On the Morphology of Group II Metal Fluoride Nanocrystals at Finite Temperature and Partial Pressure of HF
Abstract
:1. Introduction
2. Results
2.1. Bulk Calculations
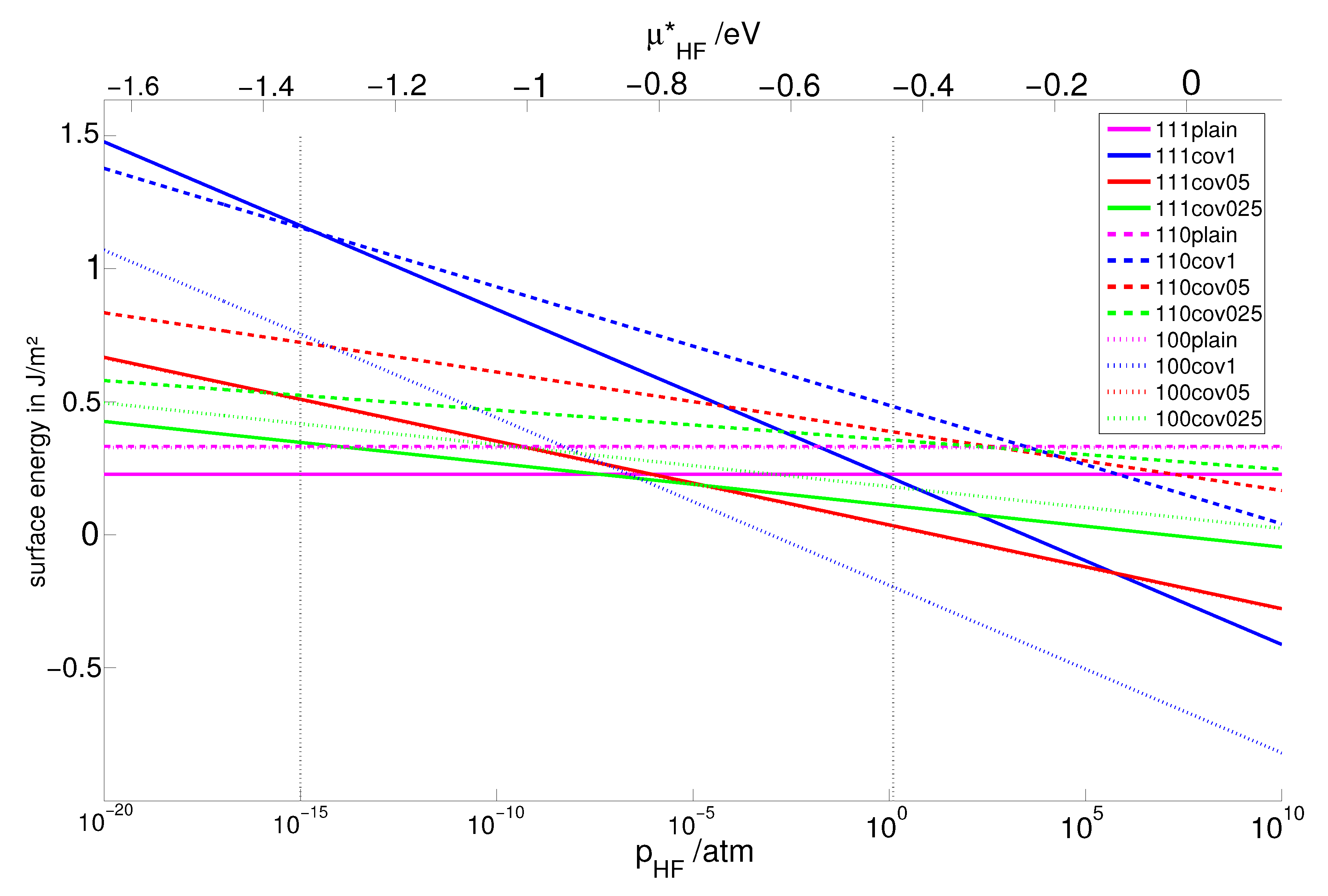
2.2. Clean Surfaces
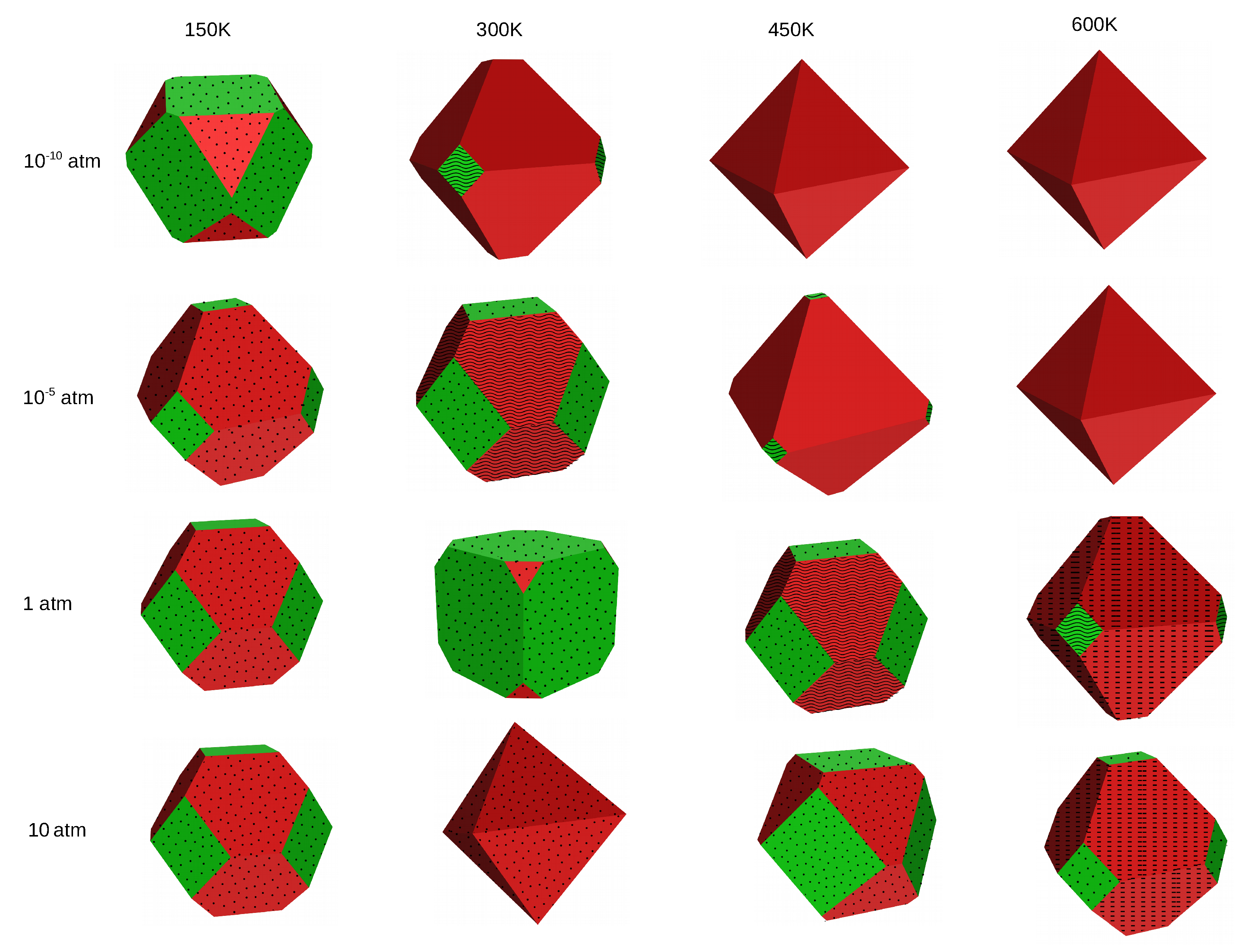
2.3. Crystal Shapes at Thermodynamic Equilibrium
2.4. Comparison Experimental Found Crystal Shapes
3. Material and Methods
3.1. Computational Details
3.2. Synthesis of Nanoparticles
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schmidt, L.; Emmerling, F.; Kirmse, H.; Kemnitz, E. Sol-gel synthesis and characterisation of nanoscopic strontium fluoride. RSC Adv. 2014, 4, 32–38. [Google Scholar] [CrossRef]
- Ling, L.; Xu, X.; Choi, G.Y.; Billodeaux, D.; Guo, G.; Diwan, R.M. Novel F-releasing Composite with Improved Mechanical Properties. J. Dent. Res. 2009, 88, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Moreau, J.L.; Sun, L.; Chow, L.C. Novel CaF2 Nanocomposite with High Strength and Fluoride Ion Release. J. Dent. Res. 2010, 89, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Kawano, K.; Ohya, T.; Tsurumi, T.; Katoh, K.; Nakata, R. X-ray excitation fluorescence spectra of the Eu2+-stabilized VK center in alkaline-earth fluoride mixed-crystal systems. Phys. Rev. B 1999, 60, 11984–11992. [Google Scholar] [CrossRef]
- Ayala, A.P. Atomistic simulations of the pressure-induced phase transitions in BaF2 crystals. J. Phys. Condens. Matter 2001, 13, 11741–11749. [Google Scholar] [CrossRef]
- Wojciechowska, M.; Zielinski, M.; Pietrowski, M. MgF2 as a non-conventional catalyst support. J. Fluor. Chem. 2003, 120, 1–11. [Google Scholar] [CrossRef]
- Kemnitz, E.; Rüdiger, S. High surface area metal fluorides as catalysts. In Functionalized Inorganic Fluorides; Tressaud, A., Ed.; Wiley: Hoboken, NJ, USA, 2010. [Google Scholar]
- Kemnitz, E.; Groß, U.; Rüdiger, S.; Shekar, C.S. Amorphous Metal Fluorides with Extraordinary High Surface Areas. Angew. Chem. Int. Ed. 2003, 42, 4251–4254. [Google Scholar] [CrossRef] [PubMed]
- Barth, J.; Johnson, R.L.; Cardona, M.; Fuchs, D.; Bradshaw, A.M. Dielectric function of CaF2 between 10 and 35 eV. Phys. Rev. B 1990, 41, 3291–3294. [Google Scholar] [CrossRef]
