Sulfur-Doped Carbon Nitride Polymers for Photocatalytic Degradation of Organic Pollutant and Reduction of Cr(VI)
Abstract
:1. Introduction
2. Results and Discussion
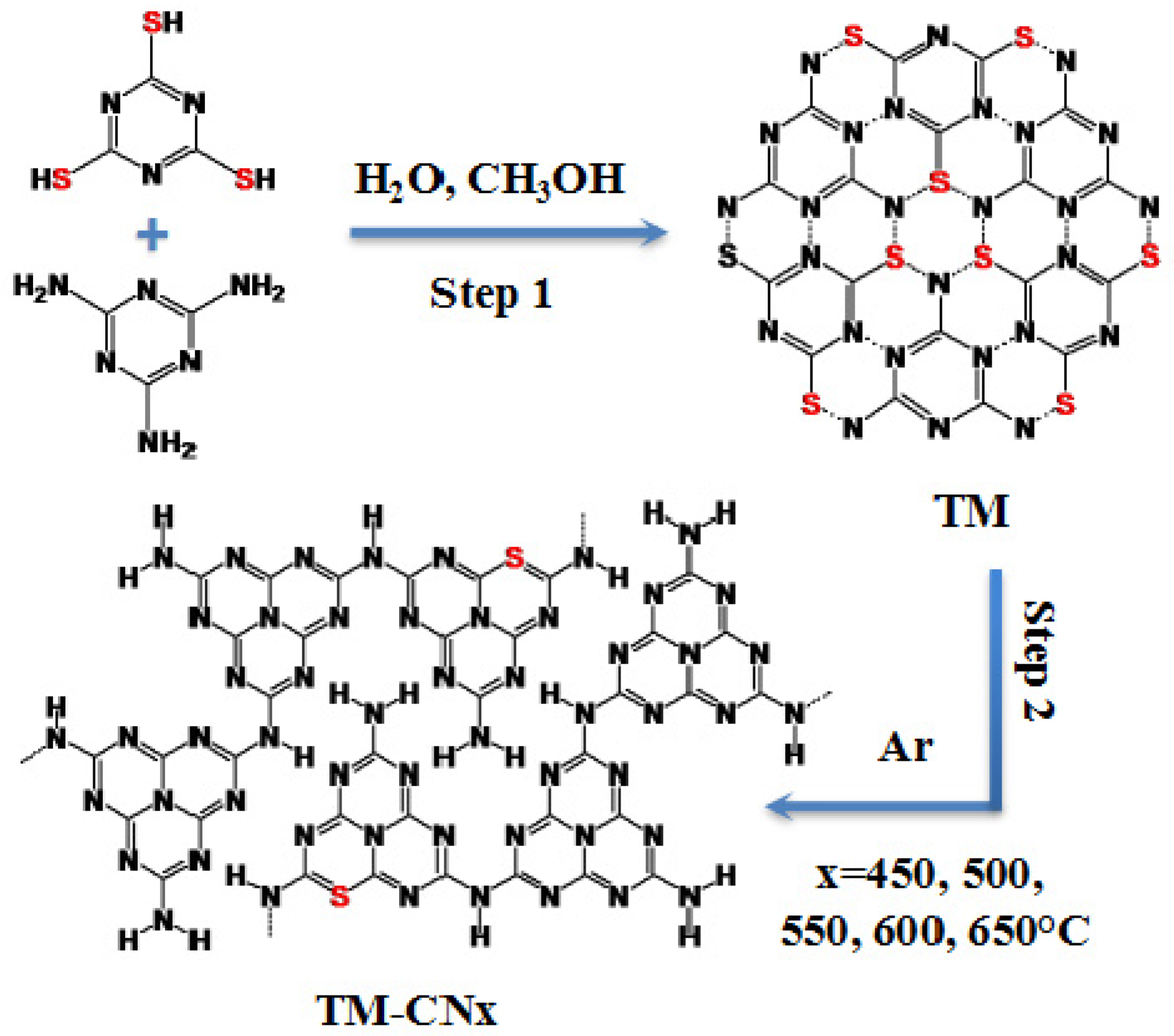
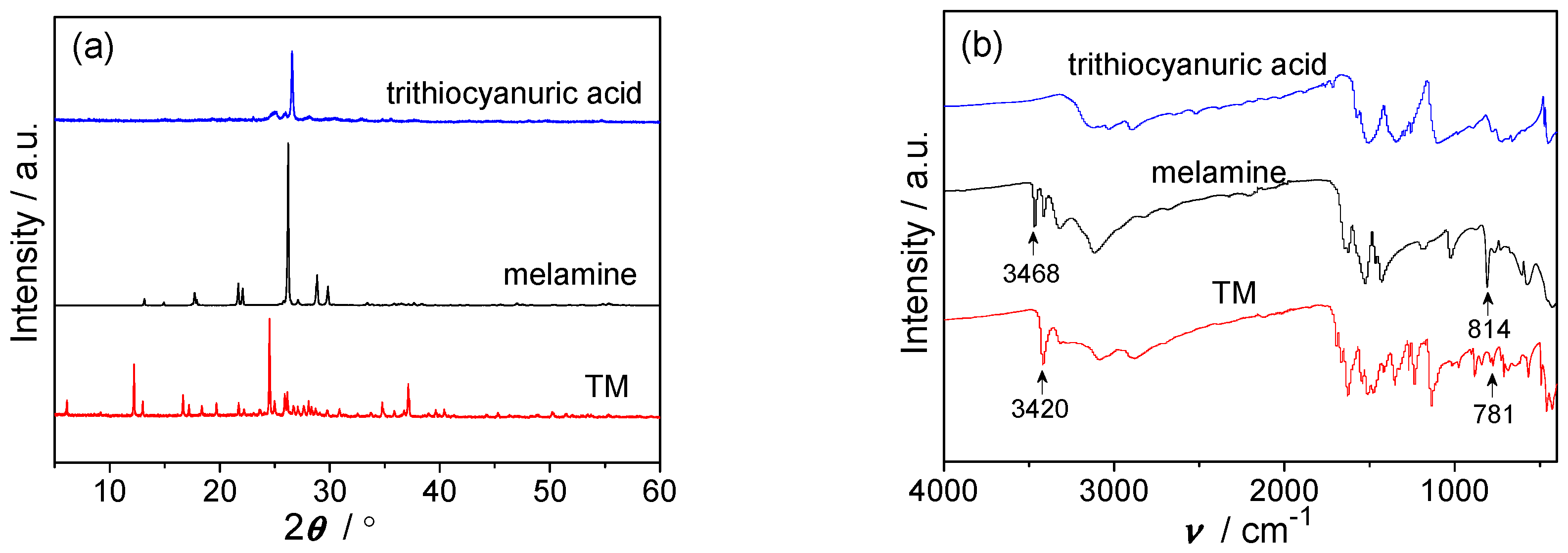
2.1. Characterization of TM Supramolecular Aggregates
2.2. Morphology of TM-CNx
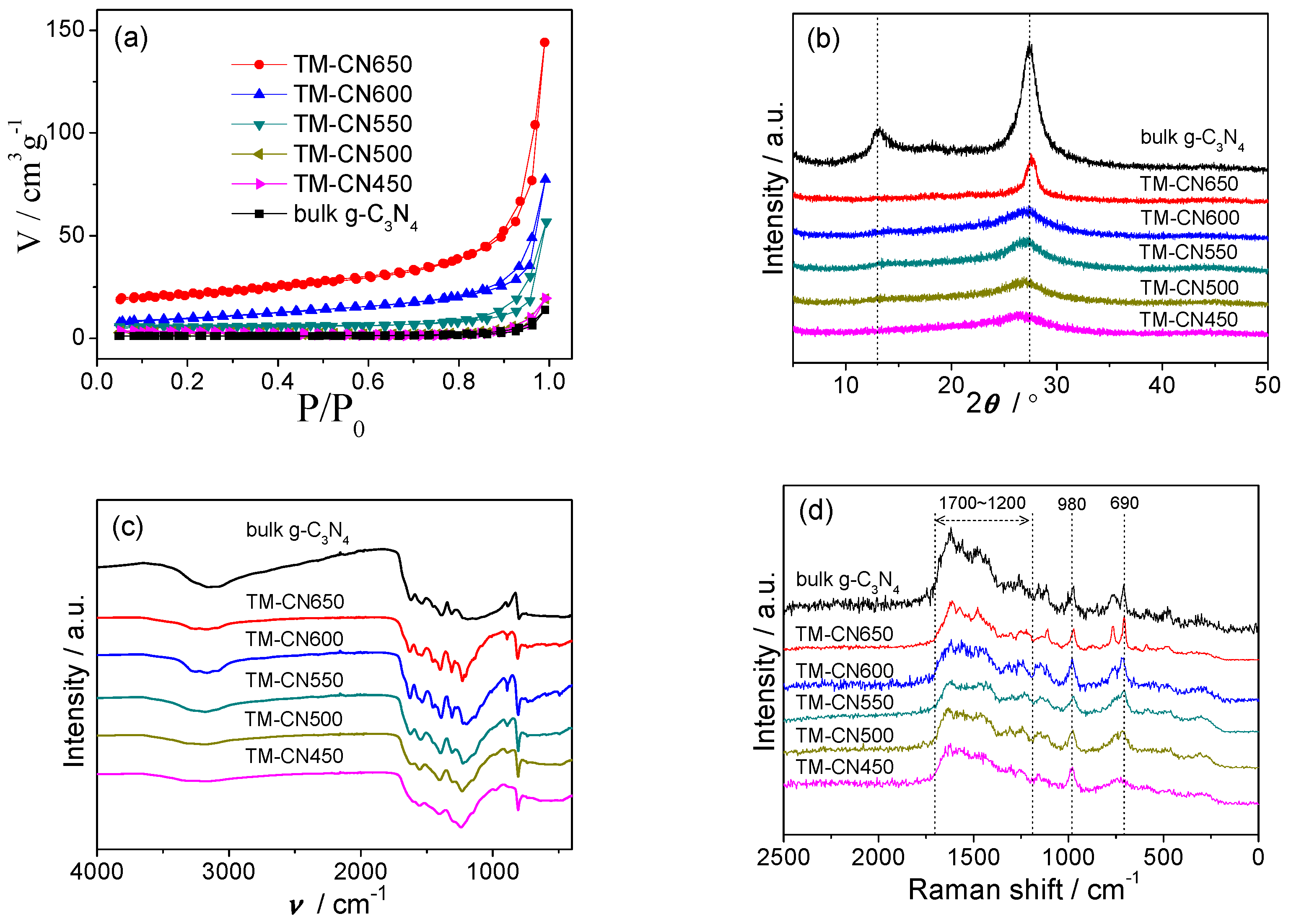
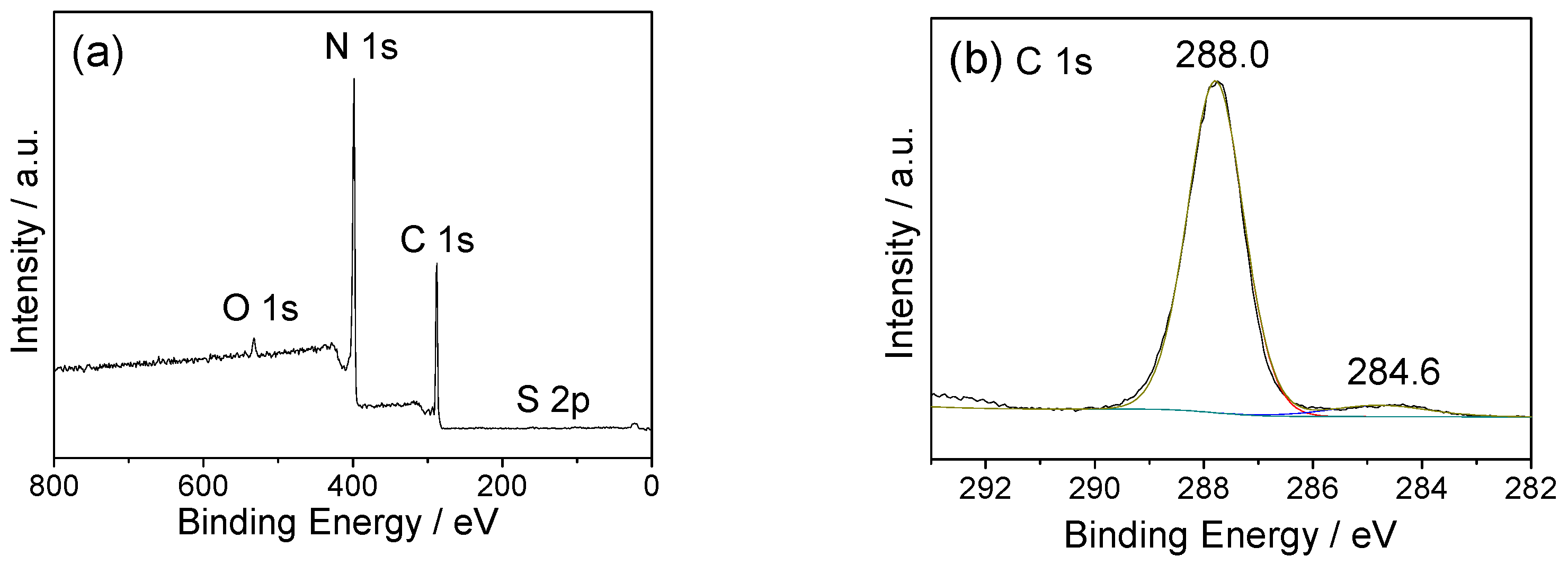
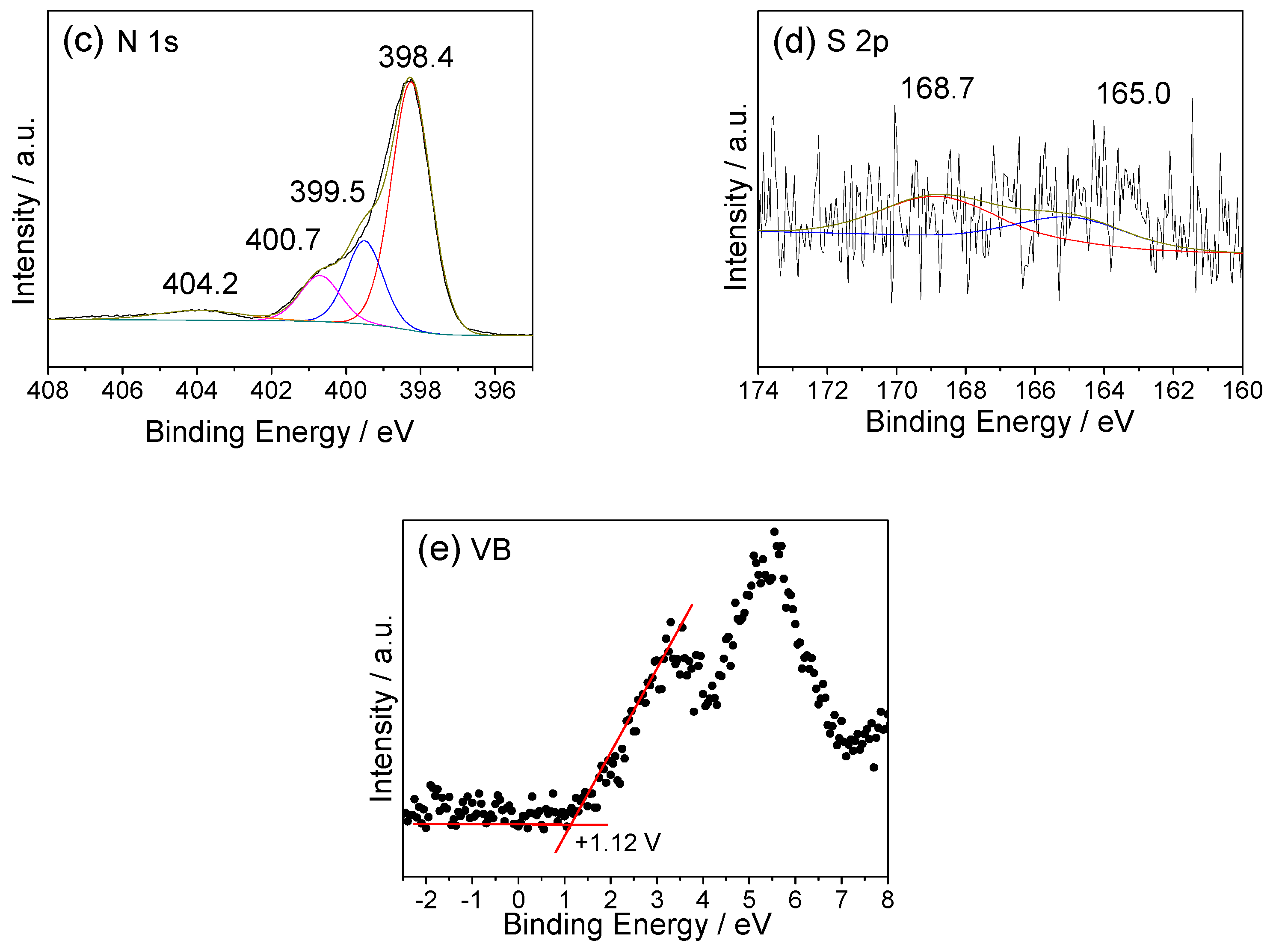
2.3. Texture and Chemical Structure of TM-CNx
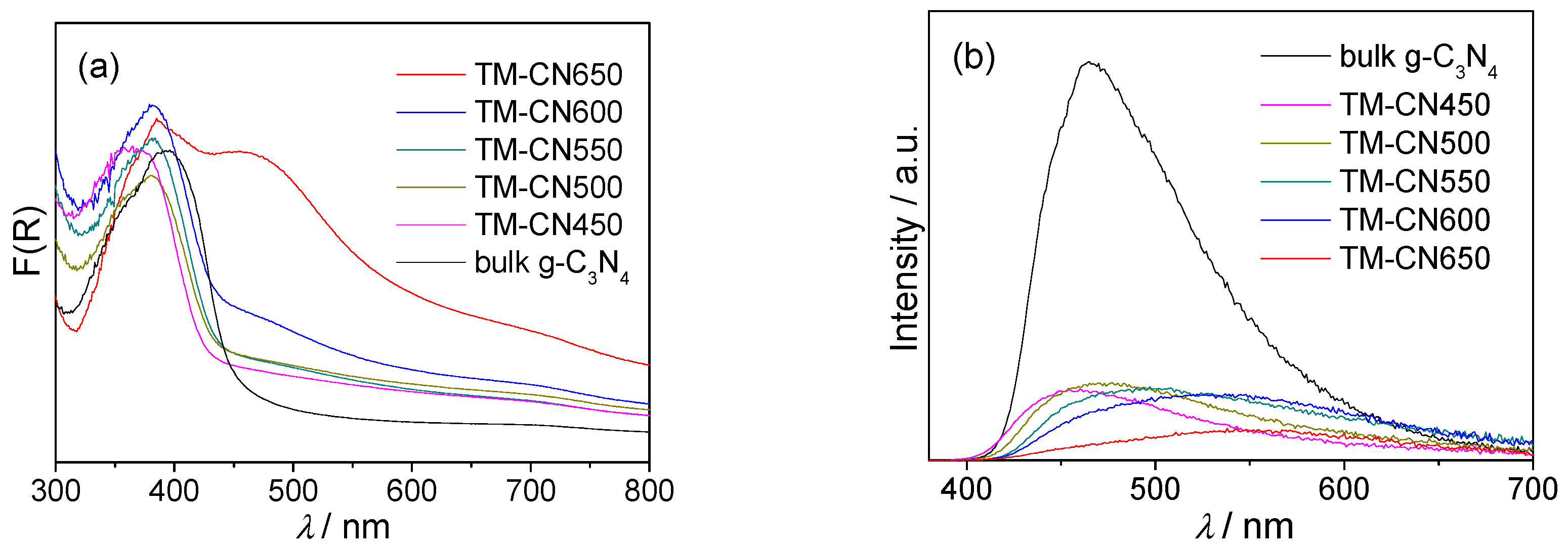
2.4. Optical Property and Band Structure of TM-CNx
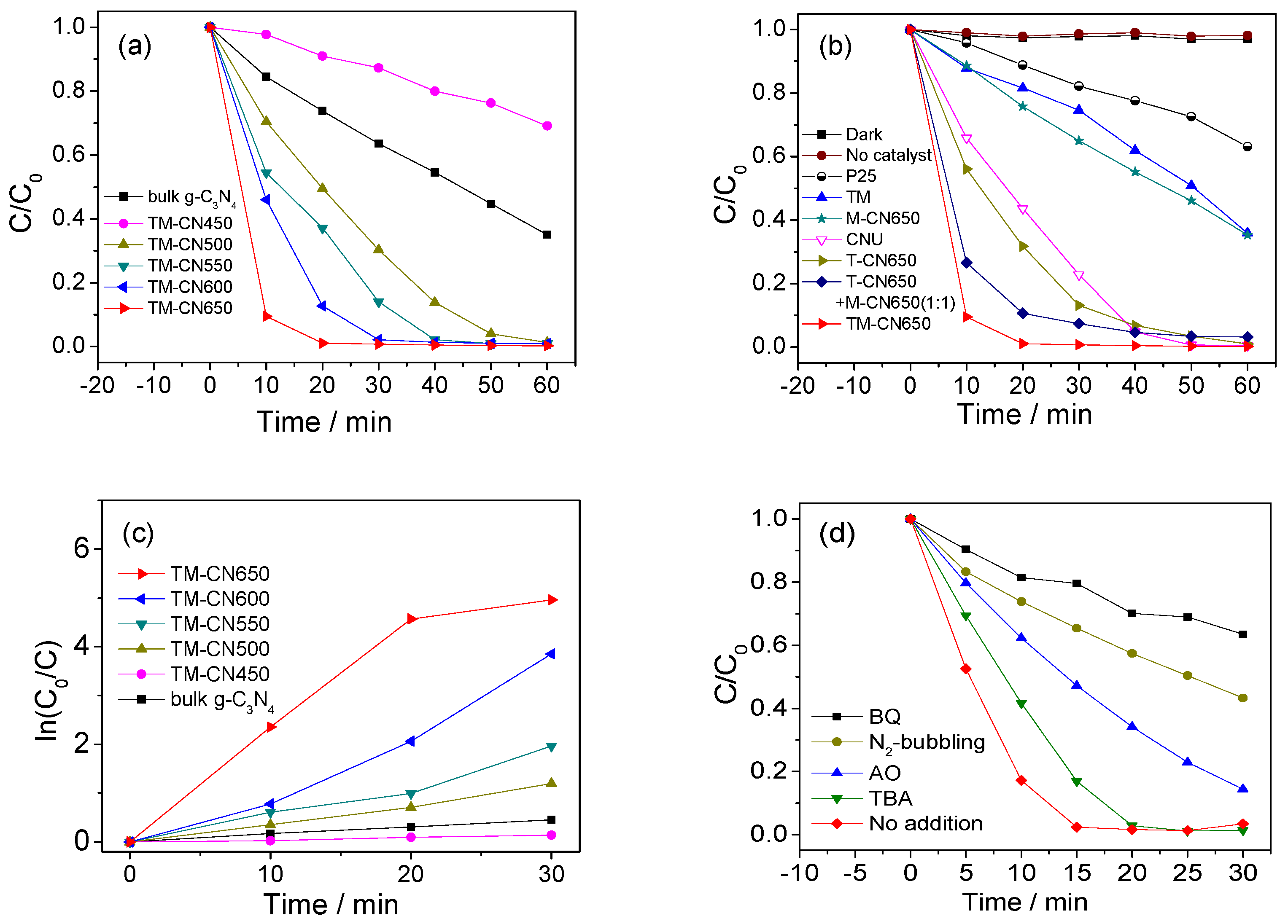
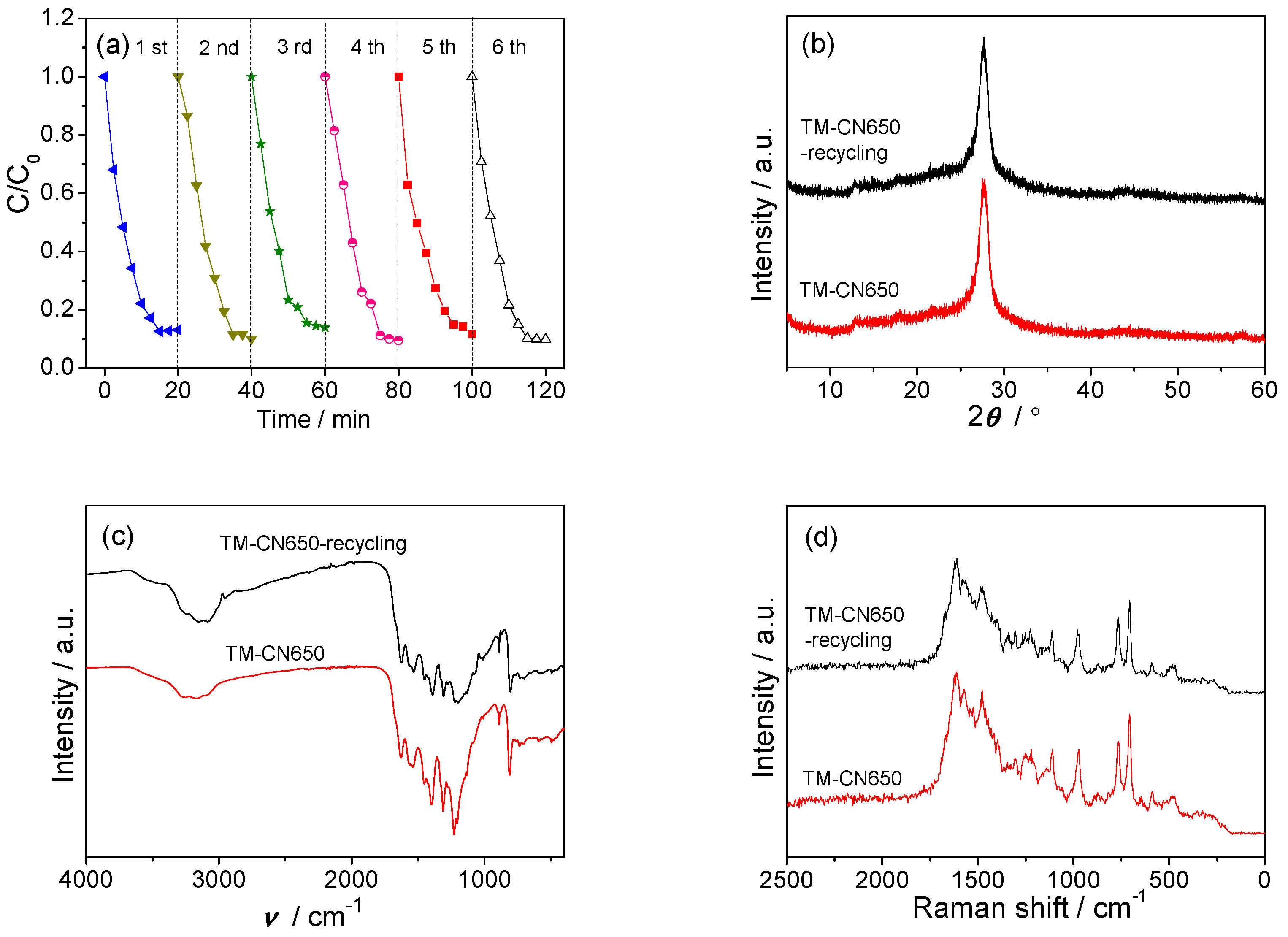
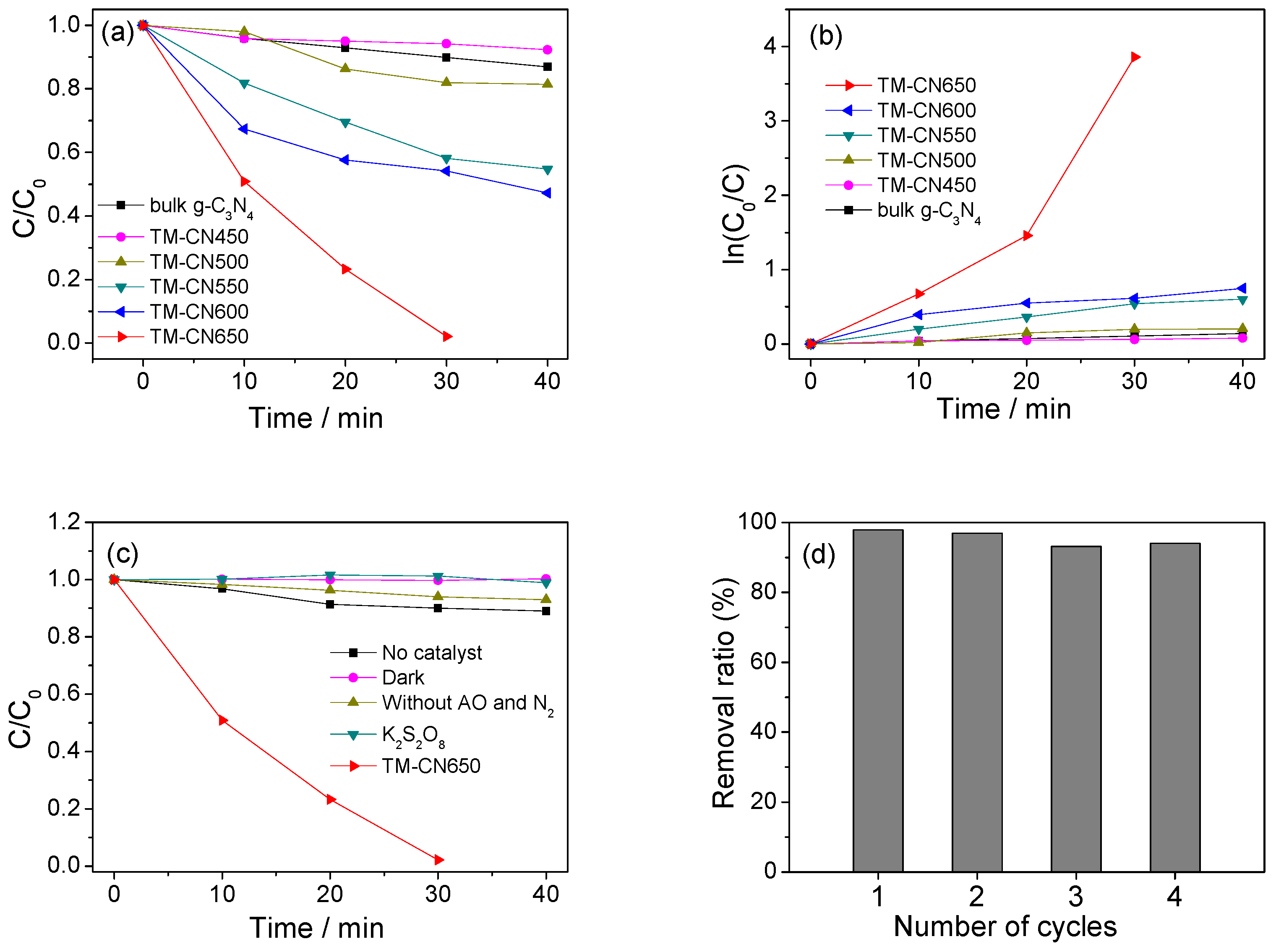
2.5. Photocatalytic Activity of TM-CNx in Degradation of RhB
2.6. Photocatalytic Activity of TM-CNx in Reduction of Cr(VI)
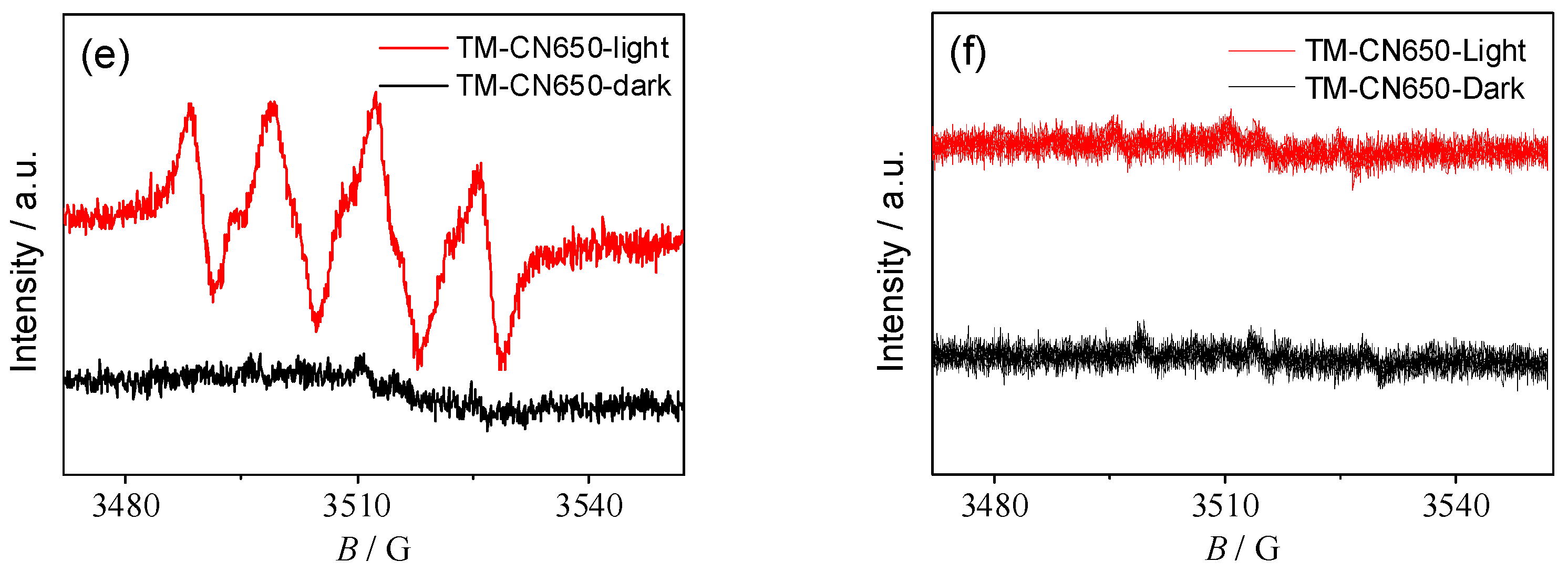
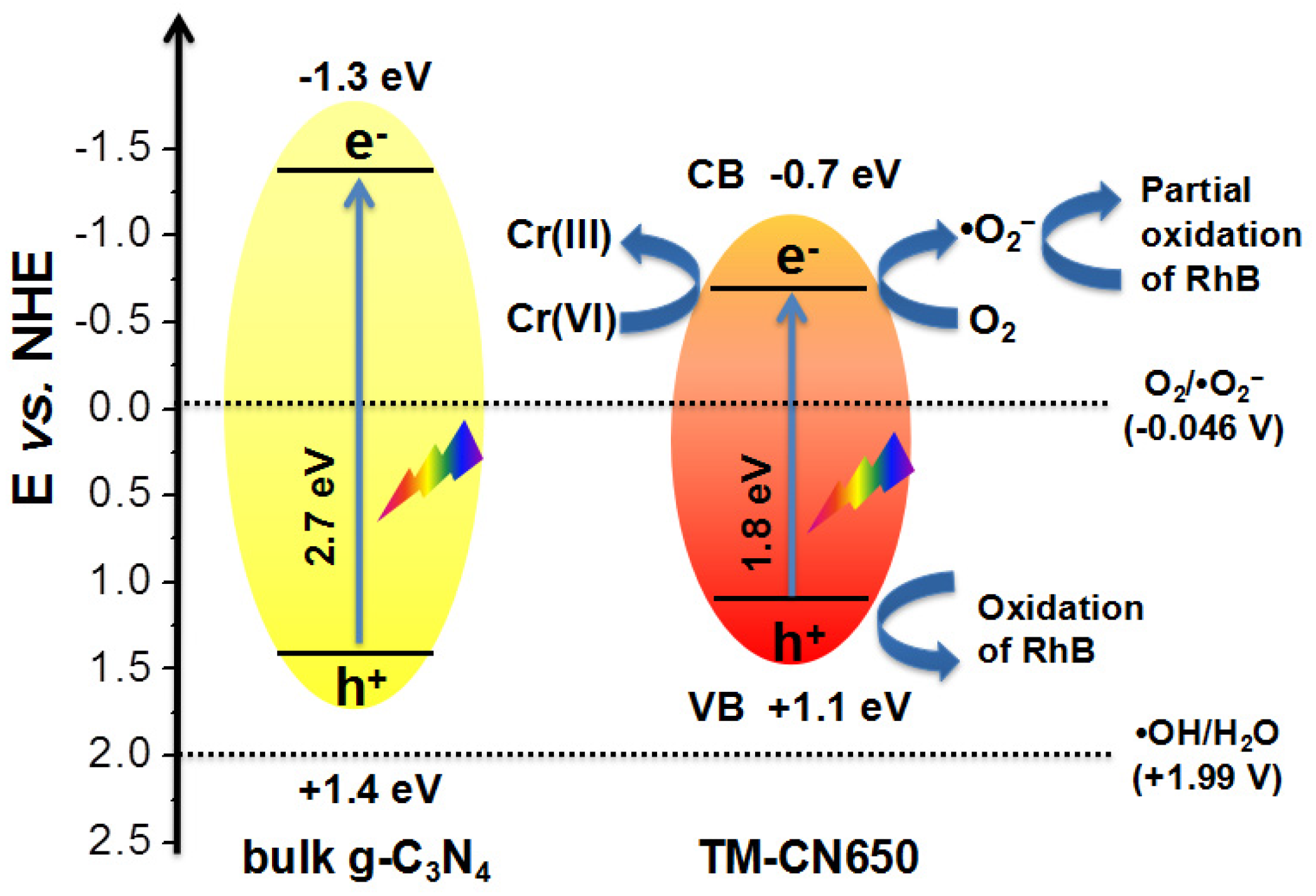
2.7. Mechanism of Photocatalytic Degradation of RhB and Reduction of Cr(VI) over TM-CN650
3. Materials and Methods
3.1. Chemicals
3.2. Synthesis of TM
3.3. Preparation of TM-CNx from TM
3.4. Preparation of Bulk g-C3N4
3.5. Preparation of CNU
3.6. Preparation of T-CN650
3.7. Preparation of M-CN650
3.8. Characterization
3.9. Photocatalytic Degradation of RhB
3.10. Photocatalytic Activity for Reduction of Cr(VI)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pichat, P. (Ed.) Photocatalysis and Water Purification: From Fundamentals to Recent Applications; Wiley-VCH: Weinheim, Germany, 2013; pp. 1–406. [Google Scholar]
- Schneider, J.; Bahnemann, D.; Ye, J.; Puma, G.L.; Dionysiou, D.D. (Eds.) Photocatalysis: Fundamentals and Perspectives; Royal Society of Chemistry: London, UK, 2016; Volume 1. [Google Scholar]
- Dionysiou, D.D.; Puma, G.L.; Ye, J.; Schneider, J.; Bahnemann, D. (Eds.) Photocatalysis: Applications; Royal Society of Chemistry: London, UK, 2016; Volume 2. [Google Scholar]
- Pichat, P. (Ed.) Photocatalysis: Fundamentals, Materials and Potential; MDPI: Basel, Switzerland, 2016. [Google Scholar]
