnett, A.J. Incidence of white spot formation after bonding and banding. Am. J. Orthod. 1982, 81, 93–98. [Google Scholar] [CrossRef]
- Julien, K.C.; Buschang, P.H.; Campbell, P.M. Prevalence of white spot lesion formation during orthodontic treatment. Angl. Orthod. 2013, 83, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, D.A.; Clark, A.E.; Rody, W.J.; McGorray, S.P.; Wheeler, T.T. A prospective randomized clinical trial into the capacity of a toothpaste containing NovaMin to prevent white spot lesions and gingivitis during orthodontic treatment. Prog. Orthod. 2015, 16, 25. [Google Scholar] [CrossRef] [PubMed]
- Magno, A.; Enoki, C.; Yoko Ito, I.; Nakane, M.; Faria, G.; Nelson-Filho, P. In-vivo evaluation of the contamination of super slick elastomeric rings by Streptococcus mutans in orthodontic patients. Am. J. Orthod. Dentofac. Orthop. 2008, 133, S104–S109. [Google Scholar] [CrossRef] [PubMed]
- Ahrari, F.; Jalaly, T.; Zebarjad, M. Tensile properties of orthodontic elastomeric ligatures. Indian J. Dent. Res. 2010, 21, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.P.; Vaz, A.C. Comparative evaluation of surface modified elastomeric ligatures for microbial colonization. An in vivo study. Indian J. Dent. Res. 2015, 26, 180–185. [Google Scholar] [PubMed]
- Forsberg, C.M.; Brattström, V.; Malmberg, E.; Nord, C.E. Ligature wires and elastomeric rings: two methods of ligation, and their association with microbial colonization of Streptococcus mutans and Lactobacilli. Eur. J. Orthod. 1991, 13, 416–420. [Google Scholar] [CrossRef] [PubMed]
- Türkkahraman, H.; Sayin, M.O.; Bozkurt, F.Y.; Yetkin, Z.; Kaya, S.; Onal, S. Archwire ligation techniques, microbial colonization, and periodontal status in orthodontically treated patients. Angl. Orthod. 2005, 75, 231–236. [Google Scholar]
- Aghili, H.; Jafari Nadoushan, A.A.; Herandi, V. Antimicrobial effect of zataria multiflora extract in comparison with chlorhexidine mouthwash on experimentally contaminated orthodontic elastomeric ligatures. J. Dent. 2015, 12, 1–10. [Google Scholar]
- Rakhshan, H.; Rakhshan, V. Effects of the initial stage of active fixed orthodontic treatment and sex on dental plaque accumulation: a preliminary prospective cohort study. Saudi Dent. J. 2014, 6, 86–90. [Google Scholar] [CrossRef]
- Miura, K.K.; Ito, I.Y.; Enoki, C.; Elias, A.M.; Matsumoto, M.A. Anticariogenic effect of fluoride-releasing elastomers in orthodontic patients. Braz. Oral Res. 2007, 21, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, B.W.; Rottwinkel, Y. Assessing patient-specific decalcification risk in fixed orthodontic treatment and its impact on prophylactic procedures. Am. J. Orthod. Dentofac. Orthop. 2004, 126, 318–324. [Google Scholar] [CrossRef]
- Srivastava, K.; Tikku, T.; Khanna, R.; Sachan, K. Risk factors and management of white spot lesions in orthodontics. J. Orthod. Sci. 2013, 2, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Seino, P.Y.; Freitas, P.M.; Marques, M.M.; de Souza Almeida, F.C.; Botta, S.B.; Moreira, M.S. Influence of CO2 (10.6 µm) and Nd: Yag laser irradiation on the prevention of enamel caries around orthodontic brackets. Lasers Med. Sci. 2015, 30, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Benson, P.E.; Douglas, C.W.; Martin, M.V. Fluoridated elastomers: effect on the microbiology of plaque. Am. J. Orthod. Dentofac. Orthop. 2004, 126, 325–330. [Google Scholar] [CrossRef]
- O’Dwyer, J.J.; Tinsley, D.; Benson, P.E. The effect of stretching on the release of fluoride from fluoridated elastomeric ligatures. Am. J. Orthod. Dentofac. Orthop. 2005, 128, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Lee, D.; Lee, J.; Lim, Y. The effect of silver ion-releasing elastomers on mutans streptococci in dental plaque. Korean J. Orthod. 2012, 42, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Won, Y.A. The Antibacterial Activities of Silver ion Releasing Elastomers on Oral Pathogenic Microbes. Master’s Thesis, Korea University, Seoul, Korea, 2010. [Google Scholar]
- O’Dell, M. Antimicrobial effectiveness of silver releasing elastomeric ligature ties. Master’s Thesis, University of Alabama, Birmingham, United States of America, 2010. Available online: http://www.mhsl.uab.edu/dt/2011m/odell.pdf (accessed on 28 March 2017).