- Camy, P.; Doualan, J.L.; Renard, S.; Braud, A.; Menard, V.; Moncorge, R. Tm3+: CaF2 for 1.9 μm laser operation. Opt. Commun. 2004, 236, 395–402. [Google Scholar] [CrossRef]
- Kumar, G.A.; Riman, R.; Chae, S.C.; Yang, Y.N.; Bae, I.K.; Moon, H.S. Synthesis and spectroscopic characterization of CaF2:Er3+ single crystal for highly efficient 1.53 μm amplification. J. Appl. Phys. 2004, 95, 3243–3249. [Google Scholar] [CrossRef]
- Tsujibayashi, T.; Toyoda, K.; Sakuragi, S.; Kamada, M.; Itoh, M. Spectral profile of the two-photon absorption coefficients in CaF2 and BaF2. Appl. Phys. Lett. 2002, 80, 2883–2885. [Google Scholar] [CrossRef]
- Giessibl, F.; Reichling, M. Investigating atomic details of the CaF2(111) surface with a qPlus sensor. Nanotechnology 2005, 16, S118–S124. [Google Scholar] [CrossRef]
- Catti, M.; Dovesi, R.; Pavese, A.; Saunders, V.R. Elastic constants and electronic structure of fluorite (CaF2): An ab initio Hartree-Fock study. J. Phys. Condens. Matter 1991, 3, 4151–4164. [Google Scholar] [CrossRef]
- Shi, H.; Eglitis, R.I.; Borstel, G. First-principles calculations of the CaF2 bulk and surface electronic structure. Phys. Status Solidi (b) 2005, 242, 2041–2050. [Google Scholar] [CrossRef]
- Shi, H.; Eglitis, R.I.; Borstel, G. Ab initio calculations of the BaF2 bulk and surface F centres. J. Phys. Condens. Matter 2006, 18, 8367–8381. [Google Scholar] [CrossRef]
- De Leeuw, N.H.; Cooper, T.G. A computational study of the surface structure and reactivity of calcium fluoride. J. Mater. Chem. 2003, 13, 93–101. [Google Scholar] [CrossRef]
- Kanchana, V.; Vaitheeswaran, G.; Rajagopalan, M. Structural phase stability of CaF2 and SrF2 under pressure. Physica B 2003, 328, 283–290. [Google Scholar] [CrossRef]
- Kanchana, V.; Vaitheeswaran, G.; Rajagopalan, M. Pressure induced structural phase transitions and metallization of BaF2. J. Alloys Compd. 2003, 359, 66–72. [Google Scholar] [CrossRef]
- Weast, R.C. CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, NJ, USA, 1976. [Google Scholar]
