A Protein Isolate from Moringa oleifera Leaves Has Hypoglycemic and Antioxidant Effects in Alloxan-Induced Diabetic Mice
Abstract
:1. Introduction
2. Results
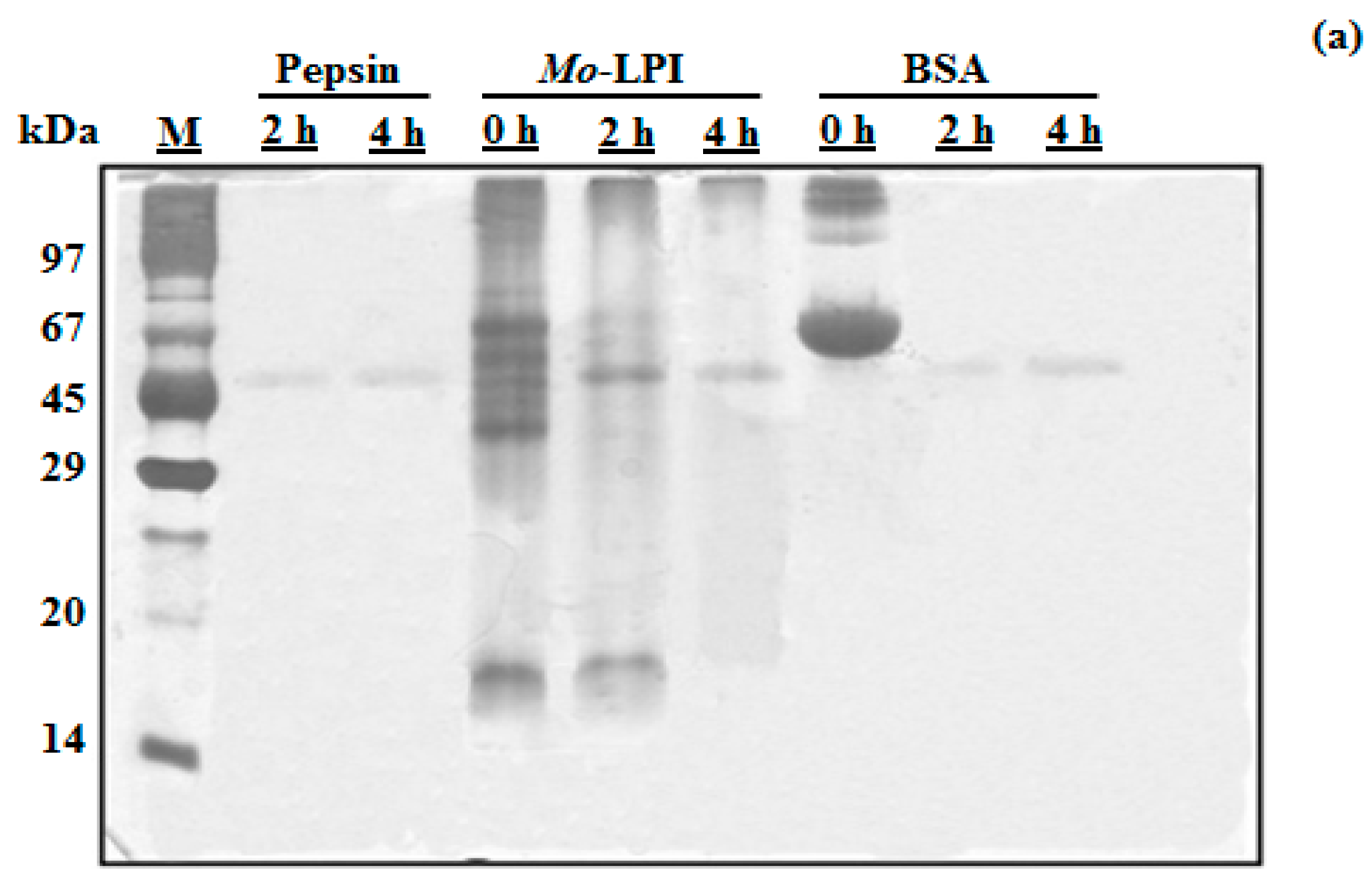
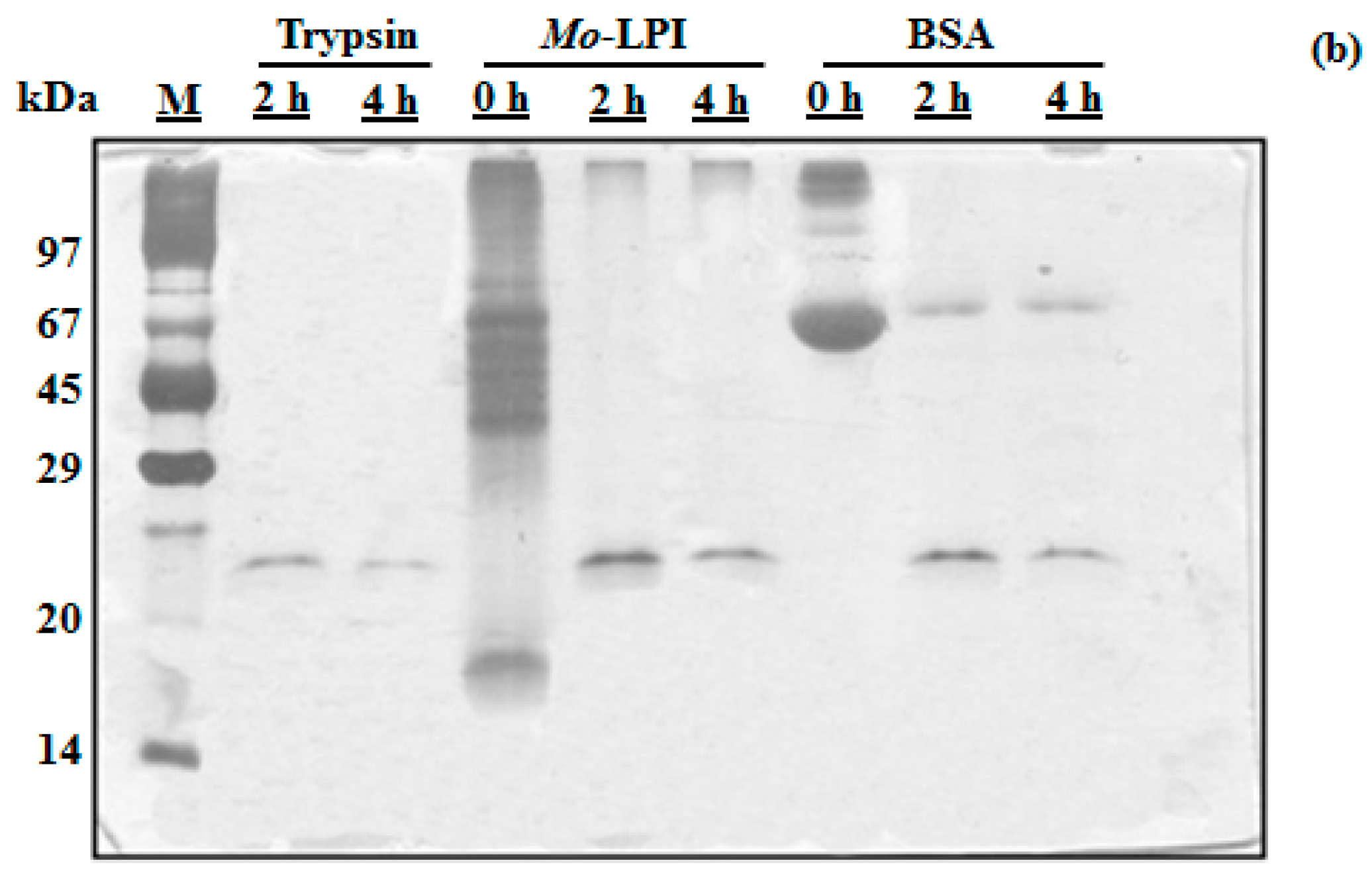
2.1. Biochemical Properties of Mo-LPI
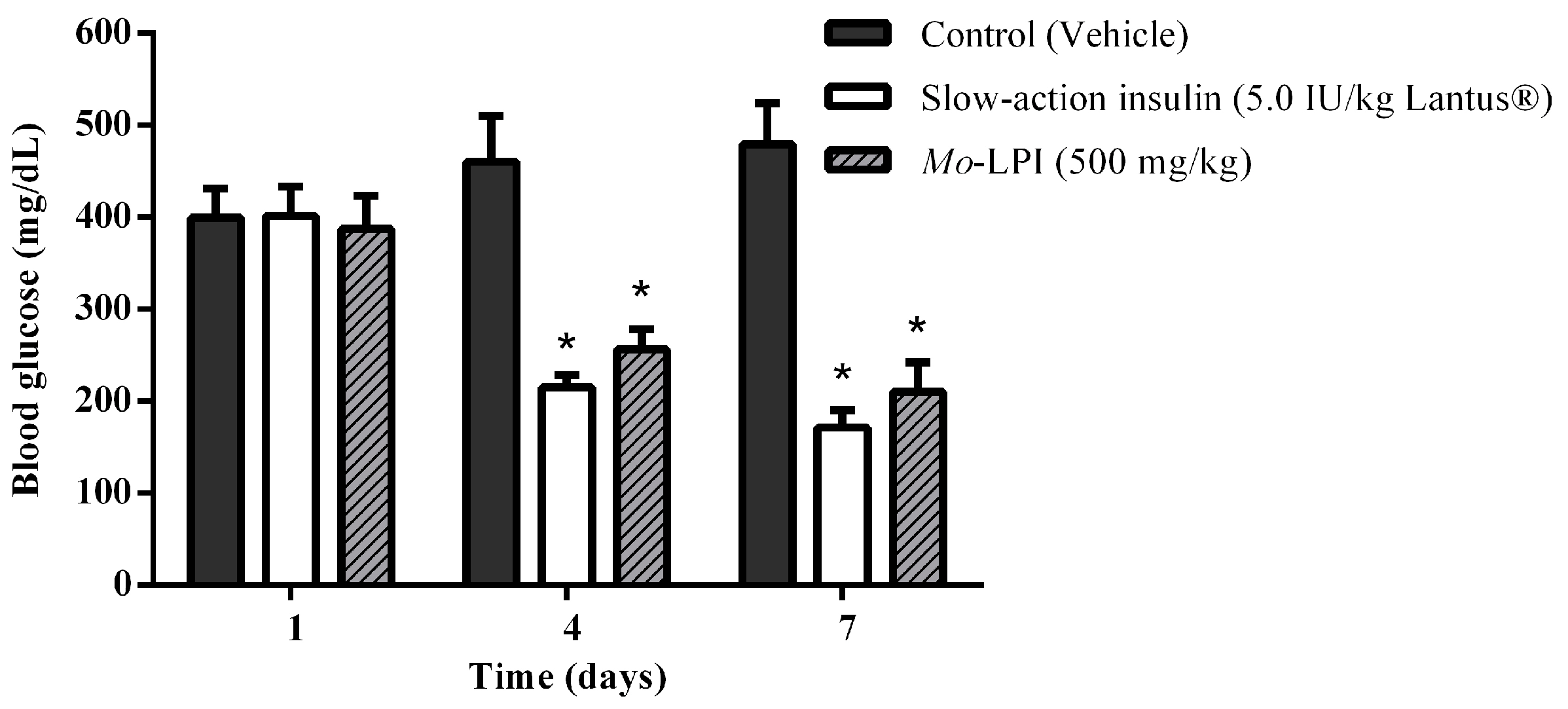
2.2. Effect of Mo-LPI on Fasting Blood Glucose in Alloxan-Induced Diabetic Mice
2.3. Effect of Mo-LPI on Serum Insulin Level
2.4. Effect of Mo-LPI on Lipid Peroxidation and Antioxidant Enzymes in Alloxan-Induced Diabetic Mice
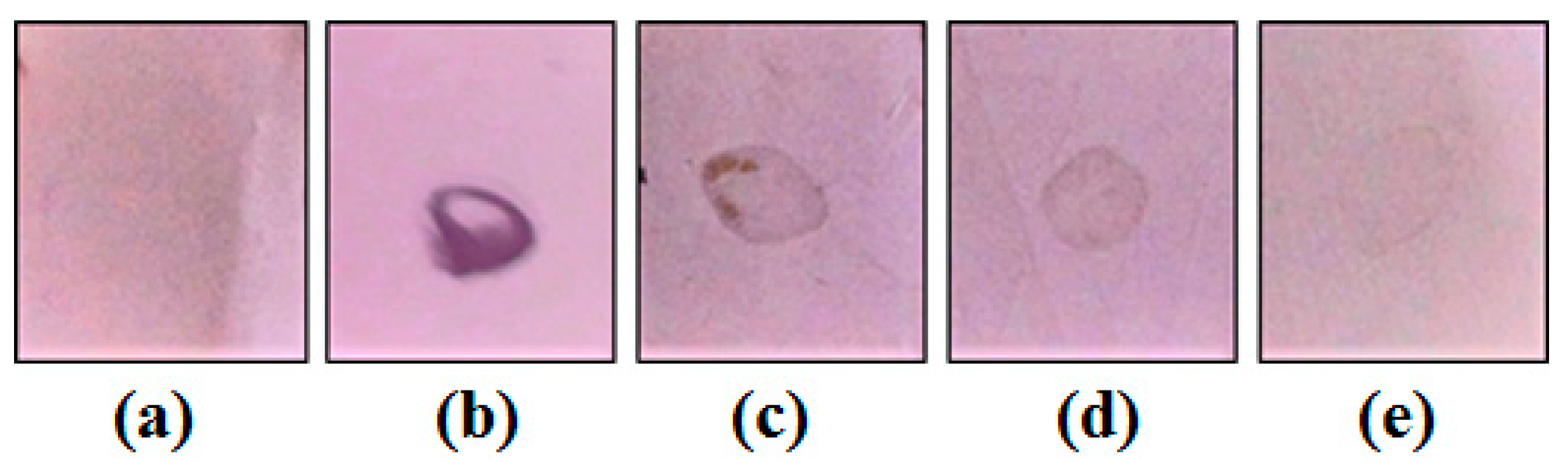
2.5. Behavioral Effect and Toxicity of Mo-LPI
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Animals
4.3. Obtention of Mo-LPI
4.4. Protein Determination
4.5. In Vitro Digestibility
4.6. Hemagglutinating Activity
4.7. Dot Blot
4.8. Evaluation of Potential for Precipitation with Zinc
4.9. Induction of Experimental Diabetes
4.10. Single Dose Test in Alloxan Induced Diabetic Mice
4.11. Repeated Dose Test in Alloxan Induced Diabetic Mice
4.12. Serum Insulin Measurement
4.13. Antioxidant Effect Evaluation
4.14. Acute Toxicity Assessment
4.15. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- American Diabetes Association. Diagnosis and classification of diabetes. Diabetes Care 2016, 39, S13–S22. [Google Scholar]
- Teugwa, C.M.; Boudjeko, T.; Tchinda, B.T.; Mejiato, P.C.; Zofou, D. Anti-hyperglycaemic globulins from selected Cucurbitaceae seeds used as antidiabetic medicinal plants in Africa. BMC Complement. Altern. Med. 2013, 13, 63. [Google Scholar] [CrossRef] [PubMed]
- Li, W.L.; Zheng, H.C.; Bukuru, J.; De Kimpe, N. Natural medicines used in the traditional Chinese medical system for therapy of diabetes mellitus. J. Ethnopharmacol. 2004, 92, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Xavier-Filho, J.; Oliveira, A.E.A.; Silva, L.B.; Azevedo, C.R.; Venâncio, T.M.; Machado, O.L.T.; Oliva, M.L.; Fernandes, K.V.S.; Xavier-Neto, J. Plant insulin or glucokinin: A conflicting issue. Braz. J. Plant Physiol. 2003, 15, 67–78. [Google Scholar] [CrossRef]
