The Pharmacological Effects of Lutein and Zeaxanthin on Visual Disorders and Cognition Diseases
Abstract
:1. Introduction
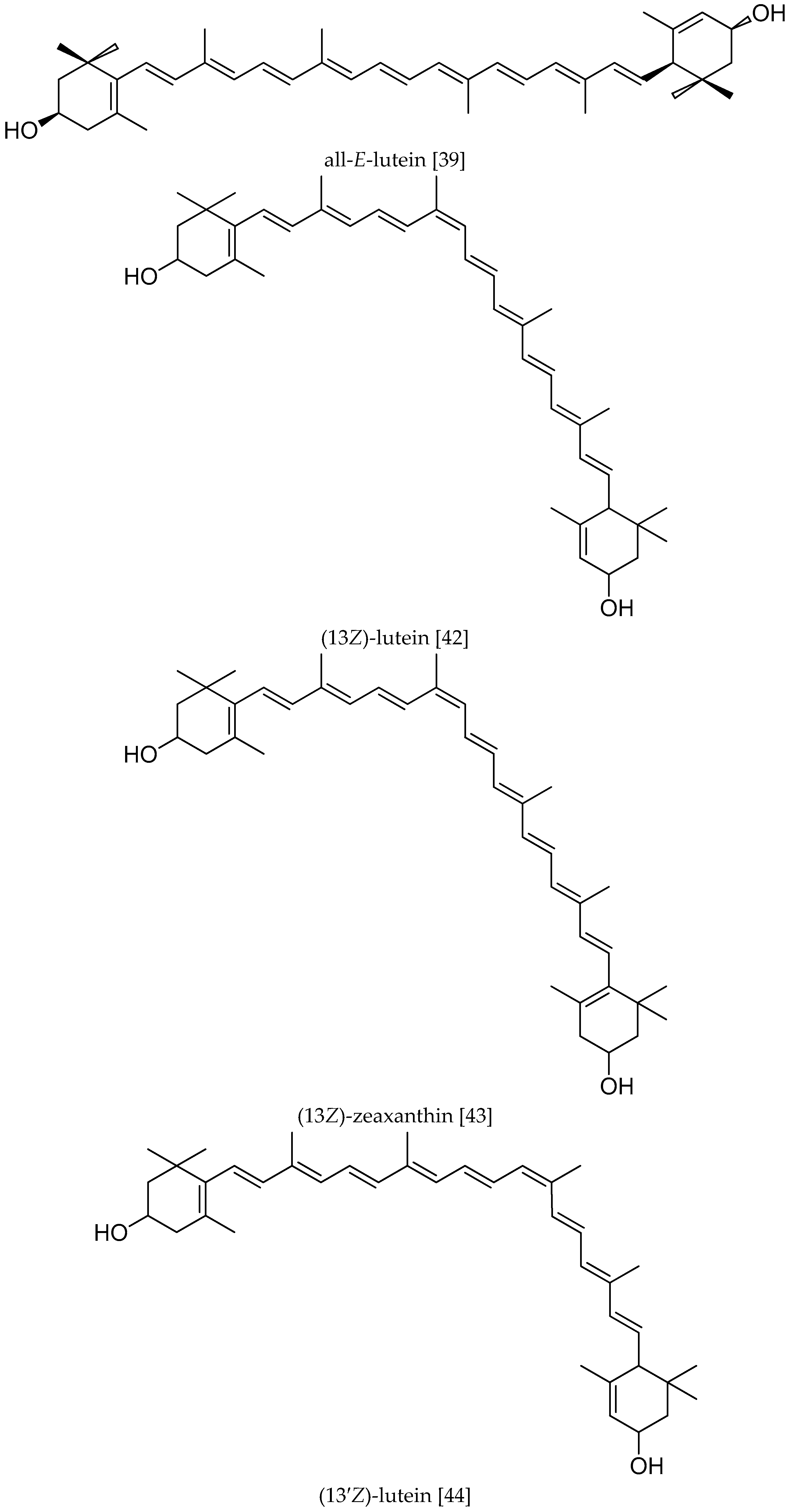
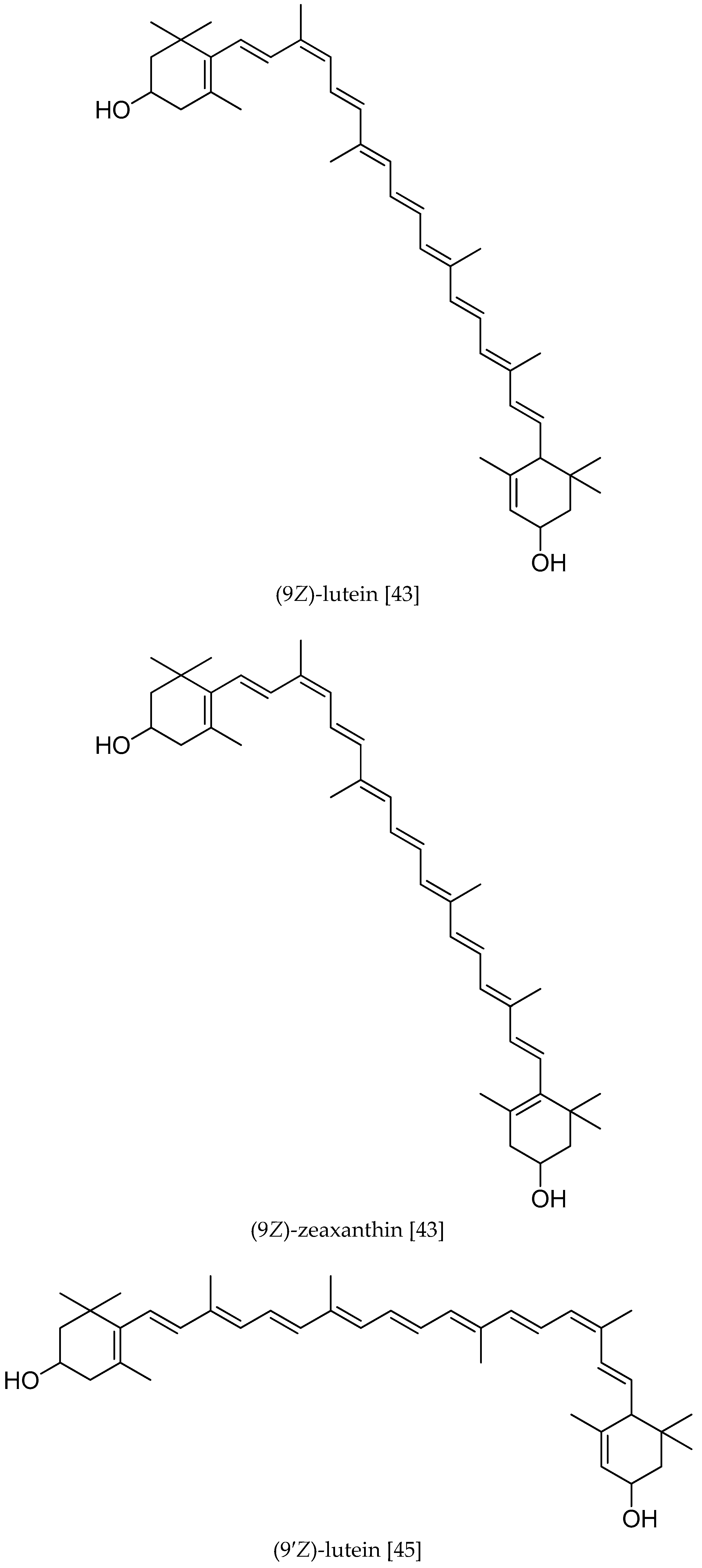
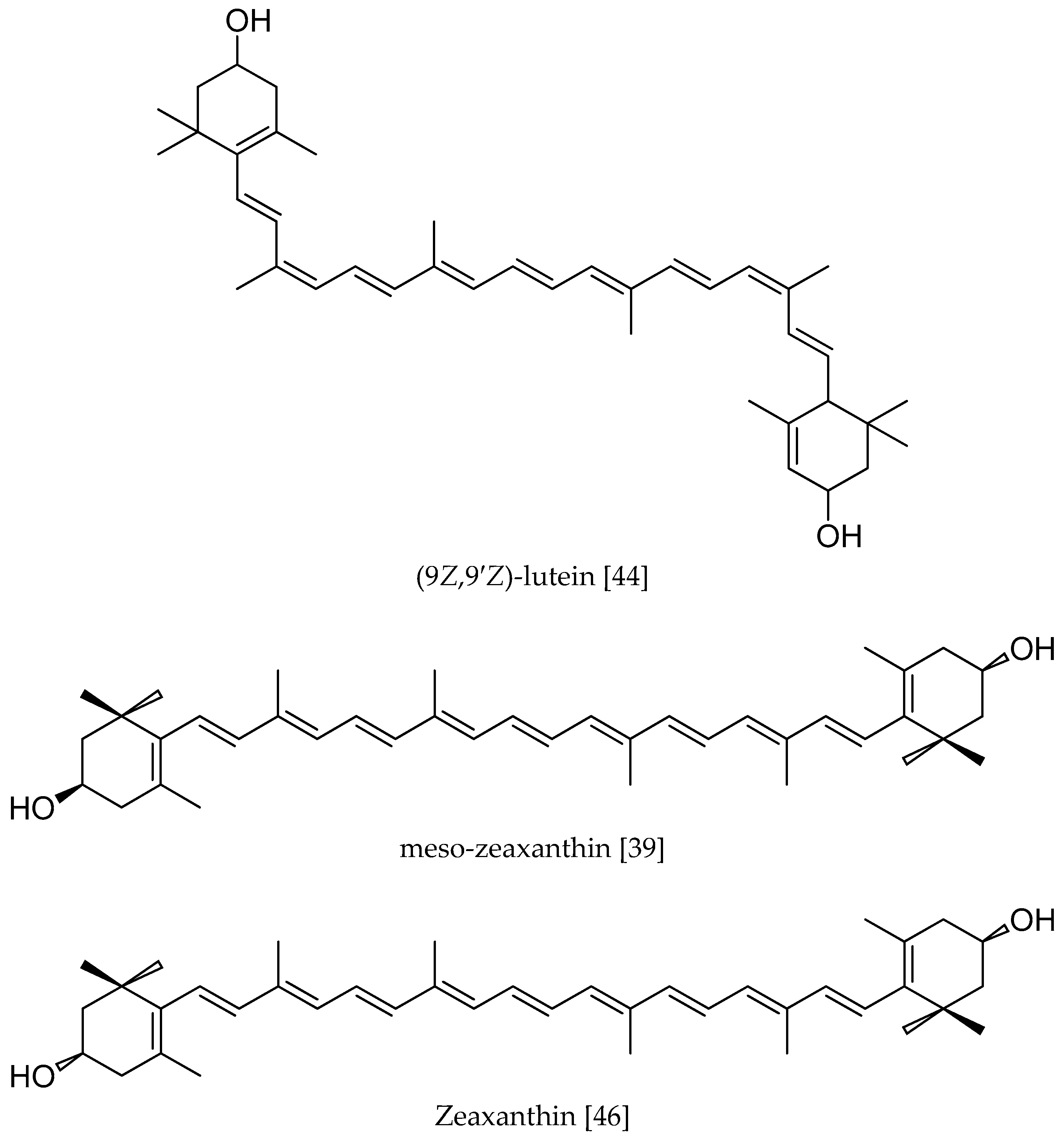
2. The Structure and Distribution of L and Z
3. The Effects on AMD
4. The Effects on ARC
5. The Effects on Cognitive Function
5.1. The Effects on Infancy Cognition Function
5.2. The Effects on Adult Cognition Function
5.3. The Effects on Alzheimer’s Disease (AD)
6. The Consumption and Edible Safety of Lutein
6.1. The Intervention of Lutein in AMD Study
6.2. Intervention Doses in Lutein and ARC Studies
6.3. Safe Consumption of Lutein
7. Conclusions and Future Directions
Acknowledgments
Conflicts of Interest
Abbreviations
| L | lutein |
| Z | zeaxanthin |
| MZ | meso-zeaxanthin |
| MP | macular pigment |
| AMD | age-related Macular degeneration |
| ARC | age-related Cataract |
| AD | Alzheimer’s disease |
| MPOD | macular pigment optical density |
| ADI | acceptable daily intake |
| mg/d | mg/day |
| kg·d | kg·day |
References
- Bone, R.A.; Landrum, J.T.; Hime, G.W.; Cains, A.; Zamor, J. Stereochemistry of the human macular carotenoids. Investig. Ophthalmol. Vis. Sci. 1993, 34, 2033–2040. [Google Scholar]
- Carotenoids. Handbook; Britton, G.; Liaaen-Jensen, S.; Pfander, H. (Eds.) Birkhäuser Basel: Basel, Switzerland, 2004. [Google Scholar]
- Khachik, F.; Beecher, G.R.; Goli, M.B.; Lusby, W.R. Separation, identification, and quantification of carotenoids in fruits, vegetables, and human plasma by high performance liquid chromatography. Pure Appl. Chem. 1991, 63, 71–80. [Google Scholar] [CrossRef]
- Khachik, F.; Spangler, C.J.; Smith, J.C.; Canfield, L.M.; Steck, A.; Pfander, H. Identification, quantification, and relative concentrations of carotenoids and their metabolites in human milk and serum. Anal. Chem. 1997, 69, 1873–1881. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, P.S. The role of lutein and zeaxanthin in protection against age-related macular degeneration. Acta Hortic. 2015, 1106, 153–160. [Google Scholar] [CrossRef]
- Van-De-Kraats, J.; Kanis, M.S.; Van-Norren, D. Lutein and zeaxanthin measured separately in the living human retina with fundus reflectometry. Investig. Ophthalmol. Vis. Sci. 2008, 49, 5568–5573. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Rubin, L.P. Role of macular xanthophylls in prevention of common neovascular retinopathies: Retinopathy of prematurity and diabetic retinopathy. Arch. Biochem. Biophys. 2015, 572, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Renzi, L.M.; Hammond, B.R. The relation between the macular carotenoids, lutein and zeaxanthin, and temporal vision. Ophthalmic Physiol. Opt. 2010, 30, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Kijlstra, A.; Tian, Y.; Kelly, E.R.; Berendschot, T.T. Lutein: More than just a filter for blue light. Prog. Retin. Eye Res. 2012, 31, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Tarsis, S.L. Preliminary identification of the human macular pigment. Vis. Res. 1985, 25, 1531–1535. [Google Scholar] [CrossRef]
- Arnold, C.; Winter, L.; Fröhlich, K.; Jentsch, S.; Dawczynski, J.; Jahreis, G.; Böhm, V. Macular xanthophylls and ω-3 long-chain polyunsaturated fatty acids in age-related macular degeneration: A randomized trial. JAMA Ophthalmol. 2013, 131, 564–572. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Friedes, L.M.; Gomez, C.M.; Kilburn, M.D.; Menendez, E.; Vidal, I.; Wang, W. Distribution of lutein and zeaxanthin stereoisomers in the human retina. Exp. Eye Res. 1997, 64, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Landrum, J.T.; Bone, R.A. Lutein, zeaxanthin, and the macular pigment. Arch. Biochem. Biophys. 2001, 385, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Fernandez, L.; Tarsis, S.L. Analysis of the macular pigment by HPLC: Retinal distribution and age study. Investig. Ophthalmol. Vis. Sci. 1988, 29, 843–849. [Google Scholar]
- Handelman, G. J.; Dratz, E.A.; Reay, C.C.; van Kuijk, F.J.G.M. Carotenoids in the human macula and whole retina. Investig. Ophthalmol. Vis. Sci. 1988, 29, 850–855. [Google Scholar]
- Wooten, B.R.; Hammond, B.R. Macular pigment: Influence on visual acuity and visibility. Prog. Retin. Eye Res. 2002, 21, 225–240. [Google Scholar] [CrossRef]
- Subczynski, W.K.; Wisniewska, A.; Widomska, J. Location of macular xanthophylls in the most vulnerable regions of photoreceptor outer-segment membranes. Arch. Biochem. Biophys. 2010, 504, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Nd, B.F.; Snodderly, D.M.; Johnson, E.J.; Schalch, W.; Koepcke, W.; Gerss, J.; Neuringer, M. Nutritional manipulation of primate retinas, V: Effects of lutein, zeaxanthin, and n-3 fatty acids on retinal sensitivity to blue-light-induced damage. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3934–3942. [Google Scholar]
- Berrow, E.J.; Bartlett, H.E.; Eperjesi, F.; Gibson, J.M. The effects of a lutein-based supplement on objective and subjective measures of retinal and visual function in eyes with age-related maculopathy—A randomised controlled trial. Br. J. Nutr. 2013, 109, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ham, W.T.; Mueller, H.A.; Sliney, D.H. Retinal sensitivity to damage from short wavelength light. Nature 1976, 260, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, P.S.; Khachik, F.; Carvalho, L.S.; Carvalho, L.S.; Muir, G.J.; Zhao, D.Y.; Katz, N.B. Identification and quantitation of carotenoids and their metabolites in the tissues of the human eye. Exp. Eye Res. 2001, 72, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Granado, F.; Olmedilla, B.; Blanco, I. Nutritional and clinical relevance of lutein in human health. Br. J. Nutr. 2003, 90, 487–502. [Google Scholar] [CrossRef] [PubMed]
