LC-ESI-MS/MS Identification of Biologically Active Phenolic Compounds in Mistletoe Berry Extracts from Different Host Trees
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Plant Material
3.2. Extraction Method
3.3. Total Phenolic and Flavonoid Content
3.4. LC-ESI-MS/MS Analysis
3.4.1. Chromatographic Conditions and Apparatus
3.4.2. Optimisation of Quantitative Analysis Parameters
3.5. Biological Activity
3.5.1. Antioxidant Activity
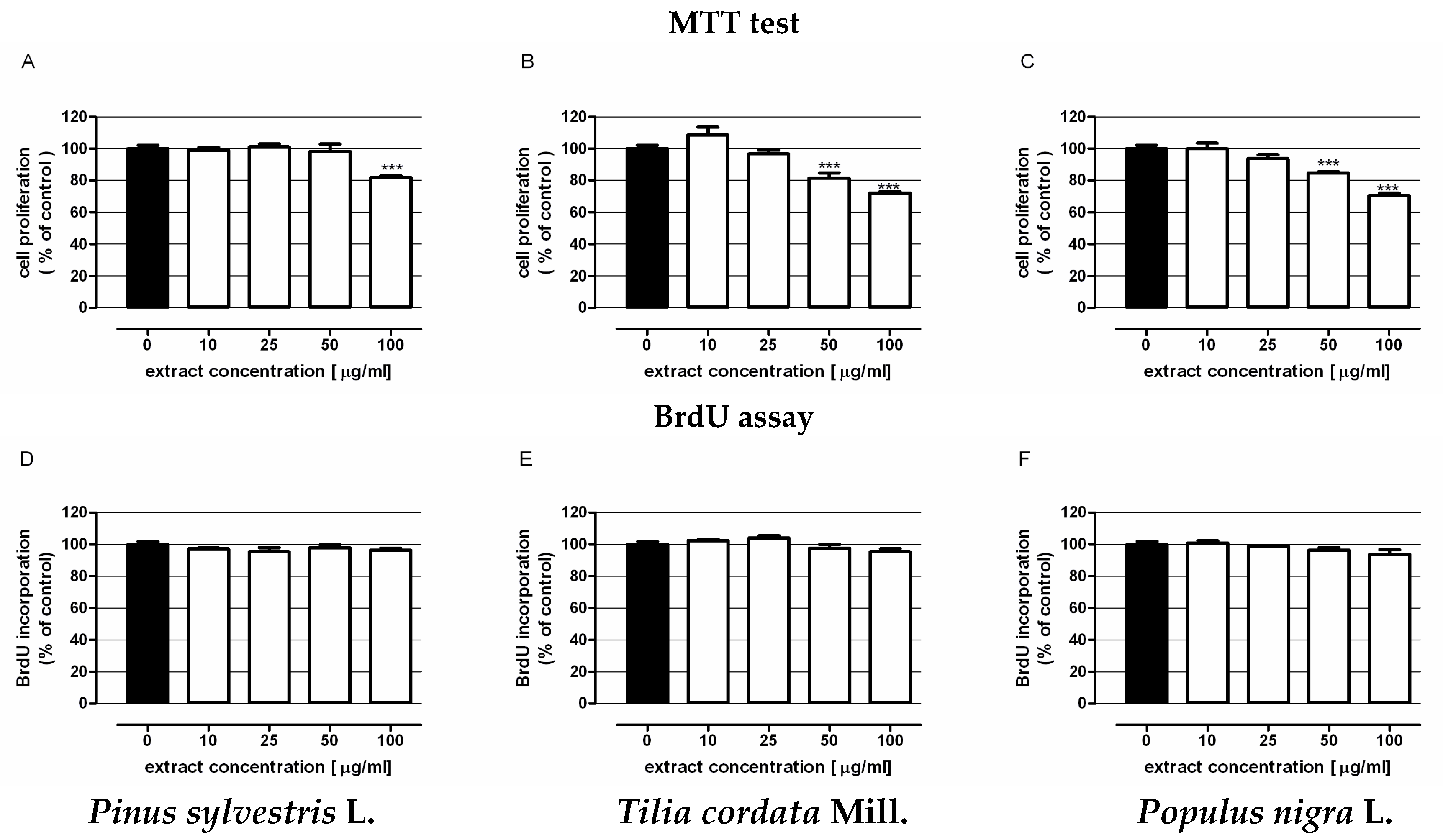
3.5.2. Examination of Extract Antiproliferative Activity
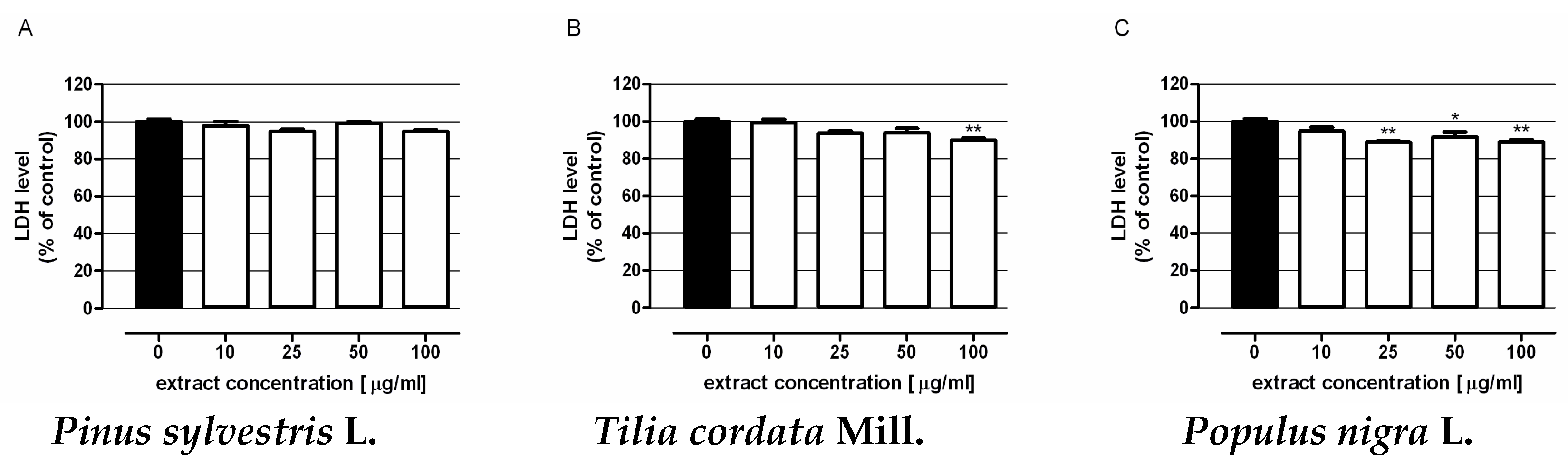
3.5.3. Assessment of Cytotoxicity of Extracts
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Onay-Uçar, E.; Karagöz, A.; Arda, N. Antioxidant activity of Viscum album spp. album. Fitoterapia 2006, 77, 556–560. [Google Scholar] [CrossRef] [PubMed]
- Orhan, D.D.; Aslan, M.; Sendogdu, N.; Ergun, F.; Yesilada, E. Evaluation of the hypoglycemic effect and antioxidant activity of three Viscum album subspecies (European mistletoe) in streptozotocin-diabetic rats. J. Ethnopharmacol. 2005, 98, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Deeni, Y.Y.; Sadiq, N.M. Antimicrobial properties and phytochemical constituents of the leaves of African mistletoe (Tapinanthus dodoneifolius (DC) Danser) (Loranthaceae): An ethnomedicinal plant of Hausaland, Northern Nigeria. J. Ethnopharmacol. 2002, 83, 235–240. [Google Scholar] [CrossRef]