- Colmenares Quintero, J.C.; Xu, Y. (Eds.) Heterogeneous Photocatalysis: From Fundamentals to Green Applications; Springer: Berlin, Germany, 2016. [Google Scholar]
- Nosaka, Y.; Nosaka, A. (Eds.) Introduction to Photocatalysis: From Basic Science to Applications; Royal Society of Chemistry: London, UK, 2016. [Google Scholar]
- Yang, C.; Ma, B.C.; Zhang, L.; Lin, S.; Ghasimi, S.; Landfester, K.; Zhang, K.A.I.; Wang, X.C. Molecular engineering of conjugated polybenzothiadiazoles for enhanced hydrogen production by photosynthesis. Angew. Chem. Int. Ed. 2016, 55, 9202–9206. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.G.; Lan, Z.A.; Lin, L.H.; Lin, S.; Wang, X.C. Overall water splitting by Pt/g-C3N4 photocatalysts without using sacrificial agents. Chem. Sci. 2016, 7, 3062–3066. [Google Scholar] [CrossRef]
- Wang, X.C.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Carlsson, J.M.; Domen, K.; Antonietti, M. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat. Mater. 2009, 8, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.M.; Zheng, Y.; Guo, F.S.; Niu, P.P.; Wang, X.C. Decorating CoP and Pt nanoparticles on graphitic carbon nitride nanosheets to promote overall water splitting by conjugated polymers. ChemSusChem 2017, 10, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.G.; Lan, Z.A.; Wang, X.C. Conjugated polymers: Catalysts for photocatalytic hydrogen evolution. Angew. Chem. Int. Ed. 2016, 55, 15712–15727. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Fang, S.; Zheng, Y.; Sun, J.; Lv, K. Thiourea-modified TiO2 nanorods with enhanced photocatalytic activity. Molecules 2016, 21, 181. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; Zhou, Z.; Wu, X.; Zhu, S.; Bi, J.; Zhou, L.; Liu, M.; Wu, L. Constructing a MoS2 QDs/CdS core/shell flowerlike nanosphere hierarchical heterostructure for the enhanced stability and photocatalytic activity. Molecules 2016, 21, 213. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.D.; Cao, X.N.; Wang, X.C. Precise formation of a hollow carbon nitride structure with a Janus surface to promote water splitting by photoredox catalysis. Angew. Chem. Int. Ed. 2016, 55, 11512–11516. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Lin, L.H.; Ye, X.J.; Guo, F.S.; Wang, X.C. Helical graphitic carbon nitrides with photocatalytic and optical activities. Angew. Chem. Int. Ed. 2014, 53, 11926–11930. [Google Scholar] [CrossRef] [PubMed]
- Long, B.H.; Zheng, Y.; Lin, L.H.; Alamry, K.A.; Asiri, A.M.; Wang, X.C. Cubic Mesoporous Carbon Nitride Polymers with Large Cage Type Pores for Visible Light Photocatalysis. J. Mater. Chem. A 2017. [Google Scholar] [CrossRef]
- Li, X.; Zhang, J.; Chen, X.; Fischer, A.; Thomas, A.; Antonietti, M.; Wang, X.C. Condensed graphitic carbon nitride nanorods by nanoconfinement: Promotion of crystallinity on photocatalytic conversion. Chem. Mater. 2011, 23, 4344–4348. [Google Scholar] [CrossRef]
- Li, X.H.; Wang, X.C.; Antonietti, M. Mesoporous g-C3N4 nanorods as multifunctional supports of ultrafine metal nanoparticles: Hydrogen generation from water and reduction of nitrophenol with tandem catalysis in one step. Chem. Sci. 2012, 3, 2170–2174. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.C.; Antonietti, M.; Zhang, Y. Facile one-pot synthesis of nanoporous carbon nitride solids by using soft templates. ChemSusChem 2010, 3, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Yan, H. Soft-templating synthesis of mesoporous graphitic carbon nitride with enhanced photocatalytic H2 evolution under visible light. Chem. Commun. 2012, 48, 3430–3432. [Google Scholar] [CrossRef] [PubMed]
- Aida, T.; Meijer, E.W.; Stupp, S.I. Functional supramolecular polymers. Science 2012, 335, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhao, Y.; Antonietti, M.; Shalom, M. New organic semiconducting scaffolds by supramolecular preorganization: Dye intercalation and dye oxidation and reduction. Small 2016, 12, 6090–6097. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhu, J.; Yang, X.; Cao, S.; Yu, J.; Shalom, M.; Antonietti, M. Synthesis of organized layered carbon by self-templating of dithiooxamide. Adv. Mater. 2016, 28, 6727–6733. [Google Scholar] [CrossRef] [PubMed]
