Essential Oils of Oregano: Biological Activity beyond Their Antimicrobial Properties
Abstract
:1. Introduction
2. Essential Oils Composition of Oregano Species
3. Biological Activities of Essential Oils of Oregano Species
3.1. Antimicrobial Effect of Essential Oils of Oregano
3.2. Essential Oils of Oregano as Antioxidants
3.3. Anti-Inflammatory Activity of Essential Oils of Oregano Species
3.4. Essential Oils of Oregano Species and Cardiovascular Diseases
3.5. Essential Oils of Oregano and Their Effect on Metabolic Syndrome
3.6. Antiprolifertive and Citotoxic Activity of Essential Oils of Oregano
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gooch, J.W. Essential oils. In Encyclopedic Dictionary of Polymers; Gooch, J.W., Ed.; Springer: New York, NY, USA, 2011; p. 274. [Google Scholar]
- Dima, C.; Dima, S. Essential oils in foods: Extraction, stabilization, and toxicity. Curr. Opin. Food Sci. 2015, 5, 29–35. [Google Scholar] [CrossRef]
- Shaaban, H.A.E.; El-Ghorab, A.H.; Shibamoto, T. Bioactivity of essential oils and their volatile aroma components: Review. J. Essent. Oil Res. 2012, 24, 203–212. [Google Scholar] [CrossRef]
- Baser, K.H.C.; Buchbauer, G. Handbook of Essential Oils: Science, Technology, and Applications; CRC Press: Boca Raton, FL, USA, 2015; p. 975. ISBN 978-1-4200-6315-8. [Google Scholar]
- Franz, C.; Novak, J. 3 Sources of essential oils. In Handbook of Essential Oils: Science, Technology, and Applications; Baser, K.H.C., Buchbauer, G., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 39–82. ISBN 978-1-4200-6315-8. [Google Scholar]
- Patel, S.; Gogna, P. Tapping botanicals for essential oils: Progress and hurdles in cancer mitigation. Ind. Crop. Prod. 2015, 76, 1148–1163. [Google Scholar] [CrossRef]
- Zuzarte, M.; Salgueiro, L. Essential oils chemistry. In Bioactive Essential Oils and Cancer; De Sousa, D.P., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 19–61. [Google Scholar]
- Islam, M.T.; Da Mata, A.M.O.F.; de Aguiar, R.P.S.; Paz, M.F.C.J.; de Alencar, M.V.O.B.; Ferreira, P.M.P.; de Carvalho Melo-Cavalcante, A.A. Therapeutic potential of essential oils focusing on diterpenes. Phytother. Res. 2016, 30, 1420–1444. [Google Scholar] [CrossRef] [PubMed]
- Lenardão, E.J.; Savegnago, L.; Jacob, R.G.; Victoria, F.N.; Martinez, D.M. Antinociceptive effect of essential oils and their constituents: An update review. J. Braz. Chem. Soc. 2016, 27, 435–474. [Google Scholar] [CrossRef]
- Adorjan, B.; Buchbauer, G. Biological properties of essential oils: An updated review. Flavour Fragr. J. 2010, 25, 407–426. [Google Scholar] [CrossRef]
- Edris, A.E. Pharmaceutical and therapeutic potentials of essential oils and their individual volatile constituents: A review. Phytother. Res. 2007, 21, 308–323. [Google Scholar] [CrossRef] [PubMed]
- Miguel, M.G. Antioxidant and anti-inflammatory activities of essential oils: A short review. Molecules 2010, 15, 9252–9287. [Google Scholar] [CrossRef] [PubMed]
- Calpouzos, L. Botanical aspects of oregano. Econ. Bot. 1954, 8, 222–233. [Google Scholar] [CrossRef]
- Kintzios, S.E. 21 Oregano. In Handbook of Herbs and Spices, 2nd ed.; Peter, K.V., Ed.; Woodhead Publishing: Cambridge, UK, 2012; pp. 417–436. [Google Scholar]
- Pascual, M.E.; Slowing, K.; Carretero, E.; Sánchez Mata, D.; Villar, A. Lippia: Traditional uses, chemistry and pharmacology: A review. J. Ethnopharmacol. 2001, 76, 201–214. [Google Scholar] [CrossRef]
- Leyva-López, N.; Nair, V.; Bang, W.Y.; Cisneros-Zevallos, L.; Heredia, J.B. Protective role of terpenes and polyphenols from three species of oregano (Lippia graveolens, Lippia palmeri and Hedeoma patens) on the suppression of lipopolysaccharide-induced inflammation in raw 264.7 macrophage cells. J. Ethnopharmacol. 2016, 187, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Karioti, A.; Milošević-Ifantis, T.; Pachopos, N.; Niryiannaki, N.; Hadjipavlou-Litina, D.; Skaltsa, H. Antioxidant, anti-inflammatory potential and chemical constituents of Origanum dubium boiss., growing wild in cyprus. J. Enzym. Inhib. Med. Chem. 2015, 30, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Economou, G.; Panagopoulos, G.; Tarantilis, P.; Kalivas, D.; Kotoulas, V.; Travlos, I.S.; Polysiou, M.; Karamanos, A. Variability in essential oil content and composition of Origanum hirtum L., Origanum onites L., Coridothymus capitatus (L.) and Satureja thymbra L. populations from the Greek island Ikaria. Ind. Crop. Prod. 2011, 33, 236–241. [Google Scholar] [CrossRef]
- Lukas, B.; Schmiderer, C.; Novak, J. Essential oil diversity of European Origanum vulgare L. (Lamiaceae). Phytochemistry 2015, 119, 32–40. [Google Scholar] [CrossRef] [PubMed]
- González-Fuentes, F.; López-Gil, M.Á.; Mendoza, S.; Escarpa, A. Electrochemical screening of biomarkers in chemotype Mexican oregano oils on single-walled carbon nanotubes screen-printed electrodes. Electroanalysis 2011, 23, 2212–2216. [Google Scholar] [CrossRef]
- Arslan, M.; Uremis, I.; Demirel, N. Effects of sage leafhopper feeding on herbage colour, essential oil content and compositions of turkish and Greek oregano. Exp. Agric. 2012, 48, 428–437. [Google Scholar] [CrossRef]
- Asensio, C.M.; Grosso, N.R.; Juliani, H.R. Quality characters, chemical composition and biological activities of oregano (Origanum spp.) essential oils from central and southern Argentina. Ind. Crop. Prod. 2015, 63, 203–213. [Google Scholar] [CrossRef]
- Azizi, A.; Yan, F.; Honermeier, B. Herbage yield, essential oil content and composition of three oregano (Origanum vulgare L.) populations as affected by soil moisture regimes and nitrogen supply. Ind. Crop. Prod. 2009, 29, 554–561. [Google Scholar] [CrossRef]
- Baranauskienė, R.; Venskutonis, P.R.; Dambrauskienė, E.; Viškelis, P. Harvesting time influences the yield and oil composition of Origanum vulgare L. ssp. vulgare and ssp. hirtum. Ind. Crop. Prod. 2013, 49, 43–51. [Google Scholar] [CrossRef]
- Gerami, F.; Moghaddam, P.R.; Ghorbanim, R.; Hassani, A. Effects of irrigation intervals and organic manure on morphological traits, essential oil content and yield of oregano (Origanum vulgare L.). An. Acad. Bras. Cienc. 2016, 88, 2375–2385. [Google Scholar] [CrossRef] [PubMed]
- De Falco, E.; Mancini, E.; Roscigno, G.; Mignola, E.; Taglialatela-Scafati, O.; Senatore, F. Chemical composition and biological activity of essential oils of Origanum vulgare L. subsp. vulgare L. under different growth conditions. Molecules 2013, 18, 14948. [Google Scholar] [PubMed]
- Bolechowski, A.; Moral, R.; Bustamante, M.A.; Paredes, C.; Agullo, E.; Bartual, J.; Carbonell-Barrachina, A.A. Composition of oregano essential oil (Origanum vulgare) as affected by the use of winery-distillery composts. J. Essent. Oil Res. 2011, 23, 32–38. [Google Scholar] [CrossRef]
- Napoli, E.; Mazzaglia, A.; Restuccia, C.; Ragni, P.; Lanza, C.M.; Ruberto, G. The effect of γ-irradiation on chemical composition, microbial load and sensory properties of sicilian oregano. LWT Food Sci. Technol. 2016, 72, 566–572. [Google Scholar] [CrossRef]
- Morshedloo, M.R.; Craker, L.E.; Salami, A.; Nazeri, V.; Sang, H.; Maggi, F. Effect of prolonged water stress on essential oil content, compositions and gene expression patterns of mono- and sesquiterpene synthesis in two oregano (Origanum vulgare L.) subspecies. Plant Physiol. Biochem. 2017, 111, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Novak, I.; Sipos, L.; Kokai, Z.; Szabo, K.; Pluhar, Z.; Sarosi, S. Effect of the drying method on the composition of Origanum vulgare L. subsp hirtum essential oil analysed by gc-ms and sensory profile method. Acta Aliment. 2011, 40, 130–138. [Google Scholar] [CrossRef]