- Amabeoku, G.J.; Leng, M.J.; Syce, J.A. Antimicrobial and anticonvulsant activities of Viscum capense. J. Ethnopharmacol. 1998, 61, 237–241. [Google Scholar] [CrossRef]
- Gorter, R.; Khwaja, T.; Linder, M. Anti-HIV and immunomodulating activities of Viscum album (mistletoe). Int. Conf. AIDS 1992, 84, 19–24. [Google Scholar]
- Simirgiotis, M.J.; Quispe, C.; Areche, C.; Sepúlveda, B. Phenolic Compounds in Chilean Mistletoe (Quintral, Tristerix tetrandus) Analyzed by UHPLC–Q/Orbitrap/MS/MS and Its Antioxidant Properties. Molecules 2016, 21, 245. [Google Scholar] [CrossRef] [PubMed]
- Son, G.S.; Ryu, W.S.; Kim, H.Y.; Woo, S.U.; Park, K.H.; Bae, J.W. Immunologic response to mistletoe extract (Viscum album L.) after conventional treatment in patientswith operable breast cancer. J. Breast Cancer 2010, 13, 14–18. [Google Scholar] [CrossRef]
- Beuth, J.; Schneider, B.; Schierholz, J.M. Impact of complementary treatment of breast cancer patients with standardized mistletoe extract during aftercare: A controlled multicenter comparative epidemiological cohort study. Anticancer Res. 2008, 28, 523–527. [Google Scholar] [PubMed]
- Mistletoe Extracts (PDQ®)-Health Professional Version. Available online: https://www.cancer.gov/about-cancer/treatment/cam/hp/mistletoe-pdq#cit/section_5.28 (accessed on 20 January 2017 ).