- Reading, V.M.; Weale, R.A. Macular pigment and chromatic aberration. J. Opt. Soc. Am. 1974, 64, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Renzi, L.M.; Hammond, B.R. The effect of macular pigment on heterochromatic luminance contrast. Exp. Eye Res. 2010, 91, 896–900. [Google Scholar] [CrossRef] [PubMed]
- Karppi, J.; Laukkanen, J.A.; Kurl, S. Plasma lutein and zeaxanthin and the risk of age-related nuclear cataract among the elderly Finnish population. Br. J. Nutr. 2012, 108, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J. A possible role for lutein and zeaxanthin in cognitive function in the elderly. Am. J. Clin. Nutr. 2012, 96, 1161S–1165S. [Google Scholar] [CrossRef] [PubMed]
- Alassane, S.; Binquet, C.; Cottet, V.; Fleck, O.; Acar, N.; Daniel, S.; Delcourt, C.; Bretillon, L.; Bron, A.M.; Creuzot, G.C. Relationships of macular pigment optical density with plasma lutein, zeaxanthin, and diet in an elderly population: The montrachet study. Investig. Ophthalmol. V. Sci. 2016, 57, 1160. [Google Scholar] [CrossRef] [PubMed]
- Landrum, J.T.; Bone, R.A.; Moore, L.L.; Gomez, C.M. Analysis of zeaxanthin distribution within individual human retinas. Methods Enzymol. 1999, 299, 457–467. [Google Scholar] [PubMed]
- Snodderly, D.M.; Auran, J.D.; Delori, F.C. The macular pigment. II. Spatial distribution in primate retinas. Investig. Ophthalmol. V. Sci. 1984, 25, 674–685. [Google Scholar]
- Sommerburg, O.; Siems, W.G.; Hurst, J.S.; Lewis, J.W.; Kliger, D.S.; van Kuijk, F.J. Lutein and zeaxanthin are associated with photoreceptors in the human retina. Curr. Eye Res. 1999, 19, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Gass, J.D.M. Müller cell cone, an overlooked part of the anatomy of the fovea centralis: Hypotheses concerning its role in the pathogenesis of macular hole and foveomacualr retinoschisis. Arch. Ophthalmol. 1999, 117, 821–823. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J. The role of carotenoids in human health. Nutr. Clin. Care 2002, 5, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Nakanishi, K.; Itagaki, Y.; Sparrow, J.R. Photooxidation of A2-PE, a photoreceptor outer segment fluorophore, and protection by lutein and zeaxanthin. Exp. Eye Res. 2006, 82, 828–839. [Google Scholar] [CrossRef] [PubMed]
- Junghans, A.; Sies, H.; Stahl, W. Macular pigments lutein and zeaxanthin as blue light filters studied in liposomes. Arch. Biochem. Biophys. 2001, 391, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Elsm, A.A.; Akhtar, H.; Zaheer, K.; Ali, R. Dietary sources of lutein and zeaxanthin carotenoids and their role in eye health. Nutrients 2013, 5, 1169–1185. [Google Scholar]
- Choi, R.Y.; Chortkoff, S.C.; Gorusupudi, A.; Bernstein, P.S. Crystalline maculopathy associated with high-dose lutein supplementation. Jama Ophthalmol. 2016, 134, 1445–1448. [Google Scholar] [CrossRef] [PubMed]
- Harikumar, K.B.; Nimita, C.V.; Preethi, K.C.; Kuttan, R.; Deshpande, J. Toxicity profile of lutein and lutein ester isolated from marigold flowers (Tagetes erecta). Int. J. Toxicol. 2008, 27, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Snodderly, D.M. Evidence for protection against age-related macular degeneration by carotenoids and antioxidant vitamins. Am. J. Clin. Nutr. 1995, 62, 1448S–1461S. [Google Scholar] [PubMed]
- Krinsky, N.I.; Landrum, J.T.; Bone, R.A. Biologic mechanisms of the protective role of lutein and zeaxanthin in the eye. Annu. Rev. Nutr. 2003, 23, 171–201. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.W. Age-related macular degeneration revisited—Piecing the puzzle: The LXIX Edward Jackson memorial lecture. Am. J. Ophthalmol. 2013, 155, 1–35. [Google Scholar] [CrossRef] [PubMed]
- Moukarzel, A.A.; Bejjani, R.A.; Fares, F.N. Xanthophylls and eye health of infants and adults. Leban. Med. J. 2009, 57, 261–267. [Google Scholar]
- El-Sm, A.A.; Young, J.C.; Akhtar, H.; Rabalski, I. Stability of lutein in wholegrain bakery products naturally high in lutein or fortified with free lutein. J. Agric. Food Chem. 2010, 58, 10109–10117. [Google Scholar]
- Dachtler, M.; Glaser, T.; Kohler, K.; Albert, K. Combined HPLC-MS and HPLC-NMR on-line coupling for the separation and determination of lutein and zeaxanthin stereoisomers in spinach and in retina. Anal. Chem. 2001, 73, 667–674. [Google Scholar] [CrossRef] [PubMed]
- Kull, D.R.; Pfander, H. Isolation and structure elucidation of two (Z)-isomers of lutein from the petals of rape (Brassica napus). J. Agric. Food Chem. 1997, 45, 4201–4203. [Google Scholar] [CrossRef]
- Calvo, M.M. Lutein: A valuable ingredient of fruit and vegetables. Crit. Rev. Food Sci. Nutr. 2005, 45, 671–696. [Google Scholar] [CrossRef] [PubMed]
- Khachik, F.; de Moura, F.F.; Zhao, D.Y.; Aebischer, C.P.; Bernstein, P.S. Transformations of selected carotenoids in plasma, liver, and ocular tissues of humans and in nonprimate animal models. Investig. Ophthalmol. V. Sci. 2002, 43, 3383–3392. [Google Scholar]
- Lim, L.S.; Mitchell, P.; Seddon, J.M.; Holz, F.G.; Wong, T.Y. Age-related macular degeneration. Insight J. Am. Soc. Ophthalmic Regist. Nurses 2012, 379, 1728–1738. [Google Scholar] [CrossRef]
- Sparrow, J.R.; Ueda, K.; Zhou, J. Complement dysregulation in AMD: RPE-Bruch’s membrane-choroid. Mol. Asp. Med. 2012, 33, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.R.; Fletcher, A.E.; Wormald, R.P. Age-related macular degeneration causing visual impairment in people 75 years or older in Britain: An add-on study to the Medical Research Council Trial of Assessment and Management of Older People in the Community. Ophthalmology 2004, 111, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Jager, R.D. Age-related macular degeneration. N. Engl. J. Med. 2008, 358, 2606–2617. [Google Scholar] [CrossRef] [PubMed]