- Caccianiga, G.; Ordesi, P.; Cambini, A.; Stanizzi, A.; Denotti, G.; Fumagalli, T. Evaluation about antimicrobial activity of silver in Orthoshield-Safe-T-Ties ligature used in orthodontic patients. Eur. J. Inflamm. 2012, 10, 93–96. [Google Scholar] [CrossRef]
- Melo, M.; Guedes, S.; Xu, H.; Rodrigues, L. Nanotechnology-based restorative materials for dental caries management. Trends Biotechnol. 2013, 31, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Correa, J.M.; Mori, M.; Sanches, H.L.; da Cruz, A.D.; Poiate, E., Jr.; Poiate, I.A. Silver nanoparticles in dental biomaterials. Int. J. Biomater. 2015. [Google Scholar] [CrossRef] [PubMed]
- Salem, W.; Leitner, D.; Zingl, F.; Schratter, G.; Prassl, R.; Goessler, W.; Reidl, J.; Schild, S. Antibacterial activity of silver and zinc nanoparticles against Vibrio cholera and enterotoxic Escherichia coli. Int. J. Med. Microbiol. 2014, 305, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Selvam, K.; Sudhakar, C.; Govarthanan, M.; Thiyagarajan, P.; Sengottaiyan, A.; Senthilkumar, B.; Selvankumar, T. Eco-friendly biosynthesis and characterization of silver nanoparticles using Tinospora cordifolia (Thunb.) Miers and evaluate its antibacterial, antioxidant potential. J. Radiat. Res. Appl. Sci. 2017, 10, 1–8. [Google Scholar] [CrossRef]
- Ajitha, B.; Ashok, Y.; Sreedhara, P.; Suneetha, Y.; Hwan-Jin, J.; Won, C. Instant biosynthesis of silver nanoparticles using Lawsonia inermis leaf extract: Innate catalytic, antimicrobial and antioxidant activities. J. Mol. Liq. 2016, 219, 474–481. [Google Scholar] [CrossRef]
- Espenti, C.S.; Rao, K.S.V.; Rao, M. Bio-synthesis and characterization of silver nanoparticles using Terminalia chebula leaf extract and evaluation of its antimicrobial potential. Mater. Lett. 2016, 174, 129–133. [Google Scholar] [CrossRef]
- Pimpang, P.; Sutham, W.; Mangkorntong, N.; Mangkorntong, P.; Choopun, S. Effect of stabilizer on preparation of silver and gold nanoparticles using grinding method. Chiang Mai J. Sci. 2008, 35, 250–257. [Google Scholar]
- Zschech, D.; Ha Kim, D.; Alexey, P.; Hopfe, S.; Scholz, R.; Goring, P.; Hillebrand, R.; Senz, S.; Hawker, J.C.; Russell, T.P. High-temperature resistant, ordered gold nanoparticles array. Nanotechnology. 2006, 17, 2122–2126. [Google Scholar] [CrossRef]
- Giorgetti, E.; Giusti, A.; Laza, S.; Marsili, P.; Giammanco, F. Production of colloidal gold nanoparticles by picoseconds laser ablation in liquids. Phys. Status Solidi A. 2007, 204, 1693–1698. [Google Scholar] [CrossRef]
- Turner, S.; Tavernier, S.; Huyberechts, G.; Biermans, E.; Bals, S.; Batenburg, K.; Tendeloo, G. Assisted spray pyrolysis production and characterization of ZnO nanoparticles with narrow size distribution. J. Nanopart. Res. 2010, 12, 615–622. [Google Scholar] [CrossRef]
