General
Melting points were determined in open capillary tubes with a MFB 595010M Gallenkamp melting point apparatus (Weiss Gallenkamp, Loughborough, UK). All new compounds were fully characterized by their analytical [melting point, elemental analysis and/or accurate mass measurement, spectroscopic data (IR,
1H-NMR and
13C-NMR, see
supplementary)] and, in the case of compound
24, also X-ray diffraction analysis. Assignments given for the NMR spectra are based on DEPT, COSY,
1H/
13C single quantum correlation (gHSQC sequence) and
1H/
13C multiple bond correlation (gHMBC sequence) spectra and by comparison with previous assignments for the benzyloxymethyl series.
1H-NMR and
13C-NMR spectra were recorded on a Varian Mercury 400 (400 MHz for
1H and 100.6 MHz for
13C, Varian, Palo Alto, CA, USA) spectrometer. Unless otherwise stated, the NMR spectra have been performed in CDCl
3. Chemical shifts (δ) are reported in parts per million related to internal TMS or CDCl
3 for
1H- and
13C-NMR, respectively. Multiplicities are reported using the following abbreviations: s, singlet; d, doublet; t, triplet; m, multiplet; br, broad; or their combinations. IR spectra were registered on a FTIR Perkin-Elmer Spectrum RX1 spectrometer (Perkin-Elmer, Seer Green, UK) using the attenuated total reflectance (ATR) technique. Absorption values are given as wavenumbers (cm
−1), the intensity of the absorptions are given as strong (s), medium (m) or weak (w). IR and NMR spectra of all new compounds are provided in the
Supplementary Materials section of this paper.High resolution mass spectra (HRMS) were carried out at the Mass Spectrometry Unity of the Centres Científics i Tecnològics of the Universitat de Barcelona (CCiTUB, Barcelona, Spain) and are reported as
m/
z. LC/MSD-TOF spectrometer with electrospray ionization (ESI-TOF-MS) from Agilent Technologies (Santa Clara, CA, USA) was used. The elemental analyses were carried out at the IQAC (CSIC) of Barcelona, Spain, in elemental microanalyzers (A5) model Flash 1112 series from Thermofinnigan for (C, H, N) determinations (Thermo Fisher Scientific SL, Barcelona, Spain) and in a titroprocessor Methrom model 808 (Massó Analítica, Barcelona, Spain) for the halogen determination. For the flash column chromatography, silica gel 60 AC (35–70 μm, SDS, reference 2000027) or neutral Al
2O
3 (50–200 μm) was used. The eluents employed are reported as volume/volume percentages. Thin-layer chromatography (TLC) was performed on aluminum-backed sheets with silica gel 60 F254 (Sigma Aldrich Química, S.L., Madrid, Spain, Merck reference 1.05554) and spots were visualized with UV light or a 1% aqueous solution of KMnO
4. X-ray diffraction analysis of compounds 24 was performed in a Bruker D8 Venture diffractometer at the CCiTUB of the University of Barcelona (Bruker Española S.A., Madrid, Spain). The compounds and reagents were purchased to the following companies: methyl 2-oxocyclopentanecarboxylate, methyl chloromethyl ether, silica gel, 60% NaH and 30% KH, both in mineral oil, and
p-toluenesulfonic acid to Sigma-Aldrich; anthrone and Pd(OAc)
2 to Alfa Aesar;
N-methylmaleimide and NaBH
4 to TCI; 3-chloro-2-chloromethyl-1-propene to Secant Chemicals, Inc., Winchendon, MA, USA; borane THF complex,
n-BuLi in hexanes, neutral aluminum oxide Brockman I (50–200 μm), methanesulfonyl chloride, and NaI to ACROS Organics; diisopropylamine and 35% H
2O
2 to Panreac; trimethylsilyl triflate, CeCl
3·7H
2O, and NaNO
2 to Fluka. All of them were used without further purification.
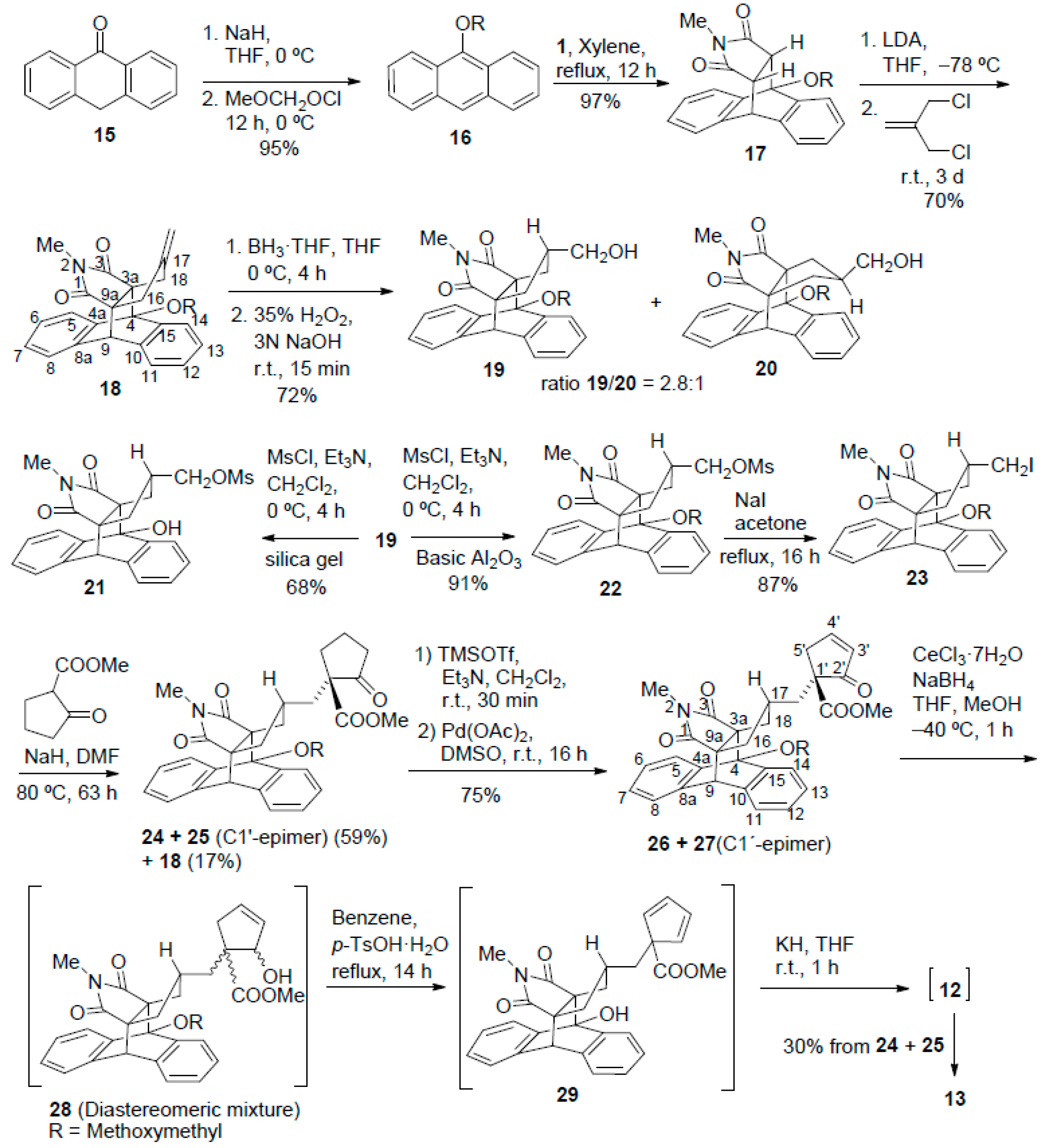
9-(Methoxymethoxy)anthracene (16). NaH (60% in mineral oil, 3.09 g, 77 mmol) was added in portions to a cold (0 °C, ice-water bath) solution of anthrone 15 (10.0 g, 51.5 mmol) in anhydrous THF (600 mL) and the mixture was stirred for 45 min at this temperature. Methoxymethyl chloride (5.87 mL, 6.22 g, 77.3 mmol) were added at 0 °C and the mixture was stirred for 12 h at room temperature. Water (250 mL) and EtOAc (300 mL) were added, the organic phase was separated and the aqueous one was extracted with EtOAc (2 × 300 mL). The combined organic phases were washed with water (250 mL) and brine (250 mL), dried (anhydrous Na2SO4) and concentrated in vacuo to give 16 (11.6 g, 95% yield). An analytical sample of 16 (105 mg) was obtained as yellow solid by crystallization of a sample of the above product (300 mg) from a mixture of hexane (5 mL) and EtOAc (2 mL). m.p. 78–79 °C (EtOAc/hexane); 1H-NMR (400 MHz, CDCl3): δ = 3.76 (s, 3H, OCH3), 5.38 (s, 2H, OCH2O), 7.45–7.52 [complex signal, 4H, 2(7)-H and 3(6)-H], 7.98–8.01 [m, 2H, 4(5)-H], 8.26 (s, 1H, 10-H), 8.31–8.34 ppm [m, 2H, 1(8)-H]; 13C-NMR (100.6 MHz, CDCl3): δ = 58.1 (CH3, OCH3), 101.2 (CH2, OCH2O), 122.6 [CH, C1(8)], 122.7 (CH, C10), 125.0 [C, C8a(9a)], 125.3 [CH, C2(7)], 125.5 [CH, C3(6)], 128.3 [CH, C4(5)], 132.3 (C, C4a(4b)], 149.7 ppm (C, C9); IR (ATR): ṽ = 1675 (w), 1343 (m), 1309 (m), 1285 (m), 1157 (m), 1098 (m), 1040 (s), 1008 (m), 933 (s), 924 (s), 884 (m), 846 (m), 791 (m), 732 (s), 693 (m), 658 cm−1 (m); accurate mass measurement: m/z calcd. for C16H14O2 + H+: 239.1067; found: 239.1056; elemental analysis calcd. (%) for C16H14O2: C 80.65, H 5.92; found: C 80.65, H 5.84.