- Friedman, D.S.; O’Colmain, B.J.; Muñoz, B.; Tomany, S.C.; McCarty, C.; de Jong, P.T.; Nemesure, B.; Mitchell, P.; Kempen, J. Prevalence of age-related macular degeneration in the United States. Arch. Ophthalmol. 2004, 122, 564–572. [Google Scholar] [PubMed]
- Fernández-Robredo, P.; Sádaba, L.M.; Salinas-Alamán, A.; Recalde, S.; Rodríguez, J.A.; García-Layana, A. Effect of lutein and antioxidant supplementation on VEGF expression, MMP-2 activity, and ultrastructural alterations in apolipoprotein E-deficient mouse. Oxid. Med. Cell. Longev. 2013, 2013, 433–454. [Google Scholar] [CrossRef] [PubMed]
- Chew, E.Y.; Clemons, T.E.; Agrón, E.; Sangiovanni, J.P.; Davis, M.D.; Ferris, F.L. Ten-year follow-up of age-related macular degeneration in the age-related eye disease study: AREDS report no. 36. JAMA Ophthalmol. 2014, 132, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Richer, S.P.; Stiles, W.; Grahamhoffman, K.; Levin, M.; Ruskin, D.; Wrobel, J.; Park, D.W.; Thomas, C. Randomized, double-blind, placebo-controlled study of zeaxanthin and visual function in patients with atrophic age-related macular degeneration: The Zeaxanthin and Visual Function Study (ZVF) FDA IND #78, 973. Optom. J. Am. Optom. Assoc. 2011, 82, 667–680. [Google Scholar]
- Ma, L.; Yan, S.F.; Huang, Y.M.; Lu, X.R.; Qian, F.; Pang, H.L.; Xu, X.R.; Zou, Z.Y.; Dong, P.C.; Xiao, X.; et al. Effect of lutein and zeaxanthin on macular pigment and visual function in patients with early age-related macular degeneration. Ophthalmology 2012, 119, 2290–2297. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Gurunadh, V.S. Is sunlight exposure a risk factor for age-related macular degeneration? A systematic review and meta-analysis. Br. J. Ophthalmol. 2013, 97, 389–394. [Google Scholar] [CrossRef]
- Bian, Q.; Gao, S.; Zhou, J.; Qin, J.; Taylor, A.; Johnson, E.J.; Tang, G.; Sparrow, J.R.; Gierhart, D.; Shang, F. Lutein and zeaxanthin supplementation reduces photooxidative damage and modulates the expression of inflammation-related genes in retinal pigment epithelial cells. Free Radic. Biol. Med. 2012, 53, 1298–1307. [Google Scholar] [CrossRef] [PubMed]
- Obermajer, N.; Jevnikar, Z.; Doljak, B.; Kos, J. Role of cysteine cathepsins in matrix degradation and cell signalling. Connect. Tissue Res. 2008, 49, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Hang, L.; Huang, B.; Wei, Y.; Zheng, S.; Li, W. Efficacy of ethanol extract of Fructus lycii and its constituents lutein/zeaxanthin in protecting retinal pigment epithelium cells against oxidative stress: In vivo and in vitro models of age-related macular degeneration. J. Ophthalmol. 2013, 2013, 862806. [Google Scholar] [CrossRef] [PubMed]
- Paraoan, L.; Hiscott, P.; Gosden, C.; Grierson, I. Cystatin C in macular and neuronal degenerations: Implications for mechanism(s) of age-related macular degeneration. Vis. Res. 2010, 50, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Treumer, F.; Klettner, A.; Baltz, J.; Hussain, A.A.; Miura, Y.; Brinkmann, R.; Roider, J.; Hillenkamp, J. Vectorial release of matrix metalloproteinases (MMPs) from porcine RPE-choroid explants following selective retina therapy (SRT): Towards slowing the macular ageing process. Exp. Eye Res. 2012, 97, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Muni, R.H.; Altaweel, M.; Tennant, M.; Weaver, B.; Kertes, P.J. Agreement among Canadian retina specialists in the determination of treatment eligibility for photodynamic therapy in age-related macular degeneration. Retina 2008, 28, 1421–1426. [Google Scholar] [CrossRef] [PubMed]
- Veritti, D.; Sarao, V.; Lanzetta, P. Neovascular age-related macular degeneration. Ophthalmologica 2012, 227, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Dhoot, D.S.; Kaiser, P.K. Ranibizumab for age-related macular degeneration. Expert Opin. Biol. Ther. 2012, 12, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Chakravarthy, U. Bevacizumab for the treatment of neovascular age-related macular degeneration. Ann. Pharmacother. 2012, 46, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Jarrett, S.G.; Boulton, M.E. Consequences of oxidative stress in age-related macular degeneration. Mol. Asp. Med. 2012, 33, 399–417. [Google Scholar] [CrossRef] [PubMed]
- Sugino, I.K.; Wang, H.; Zarbin, M.A. Age-related macular degeneration and retinal pigment epithelium wound healing. Mol. Neurobiol. 2003, 28, 177–194. [Google Scholar] [CrossRef]
- Mettu, P.S.; Wielgus, A.R.; Ong, S.S.; Cousins, S.W. Retinal pigment epithelium response to oxidant injury in the pathogenesis of early age-related macular degeneration. Mol. Asp. Med. 2012, 33, 376–398. [Google Scholar] [CrossRef] [PubMed]
- Zuhal, Y.; Irem, U.N.; Filiz, Y. The role of oxidative stress and antioxidants in the pathogenesis of age-related macular degeneration. Clinics 2011, 66, 743–746. [Google Scholar]
- Thornton, J.; Edwards, R.; Mitchell, P.; Harrison, R.A.; Buchan, I.; Kelly, S.P. Smoking and age-related macular degeneration: A review of Association. Eye 2005, 19, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A.; Menon, B.; Gierhart, D.L. Beneficial effect of zeaxanthin on retinal metabolic abnormalities in diabetic rats. Investig. Ophthalmol. Vis. Sci. 2008, 49, 1645–1651. [Google Scholar] [CrossRef] [PubMed]
- Pons, M.; Cousins, S.W.; Csaky, K.G.; Striker, G.; Marin-Castaño, M.E. Cigarette smoke-related hydroquinone induces ilamentous actin reorganization and heat shock protein 27 phosphorylation through p38 and extracellular signal-regulated kinase 1/2 in retinal pigment epithelium: implications for age-related macular degeneration. Am. J. Pathol. 2010, 177, 1198–1213. [Google Scholar] [PubMed]