- Ko, T.S.; Yang, S.; Hsu, H.C.; Chu, C.P.; Lin, H.F.; Liao, S.C.; Lu, T.C.; Kuo, H.C.; Hsieh, W.H.; Wang, S.C. ZnO nanopowders fabricated by dc thermal plasma synthesis. Mater. Sci. Eng., B. 2006, 134, 54–58. [Google Scholar] [CrossRef]
- Breitwieser, D.; Moghaddam, M.M.; Spirk, S.; Baghbanzadeh, M.; Pivec, T.; Fasl, H.; Ribitsch, V.; Kappe, C.O. In situ preparation of silver nanocomposites on cellulosic fibers microwave vs conventional heating. Carbohydr. Polym. 2013, 94, 677–686. [Google Scholar] [CrossRef] [PubMed]
- Padalia, H.; Moteriya, P.; Chanda, S. Green synthesis of silver nanoparticles from marigold flower and its synergistic antimicrobial potential. Arabian J. Chem. 2014, 8, 732–741. [Google Scholar] [CrossRef]
- Manjumeena, R.; Duraibabu, D.; Sudha, J.; Kalaichelvan, P.T. Biogenic nanosilver incorporated reverse osmosis membrane for antibacterial and antifungal activities against selected pathogenic strains: an enhanced eco-friendly water disinfection approach. J. Environ. Sci. Health. Part A Toxic/Hazard. Subst. Environ. Eng. 2014, 49, 1125–1133. [Google Scholar] [CrossRef] [PubMed]
- Velusamy, P.; Das, J.; Pachaiappan, R.; Vaseeharan, B.; Pandian, K. Greener approach for synthesis of antibacterial silver nanoparticles using aqueous solution of neem gum (Azadirachta indica L.). Ind. Crops Prod. 2015, 66, 103–109. [Google Scholar] [CrossRef]
- Gené, R.M.; Segura, L.; Adzet, T.; Marin, E.; Iglesias, J. Heterotheca inuloides: Anti-inflamatory and analgesic effect. J. Ethnopharmacol. 1998, 60, 157–162. [Google Scholar] [CrossRef]
- Kamatani, J.; Iwadate, T.; Tajima, R.; Kimoto, H.; Yamada, Y.; Masuoka, N.; Kubo, I.; Nihei, K. Stereochemical investigation and total synthesis of inuloidin, a biologically active sesquiterpenoid from Heterotheca inuloides. Tetrahedron 2014, 70, 3141–3145. [Google Scholar] [CrossRef]
- Lin, L.; Harnly, J. LC-PDA-ESI/MS Identification of the phenolic components of three compositae spices: Chamomile, Tarragon and Mexican Arnica. Nat. Prod. Commun. 2012, 7, 749–752. [Google Scholar] [PubMed]
- Sudhakar, C.; Selvam, K.; Govarthanan, M.; Senthilkumar, B.; Sengottaiyan, A.; Stalin, M.; Selvankumar, T. Acorus calamus rhizome extract mediated biosynthesis of silver nanoparticles and their bactericidal activity against human pathogens. Genet. Eng. Biotechnol. J. 2015, 13, 93–99. [Google Scholar] [CrossRef]
- Prabu, H.; Johnson, I. Plant-mediated biosynthesis and characterization of silver nanoparticles by leaf extracts of Tragia involucrate, Cymbopogon citronella, Solanum verbascifolium and Tylophara ovata. Karbala Int. J. Mod. Sci. 2015, 1, 237–246. [Google Scholar] [CrossRef]