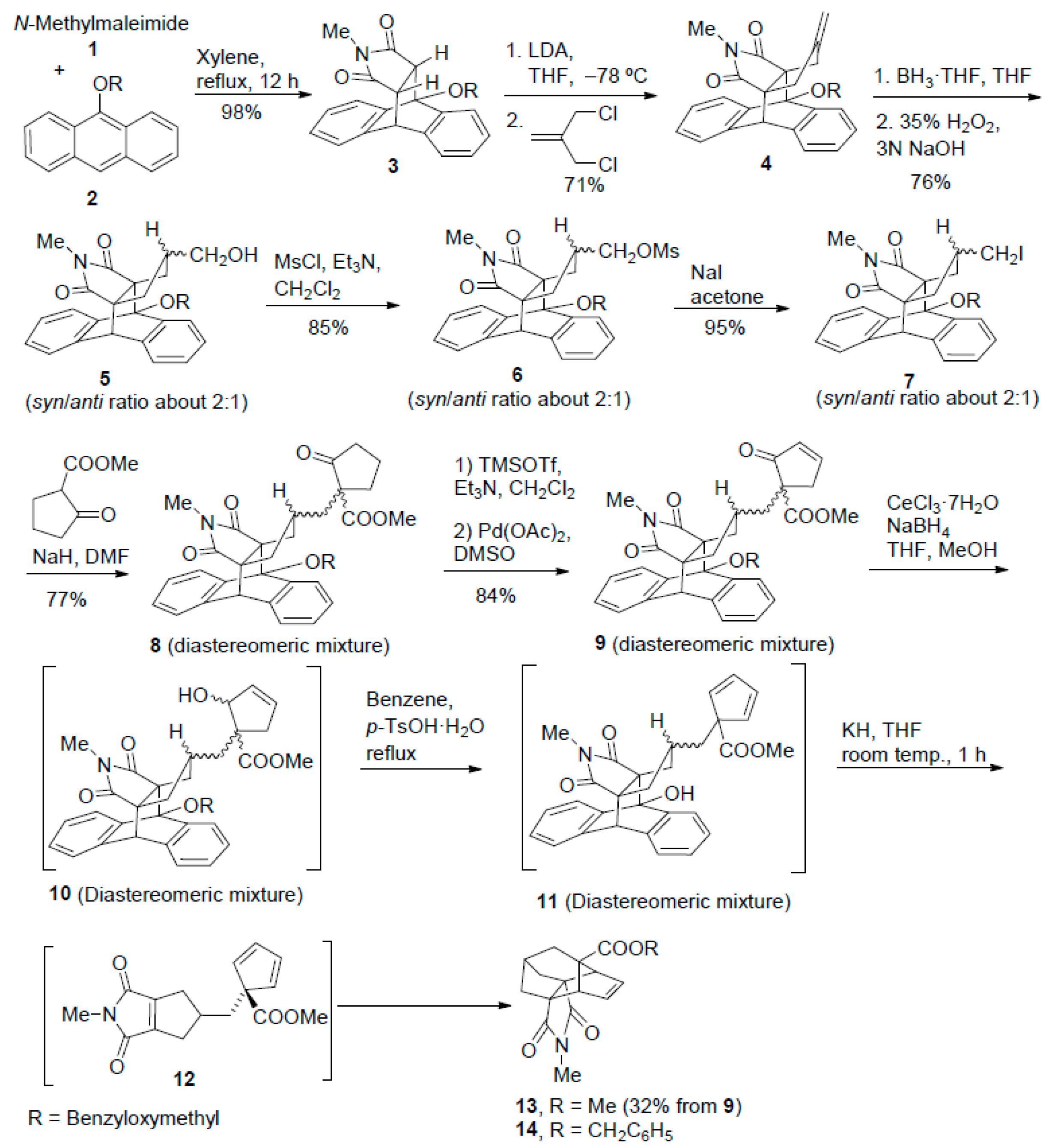
4-(Methoxymethoxy)-2-methyl-3a,4,9,9a-tetrahydro-4,9[1′,2′]benzeno-1H-benz[f]isoindole-1,3(2H)-dione (17). A solution of 16 (8.20 g, 34.2 mmol) and N-methylmaleimide 1 (3.80 g, 34.2 mmol) in xylene (350 mL) was heated at 140 °C for 12 h. The solution was allowed to cool to room temperature and was concentrated in vacuo to give adduct 17 as yellow solid (11.6 g, 97% yield). An analytical sample of 17 (83 mg) was obtained as yellow solid by crystallization of a sample of the above product (200 mg) from a mixture of hexane (3 mL) and EtOAc (1 mL). m.p. 168–169 °C (EtOAc/hexane); 1H-NMR (400 MHz, CDCl3): δ = 2.50 (s, 3H, N-CH3), 3.31 (dd, 3J(H,H) = 8.4 Hz, 3J(H,H) = 3.2 Hz, 1H, 9a-H), 3.44 (d, 3J(H,H) = 8.4 Hz, 1H, 3a-H), 3.82 (s, 3H, OCH3), 4.70 (d, 3J(H,H) = 3.2 Hz, 1H, 9-H), 5.53 (d, 2J(H,H) = 5.4 Hz, 1H) and 5.55 (d, 2J(H,H) = 5.4 Hz, 1H) (OCH2O), 7.12 (pseudo dt, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.4 Hz, 1H, 7-H), 7.17 (overlapped pseudo dt, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.2 Hz, 1H, 6-H), 7.20 (overlapped pseudo dt, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.3 Hz, 1H, 12-H), 7.21–7.26 (complex signal, 2H, 8-H and 13-H), 7.38 (dd, 3J(H,H) = 7.4 Hz, 4J(H,H) = 1.4 Hz, 1H, 11-H), 7.65 (dd, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.2 Hz, 1H, 5-H), 7.68 ppm (dd, 3J(H,H) = 7.6 Hz, 4J(H,H) = 1.2 Hz, 1H, 14-H); 13C-NMR (100.6 MHz, CDCl3): δ = 24.3 (CH3, N-CH3), 44.6 (CH, C9), 47.4 (CH, C3a), 47.9 (CH, C9a), 57.1 (CH3, OCH3), 81.8 (C, C4), 93.8 (CH2, OCH2O), 121.8 (CH, C5), 122.2 (CH, C14), 124.0 (CH, C11), 124.4 (CH, C8), 126.6 (CH, C13), 126.9 (CH, C12), 127.00 (CH, C7), 127.03 (CH, C6), 136.2 (C, C8a), 139.7 (C, C4a), 140.2 (C, C10), 141.6 (C, C15), 174.1 (C, C1), 176.2 ppm (C, C3); IR (ATR): ṽ = 1692 (s), 1457 (m), 1430 (m), 1292 (m), 1276 (m) 1163 (m), 1124 (m), 1050 (m), 1013 (m), 963 (m), 771 (m), 738 cm−1 (s); accurate mass measurement: m/z calcd. for C21H19NO4 + Na+: 372.1206; found: 372.1198; elemental analysis calcd. (%) for C21H19NO4: C 72.19, H 5.48, N 4.01; found: C 72.08, H 5.42, N 3.79.
4-[(Methoxy)methoxy)-2-methyl-17-methylene-4,9-dihydro-4,9[1′,2′]benzeno-3a,9a-propano-1H-benz[f]isoindole-1,3(2H)-dione (18). A solution of n-BuLi in hexanes (2.75 mL, 2.5 M, 6.87 mmol) was added dropwise to a cold (−78 °C, acetone/solid CO2 bath) and magnetically-stirred solution of diisopropylamine (0.97 mL, 6.9 mmol) in anhydrous THF (18 mL) under an Ar atmosphere. When n-BuLi addition was finished, the solution was allowed to warm to 0 °C for 1 h, it was cooled again to −78 °C, and a solution of 17 (1.00 g, 2.86 mmol) in anhydrous THF (18 mL) was added dropwise. Then, the solution was stirred a −78 °C for 15 min and allowed to warm to 0 °C for 1 h. The solution was again cooled to −78 °C and 3-chloro-2-(chloromethyl)-1-propene (0.48 mL 96% content, 498 mg, 3.98 mmol) was added dropwise. The reaction mixture was allowed to warm to room temperature and it was stirred for three days at this temperature. The mixture was made acidic with aqueous 2 N HCl (8 mL) and was extracted with Et2O (3 × 40 mL). The combined organic extracts were dried (anhydrous Na2SO4) and concentrated in vacuo to give a brown waxy residue (1.29 g) that was subjected to column chromatography (silica gel 35–70 μm, 65 g, hexane/EtOAc mixtures) to give an elution with hexane/EtOAc 19:1 to 9:1, product 18 (801 mg, 70% yield) as yellow solid. m.p 150–152 °C (EtOAc/hexane); 1H-NMR (400 MHz, CDCl3): δ = 1.99–2.04 (dm, 2J(H,H) = 15.2 Hz, 1H, 16-Hn), 2.25–2.31 (dm, 2J(H,H) = 15.8 Hz, 1H, 18-Hn), 2.45 (s, 3H, N-CH3), 2.59 (broad d, 2J(H,H) = 15.2 Hz, 1H, 16-Hx), 2.68 (broad d, 2J(H,H) = 15.8 Hz, 1H, 18-Hx), 3.80 (s, 3H, OCH3), 4.44 (s, 1H, 9-H), 4.58–4.60 (m, 1H, 17=CHa), 4.60–4.62 (m, 1H, 17=CHs), 5.37 (d, 2J(H,H) = 5.6 Hz, 1H, OCHaO), 5.40 (d, 2J(H,H) = 5.6 Hz, 1H, OCHbO), 7.13 (pseudo dt, 4J(H,H) = 1.4 Hz, 3J(H,H) = 7.2 Hz, 1H, 7-H), 7.18 (pseudo dt, 4J(H,H) = 1.5 Hz, 3J(H,H) = 7.6 Hz, 1H, 6-H), 7.22–7.27 (complex signal, 2H, 8-H, 12-H), 7.28 (overlapped pseudo dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.2 Hz, 1H, 13-H), 7.37–7.39 (ddm, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.6 Hz, 1H, 11-H), 7.74–7.77 ppm (broad d, 3J(H,H) = 7.6 Hz, 2H, 5-H and 14-H); 13C-NMR (100.6 MHz, CDCl3): δ = 24.6 (CH3, N-CH3), 38.2 (CH2, C18), 39.0 (CH2, C16), 48.7 (CH, C9), 56.4 (CH3, OCH3), 64.1 (C, C3a), 64.8 (C, C9a), 87.1 (C, C4), 94.2 (CH2, OCH2O), 108.9 (CH2, 17=CH2), 122.7 (CH, C5), 124.3 (CH, C14), 124.8 (CH, C8), 126.3 (CH, C11), 126.9 (CH, C13), 127.0 (CH, C6), 127.20 (CH) and 127.21 (CH) (C7 and C12), 138.15 (C, C8a), 138.21 (C, C10), 139.4 (C, C4a), 139.9 (C, C15), 146.6 (C, C17), 177.2 (C, C3), 179.4 ppm (C, C1); IR (ATR): ṽ = 1696 (s), 1456 (m), 1428 (m), 1373 (m), 1304 (m) 1158 (m), 1092 (m), 1020 (m), 987 (m), 930 (m), 773 (m), 758 cm−1 (m); accurate mass measurement: m/z calcd. for C25H23NO4 + Na+: 424.1519. found: 424.1521; elemental analysis calcd. (%) for C25H23NO4: C 74.80, H 5.77, N 3.49; found: C 74.92, H 5.74, N 3.39.
