DENV particles are about 50 nm in diameter. The DENV RNA genome of 10,723 nucleotides encodes an uninterrupted open reading frame, which directs the synthesis of the polyprotein precursor NH2-C-prM-E-NS1-NS2A-NS2B-NS3-NS4A-NS4B-NS5-COOH, where C is the capsid protein, M is a membrane-associated protein, E is the envelope protein and NS1-NS5 are nonstructural proteins [
25,
26,
27].
3.1. NS3
NS3 is a multifunctional enzyme, which acts as a protease for polyprotein processing, an RNA triphosphatase for capping nascent viral RNA and a helicase, along with cofactor NS2B, for unwinding the double-stranded, replicative form of RNA [
28]. NS3 is sufficiently conserved within the four DENV serotypes [
28]. Dissecting its structural and functional domains is thus critical to improving the understanding of the flavivirus life cycle and assisting in the design of effective antiviral drugs. Furthermore, the conservation of domain structures across the Flaviviridae family suggests conservation of functions [
22,
26].
The NS3 protease is required for the production of mature viruses and plays a key role in maintaining infectivity. This enzyme mediates the cleavage of polyproteins into functional proteins that are required for viral propagation. The dimeric protease NS2B-NS3 is responsible for protein processing at the junctions of NS2A/NS2B, NS2B/NS3, NS3/NS4A and NS4B/NS5, as well as internal DENV sites within C, 2A, NS3 and NS4A, thus making the nonstructural NS2B-NS3 protease an ideal target for drug design against DENV infection [
29,
30,
31].
The helicase domain of NS3 has seven structural motifs that are reminiscent of superfamily 2 helicases. It has three subdomains with significant sequence identity and structural similarity to other flavivirus helicases. Subdomains I and II are also structurally similar to the corresponding domains in the hepatitis C virus, suggesting a common mechanism of action [
26,
32,
33,
34].
The combined activities of the polynucleotide-stimulated helicase and nucleoside triphosphatase (NTPase) regions in the C-terminal domain are required for both melting secondary structures prior to the initiation of RNA synthesis and unwinding RNA duplexes; these actions occur by either separating double-stranded RNA intermediates that are formed during viral RNA synthesis or removing proteins bound to viral RNA using a translocase function. DENV with impaired helicase activity loses its ability to replicate, demonstrating the importance of the NS3 helicase domain in the viral life cycle. For this reason, inhibitors or modulators of these enzymes are of great interest as therapeutic agents [
26,
35,
36,
37].
3.2. NS5
NS5 is the largest (104 kDa) and most conserved protein in DENV. Specifically, residues 320–368 are strictly conserved among the flaviviruses. These residues have also been implicated in the interaction between NS5 and NS3 [
38,
39]. The important role of NS5 in DENV replication makes these proteins interesting targets for virus inhibition [
26]. Similar to NS3, NS5 possesses two major activities, an RNA-dependent RNA polymerase (RdRp; residues 320–900) at its C-terminal end and a methyltransferase (MTase; residues 1–296) at its N-terminal end [
26,
40]. The NS5 MTase is a surface polyprotein that is essential for the attachment of the virus to the host cell. Thus, the ligand-bound crystal structure of the N-terminal domain of the NS5 MTase has become a crucial tool for current drug discovery efforts. The crystal structure of the RdRp shows an active site with two zinc ion-binding motifs, which are ideal targets for designing novel RdRp inhibitors [
4,
41,
42].
The NS5 RdRp plays a vital role in the replication of viral RNA. Following viral entry and translation of the viral genome, the NS5 RdRp performs de novo RNA synthesis, generating negative-sense RNA from the positive-sense viral RNA template. The latter then serves as a template for the synthesis of more positive-sense RNA strands, which are either used for protein translation or packaged into infectious virions [
43,
44,
45].
The flavivirus MTase catalyzes two very distinct methylation reactions, but there is only a single binding site for the methyl donor. It is likely that the RNA substrate repositions itself after the first reaction so that the second reaction can proceed [
45,
46].
Natural compounds that display inhibitory effects against DENV enzymes are described below. The evaluation of a compound’s activity against viral enzymes can be investigated in two ways. First, the effect of a substance can be assessed directly on the enzyme (enzymatic assay). Second, the evaluation can be carried out in replicon cell lines (which are unable to produce infectious particles, but are capable of RNA replication) using a virus strain (post-treatment assay). For the post-treatment assay, host cells are initially infected with viruses, which can attach to and enter the cell. Then, the viruses that did not attach to the host cells are removed. Finally, the compound under investigation is added. An observation of virus inhibition indicates that the compound acts on the virus replication process, which involves several enzymes. The compound is thus assumed to act on one or more viral enzymes. Molecular docking is another important approach that has been used to investigate inhibitors of viral enzymes. The results from molecular docking experiments and in vitro analyses help streamline the screening processes.
3.3. Direct Inhibitory Activities of Natural Compounds against DENV Enzymes
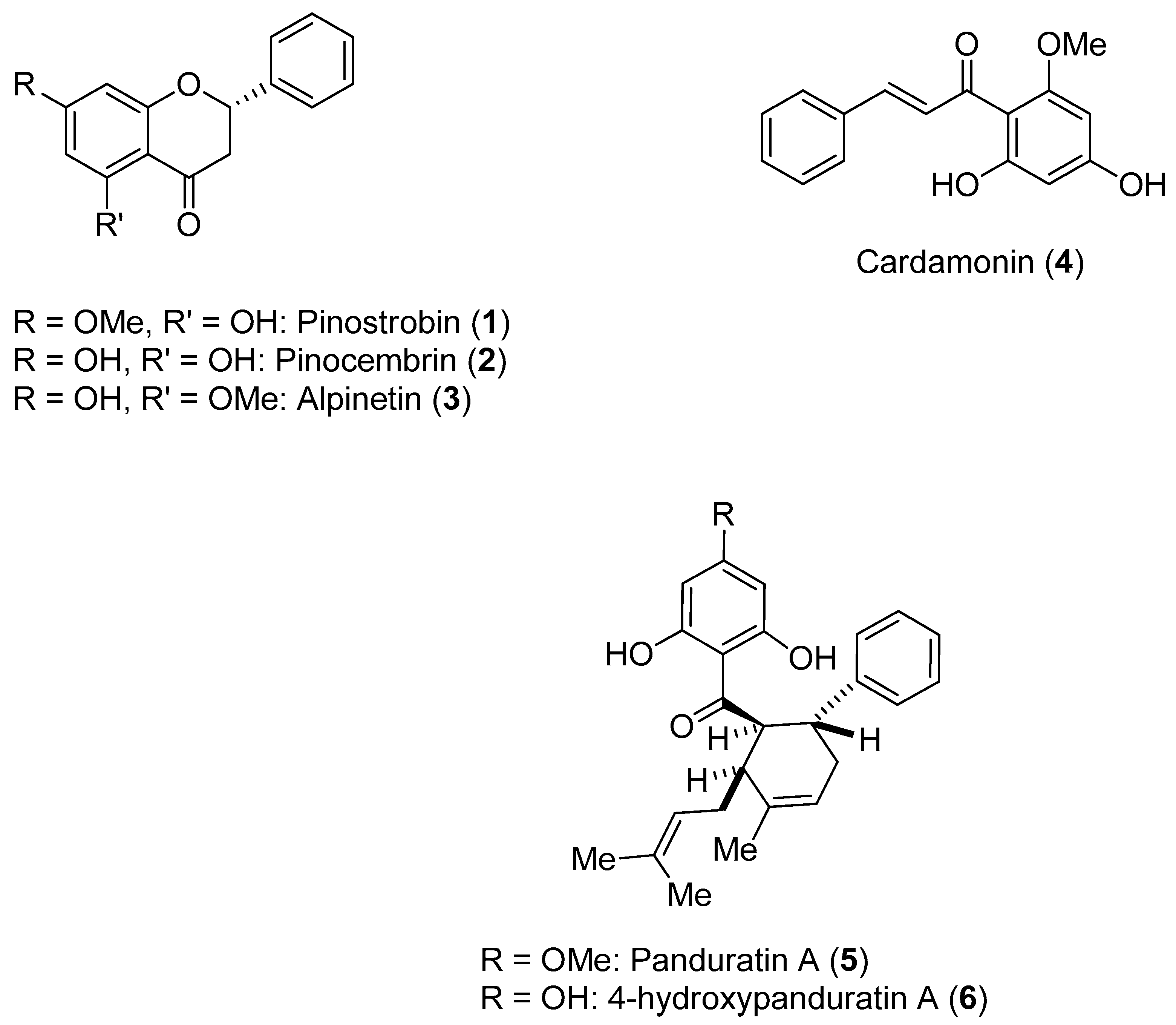
Boesenbergia rotunda (L.) Mansf. Kulturpfl (Br) is a member of the ginger family. A phytochemical investigation of yellow rhizomes of this species from Thailand led to the isolation of six compounds, which are shown in
Figure 1.