- Figiel, A.; Szumny, A.; Gutiérrez-Ortíz, A.; Carbonell-Barrachina, Á.A. Composition of oregano essential oil (Origanum vulgare) as affected by drying method. J. Food Eng. 2010, 98, 240–247. [Google Scholar] [CrossRef]
- Calín-Sánchez, Á.; Figiel, A.; Lech, K.; Szumny, A.; Martínez-Tomé, J.; Carbonell-Barrachina, Á.A. Dying methods affect the aroma of Origanum majorana L. analyzed by gc–ms and descriptive sensory analysis. Ind. Crop. Prod. 2015, 74, 218–227. [Google Scholar] [CrossRef]
- Sözmen, F.; Uysal, B.; Köse, E.O.; Aktaş, Ö.; Cinbilgel, I.; Oksal, B.S. Extraction of the essential oil from endemic Origanum bilgeri P.H.Davis with two different methods: Comparison of the oil composition and antibacterial activity. Chem. Biodivers. 2012, 9, 1356–1363. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, S.M.B.; Nikmaram, N.; Esteghlal, S.; Mousavi Khaneghah, A.; Niakousari, M.; Barba, F.J.; Roohinejad, S.; Koubaa, M. Efficiency of ohmic assisted hydrodistillation for the extraction of essential oil from oregano (Origanum vulgare subsp. viride) spices. Innov. Food Sci. Emerg. Technol. 2017, 41, 172–178. [Google Scholar] [CrossRef]
- Zheljazkov, V.D.; Astatkie, T.; Schlegel, V. Distillation time changes oregano essential oil yields and composition but not the antioxidant or antimicrobial activities. HortScience 2012, 47, 777–784. [Google Scholar]
- Stamenic, M.; Vulic, J.; Djilas, S.; Misic, D.; Tadic, V.; Petrovic, S.; Zizovic, I. Free-radical scavenging activity and antibacterial impact of Greek oregano isolates obtained by sfe. Food Chem. 2014, 165, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Han, F.; Ma, G.-Q.; Yang, M.; Yan, L.; Xiong, W.; Shu, J.-C.; Zhao, Z.-D.; Xu, H.-L. Chemical composition and antioxidant activities of essential oils from different parts of the oregano. J. Zhejiang Univ. Sci. B 2017, 18, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Sarrazin, S.L.F.; Oliveira, R.B.; Barata, L.E.S.; Mourão, R.H.V. Chemical composition and antimicrobial activity of the essential oil of Lippia grandis schauer (verbenaceae) from the western amazon. Food Chem. 2012, 134, 1474–1478. [Google Scholar] [CrossRef] [PubMed]
- Betancourt, L.; Phandanauvong, V.; Patiño, R.; Ariza-Nieto, C.; Afanador-Téllez, G. Composition and bactericidal activity against beneficial and pathogenic bacteria of oregano essential oils from four chemotypes of Origanum and Lippia genus. Rev. Med. Vet. Zootec 2012, 59, 21–31. [Google Scholar]
- Ortega-Nieblas, M.M.; Robles-Burgueño, M.R.; Acedo-Félix, E.; González-León, A.; Morales-Trejo, A.; Vázquez-Moreno, L. Chemical composition and antimicrobial activity of oregano (Lippia palmeri S. Wats) essential oil. Rev. Fitotec. Mex. 2011, 34, 11–17. [Google Scholar]
- Figueredo, G.; Ozcan, M.M.; Chalchat, J.C.; Bagci, Y.; Chalard, P. Chemical composition of essential oil of Hyssopus officinalis L. and Origanum acutidens. J. Essent. Oil Bear. Plant 2012, 15, 300–306. [Google Scholar] [CrossRef]
- Goze, I.; Alim, A.; Cetinus, S.A.; Cetin, A.; Durmus, N.; Atas, A.T.; Vural, N. In vitro antimicrobial, antioxidant, and antispasmodic activities and the composition of the essential oil of Origanum acutidens (hand.-mazz.) ietswaart. J. Med. Food 2010, 13, 705–709. [Google Scholar] [CrossRef] [PubMed]
- Dambolena, J.S.; Zunino, M.P.; Lucini, E.I.; Olmedo, R.; Banchio, E.; Bima, P.J.; Zygadlo, J.A. Total phenolic content, radical scavenging properties, and essential oil composition of origanum species from different populations. J. Agric. Food Chem. 2010, 58, 1115–1120. [Google Scholar] [CrossRef] [PubMed]
- Al Hafi, M.; El Beyrouthy, M.; Ouaini, N.; Stien, D.; Rutledge, D.; Chaillou, S. Chemical composition and antimicrobial activity of Origanum libanoticum, Origanum ehrenbergii, and Origanum syriacum growing wild in Lebanon. Chem. Biodivers. 2016, 13, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Waller, S.B.; Madrid, I.M.; Silva, A.L.; Dias de Castro, L.L.; Cleff, M.B.; Ferraz, V.; Meireles, M.C.A.; Zanette, R.; de Mello, J.R.B. In vitro susceptibility of Sporothrix brasiliensis to essential oils of Lamiaceae family. Mycopathologia 2016, 181, 857–863. [Google Scholar] [CrossRef] [PubMed]
- Karabörklü, S.; Ayvaz, A.; Yilmaz, S.; Akbulut, M. Chemical composition and fumigant toxicity of some essential oils against Ephestia kuehniella. J. Econ. Entomol. 2011, 104, 1212–1219. [Google Scholar] [CrossRef] [PubMed]
- Ocak, I.; Celik, A.; Ozel, M.Z.; Korcan, E.; Konuk, M. Antifungal activity and chemical composition of essential oil of Origanum hypericifolium. Int. J. Food Prop. 2012, 15, 38–48. [Google Scholar] [CrossRef]
- Rodríguez-Solana, R.; Daferera, D.J.; Mitsi, C.; Trigas, P.; Polissiou, M.; Tarantilis, P.A. Comparative chemotype determination of lamiaceae plants by means of gc–ms, ft-ir, and dispersive-raman spectroscopic techniques and gc-fid quantification. Ind. Crop. Prod. 2014, 62, 22–33. [Google Scholar] [CrossRef]
- Stefanaki, A.; Cook, C.M.; Lanaras, T.; Kokkini, S. The oregano plants of chios island (Greece): Essential oils of Origanum onites L. growing wild in different habitats. Ind. Crop. Prod. 2016, 82, 107–113. [Google Scholar] [CrossRef]
- Ozkan, G.; Baydar, H.; Erbas, S. The influence of harvest time on essential oil composition, phenolic constituents and antioxidant properties of turkish oregano (Origanum onites L.). J. Sci. Food Agric. 2010, 90, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Gendy, A.N.E.; Leonardi, M.; Mugnaini, L.; Bertelloni, F.; Ebani, V.V.; Nardoni, S.; Mancianti, F.; Hendawy, S.; Omer, E.; Pistelli, L. Chemical composition and antimicrobial activity of essential oil of wild and cultivated Origanum syriacum plants grown in Sinai, Egypt. Ind. Crop. Prod. 2015, 67, 201–207. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; El Gendy, A.E.-N.G.S.; Sendra, E.; Fernández-López, J.; Abd El Razik, K.A.; Omer, E.A.; Pérez-Alvarez, J.A. Chemical composition and antioxidant and anti-listeria activities of essential oils obtained from some Egyptian plants. J. Agric. Food Chem. 2010, 58, 9063–9070. [Google Scholar] [CrossRef] [PubMed]
- Berrehal, D.; Boudiar, T.; Hichem, L.; Khalfallah, A.; Kabouche, A.; Al-Freihat, A.; Ghannadi, A.; Sajjadi, E.; Mehrabani, M.; Safaei-Ghomi, J.; et al. Comparative composition of four essential oils of oregano used in Algerian and Jordanian folk medicine. Nat. Prod. Commun. 2010, 5, 957–960. [Google Scholar] [PubMed]
- Werdin González, J.O.; Gutiérrez, M.M.; Murray, A.P.; Ferrero, A.A. Composition and biological activity of essential oils from labiatae against nezara viridula (hemiptera: Pentatomidae) soybean pest. Pest Manag. Sci. 2011, 67, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Martucci, J.F.; Gende, L.B.; Neira, L.M.; Ruseckaite, R.A. Oregano and lavender essential oils as antioxidant and antimicrobial additives of biogenic gelatin films. Ind. Crop. Prod. 2015, 71, 205–213. [Google Scholar] [CrossRef]