Syn-17-(hydroxymethyl)-4-(methoxymethoxy)-2-methyl-4,9-dihydro-4,9[1′,2′]benzeno-3a,9a-propano-1H-benz[f]isoindole-1,3(2H)-dione (19) and anti-17-(hydroxymethyl)-4-(methoxymethoxy)-2-methyl-4,9-dihydro-4,9[1’,2’]benzeno-3a,9a-propano-1H-benz[f]isoindole-1,3(2H)-dione (20). A solution of the BH3 THF complex in anhydrous THF (26.5 mL, 1 M in THF, 26.5 mmol) was added dropwise to a cold (0 °C, ice-water bath) and magnetically-stirred solution of compound 18 (4.62 g, 11.5 mmol) in anhydrous THF (150 mL) under an Ar atmosphere, and the reaction mixture was stirred at 0 °C for 4 h. After addition of EtOH (12.7 mL), the mixture was allowed to warm to room temperature, and aqueous solutions of 35% H2O2 (9.4 mL) and 3 M NaOH (14.8 mL) were simultaneously added dropwise in 15 min, occasionally cooling with a water bath, and the reaction mixture was stirred at room temperature for 15 min. Water (50 mL) and EtOAc (100 mL) were added, the organic phase was separated and the aqueous one was extracted with EtOAc (2 × 100 mL). The combined organic phases were dried (anhydrous Na2SO4) and concentrated to dryness in vacuo to give a white solid (5.5 g) that was subjected to column chromatography (silica gel 35–70 μm, 165 g, hexane/EtOAc mixtures). Alcohol 19 (1.40 g), a 2:1 mixture of 19 and 20 (1.174 g) and alcohol 20 (330 mg) were successively eluted as white solids with hexane/EtOAc 3:2, 1:1, and 2:3, respectively (overall yield: 2.56 g, 72%, approximate ratio 19/20 = 2.8/1 (1H-NMR).
Analytical and spectroscopic data of 19. White solid, m.p. 216–222 °C (EtOAc/hexane); 1H-NMR (400 MHz, CDCl3): δ = 0.92 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 16-Hn), 1.14 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 18-Hn), 1.20–1.34 (broad s, OH), 1.84–1.97 (m, 1H, 17-H), 2.16 (ddd, 2J(H,H) = 12.8 Hz, 3J(H,H) = 6.0 Hz, 4J(H,H) = 1.6 Hz, 1H, 16-Hx), 2.28 (ddd, 2J(H,H) = 12.8 Hz, 3J(H,H) = 6.0 Hz, 4J(H,H) = 1.2 Hz, 1H, 18-Hx), 2.46 (s, 3H, N-CH3), 3.20 (dd, 2J(H,H) = 11.2 Hz, 3J(H,H) = 5.6 Hz, 1H, CHaOH), 3.23 (dd, 2J(H,H) = 11.2 Hz, 3J(H,H) = 5.6 Hz, 1H, CHbOH), 3.78 (s, 3H, OCH3), 4.40 (s, 1H, 9-H), 5.36 (d, 2J(H,H) = 5.6 Hz, 1H, OCHaO), 5.39 (d, 2J(H,H) = 5.6 Hz, 1H, OCHbO), 7.11 (pseudo dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.2 Hz, 1H, 7-H), 7.16 (pseudo dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.4 Hz, 1H, 6-H), 7.21 (overlapped dd, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.6 Hz, 1H, 8-H), 7.24 (overlapped pseudo dt, 4J(H,H) = 1.2 Hz, 3J(H,H) = 6.8 Hz, 1H, 12-H), 7.28 (overlapped pseudo dt,4J(H,H) = 1.4 Hz, 3J(H,H) = 7.3 Hz, 1H, 13-H), 7.37 (dd, 3J(H,H) = 6.6 Hz, 4J(H,H) = 1.4 Hz, 1H, 11-H), 7.70–7.73 (ddm, 3J(H,H) = 7.4 Hz, 4J(H,H) = 1.6 Hz, 1H, 5-H), 7.75–7.78 ppm (ddm, 3J(H,H) = 7.6 Hz, 4J(H,H) = 1.6 Hz, 1H, 14-H); 13C-NMR (100.6 MHz, CDCl3): δ = 24.6 (CH3, N-CH3), 34.4 (CH2, C18), 35.0 (CH2, C16), 42.5 (CH, C17), 48.6 (CH, C9), 56.4 (CH3, OCH3), 64.9 (C, C9a), 65.0 (CH2, CH2OH), 65.4 (C, C3a), 87.0 (C, C4), 94.1 (CH2, OCH2O), 122.7 (CH, C5), 124.3 (CH, C14), 124.7 (CH, C8), 126.4 (CH, C11), 126.7 (CH, C13), 126.9 (CH, C6), 127.1 (2CH, C7 and C12), 138.3 (C, C8a), 138.4 (C, C10), 139.6 (C, C4a), 139.9 (C, C15), 177.5 (C, C3), 179.7 ppm (C, C1); IR (ATR): ṽ = 3457 (m), 1767 (w), 1685 (s), 1433 (m), 1378 (m), 1299 (m), 1165 (m), 1145 (m), 1072 (m), 1053 (s), 1004 (s), 794 (m), 760 cm−1 (s); accurate mass measurement: m/z calcd. for C25H25NO5 + Na+ 442.1625; found: 442.1616; elemental analysis calcd. (%) for C25H25NO5: C 71.58, H 6.01, N 3.34; found: C 71.87, H 5.99, N 3.24.
Analytical and spectroscopic data of 20. White solid, m.p. 186–187 °C (EtOAc/hexane); 1H-NMR (400 MHz, CDCl3): δ = 1.09–1.20 (m, 1H, 17-H), 1.27–1.35 (broad s, 1H, OH), 1.71 (dd, 2J(H,H) = 14.0 Hz, 3J(H,H) = 7.6 Hz, 1H, 16-Hn), 1.79–1.85 (overlapped dd, 2J(H,H) = 14.2 Hz, 3J(H,H) = 6.8 Hz, 1H, 18-Hx), 1.83–1.89 (overlapped dd, 2J(H,H) = 14.0 Hz, 3J(H,H) = 6.8 Hz, 1H, 16-Hx), 2.12 (dd, 2J(H,H) = 14.2 Hz, 3J(H,H) = 7.8 Hz, 1H, 18-Hn), 2.41 (s, 3H, N-CH3), 3.19 (broad d, 3J(H,H) = 5.2 Hz, 2H, CH2OH), 3.79 (s, 3H, OCH3), 4.43 (s, 1H, 9-H), 5.36 (d, 2J(H,H) = 5.8 Hz, 1H) and 5.39 (d, 2J(H,H) = 5.8 Hz) (OCH2O), 7.11–7.16 (overlapped pseudo dt, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.6 Hz, 1H, 7-H), 7.14–7.19 (overlapped pseudo dt, 4J(H,H) = 1.4 Hz, 3J(H,H) = 7.6 Hz, 1H, 6-H), 7.22–7.24 (overlapped dd, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.0 Hz, 1H, 8-H), 7.24–7.28 (overlapped pseudo dt, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.6 Hz, 1H, 12-H), 7.28–7.32 (overlapped pseudo dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 8.0 Hz, 1H, 13-H), 7.39 (dd, 4J(H,H) = 1.4 Hz, 3J(H,H) = 7.4 Hz, 1H, 11-H), 7.74 (dd, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.2 Hz, 1H, 5-H), 7.79 ppm (dd, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.6 Hz, 1H, 14-H); 13C-NMR (100.6 MHz, CDCl3): δ = 24.4 (CH3, N-CH3), 33.0 (CH2, C18), 35.2 (CH2, C16), 43.5 (CH, C17), 49.9 (CH, C9), 56.4 (CH3, OCH3), 65.4 (C, C9a), 65.5 (CH2, CH2OH), 66.5 (C, C3a), 87.5 (C, C4), 94.4 (CH2, OCH2O), 122.7 (CH, C5), 124.0 (CH, C14), 124.8 (CH, C8), 125.8 (CH, C11), 126.9 (2 CH, C6 and C13), 127.2 (CH, C7), 127.3 (CH, C12), 138.3 (C, C10), 138.4 (C, C8a), 139.1 (C, C4a), 140.0 (C, C15), 177.6 (C, C3), 179.7 ppm (C, C1); IR (ATR): ṽ = 3471 (m), 2946 (w), 2935 (w), 2855 (w), 1768 (w), 1691 (s), 1456 (m), n 1434 (m), 1381 (m), 1300 (m), 1162 (m), 1070 (m), 1043 (s), 1014 (m), 990 (s), 956 (s), 929 (m), 773 (m), 756 (m), 720 cm−1 (m); accurate mass measurement: m/z calcd. for C25H25NO5 + H+ 420.1805; found: 420.1815; elemental analysis calcd. (%) for C25H25NO5: C 71.58, H 6.01, N 3.34; found: C 71.32, H 5.81, N 3.11.
