A New N-methoxypyridone from the Co-Cultivation of Hawaiian Endophytic Fungi Camporesia sambuci FT1061 and Epicoccum sorghinum FT1062
Abstract
:1. Introduction
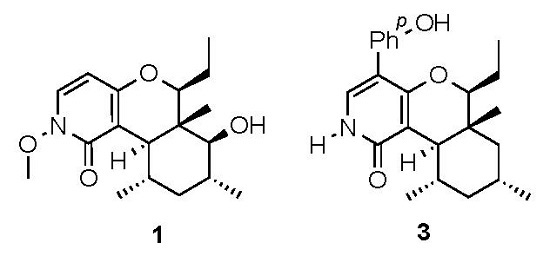
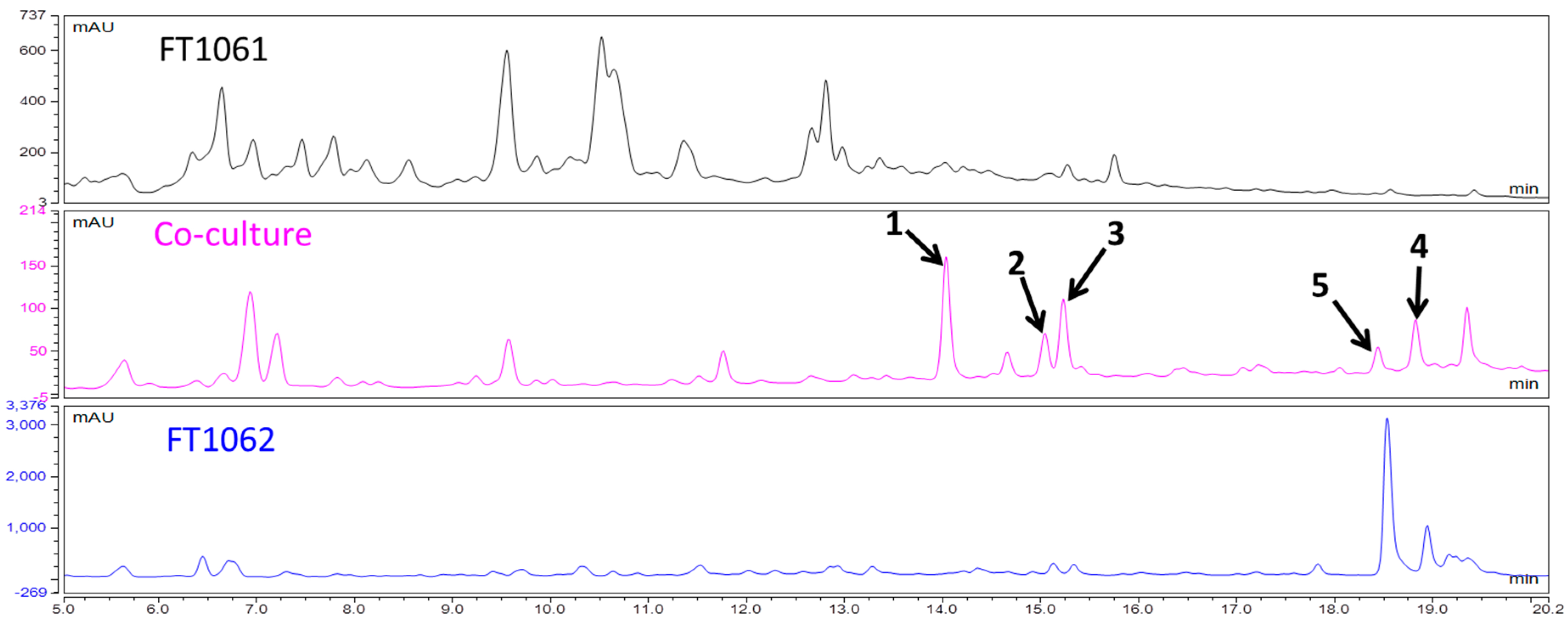
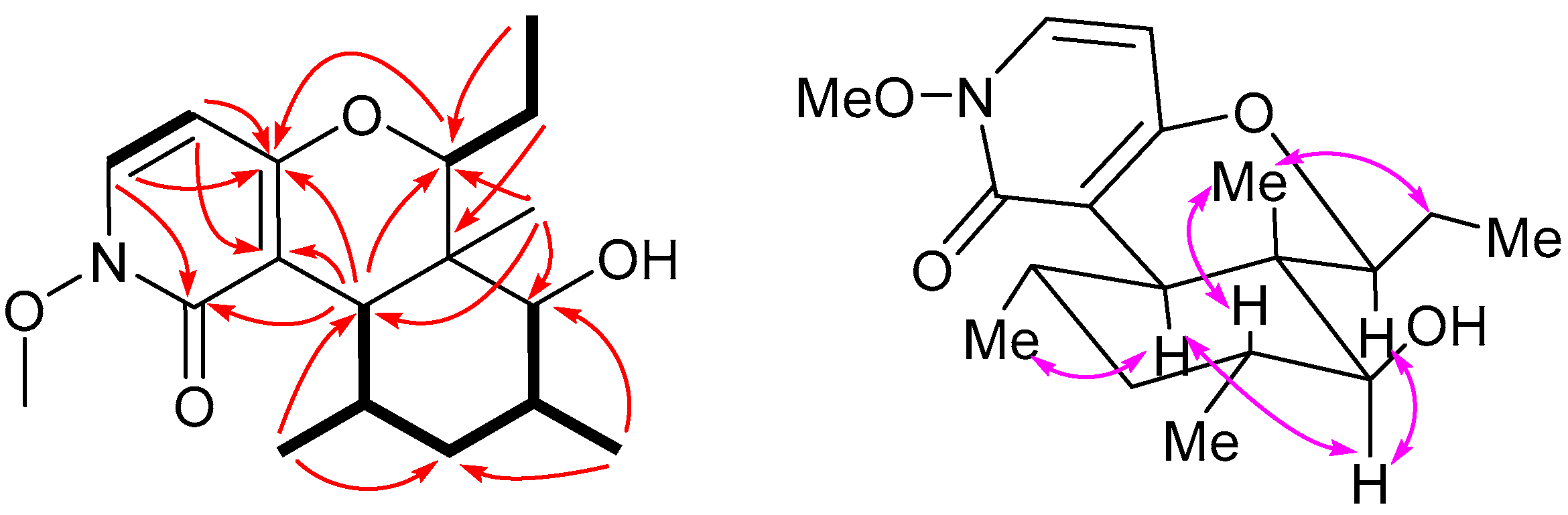
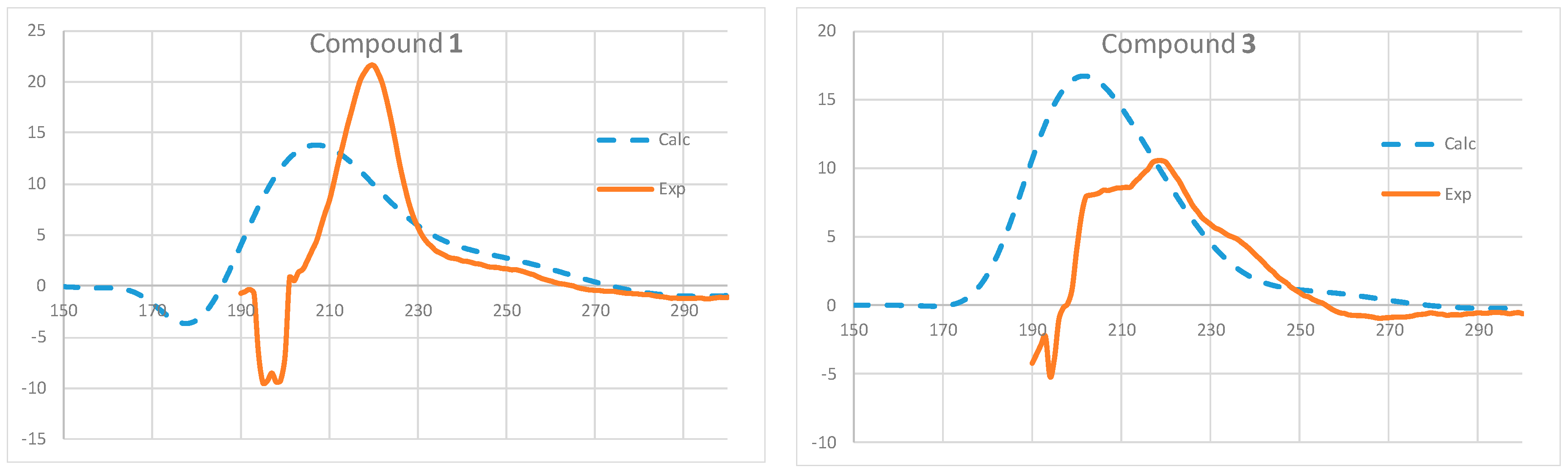
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Isolation and Identification of Fungal Strain
3.3. Cultivation
3.4. Isolation of Compounds 1–5
3.5. Charaterization of Compound 1
3.6. Anti-Microbial Activity
3.7. Anti-Proliferative Activity
3.8. ECD Calculations
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Barakat, F.; Vansteelandt, M.; Triastuti, A.; Rieusset, L.; Cabanillas, B.; Haddad, M.; Fabre, N. Co-cultivation approach and untargeted metabolomics in the search for new secondary metabolites from endophytic fungi. Planta Med. 2016, 82, S1–S381. [Google Scholar] [CrossRef]
- Brakhage, A.A.; Schroeckh, V. Fungal secondary metabolites—Strategies to activate silent gene clusters. Fungal Genet. Biol. 2011, 48, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Scherlach, K.; Hertweck, C. Triggering cryptic natural product biosynthesis in microorganisms. Org. Biomol. Chem. 2009, 7, 1753–1760. [Google Scholar] [CrossRef] [PubMed]
- Adnani, N.; Vazquez-Rivera, E.; Adibhatla, S.N.; Ellis, G.A.; Braun, D.R.; Bugni, T.S. Investigation of Interspecies Interactions within Marine Micromonosporaceae Using an Improved Co-Culture Approach. Mar. Drugs 2015, 13, 6082–6098. [Google Scholar] [CrossRef] [PubMed]