- Olmedo, R.; Nepote, V.; Grosso, N.R. Antioxidant activity of fractions from oregano essential oils obtained by molecular distillation. Food Chem. 2014, 156, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Quiroga, P.R.; Grosso, N.R.; Lante, A.; Lomolino, G.; Zygadlo, J.A.; Nepote, V. Chemical composition, antioxidant activity and anti-lipase activity of Origanum vulgare and Lippia turbinata essential oils. Int. J. Food Sci. Technol. 2013, 48, 642–649. [Google Scholar] [CrossRef]
- Wei, H.-K.; Chen, G.; Wang, R.-J.; Peng, J. Oregano essential oil decreased susceptibility to oxidative stress-induced dysfunction of intestinal epithelial barrier in rats. J. Funct. Foods 2015, 18, 1191–1199. [Google Scholar] [CrossRef]
- Elizalde, J.J.; Espinoza, M. Effect of ionizing irradiation on Origanum leaves (Origanum vulgare L.) essential oil composition. J. Essent. Oil Bear. Plants 2011, 14, 164–171. [Google Scholar] [CrossRef]
- Gong, H.Y.; Liu, W.H.; Lv, G.Y.; Zhou, X. Analysis of essential oils of Origanum vulgare from six production areas of china and Pakistan. Rev. Bras. Farmacogn. 2014, 24, 25–32. [Google Scholar] [CrossRef]
- Thomidis, T.; Filotheou, A. Evaluation of five essential oils as bio-fungicides on the control of pilidiella granati rot in pomegranate. Crop Protect. 2016, 89, 66–71. [Google Scholar] [CrossRef]
- Verma, R.S.; Rahman, L.; Verma, R.K.; Chanotiya, C.S.; Chauhan, A.; Yadav, A.; Yadav, A.K.; Singh, A. Changes in the essential oil content and composition of Origanum vulgare L. during annual growth from kumaon himalaya. Curr. Sci. 2010, 98, 1010–1012. [Google Scholar]
- Pirigharnaei, M.; Zare, S.; Heidari, R.; Khara, J.; Sabzi, R.E.; Kheiry, F. The essential oil composition of Iranian oregano (Origanum vulgare L.) populations in field and provenance from piranshahr district, west azarbaijan province, Iran. J. Food Agric. Environ. 2011, 9, 89–93. [Google Scholar]
- Fratini, F.; Mancini, S.; Turchi, B.; Friscia, E.; Pistelli, L.; Giusti, G.; Cerri, D. A novel interpretation of the fractional inhibitory concentration index: The case Origanum vulgare L. and Leptospermum scoparium J.R. et G. forst essential oils against Staphylococcus aureus strains. Microbiol. Res. 2017, 195, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Pesavento, G.; Calonico, C.; Bilia, A.R.; Barnabei, M.; Calesini, F.; Addona, R.; Mencarelli, L.; Carmagnini, L.; Di Martino, M.C.; Lo Nostro, A. Antibacterial activity of oregano, rosmarinus and thymus essential oils against Staphylococcus aureus and Listeria monocytogenes in beef meatballs. Food Control 2015, 54, 188–199. [Google Scholar] [CrossRef]
- Fouad, R.; Bousta, D.; Lalami, A.E.; Chahdi, F.O.; Amri, I.; Jamoussi, B.; Greche, H. Chemical composition and herbicidal effects of essential oils of Cymbopogon citratus (dc) stapf, Eucalyptus cladocalyx, Origanum vulgare L and Artemisia absinthium L cultivated in Morocco. J. Essent. Oil Bear. Plants 2015, 18, 112–123. [Google Scholar] [CrossRef]
- Teixeira, B.; Marques, A.; Ramos, C.; Serrano, C.; Matos, O.; Neng, N.R.; Nogueira, J.M.F.; Saraiva, J.A.; Nunes, M.L. Chemical composition and bioactivity of different oregano (Origanum vulgare) extracts and essential oil. J. Sci. Food Agric. 2013, 93, 2707–2714. [Google Scholar] [CrossRef] [PubMed]
- Ličina, B.Z.; Stefanović, O.D.; Vasić, S.M.; Radojević, I.D.; Dekić, M.S.; Čomić, L.R. Biological activities of the extracts from wild growing Origanum vulgare L. Food Control 2013, 33, 498–504. [Google Scholar] [CrossRef]
- Soković, M.; Glamočlija, J.; Marin, P.D.; Brkić, D.; Van Griensven, L.J. Antibacterial effects of the essential oils of commonly consumed medicinal herbs using an in vitro model. Molecules 2010, 15, 7532–7546. [Google Scholar] [CrossRef] [PubMed]
- Stojković, D.; Glamočlija, J.; Ćirić, A.; Nikolić, M.; Ristić, M.; Šiljegović, J.; Soković, M. Investigation on antibacterial synergism of Origanum vulgare and Thymus vulgaris essential oils. Arch. Biol. Sci. 2013, 65, 639–643. [Google Scholar] [CrossRef]
- Boskovic, M.; Zdravkovic, N.; Ivanovic, J.; Janjic, J.; Djordjevic, J.; Starcevic, M.; Baltic, M.Z. Antimicrobial activity of thyme (Thymus vulgaris) and oregano (Origanum vulgare) essential oils against some food-borne microorganisms. Procedia Food Sci. 2015, 5, 18–21. [Google Scholar] [CrossRef]
- Mechergui, K.; Coelho, J.A.; Serra, M.C.; Lamine, S.B.; Boukhchina, S.; Khouja, M.L. Essential oils of Origanum vulgare L. subsp. glandulosum (desf.) ietswaart from Tunisia: Chemical composition and antioxidant activity. J. Sci. Food Agric. 2010, 90, 1745–1749. [Google Scholar] [PubMed]
- Mechergui, K.; Jaouadi, W.; Coelho, J.P.; Khouja, M.L. Effect of harvest year on production, chemical composition and antioxidant activities of essential oil of oregano (Origanum vulgare subsp glandulosum (desf.) ietswaart) growing in north africa. Ind. Crop. Prod. 2016, 90, 32–37. [Google Scholar] [CrossRef]
- Béjaoui, A.; Chaabane, H.; Jemli, M.; Boulila, A.; Boussaid, M. Essential oil composition and antibacterial activity of Origanum vulgare subsp. glandulosum desf. at different phenological stages. J. Med. Food 2013, 16, 1115–1120. [Google Scholar] [PubMed]
- Kilic, Ö.; Özdemir, F.A. Variability of essential oil composition of Origanum vulgare L. subsp. gracile populations from Turkey. J. Essent. Oil Bear. Plants 2016, 19, 2083–2090. [Google Scholar] [CrossRef]
- Grondona, E.; Gatti, G.; Lopez, A.G.; Sanchez, L.R.; Rivero, V.; Pessah, O.; Zunino, M.P.; Ponce, A.A. Bio-efficacy of the essential oil of oregano (Origanum vulgare lamiaceae. Ssp hirtum). Plant Food Hum. Nutr. 2014, 69, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Paraskevakis, N.; Tsiplakou, E.; Daferera, D.; Sotirakoglou, K.; Polissiou, M.; Zervas, G. Changes in essential oil content and composition of Origanum vulgare spp. hirtum during storage as a whole plant or after grinding and mixing with a concentrate ruminant diet. J. Essent. Oil Res. 2015, 27, 264–270. [Google Scholar]
- Karamanos, A.J.; Sotiropoulou, D.E.K. Field studies of nitrogen application on Greek oregano (Origanum vulgare ssp. hirtum (link) ietswaart) essential oil during two cultivation seasons. Ind. Crop. Prod. 2013, 46, 246–252. [Google Scholar]
- Tibaldi, G.; Fontana, E.; Nicola, S. Growing conditions and postharvest management can affect the essential oil of Origanum vulgare L. ssp. hirtum (link) ietswaart. Ind. Crop. Prod. 2011, 34, 1516–1522. [Google Scholar] [CrossRef]
- Spagnoletti, A.; Guerrini, A.; Tacchini, M.; Vinciguerra, V.; Leone, C.; Maresca, I.; Simonetti, G.; Sacchetti, G.; Angiolella, L. Chemical composition and bio-efficacy of essential oils from italian aromatic plants: Mentha suaveolens, Coridothymus capitatus, Origanum hirtum and Rosmarinus officinalis. Nat. Prod. Commun. 2016, 11, 1517–1520. [Google Scholar]
- Bonfanti, C.; Iannì, R.; Mazzaglia, A.; Lanza, C.M.; Napoli, E.M.; Ruberto, G. Emerging cultivation of oregano in sicily: Sensory evaluation of plants and chemical composition of essential oils. Ind. Crop. Prod. 2012, 35, 160–165. [Google Scholar] [CrossRef]
- Mancini, E.; Camele, I.; Elshafie, H.S.; de Martino, L.; Pellegrino, C.; Grulova, D.; de Feo, V. Chemical composition and biological activity of the essential oil of Origanum vulgare ssp. hirtum from different areas in the southern apennines (Italy). Chem. Biodivers. 2014, 11, 639–651. [Google Scholar] [PubMed]
- Sarikurkcu, C.; Zengin, G.; Oskay, M.; Uysal, S.; Ceylan, R.; Aktumsek, A. Composition, antioxidant, antimicrobial and enzyme inhibition activities of two Origanum vulgare subspecies (subsp. vulgare and subsp. hirtum) essential oils. Ind. Crop. Prod. 2015, 70, 178–184. [Google Scholar] [CrossRef]
