Abstract
Strategies for the control of sulfate-reducing bacteria (SRB) in the oil industry involve the use of high concentrations of biocides, but these may induce bacterial resistance and/or be harmful to public health and the environment. Essential oils (EO) produced by plants inhibit the growth of different microorganisms and are a possible alternative for controlling SRB. We aimed to characterize the bacterial community of produced water obtained from a Brazilian petroleum facility using molecular methods, as well as to evaluate the antimicrobial activity of EO from different plants and their major components against Desulfovibrio alaskensis NCIMB 13491 and against SRB growth directly in the produced water. Denaturing gradient gel electrophoresis revealed the presence of the genera Pelobacter and Marinobacterium, Geotoga petraea, and the SRB Desulfoplanes formicivorans in our produced water samples. Sequencing of dsrA insert-containing clones confirmed the presence of sequences related to D. formicivorans. EO obtained from Citrus aurantifolia, Lippia alba LA44 and Cymbopogon citratus, as well as citral, linalool, eugenol and geraniol, greatly inhibited (minimum inhibitory concentration (MIC) = 78 µg/mL) the growth of D. alaskensis in a liquid medium. The same MIC was obtained directly in the produced water with EO from L. alba LA44 (containing 82% citral) and with pure citral. These findings may help to control detrimental bacteria in the oil industry.
1. Introduction
Oil exploration and production generates a large amount of produced water (defined as the water that exists in subsurface formations and is brought to the surface during oil and gas production) [1]. Typically, a new field yields about 5 to 15% produced water of the total volume of petroleum. However, as the field matures, the produced water volume can reach up to 90% of production because additional water is usually injected into the reservoir to sustain the pressure necessary to maintain or increase oil recovery levels [2].
At the surface, produced water is separated from hydrocarbons, but small suspended oil particles and dissolved organic and inorganic compounds (mainly chloride and sulfide ions) still remain, making the discharge of produced water an environmental problem [3,4]. Different strategies have been developed to reuse and recycle produced water, including re-injecting it back into reservoirs to increase oil production and many other industrial uses [1,5,6,7].
Water-injection systems are frequently contaminated by bacteria that can cause severe plugging of surface and downhole equipment and injection-well formations, and also generate H2S that can indirectly cause pitting corrosion [8,9]. Biogenic sulfide production (biogenic souring) results from sulfate-reducing bacteria (SRB, [10]) or other sulfidogenic bacterial activity. In addition to the corrosion of metal surfaces, H2S is both toxic and explosive [11,12]. To counter these effects, bacterial growth in water-injection systems is controlled mainly by chemical biocides, such as chloride, glutaraldehyde and quaternary ammonium salts [13,14]. Several other water-based fluids used in oil and natural gas drilling and production operations benefit from the use of biocides, as well as the water holding tanks. However, biocides may fail because of difficulties in penetrating bacterial biofilms and also due to bacterial biocide-resistance [15]. Moreover, residual concentrations, toxicity and persistence of biocides in industrial effluents are known to be detrimental to public health and the environment [14]. Hence, alternative biocides against harmful bacteria, with a particular focus on SRB control, are of great interest to the petroleum industry [14,16,17].
Several essential oils (EO)—complex mixtures of volatile, lipophilic and odiferous substances arising from the secondary metabolism of plants—have been used as therapeutic agents since ancient times, and some of them have been scientifically proven to possess medicinal properties [18,19,20] and antimicrobial activities [21]. They are mainly composed of monoterpenes and sesquiterpenes, and their oxygenated derivatives (alcohols, aldehydes, esters, ketones, phenols and oxides). Korenblum et al. [22] previously demonstrated that lemongrass (Cymbopogon citratus (DC.) Stapf) essential oil (LEO) or citral (the principal compound of LEO) is able to control planktonic growth of Desulfovibrio alaskensis strain NCIMB 13491 and/or prevent its biofilm formation and sulfide-induced corrosion of metal surfaces. However, though a wide assortment of EO have already been described in the literature, their antimicrobial activity against SRB growth is understudied.
In this study, we first characterized (using molecular methods) the bacterial community present in produced water samples from a Brazilian petroleum industrial facility. We then tested different essential oils and some of their major components against D. alaskensis (to assess control of SRB growth) and also directly, to elucidate their antimicrobial properties on our produced water samples.