- Siegel, R.; Desantis, C.; Jemal, A. Colorectal cancer statistics, 2014. CA Cancer J. Clin. 2014, 64, 104–117. [Google Scholar] [CrossRef] [PubMed]
- Li, W.-Y.; Chan, S.-W.; Guo, D.-J.; Yu, P.H.-F. Correlation between Antioxidative Power and Anticancer Activity in Herbs from Traditional Chinese Medicine Formulae with Anticancer Therapeutic Effect. Pharm. Biol. 2008, 45, 541–546. [Google Scholar] [CrossRef]
- Rosa, L.S.; Silva, N.J.A.; Soares, N.C.P.; Monteiro, M.C.; Teodoro, A.J. Anticancer Properties of Phenolic Acids in Colon Cancer A Review. J. Nutr. Food Sci. 2016, 6. [Google Scholar] [CrossRef]
- Batra, P.; Sharma, A.K. Anti-cancer potential of flavonoids: Recent trends and future perspectives. Biotech 2013, 3, 439–459. [Google Scholar] [CrossRef] [PubMed]
- Friedel, W.E.; Matthes, H.; Bock, P.R.; Zänker, K.S. Systematic evaluation of the clinical effects of supportive mistletoe treatment within chemo- and/or radiotherapy protocols and long-term mistletoe application in nonmetastatic colorectal carcinoma: Multicenter, controlled, observational cohort study. J. Soc. Integr. Oncol. 2009, 7, 29–43. [Google Scholar]
- Edlund, U.; Hensel, A.; Frӧse, D.; Pfűller, U.; Scheffler, A. Polysaccharides from fresh Viscum album L. Berry extract and their interaction with Viscum album Agglutinin I. Arzneimittel-Forschung/Drug Res. 2000, 7, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Azuma, J.I.; Kim, N.H.; Heux, L.; Vuong, R.; Chanzy, H. Cellulose system in viscin from mistletoe berries. Cellulose 2000, 1, 3–19. [Google Scholar] [CrossRef]
- Wagner, H.; Jordan, E. An immunologically active arabinogalactan from Viscum album ‘berries’. Phytochemistry 1988, 8, 2511–2517. [Google Scholar] [CrossRef]
- Cammarata, P.L.; Cajelli, E. Free amino acid content of Viscum album L. berries parasitizing the Pinus silvestris L. and Pinus nigra Arnold var. austriaca. Boll. Chim. Farm. 1967, 8, 521–526. [Google Scholar]
- Carbonaro, M.; Virgili, F.; Carnovale, E. Evidence for protein-tannin interaction in legumes: Implications in the antioxidant properties of faba bean tannins. Lebens-Wiss Technol. 1996, 29, 743–750. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Okuda, T.; Yoshida, T.; Hatano, T. Antioxidant effects of tannins and related polyphenols. In Phenolic Compounds in Food and their Effects on Health. II. Antioxidants and Cancer prevention; Ho, C.T., Lee, C.Y., Huang, M.T., Eds.; American Chemical Society: Washington DC, USA, 1992; pp. 87–97. [Google Scholar]
- Amzad Hossain, M.; Mizanur Rahman, S.M. Isolation and characterization of flavonoids from the leaves of medicinal plant Orthosiphon stamineus. Arabian J. Chem. 2015, 8, 218–221. [Google Scholar] [CrossRef]
- Williamson, G.; Manach, C. Bioavailability and bioefficacy of polyphenols in humans. II. Review of 93 intervention studies. Am. J. Clin. Nutr. 2005, 81, 243S–255S. [Google Scholar] [PubMed]
- Wilkins, C. Impressive development in mass spektrometry. Trends Anal. Chem. 2007, 26, 65–67. [Google Scholar] [CrossRef]