- Seto, C.T.; Whitesides, G.M. Self-assembly based on the cyanuric acid-melamine lattice. J. Am. Chem. Soc. 1990, 112, 6409–6411. [Google Scholar] [CrossRef]
- Seto, C.T.; Whitesides, G.M. Molecular self-assembly through hydrogen bonding: Supramolecular aggregates based on the cyanuric acid-melamine lattice. J. Am. Chem. Soc. 1993, 115, 905–916. [Google Scholar] [CrossRef]
- Mathias, J.P.; Simanek, E.E.; Zerkowski, J.A.; Seto, C.T.; Whitesides, G.M. Structural preferences of hydrogen-bonded networks in organic solution-the cyclic CA3·M3 “rosette”. J. Am. Chem. Soc. 1994, 116, 4316–4325. [Google Scholar] [CrossRef]
- Ishida, Y.; Chabanne, L.; Antonietti, M.; Shalom, M. Morphology control and photocatalysis enhancement by the one-pot synthesis of carbon nitride from preorganized hydrogen-bonded supramolecular precursors. Langmuir 2014, 30, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Shalom, M.; Guttentag, M.; Fettkenhauer, C.; Inal, S.; Neher, D.; Llobet, A.; Antonietti, M. In situ formation of heterojunctions in modified graphitic carbon nitride: Synthesis and noble metal free photocatalysis. Chem. Mater. 2014, 26, 5812–5818. [Google Scholar] [CrossRef]
- Xu, J.; Brenner, T.J.K.; Chabanne, L.; Neher, D.; Antonietti, M.; Shalom, M. Liquid-based growth of polymeric carbon nitride layers and their use in a mesostructured polymer solar cell with Voc exceeding 1 V. J. Am. Chem. Soc. 2014, 136, 13486–13489. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Deng, Z.; Li, M.; Jiang, B.; Tian, C.; Pan, Q.; Fu, H. Phosphorus-doped carbon nitride tubes with a layered micro-nanostructure for enhanced visible-light photocatalytic hydrogen evolution. Angew. Chem. Int. Ed. 2016, 55, 1830–1834. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Tang, H.; Xu, J.; Antonietti, M.; Shalom, M. Silver phosphate/graphitic carbon nitride as an efficient photocatalytic tandem system for oxygen evolution. ChemSusChem 2015, 8, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Silva, R.; Goswami, A.; Asefa, T. Cu-doped carbon nitride: Bio-inspired synthesis of H2-evolving electrocatalysts using graphitic carbon nitride (g-C3N4) as a host material. Appl. Surf. Sci. 2015, 357, 221–228. [Google Scholar] [CrossRef]
- Cui, Q.; Xu, J.; Wang, X.; Li, L.; Antonietti, M.; Shalom, M. Phenyl-modified carbon nitride quantum dots with distinct photoluminescence behavior. Angew. Chem. Int. Ed. 2016, 55, 3672–3676. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Li, J.; Yuan, Y.; Li, L.; Zhang, M.; Zhou, C.; Lin, Z. A Rapid microwave-assisted thermolysis route to highly crystalline carbon nitrides for efficient hydrogen generation. Angew. Chem. Int. Ed. 2016, 55, 14693–14697. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Brenner, T.J. K.; Chen, Z.; Neher, D.; Antonietti, M.; Shalom, M. Upconversion-agent induced improvement of g-C3N4 photocatalyst under visible light. ACS Appl. Mater. Interfaces 2014, 6, 16481–16486. [Google Scholar] [CrossRef] [PubMed]
- Shalom, M.; Inal, S.; Fettkenhauer, C.; Neher, D.; Antonietti, M. Improving carbon nitride photocatalysis by supramolecular preorganization of monomers. J. Am. Chem. Soc. 2013, 135, 7118–7121. [Google Scholar] [CrossRef] [PubMed]
- Jun, Y.S.; Lee, E.Z.; Wang, X.; Hong, W.H.; Stucky, G.D.; Thomas, A. From melamine-cyanuric acid supramolecular aggregates to carbon nitride hollow spheres. Adv. Funct. Mater. 2013, 23, 3661–3667. [Google Scholar] [CrossRef]
- Li, Y.; Yang, L.; Dong, G.; Ho, W. Mechanism of NO photocatalytic oxidation on g-C3N4 was changed by Pd-QDs modification. Molecules 2016, 21, 36. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, B.; Lin, S.; Zhang, Y.; Wang, X. Activation of n→π* transitions in two-dimensional conjugated polymers for visible light photocatalysis. J. Phys. Chem. C 2014, 118, 29981–29989. [Google Scholar] [CrossRef]