- Bindhu, M.R.; Umadevi, M. Antibacterial and catalytic activities of green synthesized silver nanoparticles. Spectrochim. Acta Part A 2015, 135, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lee, Y. Measurement of discolouration of orthodontic elastomeric modules with a digital camera. Eur. J. Orthod. 2009, 31, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Cristóbal, L.F.; Martínez-Castañon, G.A.; Martínez-Martínez, R.E.; Loyola-Rodríguez, J.P.; Patiño-Marín, N.; Reyes-Macías, J.F.; Ruiz, F. Antimicrobial sensitivity of Streptococcus mutans serotypes to silver nanoparticles. Mater. Sci. Eng. C Mater. Biol. Appl. 2012, 32, 896–901. [Google Scholar] [CrossRef]
- Kasraei, S.; Sami, L.; Hendi, S.; Alikhani, M.; Rezaei-Soufi, L.; Khamverdi, Z. Antibacterial properties of composite resins incorporating silver and zinc oxide nanoparticles on Streptococcus mutans and Lactobacillus. Restor. Dent. Endod. 2014, 39, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Emmanuel, R.; Palanisamy, S.; Chen, S.M.; Chelladurai, K.; Padmavathy, S.; Saravanan, M.; Prakash, P.; Ali, M.A.; Al-Hemaid, F.M.A. Antimicrobial efficacy of green synthesized drug blended silver nanoparticles against dental caries and periodontal disease causing microorganisms. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 56, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Cavadas, F.; de Oliveira, L.; Monteiro, F.; da Silva, E. Preparation, characterization and antibacterial properties of silver nanoparticles-hidoxyapatite composites by a simple and eco-friendly method. Ceram. Int. 2016, 42, 2271–2280. [Google Scholar]
- Hernández-Sierra, J.F.; Ruiz, F.; Pena, D.C.; Martínez-Gutiérrez, F.; Martínez, A.E.; Guillén, A.; Tapia-Pérez, H.; Castañón, G.M. The antimicrobial sensitivity of Streptococcus mutans to nanoparticles of silver, zinc oxide, and gold. Nanomedicine 2008, 4, 237–240. [Google Scholar]
- Zhang, K.; Melo, M.A.; Cheng, L.; Weir, M.D.; Bai, Y.; Xu, H.H. Effect of quaternary ammonium and silver nanoparticles-containing adhesives on dentin bond strength and dental plaque microcosm biofilms. Den. Mater. 2012, 28, 842–852. [Google Scholar] [CrossRef] [PubMed]
- Argueta-Figueroa, L.; Scougall-Vilchis, R.J.; Morales-Luckie, R.A.; Olea-Mejía, O.F. An evaluation of the antibacterial properties and shear bond strength of copper nanoparticles as a nanofiller in orthodontic adhesive. Aust. Orthod. J. 2015, 31, 42–48. [Google Scholar] [PubMed]
- Samiei, M.; Farjami, A.; Dizaj, S.M.; Lotfipour, F. Nanoparticles for antimicrobial purposes in Endodontics: A systematic review of in vitro studies. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 58, 1269–1278. [Google Scholar] [CrossRef] [PubMed]
- Afkhami, F.; Akbari, S.; Chiniforush, N. Entrococcus faecalis elimination in root canals using silver nanoparticles, photodynamic therapy, diode laser, or laser activated nanoparticles: an in vitro study. J. Endod. 2017, 43, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, R.D.; Bhide, S.V.; Prasad, B.; Shimpi, J. Applications of silver nanoparticles in prosthodontics: An overview. Eur. J. Prosthodont. 2016, 4, 45–50. [Google Scholar] [CrossRef]