- Nicolav, M. Shaped single crystals of CaF2. J. Cryst. Growth 2000, 218, 62–66. [Google Scholar] [CrossRef]
- Samara, G.A. Temperature and pressure dependences of the dielectric properties of PbF2 and the alkaline-earth fluorides. Phys. Rev. B 1976, 13, 4529–4544. [Google Scholar] [CrossRef]
- Wyckoff, R.W.G. Crystal Structures, 2nd ed.; Interscience Publishers: New York, NY, USA, 1982; Volume 1. [Google Scholar]
- Leger, J.M.; Haines, J.; Atouf, A.; Schuete, O.; Hull, S. High-pressure X-ray- and neutron-diffraction studies of BaF2: An example of a coordination number of 11 in AX2 compounds. Phys. Rev. B 1995, 52, 13247–13256. [Google Scholar] [CrossRef]
- Bennewitz, R.; Reichling, M.; Matthias, E. Force microscopy of cleaved and electron-irradiated CaF2(111) surfaces in ultra-high vacuum. Surf. Sci. 1997, 387, 69–77. [Google Scholar] [CrossRef]
- Wulff, G. Zur Frage der Geschwindigkeit des Wachsthums und der Auflösung der Krystallflächen. Z. Kristallogr. Mineral. 1901, 34, 449–530. [Google Scholar] [CrossRef]
- Gilman, J.J. Direct Measurements of the Surface Energies of Crystals. J. Appl. Phys. 1960, 31, 2208–2218. [Google Scholar] [CrossRef]
- Puchin, V.E.; Puchina, A.V.; Huisinga, M.; Reichling, M. Theoretical modelling of steps on the CaF2(111) surface. J. Phys. Condens. Matter 2001, 13, 2081–2094. [Google Scholar] [CrossRef]
- Tasker, P.W. The structure and properties of fluorite crystal surfaces. J. Physique Coll. 1980, 41, C6-488–C6-491. [Google Scholar] [CrossRef]
- Kraatz, P.; Zoltai, T. Effects of ionizing radiation on cleavage surface energy of SrF2. J. Appl. Phys. 1974, 45, 5093–5095. [Google Scholar] [CrossRef]
- Morris, E.; Groy, T.; Leinenweber, K. Crystal structure and bonding in the high-pressure form of fluorite (CaF2). J. Phys. Chem. Solids 2001, 62, 1117–1122. [Google Scholar] [CrossRef]
- Dovesi, R.; Orlando, R.; Civalleri, B.; Roetti, C.; Saunders, V.R.; Zicovich-Wilson, C.M. CRYSTAL: A computational tool for the ab initio study of the electronic properties of crystals. Z. Kristallogr. 2005, 220, 571–573. [Google Scholar] [CrossRef]