- Ranganathan, A.; Pedireddi, V.R.; Rao, C.N.R. Hydrothermal synthesis of organic channel structures: 1:1 hydrogen-bonded adducts of melamine with cyanuric and trithiocyanuric acids. J. Am. Chem. Soc. 1999, 121, 1752–1753. [Google Scholar] [CrossRef]
- Fan, Q.; Liu, J.; Yu, Y.; Zuo, S.; Li, B. A simple fabrication for sulfur doped graphitic carbon nitride porous rods with excellent photocatalytic activity degrading RhB dye. Appl. Surf. Sci. 2017, 391, 360–368. [Google Scholar] [CrossRef]
- Feng, L.; Zou, Y.; Li, C.; Gao, S.; Zhou, L.; Sun, Q.; Fan, M.; Wang, H.; Wang, D.; Li, G.; Zou, X. Nanoporous sulfur-doped graphitic carbon nitride microrods: A durable catalyst for visible-light-driven H2 evolution. Int. J. Hydrog. Energy 2014, 39, 15373–15379. [Google Scholar] [CrossRef]
- Lan, Z.A.; Zhang, G.G.; Wang, X.C. A facile synthesis of Br-modified g-C3N4 semiconductors for photoredox water splitting. Appl. Catal. B 2016, 192, 116–125. [Google Scholar] [CrossRef]
- Long, B.H.; Lin, J.L.; Wang, X.C. Thermally-induced desulfurization and conversion of guanidine thiocyanate into graphitic carbon nitride catalysts for hydrogen photosynthesis. J. Mater. Chem. A 2014, 2, 2942–2951. [Google Scholar] [CrossRef]
- Wang, Y.; Lin, F.; Peng, J.; Dong, Y.; Li, W.; Huang, Y. A robust bilayer nanofilm fabricated on copper foam for oil-water separation with improved performances. J. Mater. Chem. A 2016, 4, 10294–10303. [Google Scholar] [CrossRef]
- Sun, J.; Xu, J.; Grafmueller, A.; Huang, X.; Liedel, C.; Algara-Siller, G.; Willinger, M.; Yang, C.; Fu, Y.; Wang, X.C.; et al. Self-assembled carbon nitride for photocatalytic hydrogen evolution and degradation of p-nitrophenol. Appl. Catal. B 2017, 205, 1–10. [Google Scholar] [CrossRef]
- Liang, R.; Jing, F.; Shen, L.; Qin, N.; Wu, L. MIL-53(Fe) as a highly efficient bifunctional photocatalyst for the simultaneous reduction of Cr(VI) and oxidation of dyes. J. Hazard. Mater. 2015, 287, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Rao, T.N.; Tryk, D.A. Titanium dioxide photocatalysis. J. Photochem. Photobiol. C 2000, 1, 1–21. [Google Scholar] [CrossRef]