- Mentreddy, S.R. Medicinal plant species with potential antidiabetic properties. J. Sci. Food Agric. 2007, 87, 743–750. [Google Scholar] [CrossRef]
- Rajasekhar, M.D.; Badri, K.R.; Kumar, K.V.; Kassetti, R.B.; Fatima, S.S.; Kumar, M.T.S.; Appa Rao, C. Isolation and characterization of a novel antihyperglycemic protein from the fruits of Momordica cymbalaria. J. Ethnopharmacol. 2010, 128, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.E.A.; Machado, O.L.T.; Gomes, V.M.; Xavier-Neto, J.; Pereira, A.C.; Vieira, J.G.H.; Fernandes, K.V.S.; Xavier-Filho, J. Jack bean seed coat contains a protein with complete sequence homology to bovine insulin. Protein Pept. Lett. 1999, 6, 15–21. [Google Scholar]
- Sheng, Q.; Yao, H.; Xu, H.; Ling, X.; He, T. Isolation of plant insulin from Momordica charantia seeds by gel filtration and RP-HPLC. Zhong Yao Cai 2004, 27, 414–416. [Google Scholar] [PubMed]
- Kavalali, G.; Tuncel, H.; Göksel, S.; Hatemi, H.H. Hypoglycemic activity of Urtica pilulifera in streptozotocin-diabetic rats. J. Ethnopharmacol. 2003, 84, 241–245. [Google Scholar] [CrossRef]
- Li, Q.; Ye, X.L.; Zeng, H.; Chen, X.; Li, X.G. Study on the extraction technology and hypoglycemic activity of lectin from Trichosanthes kirilowi. Zhong Yao Cai 2012, 35, 475–479. [Google Scholar] [PubMed]
- Rocha, A.A.; Araújo, T.F.S.; Fonseca, C.S.M.; Mota, D.L.; Medeiros, P.L.; Paiva, P.M.G.; Coelho, L.C.B.B.; Correia, M.T.S.; Lima, V.L.M. Lectin from Crataeva tapia bark improves tissue damages and plasma hyperglycemia in alloxan-induced diabetic mice. Evid.-Based Complement. Altern. Med. 2013, 2013, 869305. [Google Scholar] [CrossRef] [PubMed]
- Kar, A.; Choudhary, B.K.; Bandyopadhyay, N.G. Comparative evaluation of hypoglycaemic activity of some Indian medicinal plants in alloxan diabetic rats. J. Ethnopharmacol. 2003, 84, 105–108. [Google Scholar] [CrossRef]