2. Results
2.1. Bacterial Community in the Produced Water Samples
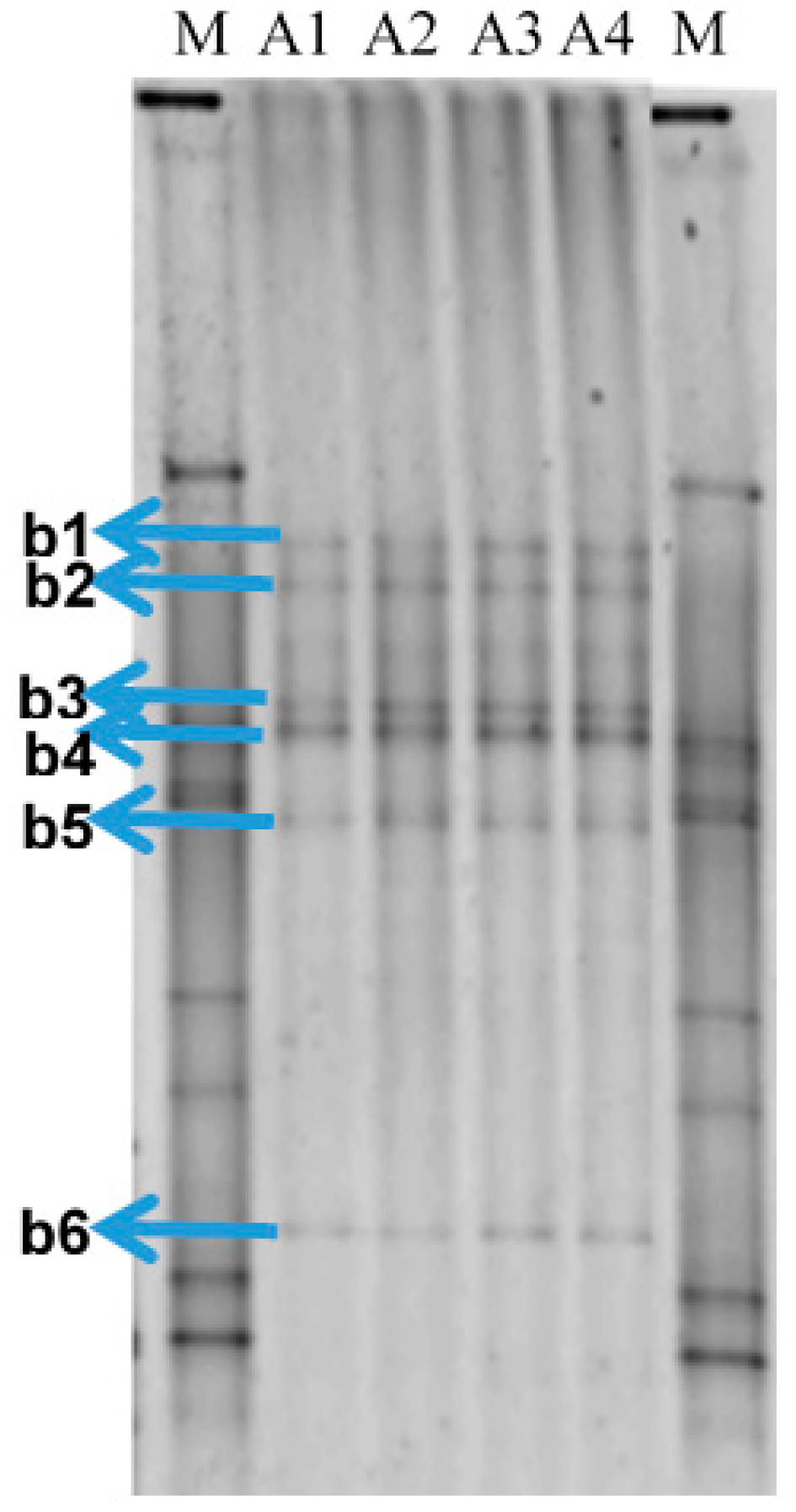
DNA from the four samples of produced water received from Petrobras Ilha Grande Bay Oil Terminal (TEBIG) was analyzed by PCR followed by denaturing gradient gel electrophoresis (PCR-DGGE) of 16S rRNA-encoding gene fragments. Fingerprint analysis showed that the bacterial community structure was very similar between replicates (Figure 1). Unweighted pair group method with arithmetic mean (UPGMA) cluster analysis revealed that the profiles were clustered with a similarity >92% (Figure S1). Six dominant and common bands for the four profiles were extracted from the gel, cloned, sequenced and further identified (see Table 1). Nucleotide sequences have been deposited in GenBank under accession numbers: KY859414–KY859419. Two bands corresponded to the genus Pelobacter (belonging to the Deltaproteobacteria class, Desulfuromonadales order), one to Geotoga (Thermotogae class, Petrotogales order), and another two bands to the genus Marinobacterium (Gammaproteobacteria class, Alteromonadales order). Only one band corresponded to an SRB, which was identified as Desulfoplanes formicivorans (Deltaproteobacteria class, Desulfovibrionales order).