- Dovesi, R.; Saunders, V.; Roetti, C.; Orlando, R.; Zicovich-Wilson, C.M.; Pascale, F.; Civalleri, B.; Doll, K.; Harrison, N.; Bush, I.; et al. CRYSTAL09 User’s Manual; University of Torino: Torino, Italy, 2010. [Google Scholar]
- Dirac, P.A.M. Note on Exchange Phenomena in the Thomas Atom. Proc. Camb. Philos. Soc. 1930, 26, 376–385. [Google Scholar] [CrossRef]
- Slater, J.C. A simplification of the Hartree-Fock method. Phys. Rev. 1951, 81, 385–390. [Google Scholar] [CrossRef]
- Vosko, S.H.; Wilk, L.; Nusair, M. Accurate spin-dependent electron liquid correlation energies for local spin density calculations: a critical analysis. Can. J. Phys. 1980, 58, 1200–1211. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed]
- Perdew, J.P.; Wang, Y. Accurate and simple analytic representation of the electron-gas correlation energy. Phys. Rev. B Condens. Matter 1992, 45, 13244–13249. [Google Scholar] [CrossRef] [PubMed]
- Becke, A.D. Density functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B Condens. Matter 1988, 37, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Huesges, Z.; Müller, C.; Paulus, B.; Hough, C.; Harrison, N.; Kemnitz, E. Characterising MgF2 surfaces with CO adsorption calculations. Surf. Sci. 2013, 609, 73–77. [Google Scholar] [CrossRef]
- Belger, D.; Huesges, Z.; Voloshina, E.; Paulus, B. The role of electron correlations in the binding properties of Ca, Sr, and Ba. J. Phys. Condens. Matter 2010, 22, 275504–275510. [Google Scholar] [CrossRef] [PubMed]
- Kaupp, M.; Schleyer, P.v.R.; Stoll, H.; Preuss, H. Pseudopotential approaches to Ca, Sr, and Ba hydrides. Why are some alkaline earth MX2 compounds bent? J. Chem. Phys. 1991, 94, 1360–1366. [Google Scholar] [CrossRef]
- Bailey, C.L.; Mukhopadhyay, S.; Wander, A.; Searle, B.G.; Harrison, N.M. Structure and Stability of α-AlF3 Surfaces. J. Phys. Chem. C 2009, 113, 4976–4983. [Google Scholar] [CrossRef]
- Reuter, K.; Scheffler, M. Composition, structure, and stability of RuO2(110) as a function of oxygen pressure. Phys. Rev. B 2001, 65, 035406, 1–11. [Google Scholar] [CrossRef]
- NIST Standard Reference Database Number 69. Available online: http://webbook.nist.gov/chemistry (accessed on 10 December 2016).