Syn-4-hydroxy-17-(methanesulfonyloxymethyl)-2-methyl-4,9-dihydro-4,9[1′,2′]benzeno-3a,9a-propano-1H-benz[f]isoindole-1,3(2H)-dione (21). MsCl (60 μL, 86 mg, 0.75 mmol) was added dropwise to a cold solution (0 °C, ice-water bath) of alcohol 19 (300 mg, 0.72 mmol) and anhydrous Et3N (0.23 mL, 166 mg, 1.65 mmol) in anhydrous CH2Cl2 (14 mL) under an Ar atmosphere, and the mixture was stirred for 4 h at 0 °C. Saturated aqueous solution of NaHCO3 (4 mL) and water (14 mL) were successively added to the reaction mixture. The organic phase was separated and the aqueous one was extracted with CH2Cl2 (2 × 20 mL). The combined organic phases were successively washed with aqueous 1 N HCl (3 × 15 mL), water (20 mL) and brine (20 mL), dried (anhydrous Na2SO4) and concentrated to dryness in vacuo to give a white solid (370 mg), that was subjected to column chromatography (silica gel 35–70 μm, 12 g, hexane/EtOAc mixtures) to give mesylate 21 (223 mg, 68% yield) as white solid, on elution with hexane/EtOAc 3:2 to 1:1. An analytical sample of 21 (65 mg) was obtained as white solid by crystallization of a sample of the above product (120 mg) from EtOAc (8 mL). m.p. 228–232 °C (EtOAc/hexane); 1H-NMR (400 MHz, CDCl3): δ = 1.13 (t, 2J(H,H) = 3J(H,H) = 12.6 Hz, 1H, 16-Hn), 1.23 (t, 2J(H,H) = 3J(H,H) = 12.6 Hz, 1H, 18-Hn), 2.09–2.21 (m, 1H, 17-H), 2.17–2.23 (ddm, 2J(H,H) = 12.8 Hz, 3J(H,H) = 6.0 Hz, 1H, 18-Hx), 2.30–2.36 (ddm, 2J(H,H) = 12.8 Hz, 3J(H,H) = 6.0 Hz, 1H, 16-Hx), 2.50 (s, 3H, N-CH3), 2.85 (s, 3H, CH3SO3), 3.81 (d, 3J(H,H) = 5.6 Hz, 2H, CH2OMs), 4.17 (s, 1H, OH), 4.43 (s, 1H, 9-H), 7.11 (pseudo dt, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.2 Hz, 1H, 7-H), 7.18 (pseudo dt, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.6 Hz, 1H, 6-H), 7.19–7.21 (overlapped dm, 3J(H,H) = 7.2 Hz, 1H, 8-H), 7.20–7.24 (overlapped pseudo dt, 4J(H,H) = 1.2 Hz, 3J(H,H) = 7.6 Hz, 1H, 12-H), 7.30 (pseudo dt, 4J(H,H) = 1.3 Hz, 3J(H,H) = 7.5 Hz, 1H, 13-H), 7.36 (dd, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.2 Hz, 1H, 11-H), 7.41–7.43 (dm, 3J(H,H) = 7.2 Hz, 1H, 5-H), 7.68–7.71 ppm (dm, 3J(H,H) = 7.2 Hz, 1H, 14-H); 13C-NMR (100.6 MHz, CDCl3): δ = 24.7 (CH3, N-CH3), 33.1 (CH2, C18), 35.2 (CH2, C16), 37.4 (CH3, CH3SO3), 39.2 (CH, C17), 48.0 (CH, C9), 63.6 (C, C9a), 63.9 (C, C3a), 69.4 (CH2, CH2OMs), 79.6 (C, C4), 121.0 (CH, C5), 123.1 (CH, C14), 124.5 (CH, C8), 126.1 (CH, C11), 126.9 (CH, C12), 127.0 (2 CH, C6 and C13), 127.1 (CH, C7), 137.6 (C, C10), 137.7 (C, C8a), 140.3 (C, C15), 141.4 (C, C4a), 179.5 (C, C1), 180.9 ppm (C, C3); IR (ATR): ṽ = 3450–3350 (w, max. at 3498 and 3385), 3028 (w), 2940 (w), 1767 (w), 1684 (s), 1454 (m), 1436 (m), 1351 (s), 1173 (s), 1072 (m), 1013 (m), 977 (s), 956 (s), 832 (m), 793 (m), 754 cm−1 (s); accurate mass measurement: m/z calcd. for C24H23NO6S + Na+: 476.1138; found: 476.1143; elemental analysis calcd. (%) for C24H23NO6S: C 63.56, H 5.11, N 3.09, S 7.07; found: C 63.26, H 5.23, N 2.80, S 6.22.
Syn-17-(methanesulfonyloxymethyl)-4-(metoxymethoxy)-2-methyl-4,9-dihydro-4,9[1′,2′]benzeno-3a,9a-propano-1H-benz[f]isoindole-1,3(2H)-dione (22). MsCl (0.23 mL, 334 mg, 2.92 mmol) was added dropwise to a cold solution (0° C, ice-water bath) of alcohol 19 (1.16 g, 2.77 mmol) and anhydrous Et3N (0.89 mL, 646 mg, 6.4 mmol) in anhydrous CH2Cl2 (60 mL) under an Ar atmosphere and the mixture was stirred for 4 h at 0 °C. Saturated aqueous solution of NaHCO3 (15 mL) and water (30 mL) were successively added to the reaction mixture. The organic phase was separated and the aqueous one was extracted with CH2Cl2 (2 × 30 mL). The combined organic phases were successively washed with aqueous 1 N HCl (3 × 30 mL), water (30 mL) and brine (30 mL), dried (anhydrous Na2SO4) and concentrated to dryness in vacuo to give a white solid (1.32 g), that was subjected to column chromatography (40 g neutral Al2O3, hexane/EtOAc mixtures) to give mesylate 22 (1.25 g, 91% yield as a white solid on elution with hexane/EtOAc 4:1 to 7:3. An analytical sample of 22 (54 mg) was obtained as white solid by crystallization of a sample of the above product (130 mg) from EtOAc (3 mL). m.p. 71–76 °C (dec) (EtOAc); 1H-NMR (400 MHz, CDCl3): δ = 0.96 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 16-Hn), 1.20 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 18-Hn), 2.03–2.15 (m, 1H, 17-H), 2.17–2.23 (ddm, 2J(H,H) = 12.8 Hz, 3J(H,H) = 6.0 Hz, 1H, 16-Hx), 2.30–2.36 (ddm, 2J(H,H) = 12.8 Hz, 3J(H,H) = 6.0 Hz, 1H, 18-Hx), 2.47 (s, 3H, N-CH3), 2.85 (s, 3H, CH3SO3), 3.79 (overlapped s, 3H, OCH3), 3.79–3.80 (overlapped d, 2H, CH2OMs), 4.41 (s, 1H, 9-H), 5.35 (d, 2J(H,H) = 6.0 Hz, 1H, OCHaO), 5.37 (d, 2J(H,H) = 6.0 Hz, 1H, OCHbO), 7.13 (pseudo dt, 4J(H,H) = 1.4 Hz, 3J(H,H) = 7.4 Hz, 1H, 7-H), 7.18 (pseudo dt, 4J(H,H) = 1.5 Hz, 3J(H,H) = 7.6 Hz, 1H, 6-H), 7.23 (dd, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.4 Hz, 1H, 8-H), 7.25–7.29 (pseudo dt, 4J(H,H) = 1.4 Hz, 3J(H,H) = 7.2 Hz, 1H, 12-H), 7.29–7.33 (pseudo dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.2 Hz, 1H, 13-H), 7.39 (dd, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.6 Hz, 1H, 11-H), 7.74–7.76 (overlapped dm, 3J(H,H) = 7.2 Hz, 1H, 5-H), 7.76–7.78 ppm (overlapped dm, 3J(H,H) = 7.2 Hz, 1H, 14-H); 13C-NMR (100.6 MHz, CDCl3): δ = 24.7 (CH3, N-CH3), 34.0 (CH2, C18), 34.7 (CH2, C16), 37.4 (CH3, CH3SO3), 39.3 (CH, C17), 48.5 (CH, C9), 56.4 (CH3, OCH3), 64.7 (C, C9a), 65.3 (C, C3a), 69.5 (CH2, CH2OMs), 87.3 (C, C4), 94.2 (CH2, OCH2O), 122.8 (CH, C5), 124.4 (CH, C14), 124.8 (CH, C8), 126.4 (CH, C11), 126.9 (CH, C13), 127.0 (CH, C6), 127.2 (CH) and 127.3 (CH) (C7 and C12), 138.0 (C, C8a), 138.2 (C, C10), 139.2 (C, C4a), 139.8 (C, C15), 177.0 (C, C3), 179.1 ppm (C, C1); IR (ATR): ṽ = 3012 (w), 2959 (w), 1769 (w), 1693 (s), 1455 (m), 1430 (m), 1378 (m), 1343 (s), 1171 (s), 1072 (m), 1050 (m), 1006 (m), 987 (s), 955 (s), 926 (s), 825 (s), 794 (m), 754 cm−1 (s); accurate mass measurement: m/z calcd. for C26H27NO7S + Na+: 520.1400; found: 520.1412; elemental analysis calcd. (%) for C26H27NO7S: C 62.76, H 5.47, N 2.82, S 6.44; found: C 62.87, H 5.48, N 2.69, S 6.26.