- He, Y.; Cai, J.; Li, T.; Wu, Y.; Lin, H.; Zhao, L.; Luo, M. Efficient degradation of RhB over GdVO4/g-C3N4 composites under visible-light irradiation. Chem. Eng. J. 2013, 215–216, 721–730. [Google Scholar] [CrossRef]
- Sawyer, D.T.; Valentine, J.S. How super is superoxide? Acc. Chem. Res. 1981, 14, 393–400. [Google Scholar] [CrossRef]
- Cui, Y.; Ding, Z.; Liu, P.; Antonietti, M.; Fu, X.; Wang, X. Metal-free activation of H2O2 by g-C3N4 under visible light irradiation for the degradation of organic pollutants. Phys. Chem. Chem. Phys. 2012, 14, 1455–1462. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.C.; Li, Z.S.; Zou, Z.G. Photodegradation of rhodamine B and methyl orange over boron-doped g-C3N4 under visible light irradiation. Langmuir 2010, 26, 3894–3901. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Pan, Z.M.; Wang, X.C. Advances in photocatalysis in China. Chin. J. Catal. 2013, 34, 524–535. [Google Scholar] [CrossRef]
- Zheng, Y.; Lin, L.H.; Wang, B.; Wang, X.C. Graphitic carbon nitride polymers toward sustainable photoredox catalysis. Angew. Chem. Int. Ed. 2015, 54, 12868–12884. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.H.; Ou, H.H.; Zhang, Y.; Wang, X.C. Tri-s-triazine-based crystalline graphitic carbon nitrides for highly efficient hydrogen evolution photocatalysis. ACS Catal. 2016, 6, 3921–3931. [Google Scholar] [CrossRef]
- Yuan, X.; Wang, Y.; Wang, J.; Zhou, C.; Tang, Q.; Rao, X. Calcined graphene/MgAl-layered double hydroxides for enhanced Cr(VI) removal. Chem. Eng. J. 2013, 221, 204–213. [Google Scholar] [CrossRef]
- Lazaridis, N.K.; Asouhidou, D.D. Kinetics of sorptive removal of chromium(VI) from aqueous solutions by calcined Mg-Al-CO3 hydrotalcite. Water Res. 2003, 37, 2875–2882. [Google Scholar] [CrossRef]
Sample Availability: Samples of the sulfur-doped carbon nitride microrods and bulk g-C3N4 are available from the authors. |


| Entry | Samples | Surface Area (m2 g−1) | Pore Volume (cm3 g−1) | C (wt. %) | N (wt. %) | H (wt. %) | S (wt. %) | Molar ratio C/N (%) |