- Klein, R.; Peto, T.; Bird, A.; Vannewkirk, M.R. The epidemiology of age-related macular degeneration. Am. J. Ophthalmol. 2004, 137, 486–495. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Heidmann, D.G.; Sall, J.; Hernandez, E.P.; Cousins, S.W. Basal laminar deposit formation in APO B100 transgenic mice: complex interactions between dietary fat, blue light, and vitamin E. Investig. Ophthalmol. V. Sci. 2004, 45, 260–266. [Google Scholar] [CrossRef]
- Catanuto, P.; Espinosaheidmann, D.; Pereirasimon, S.; Espinosa-Heidmann, D.; Pereira-Simon, S.; Sanchez, P.; Salas, P.; Hernandez, E.; Cousins, S.W.; Elliot, S.J. Mouse retinal pigmented epithelial cell lines retain their phenotypic characteristics after transfection with human papilloma virus: A new tool to further the study of RPE biology. Exp. Eye Res. 2009, 88, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M. Genetic and environmental underpinnings to age-related ocular diseases. Investig. Ophthalmol. Vis. Sci. 2013, 54, ORSF28–ORSF30. [Google Scholar] [CrossRef] [PubMed]
- Merle, B.M.; Maubaret, C.; Korobelnik, J.F.; Delyfer, M.N.; Rougier, M.B.; Lambert, J.C.; Amouyel, P.; Malet, F.; Le Goff, M.; Dartigues, J.F.; et al. Association of HDL-related loci with age-related macular degeneration and plasma lutein and zeaxanthin: The Alienor study. PLoS ONE 2013, 8, e79848. [Google Scholar] [CrossRef] [PubMed]
- Jeganathan, V.S.; Verma, N. Safety and efficacy of intravitreal anti-VEGF injections for age-related macular degeneration. Curr. Opin. Ophthalmol. 2009, 20, 223–225. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Dou, H.L.; Wu, Y.Q.; Huang, Y.B.; Xu, X.R.; Zou, Z.Y.; Lin, X.M. Lutein and zeaxanthin intake and the risk of age-related macular degeneration: A systematic review and meta-analysis. Br. J. Nutr. 2012, 107, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Scripsema, N.K.; Hu, D.N.; Rosen, R.B. Lutein, zeaxanthin, and meso-zeaxanthin in the clinical management of eye disease. J. Ophthalmol. 2015, 2015, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chew, E.Y.; Clemons, T.E.; Sangiovanni, J.P.; Danis, R.P.; Ferris, F.L.; Elman, M.J.; Antoszyk, A.N.; Ruby, A.J.; Orth, D.; Bressler, S.B.; et al. Secondary analyses of the effects of lutein/zeaxanthin on age-related macular degeneration progression: AREDS2 report No. 3. JAMA Ophthalmol. 2014, 132, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Charters, L. AREDS gets another look: Removing beta-carotene, adding lutein/zeaxanthin shows clear benefits. Formulary 2013, 48, 229–229. [Google Scholar]
- Chew, E.Y.; Clemons, T.E.; SanGiovanni, J.P.; Danis, R.; Ferris, F.L.; Elman, M.; Antoszyk, A.; Ruby, A.; Orth, D.; Bressler, S.; et al. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: The Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. J. Am. Med. Assoc. 2013, 309, 2005–2015. [Google Scholar]
- NIH Study Provides Clarity on Supplements for Protection against Blinding Eye Disease; National Eye Institute: Washington, DC, USA, 2013.
- Musch, D.C. Evidence for including lutein and zeaxanthin in oral supplements for age-related macular degeneration. JAMA Ophthalmol. 2014, 132, 139–141. [Google Scholar] [CrossRef] [PubMed]
- Murray, I.J.; Makridaki, M.; Rl, V.D.V.; Carden, D.; Parry, N.R.; Berendschot, T.T. Lutein supplementation over a one-year period in early AMD might have a mild beneficial effect on visual acuity: The CLEAR study. Investig. Ophthalmol. V. Sci. 2013, 54, 1781–1788. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Dou, H.L.; Huang, Y.M.; Huang, Y.M.; Lu, X.R.; Xu, X.R.; Qian, F.; Zou, Z.Y.; Pang, L.H.; Dong, P.C.; et al. Improvement of retinal function in early age-related macular degeneration after lutein and zeaxanthin supplementation: A randomized, double-masked, placebo-controlled trial. Am. J. Ophthalmol. 2012, 154, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Woo, J.H.; Sanjay, S.; Eong, K.A. Benefits of early awareness in age-related macular degeneration. Eye 2009, 23, 2271–2271. [Google Scholar] [CrossRef] [PubMed]
- Pinazodurán, M.D.; Gómezulla, F.; Arias, L.; Araiz, J.; Casaroli-Marano, R.; Gallego-Pinazo, R.; García-Medina, J.J.; López-Gálvez, M.I.; Manzanas, L.; Salas, A.; et al. Do nutritional supplements have a role in age macular degeneration prevention? J. Ophthalmol. 2014, 2014, 901686–901686. [Google Scholar]
- Chew, E.Y. Nutrition effects on ocular diseases in the aging eye. Investig. Ophthalmol. Vis. Sci. 2013, 54, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Beatty, S.; Chakravarthy, U.; Nolan, J.M.; Muldrew, K.A.; Woodside, J.V.; Denny, F.; Stevenson, M.R. Secondary outcomes in a clinical trial of carotenoids with coantioxidants versus placebo in early age-related macular degeneration. Ophthalmology 2013, 120, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Ho, L.; Van Leeuwen, R.; Witteman, J.C.M.; van Duijn, C.M.; Uitterlinden, A.G.; Hofman, A.; de Jong, P.T.V.M.; Vingerling, J.R.; Klaver, C.C. Reducing the genetic risk of age-related macular degeneration with dietary antioxidants, zinc, and omega-3 fatty acids. Arch. Ophthalmol. 2011, 129, 758–766. [Google Scholar] [CrossRef] [PubMed]
- Barker, F.M. Dietary supplementation: Effects on visual performance and occurrence of AMD and cataracts. Curr. Med. Res. Opin. 2010, 26, 2011–2023. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.L. Preliminary results in macular pigment optical density associated with and without zeaxanthin and lutein supplementation. Adv. Ophthalmol. Vis. Syst. 2015, 2, 0066. [Google Scholar] [CrossRef]
