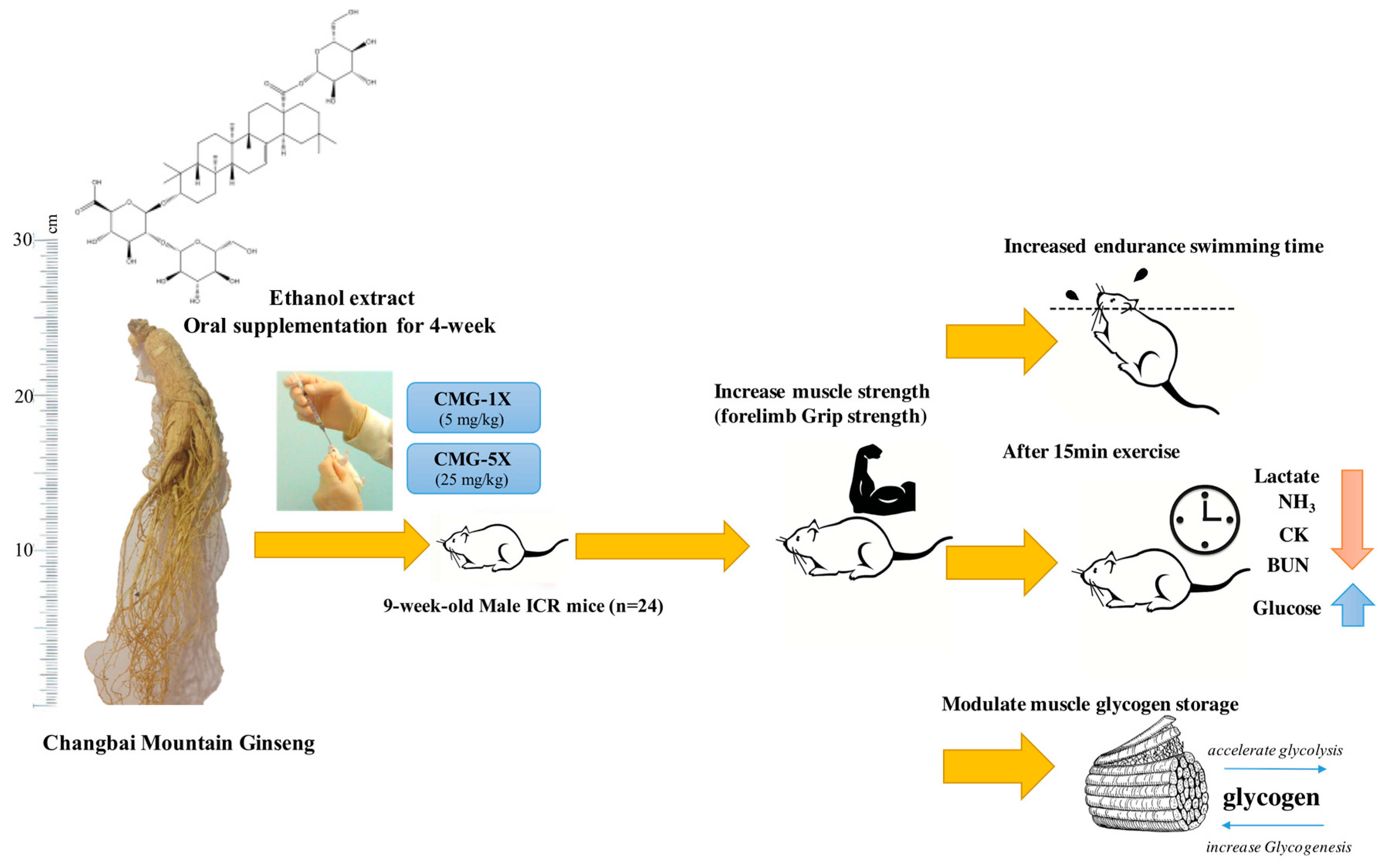
Changbai Mountain Ginseng (Panax ginseng C.A. Mey) Extract Supplementation Improves Exercise Performance and Energy Utilization and Decreases Fatigue-Associated Parameters in Mice
Abstract
:1. Introduction
2. Results
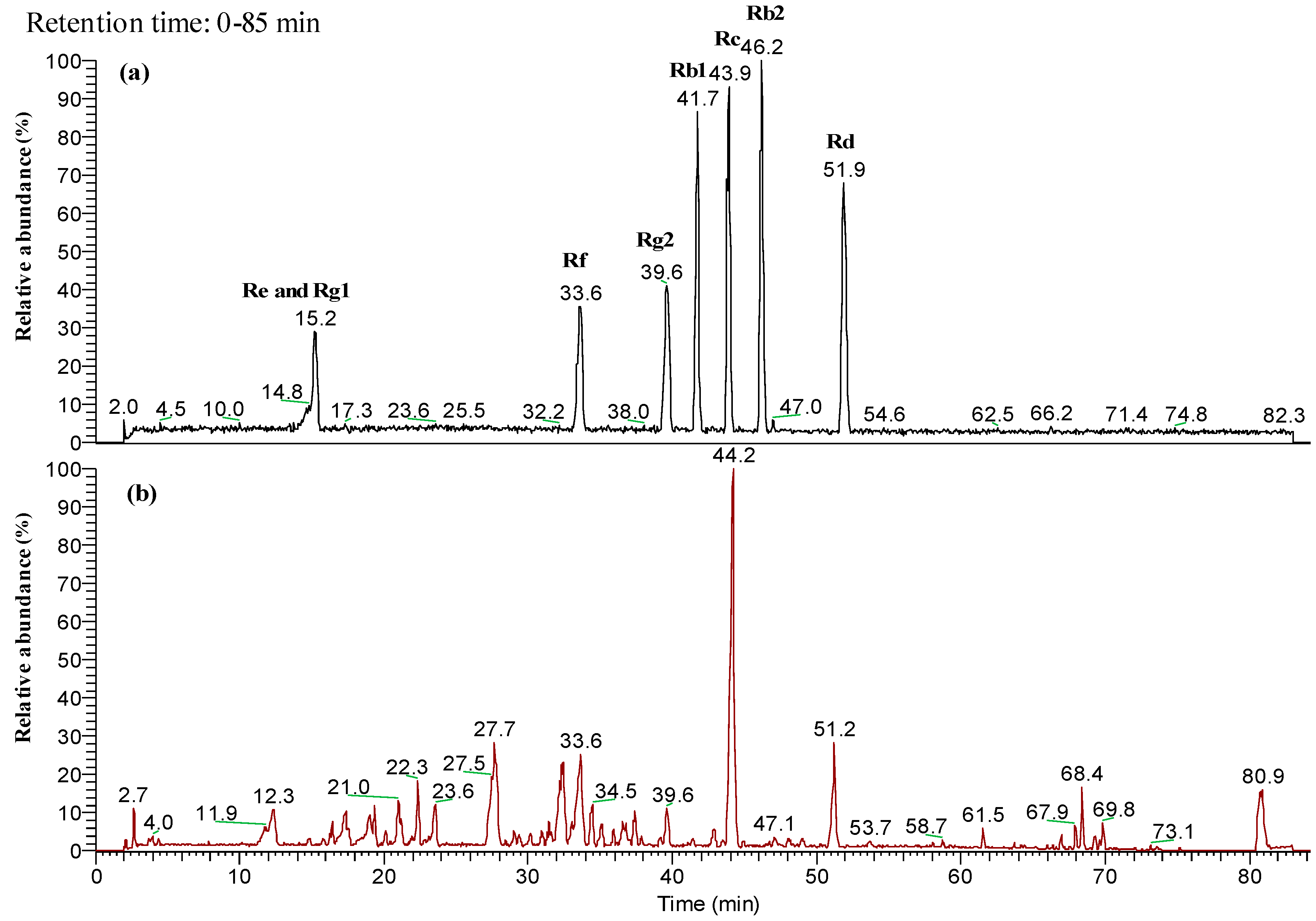
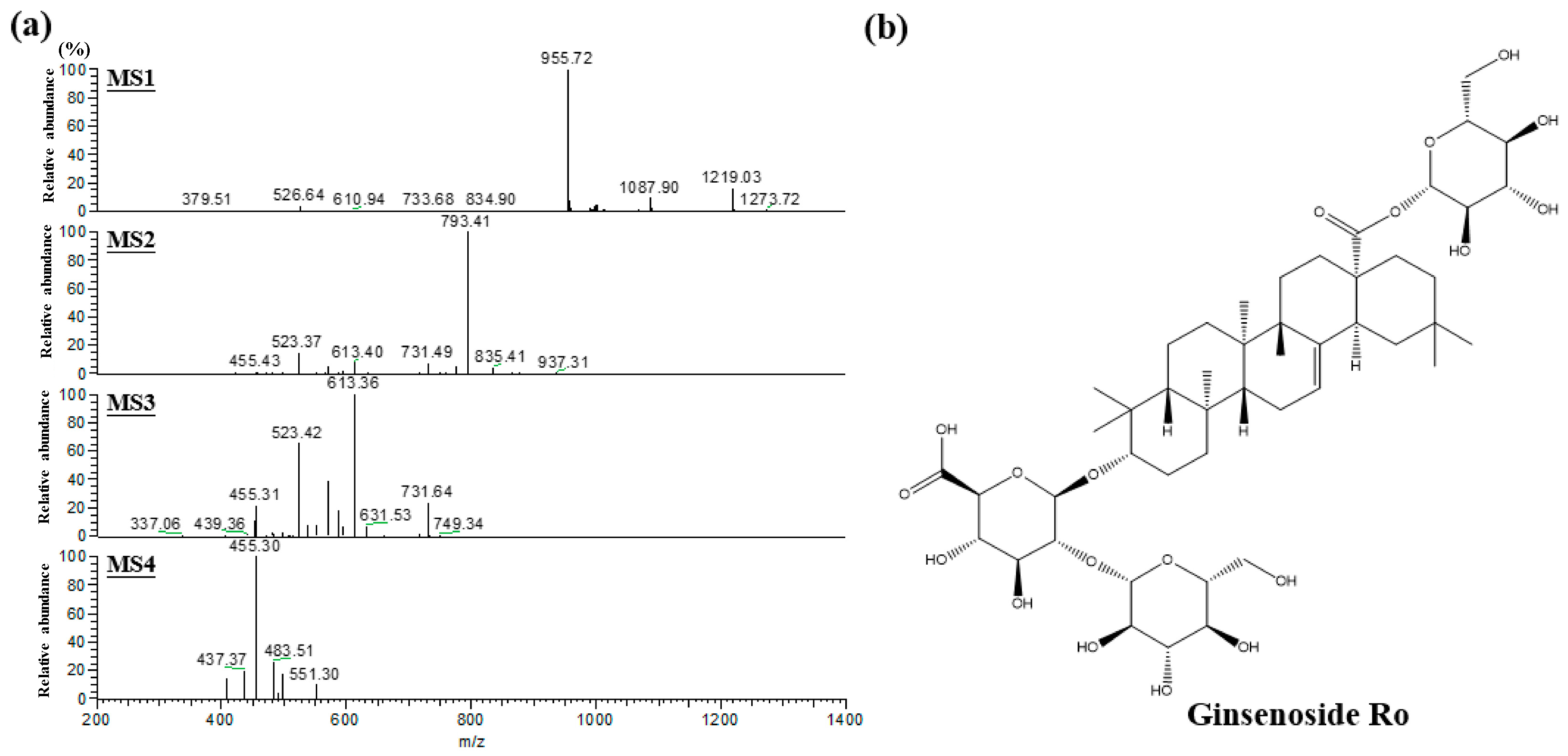
2.1. Characterized the Major Compound of CMG Extract by HPLC-ESI-MS/MS
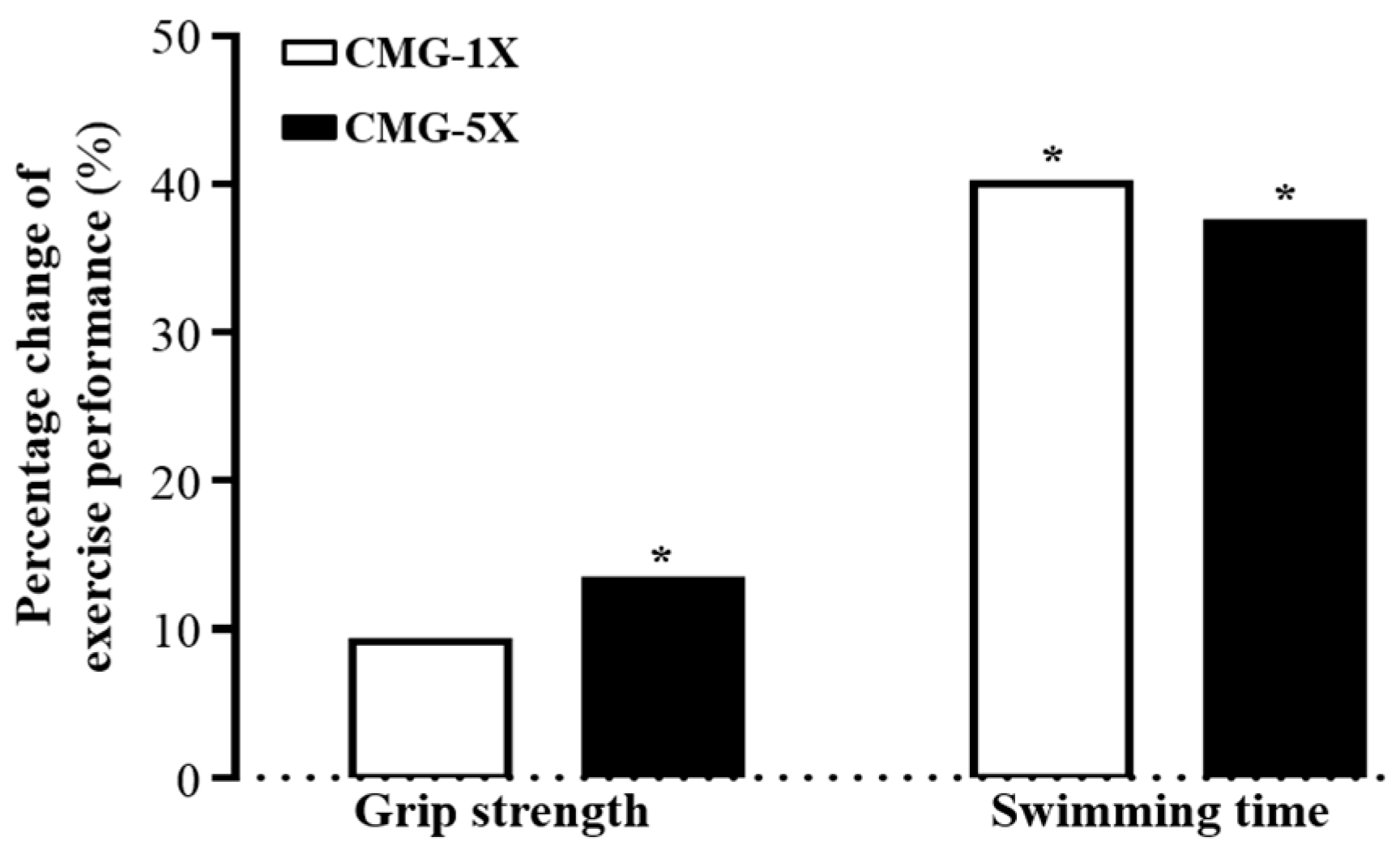
2.2. Effects of CMG on Forelimb Grip Strength
2.3. Effect of CMG on Exercise Performance in a Weight-Loaded Swimming Test
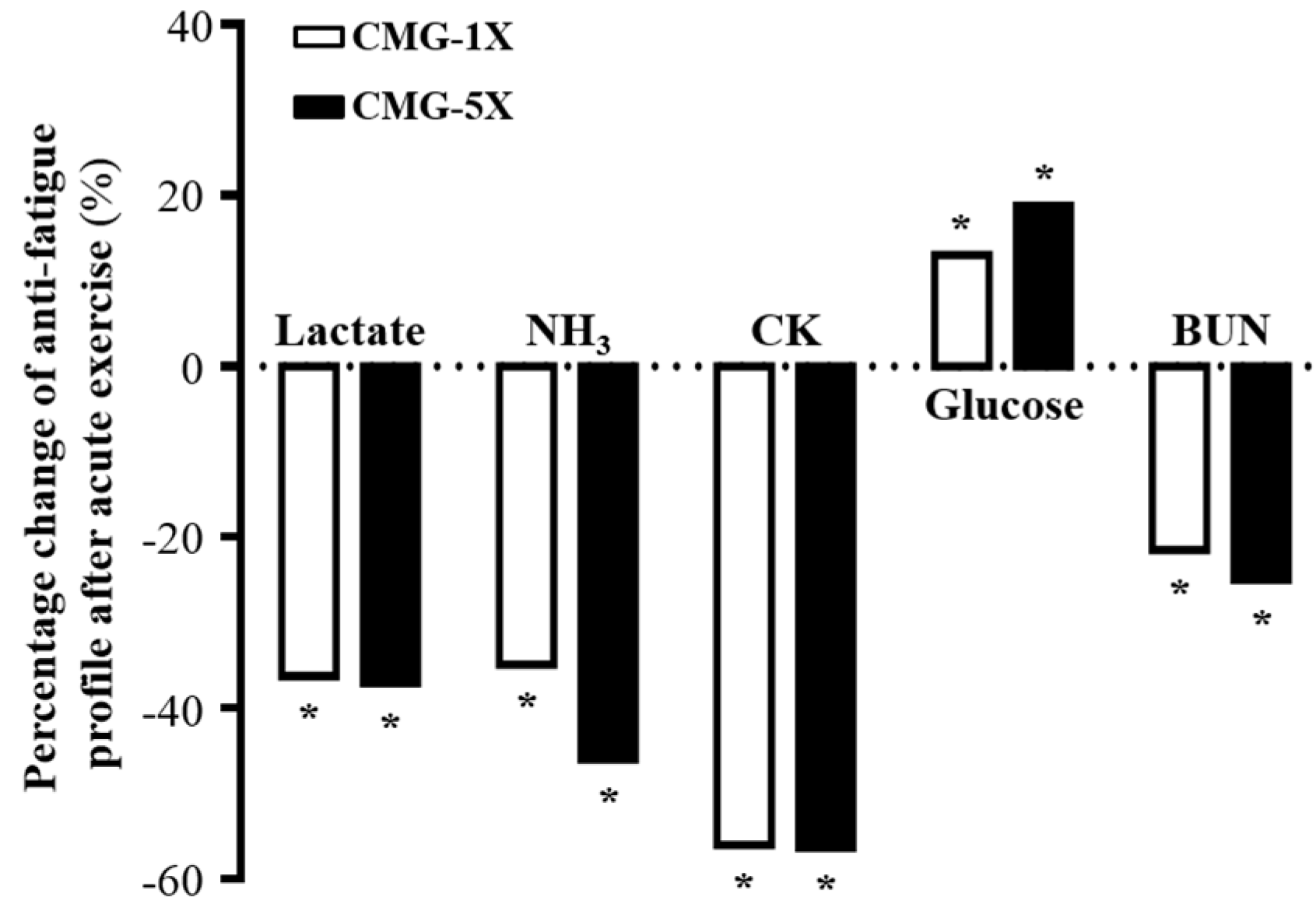
2.4. Effect of CMG Supplementation on Serum Lactate, Ammonia, Glucose, CK, and BUN Levels after Acute Exercise Challenge
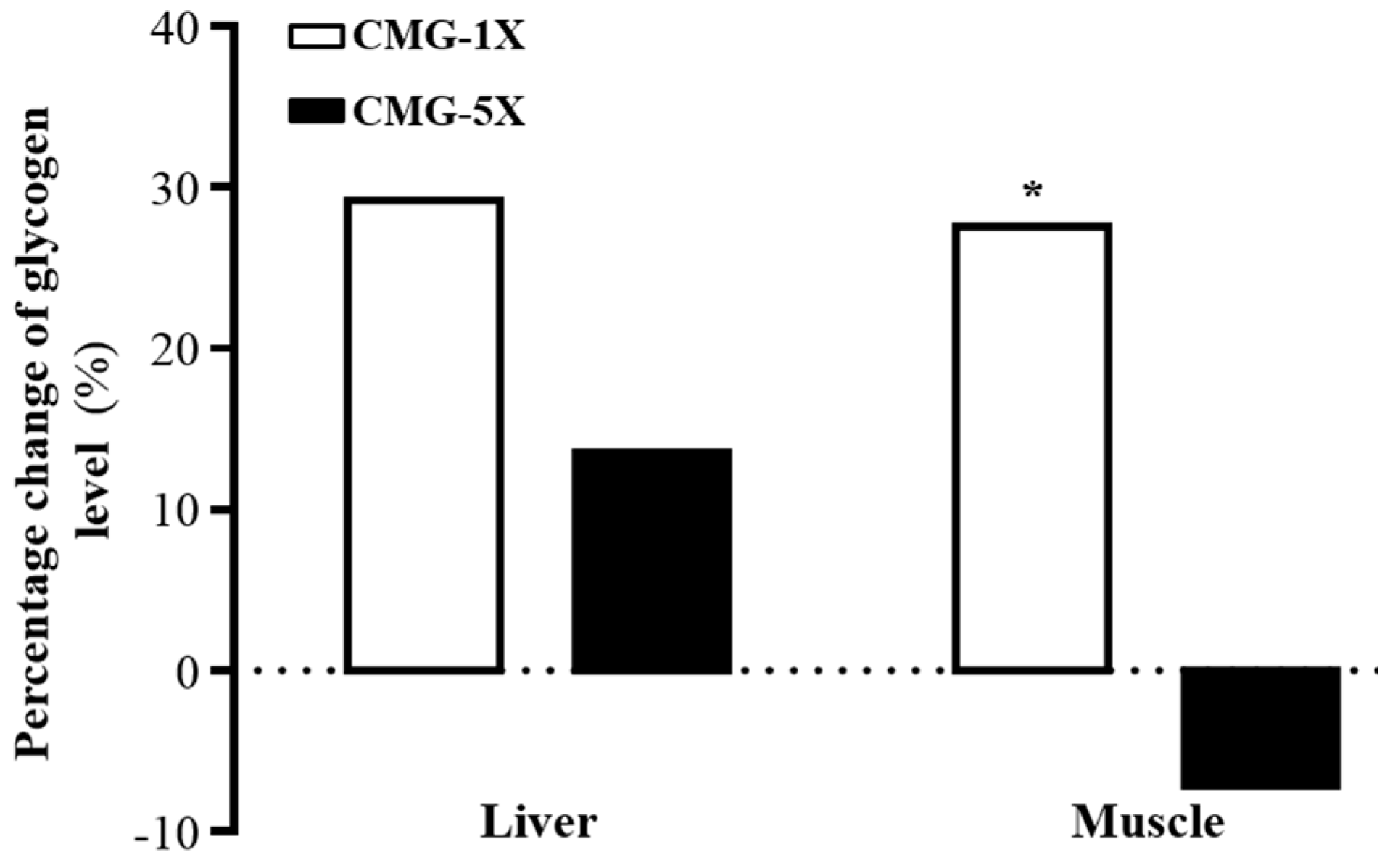
2.5. Effect of CMG Supplementation for 4 Weeks on Hepatic and Muscle Glycogen Levels
2.6. General Characteristics of Mice with CMG Supplementation for Four Weeks
2.7. Effect of CMG Supplementation on Biochemical Variables at the End of the Experiment
3. Discussion
4. Materials and Methods
4.1. Preparation of Changbai Mountain Ginseng (CMG) Extract
4.2. Liquid Chromatographic-Mass Spectrometry (LC-MS) Analysis of CMG Extract
4.3. Animals and Experiment Design
4.4. Forelimb Grip Strength Test
4.5. Swimming Exercise Performance Test
4.6. Determination of Fatigue-Associated Biochemical Variables
4.7. Clinical Biochemical Profiles
4.8. Histology of Tissues
4.9. Tissue Glycogen Determination and Visceral Organ Weight
4.10. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
References
- Shuna, L.; Zhaohuai, J. Development Models of Resource-dependent Cities’ Transformations and Its Experience and Lessons-Take Baishan City’s Development of Transformations as an Example. Energy Procedia 2011, 5, 1626–1630. [Google Scholar] [CrossRef]
- Attele, A.S.; Wu, J.A.; Yuan, C.S. Ginseng pharmacology: Multiple constituents and multiple actions. Biochem. Pharmacol. 1999, 58, 1685–1693. [Google Scholar] [CrossRef]
- Cui, J. Identification and quantification of ginsenosides in various commercial ginseng preparations. Eur. J. Pharm. Sci. 1995, 3, 77–85. [Google Scholar] [CrossRef]