These compounds were assayed against the DENV-2 NS3 protease [
47]. The enzymatic assays were carried out using the fluorogenic peptide Boc-Gly-Arg-Arg-MCA, which is an active substrate of the DENV-2 NS3 protease. Initially, the activities of the six compounds against NS3 were evaluated at three different concentrations (120, 240 and 400 ppm); all compounds inhibited NS3 protease activity in a concentration-dependent manner. The most active compounds were
5 and
6. The activities of these compounds were further evaluated at 40, 80 and 160 ppm. The inhibitory activity of Compound
5 ranged from 27.1% (40 ppm)–99.8% (400 ppm), whereas the inhibitory activity of Compound
6 ranged from 52.0% (40 ppm)–99.8% (400 ppm). Individually, Compounds
2 and
4 were found to have low inhibitory activity. However, when these flavones were combined, an increase in inhibitory activity was noticed (synergistic effect). Kinetic studies carried out with Compounds
1,
4,
5 and
6 resulted in the determination of their K
i values (345 for
1; 377 for
4; 25 for
5; and 21 for
6). Compounds
1 and
4 appeared to function via a noncompetitive mechanism, whereas Compounds
5 and
6 displayed competitive inhibition [
47].
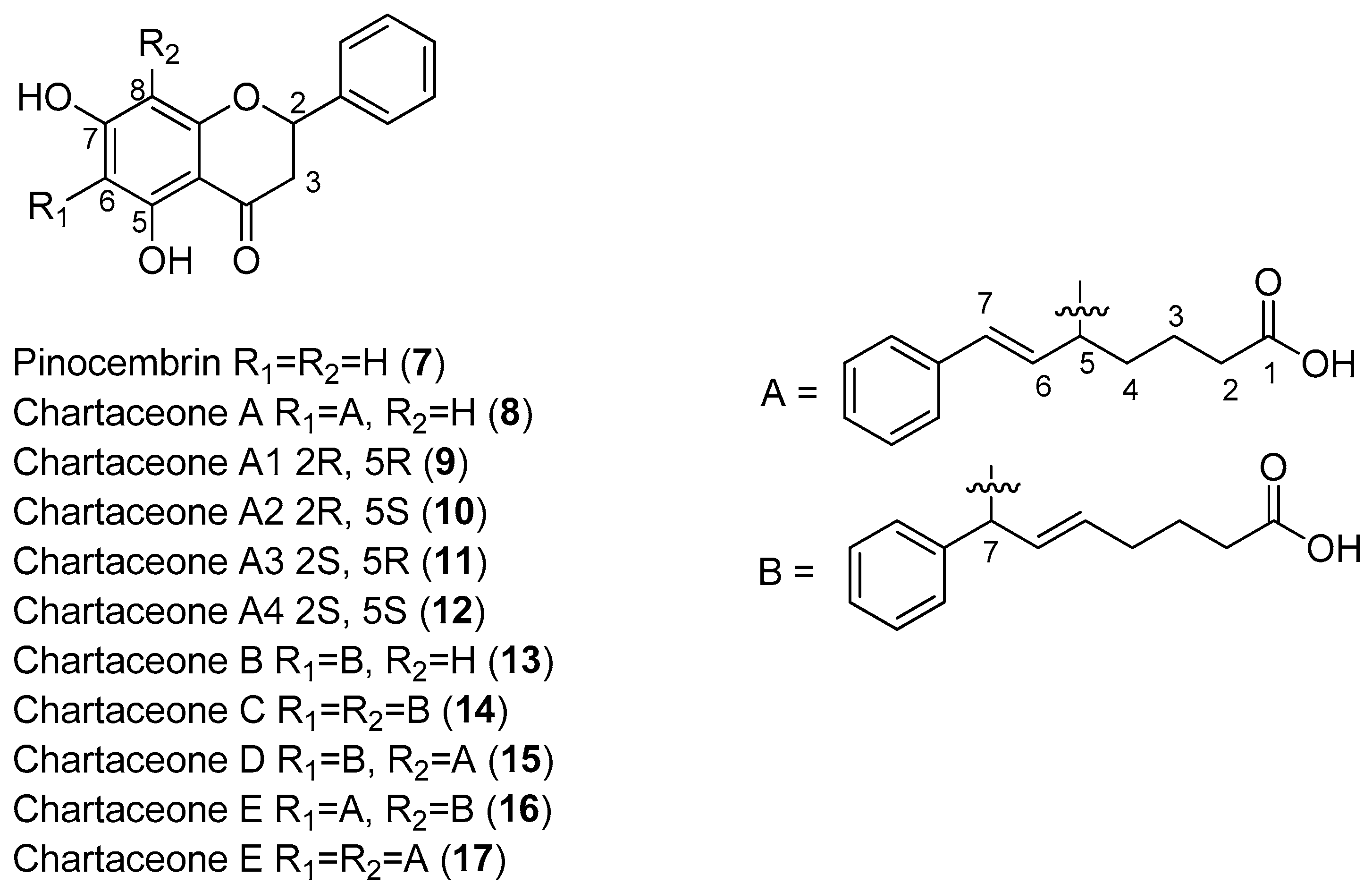
Litaudon and co-workers biologically screened 1350 ethyl acetate extracts obtained from various parts of 650 New Caledonian plants. The extracts were screened at a concentration of 50 μg/mL in a DENV polymerase assay using the RdRp domain of DENV-2 NS5. A second screen of 320 active extracts at a concentration of 10 μg/mL was then performed, which resulted in the selection of 49 extracts exhibiting enzymatic inhibition of at least 80%. An extract from the bark of
Cryptocarya chartacea displayed significant enzymatic inhibitory activity (90% at 10 μg/mL) and was subsequently selected for fractionation. Flavonoids
7–
17 (
Figure 2) were isolated from this extract [
48].