- Jaiswal, D.; Kumar Rai, P.; Kumar, A.; Mehta, S.; Watal, G. Effect of Moringa oleifera Lam. leaves aqueous extract therapy on hyperglycemic rats. J. Ethnopharmacol. 2009, 123, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Mathur, M.; Bajaj, V.K.; Katariya, P.; Yadav, S.; Kamal, R.; Gupta, R.S. Evaluation of antidiabetic and antioxidant activity of Moringa oleifera in experimental diabetes. J. Diabetes 2012, 4, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Mbikay, M. Therapeutic potential of Moringa oleifera leaves in chronic hyperglycemia and dyslipidemia: A review. Front. Pharmacol. 2012, 3, 24. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.L.; Oliveira, H.D.; Oliveira, J.T.A.; Gifoni, J.M.; Rocha, R.O.; Sousa, D.O.B.; Vasconcelos, I.M. Purification of a chitin-binding protein from Moringa oleifera seeds with potential to relieve pain and inflammation. Protein Pept. Lett. 2011, 18, 1078–1085. [Google Scholar] [CrossRef] [PubMed]
- Sinha, M.; Das, D.K.; Bhattacharjee, S.; Majumdar, S.; Dey, S. Leaf extract of Moringa oleifera prevents ionizing radiation-induced oxidative stress in mice. J. Med. Food 2011, 14, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- Mehta, K.; Balaraman, R.; Amin, A.H.; Bafna, P.A.; Gulati, O.D. Effect of fruits of Moringa oleifera on the lipid profile of normal and hypercholesterolaemic rabbits. J. Ethnopharmacol. 2003, 86, 191–195. [Google Scholar] [CrossRef]
- Tende, J.A.; Ezekiel, I.; Dikko, A.A.U.; Goji, A.D.T. Effect of ethanolic leaves extract of Moringa oleifera on blood glucose levels of streptozotocin-induced diabetes and normoglycemic Wistar rats. Br. J. Pharmacol. Toxicol. 2011, 3, 1–4. [Google Scholar]
- Francis, J.A.; Jayaprakasam, B.; Olson, L.K.; Nair, M.G. Insulin secretagogues from Moringa oleifera with cyclooxygenase enzyme and lipid peroxidation inhibitory activities. Helv. Chim. Acta 2004, 87, 317–326. [Google Scholar] [CrossRef]