- Cox, J.D.; Wagman, D.D.; Medvedev, V.A. CODATA Key Values for Thermodynamics; Hemisphere Publishing: New York, NY, USA, 1989. [Google Scholar]
- Geysermans, P.; Finocchi, F.; Goniakowski, J.; Hacquart, R.; Jupille, J. Combination of (100), (110) and (111) facets in MgO crystals shapes from dry to wet environment. Phys. Chem. Chem. Phys. 2008, 11, 2228–2233. [Google Scholar] [CrossRef] [PubMed]
- Kanaki, E.; Gohr, S.; Müller, C.; Paulus, B. Theoretical study on the morphology of MgF2 nanocrystals at finite temperature and pressure. Surf. Sci. 2015, 632, 158–163. [Google Scholar] [CrossRef]
Sample Availability: Not available. |




| CaF | SrF | BaF | ||||
|---|---|---|---|---|---|---|
| Calc. | Exp. | calc. | Exp. | Calc. | Exp. | |
| a (Å) | 5.50 | 5.46 | 5.84 | 5.8 | 6.24 | 6.20 |
| B (GPa) | 82 | 83 | 62 | 69 | 62 | 57 |
| E (eV) | −16.88 | −16.08 | −17.01 | −15.95 | −17.90 | −16.01 |
| E (eV) | −26.89 | −27.46 | −25.35 | −26.03 | −23.90 | −24.58 |
| Surface | Surface Energy in (J/m) | ||
|---|---|---|---|
| CaF | SrF | BaF | |
| 111 | 0.47 | 0.45 | 0.39 |
| 110 | 0.71 | 0.67 | 0.57 |
| 100 | 0.95 | 0.98 | 0.86 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaawar, Z.; Mahn, S.; Kemnitz, E.; Paulus, B. On the Morphology of Group II Metal Fluoride Nanocrystals at Finite Temperature and Partial Pressure of HF. Molecules 2017, 22, 663. https://doi.org/10.3390/molecules22040663
Kaawar Z, Mahn S, Kemnitz E, Paulus B. On the Morphology of Group II Metal Fluoride Nanocrystals at Finite Temperature and Partial Pressure of HF. Molecules. 2017; 22(4):663. https://doi.org/10.3390/molecules22040663
Chicago/Turabian StyleKaawar, Zeinab, Stefan Mahn, Erhard Kemnitz, and Beate Paulus. 2017. "On the Morphology of Group II Metal Fluoride Nanocrystals at Finite Temperature and Partial Pressure of HF" Molecules 22, no. 4: 663. https://doi.org/10.3390/molecules22040663
APA StyleKaawar, Z., Mahn, S., Kemnitz, E., & Paulus, B. (2017). On the Morphology of Group II Metal Fluoride Nanocrystals at Finite Temperature and Partial Pressure of HF. Molecules, 22(4), 663. https://doi.org/10.3390/molecules22040663