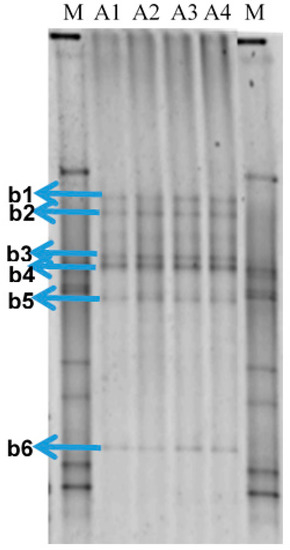
Figure 1.
Denaturing gradient gel electrophoresis (DGGE) fingerprints to compare the bacterial communities of the four produced water samples received from Petrobras Ilha Grande Bay Oil Terminal (TEBIG). The letters A and b followed by a number correspond to the water samples and to the band retrieved from the gel that was subsequently reamplified and sequenced, respectively. The letter M indicates the bacterial standard marker.

Table 1.
Identification (using the Basic Local Alignment Search Tool—BLAST-N) of partial rrs gene sequences of the different bands extracted from the DGGE gel.
To further establish the diversity of the SRB population in the produced water samples, a portion of the dsrA gene coding for dissimilatory sulfite reductase was used to construct a clone library. All of the resulting sequences from the produced water sample (from 20 clones) were highly similar and associated with D. formicivorans, indicating that this sulfate-reducing bacterial species predominates in the produced water studied here. A representative dsrA nucleotide sequence has been deposited in GenBank under the accession number KY867755. Total SRB enumeration in the produced water sample was equivalent to 1.5 × 105 MPN/mL.
2.2. Essential Oils and Major Components Against SRB
Table 2 shows the different plants from which EO were distilled and their major components. All of these EO were tested against SRB strain D. alaskensis NCIMB 13491 and also against produced water samples. Minimum inhibitory concentrations (MICs) determined using the crude EO against D. alaskensis varied from 78 to 2500 µg/mL (Table 2). The best results (MIC = 78 or 156 µg/mL) were observed for the EO extracted from “mirim” lime, “siciliano” lemon, lemon balm (Lippia alba LA10, LA22, LA44) and lemongrass (Cymbopogon citratus).

Table 2.
Determination of the minimum inhibitory concentration (MIC) of essential oils (EO) against Desulfovibrio alaskensis NCIMB 13491 and against sulfate-reducing bacteria (SRB) growth in produced water samples.
We also tested commercially-available major components of EO and found the lowest MIC values were observed for citral, linalool, geraniol and eugenol (Table 3). We selected those EO and their components exhibiting low MIC values to assess inhibition of SRB growth in produced water samples. We detected an increase in the MIC values (over our tests against D. alaskensis) for all EO or major components tested, except for L. alba LA22, LA44 and citral (Table 2 and Table 3).

Table 3.
Determination of the minimum inhibitory concentration (MIC) of commercially-available major components of EO against Desulfovibrio alaskensis NCIMB 13491 and against SRB growth in produced water samples.
3. Discussion
Various plant EO have been extensively studied because of their antimicrobial activity against a range of medically-relevant bacteria [21]. Furthermore, other biological activities have been attributed to different EO, such as anti-inflammatory, anti-diabetic, anti-mutagenic, insecticidal, molluscicidal and protozoicidal effects, among others [27]. Kerekes et al. [28] proposed the use of EO as natural preservatives and sanitizers in the food industry, and Borrego et al. [29] suggested their use in minimizing biodeterioration of documents. However, their potential for applications against detrimental bacteria in the petroleum industry is much less studied. Korenblum et al. [22] only evaluated the anti-corrosive effect and antimicrobial activity of one EO (lemongrass) against the growth of D. alaskensis. To our knowledge, the effects of other EO against SRB and their possible usage directly in produced water generated by the petroleum industry have never before been tested.
Our results demonstrate the potential to use different EO obtained from “mirim” lime, “siciliano” lemon, as well as different varieties of lemon balm and lemongrass, in the petroleum industry to control bacterial growth in produced water. The low MIC values (78–156 µg/mL) suggest that only a small amount of each EO is necessary to inhibit the growth of SRB, at least for the SRB model strain D. alaskensis NCIMB 13491 that was previously isolated from a soured oil reservoir [30]. MIC values for lemongrass essential oil (LEO) and citral of 170 µg/mL for the inhibition of SRB growth have been previously reported [22], and a comparable MIC value for LEO against other bacteria was presented by Adukwu et al. [31].