- Vale-Silva, L.; Silva, M.-J.; Oliveira, D.; Gonçalves, M.-J.; Cavaleiro, C.; Salgueiro, L.; Pinto, E. Correlation of the chemical composition of essential oils from Origanum vulgare subsp. virens with their in vitro activity against pathogenic yeasts and filamentous fungi. J. Med. Microbiol. 2012, 61, 252–260. [Google Scholar] [PubMed]
- Vazirian, M.; Mohammadi, M.; Farzaei, M.H.; Amin, G.; Amanzadeh, Y. Chemical composition and antioxidant activity of origanum vulgare subsp. vulgare essential oil from Iran. Res. J. Pharmacogn. 2015, 2, 41–46. [Google Scholar]
- Nurzynska-Wierdak, R.; Bogucka-Kocka, A.; Sowa, I.; Szymczak, G. The composition of essential oil from three ecotypes of Origanum vulgare L. ssp vulgare cultivated in Poland. Farmacia 2012, 60, 571–577. [Google Scholar]
- Rodriguez-Garcia, I.; Silva-Espinoza, B.A.; Ortega-Ramirez, L.A.; Leyva, J.M.; Siddiqui, M.W.; Cruz-Valenzuela, M.R.; Gonzalez-Aguilar, G.A.; Ayala-Zavala, J.F. Oregano essential oil as an antimicrobial and antioxidant additive in food products. Crit. Rev. Food Sci. Nutr. 2016, 56, 1717–1727. [Google Scholar] [CrossRef] [PubMed]
- Adame-Gallegos, J.R.; Andrade-Ochoa, S.; Nevarez-Moorillon, G.V. Potential use of Mexican oregano essential oil against parasite, fungal and bacterial pathogens. J. Essent. Oil Bear. Plant 2016, 19, 553–567. [Google Scholar] [CrossRef]
- Jayasena, D.D.; Jo, C. Essential oils as potential antimicrobial agents in meat and meat products: A review. Trends Food Sci. Technol. 2013, 34, 96–108. [Google Scholar] [CrossRef]
- Arana-Sánchez, A.; Estarrón-Espinosa, M.; Obledo-Vázquez, E.N.; Padilla-Camberos, E.; Silva-Vázquez, R.; Lugo-Cervantes, E. Antimicrobial and antioxidant activities of Mexican oregano essential oils (Lippia graveolens h. B.K.) with different composition when microencapsulated inβ-cyclodextrin. Lett. Appl. Microbiol. 2010, 50, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Benavides, S.; Villalobos-Carvajal, R.; Reyes, J.E. Physical, mechanical and antibacterial properties of alginate film: Effect of the crosslinking degree and oregano essential oil concentration. J. Food Eng. 2012, 110, 232–239. [Google Scholar] [CrossRef]
- Emiroğlu, Z.K.; Yemiş, G.P.; Coşkun, B.K.; Candoğan, K. Antimicrobial activity of soy edible films incorporated with thyme and oregano essential oils on fresh ground beef patties. Meat Sci. 2010, 86, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Hernández, H.; Fraňková, A.; Sýkora, T.; Klouček, P.; Kouřimská, L.; Kučerová, I.; Banout, J. The effect of oregano essential oil on microbial load and sensory attributes of dried meat. J. Sci. Food Agric. 2017, 97, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Circu, M.L.; Aw, T.Y. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic. Biol. Med. 2010, 48, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteridge, J.M.C. Chapter 4: Cellular responses to oxidative stress: Adaptation, damage, repair, senescence and death. In Free Radicals in Biology and Medicine; Halliwell, B., Gutteridge, J.M.C., Eds.; Oxford University Press: Oxford, UK, 2007; Volume 4, pp. 187–267. ISBN 978-0-19-856869-8. [Google Scholar]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vascul. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Patel, A.K.; Shah, N.; Chaudhary, A.; Jha, U.; Yadav, U.C. Oxidative stress and antioxidants in disease and cancer: A review. Asian Pac. Cancer Prev. 2014, 15, 4405–4409. [Google Scholar] [CrossRef]
- Cazzola, R.; Cestaro, B. Chapter 9: Antioxidant spices and herbs used in diabetes. In Diabetes: Oxidative Stress and Dietary Antioxidants; Preedy, V.R., Ed.; Academic Press: San Diego, CA, USA, 2014; pp. 89–97. [Google Scholar]
- Gonzalez-Burgos, E.; Gomez-Serranillos, M.P. Terpene compounds in nature: A review of their potential antioxidant activity. Curr. Med. Chem. 2012, 19, 5319–5341. [Google Scholar] [CrossRef] [PubMed]
- Conforti, F.; Marrelli, M.; Menichini, F.; Tundis, R.; Statti, G.A.; Solimene, U.; Menichini, F. Chemical composition and protective effect of oregano (Origanum heracleoticum L.) ethanolic extract on oxidative damage and on inhibition of NO in LPS-stimulated raw 264.7 macrophages. J. Enzym. Inhib. Med. Chem. 2011, 26, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Tuttolomondo, T.; La Bella, S.; Licata, M.; Virga, G.; Leto, C.; Saija, A.; Trombetta, D.; Tomaino, A.; Speciale, A.; Napoli, E.M.; et al. Biomolecular characterization of wild sicilian oregano: Phytochemical screening of essential oils and extracts, and evaluation of their antioxidant activities. Chem. Biodivers. 2013, 10, 411–433. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.; Wei, H.; Sun, H.; Ao, J.; Long, G.; Jiang, S.; Peng, J. Effects of dietary supplementation of oregano essential oil to sows on oxidative stress status, lactation feed intake of sows, and piglet performance. Biomed. Res. Int. 2015, 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, L.R. Role of plant polyphenols in genomic stability. Mut. Res. Fund. Mol. Mutagen. 2001, 475, 89–111. [Google Scholar] [CrossRef]
- León-González, A.J.; Auger, C.; Schini-Kerth, V.B. Pro-oxidant activity of polyphenols and its implication on cancer chemoprevention and chemotherapy. Biochem. Pharmacol. 2015, 98, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Llana-Ruiz-Cabello, M.; Gutiérrez-Praena, D.; Puerto, M.; Pichardo, S.; Jos, Á.; Cameán, A.M. In vitro pro-oxidant/antioxidant role of carvacrol, thymol and their mixture in the intestinal caco-2 cell line. Toxicol. In Vitro 2015, 29, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Terenina, M.B.; Misharina, T.A.; Krikunova, N.I.; Alinkina, E.S.; Fatkulina, L.D.; Vorob’yova, A.K. Oregano essential oil as an inhibitor of higher fatty acid oxidation. Appl. Biochem. Microbiol. 2011, 47, 445–449. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Menichini, F.; Conforti, F.; Tundis, R.; Bonesi, M.; Saab, A.M.; Statti, G.A.; Cindio, B.d.; Houghton, P.J.; Menichini, F.; et al. Chemical analysis, antioxidant, antiinflammatory and anticholinesterase activities of Origanum ehrenbergii boiss and Origanum syriacum L. essential oils. Food Chem. 2009, 117, 174–180. [Google Scholar] [CrossRef]
- Suhaj, M. Spice antioxidants isolation and their antiradical activity: A review. J. Food Comp. Anal. 2006, 19, 531–537. [Google Scholar] [CrossRef]
- Olmedo, R.H.; Nepote, V.; Grosso, N.R. Preservation of sensory and chemical properties in flavoured cheese prepared with cream cheese base using oregano and rosemary essential oils. LWT Food Sci. Technol. 2013, 53, 409–417. [Google Scholar] [CrossRef]
- Gavaric, N.; Mozina, S.S.; Kladar, N.; Bozin, B. Chemical profile, antioxidant and antibacterial activity of thyme and oregano essential oils, thymol and carvacrol and their possible synergism. J. Essent. Oil Bear. Plants 2015, 18, 1013–1021. [Google Scholar] [CrossRef]
- El Babili, F.; Bouajila, J.; Souchard, J.P.; Bertrand, C.; Bellvert, F.; Fouraste, I.; Moulis, C.; Valentin, A. Oregano: Chemical analysis and evaluation of its antimalarial, antioxidant, and cytotoxic activities. J. Food Sci. 2011, 76, C512–C518. [Google Scholar] [CrossRef] [PubMed]
- Alinkina, E.S.; Misharina, T.A.; Fatkullina, L.D. Antiradical properties of oregano, thyme, and savory essential oils. Appl. Biochem. Microbiol. 2013, 49, 73–78. [Google Scholar] [CrossRef]