- Paula, P.C.; Oliveira, J.T.A.; Sousa, D.O.B.; Alves, B.G.T.; Carvalho, A.F.U.; Franco, O.L.; Vasconcelos, I.M. Insulin-like plant proteins as potential innovative drugs to treat diabetes—The Moringa oleifera case study. New Biotechnol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Venancio, T.M.; Oliveira, A.E.A.; Silva, L.B.; Machado, O.L.T.; Fernandes, K.V.S.; Xavier-Filho, J. A protein with amino acid sequence homology to bovine insulin is present in the legume Vigna unguiculata (cowpea). Braz. J. Med. Biol. Res. 2003, 36, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Khanna, P.; Jain, S.C.; Panagariya, A.; Dixit, V.P. Hypoglycemic activity of polypeptide-p from a plant source. J. Nat. Prod. 1981, 44, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Aleator, O. Protein quality evaluation and in vitro multi-enzyme digestibility of some plant protein isolates and concentrates. Arch. Zootech. 2012, 15, 5–16. [Google Scholar]
- Oliveira, J.T.A.; Melo, V.M.M.; Câmara, M.F.L.; Vasconcelos, I.M.; Beltramini, L.M.; Machado, O.L.T.; Gomes, V.M.; Pereira, S.P.; Fernandes, C.F.; Nunes, E.P.; et al. Purification and physicochemical characterization of a cotyledonary lectin from Luetzelburgia auriculata. Phytochemistry 2002, 61, 301–310. [Google Scholar] [CrossRef]
- Raman, B.V.; Sravani, B.; Rekha, P.P.; Lalitha, K.V.N.; Rao, B.N. Effect of plant lectins on human blood group antigens with special focus on plant foods and juices. Int. J. Res. Ayurveda Pharm. 2012, 3, 255–263. [Google Scholar]
- Azevedo, C.R.; Maciel, F.M.; Silva, L.B.; Ferreira, A.T.S.; Da Cunha, M.; Machado, O.L.T.; Fernandes, K.V.S.; Oliveira, A.E.A.; Xavier-Filho, J. Isolation and intracellular localization of insulin-like proteins from leaves of Bauhinia variegata. Braz. J. Med. Biol. Res. 2006, 39, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Gualandi-Signorini, A.M.; Giorgi, G. Insulin formulations: A review. Eur. Rev. Med. Pharmacol. Sci. 2001, 5, 73–83. [Google Scholar] [PubMed]
- Fischer, J. Drugs and chemicals that produce diabetes. Trends Pharmacol. Sci. 1985, 6, 72–75. [Google Scholar] [CrossRef]
- Cryer, P.E.; Davis, S.N.; Shamoon, H. Hypoglycemia in diabetes. Diabetes Care 2003, 26, 1902–1912. [Google Scholar] [CrossRef] [PubMed]