When we focused our MIC assays on the produced water samples from TEBIG, in general we found that higher concentrations of EO were necessary to inhibit SRB growth in produced water compared to D. alaskensis cultures, with the MIC value at least doubling for the majority of EO tested. This effect may be explained by the bacterial community present in the produced water samples, which may be interfering with the EO activity. Our molecular analyses revealed the presence in produced water samples of a mixed culture of four predominant genera, including the SRB D. formicivorans. Geotoga spp. are fermentative bacteria capable of reducing elemental sulfur to hydrogen sulfide [32], and Pelobacter spp. are commonly found in marine sediments [33] and oil reservoirs [34,35]. The presence of these two genera has already been demonstrated in anaerobic highly corrosive biofilms recovered from the inside of a steel pipe of an offshore oil production facility [36]. Sequences related to Marinobacterium have been found in the Xinjiang Luliang water-flooding petroleum reservoir [37]. D. formicivorans was recently isolated from the sediment of meromictic Lake Harutori in Japan and described as a novel species of a new genus belonging to the family Desulfomicrobiaceae [38]. Therefore, our results broaden the SRB targets that can be subjected to EO growth inhibition.
We also considered the major components of the different EO using commercially-available products, and our findings indicate that the presence of high concentrations of linalool, citral and a mixture of limonene and citral seemed to be responsible for the effectiveness of the various EO. The results we obtained for citral corroborate those obtained by Korenblum et al. [22]. Citral, linalool, geraniol and eugenol all presented low MIC values of 78 µg/mL when tested against D. alaskensis. As geraniol is a linalool isomer, the same response of D. alaskensis to both components was expected. Moreover, as eugenol (which is used as a major ingredient in a variety of dental materials) presents antibacterial activity mainly against anaerobic bacteria [39], SRB were also likely to be inhibited by it. However, only citral demonstrated equivalent effectiveness (i.e., low MIC) against bacterial growth in both pure SRB cultures and production water samples.
Our findings suggest that use of different EO and their major constituents may be an option for replacing or at least decreasing the application of synthetic biocides to control bacterial growth in the petroleum industry. We propose that the antimicrobial activities we observed directly in produced water samples will be the basis for further investigations of the use of EO as an option for controlling SRB growth in petroleum industry facilities.
4. Materials and Methods
4.1. Produced Water
Four receptacles containing 5 L of produced water were brought from TEBIG (coordinates: 23°3′23″ S, 44°14′6″ W), off the coast of Rio de Janeiro, Brazil. The water samples were maintained at 4 °C until use. The physicochemical properties of the produced water samples were: pH of 7.6, salinity of 23.2 g/L, Chloride 15.1 g/L, Calcium 0.51 g/L, Iron 1.2 mg/L, Potassium 0.16 g/L, Strontium 5.4 mg/L, Sulfide < 0.3 mg/L, Magnesium 0.45 g/L, Sodium 9.0 g/L, Barium 8.4 mg/L.
4.2. Sulfate-Reducing Bacteria
The sulfate-reducing bacteria (SRB) Desulfovibrio alaskensis NCIMB 13491 [30] was grown at 30 °C for 3–7 days in either sealed serum bottles or BD Vacutainer serum tubes containing, respectively, 10 mL and 1 mL of Postgate C or Postgate E media [40]. The bottles were purged with a N2 flux to achieve anaerobic conditions.
4.3. EO and Major Components
Fruits of different species of Citrus were collected in local markets in Rio de Janeiro, Brazil. Fruit peels (150 g) were removed and homogenized in distilled water using a laboratory blender. The resulting mixture was immediately subjected to hydrodistillation using a glass-type Clevenger apparatus as described in Simas et al. [23]. Lemongrass (Cymbopogon citratus (DC.) Stapf, Poaceae) and lemon balm (Lippia alba (Mill) N. E. Brown, Verbenaceae) leaves were collected from the Research Farm of the Federal University of Sergipe, Brazil. One sample of L. sidoides (LSID104) was collected at Poço Redondo, Sergipe, Brazil, and the remaining L. sidoides samples were harvested from the Research Farm of the Federal University of Sergipe, Brazil. Leaves from Croton cajucara Benth (Euphorbiaceae) were obtained from the Germoplasm collection of “EMBRAPA Amazônia Ocidental,” Amazonas, Brazil. EO of lemongrass, white and red “sacaca,” lemon balm and pepper-rosmarin were obtained from the fresh leaves by hydrodistillation [22,24,25,26].
All pure compounds of the major components of the EO used in bioassays were obtained from Sigma-Aldrich (São Paulo, Brazil).
4.4. DNA Extraction
A 50 mL sample from each receptacle containing produced water was filtered through a Millipore membrane (0.45 µm), before the total DNA was extracted using a FastDNA® Spin Kit for Soil (MP Biomedicals, Santa Ana, CA, USA) and then stored at 4 °C prior to PCR amplification.