- Tadano, T.; Nakagawasai, O.; Niijima, F. The effects of traditional tonics on fatigue in mice differ from those of the antidepressant imipramine: A pharmacological and behavioral study. Am. J. Chin. Med. 2000, 28, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Barton, D.L.; Liu, H.; Dakhil, S.R.; Linquist, B.; Sloan, J.A.; Nichols, C.R.; Loprinzi, C.L. Wisconsin Ginseng (Panax quinquefolius) to improve cancer-related fatigue: A randomized, double-blind trial. J. Natl. Cancer Inst. 2013, 105, 1230–1238. [Google Scholar] [CrossRef] [PubMed]
- Bae, H.J.; Chung, S.I.; Lee, S.C.; Kang, M.Y. Influence of Aging Process on the Bioactive Components and Antioxidant Activity of Ginseng (Panax ginseng L.). J. Food Sci. 2014, 79, H2127–H2131. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.L.; Tsai, Y.H.; Lin, Y.H.; Chao, J.C. Ginseng extract and ginsenoside Rb1 attenuate carbon tetrachloride-induced liver fibrosis in rats. BMC Complement. Altern. Med. 2014, 14, 415. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Oh, M.; Park, J.; Jang, S.Y.; Cheong, S.H.; Lee, H.; Moon, S.H. Antioxidant and anti-inflammatory activities of the ethanolic extract of fermented red ginseng marc. Food Sci. Biotechnol. 2015, 24, 651–657. [Google Scholar] [CrossRef]
- Wang, J.R.; Yau, L.F.; Tong, T.T.; Feng, Q.T.; Bai, L.P.; Ma, J.; Jiang, Z.H. Characterization of Oxygenated Metabolites of Ginsenoside Rb1 in Plasma and Urine of Rat. J. Agric. Food Chem. 2015, 63, 2689–2700. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, H.Q.; Lu, L.; Fu, D.L.; Liu, A.J.; Li, J.H.; Zheng, G.Q. Ginsenoside Rg1 provides neuroprotection against blood brain barrier disruption and neurological injury in a rat model of cerebral ischemia/reperfusion through downregulation of aquaporin 4 expression. Phytomedicine 2014, 21, 998–1003. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Qi, L.W.; Du, G.J.; Mehendale, S.R.; Wang, C.Z.; Yuan, C.S. Red notoginseng: Higher ginsenoside content and stronger anticancer potential than Asian and American ginseng. Food Chem. 2011, 125, 1299–1305. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, Y.; Zhang, X.Y.; Xu, L.H.; Ouyang, D.Y.; Liu, K.P.; He, X.H. Ginsenoside Rg1 regulates innate immune responses in macrophages through differentially modulating the NF-κB and PI3K/Akt/mTOR pathways. Int. Immunopharmacol. 2014, 23, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Li, W.; Li, X.; Zhang, M.; Chen, L.; Zheng, Y.N.; Ruan, C.C. Antidiabetic effects of malonyl ginsenosides from Panax ginseng on type 2 diabetic rats induced by high-fat diet and streptozotocin. J. Ethnopharmacol. 2013, 145, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Kim, J.H. A review on the medicinal potentials of ginseng and ginsenosides on cardiovascular diseases. J. Ginseng Res. 2014, 38, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.; Mudge, K.W.; Vermeylen, F. Effects of population, age, and cultivation methods on ginsenoside content of wild American ginseng (Panax quinquefolium). J. Agric. Food Chem. 2005, 53, 8498–8505. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, S.; Fan, Y.; Chen, Y.; Liu, D.; Cheng, H.; Zhou, Y. Anti-fatigue activity of the water-soluble polysaccharides isolated from Panax ginseng C.A. Meyer. J. Ethnopharmacol. 2010, 130, 421–423. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.E.; Huang, W.C.; Liao, C.C.; Chang, Y.K.; Kan, N.W.; Huang, C.C. Resveratrol protects against physical fatigue and improves exercise performance in mice. Molecules 2013, 18, 4689–4702. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.J.; Chiu, C.C.; Li, Y.P.; Huang, W.C.; Huang, Y.T.; Huang, C.C.; Chuang, H.L. Effect of intestinal microbiota on exercise performance in mice. J. Strength Cond. Res. 2015, 29, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.P.; Zhang, Y.B.; Yang, X.W.; Yang, X.B.; Xu, W.; Xu, F.; Zhang, L.X. High-Performance Liquid Chromatography with Diode Array Detector and Electrospray Ionization Ion Trap Time-of-Flight Tandem Mass Spectrometry to Evaluate Ginseng Roots and Rhizomes from Different Regions. Molecules 2016, 21, 603. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Huang, W.C.; Chiu, C.C.; Chang, Y.K.; Huang, C.C. Whey protein improves exercise performance and biochemical profiles in trained mice. Med. Sci. Sports Exerc. 2014, 46, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.H.; Huang, C.Y.; Lee, S.D.; Hsu, M.F.; Wang, R.Y.; Kao, C.L.; Kuo, C.H. Decreased eccentric exercise-induced macrophage infiltration in skeletal muscle after supplementation with a class of ginseng-derived steroids. PLoS ONE 2014, 9, e114649. [Google Scholar] [CrossRef] [PubMed]