- McCrimmon, R.J.; Sherwin, R.S. Hypoglycemia in type 1 diabetes. Diabetes 2010, 59, 2333–2339. [Google Scholar] [CrossRef] [PubMed]
- Yibchok-Anun, S.; Adisakwattana, S.; Yao, C.Y.; Sangvanich, P.; Roengsumran, S.; Hsu, W.H. Slow acting protein extract from fruit pulp of Momordica charantia with insulin secretagogue and insulinomimetic activities. Biol. Pharm. Bull. 2006, 29, 1126–1131. [Google Scholar] [CrossRef] [PubMed]
- Gifoni, J.M.; Oliveira, J.T.A.; Oliveira, H.D.; Batista, A.B.; Pereira, M.L.; Gomes, A.S.; Oliveira, H.P.; Grangeiro, T.B.; Vasconcelos, I.M. A novel chitin-binding protein from Moringa oleifera seed with potential for plant disease control. Biopolymers 2012, 98, 406–415. [Google Scholar] [CrossRef] [PubMed]
- Katre, U.V.; Suresh, C.G.; Khan, M.I.; Gaikwad, S.M. Structure-activity relationship of a hemagglutinin from Moringa oleifera seeds. Int. J. Biol. Macromol. 2008, 42, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Li, Y.-W.; Jia, N.; Song, X.-M.; Duan, J.-L.; Weng, Y.; Guan, Y.; Zhou, D.; Wang, L.; Xi, M.M.; et al. Insulin-secretagogue activity of eleven plant extracts and twelve pure compounds isolated from Aralia taibaiensis. Life Sci. 2013, 92, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Jiang, Y.; Chen, W.; Yao, F.; Huang, G.; Sun, L. Evaluation of hypoglycemic effects of polyphenols and extracts from Penthorum chinense. J. Ethnopharmacol. 2015, 163, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Kudaravalli, J.; Vijayalakshmi, G.; Kishore, K.K. Safety and efficacy of sulfonylurea drugs in type 2 diabetes mellitus. Apollo Med. 2013, 10, 165–168. [Google Scholar] [CrossRef]
- Nishikawa, T.; Edelstein, D.; Du, X.L.; Yamagishi, S.; Matsumura, T.; Kaneda, Y.; Yorek, M.A.; Beebe, D.; Oates, P.J.; Hammes, H.P.; et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000, 404, 787–790. [Google Scholar] [PubMed]
- Del Rio, D.; Stewart, A.J.; Pellegrini, N. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr. Metab. Cardiovasc. Dis. 2005, 15, 316–328. [Google Scholar] [CrossRef] [PubMed]