4.5. PCR Amplification of Bacterial rrs and dsrA Genes
PCR amplification of 16S rRNA-encoding genes was performed using the bacterial primer set U968f and L1401r [41] in a 25 μL-mixture containing about 10 ng of DNA, 0.8 µM of each primer, 0.5 µM of each dNTP, 2.5 mM MgCl2, 5 U Taq DNA polymerase (GOTaq®Flex–Promega, Madison, WI, USA), and 5 µL of the 5X PCR buffer supplied by the manufacturer. A GC-clamp was added to the forward primer [42]. The amplification conditions were as follows: 1× (3 min at 94 °C), 35× (1 min at 94 °C, 1.5 min at 55 °C, and 1 min at 72 °C), and an extension for 10 min at 72 °C.
A portion of the dsrA gene coding for dissimilatory sulfite reductase was amplified using the primers DSR-1F [43] and DSR-R [44]. The 25 µL-reaction mixture contained about 10 ng of DNA, 100 nM of each primer, 0.2 mM of each dNTP, 1.25 U Taq DNA polymerase, 5X PCR buffer (Promega), and sterile Milli-Q water. The amplification conditions were as follows: 1× (15 s at 94 °C), 30× (15 s at 94 °C, 20 s at 54 °C, and 54 s at 72 °C), and an extension for 1 min at 72 °C. Positive (D. alaskensis strain NCIMB 13491) and negative controls (without DNA) were run in all PCR amplifications.
The products were analyzed by electrophoresis in 1.4% agarose gels, followed by ethidium bromide staining (1.2 mg/L ethidium bromide in 1X TAE buffer-20 mM Tris-acetate, pH 7.4, 10 mM acetate, 0.5 mM disodium EDTA).
4.6. Denaturing Gradient Gel Electrophoresis (DGGE) and Statistical Analyses
DGGE analysis was carried out as described previously [42] using an Ingeny PhorU2 apparatus (Ingeny International BV, Goes, The Netherlands). PCR products were loaded onto 8% (w/v) polyacrylamide gels in 1X TAE buffer. Polyacrylamide gels contained a denaturing gradient of urea and formamide varying from 46.5% to 60%. The gels were run for 17 h at 140 V and 65 °C. After this period, they were soak-stained for 1 h in SYBR Green I nucleic acid gel stain solution (10,000× g concentrated; Life Technologies, Eugene, OR, USA) and immediately photographed under UV light. Dendrograms were constructed based on the presence and absence of bands with the unweighted pair group method with mathematical averages (UPGMA) and the Pearson similarity coefficient using GelCompare II software (Applied Maths, Sint-Martens-Latem, Belgium).
4.7. Sequencing of DGGE Bands and dsrA Clone Libraries
Some bands were retrieved from the gels (marked in Figure 1) and 16S rRNA-encoding genes were reamplified as described above. These PCR products and those of dsrA PCR amplification were purified using Wizard SV Gel and PCR Clean-up System (Promega), and ligated to the pTZ57R/T plasmid vector using InsTAclone PCR Cloning Kit (Fermentas, Hanover, MD, USA), following the supplier’s instructions. The ligation products were transformed into competent Escherichia coli DH5-α cells. Sequencing of the inserts was performed by Macrogen (Seoul, Korea) using the primers U968F (16S rRNA) or DSR-1F (dsrA).
The partial rrs gene sequences were identified using the BLAST-N facility (www.ncbi.nlm.nih.gov/blast) of the National Center for Biotechnology Information (NCBI) with the GenBank non-redundant database.
4.8. Enumeration of Sulfate-Reducing Bacteria
SRB enumeration in pure cultures and in the produced water samples was performed using the most probable number (MPN) method, as described by Postgate [40] and the MPN Reference Table [45]. The four water samples were combined to perform the MPN analysis.
4.9. Determination of Minimum Inhibitory Concentration (MIC) of EO and Major Components
Macrodilution susceptibility tests were performed in Postgate E broth (1990 µL) in BD Vacutainer tubes, with the different EO or their major components (10 µL) being added to the first dilution tube. The tube contents were serially diluted twice to a lowest concentration of about 19.5 µg/mL of EO or their components to determine the minimum inhibitory concentrations. D. alaskensis was grown for 3–7 days at 32 °C in Postgate C medium to yield a final SRB inoculum of 105 cells/mL. A volume of 100 µL of the SRB culture (or the production water) was introduced into the various Vacutainer tubes. They were incubated for 7 days at 32 °C. SRB growth was detected by observing accumulation of the blackish color of the medium caused by iron sulfide precipitation in Postgate E medium. The minimum inhibitory concentration (MIC) was determined as the least amount of EO or major component added that did not result in a blackish color in the medium.