- Watson, J.T.; Sparkman, O.D. Introduction to Mass Spectrometry. Instrumentation, Applications and Strategies for Data Interpretation; John Wiley & Sons Ltd.: Anglia, UK, 2007. [Google Scholar]
- Jiang, Z.; Wang, Y.; Zheng, Y.; Yang, J.; Zhang, L. Ultra high performance liquid chromatography coupled with triple quadrupole mass spectrometry and chemometric analysis of licorice based on the simultaneous determination of saponins and flavonoids. J. Sep. Sci. 2016, 39, 2928–2940. [Google Scholar] [CrossRef] [PubMed]
- Saibabu, V.; Zeeshan, F.; Luqman, A.K.; Saif, H. Therapeutic Potential of Dietary Phenolic Acids. Adv. Pharm. Sci. 2015, 2015, 823539. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Pietta, P.G. Flavonoids as antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, M.A.; Alilou, M. Naringenin attenuates CCl4-induced hepatic inflammation by the activation of an Nrf2-mediated pathway in rats. Clin. Exp. Pharmacol. Physiol. 2014, 41, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Park, K.Y.; Moon, S.H.; Rhee, S.H.; Young, H.S. Antimutagenic effect of plant flavonoids in the Salmonella assay system. Arch. Pharm. Res. 1994, 17, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Ekambaram, G.; Rajendran, P.; Magesh, V.; Sakthisekaran, D. Naringenin reduces tumor size and weight lost in N-methyl-N′-nitro-N-nitrosoguanidine-induced gastric carcinogenesis in rats. Nutr. Res. 2008, 28, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Song, H.M.; Park, G.H.; Eo, H.J.; Jeong, J.B. Naringenin-Mediated ATF3 Expression Contributes to Apoptosis in Human Colon Cancer. Biomol. Ther. 2016, 2, 140–146. [Google Scholar]
- Song, H.M.; Park, G.H.; Eo, H.J.; Lee, J.W.; Kim, M.K.; Lee, J.R.; Lee, M.H.; Koo, J.S.; Jeong, J.B. Anti-Proliferative Effect of Naringenin through p38-Dependent Downregulation of Cyclin D1 in Human Colorectal Cancer Cells. Biomol. Ther. (Seoul) 2015, 23, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, T.; Vanamala, J.; Taddeo, S.S.; Davidson, L.A.; Murphy, M.E.; Patil, B.S.; Wang, N.; Carroll, R.J.; Chapkin, R.S.; Lupton, J.R.; et al. Apigenin and naringenin suppress colon carcinogenesis through the aberrant crypt stage in azoxymethane-treated rats. Exp. Biol. Med. 2010, 235, 710–717. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.H.; Kim, S.C.; Shin, B.Y.; Jin, S.H.; Jo, M.J.; Jegal, K.H.; Kim, Y.W.; Lee, J.R.; Ku, S.K.; Cho, I.J.; et al. O-Methylated flavonol isorhamnetin prevents acute inflammation through blocking of NF-κB activation. Food Chem. Toxicol. 2013, 59, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.H.; Shin, B.Y.; Han, J.Y.; Kim, M.G.; Wi, J.E.; Kim, Y.W.; Cho, I.J.; Kim, S.C.; Shin, S.M.; Ki, S.H. Isorhamnetin protects against oxidative stress by activating Nrf2 and inducing the expression of its target genes. Toxicol. Appl. Pharmacol. 2014, 15, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Saud, S.M.; Young, M.R.; Jones-Hall, Y.L.; Ileva, L.; Evbuomwan, M.O.; Wise, J.; Colburn, N.H.; Kim, Y.S.; Bobe, G. Chemopreventive activity of plant flavonoid isorhamnetin in colorectal cancer is mediated by oncogenic Src and β-catenin. Cancer Res. 2013, 73, 5473–5484. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.; Seo, K.; Ki, S.H.; Shin, S.M. Isorhamnetin Inhibits Reactive Oxygen Species-Dependent Hypoxia Inducible Factor (HIF)-1α Accumulation. Biol. Pharm. Bull. 2016, 39, 1830–1838. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, L.; Manu, K.A.; Shanmugam, M.K.; Li, F.; Siveen, K.S.; Vali, S.; Kapoor, S.; Abbasi, T.; Surana, R.; Smoot, D.T.; et al. Isorhamnetin Inhibits Proliferation and Invasion and Induces Apoptosis through the Modulation of Peroxisome Proliferator-activated Receptor γ Activation Pathway in Gastric Cancer. J. Biol. Chem. 2012, 287, 38028–38040. [Google Scholar] [CrossRef] [PubMed]
- Philchenkov, A.A.; Zavelevych, M.P. Rhamnazin inhibits proliferation and induces apoptosis of human jurkat leukemia cells in vitro. Ukr. Biochem. J. 2015, 87, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Cai, W.; Pei, C.G.; Shao, Y. Rhamnazin, a novel inhibitor of VEGFR2 signaling with potent antiangiogenic activity an antitumor efficacy. Biochem. Biophys. Res. Commun. 2015, 458, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Olech, M.; Nowak, R.; Los, R.; Rzymowska, J.; Malm, A.; Chruściel, K. Biological activity and composition of teas and tinctures prepared from Rosa rugosa Thunb. Cent. Eur. J. Biol. 2012, 7, 172–182. [Google Scholar] [CrossRef]
- Lamaison, J.L.C.; Carret, A. Teneurs en principaux flavonoides des fleurs de Cratageus monogyna Jacq et de Cratageus Laevigata (Poiret D.C) en Fonction de la vegetation. Plant Méd. Phytothér. 1990, 25, 12–16. [Google Scholar]
- Pietrzak, W.; Nowak, R.; Olech, M. Effect of extraction method on phenolic content and antioxidant activity of mistletoe extracts from Viscum album subsp. abietis. Chem. Pap. 2014, 68, 976–982. [Google Scholar] [CrossRef]
- Nowacka, N.; Nowak, R.; Drozd, M.; Olech, M.; Los, R.; Malm, A. Analysis of phenolic constituents, antiradical and antimicrobial activity of edible mushrooms growing wild in Poland. LWT-Food Sci. Technol. 2014, 59, 689–694. [Google Scholar] [CrossRef]
- Gawlik-Dziki, U.; Świeca, M.; Dziki, D.; Sęczyk, Ł.; Złotek, U.; Różyło, R.; Kaszuba, K.; Ryszawy, D.; Czyż, J. Anticancer and Antioxidant Activity of Bread Enriched with Broccoli Sprouts. BioMed Res. Int. 2014. [Google Scholar] [CrossRef] [PubMed]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS• radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Oyaizu, M. Studies on products of browning reaction–antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. 1984, 44, 307–315. [Google Scholar] [CrossRef]
- Guo, J.T.; Lee, H.L.; Chiang, S.H.; Lin, H.I.; Chang, C.Y. Antioxidant properties of the extracts from different parts of broccoli in Taiwan. J. Food Drug Anal. 2001, 9, 96–101. [Google Scholar]
- Kuo, J.-M.; Yeh, D.-B.; Pan, B. Rapid photometric assay evaluating antioxidative activity in edible part material. J. Agric. Food Chem. 1999, 47, 3206–3209. [Google Scholar] [CrossRef] [PubMed]