- Saio, V.; Syiem, D.; Sharma, R. Effect of Potentilla fulgens on lipid peroxidation and antioxidant status in alloxan-induced diabetic mice. J. Basic Clin. Pharm. 2012, 3, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Cencione, C.; Spallotta, F.; Greco, S.; Martelli, F.; Zeiher, A.M.; Gaetano, C. Epigenetic mechanisms of hyperglycemic memory. Int. J. Biochem. Cell Biol. 2014, 51, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Lima, V.B.S.; Sampaio, F.A.; Bezerra, D.L.C.; Neto, J.M.M.; Marreiro, D.N. Parameters of glycemic control and their relationship with zinc concentrations in blood and with superoxide dismutase enzyme activity in type 2 diabetes patients. Arq. Bras. Endocrinol. Metab. 2011, 55, 701–707. [Google Scholar]
- Zhang, L.; Yang, J.; Chen, X.-Q.; Zan, K.; Wen, X.-D.; Chen, H.; Wang, Q.; Lai, M.-X. Antidiabetic and antioxidant effects of extracts from Potentilla discolor Bunge on diabetic rats induced by high fat diet and streptozotocin. J. Ethnopharmacol. 2010, 132, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, S.; Rajasekaran, A.; Manisenthil Kumar, K.T. Antidiabetic, antihyperlipidemic and antioxidant potential of methanol extract of Tectona grandis flowers in streptozotocin induced diabetic rats. Asian Pac. J. Trop. Med. 2011, 4, 624–631. [Google Scholar] [CrossRef]
- Ramachandran, S.; Rajasekaran, A.; Manisenthil Kumar, K.T. Investigation of hypoglycemic, hypolipidemic and antioxidant activities of aqueous extract of Terminalia paniculata bark in diabetic rats. Asian Pac. J. Trop. Biomed. 2012, 2, 262–268. [Google Scholar] [CrossRef]
- Awodele, O.; Oreagba, I.A.; Odoma, S.; Silva, J.A.T.; Osunkalu, V.O. Toxicological evaluation of the aqueous leaf extract of Moringa oleifera Lam. (Moringaceae). J. Ethnopharmacol. 2012, 139, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Sathe, S.K.; Sze-Tao, K.W.C.; Wolf, W.J.; Hamaker, B.R. Biochemical characterization and in vitro digestibility of the major globulin in cashew nut (Anacardium occidentale). J. Agric. Food Chem. 1997, 45, 2854–2860. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the bacteriophage T4. Nature 1970, 227, 679–685. [Google Scholar] [CrossRef]