As can be seen in
Figure 2, flavonoids
8–
17 inhibited the DENV NS5 RdRp, with IC
50 values ranging from 1.8 μM (Compound
15)–72.5 μM (Compound
13). Pinocembrin (
7) was found to be inactive against the DENV-2 NS5 RdRp, suggesting that the aliphatic portion of the chartaceone structure plays an important role in the inhibition of the polymerase. In this investigation, the authors also evaluated the effects of the isolated compounds on nasopharyngeal carcinoma cells. No compounds were found to be toxic to this cell line at a concentration of 10 μg/mL [
48].
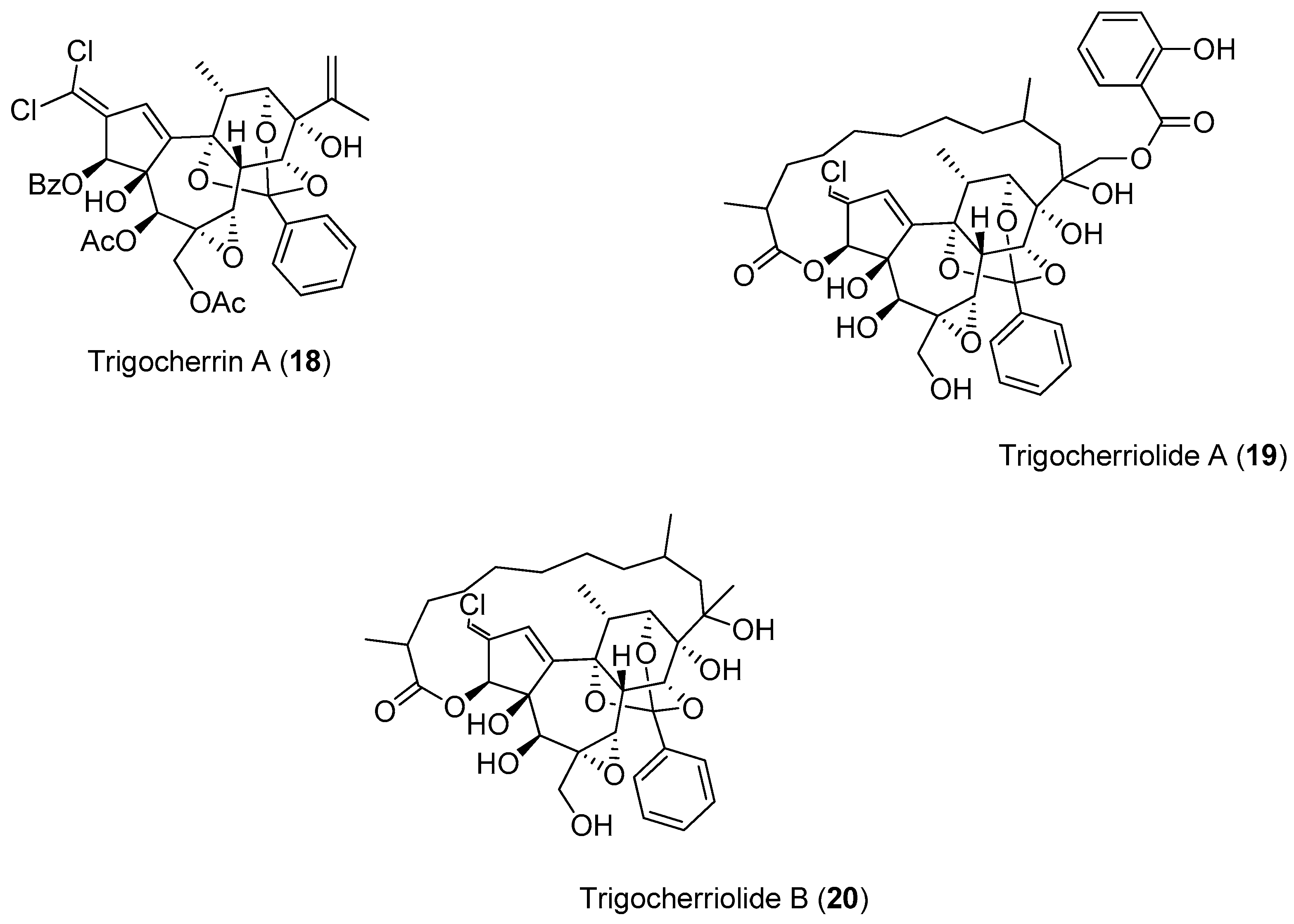
The investigation of the ethyl acetate extracts of the bark and wood of
Trigonostemon cherrieri, a rare endemic plant from New Caledonia, resulted in the isolation of a series of very complex, oxygenated diterpenoids, which included Compounds
18–
20. These compounds were screened against the DENV NS5 RdRp (
Figure 3).
Compounds
18–
20 were capable of inhibiting the DENV NS5 RdRp. Compound
19 was the most potent (IC
50 = 3.1 μM), with an inhibitory activity more than 150-times lower than 3-deoxy-Guanosine Triphosphate (3-deoxy-GTP) (0.02 μM), which was used as the reference compound [
49].