- Pettit, P.K. Mixed fermentation for natural product drug discovery. Appl. Microbiol. Biotechnol. 2009, 83, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Shang, Z.; Salim, A.A.; Capon, R.J. Chaunopyran A: co-cultivation of marine mollusk-derived fungi activates a rare class of 2-alkenyl-tetrahydropyran. J. Nat. Prod. 2017, 80, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, S.; Schumpp, O.; Bohni, N.; Monod, M.; Gindro, K.; Wolfender, J.L. De Novo production of metabolites by fungal co-culture of Trichophyton rubrum and Bionectria ochroleuca. J. Nat. Prod. 2013, 76, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.C.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Induced production of emericellamides A and B from the marine-derived fungus Emericella sp. in competing co-culture. J. Nat. Prod. 2007, 70, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Zuck, K.M.; Shipley, S.; Newman, D.J. Induced production of N-formyl alkaloids from Aspergillus fumigatus by co-culture with Streptomyces peucetius. J. Nat. Prod. 2011, 74, 1653–1657. [Google Scholar] [CrossRef] [PubMed]
- Sonnebichler, J.; Dietrich, J.; Peipp, H. Secondary fungal metabolites and their biological activities, V. Investigations concerning the induction of the biosynthesis of toxic secondary metabolites in basidiomycetes. Biol. Chem. Hoppe Seyler 1994, 375, 71–79. [Google Scholar] [CrossRef]
- Cao, S.; Ross, L.; Tamayo, G.; Clardy, J. Asterogynins: secondary metabolites from a Costa Rican endophytic fungus. Org. Lett. 2010, 12, 4661–4663. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Clardy, J. New naphthoquinones and a new δ-lactone produced by endophytic fungi from Costa Rica. Tetrahedron Lett. 2011, 52, 2206–2208. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Cryan, L.; Habeshian, K.A.; Murillo, C.; Tamayo-Castillo, G.; Rogers, M.S.; Clardy, J. Phenolic compounds as antiangiogenic CMG2 inhibitors from Costa Rican endophytic fungi. Bioorg. Med. Chem. Lett. 2012, 22, 5885–5888. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; McMillin, D.W.; Tamayo, G.; Delmore, J.; Mitsiades, C.S.; Clardy, J. Inhibition of tumor cells interacting with stromal cells by xanthones isolated from a Costa Rican Penicillium sp. J. Nat. Prod. 2012, 75, 793–797. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Yang, B.; Turkson, J.; Cao, S. Anti-proliferative ambuic acid derivatives from Hawaiian endophytic fungus Pestalotiopsis sp. FT172. Phytochemistry 2017, 140, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Li, C.S.; Sarotti, A.M.; Turkson, J.; Cao, S. Verbenanone, an octahydro-5H-chromen-5-one from a Hawaiian-Plant Associated Fungus FT431. Tetrahedron Lett. 2017, 58, 2290–2293. [Google Scholar] [CrossRef]
- Huang, P.; Li, C.S.; Sarotti, A.M.; Turkson, J.; Cao, S. Sphaerialactonam, a γ-lactam–isochromanone from the Hawaiian endophytic fungus Paraphaeosphaeria sp. FT462. Tetrahedron Lett. 2017, 58, 1330–1333. [Google Scholar] [CrossRef]
- Li, C.; Ren, G.; Yang, B.; Gabriella, M.; Turkson, J.; Fei, P.; Ding, Y.; Walker, L.; Cao, S. Meroterpenoids with Antiproliferative Activity from a Hawaiian-Plant Associated Fungus Peyronellaea coffeae-arabicae FT238. Org. Lett. 2016, 18, 2335–2338. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ding, Y.; Yang, B.; Hoffman, N.; Yin, H.Q.; Mahmud, T.; Turkson, J.; Cao, S. Eremophilane sesquiterpenes from Hawaiian endophytic fungus Chaetoconis sp. FT087. Phytochemistry 2016, 126, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.J.; Li, C.; Cao, S. Hawaii natural compounds are promising to reduce ovarian cancer deaths. Cancer Biol. Ther. 2016, 17, 709–712. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ding, Y.; Yang, B.; Miklossv, G.; Yin, H.Q.; Walker, L.A.; Turkson, J.; Cao, S. A New Metabolite with a Unique 4-Pyranone-γ-Lactam-1,4-Thiazine Moiety from a Hawaiian-Plant Associated Fungus. Org. Lett. 2015, 17, 3556–3559. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Yang, B.; Fenstemacher, R.; Turkson, J.; Cao, S. Lycopodiellactone, an unusual δ-lactone-isochromanone from a Hawaiian plant-associated fungus Paraphaeosphaeria neglecta FT462. Tetrahedron Lett. 2015, 56, 1724–1727. [Google Scholar] [CrossRef]
- Shiratake, S.; Nakahara, T.; Iwahashi, H.; Onodera, T.; Mizushina, Y. Rose myrtle (Rhodomyrtus tomentosa) extract and its component, piceatannol, enhance the activity of DNA polymerase and suppress the inflammatory response elicited by UVB-induced DNA damage in skin cells. Mol. Med. Rep. 2015, 12, 5857–5864. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.C.; Davenport, K.W.; Hovde, B.; Silva, D.; Chain, P.S.G.; Correa, B.; Rodrigues, D.F. Draft Genome Sequence of Sorghum Grain Mold Fungus Epicoccum sorghinum, a Producer of Tenuazonic Acid. Genome Announc. 2017, 5, e01495–e01516. [Google Scholar] [CrossRef] [PubMed]
- McBrien, K.D.; Gao, Q.; Huang, S.; Klohr, S.E.; Wang, R.R.; Pirnik, D.M.; Neddermann, K.M.; Bursuker, I.; Kadow, K.F.; Leet, J.E. Fusaricide, a new cytotoxic N-hydroxypyridone from Fusarium sp. J. Nat. Prod. 1996, 59, 1151–1153. [Google Scholar] [CrossRef] [PubMed]