Supplementary Materials
Supplementary Materials are available.
Acknowledgments
This study was supported by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ).
Author Contributions
L.S., J.M.M., D.S.A.M., C.S.A. conceived and designed the experiments; P.M.d.S., J.M.M., F.R.d.V.G., V.V. performed the experiments; P.M.d.S., L.S., J.M.M., D.S.A.M., C.S.A. analyzed the data; H.R.B., A.F.B., C.G., M.P.d.S. contributed with the different EO, major components and produced water; L.S. wrote the paper and all authors revised, gave suggestions and approved the manuscript in the present form.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Fakhru’l-Razi, A.; Pendashteh, A.; Abdullah, L.C.; Biak, D.R.A.; Madaeni, S.S.; Abidin, Z.Z. Review of technologies for oil and gas produced water treatment. J. Hazard. Mater. 2009, 170, 530–551. [Google Scholar] [PubMed]
- Muggeridge, A.; Cockin, A.; Webb, K.; Frampton, H.; Collins, I.; Moulds, T.; Salino, P. Recovery rates, enhanced oil recovery and technological limits. Philos. Trans. R. Soc. A 2014, 372, 20120320. [Google Scholar]
- Fontenot, B.E.; Hunt, L.R.; Hildenbrand, Z.L.; Carlton, D.D.; Oka, H.; Walton, J.L.; Hopkins, D.; Osorio, A.; Bjorndal, B.; et al. An evaluation of water quality in private drinking water wells near natural gas extraction sites in the Barnett shale formation. Environ. Sci. Technol. 2013, 47, 10032–10040. [Google Scholar] [CrossRef] [PubMed]
- Harkness, J.S.; Dwyer, G.S.; Warner, N.R.; Parker, K.M.; Mitch, W.A.; Vengosh, A. Iodide, bromide, and ammonium in hydraulic fracturing and oil and gas wastewaters: Environmental implications. Environ. Sci. Technol. 2015, 49, 1955–1963. [Google Scholar] [CrossRef] [PubMed]
- Veil, J.; Puder, M.G.; Elcock, D.; Redweik, R.J.J. A White Paper Describing Produced Water from Production of Crude Oil, Natural Gas and Coal Bed Methane. Available online: http://www.netl.doe.gov/publications/oil pubs/prodwaterpaper.pdf (accessed on 26 January 2017).
- Arthur, J.D.; Langhus, B.G.; Patel, C. Technical Summary of Oil and Gas Produced Water Treatment Technologies. Available online: http://www.rrc.state.tx.us/ commissioners/williams/environment/produced water treatment Tech.pdf (accessed on 26 January 2017).
- Igunnu, E.T.; Chen, G.Z. Produced water treatment technologies. Int. J. Low-Carbon Technol. 2014, 9, 157–177. [Google Scholar]
- Singer, M.E. Microbial biosurfactants. In Microbes and Oil Recovery; Zajic, J.E., Donaldson, E.C., Eds.; Bioresources Publications: El Paso, TX, USA, 1985; pp. 19–38. [Google Scholar]
- Popoola, L.T.; Grema, A.S.; Latinwo, G.K.; Gutti, B.; Balogun, A.S. Corrosion problems during oil and gas production and its mitigation. Int. J. Ind. Chem. 2013, 4, 35. [Google Scholar]
- Barton, L.L.; Fauque, G.D. Biochemistry, physiology and biotechnology of sulfate-reducing bacteria. Adv. Appl. Microbiol. 2009, 68, 41–98. [Google Scholar] [PubMed]
- Gevertz, D.; Telang, A.J.; Voordouw, G.; Jenneman, G.E. Isolation and characterization of strains CVO and FWKO B, two novel nitrate-reducing, sulfide-oxidizing bacteria isolated from oil field brine. Appl. Environ. Microbiol. 2000, 66, 2491–2501. [Google Scholar] [CrossRef] [PubMed]
- Hubert, C.; Nemati, M.; Jenneman, G.; Voordouw, G. Corrosion risk associated with microbial souring control using nitrate or nitrite. Appl. Microbiol. Biotechnol. 2005, 68, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Videla, H.A. Prevention and control of biocorrosion. Int. Biodeterior. Biodegrad. 2002, 49, 259–270. [Google Scholar]
- Pichtel, J. Oil and gas production wastewater: Soil contamination and pollution prevention. Appl. Environ. Soil Sci. 2016, 2016, 24. [Google Scholar] [CrossRef]
- Stewart, P.S.; Costerton, J.W. Antibiotic resistance of bacteria in biofilms. Lancet 2001, 358, 135–138. [Google Scholar] [CrossRef]