- Cabot, F.; Saad, A.; Mcalinden, C.; Haddad, N.M.; Grise-Dulac, A.; Gatinel, D. Objective assessment of crystalline lens opacity level by measuring ocular light scattering with a double-pass system. Am. J. Ophthalmol. 2013, 155, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Stevens, G.A.; White, R.A.; Flaxman, S.R.; Price, H.; Jonas, J.B.; Keeffe, J.; Leasher, J.; Naidoo, K.; Pesudovs, K.; Resnikoff, S.; et al. Global prevalence of vision impairment and blindness: Magnitude and temporal trends, 1990–2010. Ophthalmology 2013, 120, 2377–2384. [Google Scholar] [CrossRef] [PubMed]
- Ferrandez, A.M.; Blin, O. Causes and prevalence of visual impairment among adults the United States. Arch. Ophthalmol. 2004, 122, 477–485. [Google Scholar]
- Rao, G.N.; Khanna, R.; Payal, A. The global burden of cataract. Curr. Opin. Ophthalmol. 2011, 22, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.M.; Afshari, N.A. Cataracts: We have perfected the surgery, but is it time for prevention? Curr. Opin. Ophthalmol. 2011, 22, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Zhang, J.; Zhang, J.; Theves, M.; Strauch, C.; Nemet, I.; Liu, X.; Qian, J.; Giblin, F.J.; Monnier, V.M. Mechanism of lysine oxidation in human lens crystallins during aging and in diabetes. J. Biol. Chem. 2009, 284, 34618–34627. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yan, H.; Löfgren, S.; Tian, X.; Lou, M.F. Ultraviolet radiation-induced cataract in mice: The effect of age and the potential biochemical mechanism. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7276–7285. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.G.; Mitchell, P.; Flood, V.M.; Burlutsky, G.; Rochtchina, E.; Cumming, R.G.; Wang, J.J. Antioxidant nutrient intake and the long-term incidence of age-related cataract: The Blue Mountains Eye Study. Am. J. Clin. Nutr. 2008, 87, 1899–1905. [Google Scholar] [PubMed]
- Andley, U.P. Effects of alpha-crystallin on lens cell function and cataract pathology. Curr. Mol. Med. 2009, 9, 887–892. [Google Scholar] [CrossRef] [PubMed]
- Neroev, V.V.; Katargina, L.A.; Kogoleva, L.V. The prevention of blindness and visual impairment in children with retinopathy of prematurity. Curr. pediatrics 2015, 14, 265–270. [Google Scholar] [CrossRef]
- Selin, J.Z.; Rautiainen, S.; Lindblad, B.E.; Morgenstern, R.; Wolk, A. High-dose supplements of vitamins C and E, low-dose multivitamins, and the risk of age-related cataract: A population-based prospective cohort study of men. Am. J. Epidemiol. 2013, 177, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Hennis, A.; Wu, S.Y.; Nemesure, B.; Leske, M.C. Risk factors for incident cortical and posterior subcapsular lens opacities in the Barbados Eye Studies. Arch. Ophthalmol. 2004, 122, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Chew, E.Y.; Clemons, T.E.; Agrón, E.; Launer, L.J.; Grodstein, F.; Bernstein, P.S. Effect of omega-3 fatty acids, lutein/zeaxanthin, or other nutrient supplementation on cognitive function: The AREDS2 randomized clinical trial. Jama J. Am. Med. Assoc. 2015, 314, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Cruickshanks, K.J. Sunlight exposure and risk of lens opacities in a population-based study. Arch. Ophthalmol. 1998, 116, 714. [Google Scholar] [CrossRef]
- Delcourt, C.; Carriere, I.M.; Barberger, G.P.; Schalch, W. Plasma lutein and zeaxanthin and other carotenoids as modifiable risk factors for age-related maculopathy and cataract: the POLA Study. Investig. Ophthalmol. V. Sci. 2006, 47, 2329–2335. [Google Scholar] [CrossRef] [PubMed]
- Le, M.; Lin, X.M. Effects of lutein and zeaxanthin on aspects of eye health. J. Sci. Food Agric. 2010, 90, 2–12. [Google Scholar]
- Lakshminarayana, R.; Aruna, G.; Sathisha, U.V.; Dharmesh, S.M.; Baskaran, V. Structural elucidation of possible lutein oxidation products mediated through peroxyl radical inducer 2,2’-Azobis (2-methylpropionamidine) dihydrochloride: Antioxidant and cytotoxic influence of oxidized lutein in HeLa cells. Chemico-Biol. Interact. 2013, 203, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, R.D.; Vashist, P.; Gupta, S.K.; Young, I.S.; Maraini, G.; Camparini, M.; Jayanthi, R.; John, N.; Fitzpatrick, K.E.; Chakravarthy, U.; et al. Inverse association of vitamin C with cataract in older people in India. Ophthalmology 2011, 118, 1958–1965. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.H.; Yu, R.B.; Liu, R.; Hao, Z.X.; Han, C.C.; Zhu, Z.H.; Ma, L. Association between lutein and zeaxanthin status and the risk of cataract: A Meta-Analysis. Nutrients 2014, 6, 452–465. [Google Scholar] [CrossRef] [PubMed]
- Ghaem, M.H.; Tai, B.C.; Wong, T.Y.; Tai, E.S.; Li, J.; Wang, J.J.; Mitchell, P. Metabolic syndrome and risk of age-related cataract over time: An analysis of interval-censored data using a random-effects model. Investig. Ophthalmol. V. Sci. 2012, 54, 641–646. [Google Scholar] [CrossRef] [PubMed]
- Beebe, D.C.; Holekamp, N.M.; Shui, Y.B. Oxidative damage and the prevention of age-related cataracts. Ophthalmic Res. 2010, 44, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Kui-Yi, X.; Lou, M.F. Effect of age on the thioltransferase (glutaredoxin) and thioredoxin systems in the human lens. Investig. Ophthalmol. Vis. Sci. 2010, 51, 6598–6604. [Google Scholar]
- Gale, C.R.; Hall, N.F.; Phillips, D.I.; Martyn, C.N. Plasma antioxidant vitamins and carotenoids and age-related cataract. Ophthalmology 2001, 108, 1992–1998. [Google Scholar] [CrossRef]