- Quiroga, P.R.; Riveros, C.G.; Zygadlo, J.A.; Grosso, N.R.; Nepote, V. Antioxidant activity of essential oil of oregano species from Argentina in relation to their chemical composition. Int. J. Food Sci. Technol. 2011, 46, 2648–2655. [Google Scholar] [CrossRef]
- Borgarello, A.V.; Mezza, G.N.; Soltermann, A.T.; Pramparo, M.C. Use of a free radical scavenging method on extracts obtained by molecular distillation from oregano essential oil. Lat. Am. Appl. Res. 2014, 44, 25–30. [Google Scholar]
- Karakaya, S.; El, S.N.; Karagözlü, N.; Şahin, S. Antioxidant and antimicrobial activities of essential oils obtained from oregano (Origanum vulgare ssp. hirtum) by using different extraction methods. J. Med. Food 2011, 14, 645–652. [Google Scholar] [PubMed]
- Boroski, M.; Giroux, H.J.; Sabik, H.; Petit, H.V.; Visentainer, J.V.; Matumoto-Pintro, P.T.; Britten, M. Use of oregano extract and oregano essential oil as antioxidants in functional dairy beverage formulations. LWT Food Sci. Technol. 2012, 47, 167–174. [Google Scholar] [CrossRef]
- Marrelli, M.; Conforti, F.; Formisano, C.; Rigano, D.; Arnold, N.A.; Menichini, F.; Senatore, F. Composition, antibacterial, antioxidant and antiproliferative activities of essential oils from three origanum species growing wild in Lebanon and Greece. Nat. Prod. Res. 2016, 30, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Castilho, P.C.; Savluchinske-Feio, S.; Weinhold, T.S.; Gouveia, S.C. Evaluation of the antimicrobial and antioxidant activities of essential oils, extracts and their main components from oregano from Madeira island, Portugal. Food Control 2012, 23, 552–558. [Google Scholar] [CrossRef]
- Almeida, A.P.; Rodriguez-Rojo, S.; Serra, A.T.; Vila-Real, H.; Simplicio, A.L.; Delgadilho, I.; Da Costa, S.B.; Da Costa, L.B.; Nogueira, I.D.; Duarte, C.M.M. Microencapsulation of oregano essential oil in starch-based materials using supercritical fluid technology. Innov. Food Sci. Emerg. Technol. 2013, 20, 140–145. [Google Scholar] [CrossRef]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Abbas, A.K.; Aster, J.C. Robbins Basic Pathology; Elsevier Health Sciences: New York, NY, USA, 2013. [Google Scholar]
- Hansson, G.K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 2005, 352, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Trinchieri, G. Cancer and inflammation: An old intuition with rapidly evolving new concepts. Annu. Rev. Immunol. 2012, 30, 677–706. [Google Scholar] [CrossRef] [PubMed]
- Arranz, E.; Jaime, L.; López de las Hazas, M.C.; Reglero, G.; Santoyo, S. Supercritical fluid extraction as an alternative process to obtain essential oils with anti-inflammatory properties from marjoram and sweet basil. Ind. Crop. Prod. 2015, 67, 121–129. [Google Scholar] [CrossRef]
- Han, X.; Parker, T.L. Anti-inflammatory, tissue remodeling, immunomodulatory, and anticancer activities of oregano (Origanum vulgare) essential oil in a human skin disease model. Biochim. Open 2017, 4, 73–77. [Google Scholar] [CrossRef]
- Lima, M.D.S.; Quintans-Júnior, L.J.; De Santana, W.A.; Martins Kaneto, C.; Pereira Soares, M.B.; Villarreal, C.F. Anti-inflammatory effects of carvacrol: Evidence for a key role of interleukin-10. Eur. J. Pharmacol. 2013, 699, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Choi, Y.; Park, S.; Park, T. Carvacrol prevents diet-induced obesity by modulating gene expressions involved in adipogenesis and inflammation in mice fed with high-fat diet. J. Nutr. Biochem. 2012, 23, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Rajan, B.; Ravikumar, R.; Premkumar, T.; Devaki, T. Carvacrol attenuates n-nitrosodiethylamine induced liver injury in experimental wistar rats. Food Sci. Hum. Wellness 2015, 4, 66–74. [Google Scholar] [CrossRef]
- Silva, F.V.; Guimarães, A.G.; Silva, E.R.S.; Sousa-Neto, B.P.; Machado, F.D.F.; Quintans-Júnior, L.J.; Arcanjo, D.D.R.; Oliveira, F.A.; Oliveira, R.C.M. Anti-inflammatory and anti-ulcer activities of carvacrol, a monoterpene present in the essential oil of oregano. J. Med. Food 2012, 15, 984–991. [Google Scholar] [CrossRef] [PubMed]
- De Souza Siqueira Quintans, J.; Menezes, P.P.; Santos, M.R.V.; Bonjardim, L.R.; Almeida, J.R.G.S.; Gelain, D.P.; Araújo, A.A.d.S.; Quintans-Júnior, L.J. Improvement of p-cymene antinociceptive and anti-inflammatory effects by inclusion in β-cyclodextrin. Phytomedicine 2013, 20, 436–440. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.A.; Taha, M.O.; Zihlif, M.A.; Disi, A.M. β-caryophyllene causes regression of endometrial implants in a rat model of endometriosis without affecting fertility. Eur. J. Pharmacol. 2013, 702, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Alberti, T.; Barbosa, W.; Vieira, J.; Raposo, N.; Dutra, R. (−)-β-caryophyllene, a cb2 receptor-selective phytocannabinoid, suppresses motor paralysis and neuroinflammation in a murine model of multiple sclerosis. Int. J. Mol. Sci. 2017, 18, 691. [Google Scholar] [CrossRef] [PubMed]
- Klauke, A.L.; Racz, I.; Pradier, B.; Markert, A.; Zimmer, A.M.; Gertsch, J.; Zimmer, A. The cannabinoid cb2 receptor-selective phytocannabinoid beta-caryophyllene exerts analgesic effects in mouse models of inflammatory and neuropathic pain. Eur. Neuropsychopharmacol. 2014, 24, 608–620. [Google Scholar] [CrossRef] [PubMed]
- Park, K.-R.; Nam, D.; Yun, H.-M.; Lee, S.-G.; Jang, H.-J.; Sethi, G.; Cho, S.K.; Ahn, K.S. B-caryophyllene oxide inhibits growth and induces apoptosis through the suppression of pi3k/akt/mtor/s6k1 pathways and ros-mediated mapks activation. Cancer Lett. 2011, 312, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Ambriz-Pérez, D.L.; Leyva-López, N.; Gutierrez-Grijalva, E.P.; Heredia, J.B. Phenolic compounds: Natural alternative in inflammation treatment. A review. Cogent. Food Agric. 2016, 2. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Chapter 9: Reactive species and disease: Fact, fiction or filibuster. In Free Radicals in Biology and Medicine; Halliwell, B., Gutteridge, J.M.C., Eds.; Oxford University Press: Oxford, UK, 2007; Volume 4, pp. 488–613. ISBN 978-0-19-856869-8. [Google Scholar]
- Moore, K.J.; Sheedy, F.J.; Fisher, E.A. Macrophages in atherosclerosis: A dynamic balance. Nat. Rev. Immunol. 2013, 13, 709–721. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, P.N. Molecular biology of atherosclerosis. Physiol. Rev. 2013, 93, 1317–1542. [Google Scholar] [CrossRef] [PubMed]
- Dantas, B.P.V.; Alves, Q.L.; de Assis, K.S.; Ribeiro, T.P.; de Almeida, M.M.; de Vasconcelos, A.P.; de Araújo, D.A.M.; de Andrade Braga, V.; de Medeiros, I.A.; Alencar, J.L.; et al. Participation of the trp channel in the cardiovascular effects induced by carvacrol in normotensive rat. Vascul. Pharmacol. 2015, 67–69, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Ocaña-Fuentes, A.; Arranz-Gutiérrez, E.; Señorans, F.J.; Reglero, G. Supercritical fluid extraction of oregano (Origanum vulgare) essentials oils: Anti-inflammatory properties based on cytokine response on THP-1 macrophages. Food Chem. Toxicol. 2010, 48, 1568–1575. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Murakami, M.; Ohba, T.; Takahashi, Y.; Ito, H. Trp channel and cardiovascular disease. Pharmacol. Ther. 2008, 118, 337–351. [Google Scholar] [CrossRef] [PubMed]