- Engels, H.J.; Fahlman, M.M.; Wirth, J.C. Effects of ginseng on secretory IgA, performance, and recovery from interval exercise. Med. Sci. Sports Exerc. 2003, 35, 690–696. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Zhang, Y.; Gao, J.; Ding, X.; Gao, S. The anti-fatigue effect of 20(R)-ginsenoside Rg3 in mice by intranasally administration. Biol. Pharm. Bull. 2008, 31, 2024–2027. [Google Scholar] [CrossRef] [PubMed]
- Strojnik, V.; Komi, P.V. Fatigue after submaximal intensive stretch-shortening cycle exercise. Med. Sci. Sports Exerc. 2000, 32, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, M.; González-Izal, M.; Navarro-Amezqueta, I.; Calbet, J.A.; Ibañez, J.; Malanda, A.; Mallor, F.; Häkkinen, K.; Kraemer, W.J.; Gorostiaga, E.M. Effects of strength training on muscle fatigue mapping from surface EMG and blood metabolites. Med. Sci. Sports Exerc. 2011, 43, 303–311. [Google Scholar] [CrossRef] [PubMed]
- White, G.E.; Wells, G.D. The effect of on-hill active recovery performed between runs on blood lactate concentration and fatigue in alpine ski racers. J. Strength Cond. Res. 2015, 29, 800–806. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.P.; Storey, K.B. A quantitative evaluation of the effect of enzyme complexes on the glycolytic rate in vivo: Mathematical modeling of the glycolytic complex. J. Theor. Biol. 1991, 149, 361–375. [Google Scholar] [CrossRef]
- Colberg, S.R.; Sigal, R.J.; Fernhall, B.; Regensteiner, J.G.; Blissmer, B.J.; Rubin, R.R.; Braun, B. Exercise and type 2 diabetes the American College of Sports Medicine and the American Diabetes Association: Joint position statement executive summary. Diabetes Care 2010, 33, 2692–2696. [Google Scholar] [CrossRef] [PubMed]