|---|---|---|---|---|---|---|---|---|
| 1 | bulk g-C3N4 | 3 | 0.01 | 34.1 | 58.6 | 2.0 | - | 0.68 |
| 2 | TM-CN450 | 8 | 0.01 | 32.1 | 56.4 | 2.3 | <0.5 | 0.66 |
| 3 | TM-CN500 | 9 | 0.01 | 33.1 | 57.3 | 2.1 | <0.5 | 0.67 |
| 4 | TM-CN550 | 17 | 0.01 | 33.4 | 57.1 | 2.3 | <0.5 | 0.68 |
| 5 | TM-CN600 | 35 | 0.04 | 32.8 | 55.6 | 2.2 | <0.5 | 0.69 |
| 6 | TM-CN650 | 72 | 0.06 | 32.8 | 55.8 | 2.2 | <0.5 | 0.69 |
| Entry | Samples | Absorption Band Edge (nm) | Band Gap Energy (eV) | kRhB (min−1) | kCr(VI) (min−1) |
|---|---|---|---|---|---|
| 1 | bulk g-C3N4 | 460 | 2.70 | 0.0152 | 0.0036 |
| 2 | TM-CN450 | 442 | 2.81 | 0.0047 | 0.0020 |
| 3 | TM-CN500 | 452 | 2.74 | 0.0352 | 0.0066 |
| 4 | TM-CN550 | 456 | 2.72 | 0.0496 | 0.0181 |
| 5 | TM-CN600 | 463 | 2.68 | 0.1031 | 0.0204 |
| 6 | TM-CN650 | 680 | 1.82 | 0.2283 | 0.1287 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, Y.; Yu, Z.; Lin, F.; Guo, F.; Alamry, K.A.; Taib, L.A.; Asiri, A.M.; Wang, X. Sulfur-Doped Carbon Nitride Polymers for Photocatalytic Degradation of Organic Pollutant and Reduction of Cr(VI). Molecules 2017, 22, 572. https://doi.org/10.3390/molecules22040572
Zheng Y, Yu Z, Lin F, Guo F, Alamry KA, Taib LA, Asiri AM, Wang X. Sulfur-Doped Carbon Nitride Polymers for Photocatalytic Degradation of Organic Pollutant and Reduction of Cr(VI). Molecules. 2017; 22(4):572. https://doi.org/10.3390/molecules22040572
Chicago/Turabian StyleZheng, Yun, Zihao Yu, Feng Lin, Fangsong Guo, Khalid A. Alamry, Layla A. Taib, Abdullah M. Asiri, and Xinchen Wang. 2017. "Sulfur-Doped Carbon Nitride Polymers for Photocatalytic Degradation of Organic Pollutant and Reduction of Cr(VI)" Molecules 22, no. 4: 572. https://doi.org/10.3390/molecules22040572
APA StyleZheng, Y., Yu, Z., Lin, F., Guo, F., Alamry, K. A., Taib, L. A., Asiri, A. M., & Wang, X. (2017). Sulfur-Doped Carbon Nitride Polymers for Photocatalytic Degradation of Organic Pollutant and Reduction of Cr(VI). Molecules, 22(4), 572. https://doi.org/10.3390/molecules22040572