- Snider, B.B.; Smith, R.B. Total synthesis of (±)-fusaricide. Synth. Commun. 2001, 31, 2667–2679. [Google Scholar] [CrossRef]
- Isaka, M.; Tanticharoen, M.; Kongsaeree, P.; Thebtaranonth, Y. Structures of Cordypyridones A−D, Antimalarial N-Hydroxy- and N-Methoxy-2-pyridones from the Insect Pathogenic Fungus Cordyceps nipponica. J. Org. Chem. 2001, 66, 4803–4808. [Google Scholar] [CrossRef] [PubMed]
- Kemami Wangun, H.V.; Hertweck, C. Epicoccarines A, B and epipyridone: Tetramic acids and pyridone alkaloids from an Epicoccum sp. associated with the tree fungus Pholiota squarrosa. Org. Biomol. Chem. 2007, 5, 1702–1705. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.X.; Jiao, J.Y.; Li, J.; Wang, W.; Gu, Q.Q.; Zhu, T.J.; Li, D.H. Pyronepolyene C-glucosides with NF-κB inhibitory and anti-influenza A viral (H1N1) activities from the sponge-associated fungus Epicoccum sp. JJY40. Bioorg. Med. Chem. Lett. 2012, 22, 3188–3190. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.C.; Syu, W.J.; Li, S.Y.; Lin, C.H.; Lee, G.H.; Sun, C.M. Antimicrobial activities of naphthazarins from Arnebia euchroma. J. Nat. Prod. 2002, 65, 1857–1862. [Google Scholar] [CrossRef] [PubMed]
- Delazar, A.; Byres, M.; Gibbons, S.; Kumarasamy, Y.; Modarresi, M.; Nahar, L.; Shoeb, M.; Sarker, S.D. Iridoid glycosides from Eremostachys glabra. J. Nat. Prod. 2004, 67, 1584–1587. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, C.; Subbaraju, G.V.; Venkateswarlu, Y.; Venugopal, R.T. New acylated iridoid glucosides from Vitex altissima. J. Nat. Prod. 2004, 67, 2012–2016. [Google Scholar] [CrossRef] [PubMed]
- Spartan’08; Wavefunction: Irvine, CA, USA, 2008; Available online: http://www.wavefun.com/ (accessed on 11 July 2017).