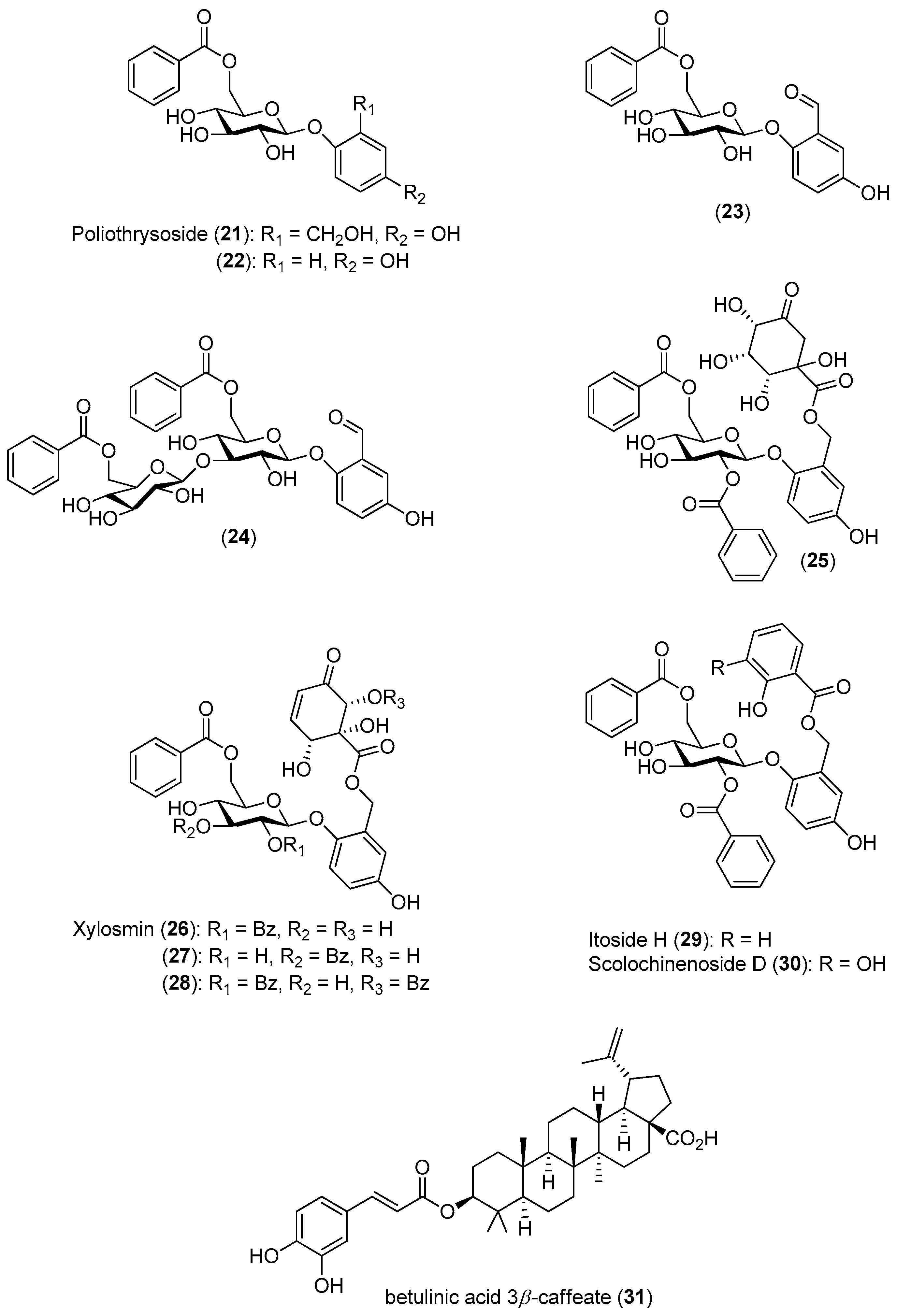
To identify new inhibitors of the DENV-2 NS5 RdRp, Litaudon and co-workers screened 820 ethyl acetate extracts from different parts of 400 plants collected in Madagascar. The bark extract of
Flacourtia ramontchi was selected for fractionation, because of its pronounced effect on the DENV-2 NS5 RdRp. The compounds shown in
Figure 4 were isolated from this extract and fully characterized [
50]. The data displayed in
Figure 4 reveal that among the isolated phenolic glycosides
21–
31, Compounds
22,
27 and
30 were the most active. The cinnamic acid derivative Compound
31 showed significant inhibitory activity, although its activity is about four-times lower than the positive control. However, no structure-activity relationships could be established for any of these compounds.
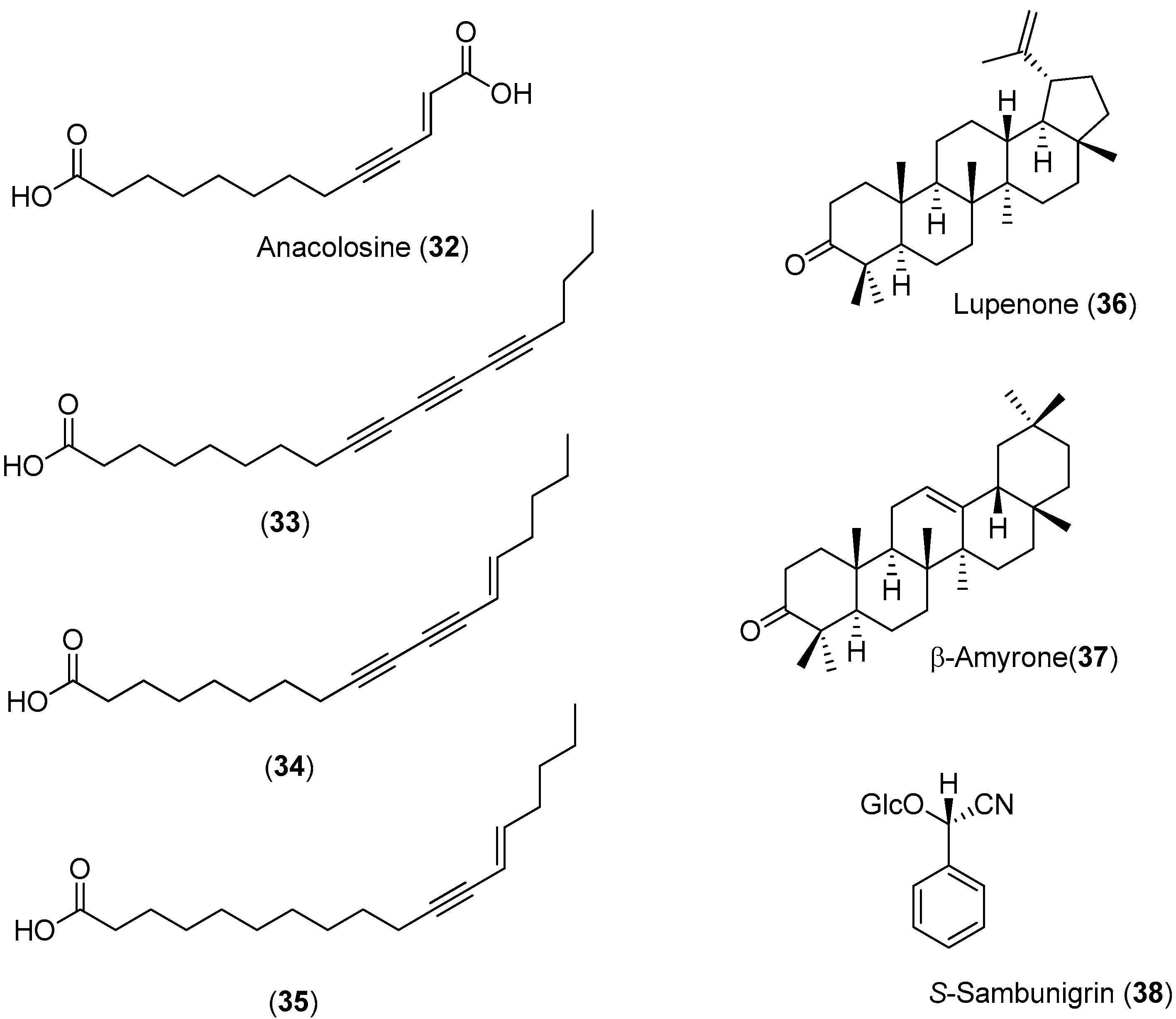
Anacolosa pervilleana is a Madagascan plant whose leaves, bark and young shoots are used in traditional medicines for the treatment of schistosomiasis, syphilis and general weakness. Four acetylenic carboxylic acids (Compounds
32–
35), two triterpenes (Compounds
36 and
37) and one aromatic compound (Compound
38) were isolated from the leaf ethyl acetate extract of this species (
Figure 5) [
51].