- Lemieszek, M.K.; Cardoso, C.; Nunes, F.H.F.M.; Barros, A.I.R.N.A.; Marques, G.; Pożarowski, P.; Rzeski, W. Boletus edulis biological active biopolymers induce cell cycle arrest in human colon adenocarcinoma cells. Food Funct. 2013, 4, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Litchfield, J.T.; Wilcoxon, F.A. A simplified method of evaluating dose-effect experiments. J. Pharmacol. Exp. Ther. 1949, 96, 99–113. [Google Scholar] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
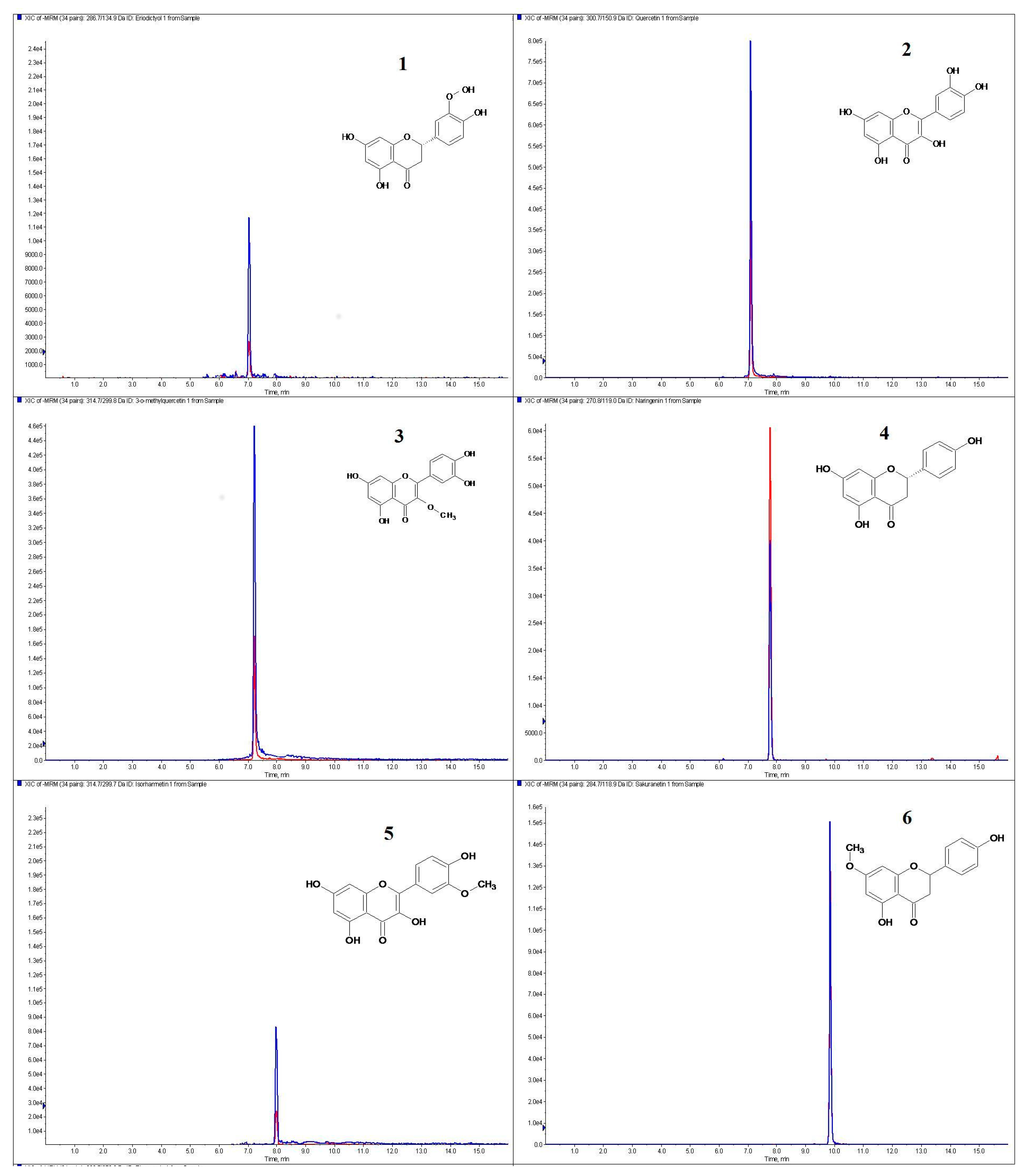

| Species | Host Tree | TPC (mg GA g−1 of dry extract) | TFC (mg Q g−1 of dry extract) |
|---|---|---|---|
| V. album | Fraxinus pensylvanica Marsh. | 8.82 ± 0.18 ns | 0.365 ± 0.005 ns |
| Malus domestica Borkh. | 6.71 ± 0.32 * | 0.356 ± 0.006 ns | |
| Sorbus aucuparia L | 9.66 ± 0.28 ns | 0.428 ± 0.007 ns | |
| Populus nigra ‘Italica’ L. | 10.43 ± 0.20 | 0.404 ± 0.017 | |
| Tilia cordata Mill. | 4.78 ± 0.09 * | 0.286 ± 0.007 ns | |
| V. austriacum | Pinus sylvestris L. | 7.11 ± 0.20 ns | 0.270 ± 0.007 ns |
| Compound | V. album | V. austriacum | ||||
|---|---|---|---|---|---|---|
| Fraxinus pensylvanica Marsh. | Malus domestica Borkh. | Sorbus aucuparia L. | Populus nigra ‘Italica’ L. | Tilia cordata Mill. | Pinus sylvestris L. | |
| Flavonoid aglycones (µg per g dry extracts) | ||||||
| Taxifolin | trace a | - b | trace a | trace a | trace a | - b |
| Myricetin | - b | - b | - b | - b | - b | - b |
| Morin | - b | - b | - b | - b | - b | - b |
| Eriodictiol | 0.011 ± 0.001 | trace a | 0.011 ± 0.001 | 0.014 ± 0.001 | 0.201 ± 0.01 | 0.001 ± 0.0001 |
| Luteolin | - b | - b | - b | trace a | - b | - b |
| Quercetin | trace a | 0.025 ± 0.0001 | 0.006 ± 0.0001 | 0.056 ± 0.003 | trace a | 0.026 ±0.0001 |
| 3-O-Methylquercetin | 0.255 ± 0.002 | 0.163 ± 0.008 | 0.510 ± 0.020 | 0.532 ± 0.004 | 0.409 ± 0.02 | 0.009 ± 0.0001 |
| Apigenin | - b | - b | trace a | trace a | trace a | trace a |
| Naringenin | 0.111 ± 0.002 | 0.102 ± 0.002 | 0.427 ± 0.0001 | 0.681 ± 0.014 | 2.319 ± 0.046 | 0.251 ± 0.013 |
| Kaempferol | - b | - b | - b | trace a | - b | trace a |
| Isorhamnetin | trace a | 0.099 ± 0.005 | 0.106 ± 0.005 | 0.297 ± 0.006 | 1.794 ± 0.09 | 0.013 ± 0.001 |
| Rhamnetin | trace a | trace a | trace a | trace a | trace a | trace a |
| Chrysin | - b | - b | - b | - b | - b | - b |
| Sakuranetin | 0.014 ± 0.0001 | 0.001 ± 0.0002 | 0.054 ± 0.003 | 0.109 ± 0.0003 | 0.009 ± 0.0001 | 0.004 ± 0.0001 |
| Prunetin | - b | - b | - b | - b | - b | - b |
| Rhamnazin | 0.009 ± 0.0001 | 0.067 ± 0.003 | 0.122 ± 0.002 | 0.472 ± 0.024 | 1.223 ± 0.061 | 0.008 ± 0.0001 |
| TOTAL | 0.4 | 0.457 | 1.236 | 2.161 | 5.955 | 0.312 |
| Compound | V. album | V. austriacum | ||||
|---|---|---|---|---|---|---|
| Fraxinus pensylvanica Marsh. | Malus domestica Borkh. | Sorbus aucuparia L. | Populus nigra ‘Italica’ L. | Tilia cordata Mill. | Pinus sylvestris L. | |
| Phenolic Acids (µg per g dry Extracts) | ||||||
| Gallic acid | 23.17 ± 0.58 | trace a | trace a | trace a | trace a | - b |
| Protocatechuic acid | 80.78 ± 0.57 | 40.88 ± 1.59 | 29.03 ± 1.45 | 31.07 ± 1.55 | trace a | trace a |