Syn-17-(iodomethyl)-4-(methoxymethoxy)-2-methyl-4,9-dihydro-4,9[1′,2′]benzeno-3a,9a-propano-1H-benz[f]isoindole-1,3(2H)-dione (23). A magnetically stirred solution of mesylate 22 (668 mg, 1.34 mmol) and NaI (2.03 g, 99%, 13.4 mmol) in anhydrous acetone (40 mL) was heated at reflux for 16 h under an Ar atmosphere. The mixture was allowed to cool to room temperature, the precipitate was filtered through a pad of Celite®, and the solid was washed with EtOAc (100 mL). The combined filtrate and washings were concentrated in vacuo, and the residue (860 mg) was subjected to column chromatography (30 g 50–200 μm silica gel, hexane/EtOAc mixtures). On elution with hexane/EtOAc 9:1, iodide 23 (617 mg, 87% yield) was obtained as yellow solid. An analytical sample of 23 (72 mg) was obtained as white solid by crystallization of a sample of the above product (100 mg) from EtOAc (0.3 mL) and hexane (0.5 mL). m.p. 176–177 °C (EtOAc/hexane); 1H-NMR (400 MHz, CDCl3): δ = 0.92 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 16-Hn), 1.15 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 18-Hn), 1.74–1.86 (m, 1H, 17-H), 2.23 (ddd, 2J(H,H) = 13.0 Hz, 3J(H,H) = 6.0 Hz, 4J(H,H) = 1.4 Hz, 1H, 16-Hx), 2.34 (ddd, 2J(H,H) = 13.2 Hz, 3J(H,H) = 6.0 Hz, 4J(H,H) = 1.4 Hz, 1H, 18-Hx), 2.47 (s, 3H, N-CH3), 2.81 (dd, 2J(H,H) = 10.0 Hz, 3J(H,H) = 5.6 Hz, 1H, CHaI), 2.84 (dd, 2J(H,H) = 10.0 Hz, 3J(H,H) = 5.6 Hz, 1H, CHbI), 3.79 (s, 3H, OCH3), 4.39 (s, 1H, 9-H), 5.36 (d, 2J(H,H) = 6.0 Hz, 1H, OCHaO), 5.37 (d, 2J(H,H) = 6.0 Hz, 1H, OCHbO), 7.12 (pseudo dt, 4J(H,H) = 1.5 Hz, 3J(H,H) = 7.2 Hz, 1H, 7-H), 7.17 (pseudo dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.4 Hz, 1H, 6-H), 7.22 (dd, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.6 Hz, 1H, 8-H), 7.27 (pseudo dt, 4J(H,H) = 1.4 Hz, 3J(H,H) = 7.2 Hz, 1H, 12-H), 7.31 (pseudo dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.6 Hz, 1H, 13-H), 7.39 (dd, 4J(H,H) = 1.4 Hz, 3J(H,H) = 7.2 Hz, 1H, 11-H), 7.73–7.76 (overlapped dm, 3J(H,H) = 7.6 Hz, 1H, 5-H), 7.75–7.77 ppm (overlapped dm, 3J(H,H) = 7.6 Hz, 1H, 14-H); 13C-NMR (100.6 MHz, CDCl3): δ = 8.4 (CH2, CH2I), 24.7 (CH3, N-CH3), 38.0 (CH2, C18), 38.7 (CH2, C16), 41.5 (CH, C17), 48.6 (CH, C9), 56.4 (CH3, OCH3), 64.8 (C, C9a), 65.6 (C, C3a), 87.3 (C, C4), 94.1 (CH2, OCH2O), 122.8 (CH, C5), 124.4 (CH, C14), 124.8 (CH, C8), 126.4 (CH, C11), 127.0 (CH, C6 and C13), 127.2 (CH, C7), 127.3 (CH, C12), 138.1 (C, C8a), 138.2 (C, C10), 139.3 (C, C4a), 139.8 (C, C15), 177.2 (C, C3), 179.3 ppm (C, C1); IR (ATR): ṽ = 2936 (w), 1769 (w), 1693 (s), 1453 (m), 1428 (m), 1376 (m), 1299 (m), 1188 (m), 1161 (m), 1093 (m), 1045 (s), 1019 (m), 1008 (m), 991 (m), 759 cm−1 (s); accurate mass measurement: m/z calcd. for C25H24INO4 + Na+: 552.0642; found: 552.0630; elemental analysis calcd. (%) for C25H24INO4: C 56.72, H 4.57, N 2.65, I 23.97; found: C 56.98, H 4.56, N 2.53, I 23.39.
(3aR*,9aR*,17S*)-17-{[(1R*)-1-(Methoxycarbonyl)-2-oxocyclopentyl]methyl}-4-(methoxymethoxy)-2-methyl-4,9-dihydro-4,9[1′,2′]benzeno-3a,9a-propano-1H-benz[f]isoindole-1,3(2H)-dione 24 and mixture of 24 and its C1′ epimer 25. A solution of methyl 2-oxocyclopentanecarboxylate (0.47 mL, 95% content, 500 mg, 3.5 mmol) was added dropwise to a cold (0 °C, ice-water bath), magnetically-stirred suspension of NaH (117 mg, 60% content, 2.9 mmol) in anhydrous DMF (5 mL) and the mixture was stirred at this temperature for 1 h. Then, a solution of iodide 23 (1.03 g, 1.95 mmol) was added at once, and the reaction mixture was stirred at 0 °C for 2 h and then it was heated at 80 °C for 63 h. A saturated aqueous solution of NH4Cl (6 mL) and water (6 mL) were added, and the mixture was extracted with EtOAc (4 × 100 mL). The combined organic extracts were dried (anhydrous Na2SO4) and concentrated in vacuo. The obtained residue (1.3 g) was subjected to column chromatography (100 g 50–200 μm neutral Al2O3, hexane/EtOAc mixtures). Alkene 18 (135 mg, 17% yield) was isolated on elution with hexane/EtOAc 4:1. A stereoisomeric mixture of 24 and 25 (about 1:1 mixture of C1′-epimers, 628 mg, 59% yield) was obtained as white solid on elution with hexane/EtOAc from 7:3 to 3:2. Crystallization of part of the above product (100 mg) from EtOAc (0.2 mL)/hexane (0.2 mL) gave a mixture of 24 and 25 in a ratio 24/25 = 7:1 (1H-NMR, 22 mg). The mother liquors were concentrated in vacuo and crystallized from EtOAc (0.4 mL)/hexane (1.4 mL) gave a mixture of 24 and 25 in a ratio 24/25 = 7.5:1 (1H-NMR, 15 mg). Recrystallization of the above product (37 mg) from EtOAc (0.4 mL)/hexane (2 mL) gave two crops (16 and 13.5 mg) of pure 24 as white solid. M.p. 159–162 °C (EtOAc/hexane); 1H-NMR (400 MHz, CDCl3): δ = 0.80 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 16-Hn), 1.01 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 18-Hn), 1.16 (dd, 2J(H,H) = 14.4 Hz, 3J(H,H) = 6.4 Hz, 1H, C1-CHa), 1.57–1.71 (complex signal, 2H, 5′-Ha and 17-H), 1.78 (dd, 2J(H,H) = 14.0 Hz, 3J(H,H) = 6.8 Hz, 1H, C1-CHb), 1.81–1.97 (complex signal, 4′-Ha and 4′-Hb), 2.07 (ddd, 2J(H,H) = 12.8 Hz, 3J(H,H) = 6.0 Hz, 4J(H,H) = 1.2 Hz, 16-Hx), 2.13–2.21 (overlapped dd, 2J(H,H) = 19.2 Hz, 3J(H,H) = 10.0 Hz, 3′-Ha), 2.18–2.23 (overlapped dd, 2J(H,H) = 12.8 Hz, 3J(H,H) = 6.0 Hz, 4J(H,H) = 1.6 Hz, 18-Hx), 2.33 (dddd, 2J(H,H) = 19.2 Hz, 3J(H,H) = 8.6 Hz, 3J(H,H) = 4.8 Hz, 4J(H,H) = 1.2 Hz, 1H, 3′-Hb), 2.42–2.52 (m, 1H, 5′-Hb), 2.45 (s, 3H, N-CH3), 3.62 (3H, COOCH3), 3.77 (s, 3H, OCH3), 4.35 (s, 1H, 9-H), 5.35 (s, 2H, OCH2O), 7.10 (dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.4 Hz, 1H, 7-H), 7.15 (dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.4 Hz, 6-H), 7.18–7.20 (m, 8-H), 7.24 (dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.2 Hz, 12-H), 7.29 (dt, 4J(H,H) = 1.6 Hz, 3J(H,H) = 7.2 Hz, 13-H), 7.33–7.36 (m, 1H, 11-H), 7.70–7.72 (m, 1H, 14-H), 7.72–7.74 ppm (m, 1H, 5-H); 13C-NMR (100.6 MHz, CDCl3): δ = 19.4 (CH2, C4′), 24.7 (CH3, N-CH3), 32.6 (CH2, C5′), 36.7 (CH, C17), 37.3 (CH2, C3′), 38.2 (CH2, C1-CH2), 38.4 (CH2, C16), 38.5 (CH2, C18), 48.6 (CH, C9), 52.6 (CH3, COOCH3), 56.3 (CH3, OCH3), 60.1 (C, C1), 64.8 (C, C9a), 65.3 (C, C3a), 87.1 (C, C4), 94.1 (CH2, OCH2O), 122.7 (CH, C5), 124.3 (CH, C14), 124.7 (CH, C8), 126.3 (CH, C11), 126.9 (2 CH, C6 and C13), 127.1 (CH, C7), 127.2 (CH, C12), 138.2 (C, C8a), 138.3 (C, C10), 139.5 (C, C4a), 139.9 (C, C15), 170.5 (C, COOCH3), 177.5 (C, C3), 179.6 (C, C1), 214.0 ppm (C, C2′); IR (ATR): ṽ = 2945 (w), 1767 (w), 1746 (w), 1713 (m), 1695 (s), 1452 (m), 1439 (m), 1429 (m), 1377 (m), 1305 (m), 1256 (m), 1238 (m), 1204 (m), 1186 (m), 1165 (m), 1046 (s), 10113 (m), 997 (m), 752 cm−1 (m); accurate mass measurement: m/z calcd. for C32H33NO7 + H+: 544.2330; found: 544.2329; elemental analysis calcd. (%) for C32H33NO7: C 70.70, H 6.12, N 2.58; found: C 70.38, H 5.95, N 2.39.