The acetylenic acids were found to be the most active compounds, inhibiting the DENV NS5 RdRp with approximately the same efficacy. The observed IC
50 values (<3 μM) were about 100-times lower than the reference compound, 3′-deoxy-GTP [
51].
3.4. Direct Inhibitory Activities of Plant Extracts against DENV Enzymes
The medicinal plants
Vernonia cinerea,
Hemigraphis reptans,
Hedyotis auricularia,
Laurentia longiflora,
Tridax procumbens and
Senna angustifolia were used to evaluate their abilities to inhibit the DENV NS3 protease. The highest inhibitory effects were observed for the ethanolic extract of
S. angustifolia leaves, the methanolic extract of
V. cinerea leaves and the ethanolic extract of
T. procumbens stems (IC
50 values: 30.1 ± 3.4, 23.7 ± 4.1 and 25.6 ± 3.8 μg/mL, respectively). The most active extracts were also tested in vitro against DENV-2-infected Vero cells, which were able to maintain a normal morphology without cytopathic effects. The percent viral inhibition of the extracts of
V. cinerea (80.6% ± 6.1%) and
T. procumbens (64.0% ± 9.4%) was significantly higher than that of
S. angustifolia extract (26.3% ± 3.8%), as measured by a plaque-forming assay and RT-qPCR [
52]. The authors of this investigation did not analyze which compounds were responsible for the observed activities.
3.5. Indirect Inhibitory Activities of Compounds and Extracts against DENV Enzymes
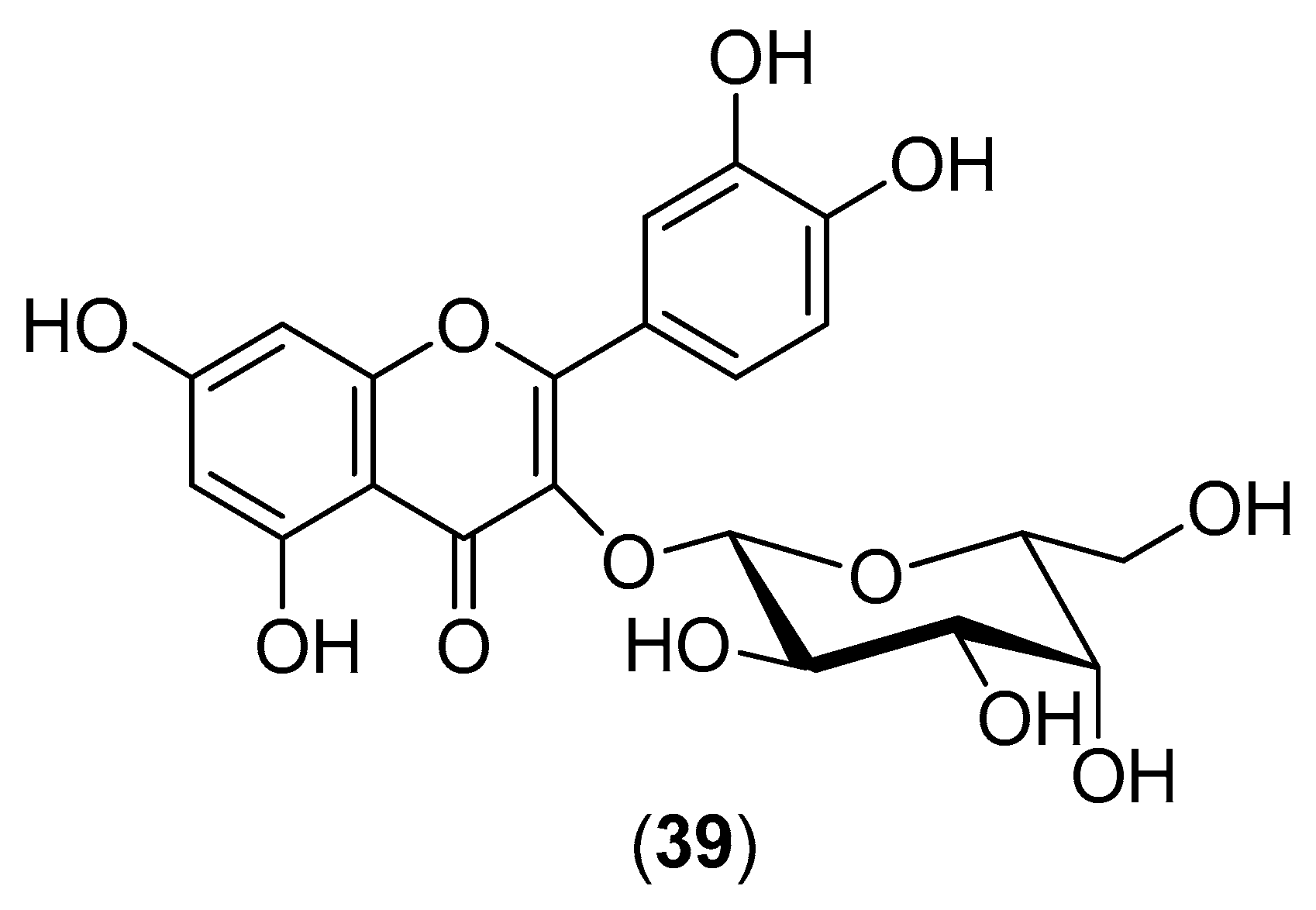
The plant
Houttuynia cordata is a popular vegetable consumed in the northern and eastern regions of Thailand. Its aqueous extract was evaluated for inhibitory activity against DENV-2 within the 10–100-mg/mL concentration range. Both pre- and post-incubation of HepG2 cells with
H. cordata extract resulted in a significant reduction in intracellular DENV-2 RNA production, correlating with a decrease in DENV-2 protein expression. The extract directly inhibited intracellular viral RNA replication, with an effective concentration (EC
50) of 0.8 mg/mL. Within the 10–40-mg/mL concentration range, the
H. cordata extract also exhibited a protective effect on virion release from infected LLC-MK2 cells (Rhesus Monkey Kidney Epithelial Cells). The reduction in RNA production and decrease in DENV-2 protein expression suggest the inhibition of viral enzymatic activity by the aqueous extract. High-performance liquid chromatography of the aqueous extract revealed that hyperoside (Compound
39) (
Figure 6) is the major component of the extract, suggesting that this flavonoid plays an important role in DENV-2 inhibition. The aqueous extract had no toxic effects on human blood cells [
53].