- Yamada, R.; Nozaki, K.; Horiuchi, N.; Yamashita, K.; Nemoto, R.; Miura, H.; Nagai, A. Ag nanoparticles-coated zirconia for antibacterial prosthesis. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 78, 1054–1060. [Google Scholar] [CrossRef] [PubMed]
- Pokrowiecki, R.; Zareba, T.; Szaraniec, B.; Mielczarec, A.; Menaszek, E.; Tyski, S. In vitro studies of nanosilver-doped titanium implants for oral and maxillofacial surgery. Int. J. Nanomedicine. 2017, 12, 4285–4297. [Google Scholar] [CrossRef] [PubMed]
- Besinis, A.; Hadi, S.D.; Le, H.R.; Tredwin, C.; Handy, R.D. Antibacterial activity and biofilm inhibition by surface modified titanium alloy medical implants following application of silver, titanium dioxide and hydroxyapatite nanocoatings. Nanotoxicology 2017, 11, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Sundeep, D.; Vijaya Kumar, T.; Rao, P.S.S.; Ravikumar, R.V.S.S.N.; Gopala Krihna, A. Green synthesis and characterization of Ag nanoparticles from Mangifera indica leaves for dental restoration and antibacterial applications. Prog. Biomater. 2017, 6, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Ai, M.; Du, Z.; Zhu, S.; Geng, H.; Zhang, X.; Cai, Q.; Yang, X. Composite resin reinforced with silver nanoparticles-laden hydroxyapatite nanowires for dental application. Dent. Mater. 2017, 33, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Reddy, A.K.; Kambalyal, P.B.; Patil, S.R.; Vankhre, M.; Khan, M.Y.; Kumar, T.R. Comparative evaluation and influence on shear bond strength of incorporating silver, zinc oxide, and titanium dioxide nanoparticles in orthodontic adhesive. J. Orthod. Sci. 2016, 5, 127–131. [Google Scholar] [PubMed]
- Degrazia, F.W.; Leitune, V.C.; García, I.M.; Arthur, R.A.; Samuel, S.M.; Collares, F.M. Effect of silver nanoparticles on the physicochemical and antimicrobial properties of an orthodontic adhesive. J. Appl. Oral Sci. 2016, 24, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Hammouda, I.M. Current perspectives of nanoparticles in medical and dental biomaterials. J. Biomed. Res. 2012, 26, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Aminian, A.; Nakhaei, S.; Agahi, R.H.; Rezaeizade, M.; Aliabadi, H.M.; Heidarpour, M. Evaluation of the effect of different stretching patterns on force decay and tensile properties of elastomeric ligatures. Dent. Res. J. 2015, 12, 589–595. [Google Scholar]
- Cheng, L.; Zhang, K.; Melo, M.A.; Weir, M.D.; Zhou, X.; Xu, H.H. Anti-biofilm dentin primer with quaternary ammonium and silver nanoparticles. J. Dent. Res. 2012, 91, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Morales-Luckie, R.A.; Lopezfuentes-Ruiz, A.A.; Olea-Mejía, O.F.; Argueta-Figueroa, L.; Sánchez-Mendieta, V.; Brostow, W.; Hinestroza, J. Synthesis of silver nanoparticles using aqueous extracts of Heterotheca inuloides as reducing agent and natural fibers as templates: Agave lechuguilla and silk. Mater. Sci. Eng. C Mat. Biol. Appl. 2016, 69, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, M.J. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Fourth Informational Supplement M100-S24; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2014; (Electronic ISBN 1–56238–898–3). [Google Scholar]