- Adams, O.P. The impact of brief high-intensity exercise on blood glucose levels. Diabetes Metab. Syndr. Obes. 2013, 6, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Baird, M.F.; Graham, S.M.; Baker, J.S.; Bickerstaff, G.F. Creatine-kinase-and exercise-related muscle damage implications for muscle performance and recovery. J. Nutr. Metab. 2012, 2012, 960363. [Google Scholar] [CrossRef] [PubMed]
- Coyle, E.F.; Hagberg, J.M.; Hurley, B.F.; Martin, W.H.; Ehsani, A.A.; Holloszy, J.O. Carbohydrate feeding during prolonged strenuous exercise can delay fatigue. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1983, 55, 230–235. [Google Scholar] [PubMed]
- Yuan, H.D.; Quan, H.Y.; Kim, S.J.; Jung, M.S.; Chung, S.H. Ginsenoside Rg2 induces orphan nuclear receptor SHP gene expression and inactivates GSK3β via AMP-activated protein kinase to inhibit hepatic glucose production in HepG2 cells. Chem. Biol. Interact. 2012, 195, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Jeong, K.J.; Kim, G.W.; Chung, S.H. AMP-activated protein kinase: An emerging target for ginseng. J. Ginseng Res. 2014, 38, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Im Chung, S.; Nam, S.J.; Xu, M.; Kang, M.Y.; Lee, S.C. Aged ginseng (Panax ginseng Meyer) reduces blood glucose levels and improves lipid metabolism in high fat diet-fed mice. Food Sci. Biotechnol. 2016, 25, 267–273. [Google Scholar] [CrossRef]
- Jensen, T.E.; Richter, E.A. Regulation of glucose and glycogen metabolism during and after exercise. J. Physiol. 2012, 590, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Coggan, A.R.; Coyle, E.F. Carbohydrate Ingestion during Prolonged Exercise: Effects on Metabolism and Performance. Exerc. Sport Sci. Rev. 1991, 19, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Radad, K.; Gille, G.; Moldzio, R.; Saito, H.; Rausch, W.D. Ginsenosides Rb1 and Rg1 effects on mesencephalic dopaminergic cells stressed with glutamate. Brain Res. 2004, 1021, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.S.; Cho, C.W.; Hong, H.D.; Lee, Y.C.; Choi, U.K.; Kim, Y.C. Hypolipidemic and antioxidant properties of phenolic compound-rich extracts from white ginseng (Panax ginseng) in cholesterol-fed rabbits. Molecules 2013, 18, 12548–12560. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhou, M.; Zhao, A.; Jia, W. Traditional Chinese medicine: Balancing the gut ecosystem. Phytother. Res. 2009, 23, 1332–1335. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Y.; Qi, L.W.; Wang, C.Z.; Li, P. Bioactivity enhancement of herbal supplements by intestinal microbiota focusing on ginsenosides. Am. J. Chin. Med. 2011, 39, 1103–1115. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Lin, C.L.; Wei, L.; Hsu, Y.J.; Chen, K.N.; Huang, C.C.; Kao, C.H. Sake Protein Supplementation Affects Exercise Performance and Biochemical Profiles in Power-Exercise-Trained Mice. Nutrients 2016, 8, 106. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Tsai, Y.H.; Tsai, T.Y.; Chiu, Y.S.; Wei, L.; Chen, W.C.; Huang, C.C. Fucoidan supplementation improves exercise performance and exhibits anti-fatigue action in mice. Nutrients 2014, 7, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Chiu, W.C.; Chuang, H.L.; Tang, D.W.; Lee, Z.M.; Wei, L.; Huang, C.C. Effect of curcumin supplementation on physiological fatigue and physical performance in mice. Nutrients 2015, 7, 905–921. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Lin, C.I.; Chiu, C.C.; Lin, Y.T.; Huang, W.K.; Huang, H.Y.; Huang, C.C. Chicken essence improves exercise performance and ameliorates physical fatigue. Nutrients 2014, 6, 2681–2696. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.J.; Li, N.; Zhou, F.; Dong, Q.T.; Zhang, X.D.; Chen, B.C.; Yu, Z. Ginsenoside Rb1 improves energy metabolism in the skeletal muscle of an animal model of postoperative fatigue syndrome. J. Surg. Res. 2014, 191, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, C.L.; Mao, X.Y.; Liu, S.; Chen, W.Z.; Huang, D.D.; Zhang, C.J.; Yu, Z. Ginsenoside Rb1 improves postoperative fatigue syndrome by reducing skeletal muscle oxidative stress through activation of the PI3K/Akt/Nrf2 pathway in aged rats. Eur. J. Pharmacol. 2014, 740, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.
| Exercise Performance | Vehicle | CMG-1X | CMG-5X | Trend Analysis |
|---|---|---|---|---|
| Forelimb grip strength (g) | 114 ± 13 a | 125 ± 10 a | 145 ± 14 b | <0.0001 (↑) |
| Weight-loaded Swimming time (min) | 2.5 ± 0.4 a | 4.1 ± 1.4 b | 4.0 ± 1.1 b | 0.0017 (↑) |