- Koo, E.; Chang, J.R.; Agrón, E.; Clemons, T.E.; Sperduto, R.D.; Ferris III, F.L.; Chew, E.Y. Ten-year incidence rates of age-related cataract in the Age-Related Eye Disease Study (AREDS): AREDS report no. 33. Ophthalmic Epidemiol. 2013, 20, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Mares, J.A.; Voland, R.; Adler, R.; Tinker, L.; Millen, A.E.; Moeller, S.M.; Blodi, B.; Gehrs, K.M.; Wallace, R.B.; Chappell, R.J.; et al. Healthy diets and the subsequent prevalence of nuclear cataract in women. Arch. Ophthalmol. 2010, 128, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Christen, W.G.; Liu, S.; Glynn, R.J.; Gaziano, J.M.; Buring, J.E. Dietary carotenoids, vitamins C and E, and risk of cataract in women: A prospective study. Arch. Ophthalmol. 2008, 126, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Von, L.J. Metabolism of carotenoids and retinoids related to vision. J. Biol. Chem. 2012, 287, 1627–1634. [Google Scholar]
- Ma, L.; Hao, Z.X.; Liu, R.R.; Yu, R.B.; Shi, Q.; Pan, J.P. A dose-response meta-analysis of dietary lutein and zeaxanthin intake in relation to risk of age-related cataract. Graefe’s Arch. Clin. Exp. Ophthalmol. 2014, 252, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; Vishwanathan, R. Relationship between serum and brain carotenoids, α-tocopherol, and retinol concentrations and cognitive performance in the oldest old from the Georgia Centenarian Study. J. Aging Res. 2012, 2013, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Vishwanathan, R.; Kuchan, M.J.; Sen, S.; Johnson, E.J. Lutein is the predominant carotenoid in infant brain: Preterm infants have decreased concentrations of brain carotenoids. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J. Role of lutein and zeaxanthin in visual and cognitive function throughout the lifespan. Nutr. Rev. 2014, 72, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Feeney, J.; Finucane, C.; Savva, G.M.; Cronin, H.; Beatty, S.; Nolan, J.M.; Kenny, R.A. Low macular pigment optical density is associated with lower cognitive performance in a large, population-based sample of older adults. Neurobiol. Aging 2013, 34, 2449–2456. [Google Scholar] [CrossRef] [PubMed]
- Vishwanathan, R.; Iannaccone, A.; Scott, T.M.; Kritchevsky, S.B.; Jennings, B.J.; Carboni, G.; Forma, G.; Satterfield, S.; Harris, T.; Johnson, K.C.; et al. Macular pigment optical density is related to cognitive function in older people. Age Ageing 2014, 43, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.Y.; Cheung, C.Y.; Li, X.; Lamoureux, E.L.; Ikram, M.K.; Ding, J.; Cheng, C.Y.; Haaland, B.A.; Saw, S.M.; Venketasubramanian, N.; et al. Visual impairment, age-related eye diseases, and cognitive function: The Singapore Malay Eye study. Arch. Ophthalmol. 2012, 130, 895–900. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.A.M.; Langa, K.M. Untreated poor vision: A contributing factor to late-life dementia. Am. J. Epidemiol. 2010, 171, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Vishwanathan, R.; Neuringer, M.; Snodderly, D.M.; Schalch, W.; Johnson, E.J. Macular lutein and zeaxanthin are related to brain lutein and zeaxanthin in primates. Nutr. Neurosci. 2013, 16, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; McDonald, K.; Caldarella, S.M.; Chung, H.Y.; Troen, A.M.; Snodderly, D.M. Cognitive findings of an exploratory trial of docosahexaenoic acid and lutein supplementation in older women. Nutr. Neurosci. 2015, 11, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Sale, A.; Berardi, N.; Maffei, L. Enrich the environment to empower the brain. Trends Neurosci. 2009, 32, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Buonocore, G.; Perrone, S.; Bracci, R. Free radicals and brain damage in the newborn. Biol. Neonate 2001, 79, 455–458. [Google Scholar]
- Knickmeyer, R.C.; Gouttard, S.; Kang, C.; Evans, D.; Wilber, K.; Smith, K.; Hamer, R.M.; Lin, W.; Gerig, G.; Gilmore, J.H. A structural MRI study of human brain development from birth to 2 years. J. Neurosci. 2008, 28, 12176–12182. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, D. Macular pigment optical density as a possible biomarker for predicting the effects of lutein and zeaxanthin on cognition among young healthy adults. Diss. Theses Gradworks 2014, 52, 2014–2027. [Google Scholar]
- Devore, E.E.; Grodstein, F.; van Rooij, F.J.; Hofman, A.; Stampfer, M.J.; Witteman, J.C.; Breteler, M.M. Dietary antioxidants and long-term risk of dementia. Arch. Neurol. 2010, 67, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Lindbergh, C.; Terry, D.; Mewborn, C.; Renzi-Hammond, L.; Hammond, B.; Miller, S. Aging and dementia-1lutein and zeaxanthin supplementation improves neurocognitive function in older adults as measured using functional magnetic resonance imaging: A randomized controlled trial. Natl. Acad. Neuropsychol. 2016, 31, 574. [Google Scholar] [CrossRef]
- Kuchan, M.; Wang, F.; Geng, Y.; Feng, B.; Lai, C. Lutein stimulates the differentiation of human stem cells to neural progenitor cells in vitro. Presented at Advances and Controversies in Clinical Nutrition, Washington, DC, USA, 5–7 December 2013. [Google Scholar]
- Terry, D.; Duda, B.; Mewborn, C.; Lindbergh, C.; Bovier, E.; Shon, D.; Puente, A.; Chu, K.; Washington, T.; Stapley, L.; et al. Brain activity associated with verbal learning and recall in older adults and its relationship to lutein and zeaxanthin concentrations. Arch. Clin. Neuropsychol. 2014, 29, 506. [Google Scholar] [CrossRef]
- Hoffmann, K.; Richer, S.; Wrobel, J.; Chen, E.; Podella, C. A prospective study of neuro-cognitive enhancement with carotenoids in elderly adult males with early age related macular degeneration. Ophthalmol. Res. Int. J. 2015, 4, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Fatani, A.J.; Parmar, M.Y.; Abuohashish, H.M.; Ahmed, M.M.; Al-Rejaie, S.S. Protective effect of lutein supplementation on oxidative stress and inflammatory progression in cerebral cortex of streptozotocin-induced diabetes in rats. Neurochem. J. 2016, 10, 69–76. [Google Scholar] [CrossRef]