| Gentisic acid | - b | - b | trace a | 3.26 ± 0.16 | trace a | trace a |
| 4-OH-benzoic acid | 11.23 ± 0.11 | 5.20 ± 0.14 | 2.72 ± 0.14 | 0.79 ± 0.04 | 3.51 ± 0.18 | trace a |
| Vanilic acid | 26.1 ± 1.02 | Trace a | 43.9 ± 1.36 | 44.1 ± 2.21 | trace a | trace a |
| Caffeic acid | 12.78 ± 0.59 | 1.57 ± 0.08 | 34.74 ± 0.52 | 9.40 ± 0.12 | trace a | trace a |
| Syringic acid | 98.60 ± 0.69 | 24.90 ± 0.05 | 121.54 ± 1.58 | 137.99 ± 1.24 | 49.35 ± 0.1 | 25.77 ± 0.05 |
| p-Coumaric acid | 1.19 ± 0.03 | trace a | trace a | trace a | trace a | 7.07 ± 0.05 |
| Ferulic acid | 4.65 ± 0.005 | 13.64 ± 0.67 | 6.94 ± 0.28 | 7.69 ± 0.08 | 6.66 ± 0.07 | 11.81 ± 0.01 |
| Salicylic acid | 4.32 ± 0.17 | 1.22 ± 0.01 | 5.18 ± 0.02 | 7.98 ± 0.07 | 0.12 ± 0.005 | 2.06 ± 0.03 |
| Veratric acid | - b | - b | - b | - b | - b | - b |
| Synapic acid | 78.50 ± 2.36 | 71.82 ± 0.43 | 107.08 ± 1.07 | 112.17 ± 1.79 | 84.80 ± 1.36 | 8.60 ± 0.09 |
| 3-OH-cinnamic acid | - b | - b | - b | - b | - b | - b |
| Rosmarinic acid | trace a | trace a | trace a | trace a | trace a | trace a |
| TOTAL | 341.32 | 159.23 | 351.13 | 354.45 | 144.44 | 55.31 |
| Species | Host Trees | Antioxidant Activity (EC50 in mg mL−1 of extract) | |||
|---|---|---|---|---|---|
| RED | ABTS• | CHEL | LPO | ||
| V. album | Fraxinus pensylvanica Marsh. | 1.04 ± 0.02 ns | 8.06 ± 0.12 ns | 0.88 ± 0.18 * | 5.30 ± 0.03 * |
| Malus domestica Borkh. | 1.21 ± 0.02 ns | 13.03 ± 0.06 ** | 0.54 ± 0.03 ns | 7.55 ± 0.19 ** | |
| Sorbus aucuparia L. | 1.05 ± 0.01 ns | 7.72 ± 0.13 ** | 0.59 ± 0.10 ns | 6.60 ± 0.33 * | |
| Populus nigra ‘Italica’ L. | 1.08 ± 0.01 | 8.17 ± 0.02 | 0.54 ± 0.03 | 5.65 ± 0.44 | |
| Tilia cordata Mill. | 0.92 ± 0.03 ns | 11.31 ± 0.14 ** | 0.45 ± 0.10 ns | 5.57 ± 0.43 ns | |
| V. austriacum | Pinus sylvestris L. | 1.33 ± 0.02 ns | 14.77 ± 0.25 ** | 0.60 ± 0.01 ns | 10.24 ± 0.25 ** |
| Content/EC50 | R (X,Y) | R2 |
|---|---|---|
| TPC & EC50 (ABTS•) | −0.70 | 0.49 |
| TFC & EC50 (ABTS•) | −0.81 | 0.65 |
| Total phenolic acids content (LC-ESI-MS/MS) & EC50 (ABTS•) | −0.97 | 0.95 |
| Total flavonoid aglycones content (LC-ESI-MS/MS) & EC50 (ABTS•) | −0.07 | 0.01 |
| Compound | Retention Time (min) | Q1 (m/z) | Q3 (m/z) | DP a (V) | EP b (V) | CEP c (V) | CE d (eV) | CXP e (V) |
|---|---|---|---|---|---|---|---|---|
| Taxifolin | 6.17 | 302.7 | 124.9 | −45 | −3.5 | −18 | −26 | 0 |
| 284.8 | −45 | −3.5 | −18 | −14 | −4 | |||
| Myricetin | 6.47 | 316.7 | 136.9 | −55 | −9 | −14 | −32 | 0 |
| 150.9 | −55 | −9 | −14 | −26 | 0 | |||
| Morin | 6.82 | 300.7 | 124.9 | −50 | −3.5 | −20 | −24 | 0 |
| 106.9 | −50 | −3.5 | −20 | −30 | 0 | |||
| Eriodictiol | 7.07 | 286.7 | 134.9 | −45 | −6 | −12 | −32 | 0 |
| 150.9 | −45 | −6 | −12 | −18 | −2 | |||
| Luteolin | 7.08 | 284.7 | 132.9 | −75 | −9 | −18 | −38 | 0 |
| 150.9 | −75 | −9 | −18 | −26 | 0 | |||
| Quercetin | 7.13 | 300.7 | 150.9 | −60 | −2.5 | −12 | −26 | 0 |
| 178.8 | −60 | −2.5 | −12 | −20 | −2 | |||
| 3-O-Methylquercetin | 7.32 | 314.7 | 299.8 | −55 | −9.5 | −22 | −18 | −4 |
| 270.8 | −55 | −9.5 | −22 | −26 | −4 | |||