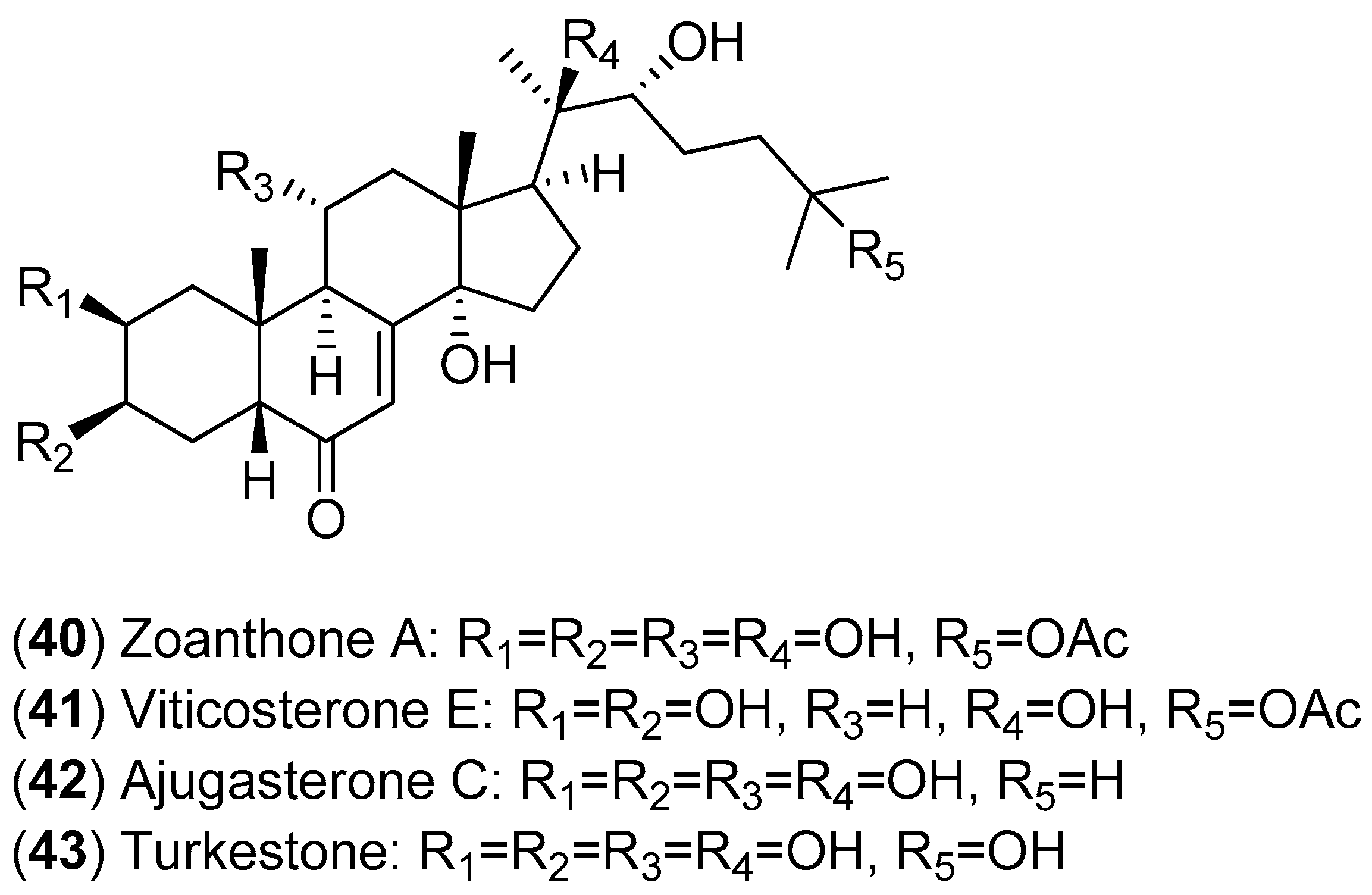
The fractionation of the ethanolic extract of
Zoanthus spp. (sea anemone) collected in Taiwan resulted in the isolation of 14 ecdysones; the inhibitory activities of these compounds against DENV-2 were evaluated. Among the isolated ecdysonoids, Compounds
40–
43 were the most potent. The results shown in
Figure 7 suggest that ajugasterone C (Compound
42) is equipotent to 2′-C-methylcytidine (positive control), with a better Seletive Index (SI). In addition, the most active ecdysone, Compound
42, was also tested against other DENV serotypes. Ecdysone
42 was found to be active against all DENV serotypes, with the following EC
50 values: DENV-1 (15.70 ± 2.36 μM), DENV-3 (9.48 ± 0.24 μM) and DENV-4 (12.15 ± 1.22 μM) with a high CC
50 (the 50% cytotoxic concentration). After analyses of both the structure-activity relationship and molecular docking, the authors proposed that ecdysone
42 impairs DENV RNA replication by blocking the viral polymerase channel [
54].
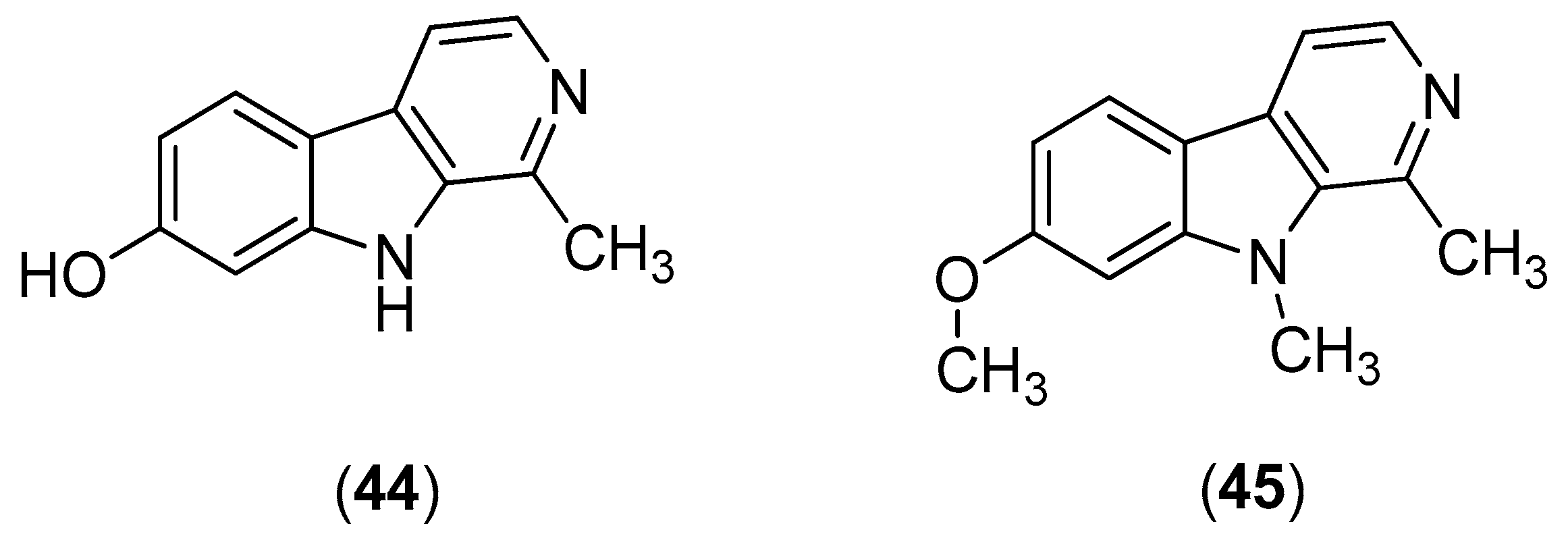
β-Carbolines are widespread plant and animal alkaloids that possess important biological activities [
55]. Quintana and co-workers evaluated the effects of natural and synthetic β-carbolines; they found that the natural product harmol (Compound
44) and its synthetic derivative 9-
N-methylharmine (Compound
45) were the most potent alkaloids, with inhibitory activities against DENV-2 and SI values of 56.2 and 61.3, respectively (
Figure 8). Vero cells were infected with DENV at a multiplicity of infection (MOI) of 1 or 0.1 PFU/cell. At 48 h post-infection, the cells were lysed; and the supernatants were harvested; virus yields were quantified by plaque assay. The results indicate that the compounds likely act via viral enzyme inhibition. Harmol (Compound
44) and 9-
N-methylharmine (Compound
45) were also found to possess inhibitory effects against DENV-1, DENV-3 and DENV-4, albeit with lower efficiencies than against DENV-2 [
56].