- Nemati, M.; Jenneman, G.E.; Voordouw, G. Mechanistic study of microbial control of hydrogen sulfide production in oil reservoirs. Biotechnol. Bioeng. 2001, 74, 424–434. [Google Scholar] [CrossRef] [PubMed]
- Stewart, P. Mechanisms of antibiotic resistance in bacterial biofilms. Int. J. Med. Microbiol. 2002, 292, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Koudou, J.; Abena, A.A.; Ngaissona, P.; Bessière, J.M. Chemical composition and pharmacological activity of essential oil of Canarium schweinfurthii. Fitoterapia 2005, 76, 700–703. [Google Scholar] [CrossRef] [PubMed]
- M’Barek, L.A.; Mouse, H.A.; Jaâfari, A.; Aboufatima, R.; Benharref, A.; Kamal, M.; Bénard, J.; El Abbadi, N.; Bensalah, M.; et al. Cytotoxic effect of essential oil of thyme (Thymus broussonettii) on the IGR-OV1 tumor cells resistant to chemotherapy. Braz. J. Med. Biol. Res. 2007, 40, 1537–1544. [Google Scholar]
- Loizzoa, M.R.; Saabb, A.; Tundisa, R.; Stattia, G.A.; Lamprontic, I.; Menichinia, F.; Gambarid, R.; Cinatle, J.; Doer, H.W. Phytochemical analysis and in vitro evaluation of the biological activity against herpes simplex virus type 1 (HSV-1) of Cedrus libani A. Rich. Phytomedicine 2008, 15, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Alviano, D.S.; Alviano, C.S. Plant extracts: Search for new alternatives to treat microbial diseases. Curr. Pharm. Biotechnol. 2009, 10, 106–121. [Google Scholar] [CrossRef] [PubMed]
- Korenblum, E.; Goulart, F.R.V.; Rodrigues, I.A.; Abreu, F.; Lins, U.; Alves, P.B.; Blank, A.F.; Valoni, E.; Sebastián, G.V.; et al. Antimicrobial action and anti-corrosion effect against sulfate reducing bacteria by lemongrass (Cymbopogon citratus) essential oil and its major component, the citral. AMB Express 2013, 3, 44. [Google Scholar] [CrossRef] [PubMed]
- Simas, D.L.R.; de Amorim, S.H.B.M.; Goulart, F.R.V.; Alviano, C.S.; Alviano, D.S.; da Silva, A.J.R. Citrus species essential oils and their components can inhibit or stimulate fungal growth in fruit. Ind. Crops Prod. 2017, 98, 108–115. [Google Scholar] [CrossRef]
- Chaves, F.M.C.; Bizzo, H.R.; Angelo, P.C.S.; Xavier, J.J.B.N.; Sá Sobrinho, A.F. Yield and chemical composition of essential oil from leaves of two varieties of sacaca (Croton cajucara Benth). Rev. Bras. Plant Med. Botucatu 2006, 8, 117–119. [Google Scholar]
- Blank, A.F.; Camêlo, L.C.A.; Arrigoni-Blank, M.F.; Pinheiro, J.B.; Andrade, T.M.; Niculau, E.S.; Alves, P.B. Chemical diversity in Lippia alba (Mill.) N.E. brown germplasm. Sci. World J. 2015, 2015, 11. [Google Scholar] [CrossRef] [PubMed]
- Farias-Junior, P.A.; Rios, M.C.; Moura, T.A.; Almeida, R.P.; Alves, P.B.; Blank, A.F.; Fernandes, R.P.M.; Scher, R. Leishmanicidal activity of carvacrol-rich essential oil from Lippia sidoides Cham. Biol. Res. 2012, 45, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Aleksic, V.; Knezevic, P. Antimicrobial and antioxidative activity of extracts and essential oils of Myrtus communis L. Microbiol. Res. 2014, 169, 240–254. [Google Scholar] [CrossRef] [PubMed]
- Kerekes, E.-B.; Deák, É.; Takó, M.; Tserennadmid, R.; Petkovits, T.; Vágvölgyi, C.; Krisch, J. Anti-biofilm forming and anti-quorum sensing activity of selected essential oils and their main components on food-related micro-organisms. J. Appl. Microbiol. 2013, 115, 933–942. [Google Scholar] [CrossRef] [PubMed]
- Borrego, B.; Valdés, O.; Vivar, I.; Lavin, P.; Guiamet, P.; Battistoni, P.; De Saravia, S.G.; Borges, P. Essential oils of plants as biocides against microorganisms isolated from Cuban and Argentine documentary heritage. ISRN Microbiol. 2012, 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Feio, M.J.; Zinkevich, V.; Beech, I.B.; Llobet-Brossa, E.; Eaton, P.; Schmitt, J.; Guezennec, J. Desulfovibrio alaskensis sp. nov., a sulfate-reducing bacterium from a soured oil reservoir. Int. J. Syst. Evol. Microbiol. 2004, 54, 1747–1752. [Google Scholar] [CrossRef] [PubMed]