| Serum Levels after 15 min Swimming | Vehicle | CMG-1X | CMG-5X | Trend Analysis |
|---|---|---|---|---|
| Lactate(mmol/L) | 7.3 ± 0.7 a | 4.7 ± 0.8 b | 4.6 ± 0.4 b | <0.0001 (↓) |
| NH3 (μmol/L) | 223 ± 95 a | 145 ± 15 b | 121 ± 20 b | <0.0001 (↓) |
| CK (U/L) | 907 ± 328 a | 400 ± 75 b | 397 ± 37 b | 0.0002 (↓) |
| Glucose (mg/dL) | 125 ± 22 a | 143 ± 19 b | 177 ± 25 b | <0.0001 (↑) |
| BUN (mg/dL) | 34.6 ± 4.5 a | 27.2 ± 1.6 b | 25.9 ± 2.2 b | <0.0001 (↓) |
| Glycogen Contents | Vehicle | CMG-1X | CMG-5X | Trend Analysis |
|---|---|---|---|---|
| Liver (µg/g) | 8.0 ± 4.0 | 11.3 ± 6.1 | 9.2 ± 2.0 | 0.3564 |
| Muscle (µg/g) | 0.16 ± 003 a | 0.23 ± 0.06 b | 0.15 ± 0.03 a | 0.4625 |
| Characteristic | Vehicle | CMG-1X | CMG-5X | Trend Analysis |
|---|---|---|---|---|
| Initial BW (g) | 37.2.6 ± 1.0 | 37.3 ± 1.3 | 37.8 ± 1.7 | 0.2231 |
| Final BW (g) | 38.5 ± 1.5 | 38.9 ± 0.9 | 38.6 ± 1.3 | 0.7732 |
| Food intake (g/day) | 7.3 ± 0.6 | 7.2 ± 0.7 | 7.2 ± 0.3 | 0.8104 |
| Water intake (mL/day) | 8.5 ± 0.6 | 8.6 ± 0.5 | 8.3 ± 0.3 | 0.1808 |
| Liver (g) | 2.09 ± 0.16 | 2.10 ± 0.10 | 2.08 ± 0.14 | 08814 |
| Kidney (g) | 0.60 ± 0.05 | 0.60 ± 0.03 | 0.61 ± 0.05 | 0.7797 |
| Heart (g) | 0.26 ± 0.04 | 0.27 ± 0.03 | 0.27 ± 0.04 | 0.4513 |
| Lung (g) | 0.22 ± 0.02 | 0.22 ± 0.01 | 0.22 ± 0.02 | 0.9438 |
| Muscle (g) | 0.37 ± 0.02 a | 0.39 ± 0.01 b | 0.40 ± 0.01 b | <0.0001 (↑) |
| EFP (g) | 0.33 ± 0.15 | 0.31 ± 0.01 | 0.29 ± 0.01 | 0.8270 |
| BAT (g) | 0.076 ± 0.011 a | 0.090 ± 0.006 b | 0.095 ± 0.009 b | <0.0001 (↑) |
| Relative liver weight (%) | 5.43 ± 0.32 | 5.40 ± 0.30 | 5.38 ± 0.22 | 0.8766 |
| Relative kidney weight (%) | 1.56 ± 0.11 | 1.55 ± 0.08 | 1.57 ± 0.13 | 0.9078 |
| Relative Heart weight (%) | 0.67 ± 0.10 | 0.70 ± 0.08 | 0.69 ± 0.10 | 0.4836 |
| Relative Lung weight (%) | 0.58 ± 0.05 | 0.56 ± 0.03 | 0.58 ± 0.05 | 1.0000 |
| Relative Muscle weight (%) | 0.95 ± 0.05 a | 1.01 ± 0.03 b | 1.03 ± 0.05 b | 0.0004 (↑) |
| Relative EFP weight (%) | 0.85 ± 0.37 | 0.79 ± 0.18 | 0.76 ± 0.20 | 0.8081 |
| Relative BAT weight (%) | 0.20 ± 0.03 a | 0.23 ± 0.02 b | 0.25 ± 0.03 b | <0.0001 (↑) |
| Variables | Vehicle | CMG-1X | CMG-5X | Trend Analysis |