Significant 1H-NMR data of 25 from the 1:1 mixture 24 and 25 and analytical data from this mixture: 1H-NMR (400 MHz, CDCl3): δ = 0.79 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 16-Hn), 1.03 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 18-Hn), 5.34 (d, 2J(H,H) = 5.2 Hz, 1H, OCHaO), 5.37 (d, 2J(H,H) = 5.2 Hz, 1H, OCHbO); accurate mass measurement: m/z calcd. for C32H33NO7 + H+: 544.2330; found: 544.2346; elemental analysis calcd. (%) for C32H33NO7: C 70.70, H 6.12, N 2.58; found: C 70.45, H 6.13, N 2.46.
(3aR*,9aR*,17S*)-17-{[(1′R*)-1-(Methoxycarbonyl)-2-oxocyclopent-3-en-1-yl]methyl}-4-(methoxymethoxy)-2-methyl-4,9-dihydro-4,9[1′,2′]benzeno-3a,9a-propano-1H-benz[f]isoindole-1,3(2H)-dione (26). Trimethylsilyl trifluoromethanesulfonate (80 μL, 98% content, 96 mg, 0.43 mmol) was added at once to a cold (0 °C, ice-water bath) solution of keto ester 24 (101 mg, 0.19 mmol) and anhydrous Et3N (0.13 mL, 94 mg, 0.93 mmol) in anhydrous CH2Cl2 (1 mL), under an Ar atmosphere, and the mixture was stirred at room temperature for 30 min. The solution was cooled to 0 °C (ice-water bath), a saturated aqueous solution of NaHCO3 (1 mL) was added, the organic phase was separated, and the aqueous one was extracted with CH2Cl2 (2 × 4 mL). The combined organic phases were dried (anhydrous Na2SO4) and concentrated in vacuo to give a brown oily residue, of the corresponding trimethylsilyl enol ether (118 mg), which was used as such in the next step. Pd(OAc)2 (43.5 mg, 98% content, 0.19 mmol) was added to a solution of the above enol ether (118 mg) in anhydrous DMSO (5 mL) and the mixture was stirred for 16 h at room temperature. The suspension was filtered through a pad of Celite®, and the solid was washed with EtOAc (10 mL). The combined filtrate and washings were concentrated in vacuo, the residue was taken in EtOAc (15 mL) and was washed with water (15 mL). The aqueous phase was extracted with EtOAc (2 × 20 mL). The combined organic phase and extracts were washed with brine (2 × 25 mL), dried (anhydrous Na2SO4) and concentrated in vacuo. The brown oily residue (120 mg) was subjected to column chromatography (10 g, 50–200 μm basic Al2O3, hexane/EtOAc mixtures). On elution with hexane/EtOAc from 9:1 to 3:2, enone 26 (69 mg, 68% yield from 24) was obtained as white solid. m.p. 160–162 °C (EtOAc/hexane); 1H-NMR (400 MHz, CDCl3): δ = 0.86 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 16-Hn), 1.04 (t, 2J(H,H) = 3J(H,H) = 12.8 Hz, 1H, 18-Hn), 1.33 (dd, 2J(H,H) = 14.4 Hz, 3J(H,H) = 6.4 Hz, 1H, C1′-CHa), 1.55–1.68 (m, 1H, 17-H), 1.84 (dd, 2J(H,H) = 14.4 Hz, 3J(H,H) = 6.4 Hz, 1H, C1′-CHb), 2.07 (ddd 2J(H,H) = 12.8 Hz, 3J(H,H) = 5.8 Hz, 4J(H,H) = 1.6 Hz, 1H, 16-Hx), 2.19 (ddd, 2J(H,H) = 13.2 Hz, 3J(H,H) = 6.0 Hz, 4J(H,H) = 1.3 Hz, 1H, 18-Hx), 2.42 (overlapped dt, 2J(H,H) = 19.2 Hz, 3J(H,H) = 2.4 Hz, 1H, 5′-Ha), 2.44 (overlapped s, 3H, N-CH3), 3.22 (dt, 2J(H,H) = 19.2 Hz, 3J(H,H) = 2.6 Hz, 1H, 5′-Hb), 3.62 (s, 3H, COOCH3), 3.77 (s, 3H, OCH3), 4.35 (s, 1H, 9-H), 5.34 (s, 2H, OCH2O), 6.09 (dt, 3J(H,H) = 5.6 Hz, 3J(H,H) = 2.0 Hz, 1H, 3′-H), 7.11 (dt, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.6 Hz, 1H, 7-H), 7.15 (dt, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.6 Hz, 1H, 6-H), 7.19 (ddm, 3J(H,H) = 6.8 Hz, 4J(H,H) = 1.6 Hz, 1H, 8-H), 7.25 (dt, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.6 Hz, 1H, 12-H), 7.28 (dt, 3J(H,H) = 7.2 Hz, 4J(H,H) = 1.6 Hz, 1H, 13-H), 7.35 (ddm, 3J(H,H) = 6.4 Hz, 4J(H,H) = 1.6 Hz, 1H, 11-H) 7.69–7.74 ppm (complex signal, 3H, 4′-H, 5-H and 14-H); 13C-NMR (100.6 MHz, CDCl3): δ = 24.7 (CH3, N-CH3), 36.8 (CH, C17), 38.3 (CH2, C1′-CH2), 38.6 (CH2, C16), 38.7 (CH2, C18), 38.9 (CH2, C5′), 48.6 (CH, C9), 52.8 (CH3, COOCH3), 56.4 (CH3, OCH3), 57.6 (C, C1′), 64.7 (C, C9a), 65.3 (C, C3a), 87.2 (C, C4), 94.1 (OCH2O), 122.7 (CH, C5), 124.3 (CH, C14), 124.7 (CH, C8), 126.3 (CH, C11), 126.89 (CH) and 126.91 (CH) (C6 and C13), 127.1 (CH, C7), 127.2 (CH, C12), 131.6 (CH, C3′), 138.20 (C) and 138.23 (C) (C8a and C10), 139.5 (C, C4a), 139.8 (C, C15), 163.9 (CH, C4′), 170.3 (COOCH3), 177.4 (C, C3), 179.6 (C, C1), 204.8 ppm (C, C2′); IR (ATR): ṽ = 2940 (w), 1767 (w), 1741 (m), 1695 (s), 1454 (m), 1423 (m), 1240 (m), 1186 (m), 1168 (m), 1054 (m), 1031 (m), 1013 (m), 984 (s), 760 (s), 749 cm−1 (m); accurate mass measurement: m/z calcd. for C32H31NO7 + Na+: 564.1993; found: 564.1993; elemental analysis calcd. (%) for C32H31NO7·0.25H2O: C 70.38, H 5.81, N 2.56; found: C 70.28, H 5.73, N 2.39.
Stereoisomeric mixture of 26 and its C1′ epimer (27). Trimethylsilyl trifluoromethanesulfonate (310 μL, 98% content, 372 mg, 1.68 mmol) was added at once to a cold (0 °C, ice-water bath) solution of a stereoisomeric mixture of keto esters 24 and 25 (approximate ratio 24/25 = 1:1, 400 mg, 0.74 mmol) and anhydrous Et3N (0.51 mL, 370 mg, 3.7 mmol) in anhydrous CH2Cl2 (3 mL) under an Ar atmosphere, and the mixture was stirred at room temperature for 30 min. The solution was cooled to 0 °C (ice-water bath), a saturated aqueous solution of NaHCO3 (3 mL) was added, the organic phase was separated and the aqueous one was extracted with CH2Cl2 (2 × 6 mL). The combined organic phases were dried (anhydrous Na2SO4) and concentrated in vacuo to give a brown oily residue, mixture of the corresponding trimethylsilyl enol ethers (412 mg), which was used as such in the next step. Accurate mass measurement: m/z calcd. for C35H41NO7Si + H+: 616.2725; found: 616.2731.