- Blum, H.; Beier, H.; Gross, H.J. Improved silver staining of plant proteins, RNA and DNA in polyacrylamide gels. Electrophoresis 1997, 8, 93–99. [Google Scholar] [CrossRef]
- Moreira, R.A.; Perrone, J.C. Purification and partial characterization of a lectin from Phaseolus vulgaris. Plant Physiol. 1977, 59, 783–787. [Google Scholar] [CrossRef]
- Lis, H.; Sharon, N. Soybean (Glycine max) agglutinin. Methods Enzymol. 1972, 28, 360–365. [Google Scholar]
- Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc. Natl. Acad. Sci. USA 1979, 76, 4350–4354. [Google Scholar] [CrossRef] [PubMed]
- Hallas-Moller, K.; Petersen, K.; Schlichtkrull, J. Crystalline and amorphous insulin-zinc compounds with prolonged action. Science 1952, 116, 394–398. [Google Scholar] [CrossRef]
- Kumar, D.; Kumar, S.; Kohli, S.; Arya, R.; Gupta, J. Antidiabetic activity of methanolic bark extract of Albizia odoratissima Benth. in alloxan induced diabetic albino mice. Asian Pac. J. Trop. Biomed. 2011, 1, 900–903. [Google Scholar] [CrossRef]
- Djomeni, P.D.D.; Tédong, L.; Asongalem, E.A.; Dimo, T.; Sokeng, S.D.; Kamtchouing, P. Hypoglycaemic and antidiabetic effect of root extracts of Ceiba pentandra in normal and diabetic rats. Afr. J. Tradit. Complement. Altern. Med. 2006, 3, 129–136. [Google Scholar] [CrossRef]
- Agar, R.; Bosnak, M.; Amanvermez, R.; Demir, S.; Ayyildiz, M.; Celik, C. The effect of ethanol on lipid peroxidation and glutathione level in the brain stem of rat. Neuroreport 1999, 10, 1799–1801. [Google Scholar] [CrossRef] [PubMed]
- Aebi, H. Catalase in vitro. Methods Enzimol. 1984, 105, 121–126. [Google Scholar]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Adedapo, A.A.; Mogbojuri, O.M.; Emikpe, B.O. Safety evaluations of the aqueous extract of the leaves of Moringa oleifera in rats. J. Med. Plants Res. 2009, 3, 586–591. [Google Scholar]