- Nolan, J.M.; Loskutova, E.; Howard, A.N.; Moran, R.; Mulcahy, R.; Stack, J.; Bolger, M.; Dennison, J.; Akuffo, K.O.; Owens, N. Macular pigment, visual function, and macular disease among subjects with Alzheimer’s disease: An exploratory study. J. Alzheimer’s Dis. 2014, 42, 1191–2002. [Google Scholar]
- Renzi, L.M.; Dengler, M.J.; Puente, A.; Miller, L.S.; Hammond, B.R. Relationships between macular pigment optical density and cognitive function in unimpaired and mildly cognitively impaired older adults. Neurobiol. Aging 2014, 35, 1695–1699. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, J.P.; Hammond, B.R., Jr. Possible influences of lutein and zeaxanthin on the developing retina. Clin. Ophthalmol. 2007, 1, 25–35. [Google Scholar] [PubMed]
- Renzi, L.M.; Bovier, E.R.; Hammond, B.R., Jr. A role for the macular carotenoids in visual motor response. Nutr. Neurosci. 2013, 16, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Crabtree, D.V.; Ojima, I.; Geng, X.; Adler, A.J. Tubulins in the primate retina: evidence that xanthophylls may be endogenous ligands for the paclitaxel-binding site. Bioorg. Med. Chem. 2001, 9, 1967–1976. [Google Scholar] [CrossRef]
- Ozawa, Y.; Sasaki, M.; Takahashi, N.; Kamoshita, M.; Miyake, S.; Tsubota, K. Neuroprotective effects of lutein in the retina. Curr. Pharm. Des. 2012, 18, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Mauer, K.; Mauer, U. Alzheimer. The life of a physician and the career of a disease. Bull. Hist. Med. 2003, 78, 728–730. [Google Scholar]
- Markesbery, W.R. The role of oxidative stress in Alzheimer disease. Arch. Neurol. 1999, 56, 1449–1452. [Google Scholar] [CrossRef] [PubMed]
- Markesbery, W.R.; Lovell, M.A. Damage to lipids, proteins, DNA, and RNA in mild cognitive impairment. JAMA Neurol. 2007, 64, 954–956. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.J.; Maras, J.E.; Rasmussen, H.M.; Tucker, K.L. Intake of lutein and zeaxanthin differ with age, sex, and ethnicity. J. Am. Dietetic Assoc. 2010, 110, 1357–1362. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.; Hankinson, S.E.; Rosner, B.; Willett, W.C.; Colditz, G.A. Prospective study of lutein/zeaxanthin intake and risk of age-related macular degeneration. Am. J. Clin. Nutr. 2008, 87, 1837–1843. [Google Scholar] [PubMed]
- O’Neill, M.E.; Carroll, Y.; Corridan, B.; Olmedilla, B.; Granado, F.; Blanco, I.; Vanden Berg, H.; Hininger, I.; Rousell, A.M.; Chopra, M.; et al. A European carotenoid database to assess carotenoid intakes and its use in a five-country comparative study. Br. J. Nutr. 2001, 85, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Nebeling, L.C.; Forman, M.R.; Graubard, B.I.; Snyder, R.A. Changes in carotenoid intake in the United States: The 1987 and 1992 National Health Interview Surveys. J. Am. Dietetic Assoc. 1997, 97, 991–996. [Google Scholar] [CrossRef]
- Seddon, J.M.; Reynolds, R.; Rosner, B. Associations of smoking, body mass index, dietary lutein, and the LIPC gene variant rs10468017 with advanced age-related macular degeneration. Mol. Vis. 2010, 16, 2412–2424. [Google Scholar] [PubMed]
- Wenzela, A.J.; Sheehanb, J.P.; Burkeb, J.D.; Lefsrudc, M.G.; Curran-Celentanob, J. Dietary intake and serum concentrations of lutein and zeaxanthin, but not macular pigment optical density, are related in spouses. Nutr. Res. 2007, 27, 462–469. [Google Scholar] [CrossRef]
- Granadolorencio, F.; Olmedillaalonso, B.; Blanconavarro, I.; Botellaromero, F.; Simal-Anton, A. Assessment of carotenoid status and the relation to glycaemic control in type I diabetics: A follow-up study. Eur. J. Clin. Nutr. 2006, 60, 1000–1008. [Google Scholar] [CrossRef] [PubMed]
- Ciulla, T.A.; Curran-Celantano, J.; Cooper, D.A.; Hammond, B.R.; Danis, R.P.; Pratt, L.M.; Riccardi, K.A.; Filloon, T.G. Macular pigment optical density in a midwestern sample. Ophthalmology 2001, 108, 730–737. [Google Scholar] [CrossRef]
- Lyle, B.J.; Mares-Perlman, J.A.; Klein, B.E.; Klein, R.; Greger, J.L. Antioxidant intake and risk of incident age-related nuclear cataracts in the Beaver Dam Eye Study. Am. J. Epidemiol. 1999, 149, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.X.; Liu, M.; Zhang, H.Z.; Gu, Y.G.; Xing, Q.Y.; Lai, J.Q. Dietary carotenoid intake in four seasons in urban and rural communities in Jining. J. Nutr. 2009, 31, 6–10. [Google Scholar]
- Wang, Z.; Lin, X. Analysis of dietary lutein and zeaxanthin intake in summer and autumn in Beijing community. Food Nutr. China 2010, 2010, 77–80. [Google Scholar]
- Song, X.; Wang, Z. Analysis of carotenoid intake in adult diet. Chin. Public Health 2007, 23, 1378–1380. [Google Scholar]
- Zhang, F.; Zhu, Y.; Cai, M. Correlation analysis between dietary intake and serum levels of level in a community of Shanghai. Environ. Occup. Med. 2012, 29, 563–565. [Google Scholar]
- Parisi, V.; Tedeschi, M.; Gallinaro, G.; Varano, M.; Saviano, S.; Piermarocchi, S. Carotenoids and antioxidants in age-related maculopathy Italian study: Multifocal electroretinogram modifications after 1 year. Ophthalmology 2008, 115, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Richer, S.; Stiles, W.; Statkute, L.; Pulido, J.; Frankowski, J.; Rudy, D.; Pei, K.; Tsipursky, M.; Nyland, J. Double-masked, placebo-controlled, randomized trial of lutein and antioxidant supplementation in the intervention of atrophic age-related macular degeneration: The Veterans LAST study (Lutein Antioxidant Supplementation Trial). Optometry 2004, 75, 216–230. [Google Scholar] [CrossRef]