3.6. In Silico Investigations of DENV Enzyme Inhibitors
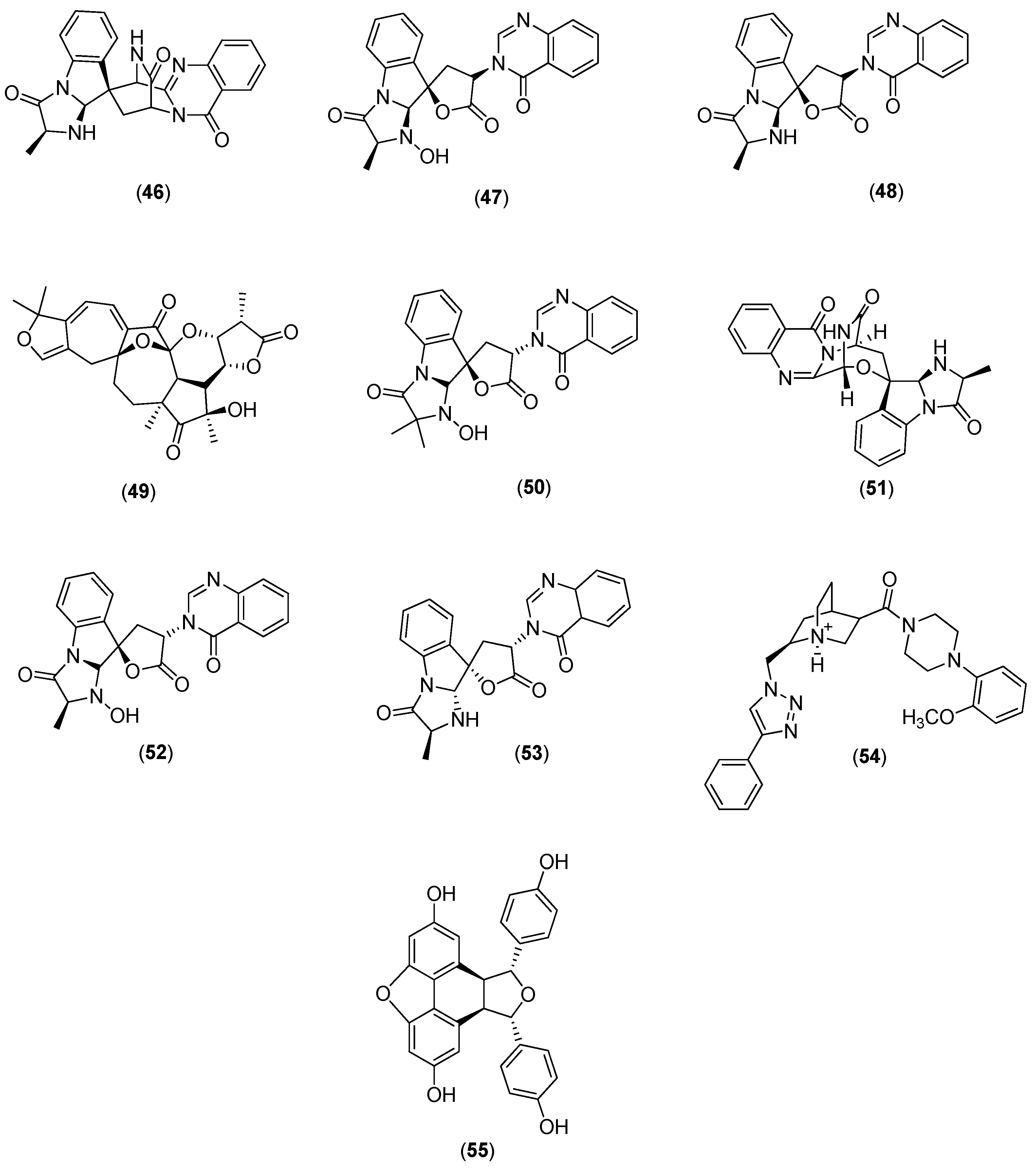
Galiano and co-authors conducted an in silico study to identify new inhibitors of the NS5 RdRp in the four DENV serotypes. They used a chemical library of 372,792 non-nucleotide compounds to perform molecular docking experiments at the binding site of the RNA template tunnel of the polymerase. After the screening process was completed, 39 compounds were identified as leading DENV RdRp inhibitor candidates. The selected compounds had a highly negative free energy variation (∆G) when docked to the binding site of the RNA template tunnel in the four DENV serotypes. In addition, the majority of the selected compounds had favorable druggability and optimal ADMET (absorption, distribution, metabolism, excretion, and toxicity) properties [
57]. Among the 39 selected compounds, 10 (Compounds
46–
55 in
Figure 9) were natural products.