- Sample Availability: Not applicable.





| Group | MDA (μmol/g Tissue) | CAT (U/µg Protein) | SOD (U/µg Protein) |
|---|---|---|---|
| Diabetic control | 57.43 ± 3.48 a | 1.92 ± 0.18 a | 0.42 ± 0.02 a |
| Non-diabetic | 33.50 ± 4.23 b | 1.88 ± 0.23 a | 1.42 ± 0.06 b |
| d + Insulin (0.7 IU/kg) | 61.13 ± 3.58 a | 2.01 ± 0.16 a | 1.20 ± 0.04 c |
| d + Mo-LPI (500 mg/kg) | 27.78 ± 2.15 b | 3.01 ± 0.35 b | 0.38 ± 0.02 a |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paula, P.C.; Sousa, D.O.B.; Oliveira, J.T.A.; Carvalho, A.F.U.; Alves, B.G.T.; Pereira, M.L.; Farias, D.F.; Viana, M.P.; Santos, F.A.; Morais, T.C.; et al. A Protein Isolate from Moringa oleifera Leaves Has Hypoglycemic and Antioxidant Effects in Alloxan-Induced Diabetic Mice. Molecules 2017, 22, 271. https://doi.org/10.3390/molecules22020271
Paula PC, Sousa DOB, Oliveira JTA, Carvalho AFU, Alves BGT, Pereira ML, Farias DF, Viana MP, Santos FA, Morais TC, et al. A Protein Isolate from Moringa oleifera Leaves Has Hypoglycemic and Antioxidant Effects in Alloxan-Induced Diabetic Mice. Molecules. 2017; 22(2):271. https://doi.org/10.3390/molecules22020271
Chicago/Turabian StylePaula, Paulo C., Daniele O. B. Sousa, Jose T. A. Oliveira, Ana F. U. Carvalho, Bella G. T. Alves, Mirella L. Pereira, Davi F. Farias, Martonio P. Viana, Flavia A. Santos, Talita C. Morais, and et al. 2017. "A Protein Isolate from Moringa oleifera Leaves Has Hypoglycemic and Antioxidant Effects in Alloxan-Induced Diabetic Mice" Molecules 22, no. 2: 271. https://doi.org/10.3390/molecules22020271
APA StylePaula, P. C., Sousa, D. O. B., Oliveira, J. T. A., Carvalho, A. F. U., Alves, B. G. T., Pereira, M. L., Farias, D. F., Viana, M. P., Santos, F. A., Morais, T. C., & Vasconcelos, I. M. (2017). A Protein Isolate from Moringa oleifera Leaves Has Hypoglycemic and Antioxidant Effects in Alloxan-Induced Diabetic Mice. Molecules, 22(2), 271. https://doi.org/10.3390/molecules22020271