|---|---|---|---|---|
| AST (U/L) | 102 ± 14 | 102 ± 11 | 101 ± 11 | 0.5837 |
| ALT (U/L) | 61 ± 16 | 59 ± 19 | 60 ± 22 | 0.7688 |
| LDH (U/L) | 534 ± 60 | 528 ± 82 | 537 ± 76 | 0.9100 |
| CK (U/L) | 560 ± 140 b | 507 ± 148 ab | 413 ± 124 a | 0.0045 (↓) |
| TP (g/dL) | 4.9 ± 0.2 a | 5.0 ± 0.4 ab | 5.2 ± 0.2 b | 0.0017 (↑) |
| Albumin (g/dL) | 3.0 ± 0.1 a | 3.0 ± 0.1 a | 3.2 ± 0.1 b | <0.0001 (↑) |
| BUN (mg/dL) | 23.6 ± 1.8 | 23.9 ± 1.6 | 22.5 ± 1.7 | 0.4280 |
| Creatinine (mg/dL) | 0.30 ± 0.03 | 0.31 ± 0.04 | 0.30 ± 0.02 | 0.8319 |
| UA (mg/dL) | 1.9 ± 0.6 | 1.7 ± 0.3 | 1.7 ± 0.3 | 0.5904 |
| TC (mg/dL) | 137 ± 13 | 133 ± 8 | 131 ± 8 | 0.3105 |
| TG (mg/dL) | 153 ± 15 b | 124 ± 22 a | 124 ± 11 a | <0.0001 (↓) |
| Glucose (mg/dL) | 148 ± 18 a | 155 ± 16 ab | 168 ± 10 b | 0.0030 (↑) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, G.-D.; Chiu, C.-H.; Hsu, Y.-J.; Hou, C.-W.; Chen, Y.-M.; Huang, C.-C. Changbai Mountain Ginseng (Panax ginseng C.A. Mey) Extract Supplementation Improves Exercise Performance and Energy Utilization and Decreases Fatigue-Associated Parameters in Mice. Molecules 2017, 22, 237. https://doi.org/10.3390/molecules22020237
Ma G-D, Chiu C-H, Hsu Y-J, Hou C-W, Chen Y-M, Huang C-C. Changbai Mountain Ginseng (Panax ginseng C.A. Mey) Extract Supplementation Improves Exercise Performance and Energy Utilization and Decreases Fatigue-Associated Parameters in Mice. Molecules. 2017; 22(2):237. https://doi.org/10.3390/molecules22020237
Chicago/Turabian StyleMa, Guo-Dong, Chun-Hui Chiu, Yi-Ju Hsu, Chien-Wen Hou, Yi-Ming Chen, and Chi-Chang Huang. 2017. "Changbai Mountain Ginseng (Panax ginseng C.A. Mey) Extract Supplementation Improves Exercise Performance and Energy Utilization and Decreases Fatigue-Associated Parameters in Mice" Molecules 22, no. 2: 237. https://doi.org/10.3390/molecules22020237
APA StyleMa, G.-D., Chiu, C.-H., Hsu, Y.-J., Hou, C.-W., Chen, Y.-M., & Huang, C.-C. (2017). Changbai Mountain Ginseng (Panax ginseng C.A. Mey) Extract Supplementation Improves Exercise Performance and Energy Utilization and Decreases Fatigue-Associated Parameters in Mice. Molecules, 22(2), 237. https://doi.org/10.3390/molecules22020237