| Apigenin | 7.77 | 268.8 | 117 | −70 | −9.5 | −12 | −44 | 0 |
| 106.8 | −70 | −9.5 | −12 | −34 | 0 | |||
| Naringenin | 7.81 | 270.8 | 119 | −50 | −11.5 | −12 | −34 | 0 |
| 150.9 | −50 | −11.5 | −12 | −22 | 0 | |||
| Kaempferol | 7.94 | 284.7 | 116.8 | −70 | −5 | −12 | −46 | 0 |
| 93 | −70 | −5 | −12 | −52 | 0 | |||
| Isorhamnetin | 8.09 | 314.7 | 299.7 | −65 | −2.5 | −26 | −20 | −4 |
| 150.9 | −65 | −2.5 | −26 | −30 | 0 | |||
| Rhamnetin | 8.85 | 314.7 | 165 | −60 | −5.5 | −18 | −24 | 0 |
| 120.9 | −60 | −5.5 | −18 | −36 | 0 | |||
| Chrysin | 9.83 | 252.8 | 208.9 | −80 | −10 | −14 | −22 | −2 |
| 142.9 | −80 | −10 | −14 | −26 | 0 | |||
| Sakuranetin | 9.89 | 284.7 | 118.9 | −60 | −5.5 | −12 | −34 | 0 |
| 164.8 | -60 | -5.5 | −12 | −20 | −2 | |||
| Prunetin | 10.18 | 282.8 | 267.7 | −55 | −12 | −18 | −20 | −4 |
| 238.7 | −55 | −12 | −18 | −26 | −2 | |||
| Rhamnazin | 10.31 | 328.7 | 270.8 | −70 | −3 | −28 | −26 | −2 |
| 313.8 | −70 | −3 | −28 | −14 | −4 |
| Compound | LOD (ng/µL) | LOQ (ng/µL) | R2 | Linearity Range (ng/µL) |
|---|---|---|---|---|
| Taxifolin | 0.015 | 0.05 | 0.9972 | 0.05–5 |
| Myricetin | 0.002 | 0.004 | 0.9959 | 0.011–3.6 |
| Morin | 0.0015 | 0.003 | 0.9951 | 0.01–5 |
| Eriodictiol | 0.005 | 0.015 | 0.9976 | 0.015–5 |
| Luteolin | 0.024 | 0.04 | 0.9961 | 0.04–4 |
| Quercetin | 0.0015 | 0.003 | 0.9960 | 0.031–3.1 |
| 3-O-Methylquercetin | 0.001 | 0.002 | 0.9962 | 0.011–3.7 |
| Apigenin | 0.003 | 0.004 | 0.9952 | 0.012–6.3 |
| Naringenin | 0.025 | 0.033 | 0.9959 | 0.033–3.3 |
| Kaempferol | 0.02 | 0.033 | 0.9973 | 0.033–20 |
| Isorhamnetin | 0.012 | 0.024 | 0.9965 | 0.04–60 |
| Rhamnetin | 0.002 | 0.006 | 0.9955 | 0.006–0.625 |
| Chrysin | 0.025 | 0.042 | 0.9954 | 0.042–2.5 |
| Sakuranetin | 0.034 | 0.046 | 0.9955 | 0.072–7.2 |
| Prunetin | 0.05 | 0.075 | 0.9967 | 0.2–20 |
| Rhamnazin | 0.054 | 0.072 | 0.9962 | 0.072–7.2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pietrzak, W.; Nowak, R.; Gawlik-Dziki, U.; Lemieszek, M.K.; Rzeski, W. LC-ESI-MS/MS Identification of Biologically Active Phenolic Compounds in Mistletoe Berry Extracts from Different Host Trees. Molecules 2017, 22, 624. https://doi.org/10.3390/molecules22040624
Pietrzak W, Nowak R, Gawlik-Dziki U, Lemieszek MK, Rzeski W. LC-ESI-MS/MS Identification of Biologically Active Phenolic Compounds in Mistletoe Berry Extracts from Different Host Trees. Molecules. 2017; 22(4):624. https://doi.org/10.3390/molecules22040624
Chicago/Turabian StylePietrzak, Wioleta, Renata Nowak, Urszula Gawlik-Dziki, Marta Kinga Lemieszek, and Wojciech Rzeski. 2017. "LC-ESI-MS/MS Identification of Biologically Active Phenolic Compounds in Mistletoe Berry Extracts from Different Host Trees" Molecules 22, no. 4: 624. https://doi.org/10.3390/molecules22040624
APA StylePietrzak, W., Nowak, R., Gawlik-Dziki, U., Lemieszek, M. K., & Rzeski, W. (2017). LC-ESI-MS/MS Identification of Biologically Active Phenolic Compounds in Mistletoe Berry Extracts from Different Host Trees. Molecules, 22(4), 624. https://doi.org/10.3390/molecules22040624