Pd(OAc)2 (169 mg, 98% content, 0.74 mmol) was added to a solution of the above stereoisomeric mixture of enol ethers (412 mg) in anhydrous DMSO (15 mL) and the mixture was stirred at room temperature for 16 h. The suspension was filtered through a pad of Celite®, and the solid was washed with EtOAc (20 mL). The combined filtrate and washings were concentrated in vacuo, the residue was taken in EtOAc (20 mL) and was washed with water (20 mL). The aqueous phase was extracted with EtOAc (2 × 20 mL). The combined organic phase and extracts were washed with brine (2 × 25 mL), dried (anhydrous Na2SO4) and concentrated in vacuo to give a brown oily residue (390 mg), which was subjected to column chromatography (10 g, 50–200 μm basic Al2O3, hexane/EtOAc mixtures). On elution with hexane/EtOAc 3:2, a stereoisomeric mixture of enones 26 and 27 in an approximate ratio 26/27 = 1:1 (301 mg, 75% yield from 24 + 25) was obtained as white solid. m.p. 76–86 °C (EtOAc/hexane); accurate mass measurement: m/z calcd. for C32H31NO7 + H+: 542.2173; found: 542.2179; elemental analysis calcd. (%) for C32H31NO7 1.5H2O: C 67.59, H 6.03, N 2.46; found: C 67.88, H 5.78, N 2.29.
Methyl 2-methyl-1,3-dioxo-2,3,5,6-tetrahydro-1H,4H-3a,7,8-(epiprop[2]ene[1,1,3]triyl)-5,8a-methanocyclo hepta[c]pyrrole-7(8H)-carboxylate 13.
- (a)
NaBH4 (67 mg, 1.72 mmol) was added portionwise to a cold (−40 °C) solution of a diastereomeric mixture of enones 26 and 27 (233 mg, 0.43 mmol) and CeCl3·7H2O (417 mg, 1.12 mmol) in a mixture of THF (4.5 mL) and MeOH (5 mL) and the mixture was stirred at this temperature for 1 h. A saturated aqueous solution of NaHCO3 (2 mL) and water (2 mL) were added and the mixture was extracted with CH2Cl2 (3 × 15 mL). The combined organic phases were dried (anhydrous Na2SO4) and concentrated in vacuo to give a complex diastereoisomeric mixture of cyclopentenols 28 (172 mg) as white solid, that was used as such in the next step.
- (b)
p-TsOH H2O (16.4 mg, 0.09 mmol) was added to a solution of the above cyclopentenols 28 (172 mg) in benzene (15 mL) and the solution was heated under reflux for 14 h with azeotropic elimination of water with a Dean-Stark equipment. Then, the solution was allowed to cool to room temperature and was treated with solid K2CO3 (about 100 mg). The suspension was filtered and the filtrate was concentrated in vacuo to give cyclopentadiene alcohol 29 as light brown solid (180 mg) that was used as such in the next step.
- (c)
A suspension of 30% KH in mineral oil (150 mg, about 1.1 mmol) was placed in a three-necked flask, provided with a low temperature thermometer and an Ar atmosphere. The mineral oil was removed by washing with anhydrous THF (5 × 5 mL) and after the last washing anhydrous THF (12 mL) was added. The mixture was cooled to 0 °C with an ice-water bath and a solution of the above product 29 (180 mg) in anhydrous THF (12 mL) was added dropwise with magnetic stirring and the mixture was stirred at room temperature for 1 h. The excess KH was destroyed by careful addition of 2 N HCl (1 mL) plus 5 N HCl (0.5 mL) under an Ar atmosphere. The solution was dried (anhydrous MgSO4, about 3 g), filtered under vacuum and concentrated under reduced pressure to give an oily residue (263 mg) that was subjected to column chromatography (20 g, 35–70 μm silica gel, hexane/EtOAc mixtures). Compound 13 (37 mg, 30% yield from 26 + 27) was obtained as a light yellow solid, on elution with hexane/EtOAc 4:1. The 1H-NMR data of this compound coincide with those previously described.
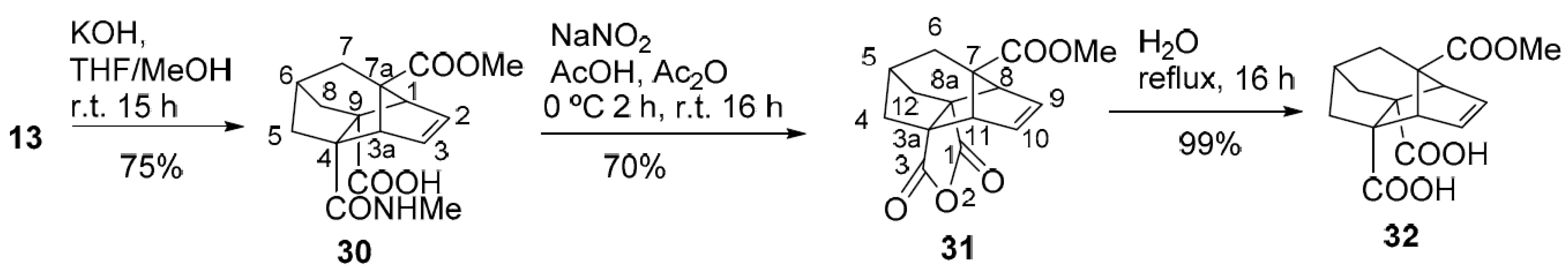
7a-(Methoxycarbonyl)-4-(methylcarbamoyl)-3a,4,5,6,7,7a-hexahydro-1H-1,4,6-(epiethane[1,1,2]triyl)indene-9-carboxylic acid (30). KOH (236 mg, 4.2 mmol) was added to a solution of compound 13 (121 mg, 0.42 mmol) in a mixture of THF (2 mL) and MeOH (2 mL) and the mixture was stirred at room temperature for 15 h. Water (8 mL) was added and the mixture was extracted with EtOAc (3 × 10 mL). The aqueous phase was made acidic with 1N HCl (6 mL) and it was extracted with EtOAc (3 × 10 mL). The combined organic phases were washed with water (2 × 10 mL), dried (anhydrous Na2SO4) and concentrated in vacuo to give amide acid (30) (96 mg, 75% yield) as yellow solid. An analytical sample of (30) (13 mg) was obtained as yellow solid by crystallization of a sample (19 mg) of the above product in MeOH (0.5 mL). m.p. 193–194 °C (MeOH); 1H-NMR (CD3OD): δ = 1.81 (d, 3J(H,H) = 2.8 Hz, 2H, 7-Ha and 7-Hb), 1.90 (dd, 2J(H,H) = 11.6 Hz, 4J(H,H) = 2.4 Hz, 1H) and 1.94 (dd, 2J(H,H) = 11.6 Hz, 4J(H,H) = 2.4 Hz, 1H) (5-Hanti and 8-Hanti), 2.23 [dd, 2J(H,H) = 11.6 Hz, 3J(H,H) = 4.0 Hz, 1H, 5-Hsyn), 2.41 [dd, 2J(H,H) = 11.6 Hz, 3J(H,H) = 4.0 Hz, 1H, 8-Hsyn), 2.46–2.49 (m, 1H, 6-H), 2.67 [s, 3H, N-CH3], 2.96–2.98 [m, 2H, 1-H and 3a-H], 3.58 [s, 3H, OCH3], 6.19 (ddd, 3J(H,H) = 5.6 Hz, 3J(H,H) = 2.8 Hz, 4J(H,H) = 1.2 Hz, 1H, 2-H), 6.34 ppm (ddd, 3J(H,H) = 5.6 Hz, 3J(H,H) = 2.8 Hz, 4J(H,H) = 1.2 Hz, 1H, 3-H); 13C-NMR (CD3OD): δ = 26.7 [CH3, N-CH3], 35.7 (CH, C6), 36.2 (CH2, C7), 44.3 (CH2, C5), 44.6 (CH2, C8), 52.2 (CH3, OCH3), 59.16 (CH, C3a), 59.20 (CH, C1), 63.4 (C, C9), 65.3 (C, C4), 68.8 (C, C7a), 136.0 (CH, C3), 138.8 (CH, C2), 176.3 (C, CON), 176.5 (C, COOCH3), 177.7 (C, COOH); IR (ATR): ṽ = 3409 (m), 3200–2500 [broad band, max. at 2981 (w), 2949 (w), 2930 (w), 2847 (w)], 1731 (s), 1709 (s), 1632 (s), 1531 (s), 1418 (m), 1294 (m), 1280 (m), 1258 (s), 1246 (s), 1209 (m), 1188 (s), 1137 (m), 1105 (m), 1034 (m), 988 (m), 829 (m), 734 (m), 707 (s), 619 cm−1 (m); accurate mass measurement: m/z calcd. for C16H19NO5 − H− 304.1190; found: 304.1188; elemental analysis calcd. (%) for C16H19NO5 0.25H2O: C 62.03, H 6.34, N 4.52; found: C 62.07, H 6.27, N 4.60.