- Oz, M.; Lozon, Y.; Sultan, A.; Yang, K.-H.S.; Galadari, S. Effects of monoterpenes on ion channels of excitable cells. Pharmacol. Ther. 2015, 152, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Saljoughian, S.; Roohinejad, S.; Bekhit, A.E.-D.A.; Greiner, R.; Omidizadeh, A.; Nikmaram, N.; Mousavi Khaneghah, A. The effects of food essential oils on cardiovascular diseases: A review. Crit. Rev. Food Sci. Nutr. 2017. [Google Scholar] [CrossRef]
- Alves-Silva, J.M.; Zuzarte, M.; Marques, C.; Salgueiro, L.; Girão, H. Protective effects of terpenes on the cardiovascular system: Current advances and future perspectives. Curr. Med. Chem. 2016, 23, 4559–4600. [Google Scholar] [CrossRef] [PubMed]
- Federation, I.D. The idf Worldwide Definition of the Metabolic Syndrome; International Diabetes Federation: Brussels, Belgium, 2016; Volume 2017. [Google Scholar]
- Alberti, K.G.M.M.; Zimmet, P.; Shaw, J. Metabolic syndrome—A new world-wide definition. A consensus statement from the international diabetes federation. Diabet. Med. 2006, 23, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Mnafgui, K.; Kchaou, M.; Ben Salah, H.; Hajji, R.; Khabbabi, G.; Elfeki, A.; Allouche, N.; Gharsallah, N. Essential oil of Zygophyllum album inhibits key-digestive enzymes related to diabetes and hypertension and attenuates symptoms of diarrhea in alloxan-induced diabetic rats. Pharm. Biol. 2016, 54, 1326–1333. [Google Scholar] [CrossRef] [PubMed]
- Aidi Wannes, W.; Marzouk, B. Research progress of Tunisian medicinal plants used for acute diabetes. J. Acute Dis. 2016, 5, 357–363. [Google Scholar] [CrossRef]
- Russo, G.L.; Russo, M.; Ungaro, P. AMP-activated protein kinase: A target for old drugs against diabetes and cancer. Biochem. Pharmacol. 2013, 86, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Yen, H.-F.; Hsieh, C.-T.; Hsieh, T.-J.; Chang, F.-R.; Wang, C.-K. In vitro anti-diabetic effect and chemical component analysis of 29 essential oils products. J. Food Drug Anal. 2015, 23, 124–129. [Google Scholar] [CrossRef]
- Dham, S.; Shah, V.; Hirsch, S.; Banerji, M.A. The role of complementary and alternative medicine in diabetes. Curr. Diab. Rep. 2006, 6, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Sebai, H.; Selmi, S.; Rtibi, K.; Souli, A.; Gharbi, N.; Sakly, M. Lavender (Lavandula stoechas L.) essential oils attenuate hyperglycemia and protect against oxidative stress in alloxan-induced diabetic rats. Lipids Health Dis. 2013, 12, 189. [Google Scholar] [CrossRef] [PubMed]
- Alef, B.; Abdennacer, B.; Mohamed, B. A-amylase inhibitory activities of Origanum glandolosum, a North African endemic species. Int. J. Adv. Res. 2013, 1, 25–32. [Google Scholar]
- Bayramoglu, G.; Senturk, H.; Bayramoglu, A.; Uyanoglu, M.; Colak, S.; Ozmen, A.; Kolankaya, D. Carvacrol partially reverses symptoms of diabetes in stz-induced diabetic rats. Cytotechnology 2014, 66, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, Y.; Gupta, V.K.; Jaitak, V. Anticancer activity of essential oils: A review. J. Sci. Food Agric. 2013, 93, 3643–3653. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Sureda, A.; Tenore, G.C.; Daglia, M.; Sharifi-Rad, M.; Valussi, M.; Tundis, R.; Sharifi-Rad, M.; Loizzo, M.R.; Ademiluyi, A.O. Biological activities of essential oils: From plant chemoecology to traditional healing systems. Molecules 2017, 22, 70. [Google Scholar] [CrossRef] [PubMed]
- Begnini, K.R.; Nedel, F.; Lund, R.G.; Carvalho, P.H.D.; Rodrigues, M.R.A.; Beira, F.T.A.; Del-Pino, F.A.B. Composition and antiproliferative effect of essential oil of Origanum vulgare against tumor cell lines. J. Med. Food 2014, 17, 1129–1133. [Google Scholar] [CrossRef] [PubMed]
- Misharina, T.A.; Burlakova, E.B.; Fatkullina, L.D.; Alinkina, E.S.; Vorob’eva, A.K.; Medvedeva, I.B.; Erokhin, V.N.; Semenov, V.A.; Nagler, L.G.; Kozachenko, A.I. Effect of oregano essential oil on the engraftment and development of lewis carcinoma in f1 dba c57 black hybrid mice. Appl. Biochem. Microbiol. 2013, 49, 432–436. [Google Scholar] [CrossRef]
- Bostancioglu, R.B.; Kurkcuoglu, M.; Baser, K.H.C.; Koparal, A.T. Assessment of anti-angiogenic and anti-tumoral potentials of Origanum onites L. essential oil. Food Chem. Toxicol. 2012, 50, 2002–2008. [Google Scholar] [CrossRef] [PubMed]
- Mosby. Mosby’s Medical Dictionary; Elsevier: St. Louis, MO, USA, 2017; p. 757. ISBN 978-0-323-41425-8. [Google Scholar]
- Ceker, S.; Nardemir, G.; Alpsoy, L.; Agar, G.; Mete, E. Anti-genotoxic and anti-oxidant effects of Origanum rotundifolium on human lymphocytes in vitro. J. Essent. Oil Bear. Plant 2012, 15, 415–423. [Google Scholar] [CrossRef]
- Mossa, A.-T.H.; Refaie, A.A.; Ramadan, A.; Bouajila, J. Antimutagenic effect of Origanum majorana L. essential oil against prallethrin-induced genotoxic damage in rat bone marrow cells. J. Med. Food 2013, 16, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.I.; Anwar, F.; Rasheed, S.; Nigam, P.S.; Janneh, O.; Sarker, S.D. Composition, antioxidant and chemotherapeutic properties of the essential oils from two Origanum species growing in Pakistan. Rev. Bras. Farmacogn. Braz. J. Pharmacogn. 2011, 21, 943–952. [Google Scholar] [CrossRef]
- De Kok, T.M.; de Waard, P.; Wilms, L.C.; van Breda, S.G. Antioxidative and antigenotoxic properties of vegetables and dietary phytochemicals: The value of genomics biomarkers in molecular epidemiology. Mol. Nutr. Food Res. 2010, 54, 208–217. [Google Scholar] [CrossRef] [PubMed]
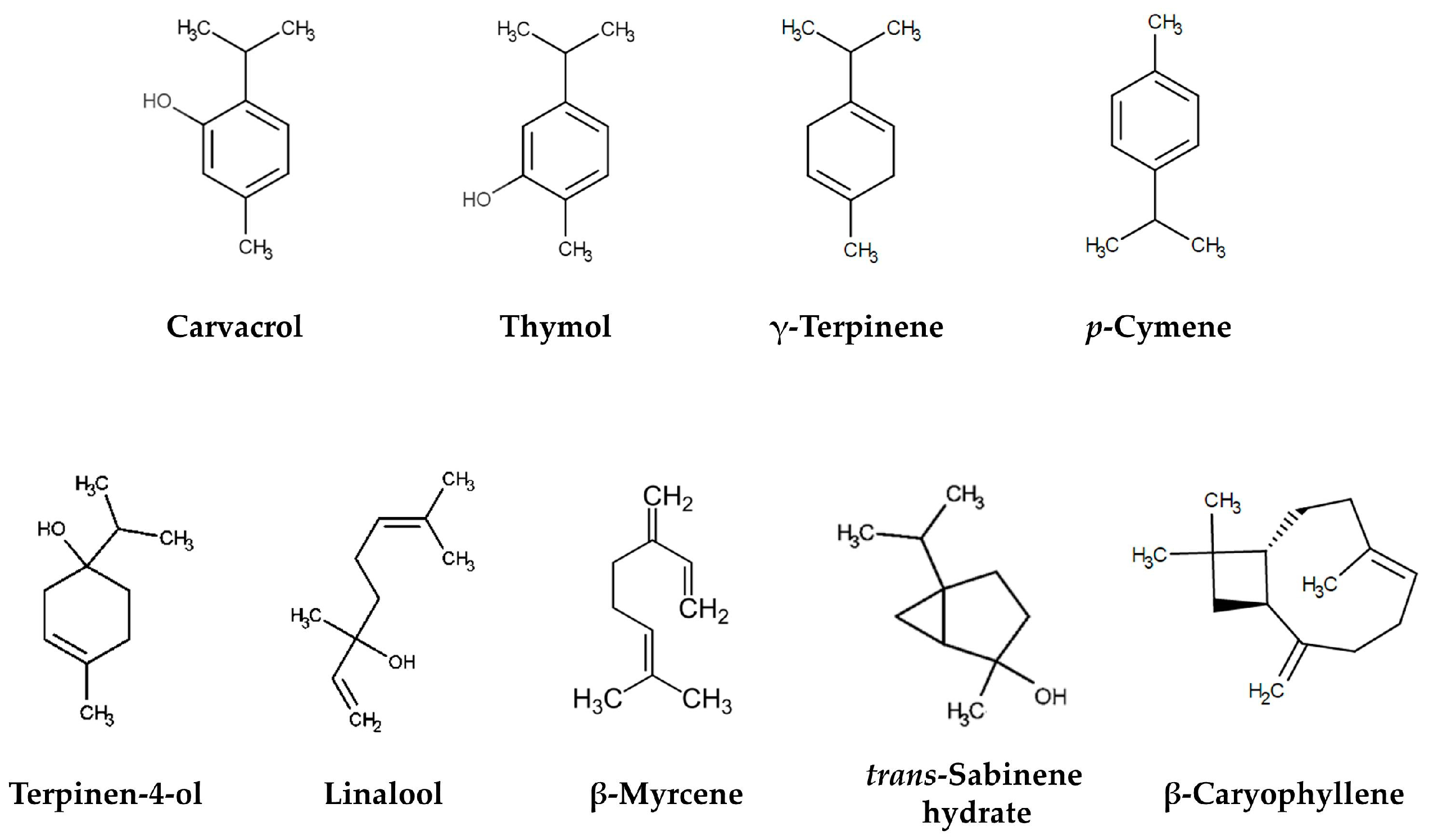
| Oregano Species | Origin | Components | Yield * | Reference |
|---|---|---|---|---|
| H. patens | Mexico | Thymol, trans-piperitol, carvacrol acetate, carvacrol, camphene, β-myrcene, γ-terpinene, cis-p-mentha-1(7), 8-dien-2-ol, α-muurolene, α-calacorene, bulnesol, cadalene, viridiflorol. | NR | [16] |
| L. grandis | Brazil | Carvacrol (37.12%), p-cymene (11.64%), thymol (7.83%), β-caryophyllene (3.93%). | 2.7% | [38] |
| L. graveolens | Mexico | Thymol, carvacrol acetate, carvacrol, camphene, β-myrcene, γ-terpinene, cis-p-mentha-1(7),8-dien-2-ol, viridiflorol. | NR | [16] |
| L. origanoides | Colombia | Thymol (78.7%), p-cymene (6.6%), γ-terpinene (2.7%), trans-β-caryophyllene (2.1%). | NR | [39] |
| L. palmeri | Mexico | Thymol, α-cedrene, trans-piperitol, eugenol, carvacrol acetate, β-selinene, γ-cadinene, spathulenol. | NR | [16] |
| O. acutidens | Mexico | Carvacrol (8.76–24.57%), p-cymene (14.25–22.37%), thymol (15.11–21.39%). | 5.0–6.0% | [40] |
| Turkey | Carvacrol (76.21%), p-cymene (7.42%), borneol (3.19%), γ-terpinene (1.38%). | 1.45% | [41] | |
| Turkey | Carvacrol (65.13%), meta-cymene (9.15%), trans-β-caryophyllene (4.43%), γ-terpinene (3.54%). | 3.1% | [42] | |
| O. x applii | Argentina | Thymol (30.77%), trans-sabinene hydrate (29.63%), γ-terpinene (4.4%), terpinen-4-ol (3.23%). | 1.83 ± 0.27 mg/g dw | [43] |
| O. ehrenbergii | Lebanon | Carvacrol (79.0%), p-cymene (4.4%), carvacrol methyl ether (2.7%), γ-terpinene (2.6%). | 3.19% | [44] |
| O. bilgeri | Turkey | Carvacrol (84.30–90.20%), p-cymene (3.40–5.85%), γ-terpinene (0.47–1.20%), thymol (0.69–1.08%). | 0.54–0.57% | [33] |
| O. libanoticum | Lebanon | β-Caryophyllene (26.8%), caryophyllene oxide (22.6%), germacrene D (17.2%), thymol methyl ether (10.5%). | 0.16% | [44] |
| O. majorana | Brazil | 1,8-Cineole (20.9%), terpinen-4-ol(20.4%), γ-terpinene (8.5%), p-cymene (7.0%). | NR | [45] |
| O. x majoricum | Colombia | trans-Sabinene hydrate (14.5%), γ-terpinene (14.0%), carvacrol methyl ether (6.0%), terpinen-4-ol (6.0%). | NR | [39] |
| Turkey | Limonene (88.01%), thymol (11.98%). | NR | [46] | |