- Adukwu, E.C.; Allen, S.C.; Phillips, C.A. The anti-biofilm activity of lemongrass (Cymbopogon flexuosus) and grapefruit (Citrus paradisi) essential oils against five strains of Staphylococcus aureus. J. Appl. Microbiol. 2012, 113, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Davey, M.E.; Wood, W.A.; Key, R.; Nakamura, K.; Stahl, D.A. Isolation of three species of Geotoga and Petrotoga: Two new genera, representing a new lineage in the bacterial line of descent distantly related to the “Thermotogales”. Syst. Appl. Microbiol. 1993, 16, 191–200. [Google Scholar] [CrossRef]
- Bowman, J.P.; Rea, S.M.; McCammon, S.A.; McMeekin, T.A. Diversity and community structure within anoxic sediment from marine salinity meromictic lakes and a coastal meromictic marine basin, Vestfold Hills, Eastern Antarctica. Environ. Microbiol. 2000, 2, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Duncan, K.E.; Gieg, L.M.; Parisi, V.A.; Tanner, R.S.; Tringe, S.G.; Bristow, J.; Suflita, J.M. Biocorrosive thermophilic microbial communities in Alaskan North slope oil facilities. Environ. Sci. Technol. 2009, 43, 7977–7984. [Google Scholar] [CrossRef] [PubMed]
- Hubert, C.R.J.; Oldenburg, T.B.P.; Fustic, M.; Gray, N.D.; Larter, S.R.; Penn, K.; Rowan, A.K.; Seshadri, R.; Sherry, A.; et al. Massive dominance of Epsilonproteobacteria in formation waters from a Canadian oil sands reservoir containing severely biodegraded oil. Environ. Microbiol. 2012, 14, 387–404. [Google Scholar] [CrossRef] [PubMed]
- Vigneron, A.; Alsop, E.B.; Chambers, B.; Lomans, B.P.; Head, I.M.; Tsesmetzis, N. Complementary microorganisms in highly corrosive biofilms from an offshore oil production facility. Appl. Environ. Microbiol. 2016, 82, 2545–2554. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Tian, H.; Li, G.; Sun, H.; Ma, T. Microbial diversity and abundance in the Xinjiang Luliang long-term water-flooding petroleum reservoir. Microbiologyopen 2015, 4, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Kojima, H.; Fukui, M. Desulfoplanes formicivorans gen. nov., sp. nov., a novel sulfate-reducing bacterium isolated from a blackish meromictic lake, and emended description of the family Desulfomicrobiaceae. Int. J. Syst. Evol. Microbiol. 2015, 65, 1902–1907. [Google Scholar] [CrossRef] [PubMed]
- Newman, M.G.; Hulem, C.; Colgate, J.; Anselmo, C. Antibacterial susceptibility of plaque bacteria. J. Dent. Res. 1979, 58, 1722–1732. [Google Scholar] [CrossRef] [PubMed]
- Postgate, J.R. The Sulphate-Reducing Bacteria, 2nd ed.; Cambridge University Press: Cambridge, UK, 1984; pp. 9–55. [Google Scholar]
- Heuer, H.; Krsek, M.; Baker, P.; Smalla, K.; Wellington, E.M.H. Analysis of actinomycete communities by specific amplification of genes encoding 16S rRNA and gel-electrophoretic separation in denaturing gradients. Appl. Environ. Microbiol. 1997, 63, 3233–3241. [Google Scholar] [PubMed]
- Muyzer, G.; de Waal, E.C.; Uitterlinden, A.G. Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl. Environ. Microbiol. 1993, 59, 695–700. [Google Scholar] [PubMed]
- Wagner, M.; Roger, A.J.; Flax, J.L.; Brusseau, G.A.; Stahl, D.A. Phylogeny of dissimilatory sulfite reductases supports an early origin of sulfate respiration. J. Bacteriol. 1998, 180, 2975–2982. [Google Scholar] [PubMed]
- Kondo, R.; Nedwell, D.B.; Purdy, K.J.; Silva, S.D. Detection and enumeration of sulphate reducing bacteria in estuarine sediments by competitive PCR. Geomicrobiol. J. 2004, 21, 145–157. [Google Scholar] [CrossRef]
- Taylor, J. The estimation of members of bacteria by tenfold dilution series. J. Appl. Microbiol. 1962, 25, 54–61. [Google Scholar]
Sample Availability: Not Available. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).