- Cangemi, F.E. TOZAL Study: An open case control study of an oral antioxidant and omega-3 supplement for dry AMD. BMC Ophthalmol. 2007, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, J.M.; Kim, J.; de Monastario, F.; Thompson, D.J.; Bone, R.A.; Landrum, J.T.; de Moura, F.F.; Khachik, F.; Chen, H.; Schleicher, R.L.; et al. Dose-ranging study of lutein supplementation in persons aged 60 years or older. Investig. Ophthalmol. Vis. Sci. 2006, 47, 5227–5233. [Google Scholar] [CrossRef] [PubMed]
- Weigert, G.; Kaya, S.; Pemp, B.; Sacu, S.; Lasta, M.; Werkmeister, R.M.; Dragostinoff, N.; Simader, C.; Garhöfer, G.; Schmidt-Erfurth, U.; et al. Effects of lutein supplementation on macular pigment optical density and visual acuity in patients with age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2011, 52, 8174–8178. [Google Scholar] [CrossRef] [PubMed]
- Alves, R.A.; Shao, A. The science behind lutein. Toxicol. Lett. 2004, 150, 57–83. [Google Scholar] [CrossRef] [PubMed]
- Olmedilla, B.; Granado, F.; Blanco, I.; Vaquero, M. Lutein, but not alpha-tocopherol, supplementation improves visual function in patients with age-related cataracts: A 2-y double-blind, placebo-controlled pilot study. Nutrition 2003, 19, 21–24. [Google Scholar] [CrossRef]
- Ribayamercado, J.D.; Blumberg, J.B. Lutein and zeaxanthin and their potential roles in disease prevention. Int. Symp. Eggs Hum. Health 2004, 23, 567S–587S. [Google Scholar]
- Olmedilla, B.; Granado, F.; Blanco, I.; Vaquero, M.; Cajigal, C. Lutein in patients with cataracts and age-related macular degeneration: A long-term supplementation study. J. Sci. Food Agric. 2001, 81, 904–909. [Google Scholar] [CrossRef]
- European Food Safety Authority. Scientific opinion on the re-evaluation of lutein (e 161b) as a food additive. EFSA J. 2010, 8, 1678. [Google Scholar] [CrossRef]
- Joint, F.A.O. Evaluation of Certain Food Additives: Sixty-Third Report of the Joint FAO/WHO Expert Committee on Food Additives; World Health Organization: Geneva, Switzerland, 2005; pp. 23–26. [Google Scholar]
- European Food Safety Authority. Safety, bioavailability and suitability of lutein for the particular nutritional use by infants and young children-Scientific Opinion of the Panel on Dietetic Products, Nutrition and Allergies. EFSA J. 2008, 6, 1–24. [Google Scholar]
- Satia, J.A.; Littman, A.; Slatore, C.G.; Galanko, J.A.; White, E. Long-term use of beta-carotene, retinol, lycopene, and lutein supplements and lung cancer risk: Results from the Vitamins and Lifestyle (VITAL) study. Am. J. Epidemiol. 2009, 170, 401–402. [Google Scholar]
- Institute of Medicine (US) Panel on Dietary Antioxidants and Related Compounds. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academies Press: Washington, DC, USA, 2000. [Google Scholar]
- Olmedilla, B.; Granado, F.; Southon, S.; Wright, A.J.; Blanco, I.; Gil-Martinez, E.; van den BERG, H.; Thurnham, D.; Corridan, B.; Chopra, M.; et al. A European multicentre, placebo-controlled supplementation study with alpha-tocopherol, carotene-rich palm oil, lutein or lycopene: Analysis of serum responses. Clin. Sci. 2002, 102, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Maharshak, N.; Shapiro, J.; Trau, H. Carotenoderma—A review of the current literature. Int. J. Dermatol. 2003, 42, 178–181. [Google Scholar] [CrossRef] [PubMed]
- Alholou, S.; Tucker, W.R.; Agron, E.; Clemons, T.E.; Chew, E.Y. The association of statin use with cataract progression using propensity matching in the Age-Related Eye Disease Study 2 (AREDS2). Investig. Ophthalmol. Vis. Sci. 2014, 55, 1675. [Google Scholar]
- Kelm, M.A.; Flanagan, V.P.; Pawlosky, R.J.; Novotny, J.A.; Clevidence, B.A.; Britz, S.J. Quantitative determination of 13C-labeled and endogenous beta-carotene, lutein, and vitamin A in human plasma. Lipids 2001, 36, 1277–1282. [Google Scholar] [CrossRef] [PubMed]
- Lienau, A.; Glaser, T.; Tang, G.; Dolnikowski, G.G.; Grusak, M.A.; Albert, K. Bioavailability of lutein in humans from intrinsically labeled vegetables determined by LC-APCI-MS. J. Nutr. Biochem. 2003, 14, 663–670. [Google Scholar]
- Burri, B.J.; Park, J.Y.K. Compartmental models of vitamin a and β-carotene metabolism in women. Adv. Exp. Med. Biol. 1998, 445, 225–237. [Google Scholar] [PubMed]
- Yao, L.H.; Liang, Y.X.; Trahanovsky, W.S.; Serfass, R.E.; White, W.S. Use of a 13C tracer to quantify the plasma appearance of a physiological dose of lutein in humans. Lipids 2000, 35, 339–348. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds lutein and zeaxanthin are available from the authors. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, Y.-P.; Sun, L.; Yu, H.-S.; Liang, L.-P.; Li, W.; Ding, H.; Song, X.-B.; Zhang, L.-J. The Pharmacological Effects of Lutein and Zeaxanthin on Visual Disorders and Cognition Diseases. Molecules 2017, 22, 610. https://doi.org/10.3390/molecules22040610
Jia Y-P, Sun L, Yu H-S, Liang L-P, Li W, Ding H, Song X-B, Zhang L-J. The Pharmacological Effects of Lutein and Zeaxanthin on Visual Disorders and Cognition Diseases. Molecules. 2017; 22(4):610. https://doi.org/10.3390/molecules22040610
Chicago/Turabian StyleJia, Yu-Ping, Lei Sun, He-Shui Yu, Li-Peng Liang, Wei Li, Hui Ding, Xin-Bo Song, and Li-Juan Zhang. 2017. "The Pharmacological Effects of Lutein and Zeaxanthin on Visual Disorders and Cognition Diseases" Molecules 22, no. 4: 610. https://doi.org/10.3390/molecules22040610
APA StyleJia, Y.-P., Sun, L., Yu, H.-S., Liang, L.-P., Li, W., Ding, H., Song, X.-B., & Zhang, L.-J. (2017). The Pharmacological Effects of Lutein and Zeaxanthin on Visual Disorders and Cognition Diseases. Molecules, 22(4), 610. https://doi.org/10.3390/molecules22040610