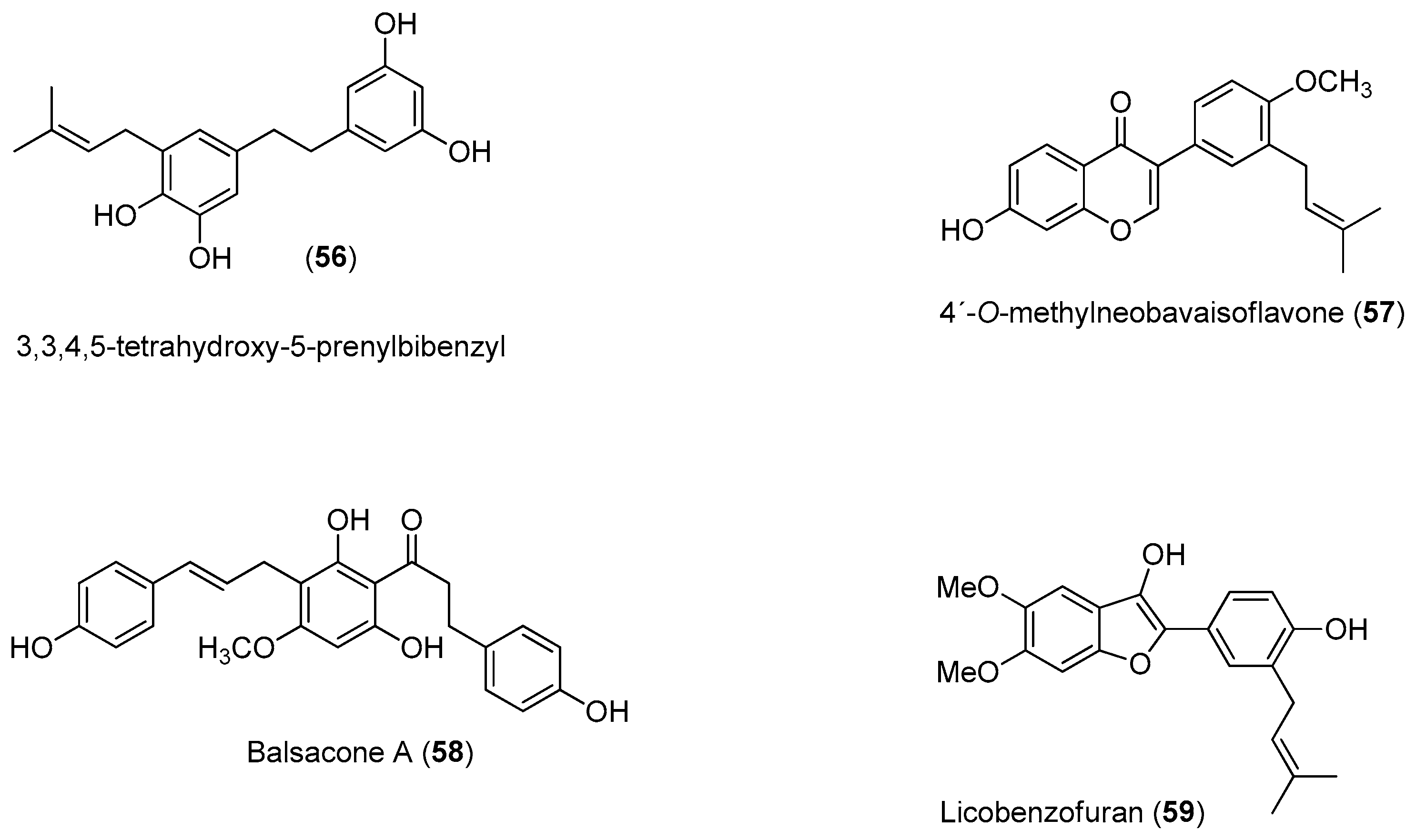
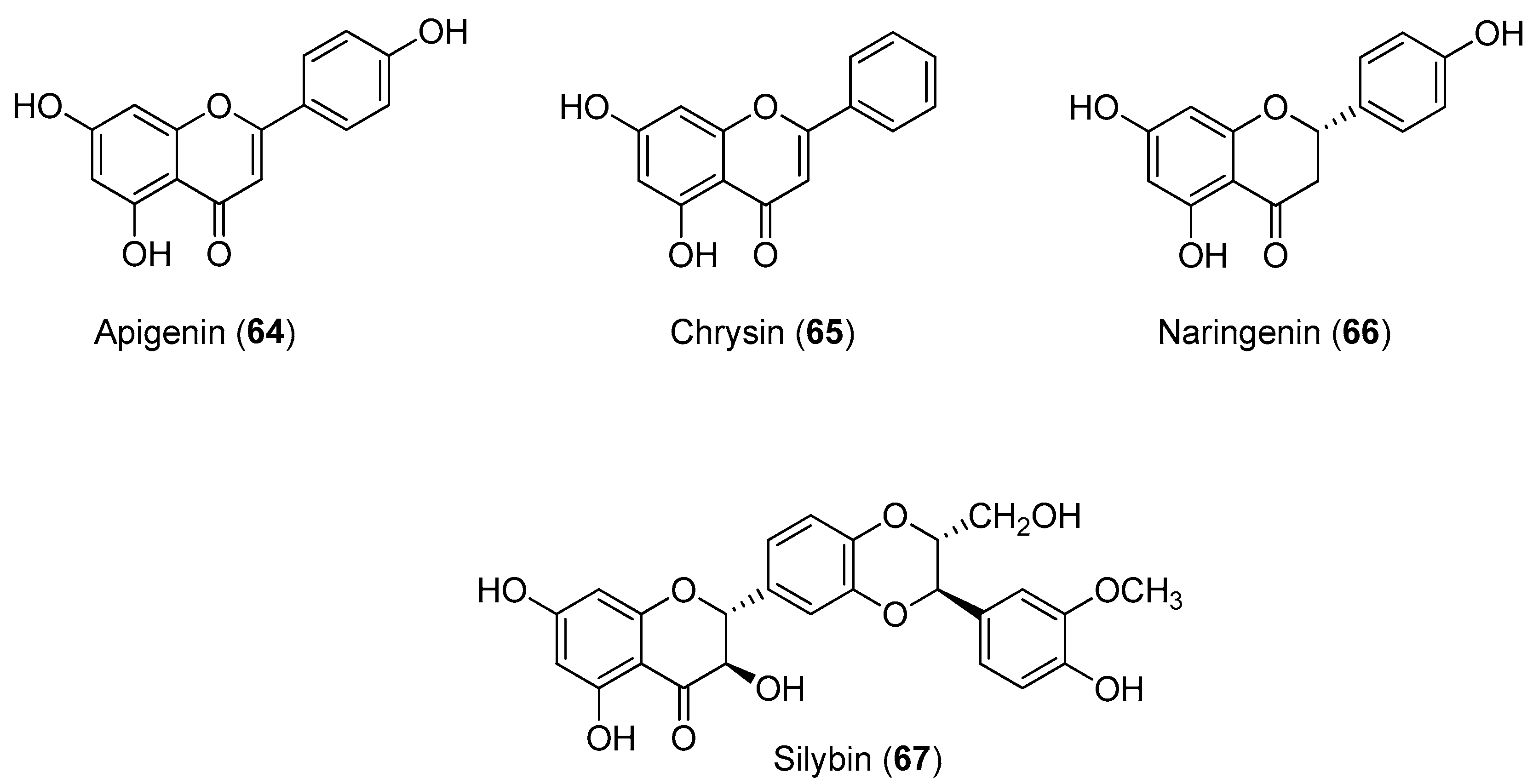
Recently, Power and Setzer reported an in silico investigation of natural products as potential antiviral agents against DENV protease (NS2B-NS3pro), helicase (NS3 helicase), MTase, RdRp and the virus envelope. A total of 2194 plant-derived natural products were docked. The compound set was composed of 290 alkaloids (68 indole alkaloids, 153 isoquinoline alkaloids, 5 quinoline alkaloids, 13 piperidine alkaloids, 14 steroidal alkaloids and 37 miscellaneous alkaloids), 678 terpenoids (47 monoterpenoids, 169 sesquiterpenoids, 265 diterpenoids, 81 steroids and 96 triterpenoids), 20 aurones, 81 chalcones, 349 flavonoids, 120 isoflavonoids, 74 lignans, 58 stilbenoids, 169 miscellaneous polyphenolic compounds, 100 coumarins, 28 xanthones, 67 quinones and 160 miscellaneous natural compounds. Polyphenolic compounds, flavonoids, chalcones and other phenolics were identified as the most strongly docking ligands for DENV protein targets [
58], as shown in
Figure 10 for selected ligands
56–
59 for DENV NS2B-NS3.