Methyl 1,3-dioxo-5,6-dihydro-1H,3H,4H-3a,7,8-(epiprop[2]ene[1,1,3]triyl)-5,8a-methanocyclohepta[c] furan-7(8H)-carboxylate (31). Solid NaNO2 (305 mg, 4.4 mmol) was added portionwise in 2 h to a cold (0 °C, ice-water bath) solution of amide acid 30 (67 mg, 0.22 mmol) in Ac2O (2.75 mL) and AcOH (1.4 mL) and the mixture was stirred at room temperature for 16 h. The mixture was cooled to 0 °C (ice-water bath), water (5 mL) was added and the solution was extracted with CH2Cl2 (3 × 8 mL). The combined organic extracts were washed with 10% aqueous solution of Na2CO3 (3 × 8 mL) and water (8 mL), dried (anhydrous Na2SO4) and concentrated in vacuo to give anhydride 31 (63.8 mg) as white solid. Sublimation of the above product (120 °C/0.2–0.5 torr) gave the analytical sample of 31 (41.5 mg, 70% yield) as white solid. m.p. 184–185 °C (EtOAc); 1H-NMR (CDCl3, 400 MHz): δ = 1.92–1.93 (dm, 3J(H,H) = 2.4 Hz, 2H, 6-H2), 2.11–2.15 [dm, 2J(H,H) = 12.0 Hz, 2H] and 2.18–2.22 [dm, 2J(H,H) = 12.0 Hz, 2H] [4(12)-Hsyn and 4(12)-Hanti], 2.71–2.75 [m, 1H, 5-H], 3.34 [t, 3J(H,H) = 4J(H,H) = 2.0 Hz, 2H, 8(11)-H], 3.62 [s, 3H, OCH3], 6.38 ppm [t, 3J(H,H) = 4J(H,H) = 2.0 Hz, 2H, 9(10)-H]; 13C-NMR (CDCl3, 100.6 MHz): δ = 35.0 (CH2, C6), 38.4 (CH, C5), 39.3 [CH2, C4(12)], 52.2 (CH3, OCH3), 56.3 [CH, C8(11)], 61.3 [C, C3a(8a)], 72.7 (C, C7), 136.4 [CH, C9(10)], 171.4 [C, C1(3)], 172.5 ppm (C, COOCH3); IR (ATR): ṽ = 2950 (w), 2918 (w), 1834 (w), 1770 (s), 1731 (s), 1696 (w), 1284 (m), 1237 (s), 1174 (m), 941 (m), 921 (vs), 899 (s), 750 (m), 741 (m), 717 cm−1 (s); accurate mass measurement: m/z calcd. for C15H14O5 + H+ 275.0914; found: 275.0920; elemental analysis calcd. (%) for C15H14O5: C 65.69, H 5.14. Found: C 65.62, H 5.12.
7a-(Methoxycarbonyl)-3a,4,5,6,7,7a-hexahydro-1H-1,4,6-(epiethane[1,1,2]triyl)indene-4,9-dicarboxylic acid (32). A mixture of anhydride (31) (17 mg, 62 μmol) in water (2 mL) was heated under reflux for 16 h. The solution was allowed to cool to room temperature and was concentrated in vacuo to give diacid 32 (18 mg, 99% yield) as white solid. m.p. 174–177 °C (water); 1H-NMR (CD3OD): δ = 1.80 (d, 3J(H,H) = 2.8 Hz, 2H, 7-H2), 1.92 [d, 2J(H,H) = 11.2 Hz, 2H, 5(8)-Hanti], 2.35 [dd, 2J(H,H) = 11.6 Hz, 3J(H,H) = 4.0 Hz, 2H, 5(8)-Hsyn], 2.44–2.46 (m, 1H, 6-H), 2.96 [t, 3J(H,H) = 4J(H,H) = 2.0 Hz, 2H, 1(3a)-H], 3.57 [s, 3H, OCH3], 4.85 (s, 12H, 2 COOH and CD3OH), 6.24 ppm [d, 3J(H,H) = 4J(H,H) = 2.0 Hz, 2H, 2(3)-H]; 13C-NMR (CD3OD): δ = 35.6 (CH, C6), 36.2 (CH2, C7), 44.1 [CH2, C5(8)], 52.2 (CH3, OCH3), 59.3 [CH, C1(3a)], 63.8 [C, C4(9)], 68.8 (C, C7a), 137.4 [CH, C2(3)], 176.6 (C, COOH), 177.2 ppm (C, COOCH3); IR (ATR): ṽ = 3200–2800 [broad band, max. at 2977 (w)], 1725 (m), 1688 (s), 1407 (m), 1303 (m), 1272 (m), 1253 (s), 1243 (s), 1190 (m), 1102 (m), 1017 (m), 935 cm−1 (m); accurate mass measurement: m/z calcd. for C15H16O6 − H− 291.0874; found: 291.0878; elemental analysis calcd. (%) for C15H16O6: C 61.64, H 5.52.
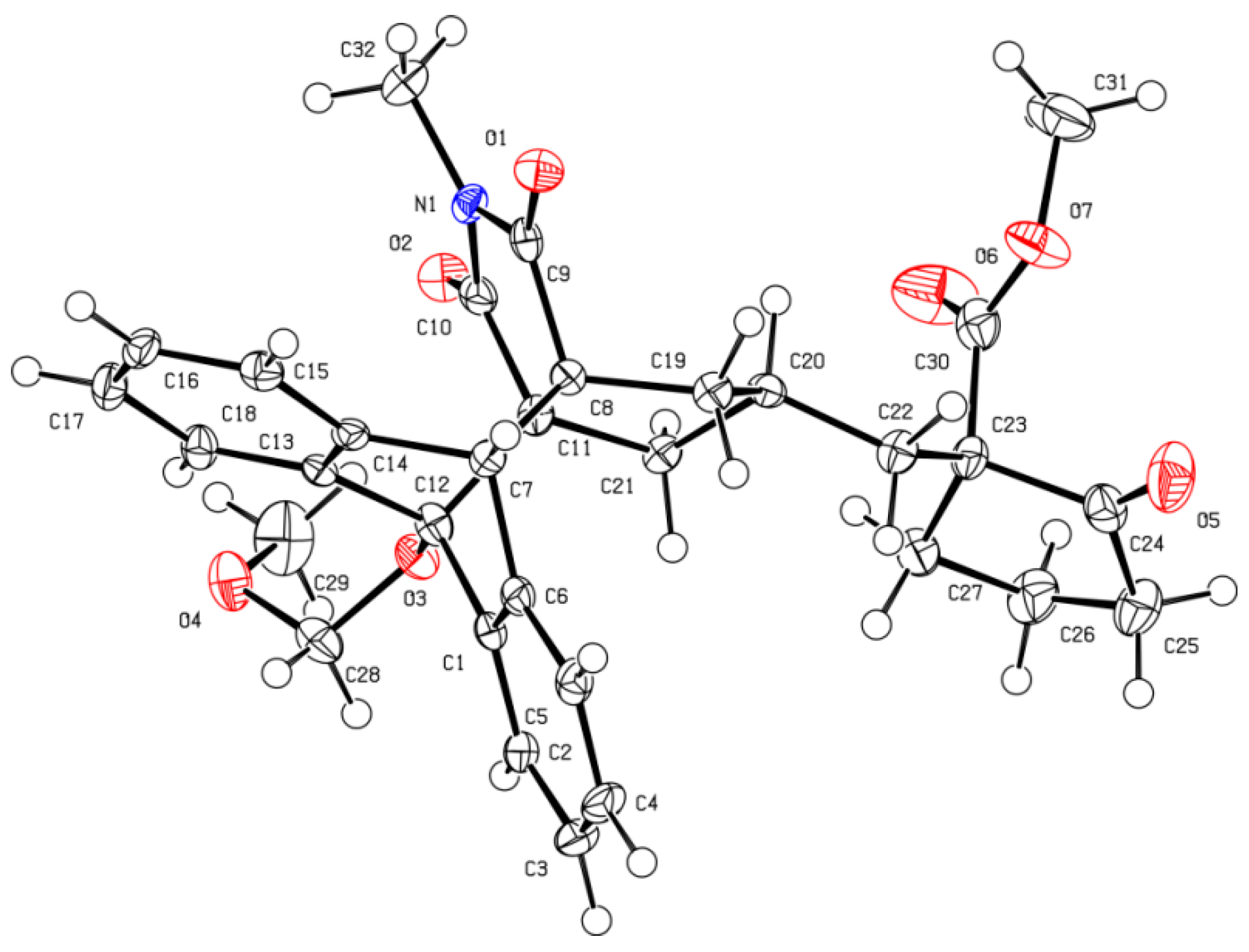
X-ray Crystal-structure determination of compound 24. A colorless prism-like specimen of C
32H
33NO
7, approximate dimensions 0.050 mm × 0.080 mm × 0.467 mm, was used for the X-ray crystallographic analysis. The X-ray intensity data were measured on a D8 Venture system equipped with a multilayer monochromator and a Mo microfocus (λ = 0.71073 Å). The frames were integrated with the Bruker SAINT software package [
14,
15] using a narrow-frame algorithm. The integration of the data using an orthorhombic unit cell yielded a total of 20021 reflections to a maximum θ angle of 26.41° (0.80 Å resolution), of which 5444 were independent (average redundancy 3.678, completeness = 99.7%, R
int = 11.85%, R
sig = 12.14%) and 3454 (63.45%) were greater than 2σ(F
2). The final cell constants of
a = 8.2651(3) Å,
b = 11.0782(6) Å,
c = 29.0497(15) Å, volume = 2659.9(2) Å
3, are based upon the refinement of the XYZ-centroids of reflections above 20 σ(I). Data were corrected for absorption effects using the multi-scan method (SADABS) [
16]. The calculated minimum and maximum transmission coefficients (based on crystal size) are 0.6511 and 0.7454. The structure was solved and refined using the Bruker SHELXTL software package [
17], using the space group P 21 21 21, with Z = 4 for the formula unit, C
32H
33NO
7. The final anisotropic full-matrix least-squares refinement on F
2 with 364 variables converged at
R1 = 5.60%, for the observed data and
wR2 = 10.62% for all data. The goodness-of-fit was 1.016. The largest peak in the final difference electron density synthesis was 0.270 e Å
−3 and the largest hole was −0.244 e Å
−3 with an RMS deviation of 0.060 e Å
−3. On the basis of the final model, the calculated density was 1.357 g cm
−3 and F(000), 1152 e.