- Argueta-Figueroa, L.; Morales-Luckie, R.A.; Scougall-Vilchis, R.; Olea-Mejía, O.F. Synthesis, characterization and antibacterial activity of copper, nickel and bimetallic Cu-Ni nanoparticles for potential use in dental materials. Prog. Nat. Sci. Mat. Int. 2014, 24, 321–328. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
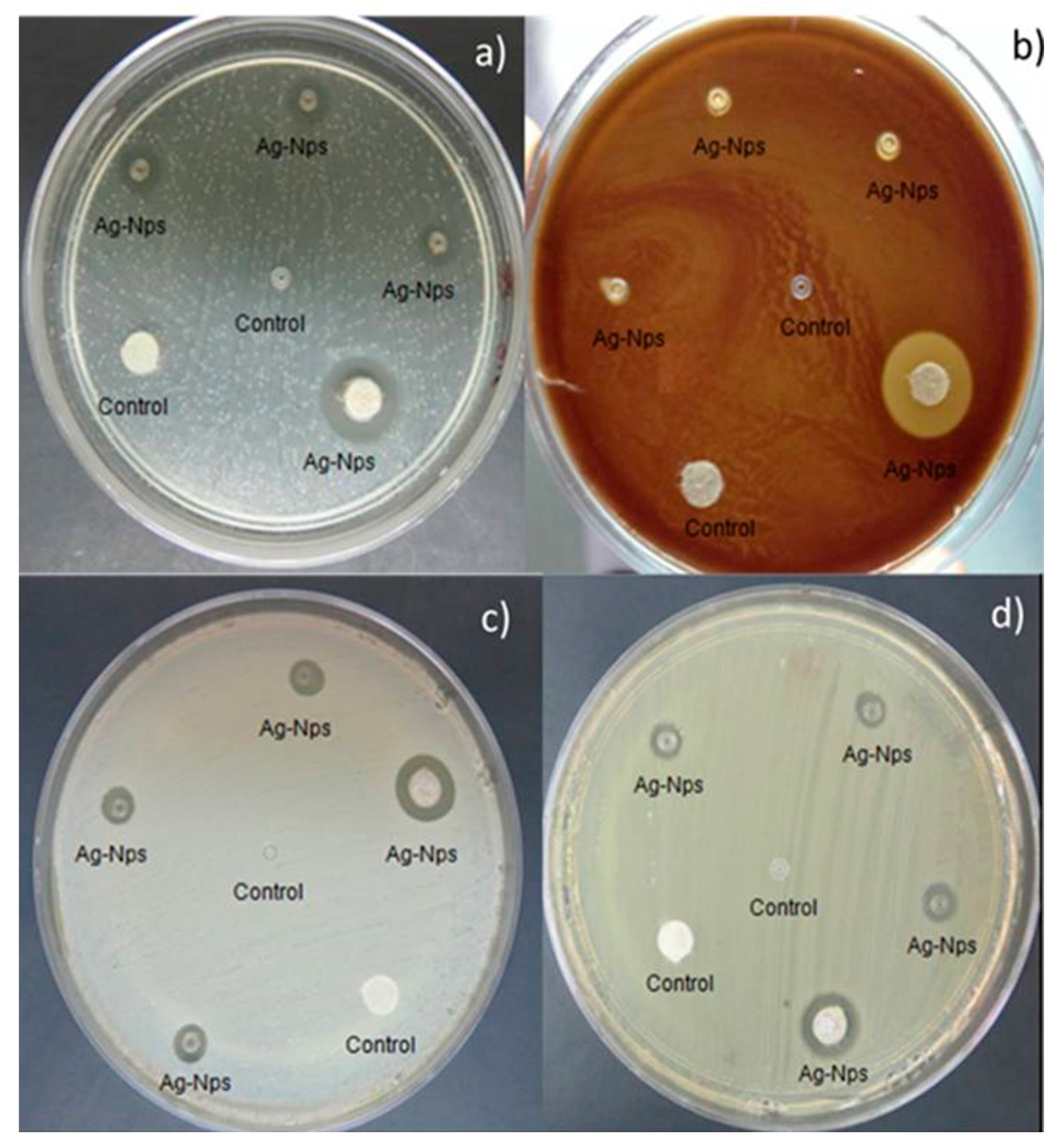
| Microorganism | Mean and Standard Deviation | |
|---|---|---|
| Ligature with AgNPs | Paper Disk with AgNPs | |
| S. mutans | 2.0 ± 0.12 mm | 4.0 ± 0.16 mm |
| L. casei | 1.0 ± 0.21 mm | 5.0 ± 0.27 mm |
| S. aureus | 2.0 ± 0.18 mm | 3.0 ± 0.22 mm |
| E. coli | 1.5 ± 0.12 mm | 2.0 ± 0.15 mm |
| Physical Properties | Mean | Range | * p | ||
|---|---|---|---|---|---|
| Control | AgNPs | Control | AgNPs | ||
| Maximum strength | 19.4897 | 20.8370 | 15.81–24.23 | 17.89–26.37 | 0.012 |
| Tension | 17.2320 | 18.1847 | 13.93–21.35 | 15.77–22.41 | 0.033 |
| Displacement | 9.0667 | 10.0733 | 7.70–10.10 | 8.72–12.39 | 0.001 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Gómora, A.E.; Lara-Carrillo, E.; Robles-Navarro, J.B.; Scougall-Vilchis, R.J.; Hernández-López, S.; Medina-Solís, C.E.; Morales-Luckie, R.A. Biosynthesis of Silver Nanoparticles on Orthodontic Elastomeric Modules: Evaluation of Mechanical and Antibacterial Properties. Molecules 2017, 22, 1407. https://doi.org/10.3390/molecules22091407
Hernández-Gómora AE, Lara-Carrillo E, Robles-Navarro JB, Scougall-Vilchis RJ, Hernández-López S, Medina-Solís CE, Morales-Luckie RA. Biosynthesis of Silver Nanoparticles on Orthodontic Elastomeric Modules: Evaluation of Mechanical and Antibacterial Properties. Molecules. 2017; 22(9):1407. https://doi.org/10.3390/molecules22091407
Chicago/Turabian StyleHernández-Gómora, Alma E., Edith Lara-Carrillo, Julio B. Robles-Navarro, Rogelio J. Scougall-Vilchis, Susana Hernández-López, Carlo E. Medina-Solís, and Raúl A. Morales-Luckie. 2017. "Biosynthesis of Silver Nanoparticles on Orthodontic Elastomeric Modules: Evaluation of Mechanical and Antibacterial Properties" Molecules 22, no. 9: 1407. https://doi.org/10.3390/molecules22091407
APA StyleHernández-Gómora, A. E., Lara-Carrillo, E., Robles-Navarro, J. B., Scougall-Vilchis, R. J., Hernández-López, S., Medina-Solís, C. E., & Morales-Luckie, R. A. (2017). Biosynthesis of Silver Nanoparticles on Orthodontic Elastomeric Modules: Evaluation of Mechanical and Antibacterial Properties. Molecules, 22(9), 1407. https://doi.org/10.3390/molecules22091407