| Argentina | trans-Sabinene hydrate (24.3–28.1%), thymol (12.1–17.4%), γ-terpinene (7.0–7.5%). | NR | [22] | |
| Argentina | trans-Sabinene hydrate (36.77%), thymol (17.77%), γ-terpinene (5.9%), α-terpinene (3.9%). | 3.9 ± 0.25 mg/g dw | [43] | |
| O. hypericifolium | Turkey | p-Cymene (34.33%), carvacrol (21.76%), thymol (19.54%), γ-terpinene (13.91%). | 2.9% | [47] |
| O. onites | Greece | Carvacrol (79.63%), γ-terpinene (3.89%), p-cymene (3.51%), β-caryophyllene (2.24%). | 3.62% | [18] |
| Greece | Carvacrol (62.6%), p-cymene (8.87%), γ-terpinene (8.45%), β-myrcene (2.92%). | NR | [48] | |
| Greece | Carvacrol (69.0–92.6%), p-cymene (0.5–9.5%), γ-terpinene (0.3–7.9%), borneol (0.8–5.5%). | 3.0–7.0% | [49] | |
| Turkey | Carvacrol (85.86%), γ-terpinene (4.43%), β-phellandrene (3.20%), p-cymene (1.83%). | 4.7 ± 0.06% | [21] | |
| Turkey | Carvacrol (83.97–88.65%), thymol (0.80–7.48%), γ-terpinene (2.63–6.15%), p-cymene (1.52–3.16%). | 2.5–3.2% | [50] | |
| O. syriacum | Egypt | Carvacrol (81.38%), p-cymene (8.48%), γ-terpinene (1.98%), β-myrcene (1.32%). | 5.5% | [51] |
| Egypt | Thymol (31.73%), γ-terpinene (14.32%), linalool (9.44%), terpinen-4-ol (7.68%). | 4.63% | [51] | |
| Egypt | Thymol (21.04%), γ-terpinene (18.96%), terpinen-4-ol (17.20%), α-terpinene (7.41%). | 0.6% | [52] | |
| Lebanon | Carvacrol (60.8%), p-cymene (8.4%), thymol (7.9%), γ-terpinene (7.5%). | 1.65% | [44] | |
| O. syriacum ssp. syriacum | Jordan | Thymol (51.8%), carvacrol (34.4%), p-cymene (3.9%). | 2.0–2.2% | [53] |
| Jordan | Thymol (72.4%), γ-terpinene (7.8%), p-cymene (5.4%), carvacrol (3.5%). | 2.0–2.2% | ||
| O. vulgare L. | Argentina | p-Cymene (26.00%), γ-terpinene (21.89%), terpinen-4-ol (16.29%), β-caryophyllene (8.25%). | NR | [54] |
| Argentina | Carvacrol (26.70%), p-cymene (15.20%), γ-terpinene (15.10%), terpinene (7.50%). | NR | [55] | |
| Argentina | γ-Terpinene (25.1%), terpinen-4-ol (16.7%), carvacrol (16.2%), α-terpinene (8.54%). | NR | [56] | |
| Argentina | γ-Terpinene (32.1%), α-terpinene (15.1%), p-cymene (8.0%), thymol (8.0%). | NR | [57] | |
| Argentina | Carvacrol (81.92%), γ-terpinene (4.49%), thymol (3.5%), p-cymene (3.07%). | NR | [58] | |
| Brazil | Carvacrol (73.9%), γ-terpinene (3.6%), thymol (3.0%), β-caryophyllene (2.8%). | NR | [45] | |
| Chile | cis-β-Terpineol (16.49%), thymol (13.26%), terpinen-4-ol (10.24%), α-terpineol (4.35%). | NR | [59] | |
| China | Carvacrol (30.73%), thymol (18.81%), p-cymene (10.88%), β-caryophyllene (8.21%). | NR | [37] | |
| China | β-Citronellol (85.3%), citronellol acetate (5.2%), β-citronellal (1.2%). | 0.7% | [60] | |
| China | Thymol (42.9%), citronellol (12.2%), β-caryophyllene (7.8%), p-cymen-2-ol (7.5%). | 0.3% | [60] | |
| China | β-Citronellol (75.0%), geraniol (7.7%), citronellol acetate (3.4%). | 0.3% | [60] | |
| China | 1,8-Cineole (20.8%), β-caryophyllene (10.2%), eugenol methyl ether (9.8%), citronellol (8.8%). | 0.3% | [60] | |
| China | Caryophyllene oxide (32.9%), β-caryophyllene (17.7%), citronellol (10.2%), germacrene D (9.8%). | 0.1% | [60] | |
| Colombia | Thymol (21.5%), p-cymene (21.0%), γ-terpinene (20.3%), α-terpinene (5.9%). | NR | [39] | |
| Greece | Carvacrol (63.03%), thymol (15.09%), p-cymene (10.47%), γ-terpinene (3.43%). | NR | [61] | |
| India | Carvacrol (35.02–62.81%), p-cymene (8.60–46.59%), γ-terpinene (2.49–19.11%). | 0.20–1.30% | [62] | |
| Iran | Carvacrol (29.85%), γ-terpinene (20.94%), α-himachalene (12.17%), β-pinene (11.67%). | 0.80% | [63] | |
| Iran | Carvacrol (23.54%), γ-terpinene (20.50%), thymol (15.41%), germacrene D-4-ol (9.26%). | 1.26% | [63] | |
| Iran | Carvacrol (59.37%), γ-terpinene (18.36%), cedrene (6.65%). | 1.66% | [63] | |
| Iran | Carvacrol (58.51%), humulene (11.46%), γ-terpinene (9.56%). | 0.93% | [63] | |
| Iran | Carvacrol (67.09%), γ-terpinene (7.71%), humulene (7.67%). | 1.36% | [63] | |
| Italy | Cavacrol (65.94%), p-cymene (9.33%), γ-terpinene (5.25%), β-caryophyllene (3.72%). | NR | [64] | |
| Italy | Carvacrol (71.8%), p-cymene (11.6%), β-caryophyllene (2.7%), linalool (1.8%). | NR | [65] | |
| Morocco | Carvacrol (34.0%), γ-terpinene (21.6%), p-cymene (9.4%), thymol (3.3%). | 2.7% | [66] | |
| Pakistan | β-Citronellol (72.7%), thymol (7.2%), citronellol acetate (5.9%). | 0.3% | [60] | |
| Poland | Carvacrol (26.38–36.72%), thymol (16.59–25.58%), γ-terpinene (10.06–16.11%), p-cymene (6.09–6.76%). | NR | [31] | |
| Portugal | Carvacrol (14.5%), β-fenchyl alcohol (12.8%), γ-terpinene (11.6%), δ-terpineol (7.5%). | NR | [67] | |
| Serbia | Sabinene (10.2%), terpinen-4-ol (9.3%), 1,8-cineole (5.8%), γ-terpinene (5.6%). | 0.17% | [68] | |
| Serbia | Carvacrol (64.5%), p-cymene (10.9%), γ-terpinene (10.8%), thymol (3.5%). | 1.5% | [69] | |
| Serbia | Carvacrol (64.5%), p-cymene (10.9%), γ-terpinene (10.8%), thymol (3.5%). | NR | [70] | |
| Serbia | Carvacrol (77.6%), p-cymene (5.14%), trans-β-caryophyllene (2.45%), linalool (2.44%). | NR | [71] | |
| Spain | Terpinen-4-ol (24.57%), carvacrol (16.09%), thymol (9.03%), γ-terpinene (6.20%). | 516 mg/plant | [27] | |
| USA | Carvacrol (17.9–81.8%), p-cymene (2.62–25.7%), γ-terpinene (2.5–19.4%), β-myrcene (0.58–6.06%). | 0.114–2.312% | [35] | |
| O. vulgare L. ssp. glandulosum | Algeria | Thymol (34.2%), carvacrol (30.5%), γ-terpinene (13.4%), p-cymene (6.6%). | 2.0–2.2% | [53] |
| Algeria | Thymol (51.1%), γ-terpinene (14.5%), p-cymene (7.5%), carvacrol (6.8%). | 2.0–2.2% | [53] | |
| Tunisia | p-Cymene (35.7–46.3%), thymol (18.4–39.1%), γ-terpinene (11.7–24.2%), carvacrol (1.7–15.1%). | 2.5–4.6% | [72] | |
| Tunisia | Thymol (31.8–46.1%), p-cymene (11.5–35.7%), γ-terpinene (24.0–27.1%), α-terpinene (1.9–3.2%). | 4.3–5.8% | [73] | |
| Tunisia | Carvacrol (65.01%), p-cymene (9.00%), γ-terpinene (4.25%), borneol (3.19%). | 1.87–3.42% | [74] | |
| O. vulgare L. ssp. gracile | Iran | Carvacrol (46.86%), γ-terpinene (14.16%), p-cymene (11.63%), carvacrol methyl ether (5.97%). | ≈2.0% | [29] |
| Turkey | Thymol (7.02–40.04%), carvacrol (8.21–33.21%), γ-terpinene (9.15–27.82%), p-cymene (3.07–23.52%). | 0.25–0.50% | [75] | |
| O. vulgare L. ssp. hirtum | Argentina | trans-Sabinene hydrate (22.9%), thymol (18.6%), γ-terpinene (7.1%), terpinen-4-ol (6.2%). | NR | [22] |
| Argentina | trans-Sabinene hydrate (17.9%), thymol (17.1%), terpinen-4-ol (9.5%), γ-terpinene (8.0%). | NR | [22] | |
| Argentina | γ-Terpinene (13.7%), terpinen-4-ol (11.2%), α-terpinene (9.9%), trans-sabinene hydrate (8.3%). | NR | [76] | |
| Colombia | Carvacrol (90.3%), thymol (3.5%), p-cymene (2.7%), γ-terpinene (1.0%). | NR | [39] | |
| Greece | Carvacrol (70.38%), p-cymene (8.17%), γ-terpinene (7.78%), β-myrcene (2.37%). | NR | [48] | |
| Greece | Carvacrol (90.29%), γ-terpinene (3.09%), p-cymene (2.25%), β-caryophyllene (1.81%). | 7.77% | [18] | |
| Greece | Carvacrol (81.28–91.21%), p-cymene (1.52–6.40%), γ-terpinene (0.49–4.01%), β-caryophyllene (0.94–2.03%). | 4.71–5.00% | [77] | |
| Greece | Carvacrol (56.46–82.70%), p-cymene (9.54–21.40%), β-disavolene (1.09–3.06%). | 0.63–4.25% | [78] | |
| Hungary | Carvacrol (82.75%), p-cymene (6.58%), γ-terpinene (5.78%). | 4.46% | [30] | |
| Italy | terpinen-4-ol (13.27–17.51%), γ-terpinene (14.58–14.95%), carvacrol (12.31–14.58%), p-cymene (8.43–10.07%). | 0.063–0.165% | [79] | |
| Italy | Thymol (37.9%), γ-terpinene (24.5%), p-cymene (16.3%), α-terpinene (4.3%). | NR | [80] | |