- Li, X.C.; Ferreiraa, D.; Dinga, Y. Determination of Absolute Configuration of Natural Products: Theoretical Calculation of Electronic Circular Dichroism as a Tool. Curr. Org. Chem. 2010, 14, 1678–1697. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09, C.01 ed.; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Netzker, T.; Fischer, J.; Weber, J.; Mattern, D.J.; König, C.C.; Valiante, V.; Schroeckh, V.; Brakhage, A.A. Microbial communication leading to the activation of silent fungal secondary metabolite gene clusters. Front. Microbiol. 2015, 6, 299. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, S.; Bohni, N.; Schnee, S.; Schumpp, O.; Gindro, K.; Wolfender, J.L. Metabolite induction via microorganism co-culture: A potential way to enhance chemical diversity for drug discovery. Biotechnol. Adv. 2014, 32, 1180–1204. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–5 are available from the authors. |

| No. | 1 in Methanol-d4 | 1 in CDCl3 | ||
|---|---|---|---|---|
| δH, J (Hz) | δC a | HMBC Correlation | δH, J (Hz) | |
| 2 | 159.4 | |||
| 3 | 114.6 | |||
| 4 | 165.8 | |||
| 5 | 6.03, d, 7.6 | 101.5 | C-3, C-4 | 5.84, d, 7.6 |
| 6 | 7.73, dd, 7.6, 0.8 | 136.6 | C-2, C-4 | 7.37, dd, 7.6, 0.8 |
| 7 | 2.07, d, 11.6 | 50.3 | C-2, C-3, C-4, C-12, | 2.04, d, 11.6 |
| C-8, C-9, C-13, C-16 | ||||
| 8 | 2.69, m | 27.3 | 2.75, m | |
| 9a | 1.84, dt, 13.5, 4.0 | 44.2 | C-7, C-11, C-8, C-10, C-17 | 1.82, dt, 13.5, 4.0 |
| 9b | 0.85, br.d, 13.5 | C-7, C-11, C-8, C-10, C-17, C-18 | 0.82, br.d, 13.5 | |
| 10 | 1.69, m | 33.7 | 1.66, m | |
| 11 | 3.03, d, 10.4 | 84.7 | C-9, C-10, C-12, C-13, C-16, C-17 | 3.08, d, 10.3 |
| 12 | 45.8 | |||
| 13 | 4.10, dd, 10.8, 1.6 | 94.9 | C-4, C-11, C-12, C-14, C-15, C-16 | 4.06, dd, 10.9, 1.7 |
| 14a | 2.05, m | 27.4 | C-12, C-13, C-15 | 2.00, m |
| 14b | 1.27, m | C-13, C-15 | 1.25, m | |
| 15 | 1.12, t, 7.4 | 11.5 | C-13, C-14 | 1.08, t, 7.3 |
| 16 | 0.71, s | 9.8 | C-7, C-11, C-12, C-13 | 0.73, s |
| 17 | 0.99, d, 6.3 | 18.7 | C-9, C-10, C-11 | 0.97, d, 6.4 |
| 18 | 1.07, d, 6.0 | 23.7 | C-7, C-8, C-9 | 1.08, d, 5.9 |
| 19 | 3.98, s | 65.0 | 4.01, s | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, C.; Sarotti, A.M.; Yang, B.; Turkson, J.; Cao, S. A New N-methoxypyridone from the Co-Cultivation of Hawaiian Endophytic Fungi Camporesia sambuci FT1061 and Epicoccum sorghinum FT1062. Molecules 2017, 22, 1166. https://doi.org/10.3390/molecules22071166
Li C, Sarotti AM, Yang B, Turkson J, Cao S. A New N-methoxypyridone from the Co-Cultivation of Hawaiian Endophytic Fungi Camporesia sambuci FT1061 and Epicoccum sorghinum FT1062. Molecules. 2017; 22(7):1166. https://doi.org/10.3390/molecules22071166
Chicago/Turabian StyleLi, Chunshun, Ariel M. Sarotti, Baojun Yang, James Turkson, and Shugeng Cao. 2017. "A New N-methoxypyridone from the Co-Cultivation of Hawaiian Endophytic Fungi Camporesia sambuci FT1061 and Epicoccum sorghinum FT1062" Molecules 22, no. 7: 1166. https://doi.org/10.3390/molecules22071166
APA StyleLi, C., Sarotti, A. M., Yang, B., Turkson, J., & Cao, S. (2017). A New N-methoxypyridone from the Co-Cultivation of Hawaiian Endophytic Fungi Camporesia sambuci FT1061 and Epicoccum sorghinum FT1062. Molecules, 22(7), 1166. https://doi.org/10.3390/molecules22071166