| Italy | γ-Terpinene (29.41%), thymol (26.86%), p-cymene (8.20%), α-terpinene (5.93%). | 5.4% | [81] | |
| Italy | Thymol (37.22%), γ-terpinene (26.37%), p-cymene (6.83%), α-terpinene (4.02%). | 2.4% | [81] | |
| Italy | Thymol (36.46%), γ-terpinene (20.77%), p-cymene (8.31%), carvacrol methyl ether (6.21%). | 3.6% | [81] | |
| Italy | Thymol (30.25%), γ-terpinene (25.89%), p-cymene (7.62%), carvacrol methyl ether (5.63%). | 4.2% | [81] | |
| Italy | Thymol y carvacrol (65.3–84.7%), linalool (0.1–2.6%), carvacrol methyl ether (0.4–1.9%). | 1.0–2.7% | [82] | |
| Italy | Thymol (18.16–56.37%), γ-terpinene (12.70–32.70%), p-cymene (8.22–10.30%). | 1.7–4.5% | [28] | |
| Lithuania | Carvacrol (72.4–88.2%), γ-terpinene (4.1–8.7%), p-cymene (2.0–3.2%), β-caryophyllene (0.9–3.0%). | 35.50–325.45 dm3/ha | [24] | |
| Serbia | Carvacrol (74.65%), p-cymene (5.87%), γ-terpinene (5.04%), trans-β-caryophyllene (1.76%). | 1.34% | [36] | |
| Turkey | Linalool (96.31%), β-caryophyllene (1.27%). | 7.31% | [83] | |
| Turkey | Carvacrol (80.09%), γ-terpinene (12.01%), p-cymene (1.72%), α-terpinene (1.58%). | 5.9 ± 0.02% | [21] | |
| O. vulgare L. ssp. virens | Argentina | trans-Sabinene hydrate (27.77%), thymol (26.1%), γ-terpinene (5.9%), α-terpinene (4.17%). | 2.17 ± 0.32 mg/g dw | [43] |
| Iran | (Z)-α-Bisabolene (39.17%), sabinene (11.52%), carvacrol (5.23%), β-bisabolene (4.24%). | ≈0.3% | [29] | |
| Portugal | α-Terpineol (0.1–65.1%), γ-terpinene (0.3–34.25), linalool (2.0–27.4%), carvacrol (0–34.2%), E-caryophyllene (2.4–11.0%). | 0.8–1.2% | [84] | |
| O. vulgare L. ssp. vulgare | Argentina | trans-Sabinene hydrate (23.4–27.2%), thymol (14.4–17.2%), terpinen-4-ol (7.8–11.0%), γ-terpinene (7.3–9.8%). | NR | [22] |
| Argentina | trans-Sabinene hydrate (32.47%), thymol (20.5%), γ-terpinene (15.47%), terpinen-4-ol (5.03%). | 1.97 ± 0.22 mg/g dw | [43] | |
| Iran | Thymol (37.13%), γ-terpinene (9.67%), carvacrol (9.57%), carvacrol methyl ether (6.88%). | 0.5% | [85] | |
| Italy | Spathulenol (18.6%), carvacrol (11.7%), β-caryophyllene (8.8%), terpinen-4-ol (5.6%). | 0.13% | [26] | |
| Italy | Carvacrol (14.3%), spathulenol (9.4%), β-caryophyllene (5.3%), terpinen-4-ol (5.0%). | 0.18% | [26] | |
| Lithuania | Sabinene (6.6–28.2%), β-caryophyllene (7.3–15.5%), E-β-ocimene (4.4–15.1%), allo-ocimene (7.7–12.1%). | 3.08–36.65 dm3/ha | [24] | |
| Turkey | Thymol (58.31%), carvacrol (16.11%), p-cymene (13.45%), γ-terpinene (4.64%). | 5.09% | [83] | |
| Poland | Sabinene (10.85–25.46%), Z-(β)-ocimene (9.10–16.33%), germacrene D (9.36–15.34%), E-caryophyllene (9.38–12.87%). | 0.66–0.86% | [86] |
| Oregano Species | Biological Activity | Effect | Reference |
|---|---|---|---|
| H. patens | Anti-inflammatory | Reduction on the levels of NO and ROS produced in murine macrophage cells. | [16] |
| L. palmeri | Anti-inflammatory | Inhibition on the production of ROS and NO by LPS-stimulated RAW 264.7 macrophages | [16] |
| L. graveolens | Antioxidant | Radical scavenging activity against DPPH | [90] |
| Anti-inflammatory | Reduction on the levels of NO and ROS produced in LPS-stimulated murine macrophage cells | [16] | |
| O. acutidens | Antioxidant | Showed scavenging activity against DPPH radical | [42] |
| O. compactum | Antioxidant | ABTS radical-scavenging activity | [111] |
| Cytotoxic | Nontoxic when used in MCF-7 cells | [111] | |
| O. dictamnus | Antioxidant | Ferric reducing/antioxidant power | [117] |
| Antiproliferative | Inhibit colon carcinoma (LoVo) and hepatocarcinoma (HepG2) cell proliferation | [117] | |
| O. ehrenbergii | Antioxidant | DPPH radical-scavenging activity | [107] |
| O. glandulosum | Antioxidant | Showed antiradical activity | [72,73] |
| O. heracleoticum | Anti-inflammatory | Inhibition of NO production | [100] |
| O. libanoticum | Antioxidant | Ferric reducing/antioxidant power | [117] |
| Antiproliferative | Inhibit HepG2 cell proliferation | [117] | |
| O. majorana | Anti-inflammatory | Reduction in the secretion of inflammatory cytokines (TNF-α, IL-1β and IL-6) in THP-1 cells | [124] |
| Anti-genotoxic | Reduces the chromosomal aberration in bone marrow cells of rats | [162] | |
| Cytotoxic | Inhibit cell viability of human breast (MCF-7) and prostate (LNCaP) cancer cell lines. | [163] | |
| O. microphyllum | Antioxidant | Showed ferric reducing power | [117] |
| O. minutiflorum | Antioxidant | Retard lipidic oxidation | [116] |
| O. onites | Antioxidant | Showed free radical scavenging against DPPH radical | [50] |
| Anti-angiogenic | Blocks in vitro tube formation | [159] | |
| O. rotundifolium | Anti-genotoxic | Reduces the effect of Aflatoxin B1 (AFB1) in human peripheral lymphocytes | [161] |
| O. syriacum | Antioxidant | DPPH radical-scavenging activity | [107] |
| O. virens | Antioxidant | Showed scavenging activity against DPPH radical | [118] |
| O. vulgare subsp. hirtum | Antioxidant | Total reducing capacity (Folin-Ciocalteu method), radical-scavenging activity in the UV radiation-induced peroxidation in liposomal membranes | [101] |
| Antioxidant | Reduces 8-hydroxy-deoxyguanosine and thiobarbituric acid reactive substances. | [102] | |
| Antioxidant | DPPH and ABTS radical-scavenging activity | [80,83,114,115] | |
| Antiproliferative | Inhibit human lung adenocarcinoma epithelial (A549) cell proliferation | [76] | |
| Cytotoxic | Decrease cell viability in a concentration-dependent manner on human keratinocyte (HaCaT) and lung cancer (A549) cell lines | [80] | |
| Hypoglycemic | α-Amylase and α-glucosidase inhibitory activity | [83] | |
| O. vulgare subsp. vulgare | Antioxidant | Radical scavenging activity (DPPH, ABTS and FRAP assays). Total reducing capacity (Folin-Ciocalteu method) | [37,55,56,57,83,110,112,85] |
| Antioxidant | Prevent autoxidation of polyunsaturated fatty acid esters | [106] | |
| Anti-inflammatory | Reduced synthesis of TNF-α, IL-1β, and IL-6 cytokines. Increased synthesis of cytokine IL-10 | [140] | |
| Anti-inflammatory | Inhibition of the levels of inflammatory biomarkers (MCP-1, VCAM-1 and ICAM-1) on activated-primary human neonatal fibroblasts | [125] | |
| Antiproliferative | Inhibit human breast adenocarcinoma (MCF-7) and human colon adenocarcinoma (HT-29) cell proliferation | [157] | |
| Antitumor | Decrease the sizes of tumors in disease mice | [158] | |
| Hypoglycemic | Inhibits α-amylase and α-glucosidase activity | [83,153] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leyva-López, N.; Gutiérrez-Grijalva, E.P.; Vazquez-Olivo, G.; Heredia, J.B. Essential Oils of Oregano: Biological Activity beyond Their Antimicrobial Properties. Molecules 2017, 22, 989. https://doi.org/10.3390/molecules22060989
Leyva-López N, Gutiérrez-Grijalva EP, Vazquez-Olivo G, Heredia JB. Essential Oils of Oregano: Biological Activity beyond Their Antimicrobial Properties. Molecules. 2017; 22(6):989. https://doi.org/10.3390/molecules22060989
Chicago/Turabian StyleLeyva-López, Nayely, Erick P. Gutiérrez-Grijalva, Gabriela Vazquez-Olivo, and J. Basilio Heredia. 2017. "Essential Oils of Oregano: Biological Activity beyond Their Antimicrobial Properties" Molecules 22, no. 6: 989. https://doi.org/10.3390/molecules22060989
APA StyleLeyva-López, N., Gutiérrez-Grijalva, E. P., Vazquez-Olivo, G., & Heredia, J. B. (2017). Essential Oils of Oregano: Biological Activity beyond Their Antimicrobial Properties. Molecules, 22(6), 989. https://doi.org/10.3390/molecules22060989









